1. Introduction
Since the beginning of our half-century of work on resistance breeding, experiments reported in publications of interest to us have been repeated to help understanding. We also took photographs of pathologies described in other authors’ publications but not shown in pictures. With these and with photographs of our own work, we aim to make the knowledge of the symptoms in this speciality more visible and clearer.
The century-old history of plant resistance breeding has been based on the rapid destruction of the attacked tissue of the host plant, the hypersensitive response (specific HR).
In the relationship between cereals and
Puccinia graminis, Ward [
1] and Stakman [
2], in the interaction between
Nicotiana glutinosa and TMV, Holmes [
3,
4], in the host–pathogen relationship between
Capsicum annum and
Xanthomonas vesicatoria, Cook and Stall [
5] described the local lesion in the
Bs1 gene, and Cook and Guevara [
6] described the local lesion in the
Bs2 gene (
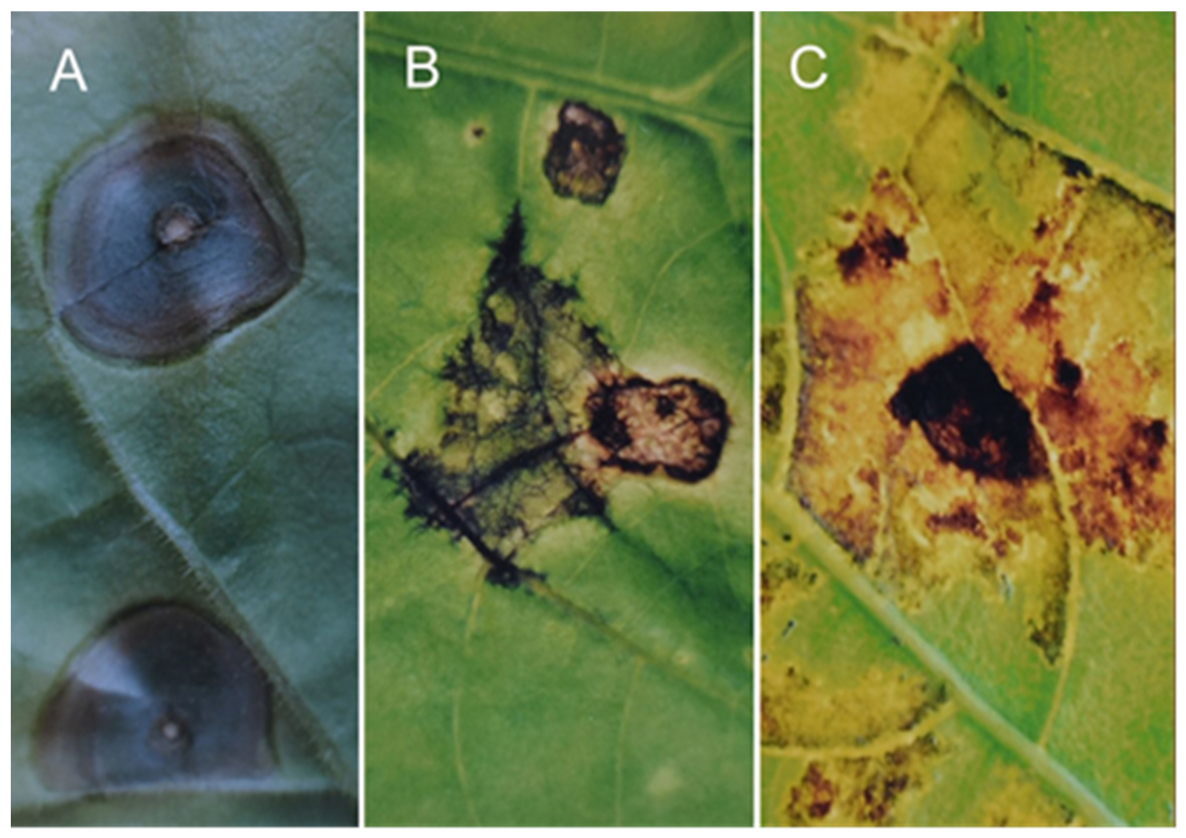
Figure 1).
In addition to the study of HR induced by specific resistance genes in the host–pathogen relationship, the observation of the non-host–pathogen relationship has also started. The leaves of the
Nicotiana tabacum L.
White Burley plant were infiltrated with inoculum concentration of 10
5, 10
6, 10
7, 10
8 cells/mL of
Pseudomonas tabaci being in a host–pathogenic relationship with tobacco, and of
Pseudomonas species as non-host–pathogenic plant pathogen. Inoculum concentration of 10
6, 10
7, 10
8 cells/mL of bacteria being in a non-host–pathogenic relationship with tobacco induced rapid tissue destruction in 24–48 h for all
Pseudomonas species. The 10
5 inoculum concentration induced partial tissue destruction only in
P. tabaci, whereas in the other pathogenic
Pseudomonas species, small local lesions were formed in the area of infected leaf spots. The authors found that phytopathogenic
Pseudomonas species can also multiply in the intercellular ducts of non-host plants, inducing (nonspecific) HR, and that this is considered a naturally occurring phenomenon, but the authors overlooked the fact that this phenomenon can only occur if the pathogen has entered the intercellular ducts in very large quantities. A very important aspect of the experiment, but not interpreted by the authors, is that the dilution series of
Pseudomonas pathogen inoculum, the reaction type switch, was in all cases triggered by increasing the inoculum concentration from 10
5 cells/mL to 10
6 cells/mL, thus setting the limit of the overall tissue retention capacity of
White Burley [
7].
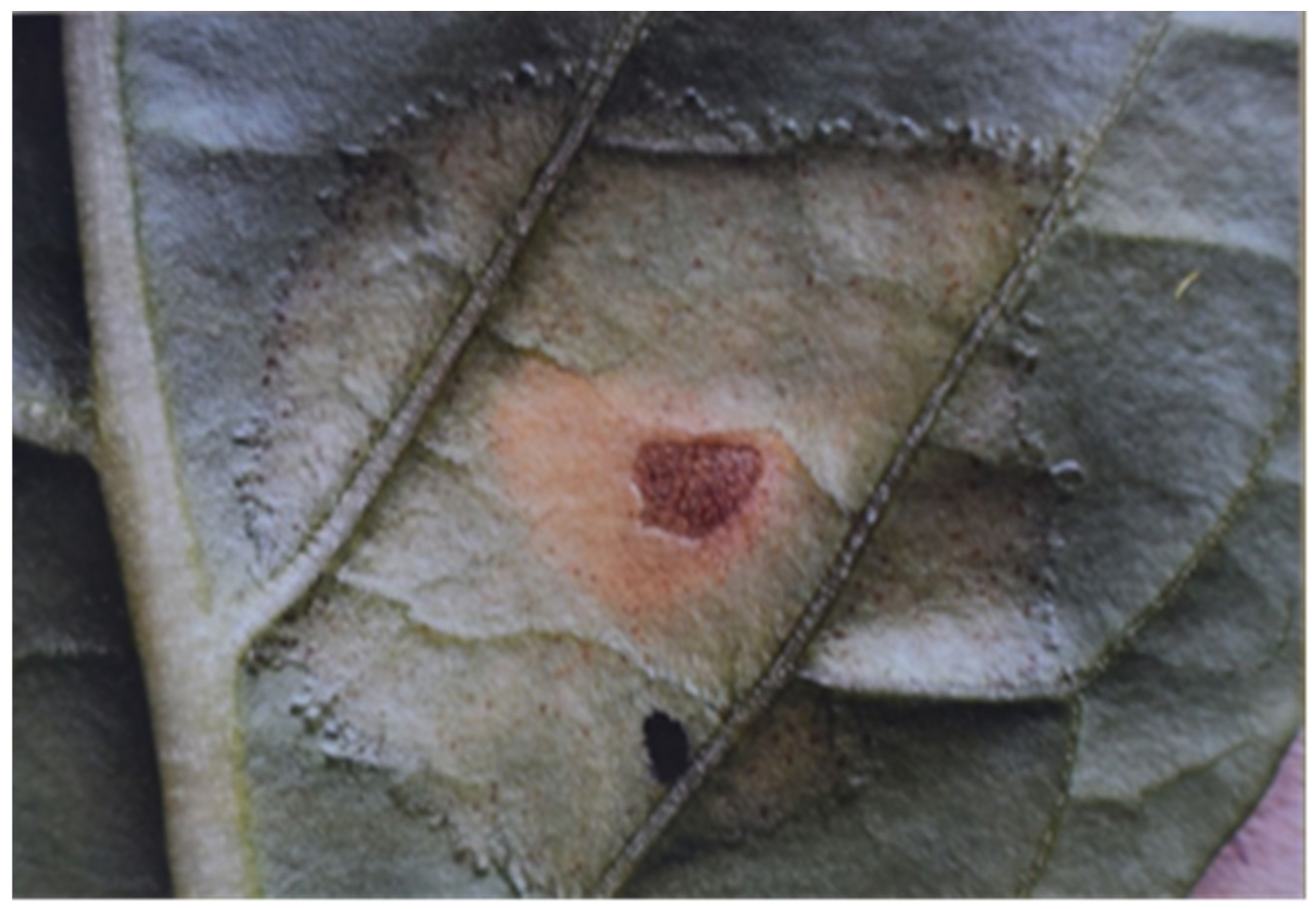
The relationship between susceptibility and HR in host–pathogen and non-host–pathogen relationships was studied by Szarka et al. [
8].
Nicotiana tabacum cv. Pallagi leaves were infiltrated with 10
1−10
9 cells/mL inoculum of
P. tabaci (
Figure 2A), and
P. phaseolicola (
Figure 2B) bacteria (
Figure 2), irrespective of the host–pathogen or non-host–pathogen relationship.
The 10
9 cells/mL inoculum induced nonspecific HR in both bacterial species in the same way. The 10
8 cells/mL inoculum of
P. tabaci induced the typical pathological symptoms of the disease on its host plant, tobacco, and 10
1–10
7 cell/mL inoculum of
P. tabaci and 10
1–10
8 cells/mL inoculum of
P. phaseolicola induced at most a slight bulging towards the front surface of the leaf and a chlorotic spot with indeterminate margins on infiltrated tissue sections. In other words, the tissue retention capacity of the
Pallagi tobacco variety was able to tolerate stress caused by a bacterial suspension of
P. tabaci at a concentration of 10
7 cell/mL and of
P. phaseolicola at a concentration of 10
8 cell/mL. From this observation, conclusions were drawn on the tissue retention capacity of tobacco [
8].
In evaluating the results, the term General Defence Reaction (GDR) was used to describe the tissue retention capacity of the plant in host–pathogen and non-host–pathogen relationships, i.e., the specific HR counterpoint, and its interpretation was described in 1995 [
9].
The 10
8 cells/mL inoculum of
Pallagi and
P. tabaci formed a host–pathogen relationship in which a delicate equilibrium was established between the stress of the pathogen and the tissue retention capacity of the host plant, which was upset by the stress effect of the pathogen’s increased proliferation. The plant tissue began to die, and this balance of power was eventually manifested in the susceptible disease. The
P. tabaci suspension at concentration of 10
7 cells/mL showed the dominance of the GDR of the plant, while the rapid tissue death caused by the suspension at concentration of 10
9 cells/mL showed the destruction of the GDR. Since susceptibility as a physiological state is intermediate between the two, it was likely that GDR levels play a dominant role in the susceptibility of plants to pathogens [
8].
To study susceptibility status and specific resistance gene activation, pepper cell lines with similar GDR levels, containing susceptible and
Bs2 resistance genes, were tested using a non-host–pathogen relationship, using
X. vesicatoria inocula at concentrations of 10
1–10
9 cells/mL [
8]. Leaf spots of susceptible and
Bs2 resistance gene-containing peppers infiltrated with suspensions of
X. vesicatoria at concentrations of 10
1–10
7 cells/mL were at most chlorotic. The inoculum at concentration of 10
8 cells/mL produced a greasy spot on the susceptible cell line and a maroon discolouration on the leaf containing the
Bs2 gene, which did not dry out and caused resistance symptoms. Since pepper cell lines with similar tissue retention were tested, it was concluded that the
Bs2 gene is only activated when one of the stress levels represented by the
X. vesicatoria dilution series breaks through the plant’s general defence system. On the basis of this observation, it is likely that the general defence response is activated first in the defence process, and that its insufficiency leads to a state of susceptibility, which induces a physiological disturbance that triggers the specific resistance gene, which, thus, only becomes active after the development of the disease.
Contrary to the “resistance = HR” theory, the “destruction is not resistance” position could not be proven until a gene encoding a resistant response without tissue death was found.
The non-hypersensitive, nonspecific recessive gene found in cell line PI 163,192 of
Capsicum Annuum became applicable for resistance identification in 1995. Based on the characteristics it encodes, it is a proposed marker of monogenic recessive trait: general defence system—
gds [
9]. In contrast to the
Bs2 gene (
Figure 3), the response regulated by the
gds gene (
Figure 4) is manifested by cell growth, cell division, tissue compaction, and bulging of the infected leaf plate.
Unlike
X. vesicatoria (
Figure 5A) being in a host–pathogen relationship with pepper,
P. phaseolicola (
Figure 5B) and
X. Phaseoli (
Figure 5C) being in a non-host relationship, and the saprophytic
P. Fluorescens (
Figure 5D) bacteria, the
gds gene responds with the same tissue lesions.
Thus, in addition to being non-hypersensitive, gds is also a non-specific gene, as it gives a generalised response (GDR) to specific stresses induced by different pathogenic species in peppers containing the gds gene.
The gene that protects pepper by producing a tissue retention defence response was published in 2002 [
11]. The gene was given the name
bs5 and another recessive tissue retention gene,
bs6, was also described at the same time.
Since the
bs5 gene was found in the same PI 163,192 cell line in which Szarka and Csilléry were the first to find and describe a tissue retention recessive gene in 1995 [
9], the authors performed allele testing of
gds and the
bs5 gene [
12]. Based on the susceptibility and tissue retention pathogenicity of
X. vesicatoria, the heritability tests confirmed that the two genes were identical. Pepper containing the
bs6 gene (ECW60) was shown to be susceptible in testing, which is presumably why it is not used in breeding today, unlike the
gds (
bs5) gene.
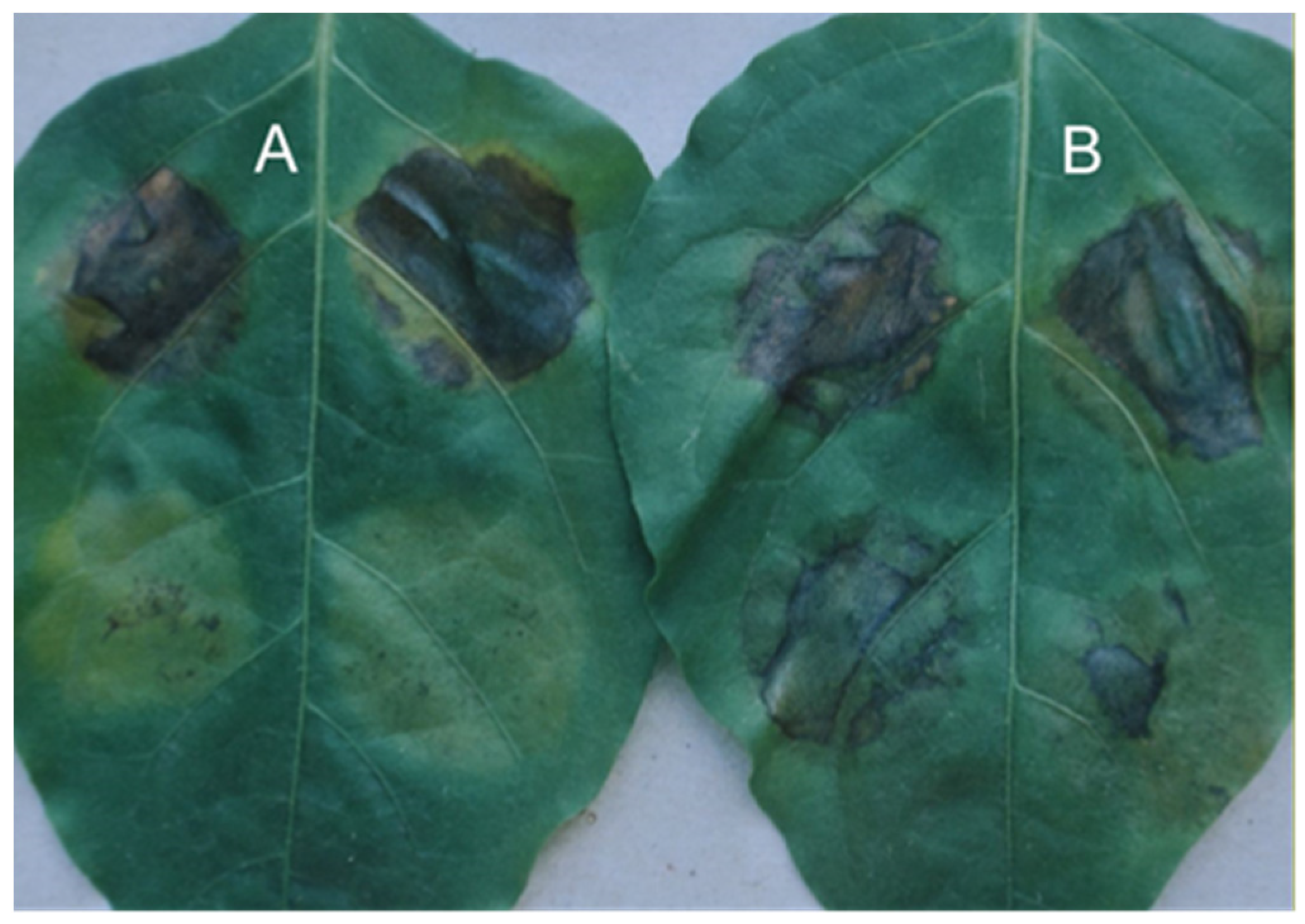
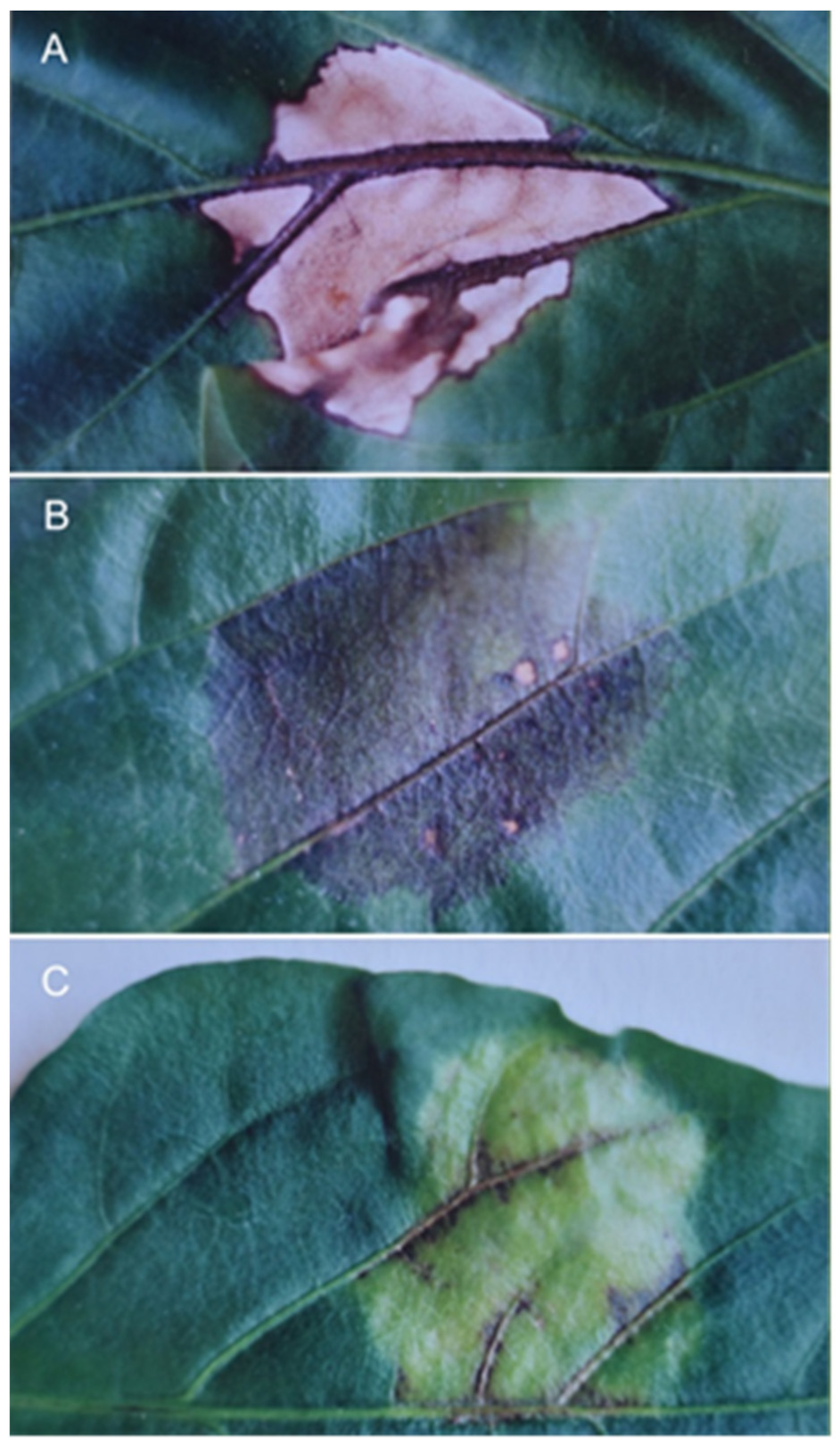
Another type of tissue retention response other than
gds has also been reported [
9]. In this experiment, pepper cell lines carrying the
Bs2 and
Bs3 genes were infected with
X. vesicatoria and the infected leaves were detached from the plants and dried 24 h after inoculation (
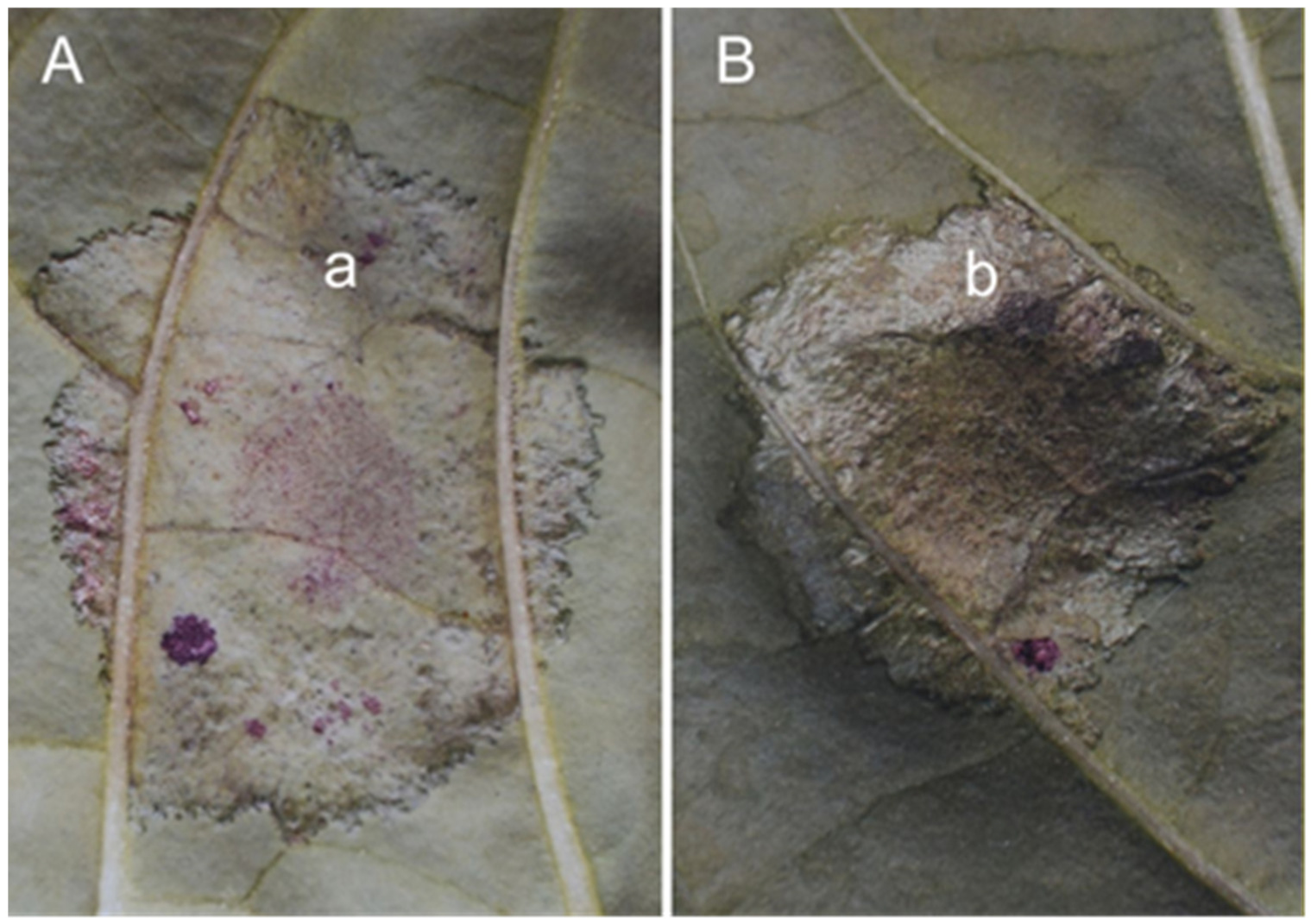
Figure 6). The leaves of the
Bs3 plant showed tissue damage indicative of HR already at the time of detachment, while the leaves of the
Bs2 plant showed only a slight purple discolouration indicating the location of the infected spots. For the
Bs3 gene, the dried infected tissue (
Figure 6A) was only half as thick as the uninfected tissue, whereas for the
Bs2 gene, in addition to the slight purple discolouration, the infected tissue was twice as thick (
Figure 6B) as the uninfected tissue.
A similar tissue retention property was observed in the non-host–pathogenic relationship between pepper containing HR genes (Bs2, Bs3) and the bean pathogen X. Phaseoli.
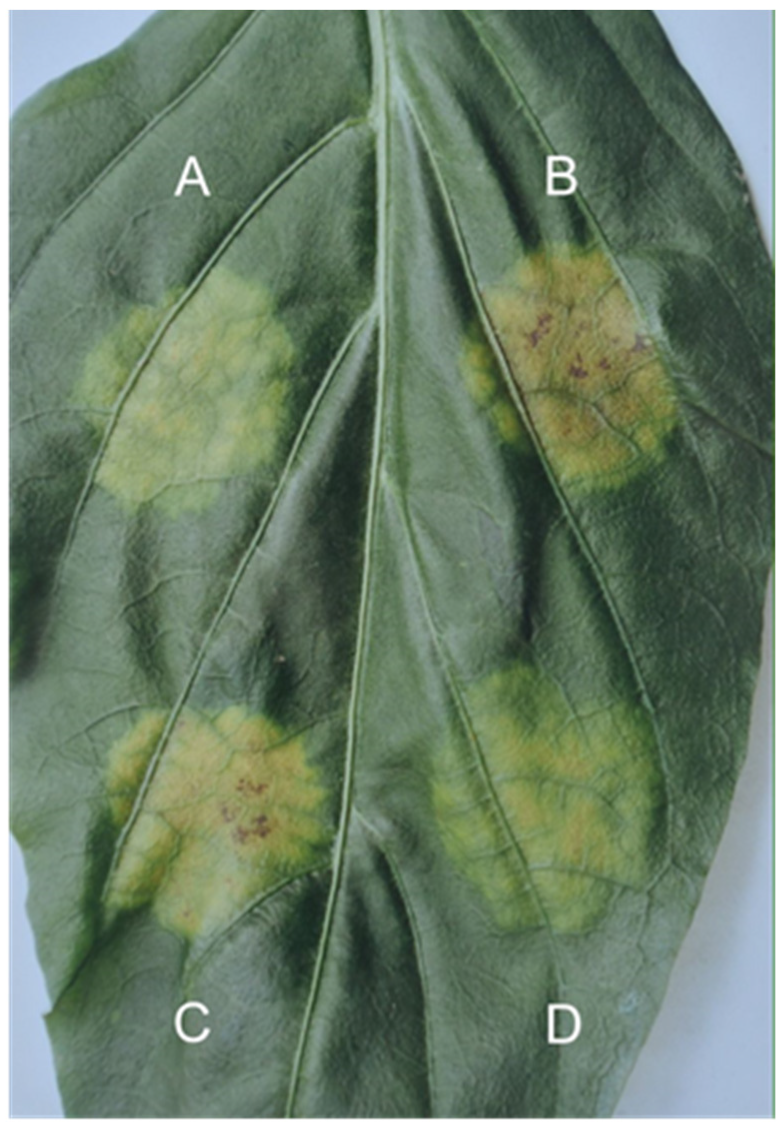
The GDR phenomenon has also been described in several host–pathogen relationships where the host plant did not contain a known resistance gene and nonetheless developed tissue retention pathology suggestive of non-susceptibility (
Figure 7). Such factors include:
Cucumis sativus–Pseudoperonospora cubensis (
Figure 7A,B),
Phaseolus vulgaris–Pseudomonas phaseolicola (
Figure 7C) [
13].
Hydrogen peroxide plays an essential role in the development of specific HR determined by specific resistance genes. H
2O
2 in plant-microbe or host–pathogen interactions, depending on its amount, either enhances or destroys host plant cells. The production of H
2O
2 by susceptible plants containing the
Bs2 and
gds genes, upon the effect of
X. vesicatoria, was as follows [
10]. The author investigated the amount of H
2O
2 by infiltration of the entire leaf surface of the plants for 10 h after infection. In the susceptible host–pathogen relationship, the pathogen enters the plant without causing a stress effect, so no significant H
2O
2 change occurs. The pathogen only becomes detectable to the plant during its accumulation. In response to H
2O
2 as a signalling molecule for the initiation of death in the susceptible phase, the
Bs2 gene is activated, causing a specific HR characterised by H
2O
2 “burst”. In the experiment, the H
2O
2 “burst” occurred within 30 min and after 8 h it was reduced to the control level, while the infected tissues were destroyed. H
2O
2 levels remained constant in plants containing the
gds gene, which encodes a strong tissue retention capacity.
H
2O
2 induces lignin synthesis and cross-linking between phenolic compounds and cellular wall proteins. This results in increased resistance to the enzymes degrading the cellular wall [
14]. The formation of cross-links between cellular wall proteins is very rapid, occurring within 2–5 min after a stimulus [
15]. Non-host resistance is primarily based on general responses linked to the cellular wall. These include thickening, lignification of the cellular wall, accumulation of phenols, flavonoids, which are highly localized responses expressed at the point of pathogen entry [
16].
The aim of our work is to describe the functional defects of specific resistance genes used in resistance breeding and to elucidate their causes. Furthermore, we will describe the pathophysiology of a previously unknown defence response without tissue destruction, its genetic analysis, and its application in resistance breeding.
2. Materials and Methods
The experiments were carried out at the Kecskemét site of Univer Product Plc. between 2017 and 2021.
A significant part of our observations on plant material for variety production were made in the framework of resistance breeding work. The plants were planted in the soil of the growing house, i.e., we worked with plants with strong roots and vigorous growth. The plants were inoculated before flowering, in the vegetative stage, which is characterised by strong growth. The leaves selected for this purpose were at 70–80% of their development. Suspensions of different concentrations of X. vesicatoria and P. phaseolicola bacteria prepared from 48 h culture were used for inoculation. To form lesions, inoculum was applied to the abaxial surface of the leaf by brushing, mimicking the natural infection process. The primary criterion of evaluation in this case was the size of the lesion. For evaluation by tissue retention, intercellular ducts were flooded by injection. This method is less dependent on the environment and allows differentiation based on the quality of the infected tissue. The assessment of symptoms was performed on days 7 and 14, with occasional continuous evaluation.
For the genetic analysis, two infected leaves of a plant, directly above each other, were inoculated (
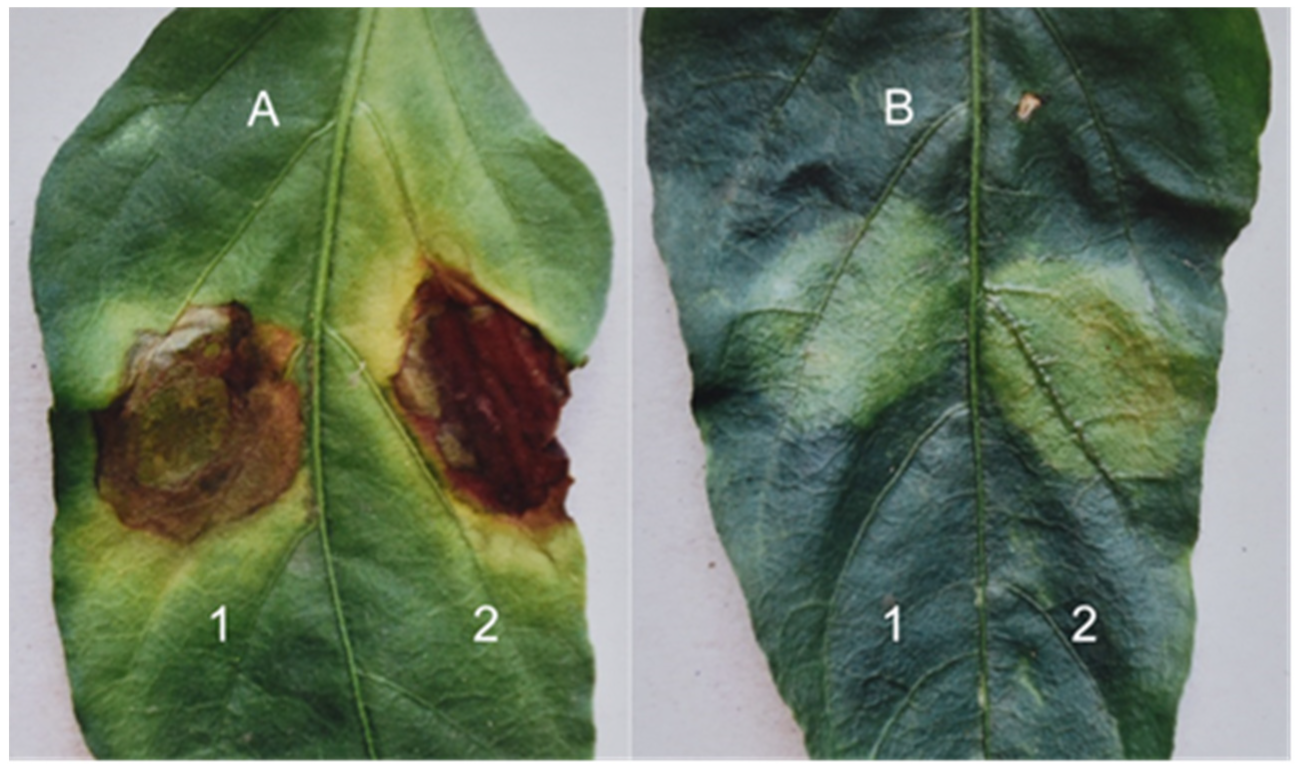
Figure 8A,B).
In both leaves, the upper left spot shows the inoculation of 108 cells/mL of X. vesicatoria bacteria in a host–pathogenic relationship with pepper, and the upper right spot shows the inoculation of 108 cells/mL of P. phaseolicola bacteria in a non-host–pathogenic relationship with pepper. The lower two spots on the upper leaf show the result of inoculation by the bacterium X. pesicatoria at concentrations of 107 (left spot) and 106 (right spot) cells/mL. The lower two spots on the lower leaf show the spots resulting from the inoculation of P. phaseolicola at concentrations of 107 (left spot) and 106 (right spot) cells/mL. Both leaves were tested with a 108 cells/mL suspension of the two pathogens to account for possible differences due to the age of the leaves.
The reaction of the leaves was classified into pathogen groups according to whether the inoculation resulted in a green, tissue-retaining spot or a drying, necrotic spot (
Figure 9,
Figure 10 and
Figure 11), as required for genetic analysis.
Figure 12A shows the design of experiments to investigate the rate of response of genes controlling specific and general defence. This involved infecting a spot with 5 mm of diameter with one pathogen (a) and then, after absorption of the watery patch, re-infecting it with the other pathogen (b), knowing the location of the first spot (a).
Figure 12B illustrates the inoculation process to study the function of specific and general defence responses in damaged cells. This involved inducing a convex tissue spot infiltrated by mechanical pressure with an object that consequently damaged the cells to different degrees (c) and inoculating it with a bacterial suspension (d).
Of the hot peppers used in the experiments,
Unihot is a commercial variety, the others are breeding lines (
Table 1).
The cleavages observed in F2 were subjected to Chi2 test regression analysis. Our null hypothesis is that the observed genetic cleavage rates correspond to a given theoretical genetic cleavage.
There is no established terminology yet for plant traits and pathological processes related to tissue retention. Hence, we see the need for an interpretation of the terms and concepts we use.
Susceptibility (S): in the case of a host-pathogen relationship, plant tissue death characteristic of the pathogen. The rate of the process depends on the plant’s capacity to retain tissue.
Hypersensitive response (HR): rapid tissue death in response to biotic stresses.
Specific hypersensitive response (sHR): in a host–pathogen relationship, the destruction of the attacked plant tissue by a specific resistance gene.
Nonspecific hypersensitive reaction (aHR): rapid tissue death in a non-host relationship due to stress induced by a pathogen, or rapid tissue death in a host–pathogen relationship without a specific resistance agent due to over-infection.
Tissue retention capacity: the genetically determined trait that protects plant tissues from destruction by biotic and abiotic stresses.
General defence response (GDR): a defence process under biotic stresses, in host–pathogen and non-host–pathogen relationships, based on the plant’s tissue retention capacity to exclude pathogens in a general response to specific stresses.
Weak/strong GDR: the degree of tissue retention manifested in pathogenicity without knowledge of the genotype.
Complete GDR: the ability of a plant containing both genes (gdr 1 + 2) to retain tissue.
Incomplete GDR: the degree of tissue retention in a plant containing either or neither of the gdr 1 and 2 genes, under stress expressed as inoculum concentration.
5. Conclusions
The inadequacies of the history of resistance breeding and our present work demonstrate that specific HR genes alone cannot safely protect the plant because they only act in a downstream manner in cells attacked by the pathogen, inducing cell death.
A decisive role in the defence of plants containing HR genes is played by the plant’s tissue retention capacity, which is expressed in the general defence response (GDR). GDR pre-emptively excludes the pathogen without tissue loss.
Specific HR genes do not protect but destroy the cells affected by pathogens. GDR genes protect the plant by strengthening the cells. The difference in the stimulus thresholds of the two responses also determines the order and effectiveness of the response. The non-pathogen specific GDR plays the role of the plant immune system due to its low stimulus threshold and high reaction rate.
Tissue retention capacity can be increased by breeding to a level where the GDR alone can provide adequate protection for the plant. In this case, the presence of specific HR genes is unnecessary; sometimes it is even a burden to the plant.
In our work on tissue retention in pepper, we found that the GDR system we studied is regulated by two recessive genes (gdr 1 + 2) that are completely independent of the HR-conferring Bs2 gene and the previously identified tissue retention gds gene.
Studies on pepper demonstrate that plant resistance can be made safer without the incorporation of specific HR genes, relying solely on GDR.
An important point is that while the function of specific HR genes is dependent on the environment, GDR is as independent as possible from environmental factors. It functions under the extreme conditions that the plant can tolerate, acting as a plant immune system.
Host plant–pathogen contacts carrying specific HR genes are very rare in nature, while there are a lot of host–pathogen contacts and plant–microbe contacts are countless. This is possible because all plants have GDRs. Consequently, if we want to integrate resistance breeding into the order of nature, our work must be based on the tissue retention capacity of plants.