Synthesis and Characterization of Linear Copolymers Based on Pharmaceutically Functionalized Monomeric Choline Ionic Liquid for Delivery of p-Aminosalicylate
Abstract
1. Introduction
2. Materials and Methods
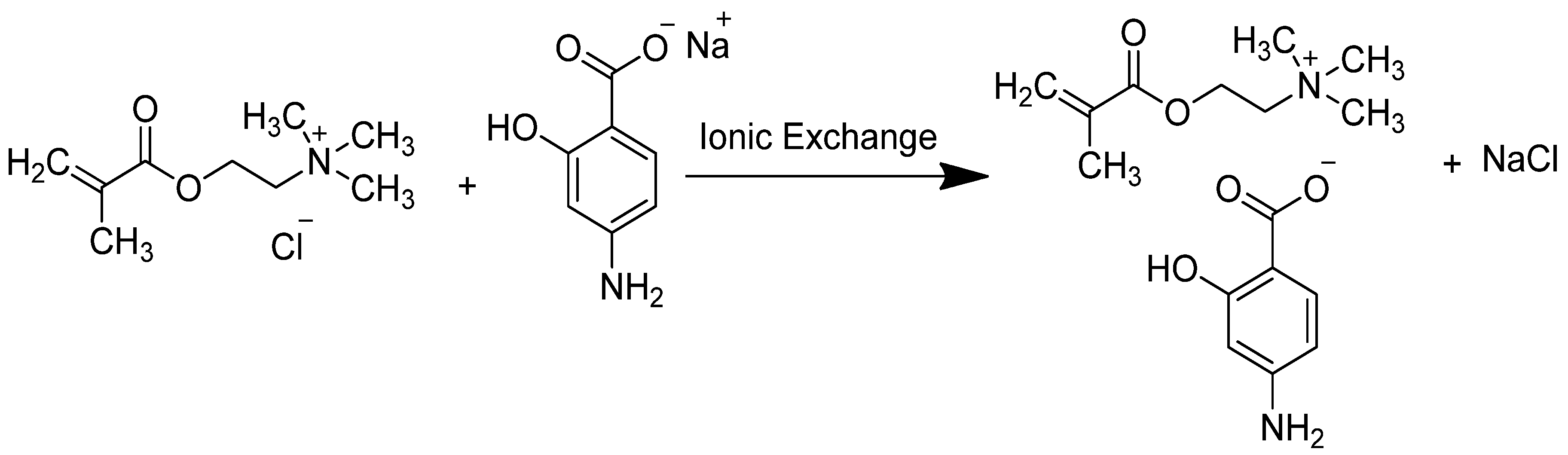
2.1. Synthesis of [2-(Methacryloyloxy)ethyl]trimethylammonium ρ-aminosalicylate (ChMAPAS)
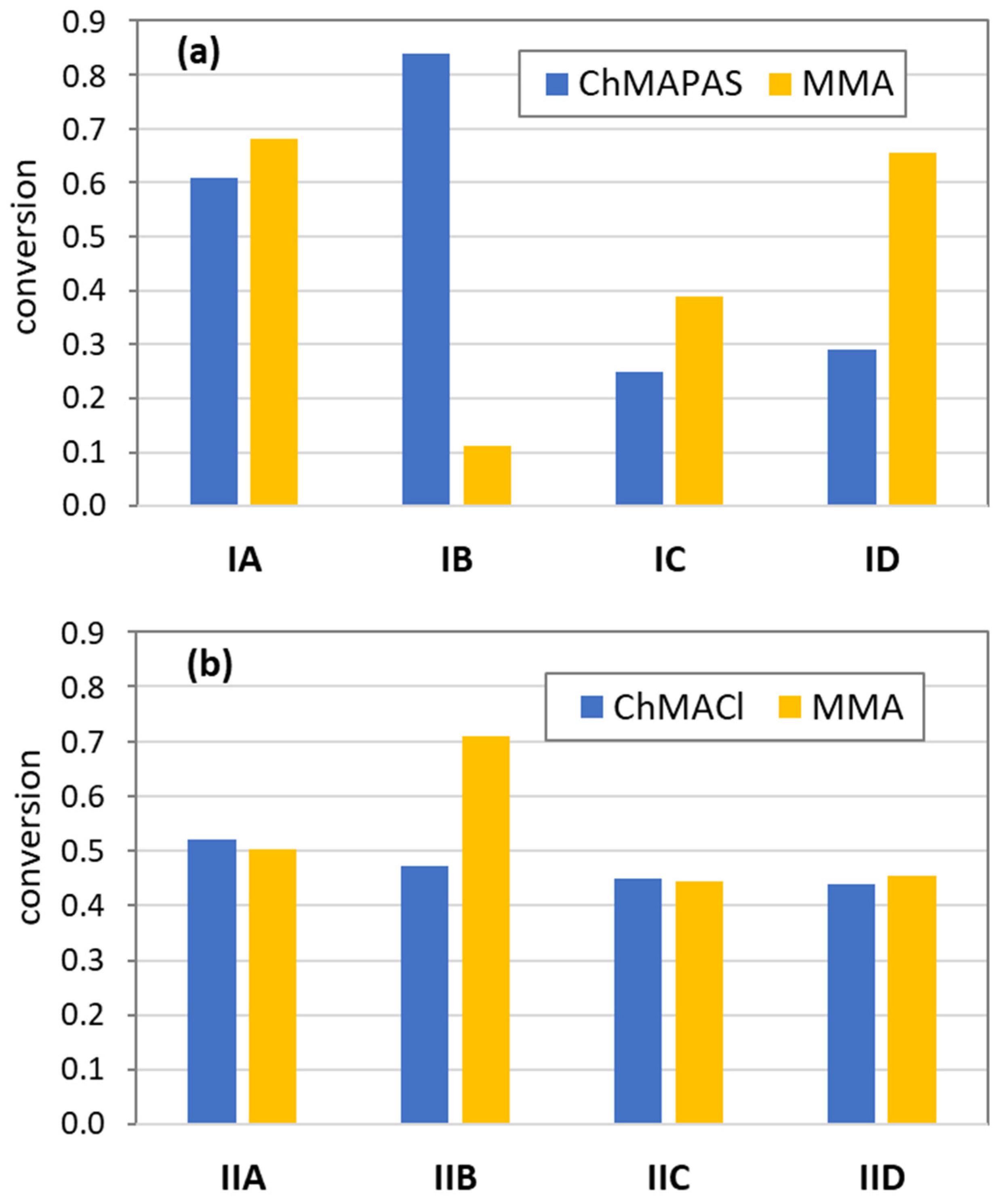
2.2. Synthesis of Linear Copolymer P(ChMAPAS-co-MMA) by ATRP (an Example for ChMAPAS/MMA (25/75) IA)
2.3. Synthesis of Linear Copolymer P(ChMACl-co-MMA) by ATRP (an Example for ChMACl/MMA (25/75) IIA)
2.4. Drug Release Studies of Pharmaceutical Anion (PAS) by Polymer Carrier
2.5. Characterization
3. Results and Discussion
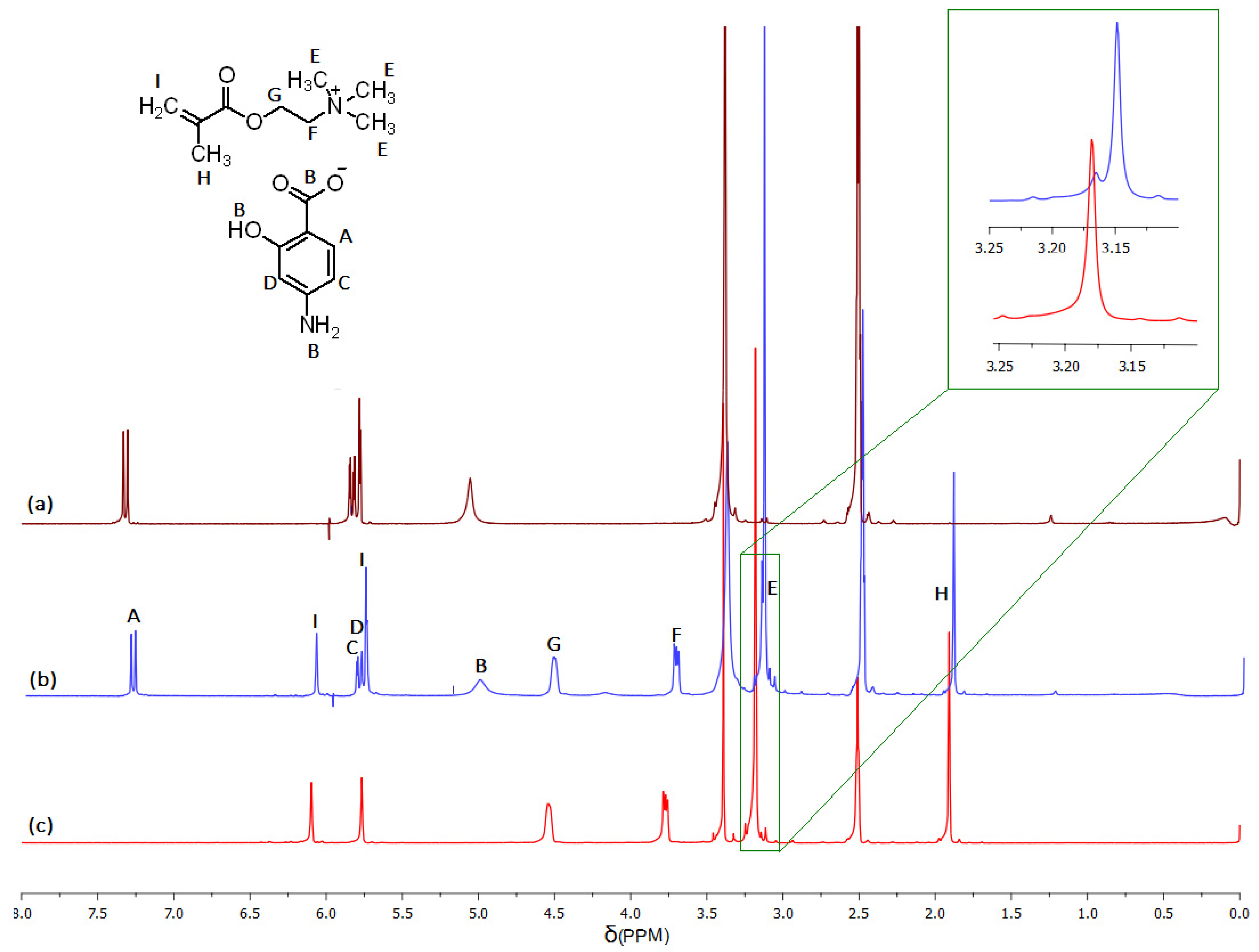
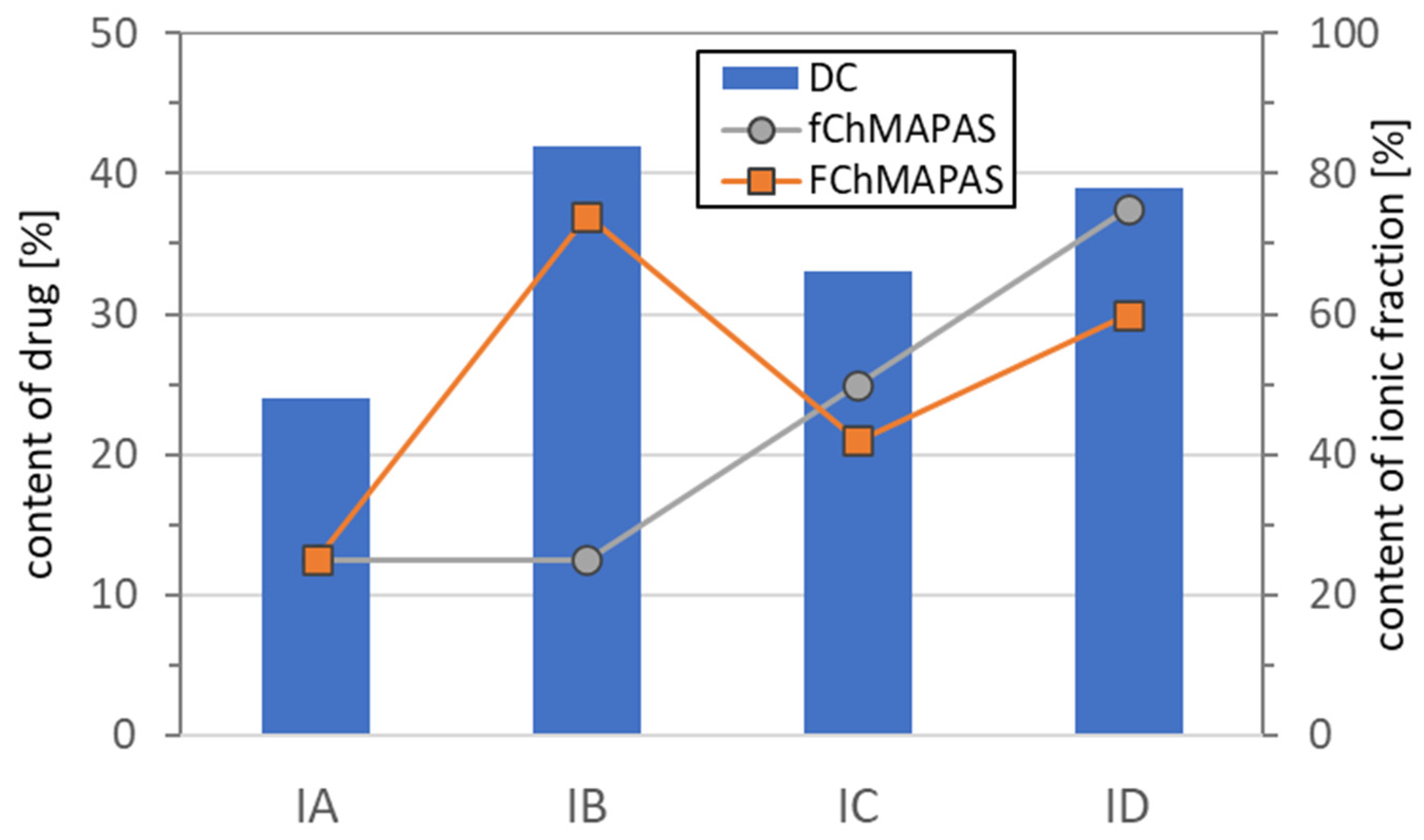
3.1. Synthesis of the Pharmaceutic Functionalized Monomer by Anion Exchange with PAS Salt
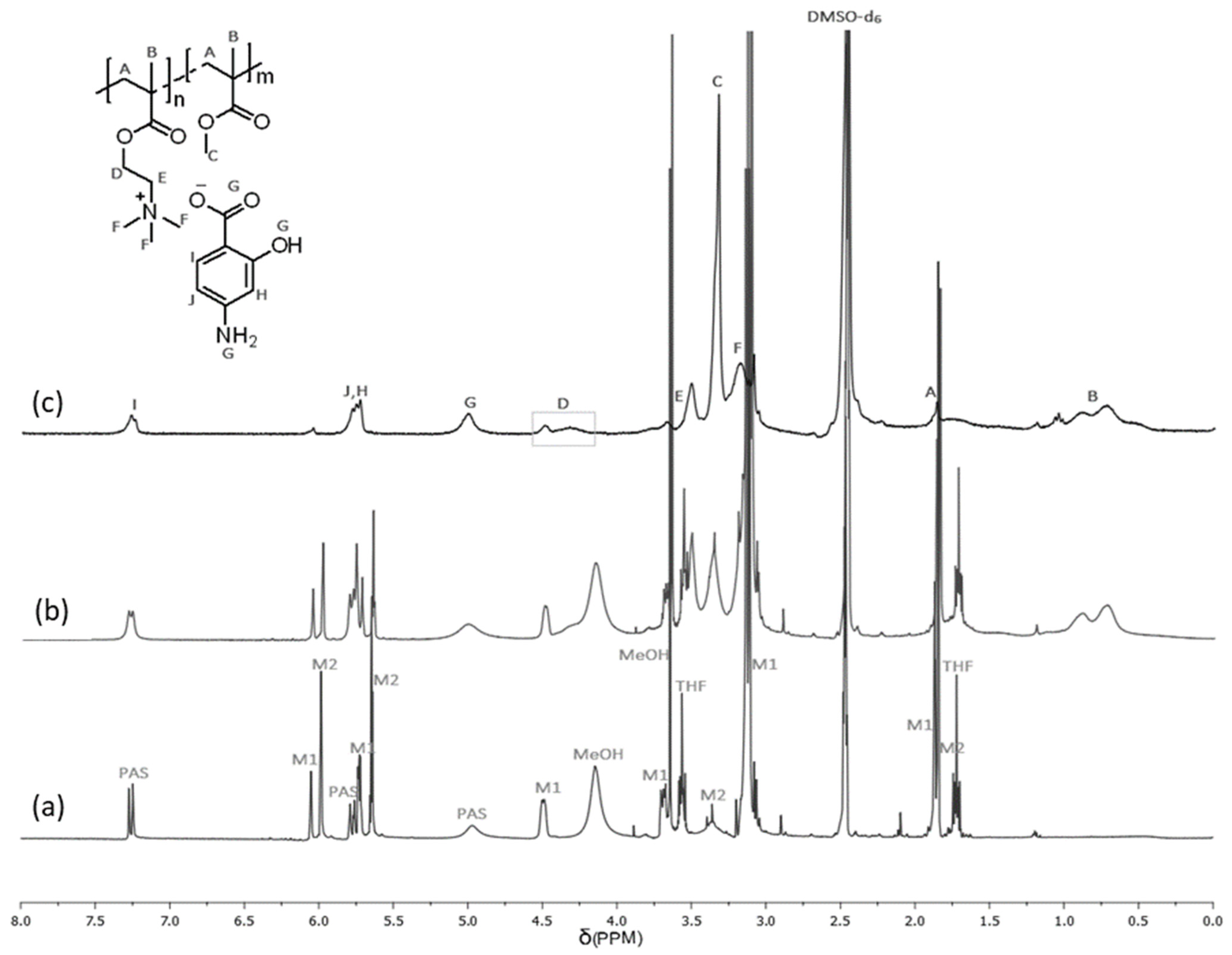
3.2. Characterization of Linear Copolymers Obtained from ChMAPAS
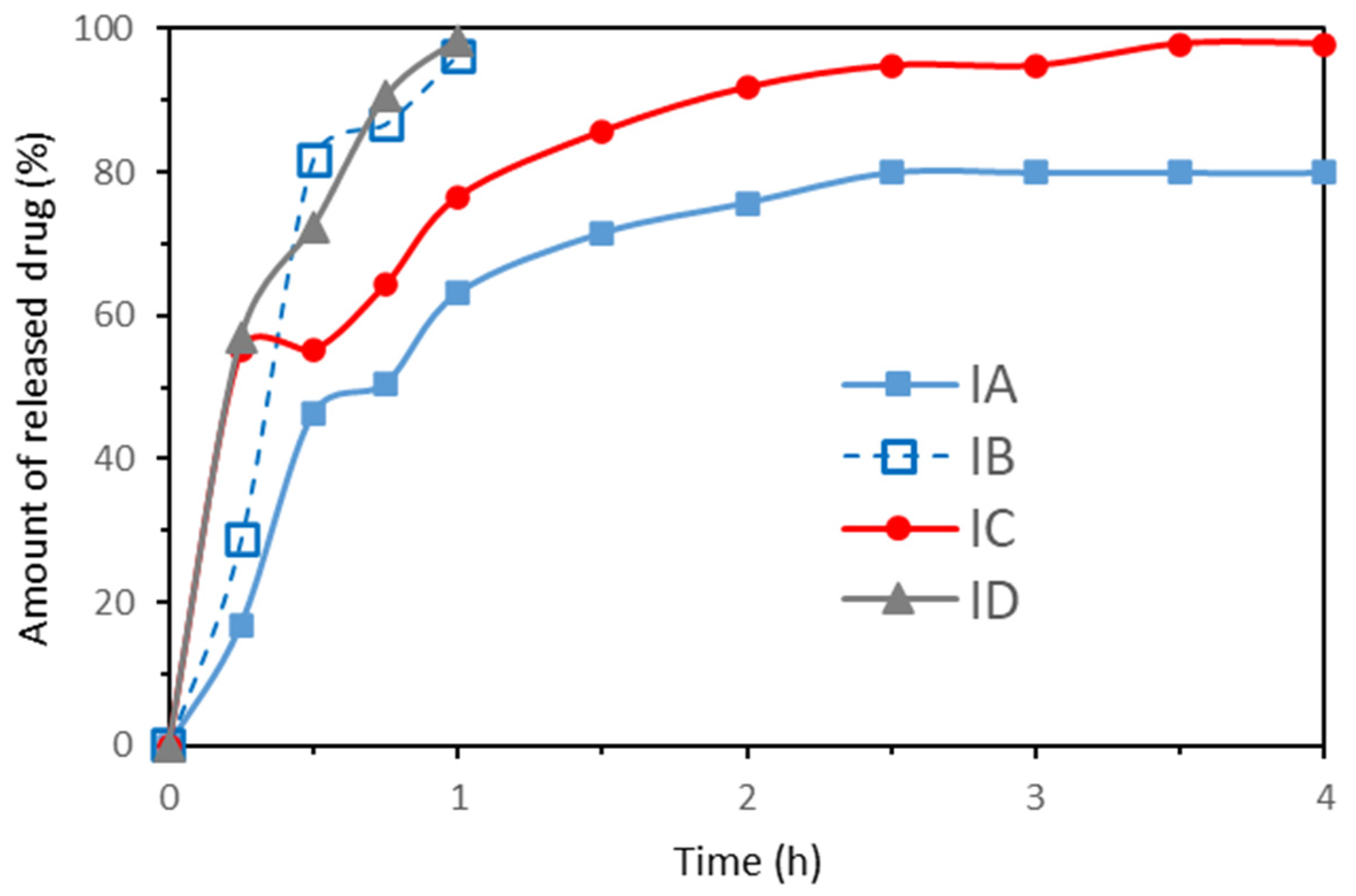
3.3. Drug Release
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Coelho, J.F.; Ferreira, P.C.; Alves, P.; Cordeiro, R.; Fonseca, A.C.; Góis, J.R.; Gil, M.H. Drug delivery systems: Advanced technologies potentially applicable in personalized treatments. EPMA J. 2010, 1, 164–209. [Google Scholar] [CrossRef]
- Zhang, Y.; Chan, H.F.; Leong, K.W. Advanced materials and processing for drug delivery: The past and the future. Adv. Drug Deliv. Rev. 2013, 65, 104–120. [Google Scholar] [CrossRef] [PubMed]
- Gaurav, I.; Thakur, A.; Iyaswamy, A.; Wang, X.; Chen, X.; Yang, Z. Factors affecting extracellular vesicles based drug delivery systems. Molecules 2021, 26, 1544. [Google Scholar] [CrossRef] [PubMed]
- Jain, K.K. Drug Delivery Systems; Springer: Berlin/Heidelberg, Germany, 2008; Volume 251. [Google Scholar]
- Visser, J.G.; Van Staden, A.D.P.; Smith, C. Harnessing macrophages for controlled-release drug delivery: Lessons from microbes. Front. Pharmacol. 2019, 10, 22. [Google Scholar] [CrossRef] [PubMed]
- Kingsley, J.D.; Dou, H.; Morehead, J.; Rabinow, B.; Gendelman, H.E.; Destache, C.J. Nanotechnology: A focus on nanoparticles as a drug delivery system. J. Neuroimmune Pharmacol. 2006, 1, 340–350. [Google Scholar] [CrossRef] [PubMed]
- Felice, B.; Prabhakaran, M.P.; Rodriguez, A.P.; Ramakrishna, S. Drug delivery vehicles on a nano-engineering perspective. Mater. Sci. Eng. C 2014, 41, 178–195. [Google Scholar] [CrossRef]
- Pandey, R.; Khuller, G. Polymer based drug delivery systems for mycobacterial infections. Curr. Drug Deliv. 2004, 1, 195–201. [Google Scholar] [CrossRef]
- Kadajji, V.G.; Betageri, G.V. Water soluble polymers for pharmaceutical applications. Polymers 2011, 3, 1972–2009. [Google Scholar] [CrossRef]
- Vega-Vásquez, P.; Mosier, N.S.; Irudayaraj, J. Nanoscale drug delivery systems: From medicine to agriculture. Front. Bioeng. Biotechnol. 2020, 8, 79. [Google Scholar] [CrossRef]
- Neugebauer, D.; Odrobińska, J.; Bielas, R.; Mielańczyk, A. Design of systems based on 4-armed star-shaped polyacids for indomethacin delivery. New J. Chem. 2016, 40, 10002–10011. [Google Scholar] [CrossRef]
- Mielanczyk, A.; Neugebauer, D. Designing drug conjugates based on sugar decorated V-shape and star polymethacrylates: Influence of composition and architecture of polymeric carrier. Bioconjug. Chem. 2015, 26, 2303–2310. [Google Scholar] [CrossRef] [PubMed]
- Bury, K.; Du Prez, F.; Neugebauer, D. Self-assembling Linear and Star Shaped Poly (ϵ-caprolactone)/poly [(meth) acrylic acid] Block Copolymers as Carriers of Indomethacin and Quercetin. Macromol. Biosci. 2013, 13, 1520–1530. [Google Scholar] [CrossRef]
- Maksym, P.; Neugebauer, D. Self-assembling polyether-b-polymethacrylate graft copolymers loaded with indomethacin. Int. J. Polym. Mater. Polym. Biomater. 2017, 66, 317–325. [Google Scholar] [CrossRef]
- Maksym, P.; Neugebauer, D. Synthesis of amphiphilic semigrafted pseudo-Pluronics for self-assemblies carrying indomethacin. RSC Adv. 2016, 6, 88444–88452. [Google Scholar] [CrossRef]
- Bury, K.; Neugebauer, D. Novel self-assembly graft copolymers as carriers for anti-inflammatory drug delivery. Int. J. Pharmaceut. 2014, 460, 150–157. [Google Scholar] [CrossRef]
- Golan, D.E.; Tashjian, A.H.; Armstrong, E.J. Principles of Pharmacology: The Pathophysiologic Basis of Drug Therapy; Wolters Kluwer Health/Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2008; Volume 396, pp. 235–258. [Google Scholar]
- Li, D.C.; Zhong, X.K.; Zeng, Z.P.; Jiang, J.G.; Li, L.; Zhao, M.M.; Yang, X.Q.; Chen, J.; Zhang, B.S.; Zhao, Q.; et al. Application of targeted drug delivery system in Chinese medicine. J. Control. Release 2009, 138, 103–112. [Google Scholar] [CrossRef]
- Sung, Y.K.; Kim, S.W. Recent advances in polymeric drug delivery systems. Biomater. Res. 2020, 24, 1–12. [Google Scholar] [CrossRef]
- Chen, K.; Liao, S.; Guo, S.; Zheng, X.; Wang, B.; Duan, Z.; Zhang, H.; Gong, Q.; Luo, K. Multistimuli-responsive PEGylated polymeric bioconjugate-based nano-aggregate for cancer therapy. Chem. Eng. J. 2020, 391, 123543. [Google Scholar] [CrossRef]
- Ofridam, F.; Ofridam, F.; Tarhini, M.; Lebaz, N.; Gagnière, É.; Mangin, D.; Elaissari, A. pH-sensitive polymers: Classification and some fine potential applications. Polym. Adv. Technol. 2021, 32, 1455–1484. [Google Scholar] [CrossRef]
- Ravi Kumar, M.N.; Kumar, N. §, Polymeric controlled drug-delivery systems: Perspective issues and opportunities. Drug Dev. Ind. Pharm. 2001, 27, 1–30. [Google Scholar] [CrossRef]
- Bhowmik, D.; Gopinath, H.; Kumar, B.P.; Duraivel, S.; Kumar, K.S. Controlled release drug delivery systems. Pharma Innov. 2012, 1, 10. [Google Scholar]
- Yessine, M.A.; Lafleur, M.; Meier, C.; Petereit, H.U.; Leroux, J.C. Characterization of the membrane-destabilizing properties of different pH-sensitive methacrylic acid copolymers. Biochim. Et Biophys. Acta (BBA)-Biomembr. 2003, 1613, 28–38. [Google Scholar] [CrossRef]
- Lu, J.; Yan, F.; Texter, J. Advanced applications of ionic liquids in polymer science. Prog. Polym. Sci. 2009, 34, 431–448. [Google Scholar] [CrossRef]
- Kowsari, E. Ionic Liquids: Applications and Perspectives; BoD–Books on Demand: Norderstedt, Germany, 2011; pp. 2–28. [Google Scholar]
- Taha, M.; Almeida, M.R.; Silva, F.A.E.; Domingues, P.; Ventura, S.P.; Coutinho, J.A.; Freire, M.G. Novel biocompatible and self-buffering ionic liquids for biopharmaceutical applications. Chem. A Eur. J. 2015, 21, 4781–4788. [Google Scholar] [CrossRef]
- Fukaya, Y.; Iizuka, Y.; Sekikawa, K.; Ohno, H. Bio ionic liquids: Room temperature ionic liquids composed wholly of biomaterials. Green Chem. 2007, 9, 1155–1157. [Google Scholar] [CrossRef]
- Araújo, J.M.; Florindo, C.; Pereiro, A.B.; Vieira, N.S.; Matias, A.A.; Duarte, C.M.; Rebelo, L.P.N.; Marrucho, I.M. Cholinium-based ionic liquids with pharmaceutically active anions. RSC Adv. 2014, 4, 28126–28132. [Google Scholar] [CrossRef]
- Li, X.; Ma, N.; Zhang, L.; Ling, G.; Zhang, P. Applications of choline-based ionic liquids in drug delivery. Int. J. Pharm. 2021, 612, 121366. [Google Scholar] [CrossRef]
- Bielas, R.; Bielas, R.; Mielańczyk, A.; Siewniak, A.; Neugebauer, D. Trimethylammonium-based polymethacrylate ionic liquids with tunable hydrophilicity and charge distribution as carriers of salicylate anions. ACS Sustain. Chem. Eng. 2016, 4, 4181–4191. [Google Scholar] [CrossRef]
- Bielas, R.; Siewniak, A.; Skonieczna, M.; Adamiec, M.; Mielańczyk, Ł.; Neugebauer, D. Choline based polymethacrylate matrix with pharmaceutical cations as co-delivery system for antibacterial and anti-inflammatory combined therapy. J. Mol. Liq. 2019, 285, 114–122. [Google Scholar] [CrossRef]
- Niesyto, K.; Neugebauer, D. Synthesis and characterization of ionic graft copolymers: Introduction and in vitro release of antibacterial drug by anion exchange. Polymers 2020, 12, 2159. [Google Scholar] [CrossRef] [PubMed]
- Niesyto, K.; Neugebauer, D. Linear Copolymers Based on Choline Ionic Liquid Carrying Anti-Tuberculosis Drugs: Influence of Anion Type on Physicochemical Properties and Drug Release. Int. J. Mol. Sci. 2020, 22, 284. [Google Scholar] [CrossRef] [PubMed]
- Niesyto, K.; Łyżniak, W.; Skonieczna, M.; Neugebauer, D. Biological In Vitro Evaluation of PIL Graft Conjugates: Cytotoxicity Characteristics. Int. J. Mol. Sci. 2021, 22, 7741. [Google Scholar] [CrossRef] [PubMed]
- Niesyto, K.; Mazur, A.; Neugebauer, D. Dual-Drug Delivery via the Self-Assembled Conjugates of Choline-Functionalized Graft Copolymers. Materials 2022, 15, 4457. [Google Scholar] [CrossRef] [PubMed]
- Bielas, R.; Łukowiec, D.; Neugebauer, D. Drug delivery via anion exchange of salicylate decorating poly (meth) acrylates based on a pharmaceutical ionic liquid. New J. Chem. 2017, 41, 12801–12807. [Google Scholar] [CrossRef]
- Bielas, R.; Bielas, R.; Mielańczyk, A.; Skonieczna, M.; Mielańczyk, Ł.; Neugebauer, D. supported poly (ionic liquid) graft copolymers as novel delivery systems of anionic pharmaceuticals for anti-inflammatory and anti-coagulant therapy. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Donald, P.R.; Diacon, A.H. Para-aminosalicylic acid: The return of an old friend. Lancet Infect. Dis. 2015, 15, 1091–1099. [Google Scholar] [CrossRef]
- Minato, Y.; Thiede, J.M.; Kordus, S.L.; McKlveen, E.J.; Turman, B.J.; Baughn, A.D. Mycobacterium tuberculosis folate metabolism and the mechanistic basis for para-aminosalicylic acid susceptibility and resistance. Antimicrob. Agents Chemother. 2015, 59, 5097–5106. [Google Scholar] [CrossRef]
- Campregher, C.; Gasche, C. Aminosalicylates. Best Pract. Res. Clin. Gastroenterol. 2011, 25, 535–546. [Google Scholar] [CrossRef]



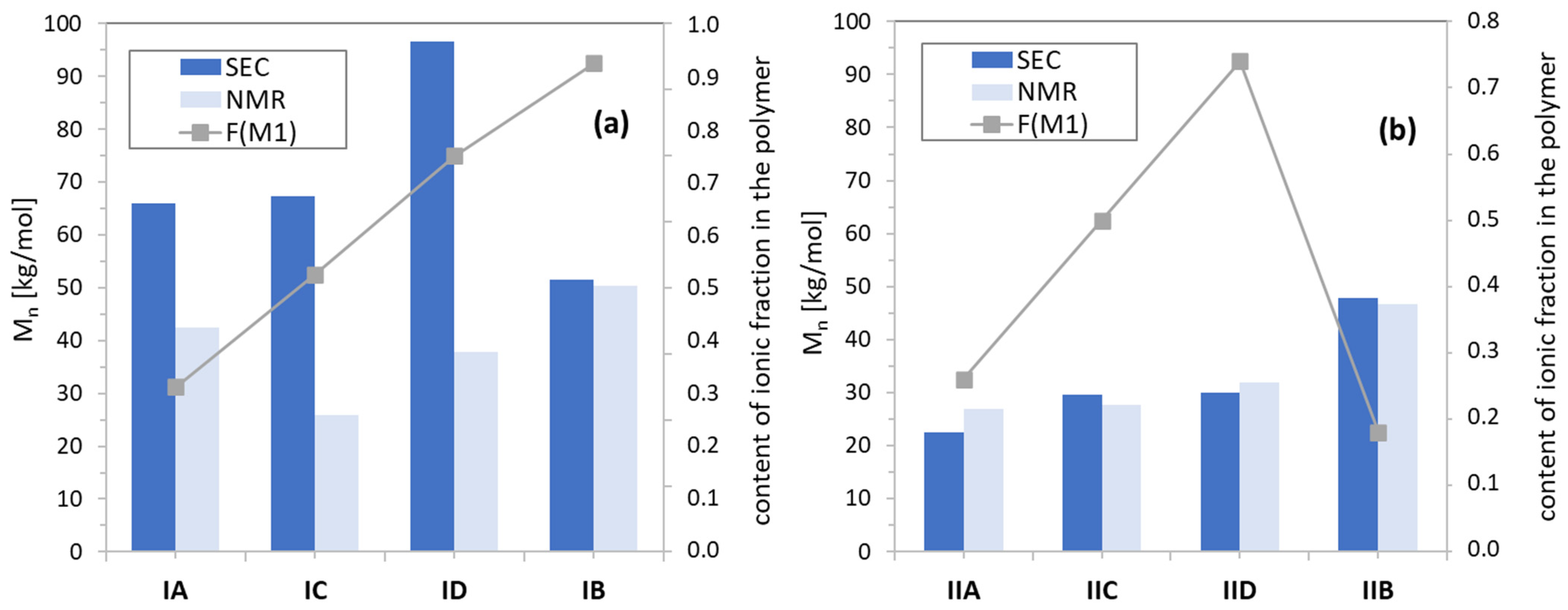
| 1H NMR | SEC | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| No | fM1/fM2 mol (%) | Time (h) | XM1 (%) | X (%) | DPM1 | DPn | FM1 (%) | Mn (g/mol) | Mn (g/mol) | Ð |
| IA | 25/75 | 0.66 | 0.61 | 0.66 | 68 | 272 | 0.25 | 42,500 | 65,900 | 1.29 |
| IIA | 1.5 | 0.52 | 0.51 | 52 | 203 | 0.26 | 26,900 | 22,400 | 1.26 | |
| IB | 25/75 | 19 | 0.84 | 0.31 | 139 | 190 | 0.74 | 50,300 | 51,600 | 1.33 |
| IIB | 5 | 0.47 | 0.65 | 71 | 390 | 0.18 | 46,700 | 47,900 | 1.12 | |
| IC | 50/50 | 0.91 | 0.25 | 0.32 | 56 | 133 | 0.42 | 25,800 | 67,300 | 1.25 |
| IIC | 1.7 | 0.45 | 0.45 | 90 | 179 | 0.50 | 27,600 | 29,600 | 1.13 | |
| ID | 75/25 | 0.73 | 0.29 | 0.37 | 97 | 162 | 0.60 | 37,900 | 96,600 | 1.36 |
| IID | 2.1 | 0.44 | 0.44 | 132 | 178 | 0.74 | 31,900 | 29,900 | 1.14 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Keihankhadiv, S.; Neugebauer, D. Synthesis and Characterization of Linear Copolymers Based on Pharmaceutically Functionalized Monomeric Choline Ionic Liquid for Delivery of p-Aminosalicylate. Pharmaceutics 2023, 15, 860. https://doi.org/10.3390/pharmaceutics15030860
Keihankhadiv S, Neugebauer D. Synthesis and Characterization of Linear Copolymers Based on Pharmaceutically Functionalized Monomeric Choline Ionic Liquid for Delivery of p-Aminosalicylate. Pharmaceutics. 2023; 15(3):860. https://doi.org/10.3390/pharmaceutics15030860
Chicago/Turabian StyleKeihankhadiv, Shadi, and Dorota Neugebauer. 2023. "Synthesis and Characterization of Linear Copolymers Based on Pharmaceutically Functionalized Monomeric Choline Ionic Liquid for Delivery of p-Aminosalicylate" Pharmaceutics 15, no. 3: 860. https://doi.org/10.3390/pharmaceutics15030860
APA StyleKeihankhadiv, S., & Neugebauer, D. (2023). Synthesis and Characterization of Linear Copolymers Based on Pharmaceutically Functionalized Monomeric Choline Ionic Liquid for Delivery of p-Aminosalicylate. Pharmaceutics, 15(3), 860. https://doi.org/10.3390/pharmaceutics15030860








