Pharmacokinetic Characterization and External Evaluation of a Quantitative Framework of Sublingual Buprenorphine in Patients with an Opioid Disorder in Puerto Rico
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Procedures
2.2. Assay Methodology
2.3. Non-Compartmental Pharmacokinetic Analysis
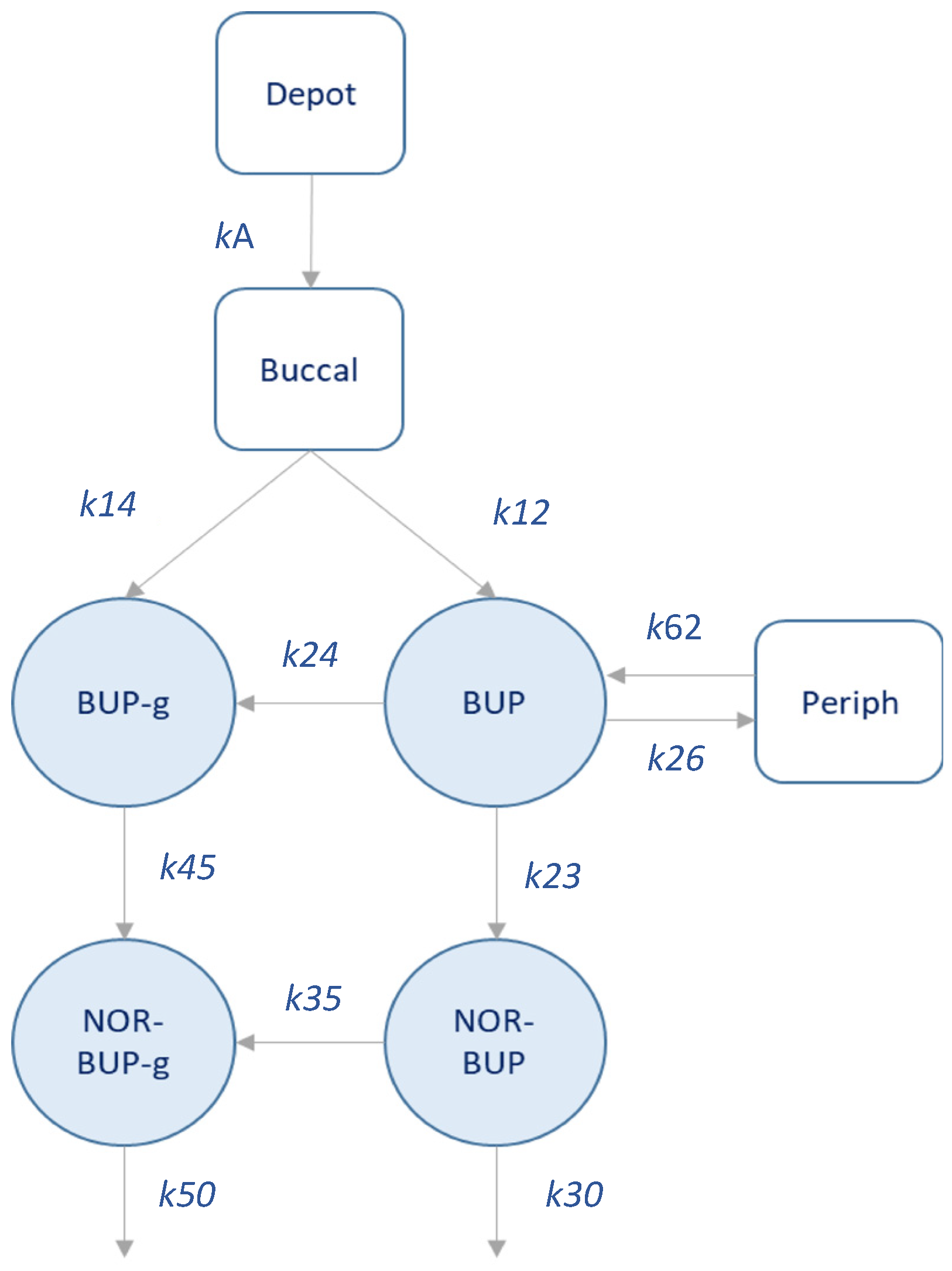
2.4. Population Pharmacokinetic Analysis
3. Results
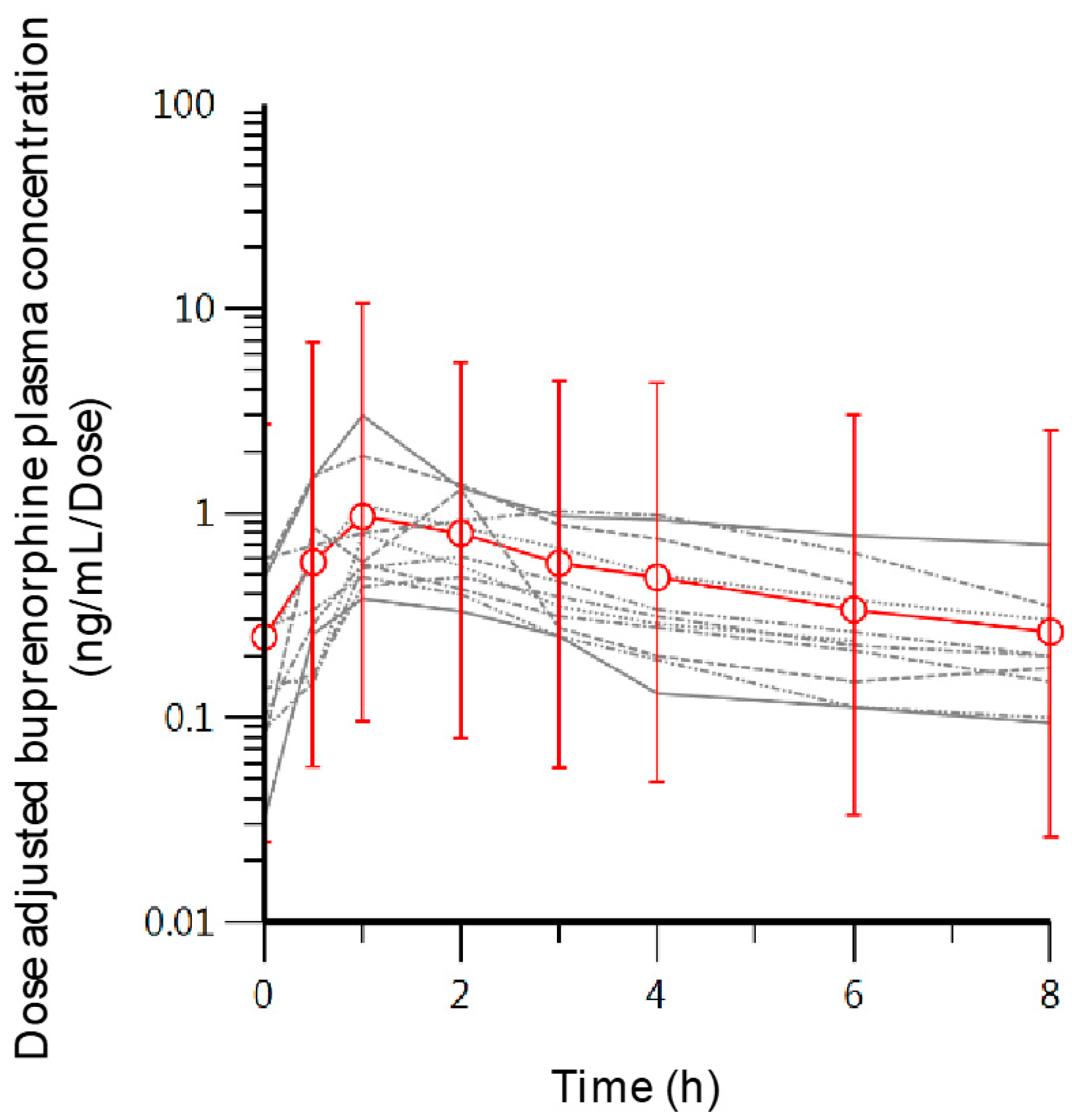
3.1. Non-Compartmental PK Analysis
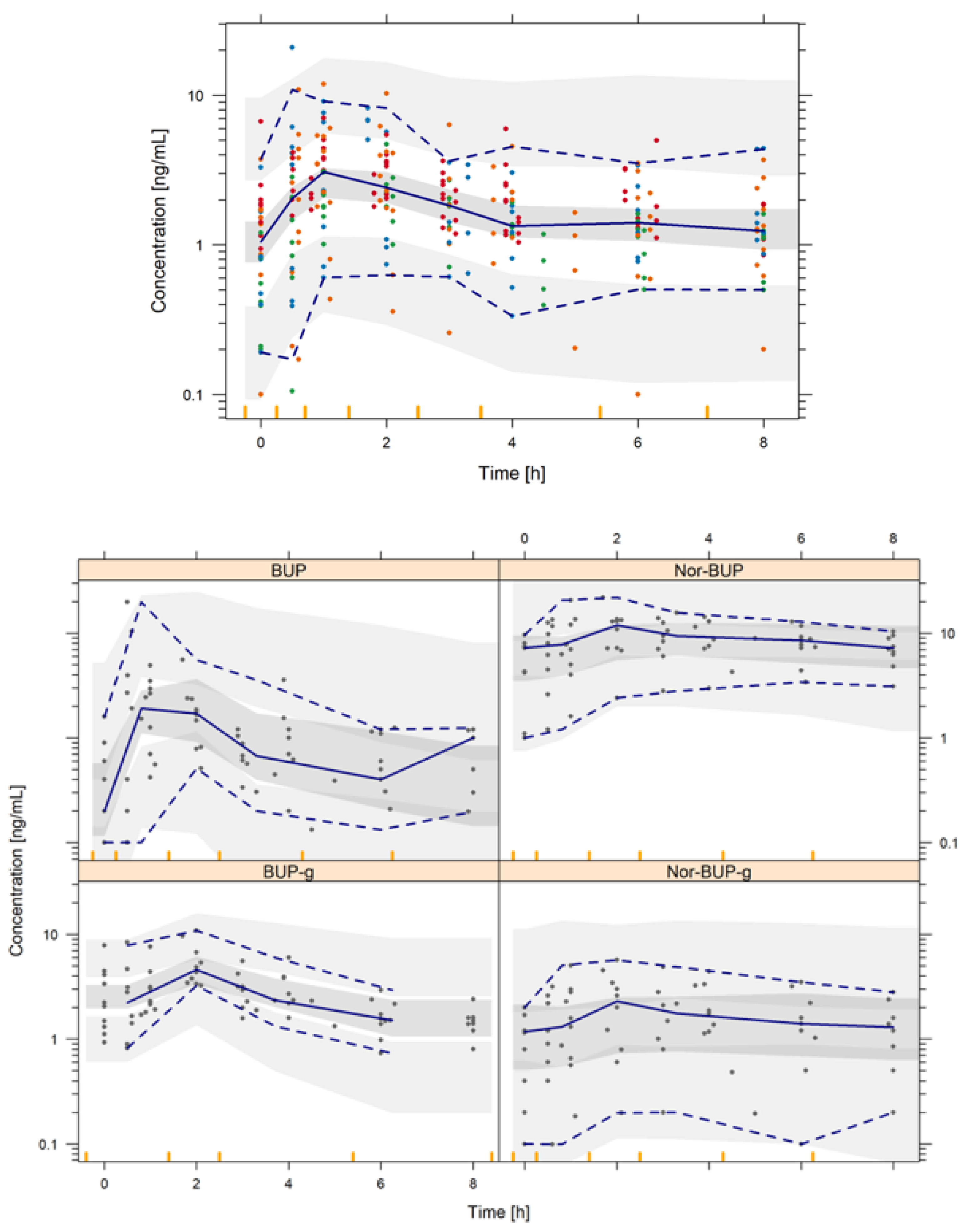
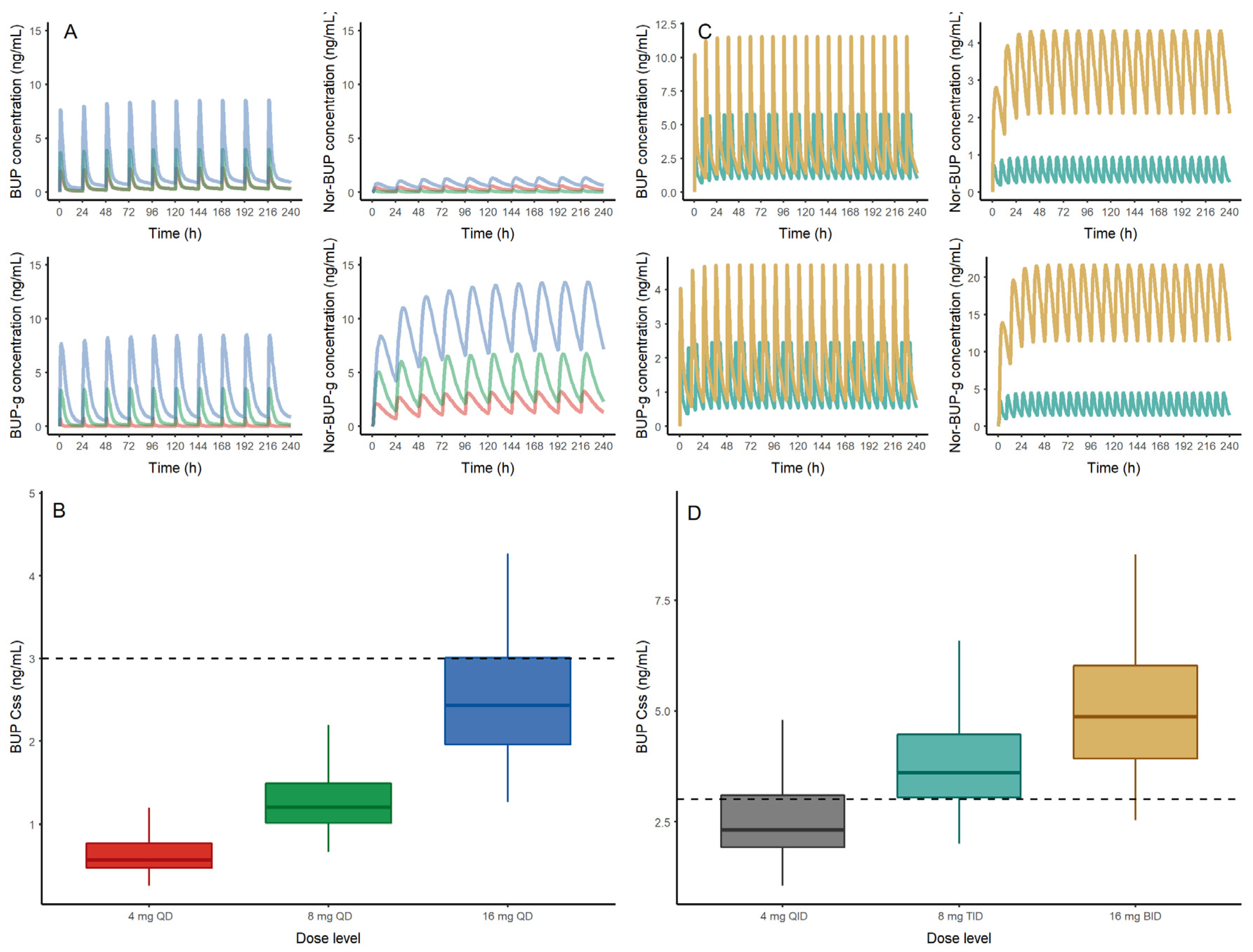
3.2. Population PK Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- National Institute on Drug Abuse Overdose Death Rates. Available online: https://www.drugabuse.gov/related-topics/trends-statistics/overdose-death-rates (accessed on 20 September 2018).
- Coto, D. Puerto Rico: Crisis de los Opioides se Siente con Fuerza. Available online: https://www.sun-sentinel.com/elsentinel/fl-es-crisis-opioides-con-fuerza-puerto-rico-20190107-story.html (accessed on 9 January 2020).
- US Department of Health and Human Services; Substance Abuse and Mental Health Services Administration; Center for Substance Abuse Treatment. Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction: A Treatment Improvement Protocol (TIP 40); SAMHSA: Rockville, MD, USA, 2004. [Google Scholar]
- FDA; CDER. Suboxone Food and Drug Administration (FDA). Center for Drug Evaluation and Research (CDER). Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2010/022410Orig1s000Lbl.pdf (accessed on 2 December 2020).
- Comer, S.; Cunningham, C.; Fishman, M.J.; Gordon, A.; Kampman, K.; Langleben, D.; Nordstrom, B.; Oslin, D.; Woody, G.; Wright, T.; et al. ASAM National Practice Guideline for the Use of Medications in the Treatment of Addiction Involving Opioid Use; ASAM: Rockville, MD, USA, 2015. [Google Scholar]
- Bruneau, J.; Ahamad, K.; Goyer, M.-È.; Poulin, G.; Selby, P.; Fischer, B.; Wild, T.C.; Wood, E. CIHR Management of opioid use disorders: A national clinical practice guideline. CMAJ 2018, 190, E247–E257. [Google Scholar] [CrossRef]
- Bonhomme, J.; Shim, R.S.; Gooden, R.; Tyus, D.; Rust, G. Opioid addiction and abuse in primary care practice: A comparison of methadone and buprenorphine as treatment options. J. Natl. Med. Assoc. 2012, 104, 342–350. [Google Scholar] [CrossRef]
- Piper, B.J.; Shah, D.T.; Simoyan, O.M.; McCall, K.L.; Nichols, S.D. Trends in Medical Use of Opioids in the US, 2006–2016. Am. J. Prev. Med. 2018, 54, 652–660. [Google Scholar] [CrossRef]
- Greenwald, M.; Johanson, C.-E.; Bueller, J.; Chang, Y.; Moody, D.E.; Kilbourn, M.; Koeppe, R.; Zubieta, J.-K. Buprenorphine duration of action: Mu-opioid receptor availability and pharmacokinetic and behavioral indices. Biol. Psychiatry 2007, 61, 101–110. [Google Scholar] [CrossRef]
- Greenwald, M.K.; Johanson, C.-E.; Moody, D.E.; Woods, J.H.; Kilbourn, M.R.; Koeppe, R.A.; Schuster, C.R.; Zubieta, J.-K. Effects of buprenorphine maintenance dose on mu-opioid receptor availability, plasma concentrations, and antagonist blockade in heroin-dependent volunteers. Neuropsychopharmacology 2003, 28, 2000–2009. [Google Scholar] [CrossRef] [PubMed]
- Greenwald, M.K.; Comer, S.D.; Fiellin, D.A. Buprenorphine maintenance and mu-opioid receptor availability in the treatment of opioid use disorder: Implications for clinical use and policy. Drug Alcohol Depend. 2014, 144, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ng, C.M.; Dombrowsky, E.; Lin, H.; Erlich, M.E.; Moody, D.E.; Barrett, J.S.; Kraft, W.K. Population Pharmacokinetic Model of Sublingual Buprenorphine in Neonatal Abstinence Syndrome. Pharmacotherapy 2015, 35, 670–680. [Google Scholar] [CrossRef] [PubMed]
- Nasser, A.F.; Heidbreder, C.; Gomeni, R.; Fudala, P.J.; Zheng, B.; Greenwald, M.K. A population pharmacokinetic and pharmacodynamic modelling approach to support the clinical development of RBP-6000, a new, subcutaneously injectable, long-acting, sustained-release formulation of buprenorphine, for the treatment of opioid dependence. Clin. Pharmacokinet. 2014, 53, 813–824. [Google Scholar] [CrossRef]
- Dong, R.; Wang, H.; Li, D.; Lang, L.; Gray, F.; Liu, Y.; Laffont, C.M.; Young, M.; Jiang, J.; Liu, Z.; et al. Pharmacokinetics of Sublingual Buprenorphine Tablets Following Single and Multiple Doses in Chinese Participants with and without Opioid Use Disorder. Drugs R D 2019, 19, 255–265. [Google Scholar] [CrossRef]
- Moody, D.E.; Fang, W.B.; Morrison, J.; McCance-Katz, E. Gender differences in pharmacokinetics of maintenance dosed buprenorphine. Drug Alcohol Depend. 2011, 118, 479–483. [Google Scholar] [CrossRef]
- Bastian, J.R.; Chen, H.; Zhang, H.; Rothenberger, S.; Tarter, R.; English, D.; Venkataramanan, R.; Caritis, S.N. Dose-adjusted plasma concentrations of sublingual buprenorphine are lower during than after pregnancy. Am. J. Obstet. Gynecol. 2017, 216, 64.e1–64.e7. [Google Scholar] [CrossRef] [PubMed]
- Food and Drug Administration. Analytical Procedures and Methods Validation for Drugs and-Biologics; FDA: Silver Spring, MD, USA, 2015. [Google Scholar]
- Zhang, H.; Bastian, J.R.; Zhao, W.; Chen, H.; Caritis, S.N.; Shaik, I.; Chaphekar, N.; Venkataramanan, R. Pregnancy Alters CYP and UGT Mediated Metabolism of Buprenorphine. Ther. Drug Monit. 2020, 42, 264–270. [Google Scholar] [CrossRef] [PubMed]
- Bergstrand, M.; Hooker, A.C.; Wallin, J.E.; Karlsson, M.O. Prediction-corrected visual predictive checks for diagnosing nonlinear mixed-effects models. AAPS J. 2011, 13, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Lindbom, L.; Pihlgren, P.; Jonsson, E.N. PsN-Toolkit--a collection of computer intensive statistical methods for non-linear mixed effect modeling using NONMEM. Comput. Methods Programs Biomed. 2005, 79, 241–257. [Google Scholar] [CrossRef]
- Jönsson, M.; Mundin, G.; Sumner, M. Pharmacokinetic and pharmaceutical properties of a novel buprenorphine/naloxone sublingual tablet for opioid substitution therapy versus conventional buprenorphine/naloxone sublingual tablet in healthy volunteers. Eur. J. Pharm. Sci. 2018, 122, 125–133. [Google Scholar] [CrossRef]
- Harris, D.S.; Mendelson, J.E.; Lin, E.T.; Upton, R.A.; Jones, R.T. Pharmacokinetics and subjective effects of sublingual buprenorphine, alone or in combination with naloxone: Lack of dose proportionality. Clin. Pharmacokinet. 2004, 43, 329–340. [Google Scholar] [CrossRef]
- McAleer, S.D.; Mills, R.J.; Polack, T.; Hussain, T.; Rolan, P.E.; Gibbs, A.D.; Mullins, F.G.P.; Hussein, Z. Pharmacokinetics of high-dose buprenorphine following single administration of sublingual tablet formulations in opioid naïve healthy male volunteers under a naltrexone block. Drug Alcohol Depend. 2003, 72, 75–83. [Google Scholar] [CrossRef]
- Ciraulo, D.A.; Hitzemann, R.J.; Somoza, E.; Knapp, C.M.; Rotrosen, J.; Sarid-Segal, O.; Ciraulo, A.M.; Greenblatt, D.J.; Chiang, C.N. Pharmacokinetics and pharmacodynamics of multiple sublingual buprenorphine tablets in dose-escalation trials. J. Clin. Pharmacol. 2006, 46, 179–192. [Google Scholar] [CrossRef]
- Compton, P.; Ling, W.; Moody, D.; Chiang, N. Pharmacokinetics, bioavailability and opioid effects of liquid versus tablet buprenorphine. Drug Alcohol Depend. 2006, 82, 25–31. [Google Scholar] [CrossRef]
- Kuhlman, J.J., Jr.; Lalani, S.; Magluilo, J., Jr.; Levine, B.; Darwin, W.D. Human pharmacokinetics of intravenous, sublingual, and buccal buprenorphine. J. Anal. Toxicol. 1996, 20, 369–378. [Google Scholar] [CrossRef]
- Food and Drug Administration (FDA)-Center for drug evaluation and research (CDER). Approval Package for Suboxone. Available online: www.accessdata.fda.gov/drugsatfda_docs/nda/2012/022410Orig1s006.pdf (accessed on 5 May 2020).
- Chiang, C.N.; Hawks, R.L. Pharmacokinetics of the combination tablet of buprenorphine and naloxone. Drug Alcohol Depend. 2003, 70, S39–S47. [Google Scholar] [CrossRef]
- Bullingham, R.E.; McQuay, H.J.; Porter, E.J.; Allen, M.C.; Moore, R.A. Sublingual buprenorphine used postoperatively: Ten hour plasma drug concentration analysis. Br. J. Clin. Pharmacol. 1982, 13, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Schuh, K.J.; Johanson, C.E. Pharmacokinetic comparison of the buprenorphine sublingual liquid and tablet. Drug Alcohol Depend. 1999, 56, 55–60. [Google Scholar] [CrossRef]
- Gruber, V.A.; Rainey, P.M.; Moody, D.E.; Morse, G.D.; Ma, Q.; Prathikanti, S.; Pade, P.A.; Alvanzo, A.A.H.; McCance-Katz, E.F. Interactions between buprenorphine and the protease inhibitors darunavir-ritonavir and fosamprenavir-ritonavir. Clin. Infect. Dis. 2012, 54, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Cone, E.J.; Gorodetzky, C.W.; Yousefnejad, D.; Buchwald, W.F.; Johnson, R.E. The metabolism and excretion of buprenorphine in humans. Drug Metab. Dispos. 1984, 12, 577–581. [Google Scholar]
- Yassen, A.; Olofsen, E.; Romberg, R.; Sarton, E.; Danhof, M.; Dahan, A. Mechanism-based pharmacokinetic-pharmacodynamic modeling of the antinociceptive effect of buprenorphine in healthy volunteers. Anesthesiology 2006, 104, 1232–1242. [Google Scholar] [CrossRef]
- Yassen, A.; Olofsen, E.; Romberg, R.; Sarton, E.; Teppema, L.; Danhof, M.; Dahan, A. Mechanism-based PK/PD modeling of the respiratory depressant effect of buprenorphine and fentanyl in healthy volunteers. Clin. Pharmacol. Ther. 2007, 81, 50–58. [Google Scholar] [CrossRef]
- Priestley, T.; Chappa, A.K.; Mould, D.R.; Upton, R.N.; Shusterman, N.; Passik, S.; Tormo, V.J.; Camper, S. Converting from Transdermal to Buccal Formulations of Buprenorphine: A Pharmacokinetic Meta-Model Simulation in Healthy Volunteers. Pain Med. 2018, 19, 1988–1996. [Google Scholar] [CrossRef]
- Doodnaught, G.M.; Monteiro, B.P.; Benito, J.; Edge, D.; Beaudry, F.; Pelligand, L.; Steagall, P. Pharmacokinetic and pharmacodynamic modelling after subcutaneous, intravenous and buccal administration of a high-concentration formulation of buprenorphine in conscious cats. PLoS ONE 2017, 12, e0176443. [Google Scholar] [CrossRef]
- Kalluri, H.V.; Zhang, H.; Caritis, S.N.; Venkataramanan, R. A physiologically based pharmacokinetic modelling approach to predict buprenorphine pharmacokinetics following intravenous and sublingual administration. Br. J. Clin. Pharmacol. 2017, 83, 2458–2473. [Google Scholar] [CrossRef]

| BUP PK | Dose 1 (mg) | tmax (h) | Cmax (ng/mL) | AUC2 (ng·h/mL) | Dose Normalized | |
|---|---|---|---|---|---|---|
| Cmax (ng/mL)/mg | AUC (ng·h/mL)/mg | |||||
| Mean | 7.64 | 1.45 | 7.18 | 26.80 | 1.06 | 3.84 |
| SD | 3.32 | 0.69 | 5.76 | 17.87 | 0.79 | 2.35 |
| RSD | 43% | 47% | 80% | 66% | 74% | 61% |
| BUP PK | Cmax (ng/mL) | Cmin2 (ng/mL) | ||||||
|---|---|---|---|---|---|---|---|---|
| BUP 1 | Nor-BUP | BUP-g | Nor-BUP-g | BUP | Nor-BUP | BUP-g | Nor-BUP-g | |
| Mean | 7.18 | 3.56 | 7.58 | 16.32 | 1.67 | 1.60 | 0.72 | 9.11 |
| SD | 5.76 | 4.9 | 10.6 | 19.7 | 1.44 | 2.03 | 1.10 | 11.61 |
| CYP 1 | UGT 2 | |
|---|---|---|
| AUCnBup+nBupG/AUCBup | AUCBupG/AUCBup | |
| Mean | 3.34 | 0.39 |
| SD | 1.87 | 0.26 |
| RSD | 55% | 66% |
| Parameter | Units | Final PK Model | Bootstrap Analysis (n = 500) | ||||
|---|---|---|---|---|---|---|---|
| Value | RSE [%] | Median | 2.5th | 97.5th | |||
| Fixed-effects | K12 | [1/h] | 2.34 | 34 | 2.41 | 0.77 | 4.11 |
| K14 | [1/h] | 1.29 | 61 | 1.27 | 0.84 | 3.98 | |
| K24 | [1/h] | 1.28 × 10−1 | 32 | 1.19 × 10−1 | 7.42 × 10−2 | 1.54 × 10−1 | |
| K23 | [1/h] | 6.43 × 10−2 | 36 | 6.32 × 10−2 | 2.69 × 10−2 | 1.07 × 10−1 | |
| K26 | [1/h] | 2.18 × 10−1 | 23 | 2.11 × 10−1 | 1.12 × 10−1 | 3.41 × 10−1 | |
| K62 | [1/h] | 4.10 × 10−2 | 14 | 4.21 × 10−2 | 3.86 × 10−2 | 4.61 × 10−2 | |
| K35 | [1/h] | 1.23 × 10−1 | 52 | 1.31 × 10−1 | 7.41 × 10−2 | 2.65 × 10−1 | |
| K45 | [1/h] | 8.73 × 10−1 | 43 | 8.81 × 10−1 | 4.55 × 10−1 | 1.31 | |
| K30 | [1/h] | 3.81 × 10−4 | 21 | 3.62 × 10−1 | 3.11 × 10−3 | 4.97 × 10−3 | |
| K50 | [1/h] | 1.27 × 10−1 | 15 | 1.39 × 10−1 | 9.91 × 10−2 | 1.54 × 10−1 | |
| KA | [1/h] | 2.54 | 58 | 2.43 | 1.71 | 4.38 | |
| V2/F | [L] | 861 | 11 | 872 | 811 | 965 | |
| V3/F | [L] | 702 | 16 | 712 | 623 | 802 | |
| Inter-individual variability | K24 | [%] | 41 | 87 | 43 | 15 | 87 |
| K23 | [%] | 63 | 78 | 65 | 11 | 76 | |
| K35 | [%] | 70 | 69 | 71 | 38 | 134 | |
| K45 | [%] | 56 | 121 | 54 | 13 | 159 | |
| K50 | [%] | 54 | 38 | 58 | 21 | 64 | |
| Residual error | BUP | [%] | 38 | 18 | 35 | 22 | 48 |
| Nor-BUP | [%] | 29 | 21 | 26 | 13 | 39 | |
| BUP-g | [%] | 80 | 61 | 78 | 42 | 187 | |
| Nor-BUP-g | [%] | 26 | 41 | 25 | 12 | 38 | |
| Doses (mg) | Predicted AUC (ng·h/mL) | Observed AUC (ng·h/mL) | AUC Ratio | Predicted Cmax (ng/mL) | Observed Cmax (ng/mL) | Cmax Ratio | Reference | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 5th | 95th | Mean | Mean | 5th | 95th | Mean | Mean | ||||
| 16 | 31.56 | 71.84 | 54.13 | 45.64 | 1.19 | 6.15 | 8.27 | 7.47 | 6.25 | 1.20 | M. Jönsson, European Journal of Pharmaceutical Sciences 122 (2018) 125–133; [21] |
| 4 | 8.54 | 22.87 | 15.86 | 17.84 | 0.89 | 1.91 | 3.54 | 2.71 | 2.44 | 1.11 | |
| 4 | 8.54 | 22.87 | 15.86 | 12.52 | 1.27 | 1.91 | 3.54 | 2.71 | 1.84 | 1.47 | Harris et al. Healthy females and male non-opioid-dependent users, age range (22–42 years); [22] |
| 8 | 19.11 | 40.34 | 29.74 | 20.22 | 1.47 | 2.51 | 5.27 | 3.87 | 3 | 1.29 | |
| 16 | 31.56 | 71.84 | 54.13 | 34.89 | 1.55 | 6.15 | 8.27 | 7.47 | 5.95 | 1.26 | |
| 16 | 31.56 | 71.84 | 54.13 | 32.63 | 1.66 | 6.15 | 8.27 | 7.47 | 5.47 | 1.37 | |
| 2 | - | - | - | - | - | 0.89 | 1.72 | 1.12 | 1.06 | 1.06 | McAleer et al. Opioid-naïve healthy male subjects, age range (19–42); [23] |
| 8 | 19.11 | 40.34 | 29.74 | 31.81 | 0.93 | 2.51 | 5.27 | 3.87 | 4 | 0.97 | |
| 12 | 29.63 | 54.22 | 42.59 | 41.61 | 1.02 | 4.29 | 7.61 | 5.84 | 5.4 | 1.08 | |
| 16 | 31.56 | 71.84 | 54.13 | 52 | 1.04 | 6.15 | 8.27 | 7.47 | 6.4 | 1.17 | |
| 8 | 19.11 | 40.34 | 29.74 | 24.55 | 1.21 | 2.51 | 5.27 | 3.87 | 3.2 | 1.21 | |
| 8 | 19.11 | 40.34 | 29.74 | 24.6 | 1.21 | 2.51 | 5.27 | 3.87 | 3.2 | 1.21 | |
| 4 | 8.54 | 22.87 | 15.86 | 9.37 | 1.69 | 1.91 | 3.54 | 2.71 | 2 | 1.36 | Ciraulo et al. Healthy non-opioid-dependent users, age range (21–45); [24] |
| 8 | 19.11 | 40.34 | 29.74 | 19.92 | 1.49 | 2.51 | 5.27 | 3.87 | 2.65 | 1.46 | |
| 16 | 31.56 | 71.84 | 54.13 | 34.94 | 1.55 | 6.15 | 8.27 | 7.47 | 4.42 | 1.69 | |
| 24 | 51.93 | 82.74 | 67.56 | 48.81 | 1.38 | 7.95 | 10.67 | 9.44 | 5.41 | 1.74 | |
| 4 | 8.54 | 22.87 | 15.86 | 13.09 | 1.21 | 1.91 | 3.54 | 2.71 | 2.33 | 1.16 | Ciraulo et al. Healthy non-opioid-dependent users, age range (21–55); [23] |
| 8 | 19.11 | 40.34 | 29.74 | 23.23 | 1.28 | 2.51 | 5.27 | 3.87 | 3.53 | 1.10 | |
| 16 | 31.56 | 71.84 | 54.13 | 39.38 | 1.37 | 6.15 | 8.27 | 7.47 | 5.83 | 1.28 | |
| 24 | 51.93 | 82.74 | 67.56 | 47.55 | 1.42 | 7.95 | 10.67 | 9.44 | 6.44 | 1.47 | |
| 16 | 31.56 | 71.84 | 54.13 | 54.7 | 0.99 | 6.15 | 8.27 | 7.47 | 6.88 | 1.09 | Compton et al. Healthy female and male ipioi-dependent-users, age range (18–65); [25] |
| 24 | 51.93 | 82.74 | 67.56 | 81.1 | 0.83 | 7.95 | 10.67 | 9.44 | 9.1 | 1.04 | |
| 32 | 68.49 | 102.37 | 84.31 | 103 | 0.82 | 12.41 | 16.29 | 14.21 | 13.93 | 1.02 | |
| 2 | 5.14 | 9.63 | 7.41 | 6.5 | 1.14 | 0.89 | 1.72 | 1.12 | 0.85 | 1.32 | Greenwald et al. Healthy female and male opioid-dependent-users, age range (34–45); [10] |
| 16 | 31.56 | 71.84 | 54.13 | 41.9 | 1.29 | 6.15 | 8.27 | 7.47 | 5.15 | 1.45 | Moody et al. Drug Alcohol Depend. (2011) 118(2–3), 479–483; [15] |
| 16 | 31.56 | 71.84 | 54.13 | 58.4 | 0.93 | 6.15 | 8.27 | 7.47 | 6.97 | 1.07 | |
| 2 | 5.14 | 9.63 | 7.41 | 10.9 | 0.68 | 0.89 | 1.72 | 1.12 | 1.65 | 0.68 | R. Dong et al. Drugs in R&D (2019) 19, 255–265; [14] |
| 4 | 8.54 | 22.87 | 15.86 | 18.1 | 0.88 | 1.91 | 3.54 | 2.71 | 2.57 | 1.05 | |
| 8 | 19.11 | 40.34 | 29.74 | 33.3 | 0.89 | 2.51 | 5.27 | 3.87 | 5 | 0.77 | |
| 12 | 29.63 | 54.22 | 42.59 | 47.7 | 0.89 | 4.29 | 7.61 | 5.84 | 7.03 | 0.83 | |
| 16 | 31.56 | 71.84 | 54.13 | 55.6 | 0.97 | 6.15 | 8.27 | 7.47 | 7.84 | 0.95 | |
| 24 | 51.93 | 82.74 | 67.56 | 73 | 0.93 | 7.95 | 10.67 | 9.44 | 11.7 | 0.81 | |
| 4 | 8.54 | 22.87 | 15.86 | 23.89 | 0.66 | 1.91 | 3.54 | 2.71 | 3.31 | 0.82 | Kuhlman et al. Journal of Analitical Toxicology Vol 20 Oct 1996, p369; [26] |
| 2 | 5.14 | 9.63 | 7.41 | 6.789 | 1.09 | 0.89 | 1.72 | 1.12 | 0.78 | 1.44 | Healthy adult male or non-pregnant, non-breastfeeding female volunteers, 18–45 years of age (inclusive), BMI between 18 and 30 kg/m2 (inclusive) and weighed a minimum of 50 kg (110 lbs); [27] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santiago, D.; Mangas-Sanjuan, V.; Melin, K.; Duconge, J.; Zhao, W.; Venkataramanan, R. Pharmacokinetic Characterization and External Evaluation of a Quantitative Framework of Sublingual Buprenorphine in Patients with an Opioid Disorder in Puerto Rico. Pharmaceutics 2020, 12, 1226. https://doi.org/10.3390/pharmaceutics12121226
Santiago D, Mangas-Sanjuan V, Melin K, Duconge J, Zhao W, Venkataramanan R. Pharmacokinetic Characterization and External Evaluation of a Quantitative Framework of Sublingual Buprenorphine in Patients with an Opioid Disorder in Puerto Rico. Pharmaceutics. 2020; 12(12):1226. https://doi.org/10.3390/pharmaceutics12121226
Chicago/Turabian StyleSantiago, Darlene, Victor Mangas-Sanjuan, Kyle Melin, Jorge Duconge, Wenchen Zhao, and Raman Venkataramanan. 2020. "Pharmacokinetic Characterization and External Evaluation of a Quantitative Framework of Sublingual Buprenorphine in Patients with an Opioid Disorder in Puerto Rico" Pharmaceutics 12, no. 12: 1226. https://doi.org/10.3390/pharmaceutics12121226
APA StyleSantiago, D., Mangas-Sanjuan, V., Melin, K., Duconge, J., Zhao, W., & Venkataramanan, R. (2020). Pharmacokinetic Characterization and External Evaluation of a Quantitative Framework of Sublingual Buprenorphine in Patients with an Opioid Disorder in Puerto Rico. Pharmaceutics, 12(12), 1226. https://doi.org/10.3390/pharmaceutics12121226







