Seed Germination Ecophysiology of Acacia dealbata Link and Acacia mearnsii De Wild.: Two Invasive Species in the Mediterranean Basin
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Area and Study Species
2.2. Seed Lot Collection and Preparation
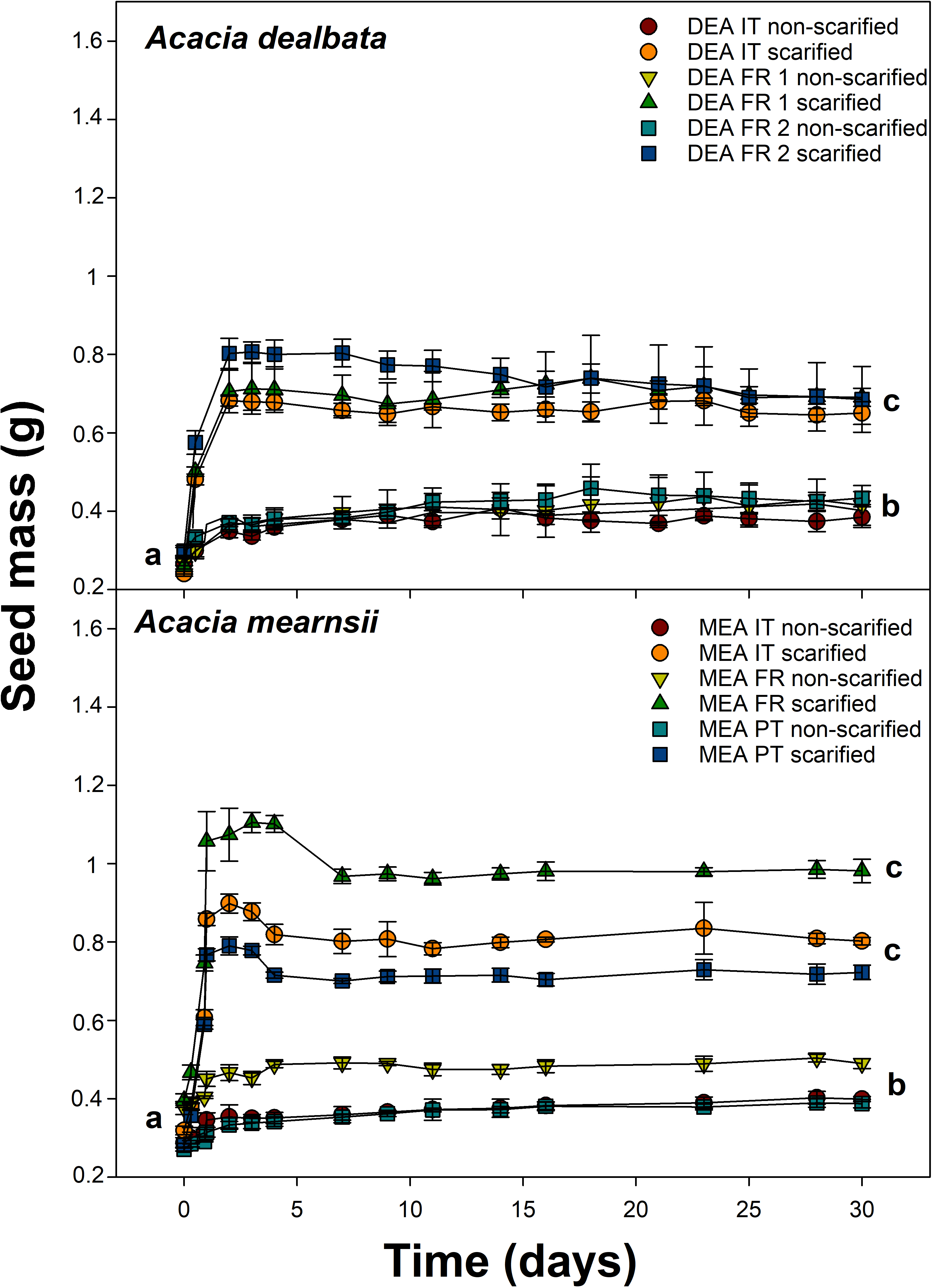
2.3. Imbibition Capacity
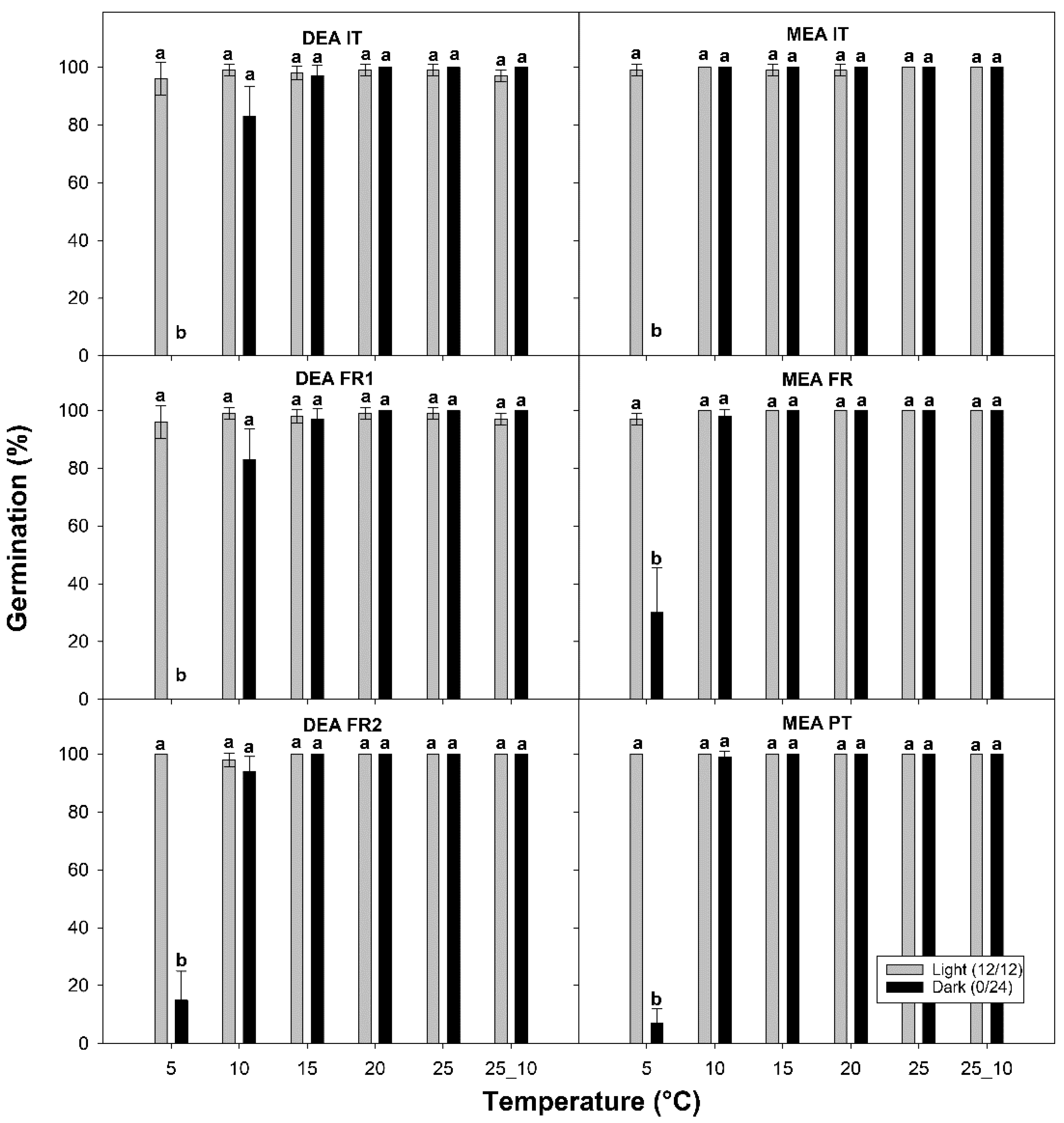
2.4. Germination Tests
2.5. Data Analysis
3. Results
3.1. Imbibition Capacity
3.2. Germination Test
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Gaertner, M.; den Breeyen, A.; Hui, C.; Richardson, D.M. Impacts of Alien Plant Invasions on Species Richness in Mediterranean-Type Ecosystems: A Meta-Analysis. Prog. Phys. Geogr. 2009, 33, 319–338. [Google Scholar] [CrossRef]
- Richardson, D.M.; Pyšek, P.; Simberloff, D.; Rejmánek, M.; Mader, A.D. Biological Invasions-the Widening Debate: A Response to Charles Warren. Prog. Hum. Geogr. 2008, 32, 295–298. [Google Scholar] [CrossRef]
- Olden, J.D.; Poff, N.L.R.; Douglas, M.R.; Douglas, M.E.; Fausch, K.D. Ecological and Evolutionary Consequences of Biotic Homogenization. Trends Ecol. Evol. 2004, 19, 18–24. [Google Scholar] [CrossRef]
- Bongaarts, J. IPBES, 2019. Summary for Policymakers of the Global Assessment Report on Biodiversity and Ecosystem Services of the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services. Popul. Dev. Rev. 2019, 45, 680–681. [Google Scholar] [CrossRef] [Green Version]
- Richardson, D.M.; Pyšek, P. Plant Invasions: Merging the Concepts of Species Invasiveness and Community Invasibility. Prog. Phys. Geogr. 2006, 30, 409–431. [Google Scholar] [CrossRef]
- Lockwood, J.L.; Cassey, P.; Blackburn, T. The Role of Propagule Pressure in Explaining Species Invasions. Trends Ecol. Evol. 2005, 20, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Gritti, E.S.; Smith, B.; Sykes, M.T. Vulnerability of Mediterranean Basin Ecosystems to Climate Change and Invasion by Exotic Plant Species. J. Biogeogr. 2006, 33, 145–157. [Google Scholar] [CrossRef]
- Myers, N.; Mittermeler, R.A.; Mittermeler, C.G.; da Fonseca, G.A.B.; Kent, J. Biodiversity Hotspots for Conservation Priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef] [PubMed]
- Fois, M.; Podda, L.; Médail, F.; Bacchetta, G. Endemic and Alien Vascular Plant Diversity in the Small Mediterranean Islands of Sardinia: Drivers and Implications for Their Conservation. Biol. Conserv. 2020, 244, 108519. [Google Scholar] [CrossRef]
- Médail, F. The Specific Vulnerability of Plant Biodiversity and Vegetation on Mediterranean Islands in the Face of Global Change. Reg. Environ. Chang. 2017. [Google Scholar] [CrossRef] [Green Version]
- Celesti-Grapow, L.; Bassi, L.; Brundu, G.; Camarda, I.; Carli, E.; D’Auria, G.; del Guacchio, E.; Domina, G.; Ferretti, G.; Foggi, B.; et al. Plant Invasions on Small Mediterranean Islands: An Overview. Plant Biosyst. 2016, 150, 1119–1133. [Google Scholar] [CrossRef]
- Brundu, G.; Lozano, V.; Manca, M.; Celesti-Grapow, L.; Sulas, L. Arctotheca calendula (L.) Levyns: An Emerging Invasive Species in Italy. Plant Biosyst. 2015, 149, 954–957. [Google Scholar] [CrossRef]
- Puddu, S.; Podda, L.; Mayoral, O.; Delage, A.; Hugot, L.; Petit, Y.; Bacchetta, G. Comparative Analysis of the Alien Vascular Flora of Sardinia and Corsica. Notulae Botanicae Horti Agrobotanici Cluj-Napoca 2016, 44, 337–346. [Google Scholar] [CrossRef] [Green Version]
- Brundu, G.; Richardson, D.M. Planted Forests and Invasive Alien Trees in Europe: A Code for Managing Existing and Future Plantings to Mitigate the Risk of Negative Impacts from Invasions. NeoBiota 2016, 30, 5–47. [Google Scholar] [CrossRef]
- Sanz Elorza, M.; Guillot Ortiz, D.; Deltoro, V. La Flora Alóctona de La Comunidad Valenciana (España). Bot. Complut. 2011, 35, 97–130. [Google Scholar] [CrossRef] [Green Version]
- Everitt, J.; Lonard, R.; Little, C. Weeds in South Texas and Northern Mexico; Texas Tech University Press: Lub-bock, TX, USA, 2007. [Google Scholar]
- Holm, L.R.G.; Plucknett, D.L.; Pancho, J.v.; Herberger, J.P. The World’s Worst Weeds. Distribution and Biology; University Press of Hawaii: Honolulu, HI, USA, 1977. [Google Scholar]
- Lowe, S.; Browne, M.; Boudjelas, S.; De Poorter, M. 100 of the World’s Worst Invasive Alien Species: A Selection from the Global Invasive Species Database; The Invasive Species Specialist Group (ISSG): Auckland, New Zealand, 2000. [Google Scholar]
- Lozano, V.; Marzialetti, F.; Carranza, M.L.; Chapman, D.; Branquart, E.; Dološ, K.; Große-Stoltenberg, A.; Fiori, M.; Capece, P.; Brundu, G. Modelling Acacia Saligna Invasion in a Large Mediterranean Island Using PAB Factors: A Tool for Implementing the European Legislation on Invasive Species. Ecol. Indic. 2020, 116, 106516. [Google Scholar] [CrossRef]
- Mayoral, O.; Mascia, F.; Podda, L.; Laguna, E.; Fraga, P.; Rita, J.; Frigau, L.; Bacchetta, G. Alien Plant Diversity in Mediterranean Wetlands: A Comparative Study within Valencian, Balearic and Sardinian Floras. Notulae Botanicae Horti Agrobotanici Cluj-Napoca 2018, 46, 317–326. [Google Scholar] [CrossRef] [Green Version]
- Meloni, F.; Dettori, C.A.; Mascia, F.; Podda, L.; Bacchetta, G. What Does the Germination Ecophysiology of the Invasive Acacia Saligna (Labill.) Wendl. (Fabaceae) Teach Us for Its Management? Plant Biosyst. 2015, 149, 242–250. [Google Scholar] [CrossRef]
- Rouget, M.; Robertson, M.P.; Mechanical, J.R.U.; Hui, C.; Essl, F.; Renteria, J.L.; Richardson, D.M. Invasion Debt—Quantifying Future Biological Invasions. Divers. Distrib. 2016, 22, 445–456. [Google Scholar] [CrossRef]
- Richardson, D.M.; Rejmánek, M. Trees and Shrubs as Invasive Alien Species—A Global Review. Divers. Distrib. 2011, 17, 788–809. [Google Scholar] [CrossRef]
- Brundu, G. Invasive alien plants in protected areas in mediterranean Islands: Knowledge gaps and main threats. In Plant Invasions in Protected Areas: Patterns, Problems and Challenges; Springer: Dordrecht, The Netherlands, 2013; pp. 395–422. ISBN 9789400777507. [Google Scholar]
- Sheppard, A.W.; Shaw, R.H.; Sforza, R. Top 20 Environmental Weeds for Classical Biological Control in Europe: A Review of Opportunities, Regulations and Other Barriers to Adoption. Weed Res. 2006, 46, 93–117. [Google Scholar] [CrossRef]
- Richardson, D.M.; Kluge, R.L. Seed Banks of Invasive Australian Acacia Species in South Africa: Role in Invasiveness and Options for Management. Perspect. Plant Ecol. Evol. Syst. 2008, 10, 161–177. [Google Scholar] [CrossRef]
- Goets, S.A.; Kraaij, T.; Little, K.M. Comparing Germination Stimuli of Two Alien Invasive Species and a Native Analogue: Towards Sustainable Management of Invasives. S. Afr. J. Bot. 2017, 112, 15–18. [Google Scholar] [CrossRef]
- Marchante, H.; Freitas, H.; Hoffmann, J.H. Seed Ecology of an Invasive Alien Species, Acacia Longifolia (Fabaceae), in Portuguese Dune Ecosystems. Am. J. Bot. 2010, 97, 1780–1790. [Google Scholar] [CrossRef] [PubMed]
- Marchante, H.; Freitas, H.; Hoffmann, J.H. Post-Clearing Recovery of Coastal Dunes Invaded by Acacia Longifolia: Is Duration of Invasion Relevant for Management Success? J. Appl. Ecol 2011, 48, 1295–1304. [Google Scholar] [CrossRef]
- Lorenzo, P.; González, L.; Roger, M.R. Allelopathic Interference of Invasive Acacia Dealbata: Physiological Effects. Allelopathy J. 2008, 2, 452–462. [Google Scholar]
- Baskin, C.C.; Baskin, J.M. Seeds: Ecology, Biogeography and Evolution of Dormancy and Germination, 2nd ed.; Academic Press: San Diego, CA, USA, 2014. [Google Scholar]
- Baskin, J.M.; Baskin, C.C. A Classification System for Seed Dormancy. Seed Sci. Res. 2004, 14, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Holmes, P.M.; Cowling, R.M. Diversity, Composition and Guild Structure Relationships between Soil-Stored Seed Banks and Mature Vegetation in Alien Plant-Invaded South African Fynbos Shrublands. Plant Ecol. 1997, 133, 107–122. [Google Scholar] [CrossRef]
- Spooner, P.G. Response of Acacia Species to Disturbance by Roadworks in Roadside Environments in Southern New South Wales, Australia. Biol. Conserv. 2005, 122, 231–242. [Google Scholar] [CrossRef]
- Fenner, M.K.; Fenner, M.; Thompson, K. The Ecology of Seeds; Cambridge University Press: Cambridge, UK, 2005. [Google Scholar]
- Ellison, A.M. Interspecific and Intraspecific Variation in Seed Size and Germination Requirements of Sarracenia (Sarraceniaceae). Am. J. Bot. 2001, 88, 429–437. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cruz, A.; Pérez, B.; Velasco, A.; Moreno, J.M. Variability in Seed Germination at the Interpopulation, Intrapopulation and Intraindividual Levels of the Shrub Erica Australis in Response to Fire-Related Cues. Plant Ecol. 2003, 169, 93–103. [Google Scholar] [CrossRef]
- Boland, D.J.; Brooker, M.I.H.; Chippendale, G.M.; Hall, N.; Hyland, B.P.M.; Johnston, R.D.; Kleinig, D.A.; Turner, J.D. Forest Trees of Australia, 4th ed.; Nelson and CSIRO: Melbourne, Australia, 1984. [Google Scholar]
- Searle, S.D. Acacia mearnsii De Wild. (black wattle) in Australia. In Black Wattle and its Utilisation; Brown, A.G., Ho, C.K., Eds.; Rural Industries Research and Development Corporation: Canberra, Australia, 1997. [Google Scholar]
- Carballeira, A.; Reigosa, M.J. Effects of Natural Leachates of Acacia Dealbata Link in Galicia (NW Spain). Bot. Bull. Acad. Sin. 1999, 40, 87–92. [Google Scholar]
- Brundu, G.; Podda, L.; Lozano, V.; Porceddu, M.; Bacchetta, G. Distribuzione Ed Invasività Di Acacia Mearnsii in Sardegna. In Proceedings of the Contributo presentato dal gruppo specie alloctone della SBI. Riunioni scientifiche dei gruppi, Milano, Italy, 27 November 2018. [Google Scholar]
- Marchante, H.; Morais, M.; Freitas, H.; Marchante, E. Guia Prático Para a Identificação de Plantas Invasoras Em Portugal; Imprensa da Universidade de Coimbra: Coimbra, Portugal, 2014. [Google Scholar]
- Minuto, L.; Casazza, G.; Dagnino, D.; Guerrina, M.; Macri, C.; Zappa, E.; Mariotti, M.G. Reproductive Traits of the Invasive Species Acacia Dealbata Link. In the Northern Mediterranean Basin. Ann. Bot. 2020, 10, 13–20. [Google Scholar] [CrossRef]
- Gibson, M.R.; Richardson, D.M.; Marchante, E.; Marchante, H.; Rodger, J.G.; Stone, G.N.; Byrne, M.; Fuentes-Ramírez, A.; George, N.; Harris, C.; et al. Reproductive Biology of Australian Acacias: Important Mediator of Invasiveness? Divers. Distrib. 2011, 17, 911–933. [Google Scholar] [CrossRef]
- Azani, N.; Babineau, M.; Bailey, C.D.; Banks, H.; Barbosa, A.R.; Pinto, R.B.; Boatwright, J.S.; Borges, L.M.; Brown, G.K.; Bruneau, A.; et al. A New Subfamily Classification of the Leguminosae Based on a Taxonomically Comprehensive Phylogeny. Taxon 2017, 66, 44–77. [Google Scholar] [CrossRef] [Green Version]
- Mabberley, D.J. Mabberley’s Plant-Book: A Portable Dictionary of Plants, Their Classification and Uses, 3th ed.; Cambridge University Press: Cambridge, UK, 2008. [Google Scholar]
- Boland, D.J. Genetic Resources and Utilisation of Australian Bipinnate Acacias (Botrycephalae). In Australian Acacias in Developing Countries, Proceedings of an International Workshop, Gympie, Australia, 4–7 August 1986; Turnbull, J.W., Ed.; ACIAR: Canberra, Australia, 1987. [Google Scholar]
- Simmons, M. Acacias of Australia; Penguin Books Australia Ltd.: Melbourne, Australia, 1988; Volume 2. [Google Scholar]
- Tame, T. Acacias of South Eastern Australia; Kangaroo Press: Sydney, Australia, 1992. [Google Scholar]
- Lorenzo, P.; González, L.; Reigosa, M.J. The Genus Acacia as Invader: The Characteristic Case of Acacia Dealbata Link in Europe. Ann. For. Sci. 2010, 67, 101. [Google Scholar] [CrossRef] [Green Version]
- Weber, E. Invasive Plant Species of the World: A Reference Guide to Environmental Weeds; CAB International Publishing: Wallingford, UK, 2003. [Google Scholar]
- Doran, J.; Turnbull, J. Australian Trees and Shrubs: Species for Land Rehabilitation and Farm Planting in the Tropics; ACIAR: Canberra, Australia, 1997. [Google Scholar]
- Grant, J.E.; Moran, G.F.; Moncur, M.W. Pollination Studies and Breeding System in Acacia Mearnsii. In Australian Tree Species Research in China, Proceedings of an International Workshop: Zhangzhou, China, 2–5 November 1992; Brown, A.G., Ed.; ACIAR Proceedings: Zhangzhou, China, 1994. [Google Scholar]
- Moncur, M.W.; Moran, G.F.; Boland, D.; Turner, J. Floral Morphology and Breeding Systems of Acacia Mearnsii De Wild. In Proceedings of the Use of Australian Trees in China Workshop, Guangzhou, China, December 1988; Chinese Academy of Forestry and ACIAR: Guangzhou, China, 1989. [Google Scholar]
- Tassin, J.; Balent, G. Le Diagnostic d’invasion d’une Essence Forestière En Milieu Rural: Exemple d’acacia Mearnsii Àla Réunion. Rev. For. Francaise 2004, 56, 132–142. [Google Scholar] [CrossRef] [Green Version]
- Baret, S.; Rouget, M.; Richardson, D.M.; Lavergne, C.; Egoh, B.; Dupont, J.; Strasberg, D. Current Distribution and Potential Extent of the Most Invasive Alien Plant Species on La Réunion (Indian Ocean, Mascarene Islands). Austral Ecol. 2006, 31, 747–758. [Google Scholar] [CrossRef]
- Raymond, C.A. Flowering Biology, Genetics and Breeding. Black Wattle and Its Utilisation; RIRDC Publication: Canberra, Australia, 1997; Volume 3. [Google Scholar]
- Bacchetta, G.; Belletti, P.; Brullo, S.; Cagelli, L.; Carasso, V.; Casas, J.L.; Cervelli, C.; Escribà, M.C.; Fenu, G.; Gorian, F. Manuale per La Raccolta, Studio, Conservazione e Gestione Ex Situ Del Germoplasma; APAT: Rome, Italy, 2006; Volume 37. [Google Scholar]
- Porceddu, M.; Santo, A.; Orrù, M.; Meloni, F.; Ucchesu, M.; Picciau, R.; Sarigu, M.; Cuena Lombrana, A.; Podda, L.; Sau, S.; et al. Seed Conservation Actions for the Preservation of Plant Diversity: The Case of the Sardinian Germplasm Bank (BG-SAR). Plant Sociol. 2017, 54, 111–117. [Google Scholar]
- Hidayati, S.N.; Baskin, J.M.; Baskin, C.C. Dormancy-Breaking and Germination Requirements for Seeds of Symphoricarpos Orbiculatus (Caprifoliaceae). Am. J. Bot. 2001, 88, 1444–1451. [Google Scholar] [CrossRef]
- Crawley, M.J. The R Book; John Wiley & Sons: Chichester, UK, 2012. [Google Scholar]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2013. [Google Scholar]
- Venier, P.; Funes, G.; Carrizo García, C. Physical Dormancy and Histological Features of Seeds of Five Acacia Species (Fabaceae) from Xerophytic Forests in Central Argentina. Flora 2012, 207, 39–46. [Google Scholar] [CrossRef]
- Passos, I.; Marchante, H.; Pinho, R.; Marchante, E. What We Don’t Seed: The Role of Long-Lived Seed Banks as Hidden Legacies of Invasive Plants. Plant Ecol. 2017, 218, 1313–1324. [Google Scholar] [CrossRef]
- Morrison, D.A.; Auldf, T.D.; Rish, S.; Porter, C.; McClay, K. Patterns of Testa-Imposed Seed Dormancy in Native Australian Legumes. Ann. Bot. 1992, 70, 157–163. [Google Scholar] [CrossRef]
- Beaury, E.M.; Fusco, E.J.; Jackson, M.R.; Laginhas, B.B.; Morelli, T.L.; Allen, J.M.; Pasquarella, V.J.; Bradley, B.A. Incorporating Climate Change into Invasive Species Management: Insights from Managers. Biol. Invasions 2020, 22, 233–252. [Google Scholar] [CrossRef] [Green Version]
- Degreef, J.; Rocha, O.J.; Vanderborght, T.; Baudoin, J.P. Soil Seed Bank and Seed Dormancy in Wild Populations of Lima Bean (Fabaceae): Considerations for in Situ and Ex Situ Conservation. Am. J. Bot. 2002, 89, 1644–1650. [Google Scholar] [CrossRef]
- Baskin, J.M.; Baskin, C.C. Evolutionary Considerations of Claims for Physical Dormancy-Break by Microbial Action and Abrasion by Soil Particles. Seed Sci. Res. 2000, 10, 409–413. [Google Scholar] [CrossRef]
- Milton, S.J.; Hall, A.V. Reproductive Biology of Australian Acacias in the South-Western Cape Province, South Africa. Trans. R. Soc. S. Afr. 1981, 44, 465–487. [Google Scholar] [CrossRef]
- Stanton, M.L. Seed Variation in Wild Radish: Effect of Seed Size on Components of Seedling and Adult Fitness. Ecology 1984, 65, 1105–1112. [Google Scholar] [CrossRef]
- Grime, J.P.; Mason, G.; Curtis, A.V.; Rodman, J.; Band, S.R. A Comparative Study of Germination Characteristics in a Local Flora. J. Ecol. 1981, 69, 1017. [Google Scholar] [CrossRef]
- Galmés, J.; Medrano, H.; Flexas, J. Germination Capacity and Temperature Dependence in Mediterranean Species of the Balearic Islands. Investigación Agraria Sistemas y Recursos Forestales 2006, 15, 88. [Google Scholar] [CrossRef] [Green Version]
- Porceddu, M.; Pritchard, H.W.; Mattana, E.; Bacchetta, G. Differential Interpretation of Mountain Temperatures by Endospermic Seeds of Three Endemic Species Impacts the Timing of in Situ Germination. Plants 2020, 9. [Google Scholar] [CrossRef]
- DelSole, T.; Tippett, M.K. Predictability in a Changing Climate. Clim. Dyn. 2018, 51, 531–545. [Google Scholar] [CrossRef] [Green Version]
- Picciau, R.; Porceddu, M.; Bacchetta, G. Can Alternating Temperature, Moist Chilling, and Gibberellin Interchangeably Promote the Completion of Germination in Clematis Vitalba Seeds? Botany 2017, 95. [Google Scholar] [CrossRef] [Green Version]
- Probert, R.J. The role of temperature in the regulation of seed dormancy and germination. In Seeds: The Ecology of Regeneration in Plant Communities; Fenner, M., Ed.; CAB International: Wallingford, UK, 2000. [Google Scholar]
- Grime, J.P.; Thompson, K. An Apparatus for Measurement of the Effect of Amplitude of Temperature Fluctuation upon the Germination of Seeds. Ann. Bot. 1976, 40, 795–799. [Google Scholar] [CrossRef]
- Cavieres, L.A.; Arroyo, M.T.K. Seed Germination Response to Cold Stratification Period and Thermal Regime in Phacelia Secunda (Hydrophyllaceae): Altitudinal Variation in the Mediterranean Andes of Central Chile. Plant Ecol. 2000, 149, 1–8. [Google Scholar] [CrossRef]
- Billings, W.D.; Mooney, H.A. The ecology of arctic and alpine plants. Biol. Rev. 1968, 43, 481–529. [Google Scholar] [CrossRef]
- Crous, C.J.; Jacobs, S.M.; Esler, K.J. Drought-Tolerance of an Invasive Alien Tree, Acacia Mearnsii and Two Native Competitors in Fynbos Riparian Ecotones. Biol. Invasions 2012, 14, 619–631. [Google Scholar] [CrossRef]
- Pauchard, A.; Maheu-Giroux, M.; Aguayo, M.; Esquivel, J. COS 10–1: Detecting Invasion Processes at the Landscape and Regional Scales: Acacia Dealbata in Chile. In Proceedings of the 93rd Ecological Society of America Annual Meeting, Milwaukee, WI, USA, 3–8 August 2008; Ecological Society of America: Milwaukee, WI, USA, 2008. [Google Scholar]
- de Wit, M.P.; Crookes, D.J.; van Wilgen, B.W. Conflicts of Interest in Environmental Management: Estimating the Costs and Benefits of a Tree Invasion. Biol. Invasions 2001, 3, 167–178. [Google Scholar] [CrossRef]
- Mayoral, O.; Podda, L.; Porceddu, M. Invasive Alien Flora on Coastal Mediterranean Habitats. In Handbook of Halophytes; Springer: New York, NY, USA, 2021; pp. 569–597. [Google Scholar] [CrossRef]
- Lorenzo, P.; Pazos-Malvido, E.; Rubido-Bará, M.; Reigosa, M.J.; González, L. Invasion by the Leguminous Tree Acacia Dealbata (Mimosaceae) Reduces the Native Understorey Plant Species in Different Communities. Aust. J. Bot. 2012, 60, 669–675. [Google Scholar] [CrossRef] [Green Version]
- Wilson, J.R.U.; Gairifo, C.; Gibson, M.R.; Arianoutsou, M.; Bakar, B.B.; Baret, S.; Celesti-Grapow, L.; DiTomaso, J.M.; Dufour-Dror, J.-M.; Kueffer, C.; et al. Risk Assessment, Eradication, and Biological Control: Global Efforts to Limit Australian Acacia Invasions. Divers. Distrib. 2011, 17, 1030–1046. [Google Scholar] [CrossRef] [Green Version]
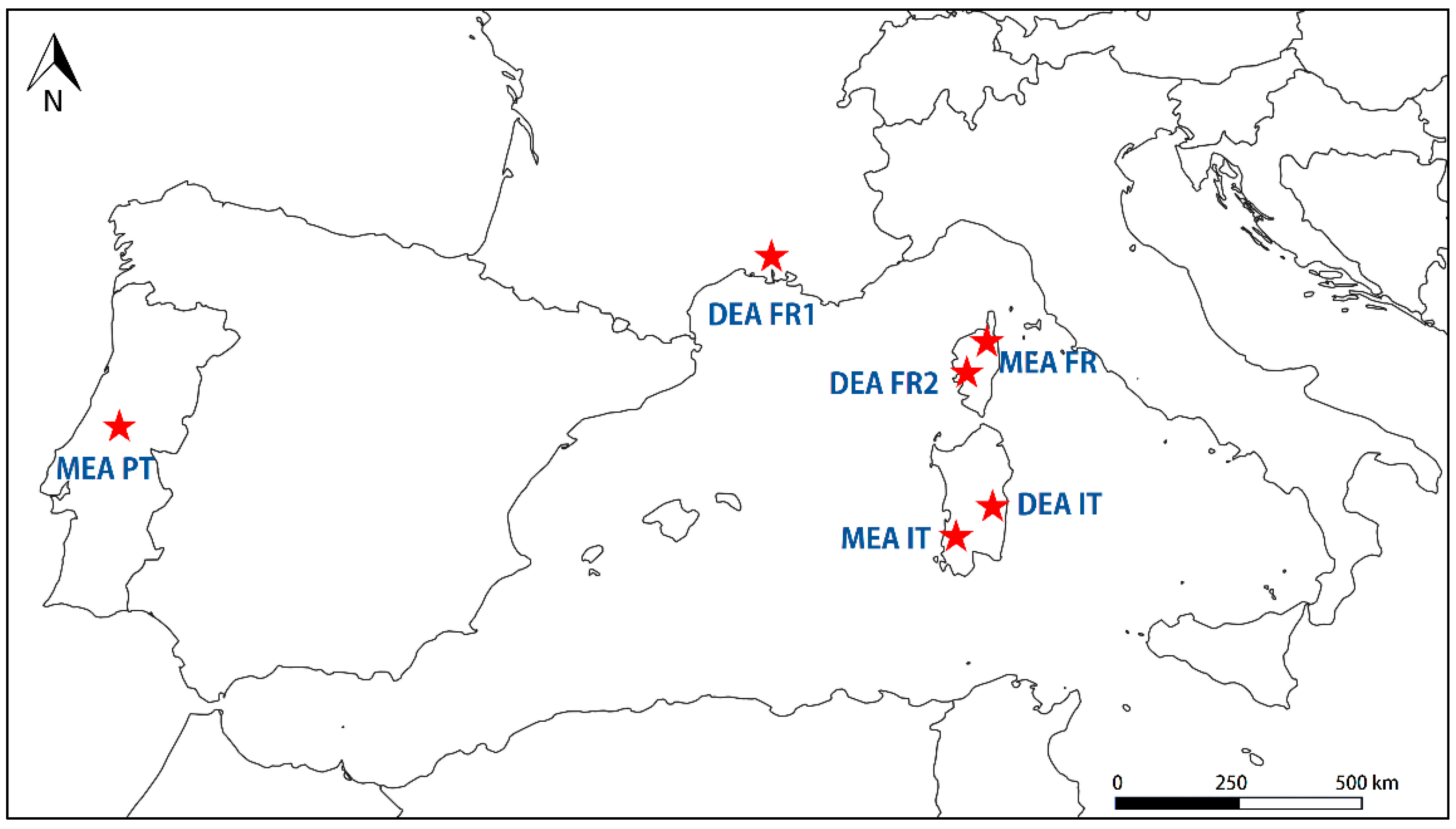


| Code | Species | Locality | Mean Coordinates (WGS 84) | Mean Altitude (m a.s.l.) | Lithology |
|---|---|---|---|---|---|
| DEA IT | Acacia dealbata | San Gregorio (Sinnai, Sardinia, Italy) | 39°17′ N 09°21′ E | 207 | Metamorphytes |
| DEA FR1 | Acacia dealbata | La Croix-Valmer (Provence-Alpes-Côte d′Azur, France) | 43°10′ N 06°36′ E | 10 | Granites |
| DEA FR2 | Acacia dealbata | Capitoro (Cauro, Corse, France) | 41°90′ N 08°82′ E | 35 | Granites |
| MEA IT | Acacia mearnsii | Rio Leni (Villacidro, Sardinia, Italy) | 39°23′ N 08°41′ E | 250 | Granites |
| MEA FR | Acacia mearnsii | Sant′Agata (Aleria, Corse, France) | 42°12′ N 09°51′ E | 12 | Limestones |
| MEA PT | Acacia mearnsii | Sarnadas de Rodão (Castelo Branco, Beira Baixa, Portugal) | 39°74′ N 07°62′ E | 100 | Metamorphytes |
| Code | No. Seeds | Avg. Mass of Seed (g) | Major Diameter (mm) | Minor Diameter (mm) |
|---|---|---|---|---|
| DEA IT | 7054 | 0.011 | 10.11 ± 0.73 | 6.13 ± 0.59 |
| DEA FR1 | 4249 | 0.010 | 9.28 ± 0.78 | 6.69 ± 0.42 |
| DEA FR2 | 14269 | 0.013 | 9.26 ± 0.84 | 5.99 ± 0.43 |
| MEA IT | 19048 | 0.013 | 8.96 ± 0.90 | 6.02 ± 0.38 |
| MEA FR | 11508 | 0.017 | 9.91 ± 0.94 | 6.44 ± 0.66 |
| MEA PT | 3387 | 0.013 | 9.27 ± 0.92 | 5.84 ± 0.51 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dessì, L.; Podda, L.; Brundu, G.; Lozano, V.; Carrouée, A.; Marchante, E.; Marchante, H.; Petit, Y.; Porceddu, M.; Bacchetta, G. Seed Germination Ecophysiology of Acacia dealbata Link and Acacia mearnsii De Wild.: Two Invasive Species in the Mediterranean Basin. Sustainability 2021, 13, 11588. https://doi.org/10.3390/su132111588
Dessì L, Podda L, Brundu G, Lozano V, Carrouée A, Marchante E, Marchante H, Petit Y, Porceddu M, Bacchetta G. Seed Germination Ecophysiology of Acacia dealbata Link and Acacia mearnsii De Wild.: Two Invasive Species in the Mediterranean Basin. Sustainability. 2021; 13(21):11588. https://doi.org/10.3390/su132111588
Chicago/Turabian StyleDessì, Ludovica, Lina Podda, Giuseppe Brundu, Vanessa Lozano, Antoine Carrouée, Elizabete Marchante, Hélia Marchante, Yohan Petit, Marco Porceddu, and Gianluigi Bacchetta. 2021. "Seed Germination Ecophysiology of Acacia dealbata Link and Acacia mearnsii De Wild.: Two Invasive Species in the Mediterranean Basin" Sustainability 13, no. 21: 11588. https://doi.org/10.3390/su132111588
APA StyleDessì, L., Podda, L., Brundu, G., Lozano, V., Carrouée, A., Marchante, E., Marchante, H., Petit, Y., Porceddu, M., & Bacchetta, G. (2021). Seed Germination Ecophysiology of Acacia dealbata Link and Acacia mearnsii De Wild.: Two Invasive Species in the Mediterranean Basin. Sustainability, 13(21), 11588. https://doi.org/10.3390/su132111588










