Chitosan-Coated Alginate Microcapsules of a Full-Spectrum Cannabis Extract: Characterization, Long-Term Stability and In Vitro Bioaccessibility
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Standards
2.2. Cannabis Raw Extract
2.3. Preparation of Alginate–Chitosan Microcapsules
2.4. Cannabinoid Content of Alginate–Chitosan Microcapsules
2.5. Encapsulation Efficiency
2.6. Physical Characterization of Alginate–Chitosan Microcapsules
2.7. Long-Term Stability of Cannabinoids in Alginate–Chitosan Microcapsules
2.8. In Vitro Gastrointestinal Release
3. Results and Discussion
3.1. Cannabinoid Content and Encapsulation Efficiency
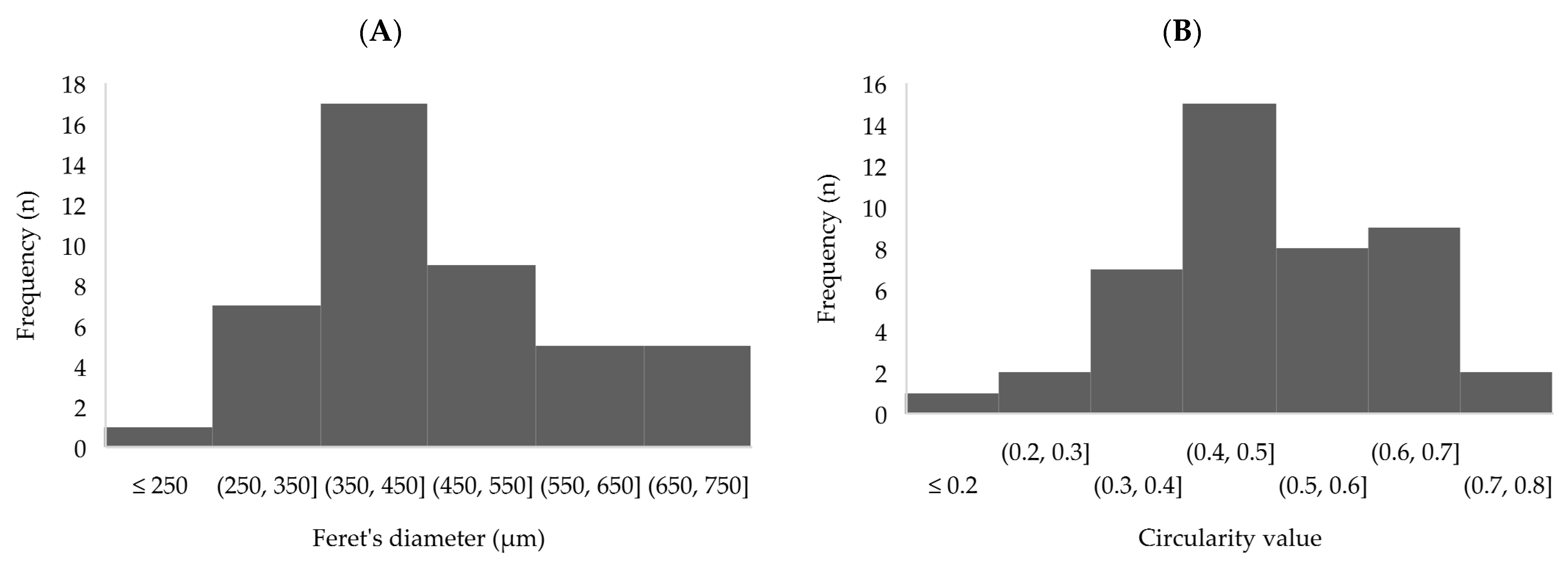
3.2. Physical Properties of Capsules: Zeta Potential, Size and Shape
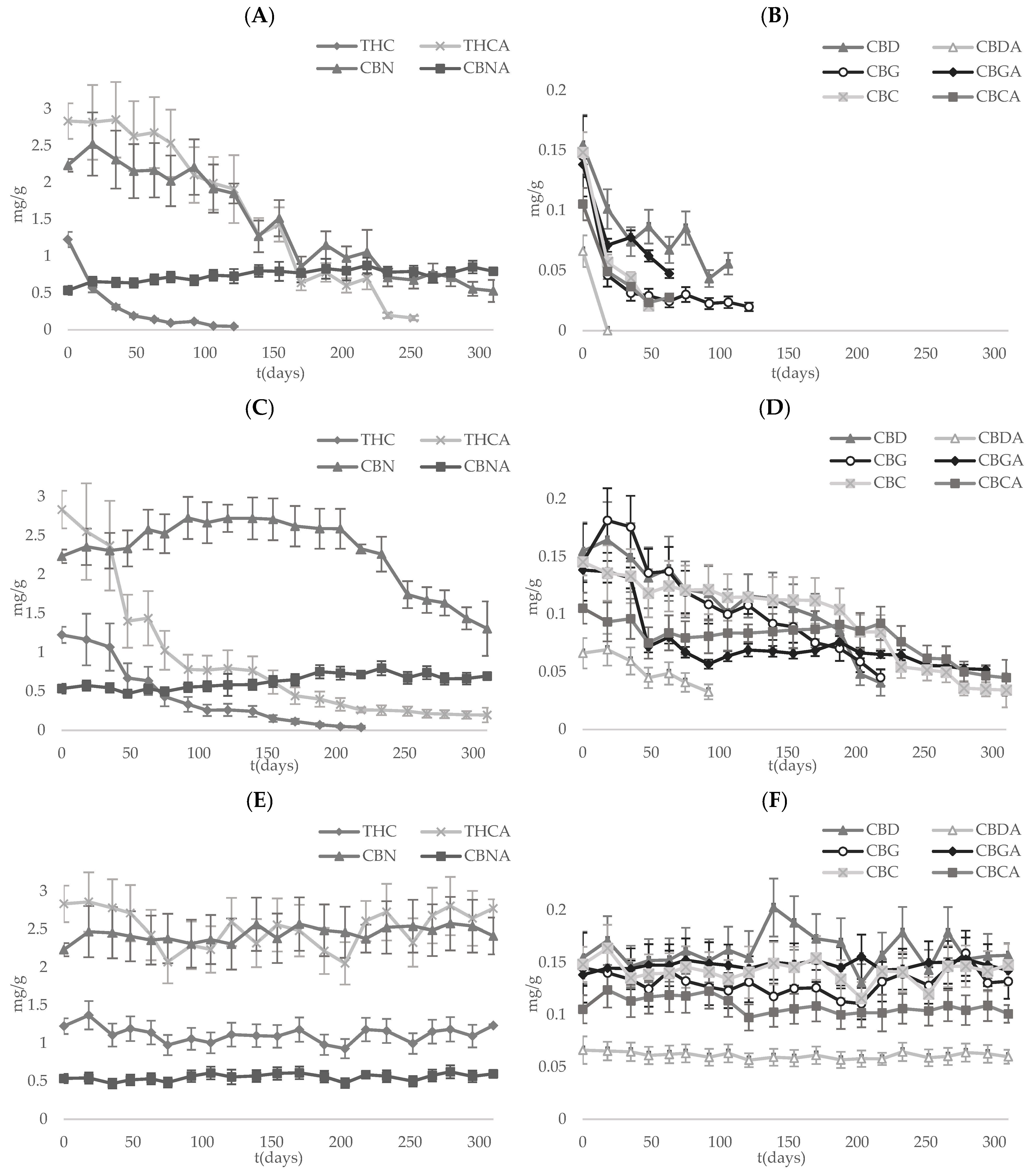
3.3. Long-Term Stability of Cannabinoids
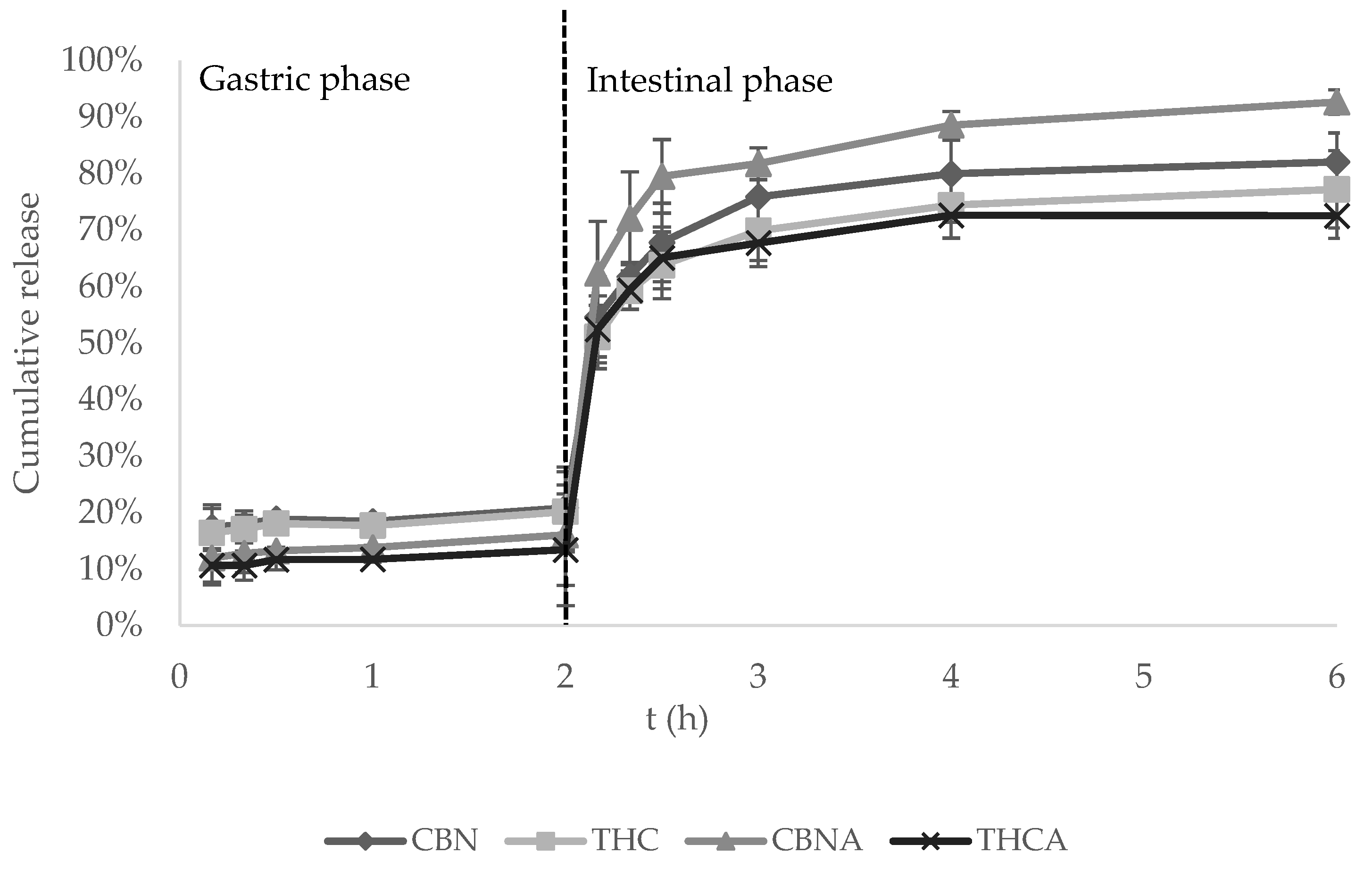
3.4. In Vitro Gastrointestinal Release
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- UNODC. World Drug Report 2022. United Nations: Office on Drugs and Crime; 2022. Available online: www.unodc.org/unodc/en/data-and-analysis/world-drug-report-2022.html (accessed on 2 March 2023).
- Hordowicz, M.; Klimkiewicz, A.; Jarosz, J.; Wysocka, M.; Jastrzębska, M. Knowledge, attitudes, and prescribing patterns of cannabis and cannabinoid-containing medicines among European healthcare workers: A systematic literature review. Drug Alcohol Depend. 2021, 221, 108652. [Google Scholar] [CrossRef]
- Kosiba, J.D.; Maisto, S.A.; Ditre, J.W. Patient-reported use of medical cannabis for pain, anxiety, and depression symptoms: Systematic review and meta-analysis. Soc. Sci. Med. 2019, 233, 181–192. [Google Scholar] [CrossRef]
- Leung, J.; Chan, G.; Stjepanović, D.; Chung, J.Y.C.; Hall, W.; Hammond, D. Prevalence and self-reported reasons of cannabis use for medical purposes in USA and Canada. Psychopharmacology 2022, 239, 1509–1519. [Google Scholar] [CrossRef] [PubMed]
- Charoenporn, V.; Charernboon, T.; Mackie, C.J. Medical Cannabis as a Substitute for Prescription Agents: A Systematic Review and Meta-analysis. J. Subst. Use 2022, 1–13. [Google Scholar] [CrossRef]
- ElSohly, M.A. (Ed.) Marijuana and the Cannabinoids; Humana Press: Totowa, NJ, USA, 2007. [Google Scholar] [CrossRef]
- Russo, E.B.; Grotenhermen, F. (Eds.) The Handbook of Cannabis Therapeutics: From Bench to Bedside; Routledge: New York, NY, USA, 2006. [Google Scholar] [CrossRef]
- Russo, E.B. Taming THC: Potential cannabis synergy and phytocannabinoid-terpenoid entourage effects. Br. J. Pharmacol. 2011, 163, 1344–1364. [Google Scholar] [CrossRef] [PubMed]
- Wallach, J. Chapter 5—Medicinal Cannabis: An overview for health-care providers. In Remington, 23rd ed.; Adejare, A., Ed.; Academic Press: Cambridge, MA, USA, 2021; pp. 75–101. [Google Scholar] [CrossRef]
- Walsh, K.B.; McKinney, A.E.; Holmes, A.E. Minor Cannabinoids: Biosynthesis, Molecular Pharmacology and Potential Therapeutic Uses. Front. Pharmacol. 2021, 12, 777804. [Google Scholar] [CrossRef] [PubMed]
- Grotenhermen, F. Clinical Pharmacokinetics of Cannabinoids. J. Cannabis Ther. 2003, 3, 3–51. [Google Scholar] [CrossRef]
- Huestis, M.A. Human Cannabinoid Pharmacokinetics. Chem. Biodivers. 2007, 4, 1770–1804. [Google Scholar] [CrossRef]
- Poyatos, L.; Pérez-Acevedo, A.; Papaseit, E.; Pérez-Mañá, C.; Martin, S.; Hladun, O.; Siles, A.; Torrens, M.; Busardo, F.; Farré, M. Oral Administration of Cannabis and Δ-9-tetrahydrocannabinol (THC) Preparations: A Systematic Review. Medicina 2020, 56, 309. [Google Scholar] [CrossRef]
- Light, K.; Karboune, S. Emulsion, hydrogel and emulgel systems and novel applications in cannabinoid delivery: A review. Crit. Rev. Food Sci. Nutr. 2022, 62, 8199–8229. [Google Scholar] [CrossRef]
- McClements, D.J. Enhancing Efficacy, Performance, and Reliability of Cannabis Edibles: Insights from Lipid Bioavailability Studies. Annu. Rev. Food Sci. Technol. 2020, 11, 45–70. [Google Scholar] [CrossRef]
- Stella, B.; Baratta, F.; Della Pepa, C.; Arpicco, S.; Gastaldi, D.; Dosio, F. Cannabinoid Formulations and Delivery Systems: Current and Future Options to Treat Pain. Drugs 2021, 81, 1513–1557. [Google Scholar] [CrossRef] [PubMed]
- Maayah, Z.H.; Takahara, S.; Ferdaoussi, M.; Dyck, J.R.B. The molecular mechanisms that underpin the biological benefits of full-spectrum cannabis extract in the treatment of neuropathic pain and inflammation. Biochim. Biophys. Acta BBA-Mol. Basis Dis. 2020, 1866, 165771. [Google Scholar] [CrossRef]
- Uziel, A.; Gelfand, A.; Amsalem, K.; Berman, P.; Lewitus, G.M.; Meiri, D.; Lewitus, D.Y. Full-Spectrum Cannabis Extract Microdepots Support Controlled Release of Multiple Phytocannabinoids for Extended Therapeutic Effect. ACS Appl. Mater. Interfaces 2020, 12, 23707–23716. [Google Scholar] [CrossRef]
- Freire, N.F.; Feuser, P.E.; Ambel, E.M.T.; Cordani, M.; De Pieri, E.; Machado-de-Ávila, R.A.; Zielinski, A.A.; Sayer, C.; de Araújo, P.H.H.; Díez, G.V.; et al. Preparation and characterization of full-spectrum cannabis extract loaded poly(thioether-ester) nanoparticles: In vitro evaluation of their antitumoral efficacy. Colloids Surf. Physicochem. Eng. Asp. 2023, 658, 130676. [Google Scholar] [CrossRef]
- Ahadian, S.; Finbloom, J.A.; Mofidfar, M.; Diltemiz, S.E.; Nasrollahi, F.; Davoodi, E.; Hosseini, V.; Mylonaki, I.; Sangabathuni, S.; Montazerian, H.; et al. Micro and nanoscale technologies in oral drug delivery. Adv. Drug Deliv. Rev. 2020, 157, 37–62. [Google Scholar] [CrossRef] [PubMed]
- Ching, S.H.; Bansal, N.; Bhandari, B. Alginate gel particles–A review of production techniques and physical properties. Crit. Rev. Food Sci. Nutr. 2017, 57, 1133–1152. [Google Scholar] [CrossRef]
- Lengyel, M.; Kállai-Szabó, N.; Antal, V.; Laki, A.J.; Antal, I. Microparticles, Microspheres, and Microcapsules for Advanced Drug Delivery. Sci. Pharm. 2019, 87, 20. [Google Scholar] [CrossRef]
- George, M.; Abraham, T.E. Polyionic hydrocolloids for the intestinal delivery of protein drugs: Alginate and chitosan—A review. J. Control. Release 2006, 114, 1–14. [Google Scholar] [CrossRef]
- Aizpurua-Olaizola, O.; Navarro, P.; Vallejo, A.; Olivares, M.; Etxebarria, N.; Usobiaga, A. Microencapsulation and storage stability of polyphenols from Vitis vinifera grape wastes. Food Chem. 2016, 190, 614–621. [Google Scholar] [CrossRef]
- Bamidele, O.P.; Emmambux, M.N. Encapsulation of bioactive compounds by ‘extrusion’ technologies: A review. Crit. Rev. Food Sci. Nutr. 2021, 61, 3100–3118. [Google Scholar] [CrossRef]
- Whelehan, M.; Marison, I.W. Microencapsulation using vibrating technology. J. Microencapsul. 2011, 28, 669–688. [Google Scholar] [CrossRef]
- Winnicki, R. Cannabinoid Formulations. US8808734B2, 19 August 2014. Available online: https://patents.google.com/patent/US8808734B2/en (accessed on 30 January 2023).
- Deladino, L.; Anbinder, P.S.; Navarro, A.S.; Martino, M.N. Encapsulation of natural antioxidants extracted from Ilex paraguariensis. Carbohydr. Polym. 2008, 71, 126–134. [Google Scholar] [CrossRef]
- Mazzoli, A.; Favoni, O. Particle size, size distribution and morphological evaluation of airborne dust particles of diverse woods by Scanning Electron Microscopy and image processing program. Powder Technol. 2012, 225, 65–71. [Google Scholar] [CrossRef]
- Lindholst, C. Long term stability of cannabis resin and cannabis extracts. Aust. J. Forensic Sci. 2010, 42, 181–190. [Google Scholar] [CrossRef]
- Zamengo, L.; Bettin, C.; Badocco, D.; Di Marco, V.; Miolo, G.; Frison, G. The role of time and storage conditions on the composition of hashish and marijuana samples: A four-year study. Forensic Sci. Int. 2019, 298, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Brodkorb, A.; Egger, L.; Alminger, M.; Alvito, P.; Assunção, R.; Ballance, S.; Bohn, T.; Bourlieu-Lacanal, C.; Boutrou, R.; Carrière, F.; et al. INFOGEST static in vitro simulation of gastrointestinal food digestion. Nat. Protoc. 2019, 14, 991–1014. [Google Scholar] [CrossRef] [PubMed]
- Minekus, M.; Alminger, M.; Alvito, P.; Ballance, S.; Bohn, T.; Bourlieu, C.; Carrière, F.; Boutrou, R.; Corredig, M.; Dupont, D.; et al. A standardised static in vitro digestion method suitable for food—An international consensus. Food Funct. 2014, 5, 1113–1124. [Google Scholar] [CrossRef]
- Aizpurua-Olaizola, O.; Soydaner, U.; Öztürk, E.; Schibano, D.; Simsir, Y.; Navarro, P.; Etxebarria, N.; Usobiaga, A. Evolution of the Cannabinoid and Terpene Content during the Growth of Cannabis sativa Plants from Different Chemotypes. J. Nat. Prod. 2016, 79, 324–331. [Google Scholar] [CrossRef]
- Maioli, C.; Mattoteia, D.; Amin, H.I.M.; Minassi, A.; Caprioglio, D. Cannabinol: History, Syntheses, and Biological Profile of the Greatest ‘Minor’ Cannabinoid. Plants 2022, 11, 2896. [Google Scholar] [CrossRef]
- Meija, J.; McRae, G.; Miles, C.O.; Melanson, J.E. Thermal stability of cannabinoids in dried cannabis: A kinetic study. Anal. Bioanal. Chem. 2022, 414, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Marangoni, I.P.; Marangoni, A.G. Cannabis edibles: Dosing, encapsulation, and stability considerations. Curr. Opin. Food Sci. 2019, 28, 1–6. [Google Scholar] [CrossRef]
- Corroon, J. Cannabinol and Sleep: Separating Fact from Fiction. Cannabis Cannabinoid Res. 2021, 6, 366–371. [Google Scholar] [CrossRef] [PubMed]
- Karniol, I.G.; Shirakawa, I.; Takahashi, R.N.; Knobel, E.; Musty, R.E. Effects of Δ9-Tetrahydrocannabinol and Cannabinol in Man. Pharmacology 1975, 13, 502–512. [Google Scholar] [CrossRef] [PubMed]
- Romano, L.; Hazekamp, A. An Overview of Galenic Preparation Methods for Medicinal Cannabis. Curr. Bioact. Compd. 2019, 15, 174–195. [Google Scholar] [CrossRef]
- Gazori, T.; Khoshayand, M.R.; Azizi, E.; Yazdizade, P.; Nomani, A.; Haririan, I. Evaluation of Alginate/Chitosan nanoparticles as antisense delivery vector: Formulation, optimization and in vitro characterization. Carbohydr. Polym. 2009, 77, 599–606. [Google Scholar] [CrossRef]
- Ho, W.-X.; Chen, W.-T.; Lien, C.-H.; Yang, H.-Y.; Chen, K.-H.; Wei, Y.-F.; Wang, M.-H.; Ko, I.-T.; Tseng, F.G.; Yin, H.S. Physical, Chemical, and Biological Properties of Chitosan-Coated Alginate Microparticles Loaded with Porcine Interleukin-1β: A Potential Protein Adjuvant Delivery System. Int. J. Mol. Sci. 2022, 23, 9959. [Google Scholar] [CrossRef]
- Khan, M.A.; Yue, C.; Fang, Z.; Hu, S.; Cheng, H.; Bakry, A.M.; Liang, L. Alginate/chitosan-coated zein nanoparticles for the delivery of resveratrol. J. Food Eng. 2019, 258, 45–53. [Google Scholar] [CrossRef]
- Ji, R.; Wu, J.; Zhang, J.; Wang, T.; Zhang, X.; Shao, L.; Chen, D.; Wang, J. Extending Viability of Bifidobacterium longum in Chitosan-Coated Alginate Microcapsules Using Emulsification and Internal Gelation Encapsulation Technology. Front. Microbiol. 2019, 10, 1389. [Google Scholar] [CrossRef]
- Gibson, N. Colloidal stability of modified nanodiamond particles. Diam. Relat. Mater. 2009, 18, 620–626. [Google Scholar] [CrossRef]
- Joseph, E.; Singhvi, G. Chapter 4—Multifunctional nanocrystals for cancer therapy: A potential nanocarrier. In Nanomaterials for Drug Delivery and Therapy; Grumezescu, A.M., Ed.; William Andrew Publishing: Norwich, NY, USA, 2019; pp. 91–116. [Google Scholar] [CrossRef]
- Shnoudeh, A.J.; Hamad, I.; Abdo, R.W.; Qadumii, L.; Jaber, A.Y.; Surchi, H.S.; Alkelany, S.Z. Chapter 15—Synthesis, Characterization, and Applications of Metal Nanoparticles. In Biomaterials and Bionanotechnology; Tekade, R.K., Ed.; Academic Press: Cambridge, MA, USA, 2019; pp. 527–612. [Google Scholar] [CrossRef]
- Khorshidian, N.; Mahboubi, A.; Kalantari, N.; Hosseini, H.; Yousefi, M.; Arab, M.; da Cruz, A.G.; Mortazavian, A.M.; Mahdavi, F.S. Chitosan-Coated Alginate Microcapsules Loaded with Herbal galactagogue Extract: Formulation Optimization and Characterization. Iran. J. Pharm. Res. IJPR 2019, 18, 1180–1195. [Google Scholar] [CrossRef]
- Heinzen, C.; Berger, A.; Marison, I. Use of Vibration Technology for Jet Break-Up for Encapsulation of Cells and Liquids in Monodisperse Microcapsules. In Fundamentals of Cell Immobilisation Biotechnology; Nedović, V., Willaert, R., Eds.; Springer: Dordrecht, The Netherlands, 2004; pp. 257–275. [Google Scholar] [CrossRef]
- Lee, B.-B.; Ravindra, P.; Chan, E.-S. Size and Shape of Calcium Alginate Beads Produced by Extrusion Dripping. Chem. Eng. Technol. 2013, 36, 1627–1642. [Google Scholar] [CrossRef]
- Fujiwara, G.M.; Campos, R.; Costa, C.K.; Dias, J.D.F.G.; Miguel, O.G.; Miguel, M.D.; Marques, F.D.A.; Zanin, S.M.W. Production and characterization of alginate-starch-chitosan microparticles containing stigmasterol through the external ionic gelation technique. Braz. J. Pharm. Sci. 2013, 49, 537–547. [Google Scholar] [CrossRef]
- McClements, D.J. Designing biopolymer microgels to encapsulate, protect and deliver bioactive components: Physicochemical aspects. Adv. Colloid Interface Sci. 2017, 240, 31–59. [Google Scholar] [CrossRef] [PubMed]
- Fraguas-Sánchez, A.I.; Torres-Suárez, A.I.; Cohen, M.; Delie, F.; Bastida-Ruiz, D.; Fernández-Carballido, A. PLGA Nanoparticles for the Intraperitoneal Administration of CBD in the Treatment of Ovarian Cancer: In Vitro and In Ovo Assessment. Pharmaceutics 2020, 12, 439. [Google Scholar] [CrossRef] [PubMed]
- de la Ossa, D.H.P.; Ligresti, A.; Gil-Alegre, M.; Aberturas, M.; Molpeceres, J.; Di Marzo, V.; Suárez, A.T. Poly-ε-caprolactone microspheres as a drug delivery system for cannabinoid administration: Development, characterization and in vitro evaluation of their antitumoral efficacy. J. Control. Release 2012, 161, 927–932. [Google Scholar] [CrossRef] [PubMed]
- Esposito, E.; Drechsler, M.; Cortesi, R.; Nastruzzi, C. Encapsulation of cannabinoid drugs in nanostructured lipid carriers. Eur. J. Pharm. Biopharm. Off. J. Arb. Pharm. Verfahr. EV 2016, 102, 87–91. [Google Scholar] [CrossRef]
- Berrocoso, E.; Rey-Brea, R.; Fernández-Arévalo, M.; Micó, J.A.; Martín-Banderas, L. Single oral dose of cannabinoid derivate loaded PLGA nanocarriers relieves neuropathic pain for eleven days. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 2623–2632. [Google Scholar] [CrossRef]
- Carbone, M.; Castelluccio, F.; Daniele, A.; Sutton, A.; Ligresti, A.; Di Marzo, V.; Gavagnin, M. Chemical characterisation of oxidative degradation products of Δ9-THC. Tetrahedron 2010, 66, 9497–9501. [Google Scholar] [CrossRef]
- Mechoulam, R. Chemistry of Cannabis. In Psychotropic Agents: Part III: Alcohol and Psychotomimetics, Psychotropic Effects of Central Acting Drugs; Hoffmeister, F., Stille, G., Eds.; Springer: Berlin/Heidelberg, Germany, 1982; pp. 119–134. [Google Scholar] [CrossRef]
- Fairbairn, J.W.; Liebmann, J.A.; Rowan, M.G. The stability of cannabis and its preparations on storage. J. Pharm. Pharmacol. 1976, 28, 1–7. [Google Scholar] [CrossRef]
- Reason, D.A.; Grainger, M.N.C.; Lane, J.R. Optimal Storage Conditions of Commercial Cannabis Crops. Ind. Eng. Chem. Res. 2022, 61, 14691–14701. [Google Scholar] [CrossRef]
- Turner, C.E.; Hadley, K.W.; Fetterman, P.S.; Doorenbos, N.J.; Quimby, M.W.; Waller, C. Constituents of Cannabis sativa L. IV: Stability of Cannabinoids in Stored Plant Material. J. Pharm. Sci. 1973, 62, 1601–1605. [Google Scholar] [CrossRef] [PubMed]
- Martone, G.; Casa, E.D. Analysis of the ageing processes in hashish samples from different geographic origin. Forensic Sci. Int. 1990, 47, 147–155. [Google Scholar] [CrossRef]
- Trofin, I.; Dabija, G.; Vaireanu, D.-I.; Laurentiu, F. The Influence of Long-term Storage Conditions on the Stability of Cannabinoids derived from Cannabis Resin. Rev. Chim. 2012, 63, 422–427. [Google Scholar]
- Smith, R.N.; Vaughan, C.G. The decomposition of acidic and neutral cannabinoids in organic solvents. J. Pharm. Pharmacol. 1977, 29, 286–290. [Google Scholar] [CrossRef] [PubMed]
- Trofin, I.; Dabija, G.; Vaireanu, D.-I.; Laurentiu, F. Long-term Storage and Cannabis Oil Stability. Rev. Chim. 2012, 53, 294. [Google Scholar]
- Marinol Monograph. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2016/205525s000lbl.pdf (accessed on 30 January 2023).
- Ribeiro, A.J.; Neufeld, R.J.; Arnaud, P.; Chaumeil, J.C. Microencapsulation of lipophilic drugs in chitosan-coated alginate microspheres. Int. J. Pharm. 1999, 187, 115–123. [Google Scholar] [CrossRef]
- Bhattacharjee, S. Understanding the burst release phenomenon: Toward designing effective nanoparticulate drug-delivery systems. Ther. Deliv. 2021, 12, 21–36. [Google Scholar] [CrossRef]
- Lunn, S.; Diaz, P.; O’Hearn, S.; Cahill, S.P.; Blake, A.; Narine, K.; Dyck, J. Human Pharmacokinetic Parameters of Orally Administered Δ9-Tetrahydrocannabinol Capsules Are Altered by Fed Versus Fasted Conditions and Sex Differences. Cannabis Cannabinoid Res. 2019, 4, 255–264. [Google Scholar] [CrossRef]
- Dima, C.; Assadpour, E.; Dima, S.; Jafari, S.M. Bioavailability and bioaccessibility of food bioactive compounds; overview and assessment by in vitro methods. Compr. Rev. Food Sci. Food Saf. 2020, 19, 2862–2884. [Google Scholar] [CrossRef]

| Cannabinoid | Cext (mg/g) | Ccap (mg/g) | EE (%) |
|---|---|---|---|
| THC | 21 ± 1 | 1.2 ± 0.1 | 98% |
| THCA | 49 ± 3 | 2.8 ± 0.2 | 96% |
| CBN | 42 ± 3 | 2.2 ± 0.1 | 87% |
| CBNA | 8.8 ± 0.6 | 0.53 ± 0.06 | 101% |
| CBD | 3.0 ± 0.2 | 0.15 ± 0.02 | 87% |
| CBDA | 1.5 ± 0.1 | 0.07 ± 0.01 | 75% |
| CBG | 2.3 ± 0.3 | 0.14 ± 0.03 | 104% |
| CBGA | 2.3 ± 0.2 | 0.14 ± 0.01 | 9 9% |
| CBC | 2.6 ± 0.2 | 0.15 ± 0.02 | 95% |
| CBCA | 2.0 ± 0.2 | 0.11 ± 0.01 | 86% |
| RT with Light Exposure | RT without Light Exposure | 4 °C without Light Exposure | |||||||
|---|---|---|---|---|---|---|---|---|---|
| λ | r2 | t1/2 | λ | r2 | t1/2 | λ | r2 | t1/2 | |
| THC | 0.0298 | 0.9815 | 23 | 0.0144 | 0.9874 | 48 | 0.0005 | 0.4471 | 1426 |
| THCA | 0.0077 | 0.8669 | 90 | 0.0098 | 0.9891 | 70 | 0.0006 | 0.4206 | 1231 |
| CBN | 0.0041 | 0.9365 | 170 | 0.0004 | 0.1155 | 1679 | −0.0005 | 0.8118 | −1466 |
| CBNA | −0.0018 | 0.8873 | −394 | −0.0010 | 0.8209 | −680 | −0.0002 | 0.2481 | −2913 |
| CBD | 0.0116 | 0.9313 | 60 | 0.0039 | 0.8540 | 177 | −0.0002 | 0.1172 | −3534 |
| CBDA | 0.0065 | 0.9302 | 107 | 0.0004 | 0.6986 | 1613 | |||
| CBG | 0.0206 | 0.8994 | 34 | 0.0038 | 0.8948 | 180 | 0.0005 | 0.4386 | 1426 |
| CBGA | 0.0176 | 0.9505 | 39 | 0.0039 | 0.8895 | 177 | −0.0003 | 0.7005 | −2170 |
| CBC | 0.0401 | 0.9855 | 17 | 0.0035 | 0.8897 | 196 | 0.0003 | 0.3161 | 2511 |
| CBCA | 0.0265 | 0.9552 | 26 | 0.0016 | 0.8016 | 440 | 0.0000 | 0.0039 | −27,211 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Villate, A.; San Nicolas, M.; Olivares, M.; Aizpurua-Olaizola, O.; Usobiaga, A. Chitosan-Coated Alginate Microcapsules of a Full-Spectrum Cannabis Extract: Characterization, Long-Term Stability and In Vitro Bioaccessibility. Pharmaceutics 2023, 15, 859. https://doi.org/10.3390/pharmaceutics15030859
Villate A, San Nicolas M, Olivares M, Aizpurua-Olaizola O, Usobiaga A. Chitosan-Coated Alginate Microcapsules of a Full-Spectrum Cannabis Extract: Characterization, Long-Term Stability and In Vitro Bioaccessibility. Pharmaceutics. 2023; 15(3):859. https://doi.org/10.3390/pharmaceutics15030859
Chicago/Turabian StyleVillate, Aitor, Markel San Nicolas, Maitane Olivares, Oier Aizpurua-Olaizola, and Aresatz Usobiaga. 2023. "Chitosan-Coated Alginate Microcapsules of a Full-Spectrum Cannabis Extract: Characterization, Long-Term Stability and In Vitro Bioaccessibility" Pharmaceutics 15, no. 3: 859. https://doi.org/10.3390/pharmaceutics15030859
APA StyleVillate, A., San Nicolas, M., Olivares, M., Aizpurua-Olaizola, O., & Usobiaga, A. (2023). Chitosan-Coated Alginate Microcapsules of a Full-Spectrum Cannabis Extract: Characterization, Long-Term Stability and In Vitro Bioaccessibility. Pharmaceutics, 15(3), 859. https://doi.org/10.3390/pharmaceutics15030859





