Adsorption and Desorption Behaviors of Four Endocrine Disrupting Chemicals in Soils from the Water-Level Fluctuation Zone of the Three Gorges Reservoir, China
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Sampling Site and Soil Samples
2.3. Adsorption and Desorption Experiments
2.4. Data Analysis
3. Results and Discussion
3.1. Characterization of Soils
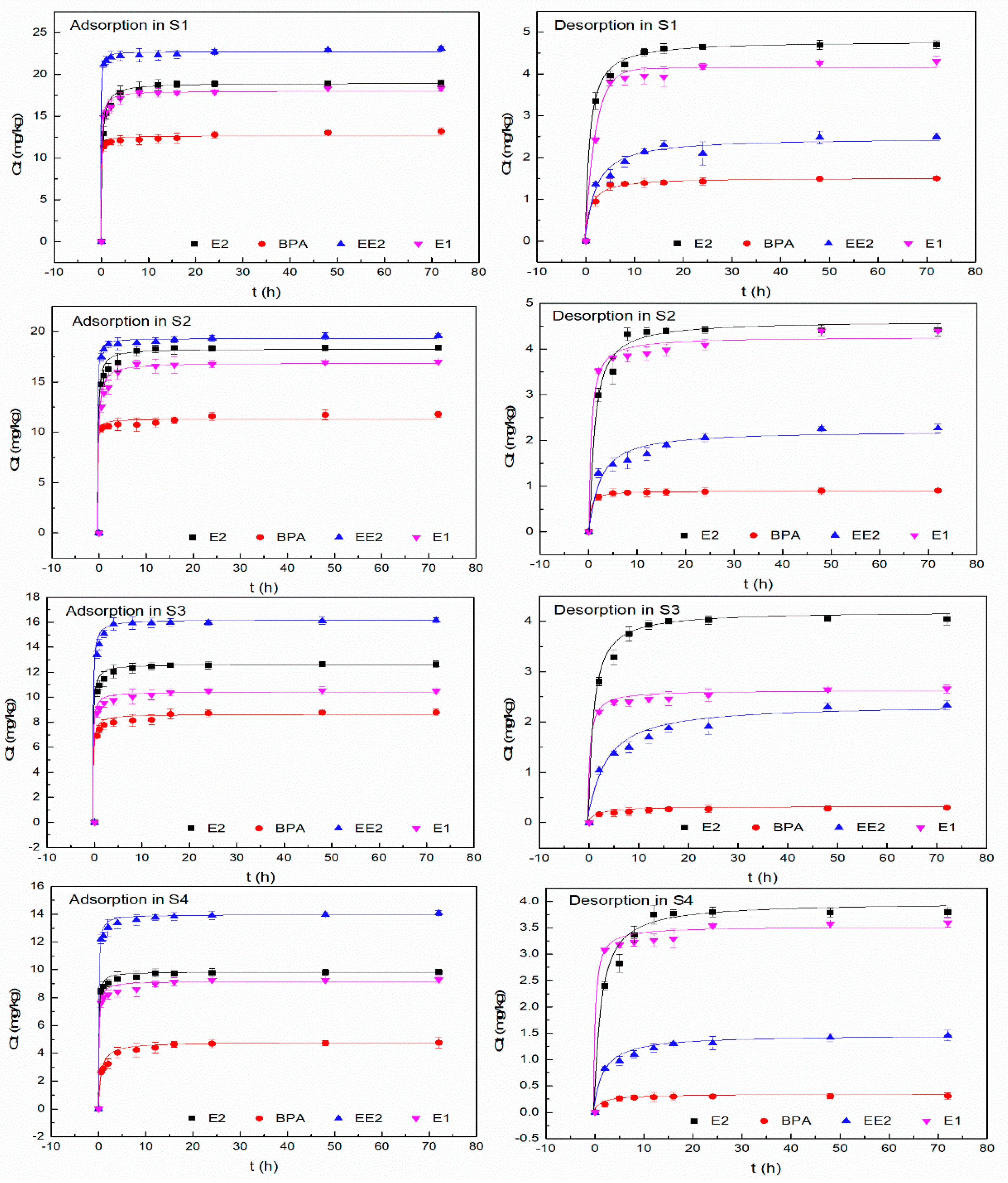
3.2. Adsorption and Desorption Kinetics
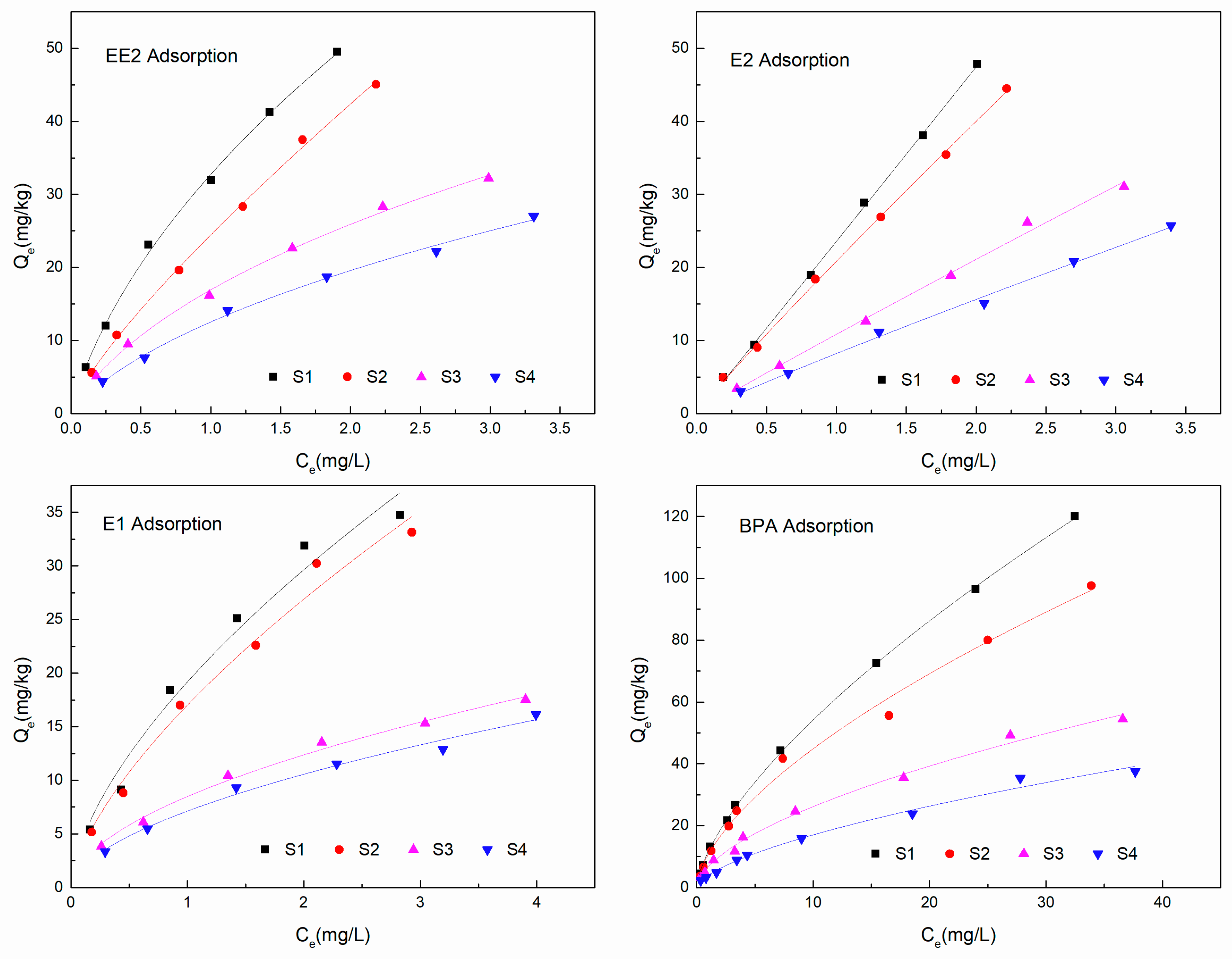
3.3. Adsorption Isotherms and Nonlinearity
3.4. Desorption Isotherms and Hysteresis
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Giulivo, M.; De Alda, M.L.; Capri, E.; Barceló, D. Human exposure to endocrine disrupting compounds: Their role in reproductive systems, metabolic syndrome and breast cancer. A review. Environ. Res. 2016, 151, 251–264. [Google Scholar] [CrossRef] [PubMed]
- Hauser, R.; Skakkebaek, N.E.; Hass, U.; Toppari, J.; Juul, A.; Andersson, A.M. Male reproductive disorders, diseases, and costs of exposure to endocrine-disrupting chemicals in the European Union. J. Clin. Endocrinol. Metab. 2015, 100, 1267–1277. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Eom, J.; Kim, J.; Lee, S.; Kim, Y. Association between some endocrine-disrupting chemicals and childhood obesity in biological samples of young girls: A cross-sectional study. Environ. Toxicol. Pharmacol. 2014, 38, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Gorga, M.; Insa, S.; Petrovic, M.; Barceló, D. Occurrence and spatial distribution of EDCs and related compounds in waters and sediments of Iberian rivers. Sci. Total Environ. 2015, 503, 69–86. [Google Scholar] [CrossRef] [PubMed]
- Thomson, B.M.; Grounds, P.R. Bisphenol A in canned foods in New Zealand: An exposure assessment. Food Addit. Contam. 2005, 22, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Rudel, R.A.; Perovich, L.J. Endocrine disrupting chemicals in indoor and outdoor air. Atmos. Environ. 2009, 43, 170–181. [Google Scholar] [CrossRef] [PubMed]
- Duong, C.N.; Jinsung, R.; Jaeweon, C.; Kim, S.D.; Choi, H.K. Estrogenic chemicals and estrogenicity in river waters of South Korea and seven Asian countries. Chemosphere 2010, 78, 286–293. [Google Scholar] [CrossRef] [PubMed]
- Weber, J.W., Jr.; Mcginley, P.M.; Katz, L.E. Sorption phenomena in subsurface systems: Concepts, models and effects on contaminant fate and transport. Water Res. 1991, 25, 499–528. [Google Scholar] [CrossRef]
- Bao, Y.; Gao, P.; He, X. The water-level fluctuation zone of Three Gorges Reservoir—A unique geomorphological unit. Earth Sci. Rev. 2015, 150, 14–24. [Google Scholar] [CrossRef]
- Peng, C.R.; Lang, Z.; Qin, H.J.; Li, D.H. Revegetation in the water level fluctuation zone of a reservoir: An ideal measure to reduce the input of nutrients and sediment. Ecol. Eng. 2014, 71, 574–577. [Google Scholar] [CrossRef]
- Ye, C.; Li, S.; Zhang, Y.; Zhang, Q. Assessing soil heavy metal pollution in the water-level-fluctuation zone of the Three Gorges Reservoir, China. J. Hazard. Mater. 2011, 191, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Jiang, T.; Huang, R.; Wang, D.; Zhang, J.; Qian, S.; Yin, D.; Chen, H. A simulation study of inorganic sulfur cycling in the water level fluctuation zone of the Three Gorges Reservoir, China and the implications for mercury methylation. Chemosphere 2017, 166, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Fang, F.; Guo, J.; Chen, Y.; Li, Z.; Guo, S. Phosphorus fractions and phosphate sorption-release characteristics relevant to the soil composition of water-level-fluctuating zone of Three Gorges Reservoir. Ecol. Eng. 2012, 40, 153–159. [Google Scholar] [CrossRef]
- Zhao, Y.; Wu, B.F.; Zeng, Y. Spatial and temporal aspects of greenhouse gas emissions from Three Gorges Reservoir, China. Biogeosci. Discuss. 2012, 9, 14503–14535. [Google Scholar] [CrossRef]
- Tang, Y.M.; Junaid, M.; Niu, A.; Deng, S.; Pei, D.S. Diverse toxicological risks of PAHs in surface water with an impounding level of 175 m in the Three Gorges Reservoir Area, China. Sci. Total Environ. 2016, 580, 1085–1096. [Google Scholar] [CrossRef] [PubMed]
- Floehr, T.; Scholz-Starke, B.; Xiao, H.; Koch, J.; Wu, L.; Hou, J.; Wolf, A.; Bergmann, A.; Bluhm, K.; Yuan, X.; et al. Yangtze Three Gorges Reservoir, China: A holistic assessment of organic pollution, mutagenic effects of sediments and genotoxic impacts on fish. J. Environ. Sci. 2015, 38, 63–82. [Google Scholar] [CrossRef] [PubMed]
- Hu, T.; Zhang, J.; Ye, C.; Zhang, L.; Xing, X.; Zhang, Y.; Wang, Y.; Sun, W.; Qi, S.; Zhang, Q. Status, source and health risk assessment of polycyclic aromatic hydrocarbons (PAHs) in soil from the water-level-fluctuation zone of the Three Gorges Reservoir, China. J. Geochem. Explor. 2017, 172, 20–28. [Google Scholar] [CrossRef]
- Loffredo, E. Fate of anthropogenic organic pollutants in soils with emphasis on adsorption/desorption processes of endocrine disruptor compounds. Pure Appl. Chem. 2006, 78, 947–961. [Google Scholar] [CrossRef]
- Sun, K.; Gao, B.; Zhang, Z.Y.; Zhang, G.X.; Liu, X.T. Sorption of endocrine disrupting chemicals by condensed organic matter in soils and sediments. Chemosphere 2010, 80, 709–715. [Google Scholar] [CrossRef] [PubMed]
- Lima, D.L.; Schneider, R.J.; Esteves, V.I. Sorption behavior of EE2 on soils subjected to different long-term organic amendments. Sci. Total Environ. 2012, 423, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Bonin, J.L.; Simpson, M.J. Sorption of steroid estrogens to soil and soil constituents in single- and multi-sorbate systems. Environ. Toxicol. Chem. 2007, 26, 2604–2610. [Google Scholar] [CrossRef] [PubMed]
- Stumpe, B.; Marschner, B. Dissolved organic carbon from sewage sludge and manure can affect estrogen sorption and mineralization in soils. Environ. Pollut. 2010, 158, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Jia, G.; Liu, X. Soil microbial biomass and metabolic quotient across a gradient of the duration of annually cyclic drainage of hillslope riparian zone in the three gorges reservoir area. Ecol. Eng. 2017, 99, 366–373. [Google Scholar] [CrossRef]
- Reoyo-Prats, B.; Aubert, D.; Menniti, C.; Ludwig, W.; Sola, J.; Pujo-Pay, M.; Conan, P.; Verneau, O.; Palacios, C. Multicontamination phenomena occur more often than expected in Mediterranean coastal watercourses: Study case of the Têt River (France). Sci. Total Environ. 2017, 579, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Luo, Y.; Wang, C.; Li, B.; Zhang, X.; Yuan, D.; Gao, X.; Zhang, H. Spatiotemporal variations and factors affecting soil nitrogen in the purple hilly area of Southwest China during the 1980s and the 2010s. Sci. Total Environ. 2016, 547, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Defersha, M.B.; Melesse, A.M. Effect of rainfall intensity, slope and antecedent moisture content on sediment concentration and sediment enrichment ratio. Catena 2012, 90, 47–52. [Google Scholar] [CrossRef]
- Zeng, F.Y.; Jia, G.M.; Chen, F.Q.; Cao, L. The Effect of Different Flooding Duration on Soil Organic Carbon and its Labile Fraction in the Riparian Zone of Three Gorges Reservoir Area. Adv. Mater. Res. 2013, 726, 3757–3761. [Google Scholar] [CrossRef]
- Tang, X.; Wu, M.; Li, Q.; Lin, L.; Zhao, W. Impacts of water level regulation on sediment physic-chemical properties and phosphorus adsorption–desorption behaviors. Ecol. Eng. 2014, 70, 450–458. [Google Scholar] [CrossRef]
- Zhang, B.; Guo, J.S.; Fang, F.; Li, Z.; Fu, C. Concentration of nutrients in the soil in water-level-fluctuating zone of three gorges reservoir. Ecohydrol. Hydrobiol. 2012, 12, 105–114. [Google Scholar] [CrossRef]
- Li, J.; Jiang, L.; Liu, X.; Lv, J. Adsorption and aerobic biodegradation of four selected endocrine disrupting chemicals in soil–water system. Int. Biodeterior. Biodegrad. 2013, 76, 3–7. [Google Scholar] [CrossRef]
- Martínezhernández, V.; Meffe, R.; Herrera, L.S.; De, B.I. The role of sorption and biodegradation in the removal of acetaminophen, carbamazepine, caffeine, naproxen and sulfamethoxazole during soil contact: A kinetics study. Sci. Total Environ. 2016, 559, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Shen, D.Z.; Fan, J.X.; Zhou, W.Z.; Gao, B.Y.; Yue, Q.Y.; Qi, K. Adsorption kinetics and isotherm of anionic dyes onto organo-bentonite from single and multisolute systems. J. Hazard. Mater. 2009, 172, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Apul, O.G.; Karanfil, T. Adsorption of synthetic organic contaminants by carbon nanotubes: A critical review. Water Res. 2015, 68, 34–55. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Thomas, M.Y.; Mark, A.S.; Yu, H., Jr.; Weber, W.J. A Distributed Reactivity Model for Sorption by Soils and Sediments. 9. General Isotherm Nonlinearity and Applicability of the Dual Reactive Domain Model. Environ. Sci. Technol. 1996, 26, 3549–3555. [Google Scholar] [CrossRef]
- And, J.J.P.; Xing, B. Mechanisms of Slow Sorption of Organic Chemicals to Natural Particles. Environ. Sci. Technol. 1996, 30, 1–11. [Google Scholar]
- Fuentes, B.; de la Luz Mora, M.; Bol, R.; San Martin, F.; Pérez, E.; Cartes, P. Sorption of inositol hexaphosphate on desert soils. Geoderma 2014, 232–234, 573–580. [Google Scholar] [CrossRef]
- Sukul, P.; Lamshöft, M.; Zühlke, S.; Spiteller, M. Sorption and desorption of sulfadiazine in soil and soil-manure systems. Chemosphere 2008, 73, 1344–1350. [Google Scholar] [CrossRef] [PubMed]
- Thielebruhn, S.; Seibicke, T.; Schulten, H.R.; Leinweber, P. Sorption of sulfonamide pharmaceutical antibiotics on whole soils and particle-size fractions. J. Environ. Qual. 2004, 33, 1331–1342. [Google Scholar] [CrossRef]
- Fei, Y.; Xing, B.; Li, X. Changes in the adsorption of bisphenol A, 17 α-ethinyl estradiol, and phenanthrene on marine sediment in Hong Kong in relation to the simulated sediment organic matter decomposition. Environ. Pollut. 2014, 192, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Pan, B.; Ning, P.; Xing, B. Part IV—sorption of hydrophobic organic contaminants. Environ. Sci. Pollut. Res. Int. 2008, 15, 554–564. [Google Scholar] [CrossRef] [PubMed]
- Hao, R.; Wang, P.; Wu, Y.; Hu, R.; Zhang, J.; Song, Y. Impacts of water level fluctuations on the physicochemical properties of black carbon and its phenanthrene adsorption-desorption behaviors. Ecol. Eng. 2017, 100, 130–137. [Google Scholar] [CrossRef]
- Oepen, B.V.; Kördel, W.; Klein, W. Sorption of nonpolar and polar compounds to soils: Processes, measurements and experience with the applicability of the modified OECD-Guideline 106. Chemosphere 1991, 22, 285–304. [Google Scholar] [CrossRef]
- Yuan, G.S.; Xing, B.S. Effects of metal cations on sorption and desorption of organic compounds in humic acids. Soil Sci. 2001, 166, 205–208. [Google Scholar] [CrossRef]
- Lesan, H.M.; Bhandari, A. Atrazine sorption on surface soils: Time-dependent phase distribution and apparent desorption hysteresis. Water Res. 2003, 37, 1644–1654. [Google Scholar] [CrossRef]
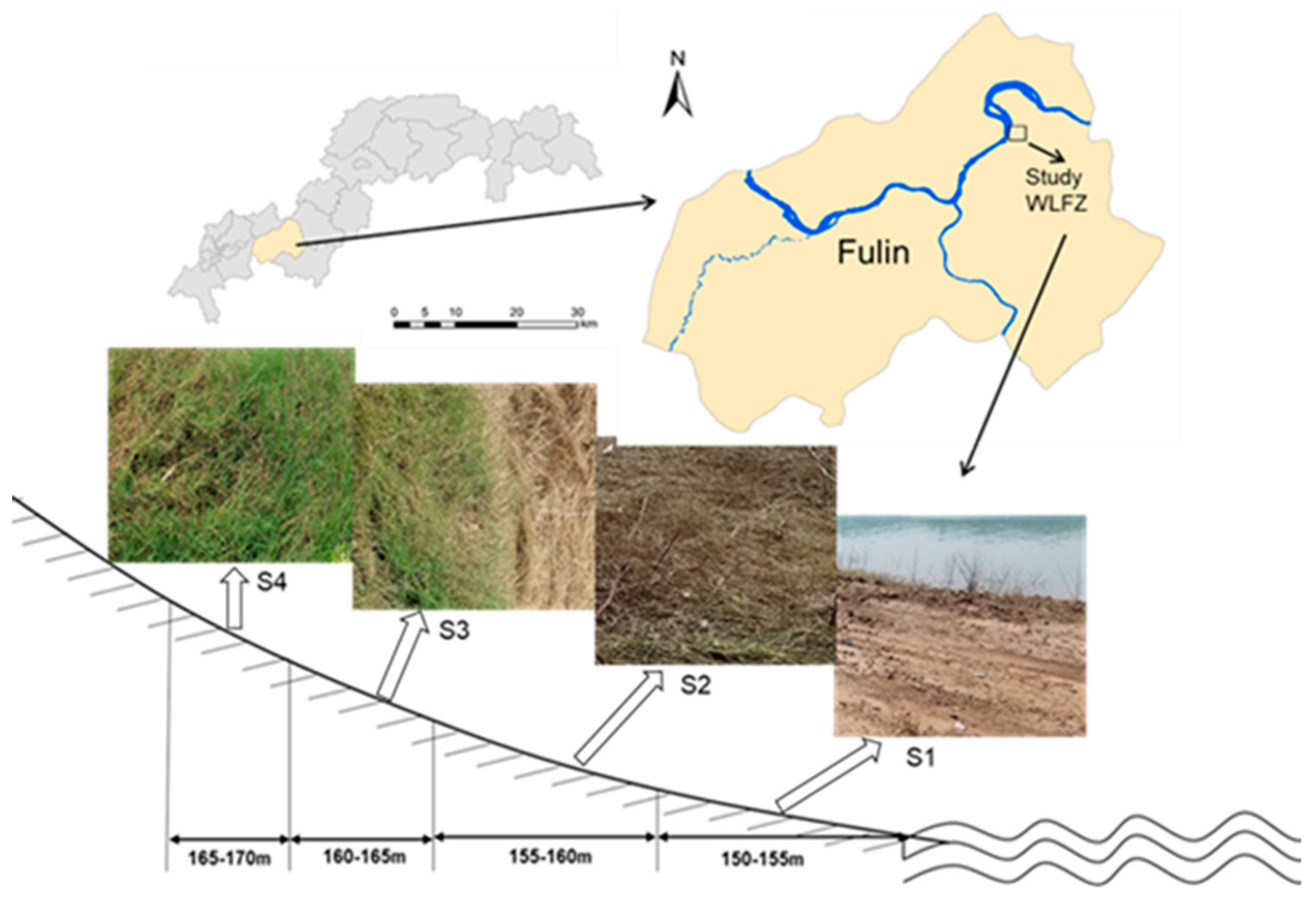


| Property | Soil Sample | |||
|---|---|---|---|---|
| S1 | S2 | S3 | S4 | |
| Elevation gradient/m | 150–155 | 155–160 | 160–165 | 165–170 |
| Average slope | 15° | 20° | 25° | 30° |
| Vegetation | none | Cynodon dactylon | Cynodon dactylon | Cynodon dactylon, Xanthium |
| pH | 7.79 | 7.60 | 8.13 | 8.30 |
| TOC/(g/kg) | 18.50 | 16.84 | 9.33 | 8.26 |
| TN/(g/kg) | 2.32 | 2.05 | 1.09 | 0.86 |
| Sand (≥0.05 mm)/% | 0.53 | 4.59 | 25.01 | 26.93 |
| Silt (0.002–0.05 mm)/% | 79.68 | 78.01 | 59.66 | 57.29 |
| Clay (≤0.002 mm)/% | 19.79 | 17.40 | 15.33 | 15.78 |
| EDC | Soil Sample | Qe (exp) mg/kg | Elovich Equation | Pseudo-First-Order | Pseudo-Second-Order | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| a | b | R2 | K1 1/h | Qe mg/kg | R2 | K2 kg/(mg·h) | Qe mg/kg | R2 | |||
| EE2 | S1 | 23.10 | 21.6 | 0.339 | 0.947 | 5.64 | 22.42 | 0.996 | 0.180 | 23.09 | 0.997 |
| S2 | 19.60 | 18.1 | 0.377 | 0.911 | 4.80 | 19.07 | 0.995 | 0.238 | 19.65 | 0.996 | |
| S3 | 16.16 | 14.4 | 0.513 | 0.792 | 3.46 | 15.79 | 0.989 | 0.411 | 16.18 | 0.998 | |
| S4 | 14.08 | 12.6 | 0.388 | 0.932 | 4.22 | 13.60 | 0.987 | 0.321 | 14.08 | 0.998 | |
| E2 | S1 | 18.90 | 15.2 | 1.126 | 0.828 | 2.13 | 18.33 | 0.977 | 0.224 | 18.97 | 0.998 |
| S2 | 18.40 | 15.8 | 0.781 | 0.884 | 3.16 | 17.78 | 0.973 | 0.288 | 18.45 | 0.997 | |
| S3 | 12.64 | 11.1 | 0.449 | 0.889 | 3.49 | 12.26 | 0.982 | 0.438 | 12.67 | 0.999 | |
| S4 | 9.84 | 8.84 | 0.285 | 0.920 | 4.07 | 9.55 | 0.987 | 0.571 | 9.86 | 0.997 | |
| E1 | S1 | 18.36 | 15.7 | 0.725 | 0.928 | 3.38 | 17.53 | 0.975 | 0.164 | 18.42 | 0.994 |
| S2 | 16.99 | 14.0 | 0.883 | 0.853 | 2.52 | 16.35 | 0.971 | 0.215 | 17.04 | 0.996 | |
| S3 | 10.50 | 9.17 | 0.378 | 0.942 | 3.61 | 10.12 | 0.980 | 0.398 | 10.55 | 0.997 | |
| S4 | 9.30 | 7.96 | 0.357 | 0.955 | 3.63 | 8.84 | 0.974 | 0.316 | 9.34 | 0.994 | |
| BPA | S1 | 13.20 | 11.6 | 0.334 | 0.941 | 4.68 | 12.44 | 0.986 | 0.152 | 13.21 | 0.993 |
| S2 | 11.80 | 10.4 | 0.304 | 0.901 | 5.09 | 11.13 | 0.982 | 0.177 | 11.85 | 0.993 | |
| S3 | 8.80 | 7.40 | 0.376 | 0.942 | 3.20 | 8.35 | 0.974 | 0.282 | 8.85 | 0.995 | |
| S4 | 4.77 | 3.10 | 0.469 | 0.925 | 1.14 | 4.45 | 0.917 | 0.268 | 4.82 | 0.998 | |
| EDC | Soil Sample | Qe (exp) mg/kg | Elovich Equation | Pseudo-First-Order | Pseudo-Second-Order | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| a | b | R2 | K1 1/h | Qe mg/kg | R2 | K2 kg/(mg·h) | Qe mg/kg | R2 | |||
| EE2 | S1 | 2.49 | 1.16 | 0.334 | 0.887 | 0.289 | 2.29 | 0.928 | 0.504 | 2.52 | 0.993 |
| S2 | 2.26 | 1.00 | 0.308 | 0.960 | 0.298 | 2.02 | 0.874 | 0.416 | 2.34 | 0.995 | |
| S3 | 2.33 | 0.77 | 0.377 | 0.985 | 0.194 | 2.10 | 0.906 | 0.306 | 2.40 | 0.988 | |
| S4 | 1.46 | 0.72 | 0.183 | 0.960 | 0.334 | 1.32 | 0.931 | 0.891 | 1.48 | 0.997 | |
| E2 | S1 | 4.70 | 3.37 | 0.369 | 0.804 | 0.602 | 4.53 | 0.981 | 0.968 | 4.74 | 0.998 |
| S2 | 4.42 | 3.10 | 0.383 | 0.628 | 0.491 | 4.36 | 0.979 | 1.487 | 4.45 | 0.992 | |
| S3 | 4.04 | 2.84 | 0.342 | 0.746 | 0.535 | 3.94 | 0.980 | 1.274 | 4.08 | 0.997 | |
| S4 | 3.79 | 2.38 | 0.402 | 0.726 | 0.397 | 3.72 | 0.968 | 1.085 | 3.85 | 0.994 | |
| E1 | S1 | 4.30 | 2.70 | 0.435 | 0.675 | 0.451 | 4.12 | 0.989 | 0.606 | 4.34 | 0.994 |
| S2 | 4.40 | 3.34 | 0.249 | 0.960 | 0.972 | 4.07 | 0.973 | 0.545 | 4.43 | 0.994 | |
| S3 | 2.66 | 2.14 | 0.123 | 0.965 | 1.031 | 2.51 | 0.985 | 1.250 | 2.67 | 0.997 | |
| S4 | 3.60 | 2.92 | 0.162 | 0.900 | 1.172 | 3.39 | 0.978 | 0.904 | 3.63 | 0.993 | |
| BPA | S1 | 1.50 | 1.03 | 0.127 | 0.689 | 0.537 | 1.43 | 0.992 | 2.008 | 1.51 | 0.998 |
| S2 | 0.90 | 0.76 | 0.035 | 0.819 | 0.997 | 0.87 | 0.996 | 6.975 | 0.90 | 0.998 | |
| S3 | 0.30 | 0.15 | 0.037 | 0.915 | 0.321 | 0.27 | 0.938 | 4.292 | 0.30 | 0.998 | |
| S4 | 0.31 | 0.18 | 0.036 | 0.659 | 0.381 | 0.30 | 0.995 | 9.602 | 0.31 | 0.998 | |
| Sample | EDCs | Linear Model | Langmuir Model | Freundlich Model | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Kd L/kg | R2 | KL L/kg | Qm mg/kg | R2 | logKF | nads | R2 | ΔG KJ/mol | ||
| S1 | EE2 | 23.60 | 0.975 | 0.545 | 95.30 | 0.994 | 1.508 | 0.714 | 0.996 | −8.603 |
| E2 | 23.71 | 0.999 | 0.120 | 217.4 | 0.995 | 1.374 | 0.976 | 0.997 | −7.838 | |
| E1 | 11.47 | 0.936 | 0.484 | 61.75 | 0.980 | 1.269 | 0.696 | 0.984 | −7.239 | |
| BPA | 3.54 | 0.979 | 0.036 | 215.2 | 0.991 | 1.061 | 0.672 | 0.998 | −6.054 | |
| S2 | EE2 | 19.45 | 0.993 | 0.224 | 136.6 | 0.994 | 1.391 | 0.774 | 0.998 | −7.938 |
| E2 | 19.46 | 0.999 | 0.269 | 99.01 | 0.989 | 1.322 | 0.906 | 0.996 | −7.542 | |
| E1 | 10.56 | 0.960 | 0.380 | 63.94 | 0.986 | 1.222 | 0.696 | 0.992 | −6.971 | |
| BPA | 2.70 | 0.965 | 0.047 | 150.6 | 0.977 | 0.980 | 0.672 | 0.993 | −5.603 | |
| S3 | EE2 | 9.67 | 0.965 | 0.438 | 56.50 | 0.993 | 1.213 | 0.661 | 0.997 | −6.910 |
| E2 | 10.28 | 0.995 | 0.079 | 151.5 | 0.998 | 1.036 | 0.953 | 0.999 | −5.910 | |
| E1 | 3.70 | 0.953 | 0.525 | 25.51 | 0.990 | 0.919 | 0.574 | 0.995 | −5.243 | |
| BPA | 1.43 | 0.954 | 0.053 | 80.90 | 0.985 | 0.811 | 0.603 | 0.995 | −4.627 | |
| S4 | EE2 | 7.09 | 0.970 | 0.358 | 48.11 | 0.989 | 1.083 | 0.678 | 0.996 | −6.178 |
| E2 | 7.32 | 0.996 | 0.131 | 74.62 | 0.996 | 0.922 | 0.903 | 0.997 | −5.262 | |
| E1 | 3.22 | 0.963 | 0.451 | 23.49 | 0.971 | 0.846 | 0.588 | 0.993 | −4.826 | |
| BPA | 0.98 | 0.961 | 0.040 | 62.71 | 0.983 | 0.594 | 0.632 | 0.994 | −3.389 | |
| EDCs | Soil Samples | logKF | ndes | R2 | HI |
|---|---|---|---|---|---|
| EE2 | S1 | 1.686 | 0.468 | 0.978 | 0.655 |
| S2 | 1.616 | 0.446 | 0.995 | 0.576 | |
| S3 | 1.609 | 0.448 | 0.979 | 0.677 | |
| S4 | 1.083 | 0.678 | 0.996 | 0.682 | |
| E2 | S1 | 1.818 | 0.636 | 0.998 | 0.652 |
| S2 | 1.646 | 0.612 | 0.998 | 0.676 | |
| S3 | 1.568 | 0.822 | 0.997 | 0.862 | |
| S4 | 1.415 | 0.775 | 0.994 | 0.858 | |
| E1 | S1 | 1.403 | 0.475 | 0.996 | 0.683 |
| S2 | 1.276 | 0.470 | 0.934 | 0.676 | |
| S3 | 0.771 | 0.450 | 0.998 | 0.784 | |
| S4 | 0.740 | 0.482 | 0.993 | 0.819 | |
| BPA | S1 | 1.204 | 0.569 | 0.997 | 0.846 |
| S2 | 1.143 | 0.573 | 0.976 | 0.853 | |
| S3 | 1.148 | 0.572 | 0.997 | 0.948 | |
| S4 | 1.025 | 0.620 | 0.990 | 0.981 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hu, Y.; Yang, Q.; Sun, J.; Chen, Q.; Fan, J.; Mei, X. Adsorption and Desorption Behaviors of Four Endocrine Disrupting Chemicals in Soils from the Water-Level Fluctuation Zone of the Three Gorges Reservoir, China. Sustainability 2018, 10, 2531. https://doi.org/10.3390/su10072531
Hu Y, Yang Q, Sun J, Chen Q, Fan J, Mei X. Adsorption and Desorption Behaviors of Four Endocrine Disrupting Chemicals in Soils from the Water-Level Fluctuation Zone of the Three Gorges Reservoir, China. Sustainability. 2018; 10(7):2531. https://doi.org/10.3390/su10072531
Chicago/Turabian StyleHu, Ying, Qingwei Yang, Jiaoxia Sun, Qingkong Chen, Jianxin Fan, and Xiaoxing Mei. 2018. "Adsorption and Desorption Behaviors of Four Endocrine Disrupting Chemicals in Soils from the Water-Level Fluctuation Zone of the Three Gorges Reservoir, China" Sustainability 10, no. 7: 2531. https://doi.org/10.3390/su10072531
APA StyleHu, Y., Yang, Q., Sun, J., Chen, Q., Fan, J., & Mei, X. (2018). Adsorption and Desorption Behaviors of Four Endocrine Disrupting Chemicals in Soils from the Water-Level Fluctuation Zone of the Three Gorges Reservoir, China. Sustainability, 10(7), 2531. https://doi.org/10.3390/su10072531





