Hydrochemical Characteristics and Groundwater Quality Assessment in the Diluvial Fan of Gaoqiao, Emei Mountain, China
Abstract
1. Introduction
2. Materials and Methods
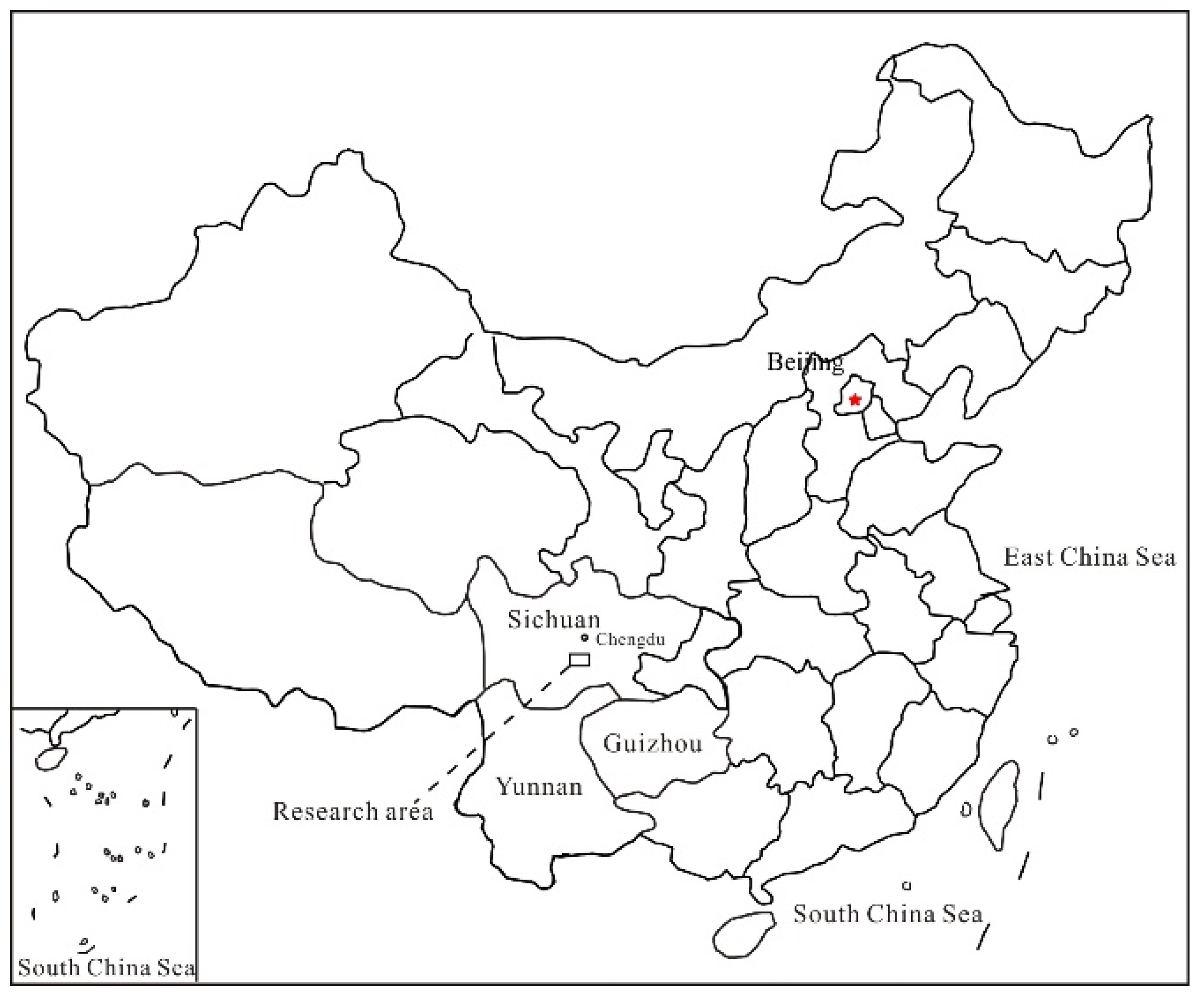
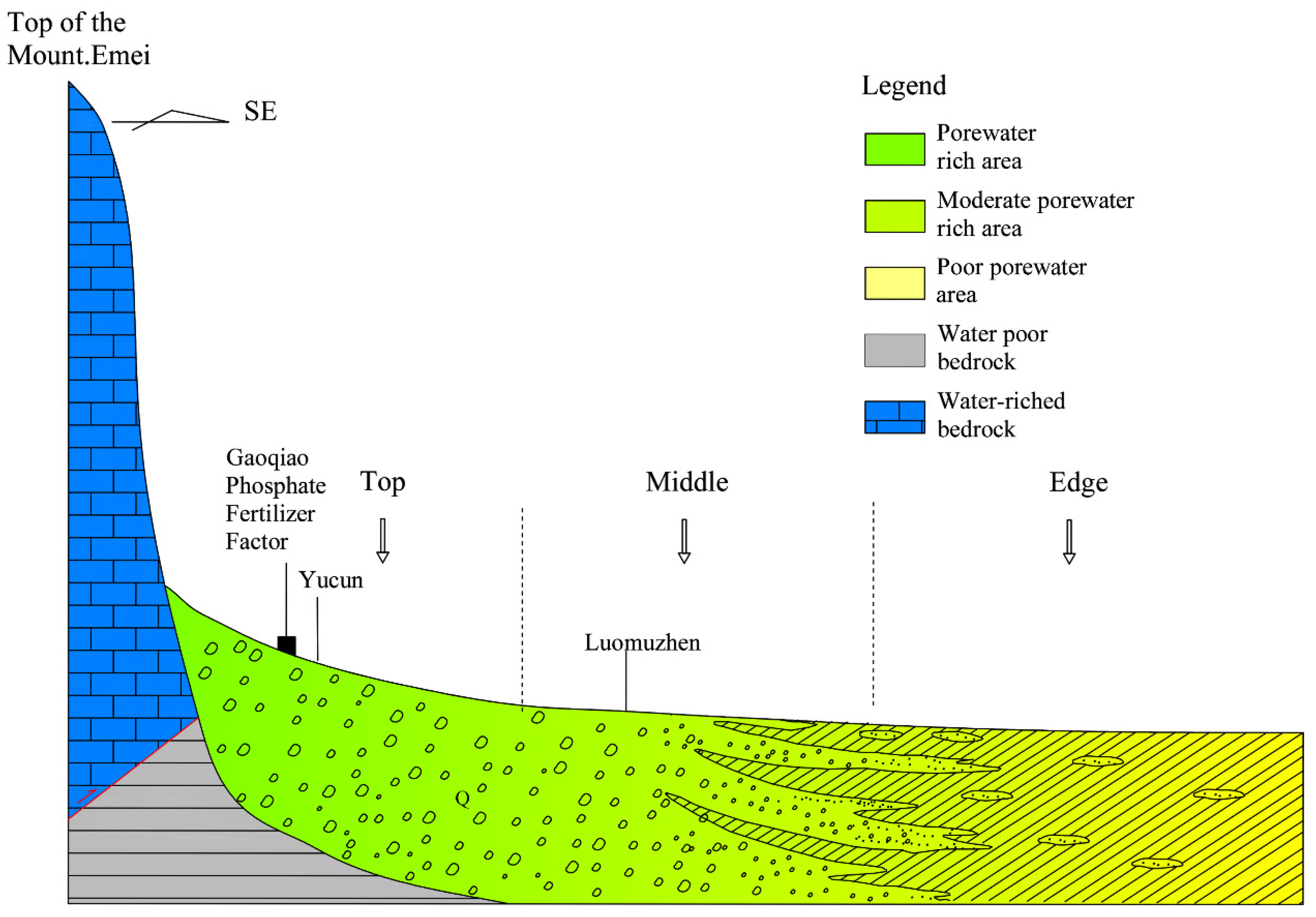
2.1. Study Area
2.2. Sample Collection and Analysis
2.3. The Principle of Five-Element Connection Number SPA in Groundwater Assessment
3. Result and Discussion
3.1. Hydrochemical Characteristics of Groundwater
3.1.1. Chemical Components of Groundwater
3.1.2. The Hydrochemical Characteristics of Groundwater
3.1.3. Total Dissolved Solid Characteristics
3.1.4. Total Hardness (TH) Characteristics
3.1.5. Characteristics of Pollutant Fluorine in Groundwater
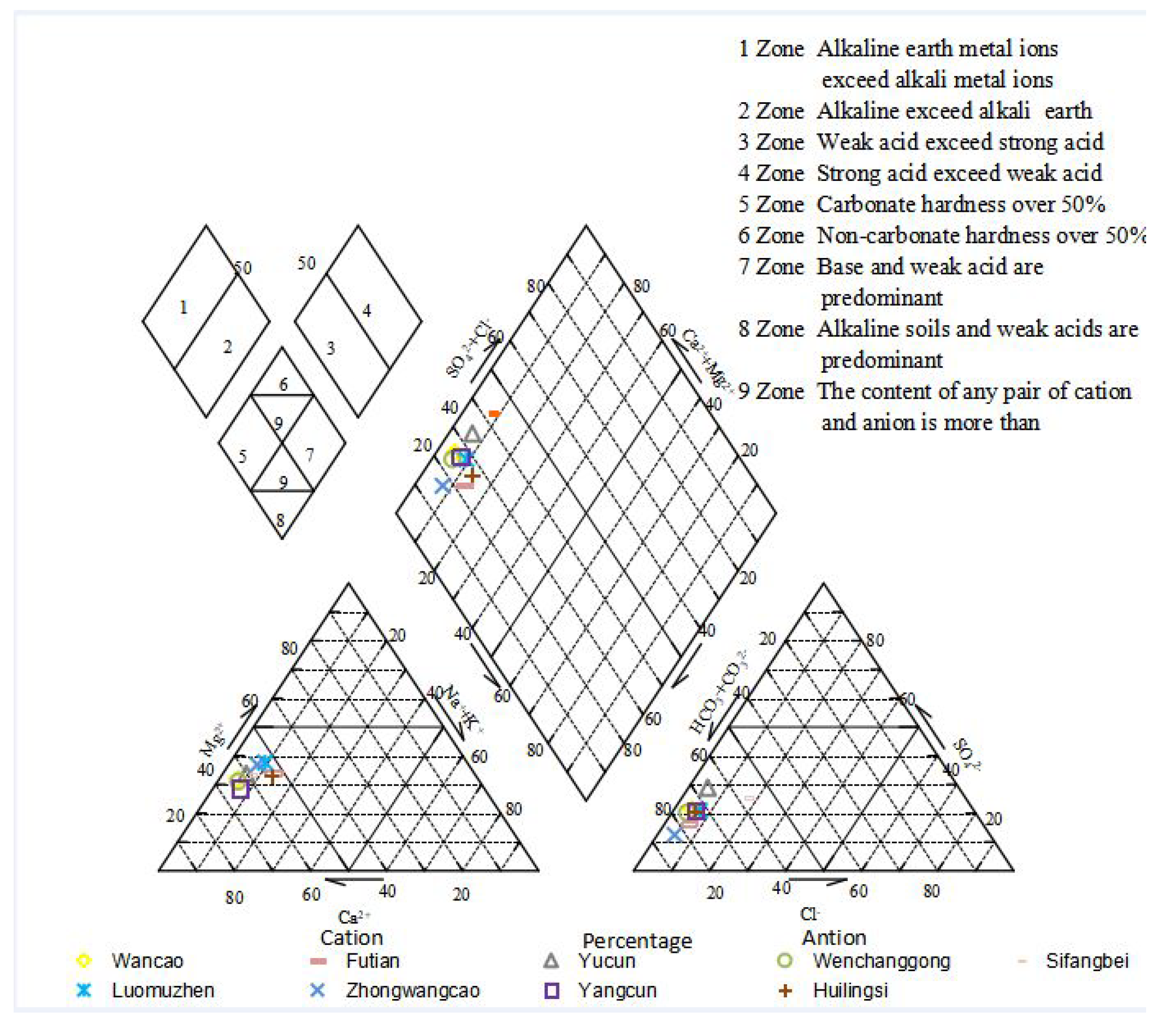
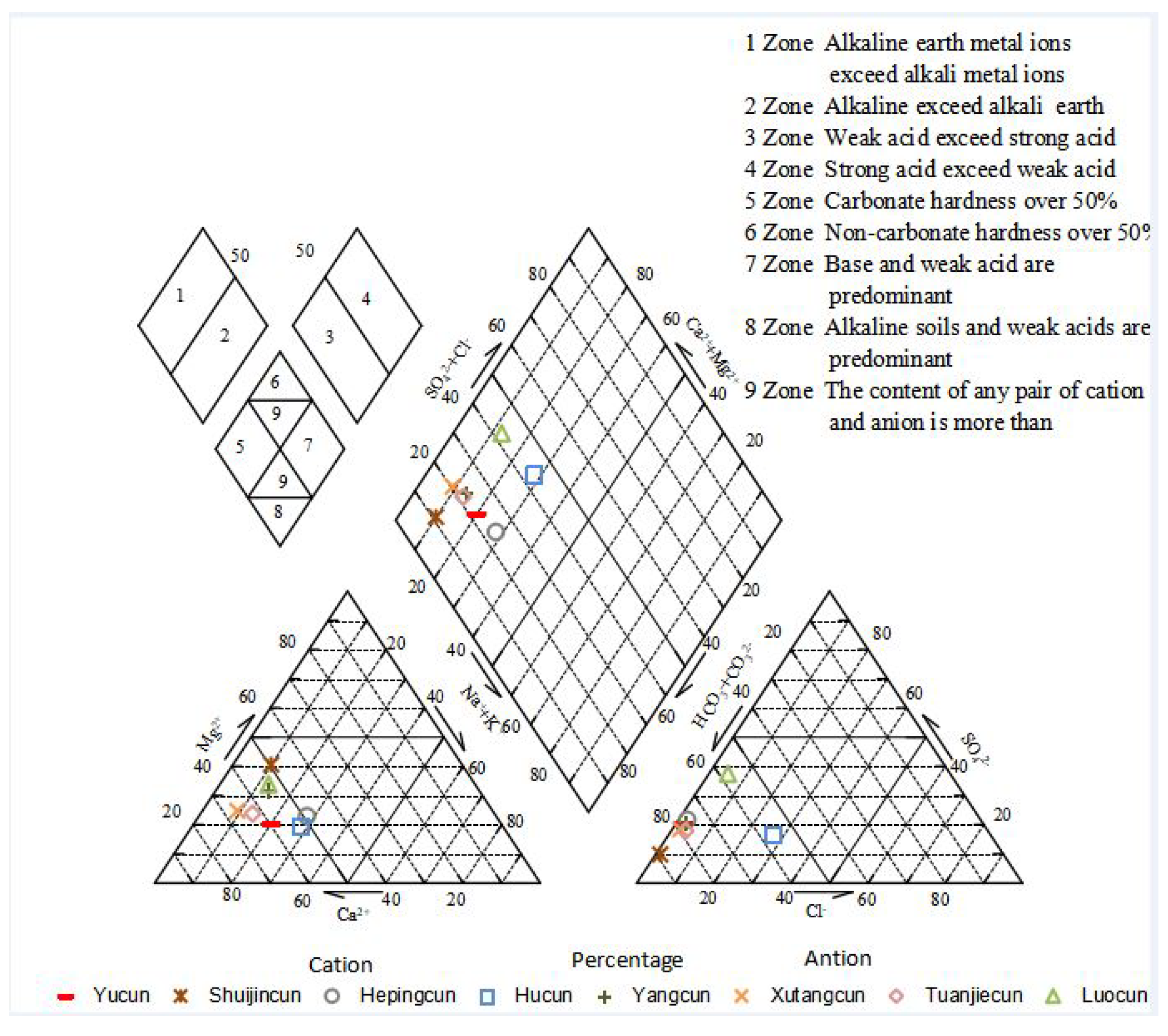
3.1.6. Characteristics of a Schoeller Diagram
3.2. Groundwater Quality Assessment Results
| μTH = 0.1 + 0.9i + 0j + 0k + 0l |
| μTDS = 0.68 + 0.32i + 0j + 0k + 0 l |
| μSO42− = 0.809 + 0.191i + 0j + 0k + 0l |
| μCl− = 1 + 0i + 0j + 0k + 0l |
| μNO3− = 0 + 0i + 0.7j + 0.3k + 0l |
| μF = 0 + 0i + 0j + 0k + 1l |
| μNH4+ = 1 + 0i + 0j + 0k + 0l |
| μFe = 1 + 0i + 0j + 0k + 0l |
| μ1 = 0.5736 + 0.1764i + 0.0875j + 0.0375k + 0.1250l |
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Subba, R.N. Factors controlling the salinity in groundwater in parts of Guntur district, Andhra Pradesh, India. Environ. Monit. Assess. 2008, 138, 327–341. [Google Scholar] [CrossRef] [PubMed]
- Sarikhani, R.; Dehnavi, A.G.; Ahmadnejad, Z.; Kalantari, N. Hydrochemical characteristics and groundwater quality assessment in Bushehr Province, SW Iran. Environ. Earth Sci. 2015, 74, 6265–6281. [Google Scholar] [CrossRef]
- Zhang, W.G.; Yang, Q.C. Effects of fluorine pollution treatment on agricultural ecological environment in Gaoqiao phosphate fertilizer plant before and after treatment. Sichuan Environ. 1986, 2, 3–9. [Google Scholar]
- Wang, W.; Jin, J.; Ding, J.; Li, Y. A new approach to water resources system assessment—Set pair analysis method. Sci. China Ser. E Technol. Sci. 2009, 52, 3017–3023. [Google Scholar] [CrossRef]
- Yao, L.L.; Gao, T.; Hu, L.-T. Application study of groundwater pollution early warning in source field: A case study in alluvial-pluvial fan of Hun River. South-to-North Water Transf. Water Sci. Technol. 2016, 1, 37–41. [Google Scholar]
- Zhu, H.; Ren, X.; Liu, Z. A new four-step hierarchy method for combined assessment of groundwater quality and pollution. Environm. Monit. Assess. 2018, 190, 50. [Google Scholar] [CrossRef] [PubMed]
- Serio, F.; Miglietta, P.P.; Lamastra, L.; Ficocelli, S.; Intini, F.; De Leo, F.; De Donno, A. Groundwater nitrate contamination and agricultural land use: A grey water footprint perspective in Southern Apulia Region (Italy). Sci. Total. Environ. 2018, 645, 1425–1431. [Google Scholar] [CrossRef] [PubMed]
- Bagordo, F.; Migoni, D.; Grassi, T.; Serio, F.; Idolo, A.; Guido, M.; Zaccarelli, N.; Fanizzi, F.P.; De Donno, A. Using the DPSIR framework to identify factors influencing the quality of groundwater in Grecìa Salentina (Puglia, Italy). Rend. Lincei 2016, 27, 113–125. [Google Scholar] [CrossRef]
- Miglietta, P.P.; Toma, P.; Fanizzi, F.P.; De Donno, A.; Coluccia, B.; Migoni, D.; Bagordo, F.; Serio, F. A grey water footprint assessment of groundwater chemical pollution: Case study in Salento (southern Italy). Sustainability 2017, 9, 799. [Google Scholar] [CrossRef]
- Duan, L.; Wang, W.K.; Cao, Y.-Q.; Wang, L.-J.; Liu, B. Hydrochemical characteristics and formation of groundwater in the middle part of the northern of Tianshan Mountain. J. Arid. Land Resour. Environ. 2007, 21, 29–34. [Google Scholar]
- Sun, L.; Gui, H. Hydro-chemical evolution of groundwater and mixing between aquifers: A statistical approach based on major ions. Appl. Water Sci. 2015, 5, 97–104. [Google Scholar] [CrossRef]
- Saka, D.; Akiti, T.T.; Osae, S.; Appenteng, M.K.; Gibrilla, A. Hydrogeochemistry and isotope studies of groundwater in the Ga West Municipal Area, Ghana. Appl. Water Sci. 2013, 3, 577–588. [Google Scholar] [CrossRef]
- Wang, Y.; Jing, H.; Yu, L.; Su, H.; Luo, N. Set pair analysis for risk assessment of water inrush in karst tunnels. Bull. Eng. Geol. Environ. 2017, 76, 1199–1207. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, W.; Li, M.; Liu, X. Risk assessment of floor water inrush in coal mines based on secondary fuzzy comprehensive evaluation. Int. J. Rock Mech. Min. Sci. 2012, 52, 50–55. [Google Scholar] [CrossRef]
- Yue, W.; Cai, Y.; Rong, Q.; Li, C.; Ren, L. A hybrid life-cycle and fuzzy-set-pair analyses approach for comprehensively evaluating impacts of industrial wastewater under uncertainty. J. Clean. Prod. 2014, 80, 57–68. [Google Scholar] [CrossRef]
- Mehra, M.; Oinam, B.; Singh, C.K. Integrated Assessment of Groundwater for Agricultural Use in Mewat District of Haryana, India Using Geographical Information System (GIS). J. Indian Soc. Remote Sens. 2016, 44, 747–758. [Google Scholar] [CrossRef]
- Liu, C.W.; Lin, K.H.; Kuo, Y.M. Application of factor analysis in the assessment of groundwater quality in a blackfoot disease area in Taiwan. Sci. Total. Environ. 2003, 313, 77–89. [Google Scholar] [CrossRef]
- An, T.D.; Tsujimura, M.; Phu, V.L.; Kawachi, A.; Hab, D.T. Chemical Characteristics of Surface Water and Groundwater in Coastal Watershed, Mekong Delta, Vietnam. Procedia Environ. Sci. 2014, 20, 712–721. [Google Scholar] [CrossRef]
- Rajesh, R.; Brindha, K.; Elango, L. Groundwater Quality and its Hydrochemical Characteristics in a Shallow Weathered Rock Aquifer of Southern India. Water Qual. Expo. Health 2015, 7, 515–524. [Google Scholar] [CrossRef]
- Arumugam, K.; Elangovan, K. Hydrochemical characteristics and groundwater quality assessment in Tirupur Region, Coimbatore District, Tamil Nadu, India. Environ. Geol. 2009, 58, 1509–1520. [Google Scholar] [CrossRef]
- Huan, H.; Wang, J.S.; Zhai, Y.Z.; Zheng, J.-Q. Chemical Characteristics and Evolution of Groundwater in the Yongding River Alluvial Fan of Beijing Plain. Acta Geosci. Sin. 2011, 32, 357–366. [Google Scholar]
- Guo, G.X.; Hou, Q.L.; Xu, L.; Liu, J.-R.; Xin, B.-D. Delamination and Zoning Characteristics of Quaternary Groundwater in Chaobai Alluvial-proluvial Fan, Beijing, Based on Hydrochemical Analysis. Acta Geosci. Sin. 2014, 35, 204–210. [Google Scholar]
- Li, Y.; Zhang, Z.; Fei, Y.; Chen, H.; Qian, Y.; Dun, Y. Investigation of quality and pollution characteristics of groundwater in the Hutuo River Alluvial Plain, North China Plain. Environ. Earth Sci. 2016, 75, 581. [Google Scholar] [CrossRef]
- Wu, Y.; You, Y.W. Assessment on water environment quality in the eastern plain of Emei Mountain. Mounta Res. 1998, 1, 64–68. [Google Scholar]
- Wu, Y.; Qin, J.X.; Zhong, P.L. Study on shallow groundwater inorganic complex in the eastern plain of Emei Mountain. J. Geol. Hazards Environ. Preserv. 1996, 3, 56–63. [Google Scholar]
- Pan, Z.; Wang, Y.; Jin, J.; Liu, X. Set pair analysis method for coordination evaluation in water resources utilizing conflict. Phys. Chem. Earth Parts A B C 2017, 101, 149–156. [Google Scholar] [CrossRef]
- Chen, L.Y.; Fu, Q.; Wei, L.L. Application of Five-Element Connection Number to the Quality Assessment ofEutrophication in Lakes. Res. Environ. Sci. 2008, 21, 82–86. [Google Scholar]
- Yu, F.; Qu, J.; Li, Z.; Gao, Z. Application of set pair analysis based on the improved five-element connectivity in the evaluation of groundwater quality in XuChang, Henan Province, China. Water Sci. Technol. Water Supply 2017, 17, 632–642. [Google Scholar] [CrossRef]
- Li, P.Y.; Qian, H.; Wu, J.H. Application of Set Pair Analysis Method Based on Entropy Weight in Groundwater Quality Assessment—A Case Study in Dongsheng City, Northwest China. J. Chem. 2014, 8, 851–858. [Google Scholar]
- Wang, X.; Wang, W.S.; Ding, J. Set pair analysis and its application to hydrology and water resources. Adv. Sci. Tech. Water Resour. 2006, 26, 9–11. [Google Scholar]
- Wang, D.; Zhu, Y.S.; Zhao, K.Q. Research and application of model based on set pair analysis and fuzzy set theory for assessment of water eutrophication. J. Hydrol. 2004, 24, 9–13. [Google Scholar]
- Standard for groundwater quality (GB/T 14848-2017). Available online: http://www.dixiashui.cn/fagui/guojiabiaozhun/20171102/5462.html (accessed on 29 November 2018).

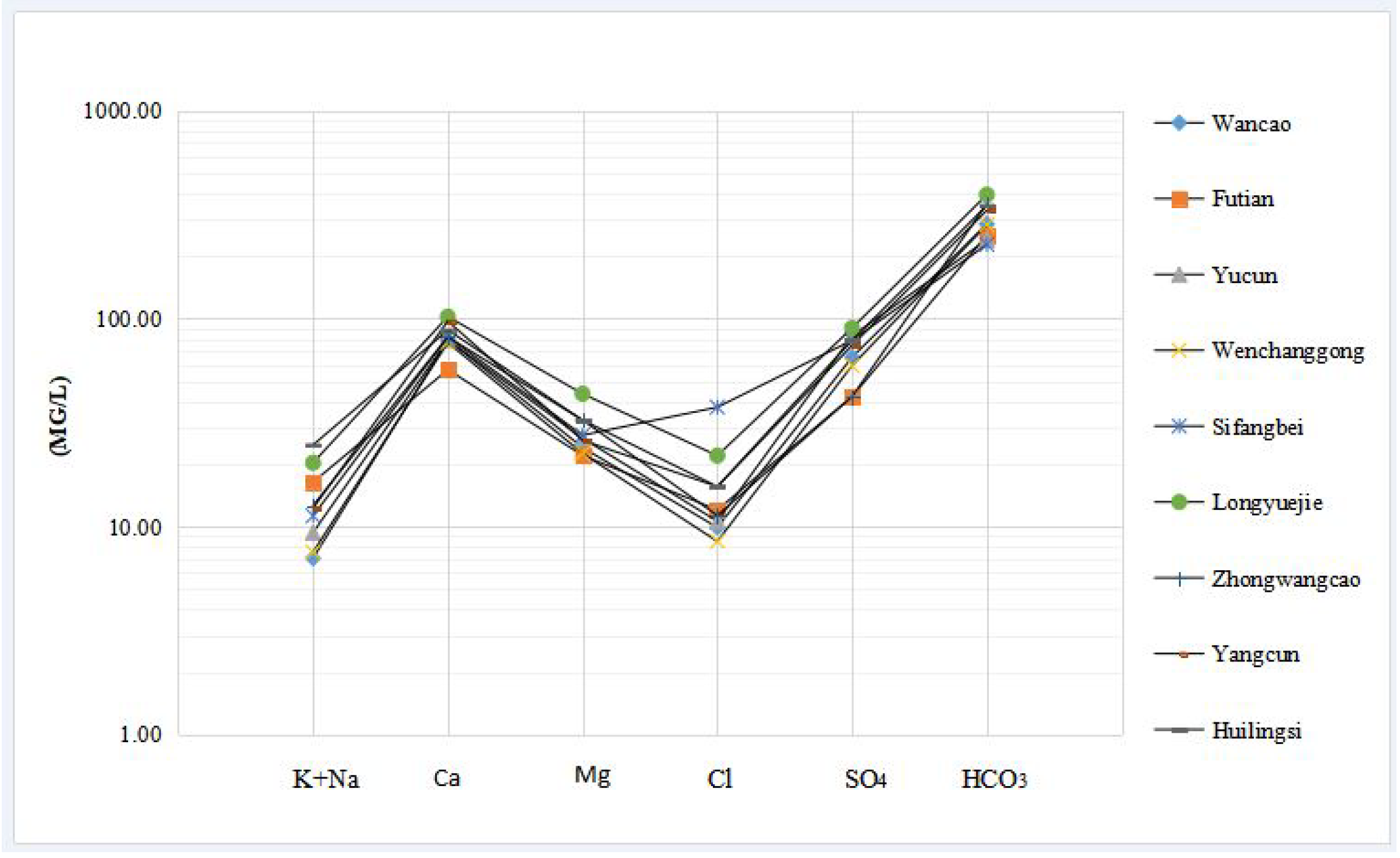
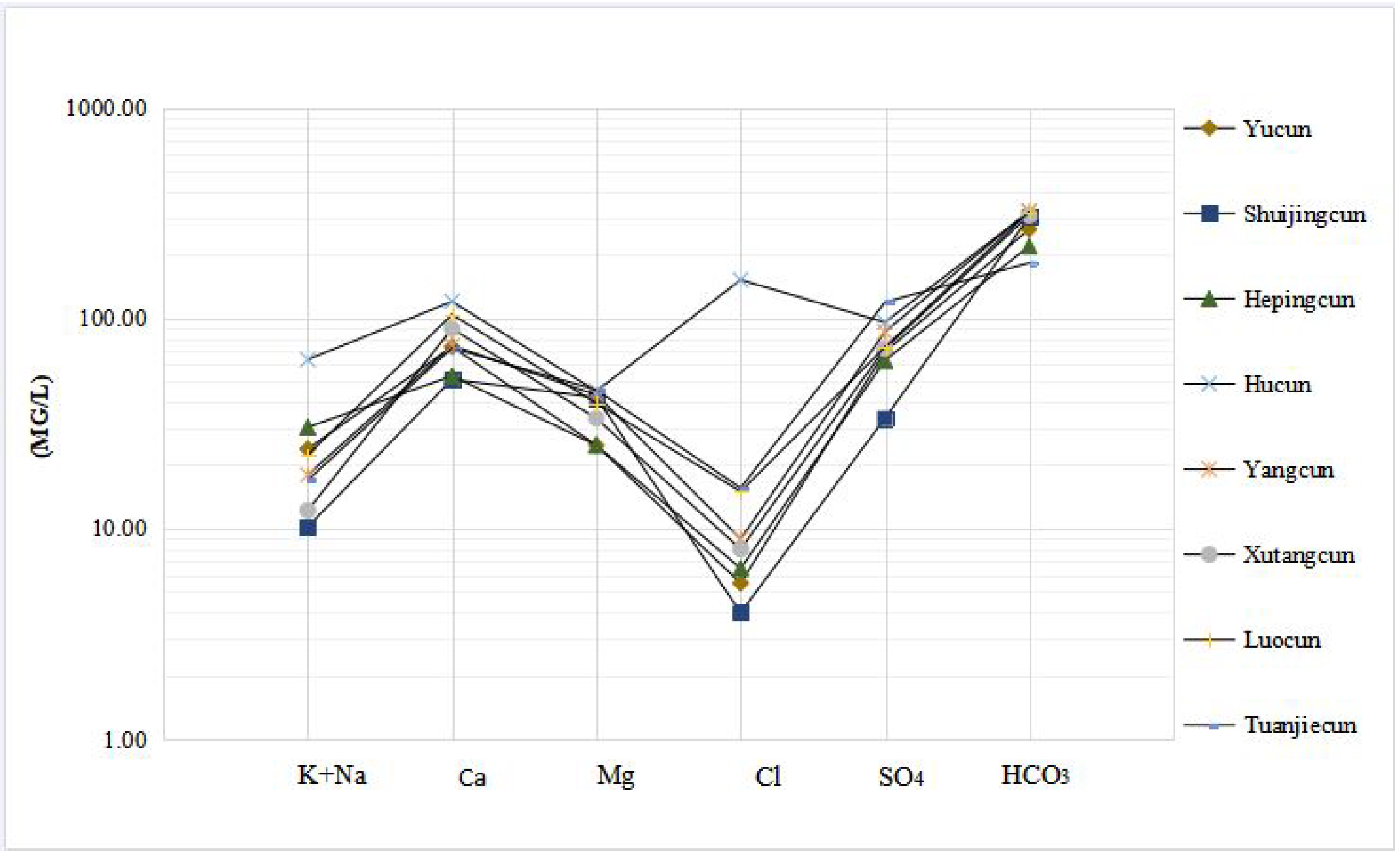
| Sampling Time | Site | Geographic Location | Characteristics of Samples |
|---|---|---|---|
| 1995 | Wancao | Top | Spring |
| Futian | Spring | ||
| Yucun | Well | ||
| Wenchanggong | Spring | ||
| Sifangbei | Well | ||
| Luouzhen | Middle | Well | |
| Zhongwangcao | Well | ||
| Yangcun | Well | ||
| Huilingsi | Edge | Well | |
| 2016 | Yucun | Top | Spring |
| Shuijincun | Well | ||
| Hepingcun | Well | ||
| Hucun | Middle | Well | |
| Yangcun | Well | ||
| Xutangcun | Edge | Spring | |
| Tuanjiecun | Well | ||
| Luocun | Well |
| Indexes | I | II | III | IV | V |
|---|---|---|---|---|---|
| TH | ≤150 | ≤300 | ≤450 | ≤650 | >650 |
| TDS | ≤300 | ≤500 | ≤1000 | ≤2000 | >2000 |
| SO42− | ≤50 | ≤150 | ≤250 | ≤350 | 350 |
| Cl− | ≤50 | ≤150 | ≤250 | ≤350 | >350 |
| NO3− | ≤2 | ≤5 | ≤20 | ≤30 | >30 |
| F | ≤1 | ≤1 | ≤1 | ≤2 | >2 |
| NH4+ | ≤0.02 | ≤0.1 | ≤0.5 | ≤1.5 | >1.5 |
| Fe | ≤0.1 | ≤0.2 | ≤0.3 | ≤2 | >2 |
| Position | Time | site | K+ + Na+ | Ca2+ | Mg2+ | Cl− | SO42− | HCO3− | CO32− | NO3− | TH | TDS | F | NH4+ | Fe |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Top | 1995 | Wancao | 7.06 | 79.26 | 24.05 | 9.93 | 65.32 | 285.4 | 0.00 | / | 296.90 | 328.74 | 0.10 | / | / |
| Futian | 16.38 | 56.97 | 22.05 | 12.10 | 42.27 | 252.18 | 0.00 | / | 233.00 | 276.55 | 0.50 | / | / | ||
| Yucun | 9.38 | 79.10 | 26.65 | 10.60 | 82.61 | 239.88 | 0.00 | / | 307.20 | 329.38 | 1.00 | / | / | ||
| Wenchanggong | 7.55 | 76.78 | 22.54 | 8.51 | 59.56 | 282.94 | 0.00 | / | 269.50 | 318.51 | 1.10 | / | / | ||
| 2016 | Yucun | 23.9 | 73.10 | 24.90 | 5.50 | 69.10 | 265.00 | 0.00 | 23.00 | 285.00 | 364.00 | 2.86 | <0.02 | <0.05 | |
| Shuijincun | 10.20 | 51.10 | 41.90 | 3.98 | 33.40 | 302.00 | 0.00 | 28.60 | 300.00 | 330.00 | 0.95 | 0.05 | <0.05 | ||
| Hepingcun | 30.30 | 53.10 | 24.90 | 6.47 | 62.90 | 219.00 | 0.00 | 32.30 | 235.00 | 332.00 | 0.34 | <0.02 | <0.05 | ||
| Middle | 1995 | Sifangbei | 11.29 | 81.74 | 27.55 | 37.6 | 42.27 | 227.58 | 0.00 | / | 317.50 | 351.54 | 0.22 | / | / |
| Luomuzhen | 20.31 | 102.40 | 43.58 | 22.00 | 90.27 | 394.88 | 0.00 | / | 435.00 | 478.10 | 0.34 | / | / | ||
| Zhongwangcao | 12.67 | 81.74 | 32.56 | 11.30 | 42.27 | 356.75 | 0.00 | / | 338.10 | 359.78 | 0.70 | / | / | ||
| Yangcun | 12.28 | 97.43 | 26.05 | 15.60 | 74.54 | 332.14 | 0.00 | / | 350.50 | 392.59 | 0.22 | / | / | ||
| 2016 | Hucun | 63.60 | 120.00 | 45.00 | 152.00 | 95.60 | 320.00 | 0.00 | 67.40 | 485.00 | 716.00 | 0.29 | <0.02 | <0.05 | |
| Yangcun | 18.00 | 74.10 | 43.20 | 8.94 | 86.10 | 323.00 | 0.00 | 32.70 | 363.00 | 435.00 | 0.31 | <0.02 | <0.05 | ||
| Edge | 1995 | Huilingsi | 24.75 | 88.34 | 32.56 | 15.60 | 78.77 | 350.60 | 0.00 | / | 354.60 | 416.22 | 0.50 | / | / |
| 2016 | Xutangcun | 12.20 | 89.10 | 33.44 | 7.99 | 71.70 | 308.00 | 0.00 | 59.60 | 360.00 | 438.00 | 0.30 | <0.02 | <0.05 | |
| Tuanjiecun | 17.00 | 72.10 | 45.60 | 15.60 | 119.00 | 183.00 | 0.00 | 124.00 | 368.00 | 494.00 | 0.24 | <0.02 | <0.05 | ||
| Luocun | 22.40 | 104.00 | 39.50 | 15.00 | 72.80 | 320.00 | 0.00 | 112.00 | 423 | 537 | 0.33 | <0.02 | <0.05 |
| TH (mg/L) | Groundwater Soft Hard Type | Samples | |
|---|---|---|---|
| In 1995 | In 2016 | ||
| 0~75 | Very soft water | 0 | 0 |
| 75~150 | Soft water | 0 | 0 |
| 150~300 | Fairly hard water | 3 | 3 |
| 300~450 | Hard water | 6 | 4 |
| >450 | Very hard water | 0 | 1 |
| Sites | a | b | c | d | e | Gm | IDC State | Set Pair Situation |
|---|---|---|---|---|---|---|---|---|
| Yucun | 0.5736 | 0.1764 | 0.0875 | 0.0375 | 0.1250 | III | a > e, a > b, b > c, c > d, d<e | identity state power (3) |
| Shuijingcun | 0.6844 | 0.1438 | 0.0175 | 0.1075 | 0.0000 | II | a > e, a > b, b > c, c<d, d > e. e = 0 | identity state power (7) |
| Hepingcun | 0.7680 | 0.1070 | 0.0000 | 0.0000 | 0.1250 | II | a > e, a > b, b > c, c<d, d > e | identity state power (6) |
| Hucun | 0.4430 | 0.2505 | 0.1596 | 0.0219 | 0.1250 | III | a > e, a<b, b > c, c > d, d<e | identity state power (49) |
| Yangcun | 0.6418 | 0.1808 | 0.0525 | 0.0000 | 0.1250 | II | a > e, a > b, b > c, c > d, d<e | identity state power (3) |
| Xutangcun | 0.6366 | 0.1884 | 0.0500 | 0.0000 | 0.1250 | II | a > e, a > b, b > c, c > d, d<e | identity state power (3) |
| Tuanjicun | 0.5965 | 0.1593 | 0.1193 | 0.0000 | 0.1250 | II | a > e, a > b, b > c, c > d, d<e | identity state power (3) |
| Luocun | 0.5425 | 0.2758 | 0.0567 | 0.0000 | 0.1250 | III | a > e, a > b, b > c, c > d, d<e | identity state power (3) |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Y.; Xu, W.; Chen, J.; Chen, Q.; Pan, Z. Hydrochemical Characteristics and Groundwater Quality Assessment in the Diluvial Fan of Gaoqiao, Emei Mountain, China. Sustainability 2018, 10, 4507. https://doi.org/10.3390/su10124507
Yang Y, Xu W, Chen J, Chen Q, Pan Z. Hydrochemical Characteristics and Groundwater Quality Assessment in the Diluvial Fan of Gaoqiao, Emei Mountain, China. Sustainability. 2018; 10(12):4507. https://doi.org/10.3390/su10124507
Chicago/Turabian StyleYang, Yanna, Wenlai Xu, Jinyao Chen, Qiang Chen, and Zhicheng Pan. 2018. "Hydrochemical Characteristics and Groundwater Quality Assessment in the Diluvial Fan of Gaoqiao, Emei Mountain, China" Sustainability 10, no. 12: 4507. https://doi.org/10.3390/su10124507
APA StyleYang, Y., Xu, W., Chen, J., Chen, Q., & Pan, Z. (2018). Hydrochemical Characteristics and Groundwater Quality Assessment in the Diluvial Fan of Gaoqiao, Emei Mountain, China. Sustainability, 10(12), 4507. https://doi.org/10.3390/su10124507




