A Review of Chemicals to Produce Activated Carbon from Agricultural Waste Biomass
Abstract
1. Introduction
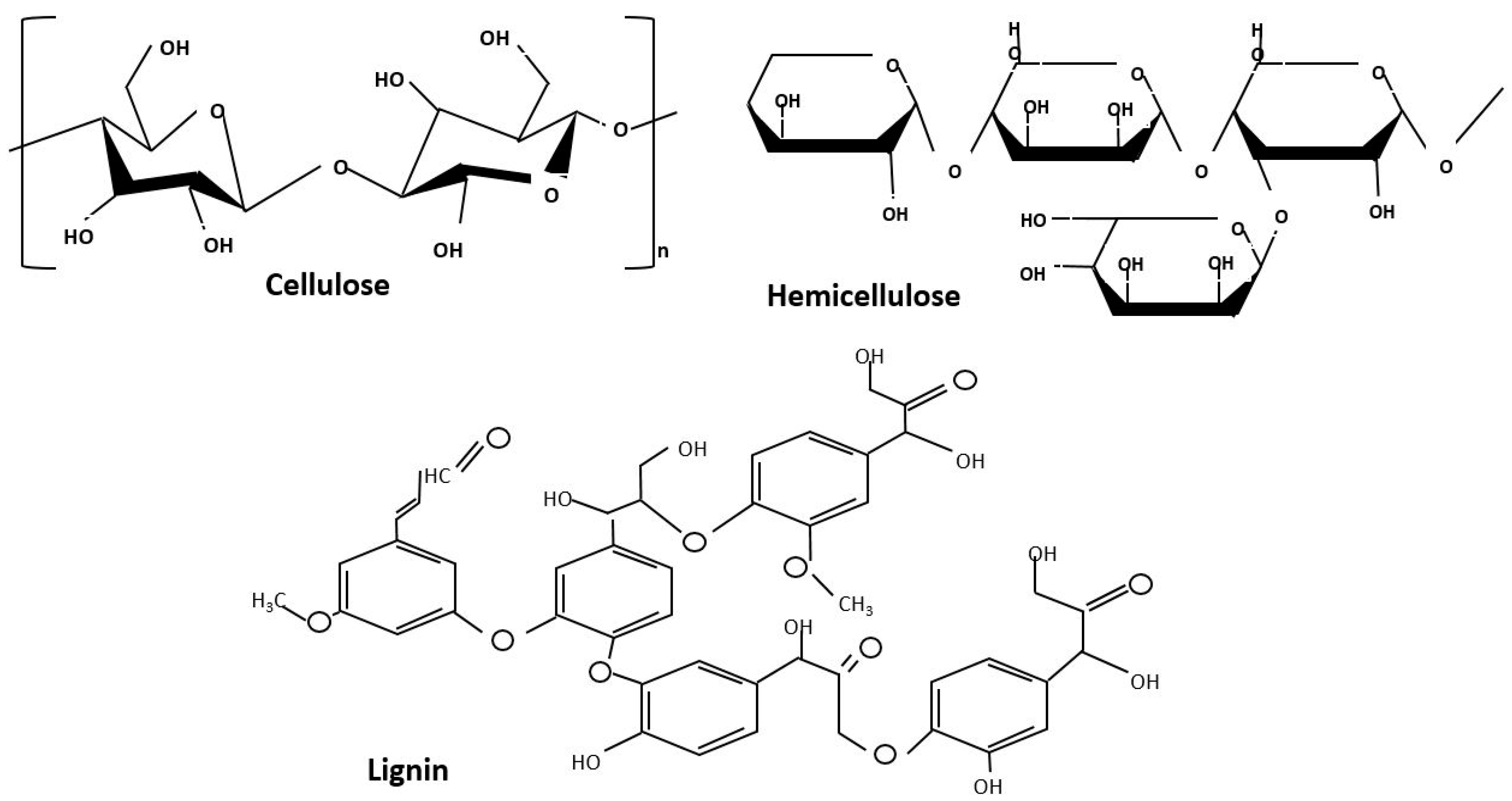
2. Fuel Characterisation for AC Production
3. Modes of Activation
3.1. Chemical Activation
3.1.1. Chemical Activation
3.1.2. Two-Step Activation Process
3.1.3. Microwave Activation Process
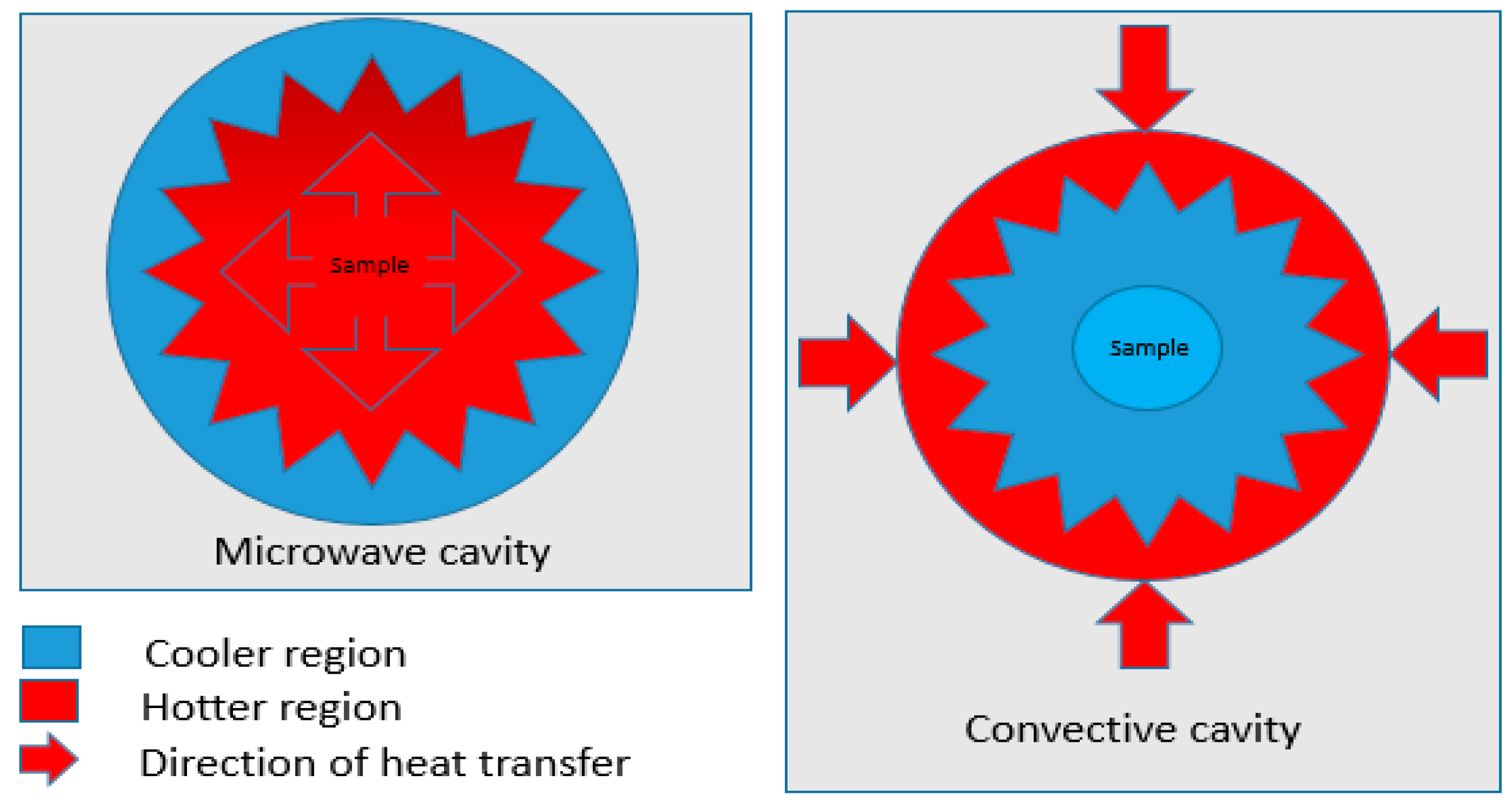
3.2. A Comparison of Activation by Microwave and the Conventional Process
4. Activating Agent Effect
4.1. The Effects of Activating Agents on Renewable and Non-Renewable Precursors
4.2. Acidic Treatment of Agricultural Residues
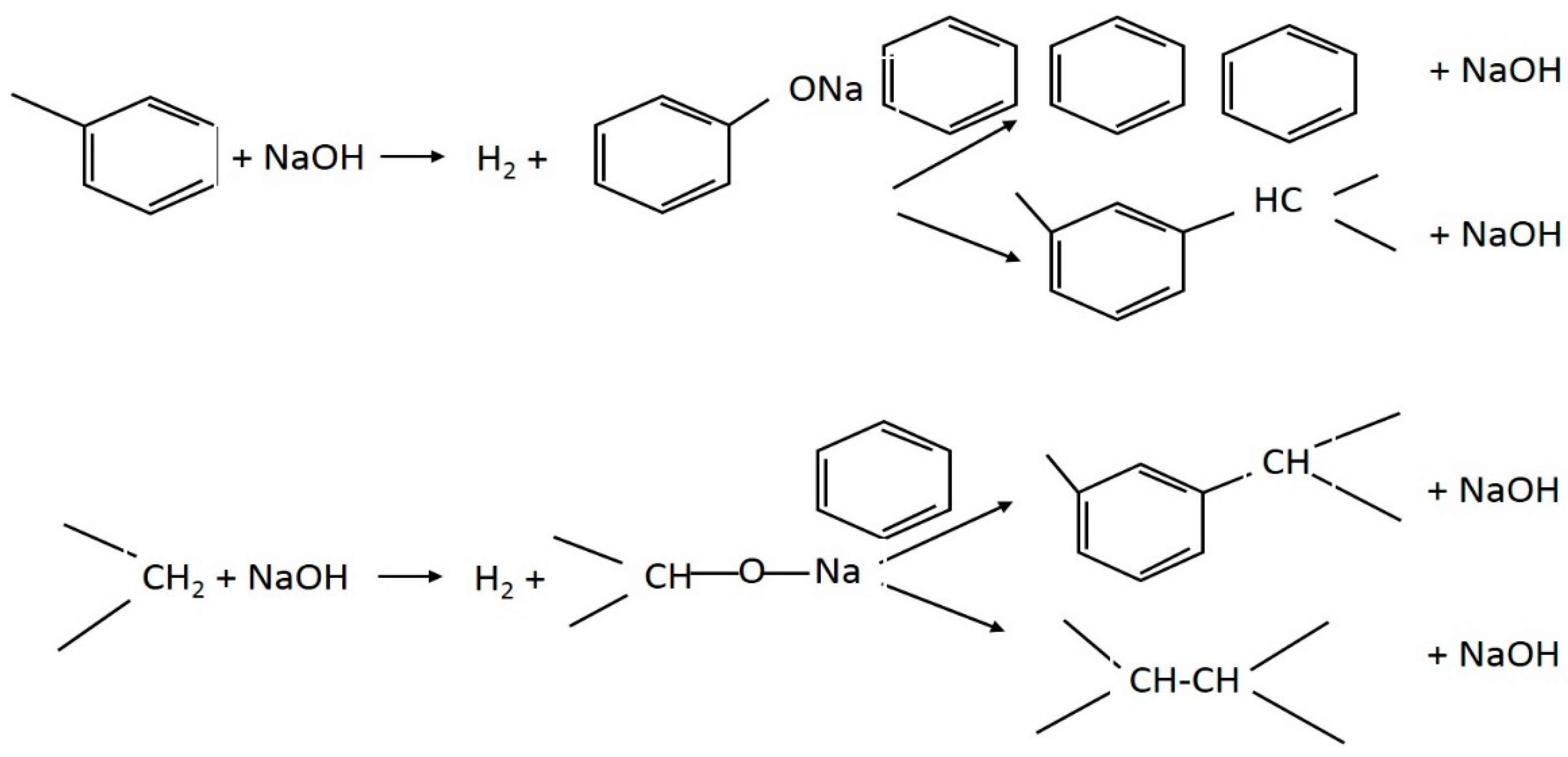
4.3. Base Treatment of Agricultural Residues
4.4. Salts Treatment of Agricultural Residues
5. Characterisation of AC
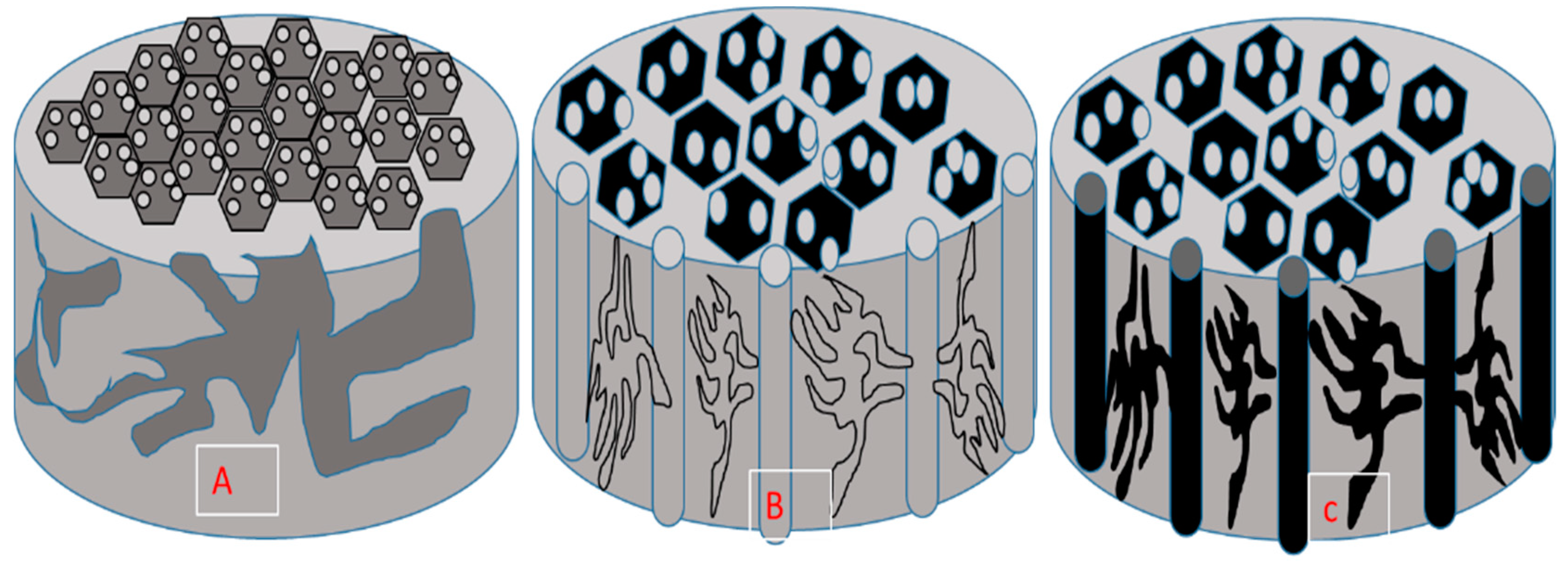
5.1. Surface Chemistry Mechanisms and Morphology
5.2. Adsorption and Kinetics Mechanism
6. Production Process Challenges
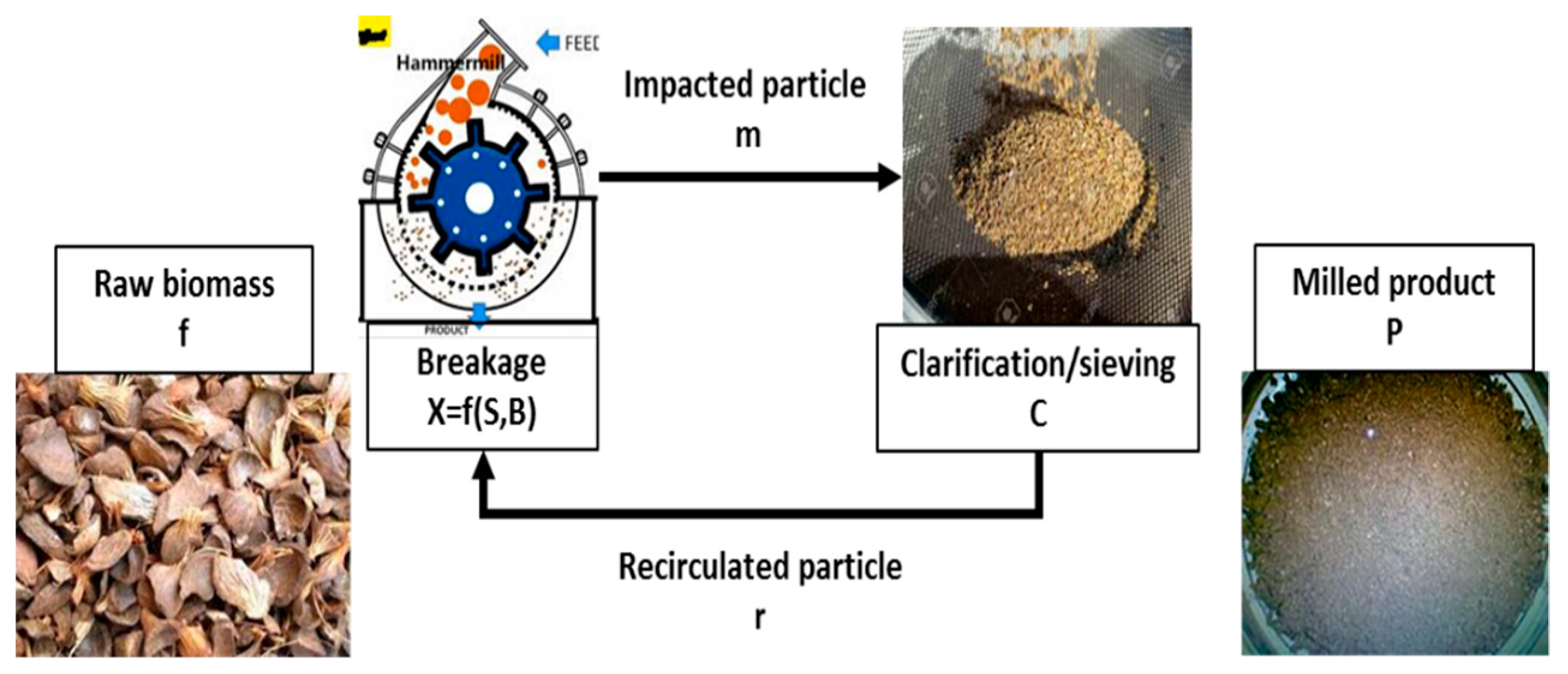
6.1. Washing and Milling
6.2. Microwave Power and Radiation Duration
6.3. Activation Interaction and Temperature
6.4. Tar Deposition
6.5. Equipment Selection and Mode
6.6. Dielectric Properties
7. Scale of Microwave for Use in Preparing AC
8. Summary
Author Contributions
Funding
Conflicts of Interest
References
- Pezoti, O.; Cazetta, A.L.; Bedin, K.C.; Souza, L.S.; Martins, A.C.; Silva, T.L.; Santos Júnior, O.O.; Visentainer, J.V.; Almeida, V.C. NaOH-activated carbon of high surface area produced from guava seeds as a high-efficiency adsorbent for amoxicillin removal: Kinetic, isotherm and thermodynamic studies. Chem. Eng. J. 2016, 288, 778–788. [Google Scholar] [CrossRef]
- Arena, N.; Lee, J.; Clift, R. Life Cycle Assessment of activated carbon production from coconut shells. J. Clean. Prod. 2016, 125, 68–77. [Google Scholar] [CrossRef]
- Lu, Y.; Yuan, J.; Lu, X.; Su, C.; Zhang, Y.; Wang, C.; Cao, X.; Li, Q.; Su, J.; Ittekkot, V.; et al. Major threats of pollution and climate change to global coastal ecosystems and enhanced management for sustainability. Environ. Pollut. 2018, 239, 670–680. [Google Scholar] [CrossRef] [PubMed]
- Rashidi, N.A.; Yusup, S.; Ahmad, M.M.; Mohamed, N.M.; Hameed, B.H. Activated Carbon from the Renewable Agricultural Residues Using Single Step Physical Activation: A Preliminary Analysis. APCBEE Procedia 2012, 3, 84–92. [Google Scholar] [CrossRef]
- Teh, C.Y.; Budiman, P.M.; Shak, K.P.Y.; Wu, T.Y. Recent Advancement of Coagulation-Flocculation and Its Application in Wastewater Treatment. Ind. Eng. Chem. Res. 2016, 55, 4363–4389. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Hogland, W.; Marques, M.; Sillanpää, M. An overview of the modification methods of activated carbon for its water treatment applications. Chem. Eng. J. 2013, 219, 499–511. [Google Scholar] [CrossRef]
- Hadi, P.; Xu, M.; Ning, C.; Sze Ki Lin, C.; McKay, G. A critical review on preparation, characterization and utilization of sludge-derived activated carbons for wastewater treatment. Chem. Eng. J. 2015, 260, 895–906. [Google Scholar] [CrossRef]
- Ling, P.; He, X.; Zhao, N.; Yu, M.; Zhang, X.; Zheng, M.; Zhu, T. Facile preparation of porous carbons from rice husk by microwave heating for supercapacitors. ICMREE 2013 - Proc. 2013 Int. Conf. Mater. Renew. Energy Environ. 2013, 2, 524–528. [Google Scholar]
- Zhenchao, J.; Yuting, Z.; Jiuming, Y.; Yedan, L.; Yang, S.; Jinyao, C.; Lishi, Z. Safety assessment of dietary bamboo charcoal powder: A 90-day subchronic oral toxicity and mutagenicity studies. Food Chem. Toxicol. 2015, 75, 50–57. [Google Scholar] [CrossRef]
- Pavlenko, D.; Giasafaki, D.; Charalambopoulou, G.; Van Geffen, E.; Gerritsen, K.G.F.; Steriotis, T.; Stamatialis, D. Carbon Adsorbents with Dual Porosity for Efficient Removal of Uremic Toxins and Cytokines from Human Plasma. Sci. Rep. 2017, 7, 1–7. [Google Scholar] [CrossRef]
- Bentsen, N.S.; Felby, C.; Thorsen, B.J. Agricultural residue production and potentials for energy and materials services. Prog. Energy Combust. Sci. 2014, 40, 59–73. [Google Scholar] [CrossRef]
- Nabais, J.M.V.; Laginhas, C.; Carrott, M.M.L.R.; Carrott, P.J.M.; Amorós, J.E.C.; Gisbert, A.V.N. Surface and porous characterisation of activated carbons made from a novel biomass precursor, the esparto grass. Appl. Surf. Sci. 2013, 265, 919–924. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, X.; Zhong, T. Pollution and health risk assessment of heavy metals in urban soil in China. Hum. Ecol. Risk Assess. An Int. J. 2016, 22, 424–434. [Google Scholar] [CrossRef]
- Research, G.V. Activated Carbon Market Analysis and Segment Forecasts to 2020; Grand View Research: San Francisco, CA, USA, 2014. [Google Scholar]
- Chand Bansal, R.; Goyal, M. Activated Carbon Adsorption; CRC Press: Boca Raton, FL, USA, 2005; ISBN 978-0-8247-5344-3. [Google Scholar]
- Xiao, H.; Peng, H.; Deng, S.; Yang, X.; Zhang, Y.; Li, Y. Preparation of activated carbon from edible fungi residue by microwave assisted K2CO3 activation-Application in reactive black 5 adsorption from aqueous solution. Bioresour. Technol. 2012, 111, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.; Lee, J.; Lee, K.-H. Microwave heating of carbon-based solid materials. Carbon Lett. 2014, 15, 15–24. [Google Scholar] [CrossRef]
- Kumar, S.B.; Mathew, K.T.; Raveendranath, U.; Augustine, P. Dielectric properties of certain biological materials at microwave frequencies. J Microw Power Electromagn Energy 2001, 36, 67–75. [Google Scholar] [CrossRef]
- Menéndes, J.A.; Menéndez, E.M.; Parra, J.B.; Pis, J.J. Thermal Treatment of active carbons: A comparison between microwave and electrical heating. pdf. J. Microw. Power Electromagn. Energy 1999, 34, 137–143. [Google Scholar] [CrossRef]
- Namazi, A.B.; Allen, D.G.; Jia, C.Q. Probing microwave heating of lignocellulosic biomasses. J. Anal. Appl. Pyrolysis 2015, 112, 121–128. [Google Scholar] [CrossRef]
- Klinger, J.L.; Westover, T.L.; Emerson, R.M.; Williams, C.L.; Hernandez, S.; Monson, G.D.; Ryan, J.C. Effect of biomass type, heating rate, and sample size on microwave-enhanced fast pyrolysis product yields and qualities. Appl. Energy 2018, 228, 535–545. [Google Scholar] [CrossRef]
- Lahti, R.; Bergna, D.; Romar, H.; Tuuttila, T.; Hu, T.; Lassi, U. Physico-chemical properties and use of waste biomass-derived activated carbons. Chem. Eng. Trans. 2017, 57, 43–48. [Google Scholar]
- Danish, M.; Ahmad, T. A review on utilization of wood biomass as a sustainable precursor for activated carbon production and application. Renew. Sustain. Energy Rev. 2018, 87, 1–21. [Google Scholar] [CrossRef]
- González-García, P. Activated carbon from lignocellulosics precursors: A review of the synthesis methods, characterization techniques and applications. Renew. Sustain. Energy Rev. 2018, 82, 1393–1414. [Google Scholar] [CrossRef]
- Martí-Rosselló, T.; Li, J.; Leo, L. Kinetic models for biomass pyrolysis. Arch Ind Biotechnol 2016, 1, 4–7. [Google Scholar]
- Aziz, M.; Kurniawan, T.; Oda, T.; Kashiwagi, T. Advanced power generation using biomass wastes from palm oil mills. Appl. Therm. Eng. 2017, 114, 1378–1386. [Google Scholar] [CrossRef]
- Ben-Iwo, J.; Manovic, V.; Longhurst, P. Biomass resources and biofuels potential for the production of transportation fuels in Nigeria. Renew. Sustain. Energy Rev. 2016, 63, 172–192. [Google Scholar] [CrossRef]
- Uemura, Y.; Omar, W.N.; Tsutsui, T.; Yusup, S.B. Torrefaction of oil palm wastes. Fuel 2011, 90, 2585–2591. [Google Scholar] [CrossRef]
- Rashidi, N.A.; Yusup, S. A review on recent technological advancement in the activated carbon production from oil palm wastes. Chem. Eng. J. 2017, 314, 277–290. [Google Scholar] [CrossRef]
- Hidayu, A.R.; Muda, N. Preparation and Characterization of Activated Carbon from Palm Kernel Shell. IOP Conf. Ser. Mater. Sci. Eng. 2017, 226, 012156. [Google Scholar]
- Abbas, A.F.; Ahmed, M.J. Mesoporous activated carbon from date stones (Phoenix dactylifera L.) by one-step microwave assisted K2CO3 pyrolysis. J. Water Process Eng. 2016, 9, 201–207. [Google Scholar]
- Chen, H. Biotechnology of lignocellulose: Theory and practice. In Biotechnology of Lignocellulose: Theory and Practice; Springer: Berlin, Germany, 2014; pp. 1–511. ISBN 9789400768987. [Google Scholar]
- Chowdhury, Z.Z.; Hamid, S.B.A.; Das, R.; Hasan, M.R.; Sharifuddin, M.Z.; Khalid, K.; Uddin, M.N. Preparation of Carbonaceous Adsorbents from Lignocellulosic Biomass and Their Use in Removal of Contaminants from Aqueous Solution. BioResources 2013, 8, 6523–6555. [Google Scholar] [CrossRef]
- Sharma, B.; Sarkar, A.; Singh, P.; Singh, R.P. Agricultural utilization of biosolids: A review on potential effects on soil and plant grown. Waste Manag. 2017, 64, 117–132. [Google Scholar] [CrossRef] [PubMed]
- Anyaoha, K.E.; Sakrabani, R.; Patchigolla, K.; Mouazen, A.M. Critical evaluation of oil palm fresh fruit bunch solid wastes as soil amendments: Prospects and challenges. Resour. Conserv. Recycl. 2018, 136, 399–409. [Google Scholar] [CrossRef]
- Shamsuddin, M.S.; Yusoff, N.R.N.; Sulaiman, M.A. Synthesis and Characterization of Activated Carbon Produced from Kenaf Core Fiber Using H3PO4 Activation. Procedia Chem. 2016, 19, 558–565. [Google Scholar] [CrossRef]
- Kongsomart, B.; Li, L.; Takarada, T. Preparation of Activated Carbons From Teak Sawdust Using Chicken Dropping Compost and Empty Fruit Bunch. Int. J. Biomass Renewables 2015, 4, 1–7. [Google Scholar]
- Kan, T.; Strezov, V.; Evans, T.J. Lignocellulosic biomass pyrolysis: A review of product properties and effects of pyrolysis parameters. Renew. Sustain. Energy Rev. 2016, 57, 126–1140. [Google Scholar] [CrossRef]
- Sun, K.; Leng, C.Y.; Jiang, J.C.; Bu, Q.; Lin, G.F.; Lu, X.C.; Zhu, G.Z. Microporous activated carbons from coconut shells produced by self-activation using the pyrolysis gases produced from them, that have an excellent electric double layer performance. Xinxing Tan Cailiao/New Carbon Mater. 2017, 32, 451–459. [Google Scholar] [CrossRef]
- Hirunpraditkoon, S.; Tunthong, N.; Ruangchai, A.; Nuithitikul, K. Adsorption Capacities of Activated Carbons Prepared from Bamboo by KOH Activation. World Acad. Sci. Eng. Technol. 2011, 5, 591–595. [Google Scholar]
- Kabenge, I.; Omulo, G.; Banadda, N.; Seay, J.; Zziwa, A.; Kiggundu, N. Characterization of Banana Peels Wastes as Potential Slow Pyrolysis Feedstock. Ccsenet.Org 2018, 11, 14–24. [Google Scholar] [CrossRef]
- Daud, Z.; Kassim, A.S.M.; Aripin, A.M.; Awang, H.; Hatta, M.Z.M. Chemical Composition and Morphological of Cocoa Pod Husks and Cassava Peels for Pulp and Paper Production. Aust. J. Basic Appl. Sci. 2013, 7, 406–411. [Google Scholar]
- Mohd Iqbaldin, M.N.; Khudzir, I.; Mohd Azlan, M.I.; Zaidi, A.G.; Surani, B.; Zubri, Z. Properties of coconut shell activated carbon. J. Trop. For. Sci. 2013, 25, 497–503. [Google Scholar]
- Abdolali, A.; Ngo, H.H.; Guo, W.; Zhou, J.L.; Du, B.; Wei, Q.; Wang, X.C.; Nguyen, P.D. Characterization of a multi-metal binding biosorbent: Chemical modification and desorption studies. Bioresour. Technol. 2015, 193, 477–487. [Google Scholar] [CrossRef] [PubMed]
- Pangavhane, D.R.; Tare, S. Grape stalk briquettes as an alternative feedstock of biomass gasifiers. Int. Energy J. 2012, 13, 11–20. [Google Scholar]
- Onochie, U.P.; Obanor, A.I.; Aliu, S.A.; Ighodaro, O.O. Proximate and Ultimate Analysis of Fuel Pellets From Oil Palm Residues. Niger. J. Technol. 2017, 36, 987–990. [Google Scholar]
- Plaza, M.G.; González, A.S.; Pis, J.J.; Rubiera, F.; Pevida, C. Production of microporous biochars by single-step oxidation: Effect of activation conditions on CO2 capture. Appl. Energy 2014, 114, 551–562. [Google Scholar] [CrossRef]
- Zhou, H. Research Method, 1st ed.; Springer: Singapore, 2017; ISBN 978-981-10-3826-6. [Google Scholar]
- Braz, C.E.; Crnkovic, P.M. Physical – Chemical Characterization of Biomass Samples for Application in Pyrolysis Process. Chem. Eng. Trans. 2014, 37, 523–528. [Google Scholar]
- Huang, Y.F.; Chiueh, P.T.; Kuan, W.H.; Lo, S.L. Microwave pyrolysis of lignocellulosic biomass: Heating performance and reaction kinetics. Energy 2016, 100, 137–144. [Google Scholar] [CrossRef]
- Gurten, I.I.; Ozmak, M.; Yagmur, E.; Aktas, Z. Preparation and characterisation of activated carbon from waste tea using K2CO3. Biomass and Bioenergy 2012, 37, 73–81. [Google Scholar] [CrossRef]
- Osman, N.B.; Shamsuddin, N.; Uemura, Y. Activated Carbon of Oil Palm Empty Fruit Bunch (EFB); Core and Shaggy. Procedia Eng. 2016, 148, 758–764. [Google Scholar] [CrossRef]
- Claoston, N.; Samsuri, A.W.; Ahmad Husni, M.H.; Mohd Amran, M.S. Effects of pyrolysis temperature on the physicochemical properties of empty fruit bunch and rice husk biochars. Waste Manag. Res. 2014, 32, 331–339. [Google Scholar] [CrossRef]
- Pathak, P.D.; Davgane, S.A.; Kulkarni, B.D. Fruit peel waste as a novel low-cost bio adsorbent. Rev. Chem. Eng. 2015, 31, 361–381. [Google Scholar] [CrossRef]
- Calvo-Muñoz, E.M.; García-Mateos, F.J.; Rosas, J.M.; Rodríguez-Mirasol, J.; Cordero, T. Biomass Waste Carbon Materials as adsorbents for CO2 Capture under Post-Combustion Conditions. Front. Mater. 2016, 3, 1–14. [Google Scholar] [CrossRef]
- Olorundare, O.F.; Msagati, T.A.M.; Krause, R.W.M.; Okonkwo, J.O.; Mamba, B.B. Activated carbon from lignocellulosic waste residues: Effect of activating agent on porosity characteristics and use as adsorbents for organic species. Water. Air. Soil Pollut. 2014, 225, 1876. [Google Scholar] [CrossRef]
- Girgis, B.S.; Soliman, A.M.; Fathy, N.A. Development of micro-mesoporous carbons from several seed hulls under varying conditions of activation. Microporous Mesoporous Mater. 2011, 142, 518–525. [Google Scholar] [CrossRef]
- Arami-Niya, A.; Wan Daud, W.M.A.; Mjalli, F.S.; Abnisa, F.; Shafeeyan, M.S. Production of microporous palm shell based activated carbon for methane adsorption: Modeling and optimization using response surface methodology. Chem. Eng. Res. Des. 2012, 90, 776–784. [Google Scholar] [CrossRef]
- Ooi, C.H.; Cheah, W.K.; Sim, Y.L.; Pung, S.Y.; Yeoh, F.Y. Conversion and characterization of activated carbon fiber derived from palm empty fruit bunch waste and its kinetic study on urea adsorption. J. Environ. Manag. 2017, 197, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Wahi, R.; Zuhaidi, N.F.Q.; Yusof, Y.; Jamel, J.; Kanakaraju, D.; Ngaini, Z. Chemically treated microwave-derived biochar: An overview. Biomass Bioenergy 2017, 107, 411–421. [Google Scholar] [CrossRef]
- Farma, R.; Fatjrin, D.; Deraman, M.A. Physical properties of activated carbon from fibers of oil palm empty fruit bunches by microwave assisted potassium hydroxide activation. AIP Conf. Proc. 2017, 1801, 040001. [Google Scholar]
- Ahmida, K.; Darmoon, M.; Al-tohami, F.; Erhayem, M.; Zidan, M. Effect of Physical and Chemical Preparation on Characteristics of Activated Carbon from Agriculture Solid Waste and their Potential Application. In Proceedings of the International Conference on Chemical, Civil and Environmental Engineering, Istanbul, Turkey, 5–6 June 2015. [Google Scholar]
- Hoseinzadeh Hesas, R.; Arami-Niya, A.; Wan Daud, W.M.A.; Sahu, J.N. Preparation of granular activated carbon from oil palm shell by microwave-induced chemical activation: Optimisation using surface response methodology. Chem. Eng. Res. Des. 2013, 91, 2447–2456. [Google Scholar] [CrossRef]
- Xin-hui, D.; Srinivasakannan, C.; Jin-hui, P.; Li-bo, Z.; Zheng-yong, Z. Comparison of activated carbon prepared from Jatropha hull by conventional heating and microwave heating. Biomass Bioenergy 2011, 35, 3920–3926. [Google Scholar] [CrossRef]
- Moreno-Castilla, C.; Carrasco-Marin, F.; Lopez-Ramon, M.V.; Alvarez-Merino M., A. Chemical and physical activation of olive-mill waste water to produce activated carbons. Carbon 2001, 39, 1415–1420. [Google Scholar] [CrossRef]
- Hernández-Montoya, V.; García-Servin, J.; Bueno-López, J.I. Thermal Treatments and Activation Procedures Used in the Preparation of Activated Carbons; Montoya, D.V.H., Ed.; InTech: Mexico City, Mexico, 2012; ISBN 978-953-51-0197-0. [Google Scholar]
- Liu, J.; Deng, Y.; Li, X.; Wang, L. Promising Nitrogen-Rich Porous Carbons Derived from One-Step Calcium Chloride Activation of Biomass-Based Waste for High Performance Supercapacitors. ACS Sustain. Chem. Eng. 2016, 4, 177–187. [Google Scholar] [CrossRef]
- Bachrun, S.; AyuRizka, N.; Annisa, S.; Arif, H. Preparation and characterization of activated carbon from sugarcane bagasse by physical activation with CO2 gas. IOP Conf. Ser. Mater. Sci. Eng. 2016, 105, 012027. [Google Scholar] [CrossRef]
- Oginni, O.; Singh, K.; Oporto, G.; Dawson-Andoh, B.; McDonald, L.; Sabolsky, E. Influence of one-step and two-step KOH activation on activated carbon characteristics. Bioresour. Technol. Reports 2019, 7, 100266. [Google Scholar] [CrossRef]
- Oghbaei, M.; Mirzaee, O. Microwave versus conventional sintering: A review of fundamentals, advantages and applications. J. Alloys Compd. 2010, 494, 175–189. [Google Scholar] [CrossRef]
- Yao, Z.T.; Ji, X.S.; Sarker, P.K.; Tang, J.H.; Ge, L.Q.; Xia, M.S.; Xi, Y.Q. A comprehensive review on the applications of coal fly ash. Earth-Science Rev. 2015, 141, 105–121. [Google Scholar] [CrossRef]
- Gan, C.; Liu, Y.; Tan, X.; Wang, S.; Zeng, G.; Zheng, B.; Li, T.; Jiang, Z.; Liu, W. Effect of porous zinc-biochar nanocomposites on Cr(VI) adsorption from aqueous solution. RSC Adv. 2015, 5, 35107–35115. [Google Scholar] [CrossRef]
- Domínguez, A.; Menéndez, J.A.; Fernández, Y.; Pis, J.J.; Nabais, J.M.V.; Carrott, P.J.M.; Carrott, M.M.L.R. Conventional and microwave induced pyrolysis of coffee hulls for the production of a hydrogen rich fuel gas. J. Anal. Appl. Pyrolysis 2007, 79, 128–135. [Google Scholar] [CrossRef]
- Fidalgo, B.; Williams, H.M.; Dawson, E.A.; Parkes, G.M.B. Conventional and microwave-heated oxygen pulsing techniques on metal-doped activated carbons. J. Porous Mater. 2014, 21, 81–89. [Google Scholar] [CrossRef]
- Nomanbhay, S.; Ong, M.Y. A Review of Microwave-Assisted Reactions for Biodiesel Production. Bioengineering 2017, 4, 57. [Google Scholar] [CrossRef]
- Gadkari, S.; Fidalgo, B.; Gu, S. Numerical investigation of microwave-assisted pyrolysis of lignin. Fuel Process. Technol. 2017, 156, 473–484. [Google Scholar] [CrossRef]
- Puziy, A.M.; Poddubnaya, O.I.; Martínez-Alonso, A.; Suárez-García, F.; Tascón, J.M.D. Surface chemistry of phosphorus-containing carbons of lignocellulosic origin. Carbon N. Y. 2005, 43, 2857–2868. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Preparation of oil palm (Elaeis) empty fruit bunch activated carbon by microwave-assisted KOH activation for the adsorption of methylene blue. Desalination 2011, 275, 302–305. [Google Scholar] [CrossRef]
- Kopac, T.; Kırca, Y.; Toprak, A. Synthesis and characterization of KOH/boron modified activated carbons from coal and their hydrogen sorption characteristics. Int. J. Hydrogen Energy 2017, 42, 23606–23616. [Google Scholar] [CrossRef]
- Danish, M.; Hashim, R.; Ibrahim, M.N.M.; Sulaiman, O. Optimized preparation for large surface area activated carbon from date (Phoenix dactylifera L.) stone biomass. Biomass Bioenergy 2014, 61, 167–178. [Google Scholar] [CrossRef]
- Köseoʇlu, E.; Akmil-Başar, C. Preparation, structural evaluation and adsorptive properties of activated carbon from agricultural waste biomass. Adv. Powder Technol. 2015, 26, 811–818. [Google Scholar] [CrossRef]
- Gao, L.; Dong, F.Q.; Dai, Q.W.; Zhong, G.Q.; Halik, U.; Lee, D.J. Coal tar residues based activated carbon: Preparation and characterization. J. Taiwan Inst. Chem. Eng. 2016, 63, 166–169. [Google Scholar] [CrossRef]
- Franca, A.S.; Oliveira, L.S.; Nunes, A.A.; Alves, C.C.O. Microwave assisted thermal treatment of defective coffee beans press cake for the production of adsorbents. Bioresour. Technol. 2010, 101, 1068–1074. [Google Scholar] [CrossRef]
- Huang, L.; Sun, Y.; Wang, W.; Yue, Q.; Yang, T. Comparative study on characterization of activated carbons prepared by microwave and conventional heating methods and application in removal of oxytetracycline (OTC). Chem. Eng. J. 2011, 171, 1446–1453. [Google Scholar] [CrossRef]
- Hoseinzadeh Hesas, R.; Wan Daud, W.M.A.; Sahu, J.N.; Arami-Niya, A. The effects of a microwave heating method on the production of activated carbon from agricultural waste: A review. J. Anal. Appl. Pyrolysis 2013, 100, 1–11. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Porous structure and adsorptive properties of pineapple peel based activated carbons prepared via microwave assisted KOH and K2CO3 activation. Microporous Mesoporous Mater. 2012, 148, 191–195. [Google Scholar] [CrossRef]
- Szymański, G.S.; Karpiński, Z.; Biniak, S.; Świątkowski, A. The effect of the gradual thermal decomposition of surface oxygen species on the chemical and catalytic properties of oxidized activated carbon. Carbon N. Y. 2002, 40, 2627–2639. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Microwave assisted preparation of activated carbon from pomelo skin for the removal of anionic and cationic dyes. Chem. Eng. J. 2011, 173, 385–390. [Google Scholar] [CrossRef]
- Somorjai, G.A.; Li, Y. Impact of surface chemistry. Proc. Natl. Acad. Sci. USA 2011, 108, 917–924. [Google Scholar] [CrossRef]
- Wang, T.; Tan, S.; Liang, C. Preparation and characterization of activated carbon from wood via microwave-induced ZnCl2 activation. Carbon N. Y. 2009, 47, 1880–1883. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Preparation, characterization and evaluation of adsorptive properties of orange peel based activated carbon via microwave induced K2CO3 activation. Bioresour. Technol. 2012, 104, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Deng, H.; Zhang, G.; Xu, X.; Tao, G.; Dai, J. Optimization of preparation of activated carbon from cotton stalk by microwave assisted phosphoric acid-chemical activation. J. Hazard. Mater. 2010, 182, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Foo, K.Y.; Hameed, B.H. Utilization of oil palm biodiesel solid residue as renewable sources for preparation of granular activated carbon by microwave induced KOH activation. Bioresour. Technol. 2013, 130, 696–702. [Google Scholar] [CrossRef] [PubMed]
- Foo, K.Y.; Hameed, B.H. Utilization of rice husks as a feedstock for preparation of activated carbon by microwave induced KOH and K2CO3 activation. Bioresour. Technol. 2011, 102, 9814–9817. [Google Scholar] [CrossRef]
- Hoseinzadeh Hesas, R.; Arami-Niya, A.; Wan Daud, W.M.A.; Sahu, J.N. Microwave-assisted production of activated carbons from oil palm shell in the presence of CO2 or N2 for CO2 adsorption. J. Ind. Eng. Chem. 2015, 24, 196–205. [Google Scholar] [CrossRef]
- Allwar, A.; Hartati, R.; Fatimah, I. Effect of nitric acid treatment on activated carbon derived from oil palm shell. In Proceedings of the International Conference on Chemistry, Chemical Process and Engineering, Yogyakarta, Indonesia, 15–16 November 2016; Volume 1823. [Google Scholar]
- Qin, C.; Clarke, K.; Li, K. Interactive forces between lignin and cellulase as determined by atomic force microscopy. Biotechnol. Biofuels 2014, 7, 65. [Google Scholar] [CrossRef]
- Yakout, S.M.; Sharaf El-Deen, G. Characterization of activated carbon prepared by phosphoric acid activation of olive stones. Arab. J. Chem. 2016, 9, S1155–S1162. [Google Scholar] [CrossRef]
- Örkün, Y.; Karatepe, N.; Yavuz, R. Influence of temperature and impregnation ratio of H3PO4 on the production of activated carbon from hazelnut shell. Acta Phys. Pol. A 2012, 121, 277–280. [Google Scholar] [CrossRef]
- Lee, H.-M.; An, K.-H.; Kim, B.-J. Effects of carbonization temperature on pore development in polyacrylonitrile-based activated carbon nanofibers. Carbon Lett. 2014, 15, 146–150. [Google Scholar] [CrossRef]
- Ello, A.S.; De Souza, L.K.C.; Trokourey, A.; Jaroniec, M. Development of microporous carbons for CO2 capture by KOH activation of African palm shells. J. CO2 Util. 2013, 2, 35–38. [Google Scholar] [CrossRef]
- Wu, F.C.; Tseng, R.L. High adsorption capacity NaOH-activated carbon for dye removal from aqueous solution. J. Hazard. Mater. 2008, 152, 1256–1267. [Google Scholar] [CrossRef]
- Tseng, R.L. Mesopore control of high surface area NaOH-activated carbon. J. Colloid Interface Sci. 2006, 303, 494–502. [Google Scholar] [CrossRef]
- Liu, Z.; Huang, Y.; Zhao, G. Preparation and characterization of activated carbon fibers from liquefied wood by ZnCl2 activation. BioResources 2016, 11, 3178–3190. [Google Scholar] [CrossRef]
- Şahin, Ö.; Saka, C.; Ceyhan, A.A.; Baytar, O. Preparation of High Surface Area Activated Carbon from Elaeagnus angustifolia Seeds by Chemical Activation with ZnCl2 in One-Step Treatment and its Iodine Adsorption. Sep. Sci. Technol. 2015, 50, 886–891. [Google Scholar] [CrossRef]
- Arampatzidou, A.C.; Deliyanni, E.A. Comparison of activation media and pyrolysis temperature for activated carbons development by pyrolysis of potato peels for effective adsorption of endocrine disruptor bisphenol-A. J. Colloid Interface Sci. 2016, 466, 101–112. [Google Scholar] [CrossRef]
- Okman, I.; Karagöz, S.; Tay, T.; Erdem, M. Activated carbons from grape seeds by chemical activation with potassium carbonate and potassium hydroxide. Appl. Surf. Sci. 2014, 293, 138–142. [Google Scholar] [CrossRef]
- Fu, K.; Yue, Q.; Gao, B.; Wang, Y.; Li, Q. Activated carbon from tomato stem by chemical activation with FeCl2. Colloids Surfaces A Physicochem. Eng. Asp. 2017, 529, 842–849. [Google Scholar] [CrossRef]
- Lee, L.Z.; Ahmad Zaini, M.A. Metal chloride salts in the preparation of activated carbon and their hazardous outlook. Desalin. Water Treat. 2016, 57, 16078–16085. [Google Scholar] [CrossRef]
- Fu, K.; Yue, Q.; Gao, B.; Sun, Y.; Wang, Y.; Li, Q.; Zhao, P.; Chen, S. Physicochemical and adsorptive properties of activated carbons from Arundo donax Linn utilizing different iron salts as activating agents. J. Taiwan Inst. Chem. Eng. 2014, 45, 3007–3015. [Google Scholar] [CrossRef]
- Rufford, T.E.; Hulicova-Jurcakova, D.; Zhu, Z.; Lu, G.Q. A comparative study of chemical treatment by FeCl3, MgCl2, and ZnCl2 on microstructure, surface chemistry, and double-layercapacitance of carbons from waste biomass. J. Mater. Res. 2010, 25, 1451–1459. [Google Scholar] [CrossRef]
- Ahmed, M.J. Preparation of Activated Carbons from Date Stones by Chemical Activation Method Using FeCl3 and ZnCl2 as Activating Agents. J. Eng. 2011, 17, 1007–1022. [Google Scholar]
- Bedia, J.; Belver, C.; Ponce, S.; Rodriguez, J.; Rodriguez, J.J. Adsorption of antipyrine by activated carbons from FeCl3-activation of Tara gum. Chem. Eng. J. 2018, 333, 58–65. [Google Scholar] [CrossRef]
- Nahil, M.A.; Williams, P.T. Pore characteristics of activated carbons from the phosphoric acid chemical activation of cotton stalks. Biomass and Bioenergy 2012, 37, 142–149. [Google Scholar] [CrossRef]
- Khedr, S.; Shouman, M.; Fathy, N.; Attia, A. Effect of Physical and Chemical Activation on the Removal of Hexavalent Chromium Ions Using Palm Tree Branches. ISRN Environ. Chem. 2014, 2014, 1–10. [Google Scholar] [CrossRef]
- Liu, Q.S.; Zheng, T.; Wang, P.; Guo, L. Preparation and characterization of activated carbon from bamboo by microwave-induced phosphoric acid activation. Ind. Crops Prod. 2010, 31, 233–238. [Google Scholar] [CrossRef]
- Lim, W.C.; Srinivasakannan, C.; Balasubramanian, N. Activation of palm shells by phosphoric acid impregnation for high yielding activated carbon. J. Anal. Appl. Pyrolysis 2010, 88, 181–186. [Google Scholar] [CrossRef]
- Jun, T.Y.; Arumugam, S.D.; Latip, N.H.A.; Latif, A.M.A.P.A. Effect of Activation Temperature and Heating Duration on Physical Characteristics of Activated Carbon Prepared from Agriculture Waste. EnvironmentAsia 2010, 7, 104–111. [Google Scholar]
- Ibrahim, T.; Moctar, B.L.; Tomkouani, K.; Gbandi, D.; Victor, D.K.; Phinthè, N. Kinetics of the Adsorption of Anionic and Cationic Dyes in Aqueous Solution by Low-Cost Activated Carbons Prepared from Sea Cake and Cotton Cake. Am. Chem. Sci. J. 2014, 4, 38–57. [Google Scholar] [CrossRef]
- Basta, A.H.; Fierro, V.; El-Saied, H.; Celzard, A. 2-Steps KOH activation of rice straw: An efficient method for preparing high-performance activated carbons. Bioresour. Technol. 2009, 100, 3941–3947. [Google Scholar] [CrossRef] [PubMed]
- Kundu, A.; Gupta, B.S.; Hashim, M.A.; Sahu, J.N.; Mujawar, M.; Redzwan, G. Optimisation of the process variables in production of activated carbon by microwave heating. RSC Adv. 2015, 5, 35899–35908. [Google Scholar] [CrossRef]
- Yagmur, E.; Ozmak, M.; Aktas, Z. A novel method for production of activated carbon from waste tea by chemical activation with microwave energy. Fuel 2008, 87, 3278–3285. [Google Scholar] [CrossRef]
- Girgis, B.S.; Smith, E.; Louis, M.M.; El-Hendawy, A.N.A. Pilot production of activated carbon from cotton stalks using H3PO4. J. Anal. Appl. Pyrolysis 2009, 86, 180–184. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Factors affecting the carbon yield and adsorption capability of the mangosteen peel activated carbon prepared by microwave assisted K2CO3 activation. Chem. Eng. J. 2012, 180, 66–74. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Mesoporous activated carbon from wood sawdust by K2CO3 activation using microwave heating. Bioresour. Technol. 2012, 111, 425–432. [Google Scholar] [CrossRef]
- Deng, H.; Li, G.; Yang, H.; Tang, J.; Tang, J. Preparation of activated carbons from cotton stalk by microwave assisted KOH and K2CO3 activation. Chem. Eng. J. 2010, 163, 373–381. [Google Scholar] [CrossRef]
- Cruz, G.; Pirilä, M.; Huuhtanen, M.; Carrión, L.; Alvarenga, E.; Keiski, R.L. Production of Activated Carbon from Cocoa (Theobroma cacao) Pod Husk. J. Civ. Environ. Eng. 2012, 02, 109. [Google Scholar] [CrossRef]
- Abechi, S.E.; Gimba, C.E.; Uzairu, A.; Dallatu, Y.A. Preparation and Characterization of Activated Carbon from Palm Kernel Shell by Chemical Activation. Res. J. Chem. Sci. 2013, 3, 54–61. [Google Scholar]
- Foo, K.Y.; Hameed, B.H. Microwave-assisted preparation of oil palm fiber activated carbon for methylene blue adsorption. Chem. Eng. J. 2011, 166, 792–795. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Coconut husk derived activated carbon via microwave induced activation: Effects of activation agents, preparation parameters and adsorption performance. Chem. Eng. J. 2012, 184, 57–65. [Google Scholar] [CrossRef]
- Cazetta, A.L.; Vargas, A.M.M.; Nogami, E.M.; Kunita, M.H.; Guilherme, M.R.; Martins, A.C.; Silva, T.L.; Moraes, J.C.G.; Almeida, V.C. NaOH-activated carbon of high surface area produced from coconut shell: Kinetics and equilibrium studies from the methylene blue adsorption. Chem. Eng. J. 2011, 174, 117–125. [Google Scholar] [CrossRef]
- Youssef, A.M.; Ahmed, A.I.; Amin, M.I.; El-Banna, U.A. Adsorption of lead by activated carbon developed from rice husk. Desalin. Water Treat. 2015, 54, 1694–1707. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. A cost effective method for regeneration of durian shell and jackfruit peel activated carbons by microwave irradiation. Chem. Eng. J. 2012, 193–194, 404–409. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Preparation of activated carbon by microwave heating of langsat (Lansium domesticum) empty fruit bunch waste. Bioresour. Technol. 2012, 116, 522–525. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Potential of jackfruit peel as precursor for activated carbon prepared by microwave induced NaOH activation. Bioresour. Technol. 2012, 112, 143–150. [Google Scholar] [CrossRef]
- Gao, J.; Qin, Y.; Zhou, T.; Cao, D.; Xu, P.; Hochstetter, D.; Wang, Y. Adsorption of methylene blue onto activated carbon produced from tea (Camellia sinensis L.) seed shells: Kinetics, equilibrium, and thermodynamics studies. J. Zhejiang Univ. Sci. B 2013, 14, 650–658. [Google Scholar]
- Dural, M.U.; Cavas, L.; Papageorgiou, S.K.; Katsaros, F.K. Methylene blue adsorption on activated carbon prepared from Posidonia oceanica (L.) dead leaves: Kinetics and equilibrium studies. Chem. Eng. J. 2011, 168, 77–85. [Google Scholar] [CrossRef]
- Theydan, S.K.; Ahmed, M.J. Optimization of preparation conditions for activated carbons from date stones using response surface methodology. Powder Technol. 2012, 224, 101–108. [Google Scholar] [CrossRef]
- Boonpoke, A.; Chiarakorn, S.; Laosiripojana, N.; Towprayoon, S.; Chidthaisong, A. Synthesis of activated carbon and MCM-41 from bagasse and rice husk and their carbon dioxide adsorption capacity. J. Sustain. Environ. 2013, 2, 77–81. [Google Scholar]
- Arami-Niya, A.; Daud, W.M.A.W.; Mjalli, F.S. Using granular activated carbon prepared from oil palm shell by ZnCl2 and physical activation for methane adsorption. J. Anal. Appl. Pyrolysis 2010, 89, 197–203. [Google Scholar] [CrossRef]
- Bhadusha, N.; Ananthabaskaran, T. Adsorptive Removal of Methylene Blue onto ZnCl2 Activated Carbon from Wood Apple Outer Shell: Kinetics and Equilibrium Studies. J. Chem. 2011, 8, 1696–1707. [Google Scholar]
- Özçimen, D.; Ersoy-Meriçboyu, A. Removal of copper from aqueous solutions by adsorption onto chestnut shell and grapeseed activated carbons. J. Hazard. Mater. 2009, 168, 1118–1125. [Google Scholar] [CrossRef]
- He, X.; Ling, P.; Qiu, J.; Yu, M.; Zhang, X.; Yu, C.; Zheng, M. Efficient preparation of biomass-based mesoporous carbons for supercapacitors with both high energy density and high power density. J. Power Sources 2013, 240, 109–113. [Google Scholar] [CrossRef]
- Deng, H.; Yang, L.; Tao, G.; Dai, J. Preparation and characterization of activated carbon from cotton stalk by microwave assisted chemical activation-Application in methylene blue adsorption from aqueous solution. J. Hazard. Mater. 2009, 166, 1514–1521. [Google Scholar] [CrossRef]
- Shafeeyan, M.S.; Daud, W.M.A.W.; Houshmand, A.; Shamiri, A. A review on surface modification of activated carbon for carbon dioxide adsorption. J. Anal. Appl. Pyrolysis 2010, 89, 143–151. [Google Scholar] [CrossRef]
- Coates, J. Interpretation of Infrared Spectra, A Practical Approach Interpretation of Infrared Spectra, A Practical Approach. In Encyclopedia of Analytical Chemistry; John Wiley & Sons Ltd.: Chichester, UK, 2000; pp. 10815–10837. [Google Scholar]
- Pathak, P.D.; Mandavgane, S.A. Preparation and characterization of raw and carbon from banana peel by microwave activation: Application in citric acid adsorption. J. Environ. Chem. Eng. 2015, 3, 2435–2447. [Google Scholar] [CrossRef]
- Dhaundiyal, A.; Singh, S.B. Study of Distributed Activation Energy Model Using Various Probability Distribution Functions for the Isothermal Pyrolysis Problem. Rud. Zb. 2017, 32, 1–14. [Google Scholar]
- Al-Anber, M.A. Thermodynamics Approach in the Adsorption of Heavy Metals. In Thermodynamics - Interaction Studies - Solids, Liquids and Gases; IntechOpen: London, UK, 2011; pp. 737–764. ISBN 978-953-307-563-1. [Google Scholar]
- Kong, S.H.; Loh, S.K.; Bachmann, R.T.; Rahim, S.A.; Salimon, J. Biochar from oil palm biomass: A review of its potential and challenges. Renew. Sustain. Energy Rev. 2014, 39, 729–739. [Google Scholar] [CrossRef]
- Maneerung, T.; Liew, J.; Dai, Y.; Kawi, S.; Chong, C.; Wang, C.H. Activated carbon derived from carbon residue from biomass gasification and its application for dye adsorption: Kinetics, isotherms and thermodynamic studies. Bioresour. Technol. 2016, 200, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Skouteris, G.; Saroj, D.; Melidis, P.; Hai, F.I.; Ouki, S. The effect of activated carbon addition on membrane bioreactor processes for wastewater treatment and reclamation - A critical review. Bioresour. Technol. 2015, 185, 399–410. [Google Scholar] [CrossRef] [PubMed]
- Demiral, H.; Baykul, E.; Gezer, M.D.; Erkoç, S.; Engin, A.; Baykul, M.C. Preparation and Characterization of Activated Carbon from Chestnut Shell and its Adsorption Characteristics for Lead. Sep. Sci. Technol. 2014, 49, 2711–2720. [Google Scholar] [CrossRef]
- Mohammad, S.G.; Ahmed, S.M. Preparation of environmentally friendly activated carbon for removal of pesticide from aqueous media. Int. J. Ind. Chem. 2017, 8, 121–132. [Google Scholar] [CrossRef]
- Tham, Y.J.; Latif, P.A.; Abdullah, A.M.; Shamala-Devi, A.; Taufiq-Yap, Y.H. Performances of toluene removal by activated carbon derived from durian shell. Bioresour. Technol. 2011, 102, 724–728. [Google Scholar] [CrossRef]
- Gokce, Y.; Aktas, Z. Nitric acid modification of activated carbon produced from waste tea and adsorption of methylene blue and phenol. Appl. Surf. Sci. 2014, 313, 352–359. [Google Scholar] [CrossRef]
- Huang, Y.; Li, S.; Lin, H.; Chen, J. Fabrication and characterization of mesoporous activated carbon from Lemna minor using one-step H3PO4 activation for Pb(II) removal. Appl. Surf. Sci. 2014, 317, 422–431. [Google Scholar] [CrossRef]
- Li, K.; Zheng, Z.; Li, Y. Characterization and lead adsorption properties of activated carbons prepared from cotton stalk by one-step H3PO4 activation. J. Hazard. Mater. 2010, 181, 440–447. [Google Scholar] [CrossRef]
- Obregón-Valencia, D.; Sun-Kou, M.D.R. Comparative cadmium adsorption study on activated carbon prepared from aguaje (Mauritia flexuosa) and olive fruit stones (Olea europaea L.). J. Environ. Chem. Eng. 2014, 2, 2280–2288. [Google Scholar] [CrossRef]
- Liu, R.L.; Liu, Y.; Zhou, X.Y.; Zhang, Z.Q.; Zhang, J.; Dang, F.Q. Biomass-derived highly porous functional carbon fabricated by using a free-standing template for efficient removal of methylene blue. Bioresour. Technol. 2014, 154, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Foo, K.Y.; Hameed, B.H. Preparation of activated carbon from date stones by microwave induced chemical activation: Application for methylene blue adsorption. Chem. Eng. J. 2011, 170, 338–341. [Google Scholar] [CrossRef]
- Mohammed, J.; Nasri, N.S.; Ahmad Zaini, M.A.; Hamza, U.D.; Ani, F.N. Adsorption of benzene and toluene onto KOH activated coconut shell based carbon treated with NH3. Int. Biodeterior. Biodegrad. 2015, 102, 245–255. [Google Scholar] [CrossRef]
- Ahmed, S.A.S.; Khalil, L.B.; El-nabarawy, T. Removal of Reactive Blue 19 dye from Aqueous Solution Using Natural and Modified Orange Peel. Carbon Lett. 2012, 13, 212–220. [Google Scholar] [CrossRef]
- Song, C.; Wu, S.; Cheng, M.; Tao, P.; Shao, M.; Gao, G. Adsorption studies of coconut shell carbons prepared by KOH activation for removal of lead(ii) from aqueous solutions. Sustainability 2014, 6, 86–98. [Google Scholar] [CrossRef]
- AL-Othman, Z.A.; Ali, R.; Naushad, M. Hexavalent chromium removal from aqueous medium by activated carbon prepared from peanut shell: Adsorption kinetics, equilibrium and thermodynamic studies. Chem. Eng. J. 2012, 184, 238–247. [Google Scholar] [CrossRef]
- García, J.R.; Sedran, U.; Zaini, M.A.A.; Zakaria, Z.A. Preparation, characterization, and dye removal study of activated carbon prepared from palm kernel shell. Environ. Sci. Pollut. Res. 2017, 1–10. [Google Scholar] [CrossRef]
- Elsayed, M.A.; Zalat, O.A. Factor Affecting Microwave Assisted Preparation of Activated Carbon from Local Raw Materials. Int. Lett. Chem. Phys. Astron. 2015, 47, 15–23. [Google Scholar] [CrossRef]
- Aghajani, Z.; Zand-Monfraed, M.-R.; Bahmani-Androod, S. Removal of lead ( II ) and mercury ( II ) from aqueous solutions and waste water using pistachio soft shell as agricultural by- products. Fresenius Environ. Bulletin 2014, 5, 170–176. [Google Scholar]
- Imamoglu, M.; Tekir, O. Removal of copper (II) and lead (II) ions from aqueous solutions by adsorption on activated carbon from a new precursor hazelnut husks. Desalination 2008, 228, 108–113. [Google Scholar] [CrossRef]
- Kula, I.; Uǧurlu, M.; Karaoǧlu, H.; Çelik, A. Adsorption of Cd(II) ions from aqueous solutions using activated carbon prepared from olive stone by ZnCl2 activation. Bioresour. Technol. 2008, 99, 492–501. [Google Scholar] [CrossRef] [PubMed]
- Ayawei, N.; Ebelegi, A.N.; Wankasi, D. Modelling and Interpretation of Adsorption Isotherms. J. Chem. 2017, 2017. [Google Scholar] [CrossRef]
- Gil, M.; Luciano, E.; Arauzo, I. Population balance model for biomass milling. Powder Technol. 2015, 276, 34–44. [Google Scholar] [CrossRef]
- Anyaoha, K.E.; Sakrabani, R.; Patchigolla, K.; Mouazen, A.M. Evaluating oil palm fresh fruit bunch processing in Nigeria. Waste Manag. Res. 2018, 36, 236–246. [Google Scholar] [CrossRef]
- Gil, M.; Arauzo, I. Hammer mill operating and biomass physical conditions effects on particle size distribution of solid pulverized biofuels. Fuel Process. Technol. 2014, 127, 80–87. [Google Scholar] [CrossRef]
- Vigneault, C.; Rothwell, T.M.; Bourgeois, G. Hammermill grinding rate and energy requirements for thin and conventional hammers. Can. Agric. Eng. 1992, 34, 203–207. [Google Scholar]
- Liu, Y.; Wang, J.; Wolcott, M.P. Assessing the specific energy consumption and physical properties of comminuted Douglas-fir chips for bioconversion. Ind. Crops Prod. 2016, 94, 394–400. [Google Scholar] [CrossRef]
- Bitra, V.S.P.; Womac, A.R.; Chevanan, N.; Miu, P.I.; Igathinathane, C.; Sokhansanj, S.; Smith, D.R. Direct mechanical energy measures of hammer mill comminution of switchgrass, wheat straw, and corn stover and analysis of their particle size distributions. Powder Technol. 2009, 193, 32–45. [Google Scholar] [CrossRef]
- Philips, E.S.; Mutuku, R.N.; Mwero, J.N. Effects of Palm Kernel Shell and Rice Husk Ash as Partial Replacements of Normal Weight Aggregate and Ordinary Portland Cement in Concrete. Eur. Int. J. Sci. Technol. 2017, 6, 42–54. [Google Scholar]
- Vogel, L.; Peukert, W. Breakage behavior of different materials - construction of a mastercurve for the breakage probability. Powder Technol. 2003, 129, 101–110. [Google Scholar] [CrossRef]
- Vogel, L.; Peukert, W. Determination of material properties relevant to grinding by practicable labscale milling tests. Int. J. Miner. Process. 2004, 74, 329–338. [Google Scholar] [CrossRef]
- Naimi, L.J.; Sokhansanj, S.; Bi, X.; Lim, C.J.; Womac, A.R.; Lau, A.K.; Melin, S. Development of Size Reduction Equations for Calculating Energy Input for Grinding Lignocellulosic Particles. Food Process Eng. Inst. Div. ASABE 2013, 29, 93–100. [Google Scholar]
- Dai, J.; Cui, H.; Grace, J.R. Biomass feeding for thermochemical reactors. Prog. Energy Combust. Sci. 2012, 38, 716–736. [Google Scholar] [CrossRef]
- Josip, Ć.; Matoši, M.; Crnek, V.; Stuli, V.; Mijatovi, I. Adsorption Characteristics of Different Adsorbents and Iron (III) Salt for Removing As (V) from Water. Food Technol. Biotechnol. 2016, 54, 250–255. [Google Scholar]
- Ji, Y.; Li, T.; Zhu, L.; Wang, X.; Lin, Q. Preparation of activated carbons by microwave heating KOH activation. Appl. Surf. Sci. 2007, 254, 506–512. [Google Scholar] [CrossRef]
- Thostenson, E.T.T.; Chou, T.-W. Microwave processing: FUndamentals and applications. Compos. Part A Appl. Sci. Manuf. 1999, 30, 1055–1071. [Google Scholar] [CrossRef]
- Lam, S.S.; Wan Mahari, W.A.; Cheng, C.K.; Omar, R.; Chong, C.T.; Chase, H.A. Recovery of diesel-like fuel from waste palm oil by pyrolysis using a microwave heated bed of activated carbon. Energy 2016, 115, 791–799. [Google Scholar] [CrossRef]
- Liew, R.K.; Nam, W.L.; Chong, M.Y.; Phang, X.Y.; Su, M.H.; Yek, P.N.Y.; Ma, N.L.; Cheng, C.K.; Chong, C.T.; Lam, S.S. Oil palm waste: An abundant and promising feedstock for microwave pyrolysis conversion into good quality biochar with potential multi-applications. Process Saf. Environ. Prot. 2018, 115, 57–69. [Google Scholar] [CrossRef]
- Mushtaq, F.; Mat, R.; Ani, F.N. A review on microwave assisted pyrolysis of coal and biomass for fuel production. Renew. Sustain. Energy Rev. 2014, 39, 555–574. [Google Scholar] [CrossRef]
- Nüchter, M.; Ondruschka, B.; Bonrath, W.; Gum, A. Microwave assisted synthesis – a critical technology overview. Green Chem. 2004, 6, 128–141. [Google Scholar] [CrossRef]
- Waheed Ul Hasan, S.; Ani, F.N. Review of limiting issues in industrialization and scale-up of microwave-assisted activated carbon production. Ind. Eng. Chem. Res. 2014, 53, 12185–12191. [Google Scholar] [CrossRef]
- Phuphuakrat, T.; Namioka, T.; Yoshikawa, K. Tar removal from biomass pyrolysis gas in two-step function of decomposition and adsorption. Appl. Energy 2010, 87, 2203–2211. [Google Scholar] [CrossRef]
- Alexieva, Z.; Gerginova, M.; Manasiev, J.; Zlateva, P.; Shivarova, N.; Krastanov, A. Phenol and cresol mixture degradation by the yeast Trichosporon cutaneum. J. Ind. Microbiol. Biotechnol. 2008, 35, 1297–1301. [Google Scholar] [CrossRef] [PubMed]
- Abu El-Rub, Z.; Bramer, E.A.; Brem, G. Experimental comparison of biomass chars with other catalysts for tar reduction. Fuel 2008, 87, 2243–2252. [Google Scholar] [CrossRef]
- Leonelli, C.; Veronesi, P. Production of Biofuels and Chemicals with Microwave. In Biofuels and Biorefineries; Springer: Berlin, Germany, 2015; Volume 3, pp. 17–39. ISBN 978-94-017-9611-8. [Google Scholar]
- Salema, A.A.; Yeow, Y.K.; Ishaque, K.; Ani, F.N.; Afzal, M.T.; Hassan, A. Dielectric properties and microwave heating of oil palm biomass and biochar. Ind. Crops Prod. 2013, 50, 366–374. [Google Scholar] [CrossRef]
- Abubakar, Z.; Salema, A.A.; Ani, F.N. A new technique to pyrolyse biomass in a microwave system: Effect of stirrer speed. Bioresour. Technol. 2013, 128, 578–585. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.S.L.; Chan, Y.S.; Law, M.C.; Leo, C.P. Comparative microstructure study of oil palm fruit bunch fibre, mesocarp and kernels after microwave pre-treatment. IOP Conf. Ser. Mater. Sci. Eng. 2017, 217. [Google Scholar] [CrossRef]
- Bykov, Y.V.; Rybakov, K.I.; Semenov, V.E. High-temperature microwave processing of materials. J. Phys. D. Appl. Phys. 2001, 34, 11. [Google Scholar] [CrossRef]
- Okahisa, Y.; Furukawa, Y.; Ishimoto, K.; Narita, C.; Intharapichai, K.; Ohara, H. Comparison of cellulose nanofiber properties produced from different parts of the oil palm tree. Carbohydr. Polym. 2018, 198, 313–319. [Google Scholar] [CrossRef]
- Oyejobi, D.O.; Abdulkadir, T.S.; Yusuf, I.T.; Badiru, M.J. Effects of Palm Kernel Shells Sizes and Mix Ratios on Lightweight Concrete. J. Res. Inf. Civ. Eng. 2012, 9, 217. [Google Scholar]
- Salema, A.A.; Ani, F.N. Microwave-assisted pyrolysis of oil palm shell biomass using an overhead stirrer. J. Anal. Appl. Pyrolysis 2012, 96, 162–172. [Google Scholar] [CrossRef]
- Leonelli, C.; Mason, T.J. Microwave and ultrasonic processing: Now a realistic option for industry. Chem. Eng. Process. Process Intensif. 2010, 49, 885–900. [Google Scholar] [CrossRef]
- Guo, Q.; Sun, D.W.; Cheng, J.H.; Han, Z. Microwave processing techniques and their recent applications in the food industry. Trends Food Sci. Technol. 2017, 67, 236–247. [Google Scholar] [CrossRef]
- Morschhäuser, R.; Krull, M.; Kayser, C.; Boberski, C.; Bierbaum, R.; Püschner, P.A.; Glasnov, T.N.; Kappe, C.O. Microwave-assisted continuous flow synthesis on industrial scale. Green Process. Synth. 2012, 1, 281–290. [Google Scholar] [CrossRef]
- Petri, I.; Martins, A.L.; Duarte, C.R.; Ataíde, C.H. Development and performance of a continuous industrial microwave dryer for remediation of drill cuttings. J. Pet. Sci. Eng. 2019, 176, 362–368. [Google Scholar] [CrossRef]
- World, C. Largest Industrial Microwave System. Available online: https://www.compositesworld.com/products/large-industrial-microwave-system (accessed on 1 June 2019).
- Bridge, I. Department of Science and Technology, South Africa. Available online: https://www.innovationbridge.info/ibportal/?q=content/activated-carbon (accessed on 16 September 2019).
- Beims, R.F.; Simonato, C.L.; Wiggers, V.R. Technology readiness level assessment of pyrolysis of trygliceride biomass to fuels and chemicals. Renew. Sustain. Energy Rev. 2019, 112, 521–529. [Google Scholar] [CrossRef]
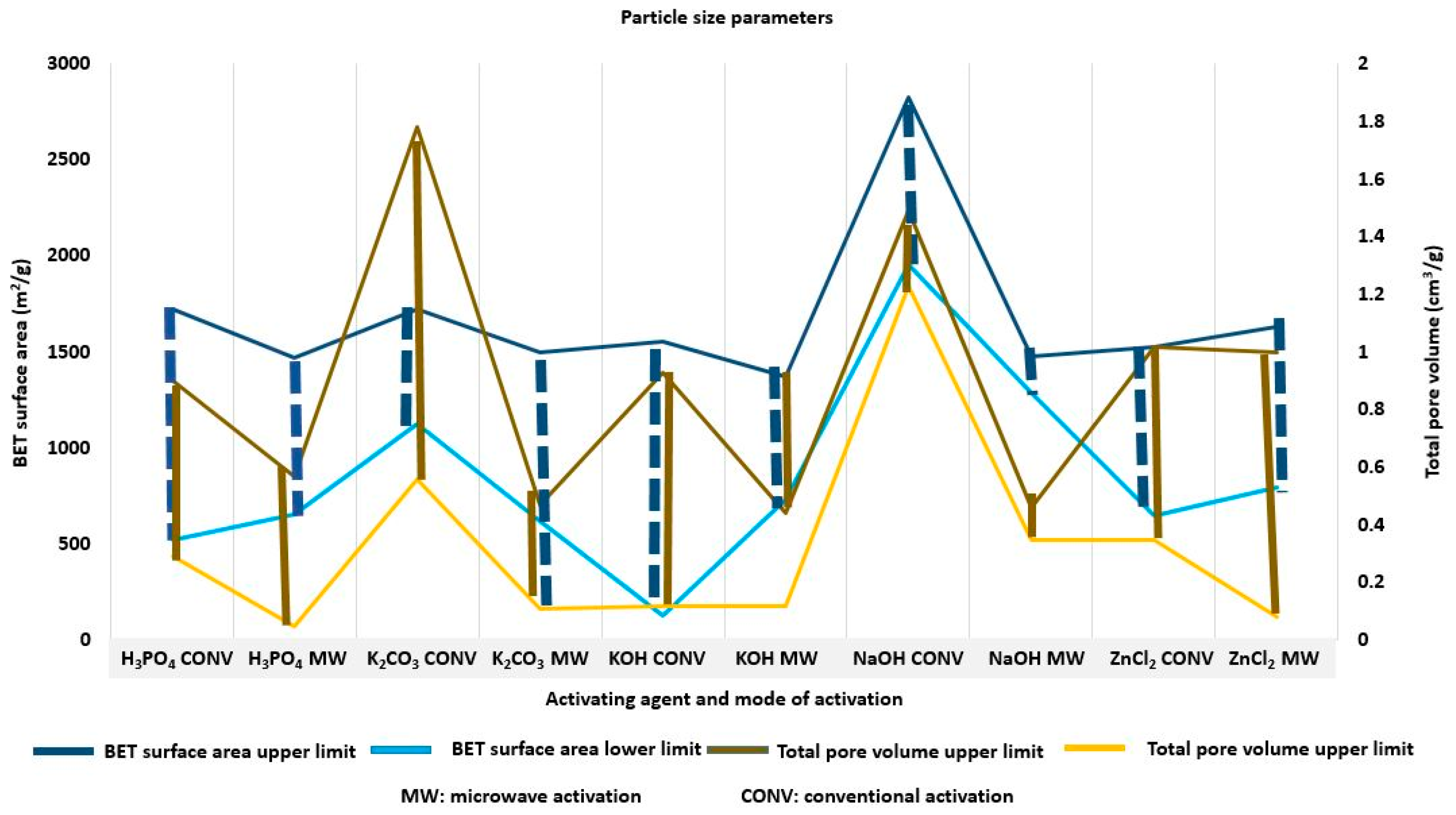
| Agricultural Waste | Proximate Analysis (% w/w) | Ultimate Analysis (% w/w) | Lignocellulosic Composition (% w/w) | Reference | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Moisture | Ash | Volatiles | C | H | N | S | O | Cellulose | Hemicellulose | Lignin | ||
| Almond stone | 11.05 | 0.76 | 77.32 | 48.76 | 7.52 | 0.48 | 0.56 | 43.68 | 21.70 | 27.70 | 36.10 | [39] |
| Bamboo | 15.30 | 1.76 | 70.12 | 34.40 | 4.61 | 0.22 | 0.07 | - | 26 | 15 | 21 | [40] |
| Banana peel | 11.56 | 9.28 | 88.02 | 35.65 | 6.19 | 1.60 | 20.75 | 45.94 | - | - | - | [41] |
| Cassava peel | 14 | 4.50 | 59.40 | 59.31 | 9.78 | 2.06 | 0.11 | 28.74 | 37.90 | 23.90 | 7.50 | [42] |
| Coconut shell | 8.21 | 0.80 | 77.82 | 49.62 | 7.31 | 0.22 | 0.10 | 42.75 | 14.00 | 32.00 | 46.0 | [43] |
| Cotton stalks | 6.00 | 6.30 | 70.50 | 43.60 | 5.80 | 0.80 | 0.00 | 49.80 | 80-95 | 5-20 | - | [44] |
| Durian shell | 11.27 | 4.84 | - | 39.30 | 5.90 | 1.00 | 0.06 | 53.74 | 60.45 | 13.09 | 15.45 | [4] |
| Grape stalk | 8.86 | 3.15 | 96.80 | 34.40 | 0.438 | 1.11 | 0.087 | 63.96 | - | - | - | [45] |
| EFB | 15.01 | 4.48 | 82.98 | 43.89 | 5.33 | 0.52 | 0.10 | 54.32 | 42.00 | 18.90 | 11.70 | [46] |
| Oil Palm MF | 11.10 | 7.90 | 84.03 | 42.20 | 5.21 | 2.21 | 0.14 | 42.34 | 42 | 22 | 14 | [46] |
| PKS | 7.96 | 1.10 | 72.47 | 50.01 | 6.90 | 1.90 | 0.03 | 41 | 20.80 | 22.70 | 50.70 | [4] |
| Olive stone | 10.40 | 1.40 | 74.40 | 44.80 | 6.00 | 0.10 | 0.01 | 49.09 | 30.80 | 17.10 | 32.60 | [47] |
| Orange peel | - | 2.15 | 77.93 | 40.28 | 6.12 | 1.08 | 0.06 | 52.46 | - | - | - | [48] |
| Peanut shell | 7.98 | 12.80 | 79.10 | 41.52 | 7.43 | 2.12 | 0.60 | 27.96 | - | - | - | [49] |
| Rice husk | 6.34 | 16.70 | 67.50 | 36.52 | 4.82 | 0.86 | - | 41.10 | 30.42 | 28.03 | 36.02 | [50] |
| Sugarcane BG | 8.61 | 4.05 | 86.02 | 47.30 | 6.20 | 0.27 | - | 44.15 | 42.16 | 36.00 | 19.30 | [50] |
| Walnut shell | 8.73 | 1.27 | 77.42 | 49.30 | 5.82 | 44.49 | 0 | - | 40.10 | 20.70 | 18.20 | [47] |
| Waste tea | 5.80 | 4.29 | - | 52.72 | 6.34 | 2.61 | 0.18 | 38.15 | 17.50 | 41.30 | 41.20 | [51] |
| One-Step Conventional | Two-Step Conventional | Microwave Process | |
|---|---|---|---|
| Temperature or power requirements | 400–1200 °C | 400–800 °C then 400–1200 °C | 300–1000 W |
| Heating duration | 1–3 h | 3–6 h | 5–30 min |
| Average yield | 30–50% | 30–40% | >40% |
| Risk of system corrosion | High | High | Low |
| Product efficiency | Low | High | High |
| Flow process | Continuous feed in and out | Batch | Batch |
| Framework | Microwave Activation | Conventional Activation | Observation |
|---|---|---|---|
| Treatment time | Shorter process time | Several hours and days | Shorter time reduces process risks |
| Heating process | Internal and volumetric heating | Surface heating Non-uniform heating | Thermal gradient is eliminated by microwave heating |
| Mode of heat transfer | Energetic coupling Coupling at molecular level | Conduction and convection Superficial and wall heating | Uniform heating is easily achieved on coupling at molecular level |
| Gas/Energy consumption | Due to short treatment time, it is low | High | Microwave process saves energy |
| Equipment size | Small | Large | Developing a large size microwave is expensive and complex |
| Preparation conditions | 300–700 W 5–15 min | 400–1200 °C 1–3 h | Same for both except when two-step activation process is required |
| AC characteristics | Higher surface area | High surface area | Choice may depend on application and process requirements |
| Complexity | High | Low | Conventional equipment is easy to build. Equipment repair is easier with conventional approaches |
| Precursor | Time (hr) | T(°C)/MW(W) | I.R | SBET (m2/g) | Vtotal (cm3/g) | Vmeso (cm3/g) | Vmicro (cm3/g) | Dp (nm) | Yield (%) | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| Result of H3PO4 activation of conventional process | ||||||||||
| Cotton stalk | 2 | 500 | 1.5 | 1720 | 0.890 | 0.710 | [114] | |||
| Peach stones | 2 | 500 | 0.4 | 1393 | 0.689 | 0.055 | 0.634 | 0.99 | 41.8 | [115] |
| Bamboo | 1 | 600 | 1.5 | 1335 | 0.625 | 0.140 | 0.485 | 1.87 | [116] | |
| Maize tassel | 1 | 500 | 1.4 | 1263 | 1.592 | [56] | ||||
| Palm shell | 0.5 | 425 | 2 | 1109 | 0.903 | 3.20 | [117] | |||
| Durian shell | 0.4 | 500 | 1021 | 0.350 | 0.210 | 63 | [118] | |||
| Cotton cake | 1.5 | 450 | 2 | 584 | 0.298 | 0.075 | 0.223 | 2.04 | 29.8 | [119] |
| Rice straw | 2 | 450 | 1 | 522 | 0.550 | 0.370 | 0.180 | 4.21 | 51.9 | [120] |
| Result of H3PO4 activation of microwave process | ||||||||||
| Palm shell | 0.28 | 800 | 2 | 1473 | [121] | |||||
| Lotus stalk | 0.25 | 700 | 2 | 1434 | 0.307 | 1.030 | 1.337 | [84] | ||
| Bamboo | 0.5 | 350 | 1 | 1432 | 0.503 | 0.193 | 0.696 | 48 | [116] | |
| Waste tea | 0.5 | 350 | 3 | 1157 | 0.573 | 0.256 | 0.829 | 35 | [122] | |
| Cotton stalk | 8 | 400 | 0.5 | 652 | 0.057 | 0.419 | 0.476 | 2.92 | [123] | |
| Result of K2CO3 activation of conventional process | ||||||||||
| Waste tea | 900 | 1 | 1722 | 0.583 | 0.039 | 0.554 | 2.0 | 22.7 | [51] | |
| Rice husk | 1.5 | 1000 | 1.5 | 1713 | 1.785 | 1.070 | 0.715 | 4.16 | [94] | |
| Mangosteen shell | 2 | 900 | 1 | 1123 | 0.560 | 0.110 | 0.450 | 1.98 | 20.7 | [124] |
| Result of K2CO3 activation of microwave process | ||||||||||
| Wood saw dust | 0.1 | 600 | 1.3 | 1496 | 0.470 | 0.394 | 0.864 | 2.30 | 80.0 | [125] |
| Rice husk | 0.11 | 600 | 1.3 | 1165 | 0.330 | 0.450 | 0.780 | 2.68 | [94] | |
| Orange peel | 0.1 | 600 | 1.3 | 1104 | 0.247 | 0.368 | 0.615 | 2.22 | 80.9 | [91] |
| Pineapple peel | 0.1 | 600 | 0.8 | 680 | 0.280 | 0.170 | 0.450 | 2.59 | [86] | |
| Cotton stalk | 0.13 | 660 | 0.8 | 621 | 0.110 | 0.270 | 0.380 | 2.43 | [126] | |
| Result of KOH activation of conventional process | ||||||||||
| Rice straw | 1 | 800 | 4 | 1554 | 0.930 | 0.340 | 0.500 | 1.14 | 13.5 | [120] |
| Bamboo | 3 | 800 | 2 | 1533 | 0.491 | [40] | ||||
| Cocoa pod husk | 1 | 800 | 1 | 490 | 0.240 | 2 | 13.5 | [127] | ||
| PKS | 0.75 | 800 | 1 | 127 | 0.120 | 0.110 | 43.4 | [128] | ||
| Result of KOH activation of microwave process | ||||||||||
| Empty FB | 0.11 | 600 | 1 | 1372 | 0.440 | 0.320 | 0.760 | 2.20 | 73.7 | [78] |
| Mesocarp Fibre | 0.1 | 600 | 0.8 | 1223 | 0.420 | 0.300 | 0.720 | 2.35 | 32.0 | [129] |
| Pineapple peel | 0.1 | 600 | 0.8 | 1006 | 0.280 | 0.310 | 0.590 | 23.44 | [86] | |
| Palm Shell | 0.16 | 600 | 0.6 | 895 | 0.491 | 2.19 | [128] | |||
| Rice husk | 0.11 | 600 | 1.3 | 752 | 0.260 | 0.380 | 0.640 | 3.41 | [94] | |
| Coconut husk | 0.11 | 600 | 1.3 | 752 | 0.260 | 0.380 | 0.640 | 3.41 | [130] | |
| Cotton stalk | 0.16 | 680 | 0.6 | 729 | 0.120 | 0.260 | 0.380 | 2.08 | [126] | |
| Result of NaOH activation of conventional process | ||||||||||
| Coconut shell | 1.5 | 700 | 3 | 2825 | 1.498 | 0.355 | 1.143 | 2.27 | 18.8 | [131] |
| Rice husk | 0.05 | 850 | 2 | 1958 | 1.230 | 0.573 | 0.550 | 1.25 | [132] | |
| Result of NaOH activation of microwave process | ||||||||||
| Durian shell | 0.1 | 600 | 1.5 | 1475 | 0.467 | 0.374 | 0.841 | 2.28 | 80.0 | [133] |
| Langsat bunch | 0.1 | 600 | 1.3 | 1293 | 0.449 | 0.303 | 0752 | 2.32 | 81.3 | [134] |
| Jackfruit peel | 0.11 | 600 | 1.5 | 1286 | 0.356 | 0.408 | 0.764 | 2.37 | 80.8 | [135] |
| Result of ZnCl2 activation of conventional process | ||||||||||
| Tea seed shell | 1 | 500 | 1 | 1530 | 0.783 | 0.184 | 0.599 | 2.05 | 44.1 | [136] |
| Oceania palm | 2 | 600 | 4.5 | 1483 | 1.022 | 0.456 | 0.494 | 2.76 | 40.0 | [137] |
| Waste apricot | 1 | 500 | 1 | 1060 | 0.790 | 0.640 | 0.150 | 2.98 | [81] | |
| Date stone | 1.2 | 500 | 2 | 1045 | 0.641 | 0.129 | 0.512 | 2.45 | 40.4 | [138] |
| Rice husk | 927 | 0.560 | 0.80 | [139] | ||||||
| Palm Shell | 900 | 926 | 0.480 | 0.470 | 2.05 | [140] | ||||
| Wood apple shell | 2 | 794 | 0.470 | [141] | ||||||
| Hazelnut shell | 647 | 0.350 | 34.0 | [142] | ||||||
| Result of ZnCl2 activation of microwave process | ||||||||||
| Peanut shell | 0.3 | 600 | 1.4 | 1634 | [143] | |||||
| Rice husk | 0.3 | 600 | 1.4 | 1527 | 2.070 | 5.99 | [143] | |||
| Cotton stalk | 0.15 | 560 | 1.6 | 794 | 0.083 | 0.547 | 0.630 | 3.20 | 37.5 | [144] |
| Adsorbent | Activator | Activation Temp /MW Power (W) | IR | Adsorbate | SBET m2/g | Adsorption Capacity (mg/g) | CR | Contact Time (min) | Contact Temp °C | Contact pH | Reference |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Durian shell | H3PO4 | 500 °C | 1:0.30 | Toluene | 1404 | 874 | 2 h | 25 | [155] | ||
| Waste tea | H3PO4 | MB | 1398 | 288.34 | [156] | ||||||
| Sugarcane bagasse | H3PO4 | 500 °C | 3:1 | Pb | 320 | 170.90 | 6 | 30 min | 25 | 4 | [157] |
| Cotton stalk | H3PO4 | 500 °C | 3:2 | Pb(II) | 1570 | 119 | 25 | 4.4 | [158] | ||
| Olive stone | H3PO4 | 600 °C | 1:1.5 | Cd | 1565 | 24.83 | 5 | [159] | |||
| Coconut husk | KOH | 700 °C | MB | 1356 | 418.15 | [130] | |||||
| Banana peel | KOH | 800 °C | MB | 2086 | 385.12 | 60 | 6 | [160] | |||
| EFB (char) | KOH | 360 W | 1:0.75 | MB | 807 | 344.80 | 50 | 4 | [78] | ||
| Date stone | KOH | 600 W | 1:1.75 | MB | 856 | 316.10 | [161] | ||||
| Coconut shell | KOH | 700 °C | 1.5:1 | Benzene | 478 | 212.77 | 30 | [162] | |||
| Orange peel | NaOH | Reactive Blue 19 | 166.60 | 100 | 24 h | 30 | 4 | [163] | |||
| Coconut shell | KOH | 800 °C | 1:2 | Pb | 1135 | 151.52 | 4 | 5 | [164] | ||
| Peanut shell | KOH | Cr(VI) | 96 | 16.26 | 40 | 7 h | 25 | 4 | [165] | ||
| Tea seed shell | ZnCl2 | 500 °C | 1:1 | MB | 1531 | 342.70 | 6.3 | [136] | |||
| Pineapple waste | ZnCl2 | 500 °C | 1:1 | MB | 915 | 288.34 | 12 h | [166] | |||
| Corn cob | K2CO3 | 600 W | 1:0.75 | MB | 765 | 275.32 | [167] | ||||
| Cotton stalk | ZnCl2 | 560 W | 1:1.6 | MB | 193.50 | 4 | 2 h | 7 | [144] | ||
| Pistachio nut shell | ZnCl2 | Hg | 1492 | 24.78 | 100 min | 25 | 6 | [168] | |||
| Hazelnut shell | ZnCl2 | 700 °C | Pb | 1067 | 13.05 | 200 | 10 min | 5.7 | [169] | ||
| Olive stone | ZnCl2 | 650 °C | Cd | 790 | 1.85 | 50 | 90 min | 25 | 9 | [170] |
| Isotherm [171] | Nonlinear Form [171] | Linear Form | Plot | Nomenclature | Description/Assumptions |
|---|---|---|---|---|---|
| Langmuir | qe=equilibrium sorption capacity for metal Ce=equilibrium solute concentration in solution b=Langmuir constant | -The simplest isotherm -Assumes that adsorption is limited to monolayer -There is insignificant interaction and change in the sorption process -The model is identical to energy being adsorbed | |||
| Freundlich | KF= equilibrium constant for relative adsorption n =adsorption constant indicative of intensity | -Relative to multi-layer adsorption -widely applied in organic compounds and highly interactive substance | |||
| Sip’s isotherm | β = Sip’s constant | -combination of Langmuir and Freundlich -It is governed by pH, temperature and concentration changes | |||
| Temkin | AT and bT =Tempkin constants | -Takes care of adsorbate-adsorbent interaction -Used for predicting the gas-phase equilibrium | |||
| Khan | bk and ak are model constant and model exponent | -General adsorption model for adsorbate from pure dilute equation solutions | |||
| Redlich-Peterson | KR, aR, and β = Redlich–Peterson parameters. β takes values between 0 and 1. For β=1 the model can be correlated to the Langmuir form | - It is anti-ideal monolayer adsorption -Has a linear relative dependence on concentration of solution | |||
| Radke-Prausnitz | The outlook of surface diffusion Relative role of pore volume. aR and rR = model constants | -Preferred in most systems at low adsorbate concentration Good for wide range of adsorbate concentration | |||
| Frenkel-Halsey-Hill | Where d, α and r = sign of the interlayer spacing (m), | ||||
| Toth | bT and nT = constants. if nT=1, Toth isotherm can be transformed to Langmuir isotherm equation | -Reduces error in experimental data -Predicts equilibrium data -Applicable in the modelling of several multilayer and heterogeneous adsorption systems | |||
| Dubinin-Radushkevich | kd = isotherm constant. ε = RT ln(1+1/Ce) R, T and Ce = gas constant CeTemperature (K) |
| Material | Frequency (GHz) | Penetration Depth (cm) | Permittivity (ε’r) | Tangent Loss (Tanδ) |
|---|---|---|---|---|
| PKS | 0.9–5.8 | 5.5–36 | 2.7 | 0.13 |
| MF | 2.5–5.8 | 10.2–24.8 | 2.0 | 0.08 |
| EFB | - | - | 6.4 | 0.3 |
| Water | 2.54 | 1–4 | - | - |
| Paper | 2.54 | 20–60 | - | - |
| Wood | 2.54 | 8–350 | 2.3 | 0.11 |
| Natural Rubber | 2.54 | 15–350 | 2.1 | 0.003 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ukanwa, K.S.; Patchigolla, K.; Sakrabani, R.; Anthony, E.; Mandavgane, S. A Review of Chemicals to Produce Activated Carbon from Agricultural Waste Biomass. Sustainability 2019, 11, 6204. https://doi.org/10.3390/su11226204
Ukanwa KS, Patchigolla K, Sakrabani R, Anthony E, Mandavgane S. A Review of Chemicals to Produce Activated Carbon from Agricultural Waste Biomass. Sustainability. 2019; 11(22):6204. https://doi.org/10.3390/su11226204
Chicago/Turabian StyleUkanwa, Kalu Samuel, Kumar Patchigolla, Ruben Sakrabani, Edward Anthony, and Sachin Mandavgane. 2019. "A Review of Chemicals to Produce Activated Carbon from Agricultural Waste Biomass" Sustainability 11, no. 22: 6204. https://doi.org/10.3390/su11226204
APA StyleUkanwa, K. S., Patchigolla, K., Sakrabani, R., Anthony, E., & Mandavgane, S. (2019). A Review of Chemicals to Produce Activated Carbon from Agricultural Waste Biomass. Sustainability, 11(22), 6204. https://doi.org/10.3390/su11226204







