1. Introduction
Maize (
Zea mays L.) is ranked among the top three important staple food crops around the globe, and South Africa comes ninth in maize production globally and second in Sub-Saharan Africa [
1]. Due to the dependence of agricultural production on climatic factors, crop yield (including maize yield) has been noted to be under threat because of climate change [
2]. Specifically, extreme weather events such as drought and flooding can adversely affect the stability of crop yields [
3]. The prevalence of extreme drought and flooding is posing a threat to the food security of Sub-Saharan African countries and in particular Southern Africa [
4,
5]. Consequently, there have been calls nationally and internationally for actions to investigate the level of resilience of existing crop cultivars to multiple abiotic stress conditions.
At present, South Africa is battling one of the worst droughts ever recorded that started in 2015. Records from the South African Weather Service show that year 2015 was the driest on record in South Africa, dating back to 1904 [
6]. Regarding flooding, most of the regions in South Africa experienced this phenomenon in year 2011, which led to crop and infrastructural loses on a number of farms [
7]. Climate projections in South Africa are indicating that under all four medium and long term climatic scenarios, a higher frequency of flooding and drought will occur [
8].
Severe droughts and excessive moisture cause considerable maize yield losses worldwide [
9]. The extent of damage on maize due to drought depends on the intensity and duration of stress, but also on the plant developmental stage at the time of exposure [
10,
11,
12,
13]. For instance, Atteya [
10] reported yield losses ranging from 32 to 92% when maize was exposed to drought during the vegetative stage. Kamara [
11] reported losses of 63–87% when the crop was exposed to drought during the reproductive stage. In a report by Monneveux [
13], losses of around 79–81% were observed when maize was exposed to drought during the grain-filling period.
Pertaining to the effects of excess moisture on plants, a number of studies have been carried out at molecular, biochemical, physiological, anatomical, and morphological levels [
14,
15]. The extent of damage just like drought is also dependent on the stage of development, the type of cultivar and the duration of waterlogging [
16,
17]. Maize is especially susceptible to excess moisture from the early seedling growth to time of tasselling [
17,
18,
19].
Tolerance to excess soil moisture has been noted in some maize genotypes having an inherent ability to produce adventitious roots and properties in root morphological adaptation like air space (aerenchyma) formation in cortical regions of adventitious roots [
18]. A deeper understanding of how different cultivars respond to extreme weather events is quite crucial as it helps in advancing knowledge in support of crop modelling and breeding activities [
20]. Grzesiak [
16], in a study similar to the present one focusing on the morphological and anatomical root traits, concluded that genotypes that could tolerate drought were also able to tolerate excess moisture. However, some genotypes might possess one of the two characteristics being either drought tolerant or tolerant to excess moisture [
9]. To date, there is a paucity of information that ascertains the agronomic and physiological performance of drought resistant genotypes under excess moisture conditions. The study by Grzesiak [
16] focused on two cultivars of different drought susceptibility though they did not investigate how growth, yield, and physiological responses were affected. It is not always the case that the negative effects on roots will translate to yield changes [
21]. Furthermore, the experiment resembled a pot experiment and was performed in a growth chamber. Ren et al. [
19], reported that roots of plants grown in pots have restricted growth, which thus buffers the natural responses of plant growth. We conclude that pot experiments may not be truly representative of what really emerges under natural conditions.
The objective of our study is to clarify growth, development, yield, yield components, and physiological responses related to the performance of alternative maize cultivars with different drought tolerance under field drought and flooding conditions imposed at different developmental stages, with a view to ascertain if maize exhibits environmental tolerance to both drought and flooding.
2. Materials and Methods
2.1. Study Site
Field experiments were conducted at University of Pretoria Hatfield Experimental Farm, Pretoria, South Africa (25°45′ S, 28°16′ E and elevation 1327 m.a.s.l.). The soil is classified as a Hutton sandy clay loam [
22] (loamy, kaolinitic, mesic, Typic Eutrustox) (
Table 1). The long-term mean annual rainfall of the study site is 670 mm, mostly concentrated during the months of October–March. Biweekly temperatures and further parameters of the study site are presented in
Table 2.
2.2. Field Trial and Treatments
Two commercial maize cultivars, PAN 413 (drought tolerant cultivar) and PAN 6Q-245 (drought intolerant cultivar, high yielder), were selected for this study (
http://www.pannar.com/agronomy).
Two seeds were planted at a depth of 0.05 m in rows using manual planters on 15 November (2015/2016) and 22 October (2016/2017). The planting density was 59,523 plants ha−1. Plots consisted of 4 rows, spaced 0.56 m apart and an intra-row spacing of 0.30 m. The rows were 5 m in length, with 1 border row on either side. Two weeks after emergence, excess plants were thinned to the desired plant density. Before planting, the field was ploughed and disked to create a level seedbed. Weed control was performed by hand. No pests or diseases occurred during either season. An automated rainout shelter was used to cover the plots for possible precipitation events. A shelter, activated by a rain sensor, moved to cover the crop within two minutes in case of rainfall (≥2 mm). Weeding was done manually when needed. At harvest, a 4 m length of the two middle rows was used for yield determination.
The maize growth period was divided into three growth stages according to the standardized growth scale Biologische Bundesanstalt, Bundessortenamt and Chemical industry (BBCH) identification keys as follows: early vegetative (GS18), mid-vegetative to tasselling (GS51), and grain filling (GS71). The seedlings grew vigorously until the first drought and flooding treatments were imposed during the vegetative stage, which commenced immediately after the unfolding of the 8th leaf.
Each cultivar was exposed to three drought-timing and three flood-timing treatments, compared to a control (C) with well-watered plants irrigated to field capacity throughout the growing season. The drought-timing treatments included water withheld during: early-vegetative stage (DV), mid-vegetative to tasselling stage (DT), and grain-filling stage (DGf). Similarly, water was applied to the flooded plots during three stages: early-vegetative stage (EmV), mid-vegetative to tasselling stage (EmT), and grain-filling stage (EmGf). A summary of the treatments is presented in
Table 3. The treatments were laid out as a randomized complete block design (RCBD) with three replications.
A drip irrigation system was used to apply water in the treatments receiving water according to crop water requirements. Crop water use was estimated from soil water content measurements conducted on a weekly basis to a depth of 1 m using a site-calibrated neutron soil water meter (Model 503 DR CPN Hydro probe; Campbell Pacific Nuclear, Concord, CA, USA). Seven drip lines with a delivery rate of 2.1 L h−1 (at a working pressure range of 120–180 kPa) were placed laterally in each irrigated plot at a distance of 0.28 m. The drip emitters were pressure compensated and were located every 0.3 m within a dripper line. During the crop establishment period (between seed germination and crop establishment), 10 mm irrigation was applied every third day. When stress was imposed to the drought-timing treatments, water was completely withheld for those specific experimental units for 21 consecutive days.
Plots exposed to flooding were zero levelled to allow even distribution of water in each plot. For flood-timing treatments, the drippers were removed, and water could flow freely to the plots, leading to ponding on the soil surface. The flooded plots were left flooded for a period of 8 consecutive days with a free water height maintained at 10 ± 2.5 cm above the ground surface. An outlet was provided in each flood experimental plots to drain excess water (above 12.5 cm high) from the plots. All plots received water according to the requirements before and after the drought and flood treatments.
2.3. Agronomic Data
Plant height was measured on eight randomly selected and marked plants on the middle rows of each plot. Measurements were taken from the ground surface to the ligule of the fully developed leaf. The measurements were taken weekly from the fourth week after emergence up to flowering and after that it was done fortnightly up to physiological maturity. Leaf area index (LAI) was calculated after measuring leaf area with an LI 3100 belt-driven leaf area meter (LI-COR, Lincoln, NE, USA). Due to the limited number of plants in each plot, only two plants per plot were used for destructive sampling. Three sampling campaigns were conducted per growing season, excluding the final harvest. The harvested plants were partitioned into stems, leaves (leaf blade), and cobs. The partitioned parts of the plant were oven dried at a temperature of 70 °C to a constant mass. At crop physiological maturity, 10 plants per plot from the two center rows were harvested for dry matter, yield and yield attributes. The cobs were then threshed to determine the seed number per pod, kernel weight (g per 1000 seeds), harvest index (HI), and seed yield. Determination of HI was done using the same plants that were used for determining yield per plot. Phenological events were determined three times per week on each plot. Dates of emergence, tasselling, and physiological maturity (black layer observed in grains of the mid-portion of the ear) were recorded when 50% of the plants reached that stage.
2.4. Photosynthesis
Photosynthesis measurements were taken three times in each season using a portable gas exchange measuring system (Li 6400, LI-COR, Lincoln, NE, USA). These measurements were taken on the last day of exposure to drought and flooding stresses. Measurements were conducted on a fully matured third leaf from the top, during a sunny day between 10:00 a.m. and 14:00 p.m. The exposure of maize to excess moisture started 12 days later, after the drought treatment had already started so that the end of drought stress could coincide with flooding stress. This was done to ensure that measurements could be done at the same time after crop exposure to the two stresses.
2.5. Statistical Analysis
Analysis of variance was conducted using Genstat 19th Edition (
https://genstat.kb.vsni.co.uk/knowledge-base/new-features-genstat-19th-edition). Univariate analysis of each variable was done separately for each cultivar with a 2 × 3 factorial arrangement of treatments, plus a control structure as described by Payne et al. [
23]. Multiple comparisons of means were performed using least significant difference (LSD) Tukey’s Studentized (HSD) range (
p < 0.05). The data from the two seasons were analysed separately because of significant (
p < 0.05) season–year interactions.
4. Discussion
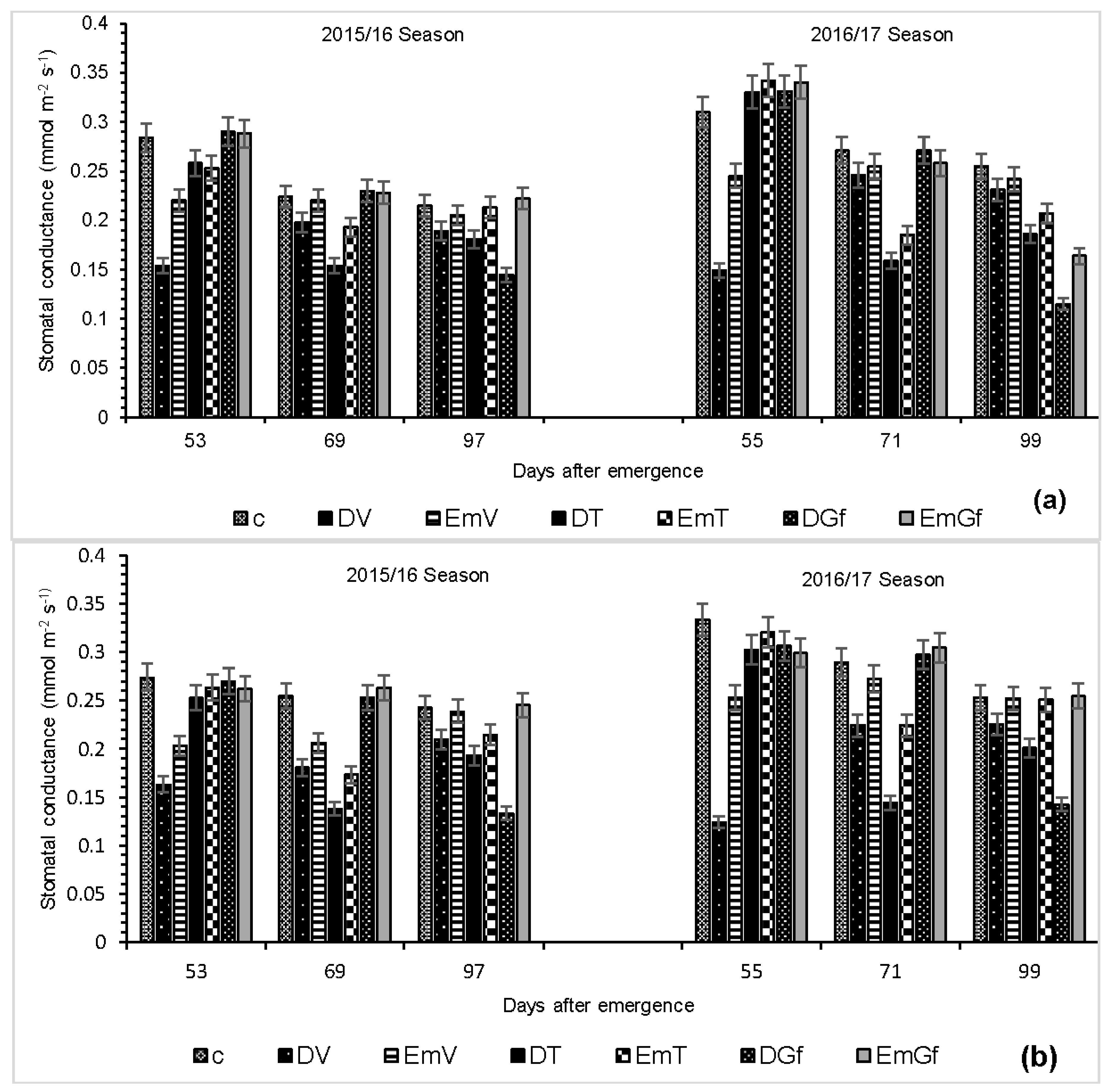
A reduction in photosynthesis due to drought and flooding is ascribed to stomatal and non-stomatal limitations [
24]. This can either be due to the physiological control (stomata closure) of the influx of atmospheric CO
2 into the mesophyll cells [
25] or the perturbation of metabolic activities, such as photo-inactivation of the PSII centres [
26]. There was a strong linear relationship between photosynthesis and stomatal conductance at most of the sampling dates 53 DAE (
r2 = 0.87), 69 DAE (
r2 = 0.83) and 91 DAE (
r2 = 0.53) for PAN 413 and for PAN 6Q 245, 53 DAE (
r2 = 0.89), 69 DAE (
r2 = 0.43), 91 DAE (
r2 = 0.71) (results not presented).
The strong relationship existing between photosynthesis and stomatal conductance implies that the reduction in photosynthesis is regulated by stomatal closure, while a weak relationship indicates that the reduction in photosynthesis is a result of regulation by non-stomatal factors [
27]. The decrease in photosynthesis due to drought is in agreement with previous reports for maize by Pelleschi et al. [
28] and Voronin et al. [
29]. Pertaining to reduced photosynthesis due to excess moisture, similar findings have been reported by Ashraf et al. [
30], Ahmed et al. [
31], and Yordanova and Popova [
32]. Ahmed et al. [
31] attributed reduction in photosynthesis to stomatal closure, although they noted that not all the reduction was due to stomatal closure, which agrees with the current findings. The same authors highlighted that damages occurred internally correlated with photo-inhibition, one of the non-stomatal factors limiting photosynthesis. The same mechanisms reported under drought stress also occur under flooding stress, whereby there is a reduction in the stomatal apertures caused by internal water deficit. Therefore, there is a reduction in CO
2 intake by the leaf, and photosynthetic carbon assimilation is decreased in favour of respiration [
32].
The effect of reduced photosynthesis is two-fold: first, water is not released through the stomata to satisfy the atmospheric demand, and then the amount of assimilation is reduced because of reduced CO
2 levels in the sub-stomatal cavity [
30]. Accordingly, the reduced CO
2 assimilation leads to reduced biomass accumulation, causing lower dry matter partitioning to the different plant organs, including leaves, consequently leading to reduced LAI. Reduced LAI translates into reduced interception of solar radiation, with reduced photosynthates production [
20].
Our findings concur with the findings of Çakir [
13] who reported a decrease in LAI as a result of drought. The results also agree with the notion that leaf elongation is one of the most sensitive plant processes to limited water [
33]. Requirement of photosynthates and energy is reduced in leaves under drought conditions and the photosynthetic assimilates from leaves are directed toward roots for their elongation to increase the water uptake [
34]. Roots act as primary sensors of water deficiency in soil and transduce signals to the aerial parts to modulate the growth and development [
35]. The consequence is a decline in the leaf area and thus the crop canopy. Like drought conditions, LAI was also negatively affected under excess moisture conditions at all growth stages. However, the impact was relatively low under flooding compared with drought imposed during the same growth stage (
Table 6). Similar findings on the effect of excess moisture on maize LAI has been reported by Guoping et al. [
36] and Jiang et al. [
37], who found out that exposing the crop to excess moisture/waterlogged conditions can result in reduced LAI.
Water deficit is one of the limiting factors for plant growth and development and has a two-fold effect on plants as it reduces the production of dry matter and causes a disorder to the partitioning of carbohydrates to grain, hence reducing HI [
38]. The reported significant (
p < 0.05) maize biomass accumulation reduction due to water stress and flooding imposed at the early vegetative stage compared with the non-stressed treatment is in line with previous studies [
12,
19]. This reduction in biomass accumulation is attributed to reduced leaf expansion and reduced stem internode elongation due to the water stress [
13] or flooding [
20], consequently affecting the dry matter accumulation. In contrast to our findings and those of Rene et al. [
19], Guoping et al. [
36], reported that excess moisture around anthesis did not have any effect on maize dry matter accumulation. The inconsistency is most probably attributed to the duration of the water logging. In our case, the plots were flooded for eight consecutive days, whereas in Guoping et al. [
36], waterlogging lasted only three days. Their findings regarding the effect of flooding during the grain filling stage on maize dry matter accumulation, however, agree with ours.
Generally maize grain yield varied between the two growing seasons. This variation is mainly attributed to the variation in air temperature between the two growing seasons. The temperature on some of the days during the 2015/2016 growing season exceeded the crop’s cut-off temperature (>30 °C). Lobell et al. [
39] observed a strong negative yield response to accumulation of temperatures above 30 °C (or extreme degree days). This is attributed to a higher vapor pressure demand (VPD) between the saturated leaf interior and the ambient air which forces the plants to close their stomata, despite the presence of enough water in the soil. At such high temperature, the plants will not be able to deliver the required amount of water to satisfy the atmospheric demand due to the resistance encountered as the water travels from the soil through the plant to the atmosphere at the soil–root interface, root endodermis resistance, root-xylem resistance, petiole resistance, and stomatal resistance.
Our results showed that drought imposed at any growth stage results in grain yield reductions, with the extent of damage depending on the developmental stage at which crop stress was imposed. This drop in yield is attributed to the decrease in the kernel number per plant and/or kernel weight resulting in the reduction of HI. Reduction in HI due to progressive water stress explains the fact that grain yield is much more affected than the total dry matter [
40]. Water stress during the tasselling stage lowered kernel number by 44%, which was twice and higher than the effect from stress imposed during other stages. This indicates that water availability at tasselling stage is very critical for maize yield. Zinselmeier et al. [
41] reported embryo formation in plants subjected to low water potential for five days around pollination, but also observed a decline in ovary starch, which consequently resulted in reduced kernel number per plant. On the other hand, the impact of water stress during the grain filling stage was more prevalent on the kernel weight, causing a 16% reduction compared with the control. This reduction is about a third of the impact caused by water stress during the tasselling stage on kernel number. The reduction in kernel weight observed in our study is most probably attributed to the presence of a weak source (leaves) that could supply the required amount of assimilates to the sink (kernels) since drought caused a drastic drop in LAI. At this stage, grain set occurred well, as can be shown by high number of grains that were formed, but the kernel weight was very low. Our findings differ from the ones that were reported by Çakir [
13], who found out that the treatments under drought had higher kernel weight. He attributed this to higher grain filling rate in the presence of fewer kernels. Such differences could most probably be due to genetic factors.
Zaidi et al. [
18] attributed grain yield reduction to the effects of excess moisture on various growth and biochemical parameters, impairment of anthesis, and silking, which eventually resulted in poor kernel development. The impact of flooding on kernel number and kernel weight varied among developmental stages during which flooding was imposed and was inconsistent between years. Flooding imposed at all developmental stages did not reduce the kernel number for both cultivars in both years, except for the flooding imposed during the vegetative stage for PAN 413 in 2016/2017. The impact of flooding on kernel weight was also inconsistent between years for each cultivar. During the 2015/2016 growing season, flooding didn’t cause any significant effect on the kernel weight of PAN 6Q-245. In 2016/2017, however, there was a 6.5% reduction in kernel weight for treatments flooded during the tasselling stages compared with the control. On the other hand, flooding during the vegetative and tasselling stages in the 2015/2016 growing season caused a 5% reduction in kernel weight compared with the control for PAN 413. While flooding during the 2016/2017 growing season did not cause any significant impact on kernel weight of PAN 413. The absence of significant reduction in kernel weight and kernel number from flooding treatments could be due to the adventitious roots which started to grow from the first and second nodes above the soil, which is in agreement with the studies of Klepper [
42].