1. Introduction
The concept of environment in the context of agroecology can be understood as: “all the natural components of the earth (air, water, soils, vegetation, animals), along with all the processes [social, economic and cultural] that occur within and among these components” [
1].
There is enough scientific evidence to prove that the activities developed by human beings (e.g., agriculture) ineluctably cause positive or negative changes over the environment. Therefore, project development decision making implies complex interdisciplinary processes to optimize resources in a sustainable way [
2,
3,
4,
5,
6,
7].
The way we perceive and interact with nature is fundamental to make decisions about the way in which an anthropogenic activity such as agriculture should be developed. That set of ideas, concepts and values with which we think and interact with the environment can be called environmental thinking [
8].
Environmental thinking in essence recognizes that humanity built a system of adaptation to the ecosystems based primarily on culture, therefore different from that of other living beings. The culture has been defined as the non-biological heritage of humanity [
9] or as all those theoretical and practical processes that are expressed in the symbolic structures, human organization and technological platforms of humanity [
10,
11,
12]. From this point of view, food production systems can be understood as sets of activities that human groups organize, direct and carry out according to their objectives and resources, influencing and being influenced by the ecosystem and biophysical environment where they are located [
13]. This aspect denotes the relevance and complexity of the ecosystem-culture interaction in the agrarian matters [
12].
On the other hand, it is widely accepted that agriculture is the result of the cultural modification of ecosystems, when these transformations are aimed at satisfying the basic needs of food, fibers and other materials according to the conditioning factors of the ecosystem-culture coevolution [
14,
15]. This type of modification interacts with some properties of the natural systems such as vulnerability. This concept must be taken into account for the planning of productive systems, for example, agroecosystems, because it describes states of susceptibility to harm, powerlessness and marginality of both physical and social systems and directs the actions to enhance well-being through the reduction of risk [
16,
17].
The study of these complex relationships is the object of agroecology [
18], a science that focuses on the environmental analysis of agroecosystems [
19,
20] and recognizes their multiple biophysical, symbolic, social, economic, political and technological interactions [
21].
Agroecology studies and proposes answers to complex problems through agrarian practices based on traditional or ancestral knowledge, the promotion of biological and cultural diversity, the autonomy of producers and the conservation and proper management of natural resources. It also proposes an interdisciplinary approach in scientific research and stimulates the biological regulations even in large-scale agriculture [
22,
23,
24,
25,
26].
This complexity also demands robust instruments and methodologies to address those issues. In a recent international meeting promoted by four French NGOs, it was recognized that “the priority is perhaps not to define a single evaluation method […] but to specify the fields of investigation and evaluation, as well as truly distinctive indicators in the case of agroecology by using the existing methods and tools” [
27]. These fields and evaluation criteria must also inevitably focus on three complementary and relevant scales: plot, farm, territory. Similarly, the authors recognize that the agricultural practices at these different scales calls for several types of indicators of technical and socio-economic performance, agro-environmental aspects, social aspects (wellbeing, culture and knowledge) and also the resilience of farms and territories facing climate or economic risks.
Within these practices or strategies that arise from agroecology, León-Sicard [
22] proposes to study what he called the Main Agroecological Structure of the major agroecosystems (MAS). This concept refers to the arrangements of the internal and external connectors of the farms (fences, hedges, living fences or patches of forest) and can relate to the probability of resilience or adaptation of agrarian systems to disturbances of different nature. The MAS can be theoretically associated with a dissipative structure, with possible beneficial effects on the establishment and development of crops at different scales, from subsistence agriculture in small areas to intensive productive processes of agro-industrial character. Likewise, the MAS may allow the design and implementation of adaptation and mitigation options to the changing climate, as a contribution to risk management.
This document presents the theoretical bases of the concept, the general methodology for its calculation and the description of some preliminary applications in Colombia.
2. Theoretical Bases of the MAS
The agroecosystem is an ecosystem deliberately modified by human beings in order to obtain goods and services with different purposes and therefore it is the place where dynamic relationships between the culture and its physical-biological environment are presented [
28,
29].
What is called here as “cultural context” refers to the human act as a whole, mediated by the thought (symbols) and by its social, economic, political and military organization, expressed in different technological platforms and intimately interrelated with the organization of societies. Thus conceived, the agroecosystem expresses an ecosystem order, molded by the cultural order.
Several authors have considered agroecosystems as units of analysis where different types of ecosystem processes and socio-economic relationships converge (energy flows, material cycles) [
19,
20]. Furthermore, the agroecosystem has been defined as “the set of relationships and interactions between soils, climates, cultivated plants, organisms of different trophic levels, adventitious plants and human groups in certain physical and geographical spaces. The study of agroecosystems includes their energy flows and information, material cycles and their symbolic, social, economic and political relations, which are expressed in different technological forms of management within specific cultural contexts” [
22]. The interactions between these components occur within diffuse limits that can go beyond the farm [
21].
The origin of the MAS is derived from the works of van der Hammen and Andrade [
30,
31] in Colombia, who proposed the idea of the Ecological Support Structure of the Nation (ESS) to facilitate the comprehension, at the country level, of the current state of its plant cover. The authors recognized two components of the EES: the Main Ecological Structure of the Landscape (MES) and the Ecological Infrastructure (EI); in the latter, they included the remnants of vegetation in the agroecosystems, although they did not elaborate the formulation of this idea. However, with these concepts the authors gave meaning to the study of landscape ecology and provided a background to articulate, in theory and practice, the management of relicts and patches of natural vegetation, including biological corridors and wooded masses.
In this way, the concept of the Main Agroecological Structure of agroecosystems is born to complement this unfinished relationship proposed by professors van der Hammen and Andrade and to provide a frame of reference for agroecology in terms of a natural property or feature of the agroecosystems: their own structure.
The MAS is therefore defined as “the internal configuration or spatial arrangement of the farm and the connectivity between its different sectors, patches and corridors of vegetation or productive systems. The MAS allows the movement and exchange of different animal and vegetable species, offers shelter, habitat and food, provides microclimatic regulation, affects the yields and the conservation of natural resources and other ecosystem and cultural aspects of the major agroecosystems” [
22].
The major agroecosystem (farm) is composed of minor agroecosystems (lots, plots, cultivation sites, forest areas, agroforestry sites or silvopastoral systems). The specific features of the MAS are given according to the way in which these minor agroecosystems relate to forests or other types of tree, herbaceous or scrub vegetation covers within the major agroecosystem. This concept, which aims at describing the structural and functional relationships of major agroecosystems in light of their determining cultural factors can be used for different applications (relationships with production, plant health, associated biodiversity, food autonomy, resilience), including future uses, such as being a taxonomic criterion for the definition of agroecosystems [
18].
When it comes to designing agroecosystems, which can be resilient to different types of disturbances, it is necessary to understand their inherent complexity [
32]. In this sense, the MAS accounts for both functional (unintentional) and planned agrobiodiversity, offering planning possibilities through the so-called ‘potential MAS,’ with repercussions at farm level (yields) and also at the level of agroecosystem matrices in landscape units at smaller scales (sidewalks, river basins, municipalities, regions) [
22].
Agroecosystems are considered as systems far from equilibrium because there are constant exchanges of matter, energy and information with the environment and it is impossible to obtain a final and permanent balance due to the continuous entropy generated in their transformation processes. However, these systems evolve and adapt to their environment until they reach the weakest possible dissipation through mechanisms of self-organization or auto-poiesis [
33,
34,
35].
When agroecosystems face fluctuations (changes in temperature, rainfall or attacks by insects or pathogens) and when these disturbances increase instead of disappearing, these systems can undergo an organized structural transformation, which would allow maintaining their functionality in time and space, towards a new temporary state called “dissipative structure.” This structure is the amplified fluctuation stabilized by the interactions with the environment and it prevails because it is continuously nourished by the energetic flow coming from the disturbance [
34,
36]. The formation and subsistence of the dissipative structure requires at least three conditions: (1) the system must be open and is able to continuously exchange matter and energy with the external environment; (2) The system must be in a state of non-equilibrium or far from equilibrium, because those states are the source of order; (3) Non-linear interactions, as well as certain nonlinear dynamic equations must exist in the system [
37].
Since its promulgation, the theory of dissipative structures has shown a wide scope of application and it is currently supported by several theoretical frameworks in engineering, medicine, psychology, agriculture and human sciences [
37,
38,
39,
40,
41,
42,
43]. For this reason, this theory is becoming an important reference in the modern scientific system [
44].
The concept of dissipative structure was applied in ecology since the 1970s to explain the relationships that exist between predators and their prey [
45] and in the study of biological structures and functions at the genetic level [
46]. In the 1980s it was used to address the evolution of ecosystems [
47] and explain the relationships between species and ecosystems [
48]. In the late twentieth century, it was applied to the analysis of the distribution of plants [
49] and in the design of processes to manage and control weed plants [
50].
In summary, the theoretical approach of dissipative structures can be applied to explain the behavior of biological, physical, chemical and social systems, to study the evolution of their structures in terms of a certain “order acquisition” [
35] and also to the analysis of resilience.
If the agroecosystem is conceived as an open system that constantly interacts with the environment (physical, biotic, social, economic and cultural), then the MAS can also be considered as a dissipative structure that allows the system to increase or improve the possibilities of maintaining its functionality by incorporating, dissipating or using the matter, energy or information coming from the disturbance. In this sense, for example, the greatest available agrobiodiversity in a farm allows different plants located in diversified strata and with various phenotypes and genotypes to have greater physical resistance to climatic disturbances but also wider ranges of response (in economic and social terms) to disturbances of different origins.
Understanding the MAS as a dissipative structure allows us to analyze the components of the agroecosystem and strengthen those that are able to dissipate the disturbance. For example, in the event of a disturbance related to the increase in evapotranspiration or the decrease in available water, it is possible to use species that are more resistant, green covers to protect the soil or other strategies, which are themselves the consequence of social or economic processes. It would also enable solutions of cultural nature such as saving water or collecting rain, mediated by the cultural features of particular agrarian societies.
Assuming this theoretical approach to thermodynamics, the MAS could be considered as a cultural dissipative structure that, insofar as it increases, it improves the possibilities of interaction and adaptation to different types of disturbances, due to the inherent complexity of greater agrobiodiversity and connectivity between sectors of agroecosystems. In this way, a higher MAS would help dispel the negative effects of external factors, whether cultural or ecosystem-related.
3. Methodological Approach
The concept of MAS is formulated precisely to encourage the necessary dialogue between landscape ecology and agroecology, a topic that had been partially addressed since the 1990s, when several authors recognized the interconnections and complexities of the spatial distribution of ecosystems and the need of acknowledging their connections with human activities [
51].
Some theorists of landscape ecology noted that the old matrices of forests or savannas had been replaced by matrices in which agricultural human activities predominated. However, the analysis of the landscape was mainly based on the management of wildlife and other natural resources [
52]. Landscape ecology has focused on studying the composition, structure, functionality and changes of the landscape and in this sense the analyses have been concentrated in three of its main components: matrices, patches and corridors [
52]. Thus, several approaches relate the size of the patches and their relative isolation from the main plant covers (theory of biogeographic islands) [
53] with the population equilibrium and the diversity of species.
However, it is above all the book “The matrix of nature: linking agriculture, conservation and food sovereignty” [
54] which clarifies the general links between conservation biology and agroecology in relationship with landscape studies. After confirming that the decrease in songbirds in eastern United States occurred in the same proportion as the reduction of shade-grown coffee crops in Latin America (which were replaced by unshaded monoculture) [
55], the authors stated that agroecosystems are important components of the natural world, intimately linked to the conservation of biodiversity. This topic had been frequently ignored in most studies on the subject. In this way, the MAS offers an additional conceptualization to the paradigm of conservation biology based on the matrix of nature, proposed by Perfecto and her colleagues [
54].
In this sense, the MAS is not part of the debate between “land sharing”
versus “land sparing” but it seeks to establish “inward” links of the agroecosystem, which facilitates the “outward” understanding of the management of biodiversity and agrobiodiversity, in line with both the ecological restoration [
56] and the new paradigm of biological conservation [
54].
The need for this change of paradigm is evident when some ecological studies, even though they appeal for landscape metrics, fail to understand the complex dynamics of agroecosystems and avoid referring to their cultural and ecosystem conditions, ignoring that among the elements of culture, we must insist on technological, symbolic and organizational factors.
Some steps in this direction have been taken by the new paradigm of ecological restoration that is considered as a strategy to guarantee the sustainability of the processes that maintain the ecosystem services that natural and semi-natural ecosystems and agroecosystem provide [
56].
Ecological restoration is defined as “the process of assisting the recovery of an ecosystem that has been degraded, damaged or destroyed” [
57] or as “a process that recovers and improves the functionality of an ecosystem within landscapes made up of agricultural production and conservation areas” [
58].
The ability to restore an agroecosystem depends on a large amount of knowledge at different scales, which includes information on the state of the landscape and the interrelation of ecological, cultural and historical factors. In other words, it is necessary to know the historical and current relationship between the natural system and the socioeconomic system, which is related, among other things, to the availability of the native biota necessary for the restoration of vegetation spots located near or within agroecosystems. In highly transformed landscapes, the matrix is a homogenous network or mosaics of agroecosystems without native vegetation spots and without vegetation corridors [
56].
Even so, many ecological restoration theorists focus more on ecosystem-type concepts and strategies or territorial protection models that optimize the relationships among habitats, species and populations in order to guarantee the conservation of biodiversity (species, habitats, landscapes) [
57,
59]. Although they consider agroecosystems as part of the landscape matrix, they still see them only as isolated crops and not as entities limited by the private property (farms, haciendas, ejidos, cooperatives) and therefore do not consider that agroecosystems can themselves contain ecosystems [
59,
60].
A conceptual approach to this process was made, debating the ideas that several researchers have in relation to the concept of agroecosystem because some of them claim that it can be a crop, whereas others argue that an agroecosystem is a cultivation site (crop), an individual farm or a set of farms distributed in the landscape [
18]. The author reviewed different definitions and concepts from the economy (which uses terms such as production systems applied to peasant, agro-industrial or capitalist units), sociology (which uses concepts related to property and land tenure when referring to agroecosystems of small, large or medium-sized landowners, tenants, smallholdings, settlers or indigenous or African-American farms), or common terms (i.e., coffee or livestock agroecosystems).
From that discussion, the author proposed to differentiate three hierarchical classes: agroecosystems of lower level (crops, meadows or forest systems), agroecosystems of higher level (farms, haciendas, ejidos, cooperatives or any other legal name that delimits and grants rights of property) and matrix of agroecosystems (aggregates of farms in the landscape).
The farm is a unit of analysis of much greater spatial and temporal stability than crops and other areas dedicated to pasture or agroforestry sites; therefore, it can be defined more easily and with greater precision, although it is recognized that change is an inherent characteristic of agroecosystems (major and minor). The truth is that the temporary mobility (monthly, biannual, annual, biannual or in lustrums and decades) of the farming plots that make up the farms is a major obstacle for its characterization and denomination as agroecosystems.
The farm subsumes not only the sites or areas of cultivation or livestock and forestry production but it also involves all the logic and production possibilities marked by the will of the owners, who are affected by other parameters, such as land titles, technical assistance, access to credit, institutional support, extension and research services, quality of infrastructure, responses to market demands or public policies. Likewise, the effects of technologies are better manifested at the farm scale than at the crop level [
18].
In any case, most of the efforts to increase the connectivity of natural vegetation are made within the major agroecosystems or farms, a situation that substantially affects the productive system.
The index of the Main Agroecological Structure emerges as the answer for this need of connecting the landscape with the elements of the farms. The MAS has five ecosystem or biological indicators (connection of the farm with the ecological structure of the landscape, extension of external connectors, extension of internal connectors, diversity of external connectors and diversity of internal connectors) and five indicators of cultural order (land use and soil conservation, weed management, other ecological or conventional management practices, perception-awareness and capacity for action).
There are no rigid boundaries that determine this categorization (biological or cultural), so the following are the arguments supporting their selection. The first five indicators, of a biological nature, represent part of the functional diversity proposed by some authors [
61], measured in terms of extension, percentage and diversity, at the scale of the farm. They give an idea of the connectivity of the edges of the farms and their communication with the surrounding landscape. The linear extension and the percentages regarding the perimeter of the farms can be easily measured, as well as their diversity (even by using other known indicators such as Shannon or Simpson).
The first indicator (connection of the farm with the ecological structure of the landscape) assesses the distance of the farm or major agroecosystem in relation to the surrounding fragments of natural vegetation (mainly forest covers) and the quality of the existing corridors. In this sense, it is worth noting that there are different kinds of fragments and corridors, as well as different types of distances that serve for the communication of several species. Thus, a given fragment will not have the same value if the assessment is made for predatory insects than for birds or bats that affect agricultural production.
Several authors recognize the importance of the links connecting elements of the farms (hedgerows or living fences) with the corridors and patches of natural vegetation, as well as their high variability of origin, floral composition and structure [
62,
63]. The literature on these topics has increased in recent years, noting how fences and hedgerows generate ecological functions of habitat for birds, small mammals and reptiles and how they play key roles in the regulation of microclimatic conditions (speed of wind, frost prevention, evapotranspiration) [
64].
But hedgerows and live fences have not only been studied from the point of view of soil protection or microclimatic regulation but also from the perspective of their influence on the regulation of insect herbivorous, the management of the associated biodiversity and, of course, in relation to agronomic factors of production and plant health [
61,
65,
66].
The extension and diversification of internal and external connectors (the other four indicators of the biological order) are useful for the integral management of the major agroecosystem and its components (production, regulation of insects and diseases, soil conditioners, microclimatic controls or germplasm banks, among other aspects). There is an extensive bibliography on the subject compiled by Altieri and Nicholls [
61] revealing that vegetation installed in rows around or within the fields of cultivation has great influence on the presence, abundance, mobility, class and lapses of permanence of phytophages and their natural enemies. A feature of the hedgerows that is evaluated by these indicators is precisely their contribution in terms of functionality for the management of the agroecosystem. This functionality is expressed in the greatest diversity possible; that is, in the presence of plants that offer pollination services, decoys or traps, allelopathy, shelter, food, effects against frost, temperature and humidity regulations, decrease in surface runoff, shade and erosion control, among other services.
The remaining five indicators belong to the cultural order, because they depend on and are explained by human behavior. The land uses (sixth indicator) of a farm are the result of factors such as educational processes, traditions, the external influence of agricultural policies (taxes, remuneration rates, incentives) and material possibilities (family composition, income, markets, availability of labor). This is also true for soil conservation practices, which depend on technological, social, economic and political factors.
The management of weeds (seventh indicator) is the direct result of the level of knowledge of the farmer and the type of the system (whether ecological or conventional), which corresponds to the eighth indicator and is referred to in this paper as “Other Management Practices.” These practices are also the result of complex cultural variables that determine the farmer’s position in favor or against the management of agrobiodiversity.
Finally, the level of awareness and knowledge of farmers about the benefits (ninth indicator of the index) and the real possibilities of planting hedges, fences or live fences (tenth indicator), also emerge from cultural processes related to training, education and financial capacities of farmers.
The parameters proposed by the authors of the methodology [
67,
68] for the evaluation of the MAS are summarized in
Table 1.
Each of the aforementioned indicators are evaluated in a 0 to 10 scale, according to the following tables and criteria developed by the authors [
18].
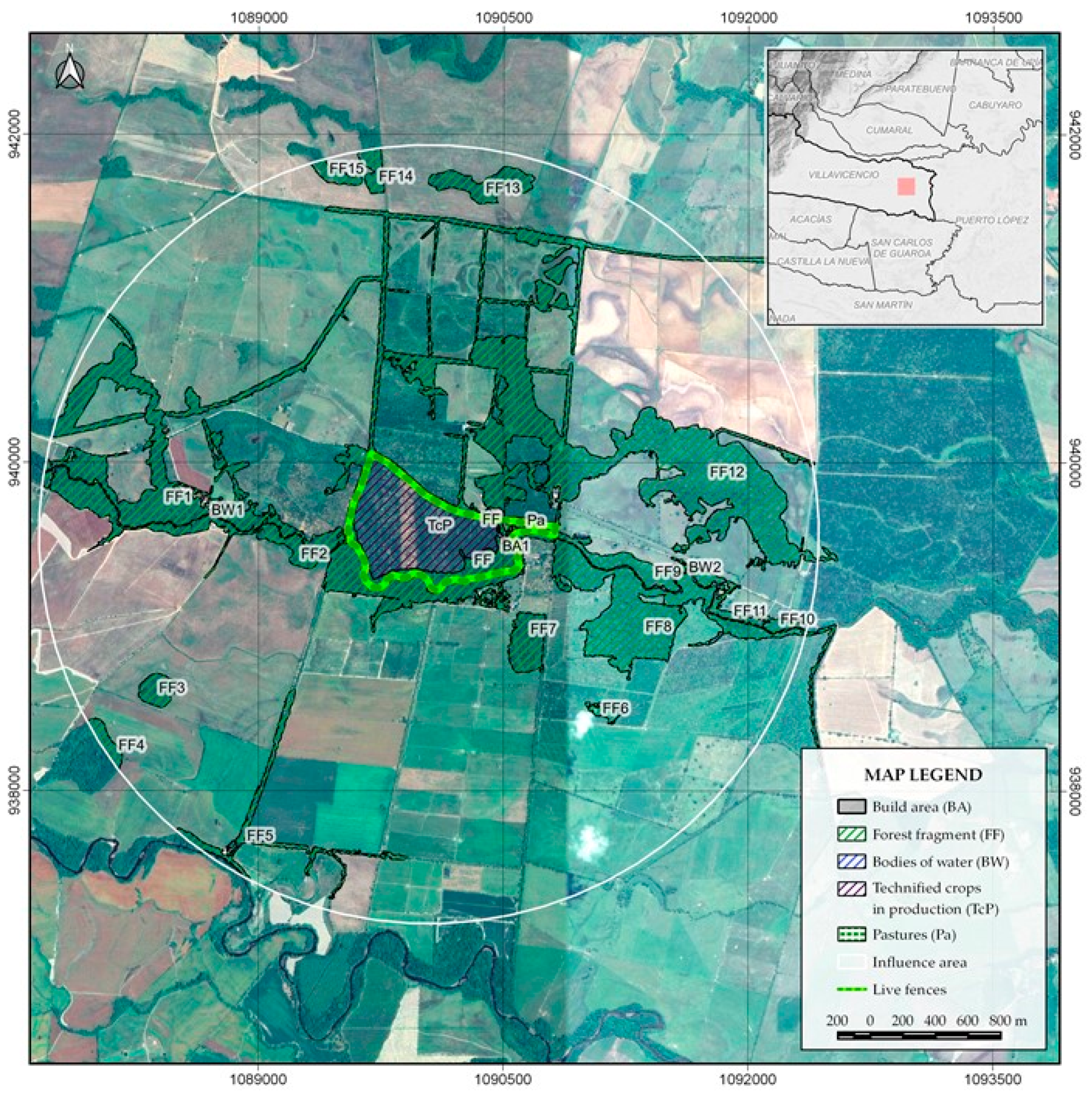
3.1. Connection of the Farm with the Main Ecological Landscape Structure (CMELS)
This parameter assesses the relationships of the agroecosystem with the elements of the surrounding landscape through metrics such as the density of patches or fragments of natural vegetation and bodies of water, as well as the distance between them and their average distance to the center of the agroecosystem. It can be determined through the following procedure by using any type of remote sensing image, such as aerial photographs, satellite or drone images (
Figure 1):
Define the area of influence of the landscape of the studied farm, starting from a circle whose center coincides with the center of the major agroecosystem. The circle has a radius that is twice the longest side of the farm.
Determine all the types of cover (especially fragments of forests, shrubs, crops and bodies of water) outside the major agroecosystem but within the area of influence of the circle. This step can be done by using the categories proposed by the Corine (Coordination of Information System on Environment) Land Cover methodology.
Determine the average distance of the identified vegetation covers to the center of the farm (which is also the center of the area of influence).
All the obtained percentages are weighted and added, on a scale of 0 to 10. The resulting value represents, in this numerical scale, the Connection of the major agroecosystem with the Main Ecological Landscape Structure (CMELS).
The scales presented in
Table 2 are intended for the interpretation of the distances and qualification of the attributes analyzed in the determination of the CMELS.
3.2. Extension of the External Connectors (EEC)
This parameter evaluates the linear extension and the surface of live fences located in the perimeter of the farms (
Table 3). A remote sensing image can also be used for estimating the EEC.
3.3. Extension of the Internal Connectors (EIC)
This parameter measures the linear extension of the rows of vegetation but at the internal level of the agroecosystem (
Table 4). Remote sensing images are also useful for this purpose.
Figure 2 presents an example of delimitation of external and internal connectors in a farm located in the Colombian Orinoquia region, made from Google—Earth images.
3.4. Diversity of External Connectors (DEC)
This parameter assesses the diversity and functionality of live fences or hedges located in the perimeter of the major agroecosystem (
Table 5). This analysis includes sampling the types of herbaceous, shrub, or tree vegetation that are part of these living fences. Samples can be taken using the same fences as transects or a random sampling can also be made in plots of different areas. The collected material is then identified according to the vernacular and scientific names (with the support of botanists and local inhabitants) and classified within the taxonomic categories of family, gender and, as far as possible, species. Other biodiversity indexes (abundance, wealth, equitability) can be used depending on the financial resources and the objectives of the research.
3.5. Diversity of the Internal Connectors (DIC)
This criterion evaluates the diversity of the live fences inside the farm (
Table 6). The same methodology described for the determination of the DEC is suggested.
3.6. Use and Soil Conservation (USC)
This parameter evaluates the distribution percentage of different covers associated with the productive activities of the farm. The Corine Land Cover methodology can be used for the calculation of the USC (
Table 7).
3.7. Weed Management (WM)
This parameter assesses the practices and management systems of weeds (
Table 8).
3.8. Other Management Practices (OP)
This indicator expresses the management types of each farm (ecological, conventional, or in the process of transition or reconversion) (
Table 9).
3.9. Perception—Awareness (PA)
It assesses the degree of conceptual clarity and awareness of farmers in relation to the use and management of agrobiodiversity (
Table 10).
3.10. Level of Capacity for Action (CA)
In addition to the commitment, it assesses the capacities and possibilities of farmers to establish, maintain or improve the MAS of their productive units (
Table 11). The evaluation of this and other parameters of the cultural order (WM, OP and PA) are determined through surveys or interviews conducted with the owners or managers of the farms.
The final calculation of the MAS of the major agroecosystems is obtained by adding the resulting value of each of the aforementioned indicators, according to the following equation (unweighted):
The scale of interpretation of the MAS is indicated in
Table 12.
4. Applications of the MAS
The MAS was applied for the first time in Colombia in a high Andean horticultural area [
67]. This study compared 6 ecological farms (from 20 to 0.16 hectares) based in the savannah of Bogotá by assessing the ten aforementioned parameters or indicators. The results revealed values of MAS between 47 (slightly developed) and 81 (strongly developed), which accounts for substantial differences in the management of the studied farms and a high degree of isolation from the ecological structure of the landscape. This first exercise suggested that the index could be applied in different socioeconomic conditions and in different natural regions of the country, so further studies were carried out in other areas of peasant production in Colombia (Anolaima, Quipile and Pulí in the department of Cundinamarca) and in coffee agroecosystems with greater biodiversity.
In this sense, we studied the ecosystem and cultural resilience of six coffee agroecosystems (three ecological and three conventional) in Anolaima (Colombian Andes) with regard to climate variability [
68]. All the ecological farms presented better MAS conditions and greater resilience than the conventional ones, although they were all were located in a region characterized by the soil susceptibility to mass movements and responded to a common history of settlement and similar socioeconomic conditions. In another study conducted in coffee agroecosystems of Quipile and Pulí (Cundinamarca) [
69], the MAS was tested in an area with different degrees of agricultural intensification and the results were compared with the typing methodology of Moguel and Toledo [
70]. The authors measured local variables associated with agricultural management in different types of covers (richness, density and average height of trees, average density and height of coffee trees, weed richness, percentage of canopy cover and distance to the nearest forest) and determined the types and frequency of application of agrochemicals and other cultural management variables.
The authors found a good correlation of the classification obtained by Moguel and Toledo with the classification proposed in the study. In addition, they determined that the floral diversity and natural spaces close to the crop substantially affected the richness of the visiting bees. They proposed that the MAS (through diversified productive arrangements and with high internal and external connectivity) generates synergistic effects that increase variety of bee species within the coffee field and enhance their conservation and ecological function of pollination.
Another study carried out to explain the socio-environmental reasons of voluntary urban migration towards intentional rural communities [
71] assessed the ecosystem effects of this migration at the level of the MAS of the farms. The study took place in three ecovillages of the department of Cundinamarca (“Varsana” located in the municipality of Granada, “El Retoño” in the municipality of Silvania and “Aldea Feliz” in the municipality of San Francisco) and it used ethnographic techniques and vegetation sampling. Results showed that the migrants contributed positively in the improvement of the agrobiodiversity of the farms, measured through the MAS. Varsana (whose inhabitants arrived in 1979) went from an initial MAS of 18 (weakly developed) to values of 80 (strongly developed) in 2013. El Retoño went from 26 to 81 in 15 years (1998–2013) and Aldea Feliz from 63 to 92 in 7 years (2006–2013). Such modifications were linked to the personal motivations of migrants who seek in the rural areas elements of healthy living, satisfaction of spiritual aspirations and vindication of values of solidarity and respect, accompanied by equalitarian administrative and economic structures.
The MAS was also applied to the case of the resilience of citrus agroecosystems to climate variability in the Colombian Orinoquia and 18 farms located in the Orinoquia foothills [
72,
73] were included in this study. We found a high correlation between the MAS and the decrease in the number of phytosanitary controls carried out by Valencia orange producers (
Citrus sinensis (L.) Osbeck), as well as positive correlations with the productivity of the studied farms.
Finally, we conducted a preliminary exercise to include agroecosystems (beyond the Corine Land Cover classification) in the national ecosystem map of Colombia and for this purpose we proposed to assess the MAS as an agroecosystem connectivity criterion that could be represented even in maps at 1:100,000 scale [
74].
The previous examples show that the MAS of the major agroecosystems can become a valuable tool for the study and planning of agroecosystems, both at the level of their internal management and also in matters related to territorial planning [
75].
At a farm scale, the MAS facilitates the understanding of the interrelationships provided by agrobiodiversity. Although specific studies have not been made yet in this field, it is possible to foresee positive relationships between MAS and the abundance of pollinators or natural enemies and of varied intra- and interspecific relationships of different organisms, which are agents or vectors of diseases. The soil biodiversity is included in this category and it can be improved by the influence of agrobiodiversity management, via the reinforcement of the MAS.
The author of the concept [
18] has noted that the MAS can help planning the activities of the farm through the so-called ‘potential MAS,’ that is, the location, layout and implementation of better and more diversified internal and external corridors, including the management of patches of natural vegetation within the farms (ecosystems within agroecosystems). The MAS allows calculating the selection of flower banks and the introduction of certain shrub or forest species, which have already been proven beneficial for the production or control of insect populations, as has been proven in the intensive silvopastoral systems [
76,
77].
On the other hand, the MAS provides explanations, at the farm scale, of landscape connectivity. It is no longer a matter of considering the biological corridors, the patches of vegetation or the remaining forests as loose pieces in the landscape. The MAS allows them to be grouped around the agricultural production units and therefore it highlights a series of cultural relations (economic, social, symbolic and technological) which are not identified by the partial analysis of landscape ecology. In other words, the MAS is the entry of agricultural producers into the maps of the environmental planning of the territory.
Socio-ecological resilience recognizes that culture has a technological component that is incorporated by farmers in response to disturbances. From this perspective, the characterization of the MAS allows determining which cultural practices are necessary to increase agrobiodiversity (i.e., the development of agroforestry, the practice of allelopathy, the harvesting of water, the management of covers and the use of entomopathogens). With these practices, it is not only possible to increase yields but also the quantity and variety of foods, creating strong connections with food security, sovereignty and autonomy and improving the nutritional processes of farmers and their communities [
78].
The above-mentioned points demonstrate the relevance of the evaluation of the MAS as a highly useful methodology for decision-making. It can help farmers make adjustments in their agroecosystems: by improving connectivity and biodiversity, the resilience and productivity of their agricultural and livestock units can increase, as well as their income, quality of life and food autonomy.
At the administrative level, the MAS can become an important input for the design of public development policies with a territorial approach, based on local needs and the active participation of farmers. Finally, the MAS can also become a useful theoretical-practical instrument to make progresses in the taxonomy of agroecosystems, an issue that the science of agroecology has not yet addressed.
Limits and Scope of the Proposed Methodology
The elements included in the formulation of the MAS refer to all those plant covers, intentionally planned or not, that are found in the major agroecosystem (crops, weeds, fences, hedgerows, corridors) or even relicts of natural forests or other types of covers (bushes, grasslands) and bodies of water. In other words, the index gathers the set of covers present in the farm, either as part of the productive system or as part of the biodiversity that has been cataloged by other authors as auxiliary or unintentional [
61]. Live fences or hedges, both internal and external, are considered as connectors under the definition of the MAS.
The novelty of this index is that it brings together biophysical indicators (length, percentage and diversity of internal and external connectors) with elements arise from the cultural order.
On the other hand, it should be understood that the MAS has no further pretensions than serving as a descriptor of the quality (structure) of agroecosystems, which is strongly related to agrobiodiversity. It does not replace other functions, features or qualities of agroecosystems that should be studied in a parallel but independent way. That is, the MAS must be separated from the obligatory study of soils or the corresponding climatic or geomorphological analyses and it does not substitute other studies which are necessary from the cultural point of view (socioeconomic, political, symbolic, technological). In this sense, the MAS can complement and relate to analyses of food security, sovereignty and autonomy, power relations, cooperative systems and markets but it cannot account for these and other cultural parameters that affect agroecosystems.
Of course, future research on the index should address its connections with multiple variables of different order: MAS—crop health; MAS—production; MAS—soil fertility; MAS—water conservation; MAS—nutritional needs of plants; MAS—food sovereignty; MAS—consumer health; or MAS—resilience. In the latter, the MAS could be used as one among many other factors that increase the capacity of transformation or response of the farms in the face of continuous change.
The proposed methodology can be improved in several aspects: first, the relationships of the farms with the surrounding landscape vary a lot, depending on key factors such as the built infrastructure (access roads, electrification), proximity to urban centers and the co-existence with other farms. These variables have not been included in this document but remain to be explored in further studies, in particular the relationship of the major agroecosystem with neighboring farms. This implies the possibility of delimiting the neighboring farms (and not only the covers) within the drawn circle, in order to make the analysis of the connection of the farm with the ecological structure of the landscape more robust and relevant. This supports the idea that forests, biological corridors and patches of natural vegetation are not loose in the landscape.
In this proposal, we have chosen to delimit a circle with a radius which is twice the size of the longest side of the farm, in order to include different covers adjacent to the major agroecosystem. However, this procedure remains arbitrary and our only argument was assuring proportionality in all measurements, regardless of the size of the farm. This proportion could be modified with the advance of new research, although in our studies (with farms of up to 350 hectares) it has provided positive results. The shape of the circle could also be modified and replaced by a square or, even better, by the existing geoforms.
In large and highly fragmented farms the MAS remains valid as a descriptive index of agrobiodiversity, since it is possible to make measurements of its internal and external connectors, without major difficulties. However, the internal relationships of agrobiodiversity change, compared to small farms. These changes should be also explored, due to the size of these agroecosystems that may affect the movement and action of many other living organisms. But this is not a failure of the index itself; it is a challenge of agroecology as a science.
Variability and change are two key aspects to understand the dynamics of agrobiodiversity within farms. In previous paragraphs, we mentioned that minor agroecosystems or crops can change over time but this does not invalidate the MAS, as it recognizes crops as a general category of land use, regardless of its nature (monthly, annual or permanent crops).
On the other hand, we have not yet considered the possibility of weighing the indicators, because we want to initially register the biophysical features of the agrobiodiversity of the major agroecosystems and this depends on measurements in the field, not on the weighting. However, the cultural determinants, which are related to land use, weed management, other management practices (ecological and conventional), perception/awareness and the ability to establish MAS, can be the subject of future studies. We can highlight the fact that the used management system (ecological or conventional) represents a key factor in the understanding of agrobiodiversity, especially in aspects related to the use of pesticides, which may be underestimated if the indicators considered in the index are not weighted.
We want to insist on the need of including in the index the two indicators most related to culture (Perception/Awareness and Capacity for Action) because the MAS is the result of a human process of intervention on the natural world. The maximum expression of this cultural intervention is agrobiodiversity and it is built according to the conscience, desires, education, economic and financial possibilities of farmers. We have grouped all these factors in only two synthetic indicators, with the epistemological risk that this implies.
Finally, the MAS encourages the dialogue between different sciences (botany, zoology, conservation biology, ethnohistory, anthropology) and allows the participation of farmers in the exchange of knowledge about the different plant covers and uses of the plants. We are currently advancing in the identification and valuation of MAS with groups of farmers and without turning to geographic information systems. The intention is bringing the studies on agrobiodiversity closer to the local communities, who are, in the end, those who know deeply their natural environments and those who have the final word on their own sowing processes.