1. Background
Allergic rhinitis and bronchial asthma are airway inflammation chronic diseases that often coexist [
1]. Moreover, allergic rhinitis is a common comorbidity of asthma that contributes to asthma severity [
2]. As a result of the co-existence of asthma and allergic rhinitis, targeting both pathologies represent a strategy for the development of new joint therapies [
3,
4,
5].
Currently, therapy consists of controlling symptoms and minimizing associated risks. Bronchodilators (short-, long-, and ultra-long acting) and anti-inflammatory drugs such as glucocorticoids (GCS), leukotriene-receptor antagonists, and theophylline are some of the existing drugs on the market.
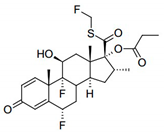
Among GCS, fluticasone propionate (FTZ) is one of the drugs delivered in a dry powder inhaler or by nasal instillation. Nasal and pulmonary delivery routes of administration are non-invasive, convenient, and comfortable, aiming the direct delivery of FTZ to the lungs [
5].
Several strategies for improving pulmonary or nasal administration of low or high molecular weight molecules have been widely described [
6,
7]. In the present work, and using FTZ as a model drug relevant for pulmonary administration [
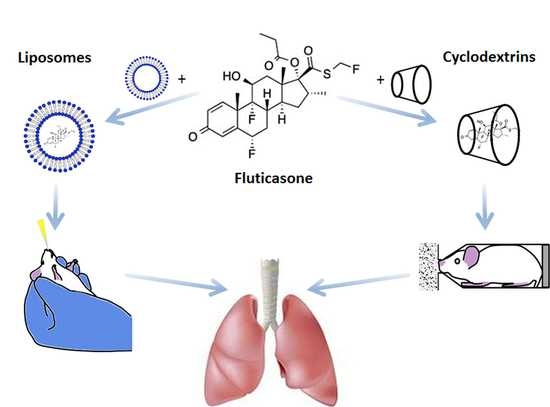
8], two strategies were addressed following its incorporation in liposomes or in Cyclodextrin (CyD) complexes.
Table 1 contains some physical properties of this molecule, namely chemical structure, molecular weight, octanol/water partition coefficient (LogP), and solubility in water.
Liposomes are undeniable biocompatible lipid systems able to increase the solubility of many lipophilic drugs, such as the case of FTZ [
9,
10,
11,
12,
13,
14] while improving delivery properties across nasal mucosa by promoting a prolonged contact between the drug and the absorptive sites in the nasal cavity [
15]. On the other hand, CyD are versatile excipients with great potential as drug carriers in pharmaceutical technology able to improve solubility, bioavailability, and stability of associated drugs [
16], particularly for pulmonary delivery [
17,
18].
In this work, liposomal formulations of FTZ were developed for further intranasal administration, while FTZ cyclodextrin (CyD) complexes previously developed [
17] were tested for pulmonary administration as dry powders. All FTZ formulations were submitted to in vitro studies in the presence of a respiratory epithelial cell line, the A549, to evaluate their influence on cell viability. The biodistribution profile of the two FTZ delivery systems was performed aiming to compare the lung accumulation levels of the glucocorticoid using liposomes and CyD complexes following intranasal and pulmonary delivery, respectively. Particularly, for the pulmonary delivery of FTZ CyD complexes, a nose-only chamber was designed, enabling the simultaneous administration for six mice [
19].
2. Materials and Methods
2.1. Materials
Fluticasone propionate (FTZ) was a kind gift from Hovione Farmacêutica, S.A. (Loures, Portugal). The CyDs used, Acetyl-γ-cyclodextrin (Acetyl-γ-CyD), was a gift from Cyclolab (Budapest, Hungary), Hydroxypropyl-γ-cyclodextrin (HP-γ-CyD) and γ-cyclodextrin (γ-CyD) were a gift from Wacker (Burghauser, Germany). Pure phospholipids, egg phosphatidyl choline (PC), and phosphatidyl glycerol (PG) were purchased from Avanti Polar Lipids (Alabaster, AL, USA). Cholesterol (CHOL), stearylamine (SA), Sodium dodecyl sulphate (SDS), dimethylsulfoxide (DMSO), Tween® 80, and phosphate buffered saline (PBS) were obtained from Sigma (St. Louis, MO, USA). Polycarbonate membranes from Nucleopore Track-Etched were purchased from Whatman, Ltd. (Clifton, NJ, USA). Dulbecco’s Modified Eagle Medium (DMEM), fetal bovine serum (FBS), TrypLE™ Express, penicilin/streptomycin, and Trypan Blue from Gibco were obtained from Invitrogen, Life Technologies Corporation (Grand Island, NY, USA). Reagents for in vitro assays were obtained from Promega (Madison, WI, USA).
Acetonitrile (HPLC grade) was from Merck (Darmstadt, Germany). Highly purified water was of Milli-Q quality and obtained from ELIX-3 equipment (Merck Millipore, Darmstadt, Germany). All other reagents and solvents were of the purest grade available, and generally were used without further treatment.
2.2. Cell Lines
The human alveolar adenocarcinoma cell line, the A549 line (ATCC® CCL-185™), was obtained from LGC Standards, Barcelona, Spain.
2.3. Animals
Male BALB/c mice were obtained from Gulbenkian Institute of Science (Oeiras, Portugal). Animals were kept on an animal room under standard hygiene conditions, with access to commercial chow and acidified drinking water ad libitum. All animal experiments were conducted according to the animal welfare organ of the Faculty of Pharmacy, Universidade de Lisboa, approved by the competent national authority Direção Geral de Alimentação e Veterinária (DGAV) on the 22/09/2016, (Project “Lung CYD-DDS (cyclodextrin-based drug delivery systems for inhalation”) and in accordance with the EU Directive (2010/63/UE) and Portuguese laws (DL 113/2013, 2880/2015, 260/2016, and 1/2019).
2.4. Preparation of Fluticasone Propionate (FTZ) Liposomes
Liposomes composed of the selected phospholipids were prepared by the de-hydration re-hydration method (DRV) [
21]. To resume, the selected phospholipids (20 µmol/mL) were mixed with FTZ at 500 µg/mL and solubilized in chloroform. The mixture was dried in a rotary evaporator (Buchi, Switzerland) until achievement of a thin lipid film in a round flask.
This film was then dispersed in deionized water and the liposomal suspension was frozen (−70 °C) and lyophilized in a freeze dryer (Edwards, CO, USA) overnight.
The re-hydration of the lyophilized powder was performed with hydroxyethyl piperazineethane sulfonic acid (HEPES) buffer (10 mM HEPES, 145 mM NaCl, pH 7.4) in two steps, in order to enhance FTZ incorporation. In the first hydration step, for 30 min, two-tenths of the volume of the original dispersion was added and subsequently the addition of HEPES buffer, up to the starting volume, was made. In order to reduce and homogenize the mean size of liposomes, the so-formed suspensions were submitted to an extrusion step through polycarbonate membranes of appropriate pore sizes until the desired mean size of liposomes was reached (0.8, 0.6, 0.4, 0.2 µm) under nitrogen pressure (10−500 1b/in2) with an Extruder device Lipex (Biomembranes Inc., Vancouver, BC, Canada). It is important to refer that the re-hydration and extrusion steps were always performed at a temperature above the transition temperature (Tc) of the phospholipids.
The separation of non-incorporated FTZ was performed by gel filtration (Econo-Pac® 10DG, Bio-Rad Laboratories, Hercules, CA, USA) followed by ultracentrifugation at 250,000 g for 120 min, at 15 °C, in a Beckman LM-80 ultracentrifuge (Beckman Instruments, Inc., Palo Alto, CA, USA). At the end, the pellet was suspended in HEPES buffer, according to the final concentration desired for FTZ liposomes. Unloaded liposomes, used in in vitro tests, were also performed for all lipid compositions tested. After rotary evaporator, the lipid film was dispersed in HEPES buffer and the resultant suspensions were extruded as above referred for FTZ loaded liposomes.
2.5. Characterization of FTZ Liposomes
The liposomal formulations were characterized in terms of lipid composition, lipids and FTZ concentrations, mean size, and zeta potential. The FTZ quantification either in liposomes or in organs was performed by HPLC (
Section 2.7), according to Couto et al. (2014) [
18], with some modifications. The lipid content of the formulations was determined using a spectrophotometric technique [
22]. Unloaded liposomes were characterized in terms of lipid content, mean size, and zeta potential.
2.5.1. Incorporation Efficiency
After quantification of lipids and FTZ, the incorporation efficiency (I.E.) was calculated using the following Equation:
where: (FTZ/Lip)
f means Final FTZ to lipid ratio; (FTZ/Lip)
i means initial FTZ to lipid ratio; I.E. is a measure of the efficiency of a lipid mixture for incorporating FTZ.
2.5.2. Liposome Size Measurements
Liposome mean diameter was determined by dynamic light scattering in a hydrodynamic sizing system (Zetasizer Nano S, Malvern Instruments, Malvern, UK). As a measure of particle size distribution of liposomes, the system reports the polydispersity index (PdI). The index ranges from 0.0 for an entirely monodisperse sample up to 1.0 for a polydisperse suspension. Liposomal formulations were previously diluted (1:100) in HEPES buffer. To ensure that appropriate mean diameter and PdI were achieved, size measurements were performed during the extrusion step and in final formulations. Each analysis was carried out at 25 °C in triplicate.
2.5.3. Zeta Potential Determination
Zeta potential of liposomal preparation was measured by Laser Doppler Electrophoresis (Zetasizer Nano Z, Malvern instruments, UK). Liposomal formulations were previously diluted (1:100) in HEPES buffer. The zeta potential of samples was recorded at a temperature of 25 °C, in triplicate.
2.6. Preparation and Characterization of FTZ Cyclodextrin Complexes
FTZ was formulated in different CyDs (Table 3) and resulting dry powders were characterized according to the method described in Drumond et al. (2014) [
17]. For in vitro studies, CyD complex dry powders were prepared using the same methodology, but without FTZ.
2.7. Chromatographic System for FTZ Quantification
The HPLC used was a System Gold (Beckman Instruments, Inc., USA) constituted by a 168 Diode-Array Detector, a 126 Solvent Module, and a Midas Spark 1.1 auto-sampler (Spark, Netherlands). The analytical column was a LiChrospher® 100 (125 × 4.0 mm) RP-18 (5 µm), (Merck, Darmstadt, Germany). The apparatus was connected to a desk computer with the 32 KARAT Software (Beckman Coulter, USA) for integration and treatment of chromatograms.
The mobile phase consisted of acetonitrile (ACN) and water (60:40, v/v) with a flow rate of 1.0 mL/min. FTZ analysis was performed by UV detection at a fixed wavelength of 236 nm and the loop was of 10 µL.
Preparation of Standard Solutions
Stock solutions of FTZ at 200 µg/mL in ACN were prepared. FTZ standards ranging from 2 to 40 µg/mL were made by diluting the respective initial stock solution with ACN. Samples from liposomes and after extraction in organs were diluted in ACN in the range of calibration curves. Standards and samples were filtered through 0.2 μm porosity polytetrafluoroethylene (PTFE) membranes before injection onto HPLC. An intermediate standard was always injected together with the analyzed samples to verify the precision of the obtained chromatograms from their peak area and concentration response.
For determining the concentration average of FTZ in each CyD complex, 5 mg of each FTZ CyD complex was weighted, diluted in ACN, and filtered through 0.2 µm porosity PTFE membranes before injection onto HPLC.
2.8. In Vitro Experiments
2.8.1. Cell Culture Maintenance
The A549 cell line was grown in culture flasks and maintained in DMEM, supplemented with 10% (
v/
v) of heat inactivated FBS (iFBS) and 1% (
v/
v) penicillin/streptomycin in a humidified atmospheric incubator at 37 °C, 5% CO
2 [
23]. The maintenance of cultures was performed every three to four days, until cells reached a confluence of about 80%. At this point, sub-culturing was performed using a solution of TripLE
TM Express. Briefly, after media removal, the cell layer was washed with PBS and incubated with TripLE
TM Express for 7–10 min at 37 °C. After cells’ detachment, complete medium was added. The cells were then centrifuged in a bench centrifuge (Beckman, Izasa, Spain) at 500×
g for 10 min and the pellet was suspended in fresh medium. Appropriate aliquots of the cell suspension were seeded in new culture flasks.
The storage of cell lines was done in liquid nitrogen with freezing medium consisting of iFBS and 10% (v/v) of DMSO.
2.8.2. Cellular Viability Assay
Cell viability was evaluated in the absence or presence of increasing concentrations of FTZ in free and liposomal forms as well as incorporated in CyD complexes, by measuring mitochondrial activity, based on the colorimetric method, the MTS (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2
H-tetrazolium), from Promega (Madison, WI, USA) [
23]. Briefly, dehydrogenase enzymes found in metabolically active cells convert tetrazolium compound to a water-soluble formazan dye, which can be quantified spectrophotometrically at λ = 490 nm, being the absorbance directly correlated to viable cells [
23].
A549 cells at a concentration of 5 × 104/mL were plated into 96-well plates (200 µL) under the culture conditions described before. Twenty-four hours after plating, medium was removed, and adherent cells were incubated with FTZ in free or incorporated in liposomes or in CyD complexes at a concentration ranging from 5 to 60 µM. Negative and positive controls were also used in parallel. Negative control was constituted by cells in the presence of complete medium. The positive control was constituted by 0.2% (w/v) SDS. In addition, as the stock free FTZ solution was prepared in DMSO, cells were also incubated with DMSO at 0.24% (v/v) corresponding to the highest concentration used for FTZ solubilization. Unloaded liposomes and CyD complexes constituted other controls, using the same lipid concentrations as in FTZ liposomes and the same amount as the one used in the respective FTZ CyD complex, respectively. All tests were performed in sextuplicate for each tested concentration and controls. After 72 h of incubation, culture medium was removed from all wells, and replaced with 100 µL of incomplete medium and 20 µL, of the combined PMS/MTS solution (1:20 v/v) freshly prepared, into each well according to manufacturer’s instructions. Plates were slightly agitated and incubated for 60 min, under the same culture conditions mentioned before.
Absorbance was measured at λ = 490 nm (Biotek Instruments®, Winooski, VT, USA). The absorbance of negative control (cells incubated with complete medium) was set as 100% viability.
A triplicate set of control wells (without cells, Blank) containing the same volumes of culture medium and combined PMS/MTS solution as in the experimental wells was also prepared. The absorbance average at λ = 490 nm from these Blank control wells was subtracted from all other absorbance values to yield corrected absorbances.
The cellular viability (%) was calculated as follows:
where
A is the absorbance obtained for each concentrations of the tested formulation;
B is the absorbance average obtained for the Blank;
M is the absorbance average for negative control (cells incubated with complete medium).
2.9. Biodistribution Studies of FTZ Formulations
Biodistribution studies were performed in healthy BALB/C mice after intranasal (i.n.) administration of FTZ in free or liposomal forms at a dose of 1 mg/kg of body weight (40 µL/nostril).
For the biodistribution studies of FTZ CyD complexes the formulations were pulmonary administered using a nose-only chamber that simultaneously allows the exposition of six animals to the dry powders [
19]. Mice were restrained with their nostrils inserted snugly into the inhalation chamber (
Figure 1). About 200 mg of CyD formulations were placed into the donor compartment of the apparatus. To prepare free FTZ in order to obtain the same amount of FTZ per gram of powder, FTZ was diluted with lactose of analytical grade (Fagron, Barcelona, Spain). The dry powders of all FTZ formulations were then dispersed by an airflow generated under an air pressure of 0.4 kg/cm
2 for 5 min.
Half an hour and three hours after i.n. administration or inhalation exposure, mice were sacrificed, blood was collected into heparinized tubes, and stored at −30 °C. Lung, spleen, and liver were removed and stored at −70 °C, until extraction procedure. At least three animals were used for each analyzed time, for each formulation tested.
2.10. Murine Model of Asthma
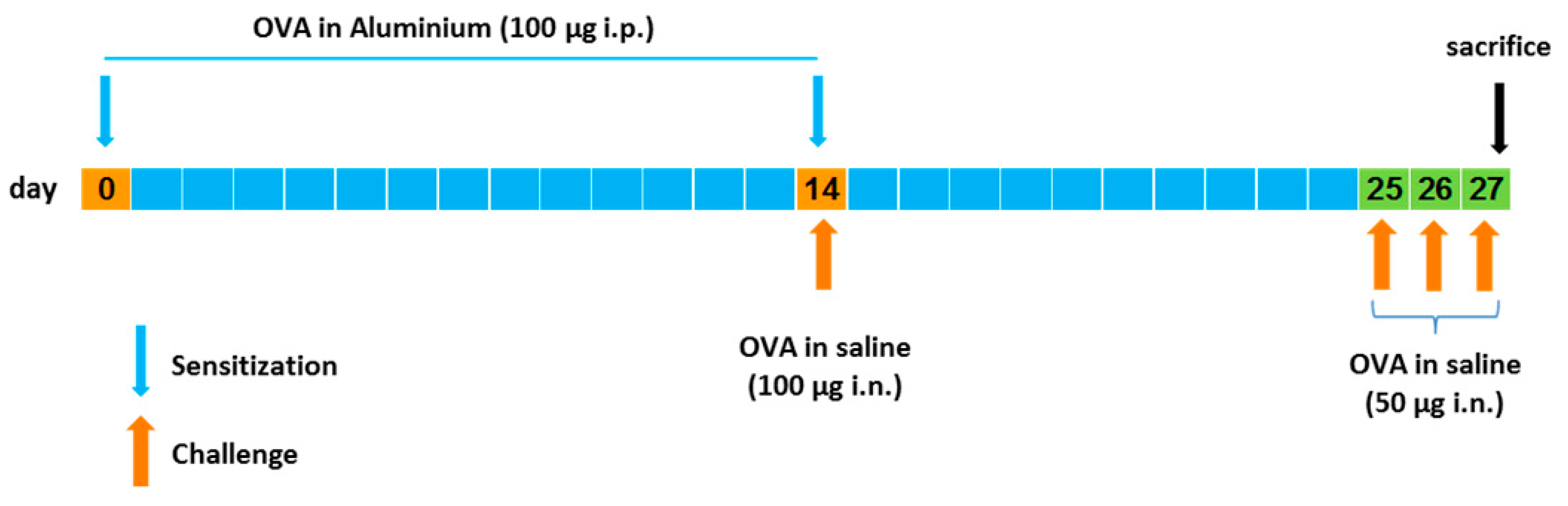
The allergic lung inflammation in mice was induced according to Henderson et al. [
24] and Oliveira et al. with some modifications [
25]. In
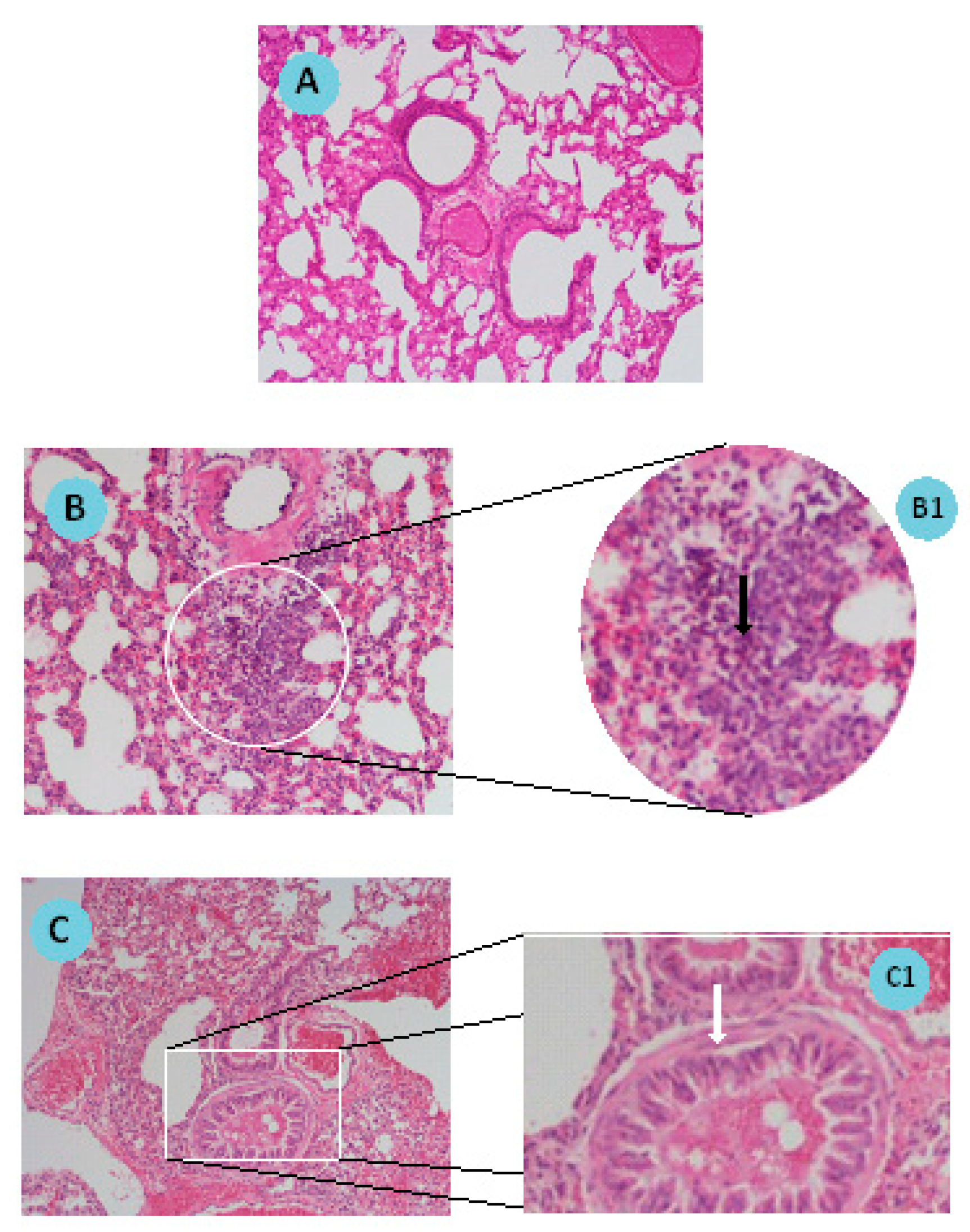
Figure 2, a schematic representation of the lung inflammation model used is shown. Briefly, mice were sensitized on days 0 and 14 by receiving intraperitoneal (i.p.) injections of 0.2 mL (100 µg) of chicken ovalbumin (OVA) in aluminum hydroxide (10 mg/mL). On days 14, 25, 26, and 27, mice were challenged with intranasal (i.n.) administrations of OVA in saline: 100 µg on day 14 and 50 µg on days 25, 26, and 27. Mice were sacrificed one day after the last OVA challenge and their lungs were removed and fixed in 10% neutral formalin. Lung sections were stained with hematoxylin and eosin for histologic analysis.
4. Discussion
FTZ is often prescribed as a first line-therapy for the effective management of pulmonary diseases such as asthma and rhinitis. Drug delivery systems, in particular liposomes and CyD complexes, are able to change in vivo biodistribution of incorporated molecules and have received great attention in recent years [
11,
12,
13]. In this work, FTZ was used as a model drug to study the biopharmaceutical advantages of its incorporation into two different systems: The incorporation of FTZ in liposomes for i.n. administration, and the association to CyD complexes, aiming to improve the in vivo profile of FTZ following pulmonary administration of resultant dry powders.
When designing FTZ liposomal formulations, it is crucial to achieve high loadings for this glucocorticoid; otherwise a high amount of lipid would be necessary to reach the desirable therapeutic dose. To achieve this goal, a combination of several factors, such as lipid composition, physicochemical properties of the drug, and liposomes preparation method should be considered. The DRV method used for incorporating FTZ in liposomes was selected, as it allows obtaining higher incorporation efficiencies than the simple thin-film hydration method [
10,
26,
29]. The preparation of FTZ liposomes with neutral, negative, and positively charged lipid compositions was performed aiming to study their influence on FTZ incorporation parameters and to select the best lipid mixtures. Due to its hydrophobic character, FTZ is assumed to be incorporated in the lipid bilayer. The main phospholipids used, PC and PG, were selected on the basis of their low
Tc (below 0 °C), meaning that at room temperature phospholipids are in liquid-crystalline phase, lipid bilayers are in a less ordered structure, and the chance of FTZ to be incorporated is higher [
31]. In fact, neutral and negatively charged liposomes prepared with PC or PC:PG, respectively, resulted in the lipid nanoformulations with higher FTZ incorporation parameters. The reduction on incorporation parameters of FTZ liposomes with PC and CHOL is based on the fact that CHOL is also inserted within lipid bilayers competing with the accommodation of the glucocorticoid. Nevertheless, for all tested FTZ nanformulations, liposomes presented small mean size values and homogeneous size distribution: Mean size ranged from 0.16 to 0.21 µm and PdI was below 0.2.
Another strategy to solve low solubility of FTZ or deficient accumulation in the lungs after inhalation involves the use of CyDs, supramolecular structures able to include water-insoluble molecules [
32]. CyDs may increase drug stability and bioavailability at the drug target site. In this sense, drug association to CyDs complexes can also increase drug action duration as drug complexes might promote a prolonged and controlled release of the incorporated material. There are a variety of CyDs derivatives in the market obtained by chemical synthesis, exhibiting different characteristics such as solubility, stability, and volume of the hydrophobic cavity, allowing controlling the activity and entrapment ability of incorporated drugs. In previous work [
17], the influence of different CyDs on physical characteristics of FTZ CyDs inhalation dry powders was evaluated. The best characteristics for pulmonary delivery were obtained with FTZ Ac-γ-CyD complex that permitted FTZ to reach the lungs in a higher percentage in relation to the emitted dose [
17].
The in vitro experiments developed in the present work aimed to investigate the potential toxic effect of FTZ formulations in the presence of a respiratory cell line, the A549. This cell line is useful for studying the mechanisms of drug delivery via the pulmonary route and in examining the transport of low molecular weight drugs and xenobiotic across the surface of the alveoli [
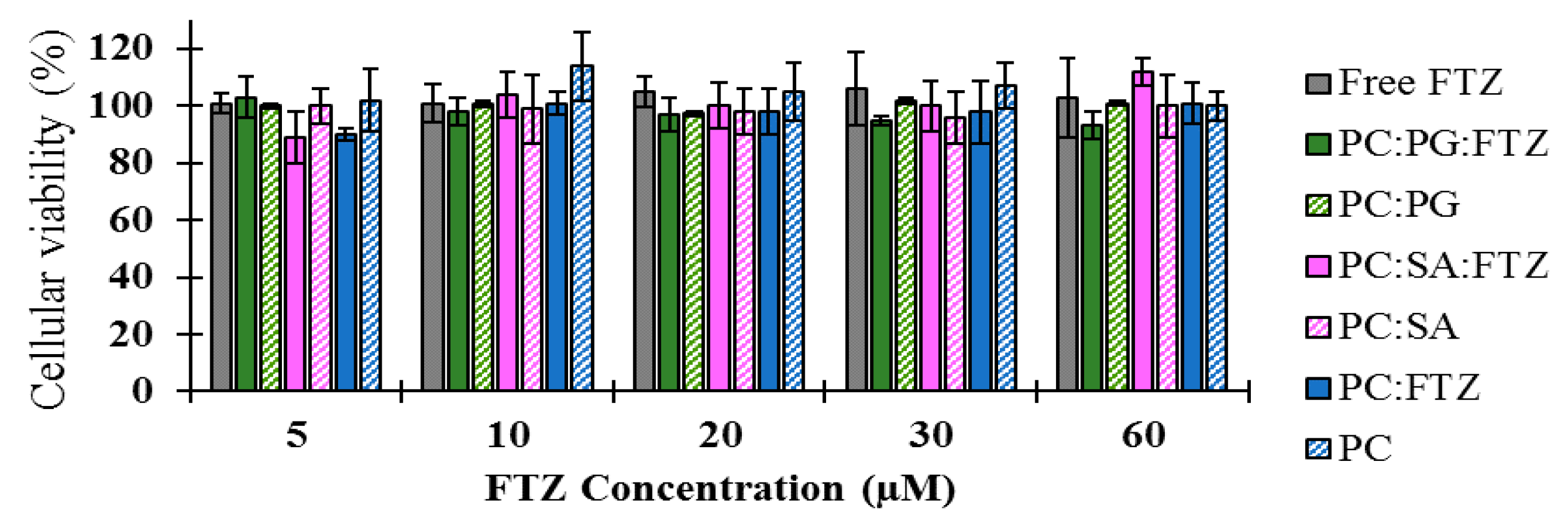
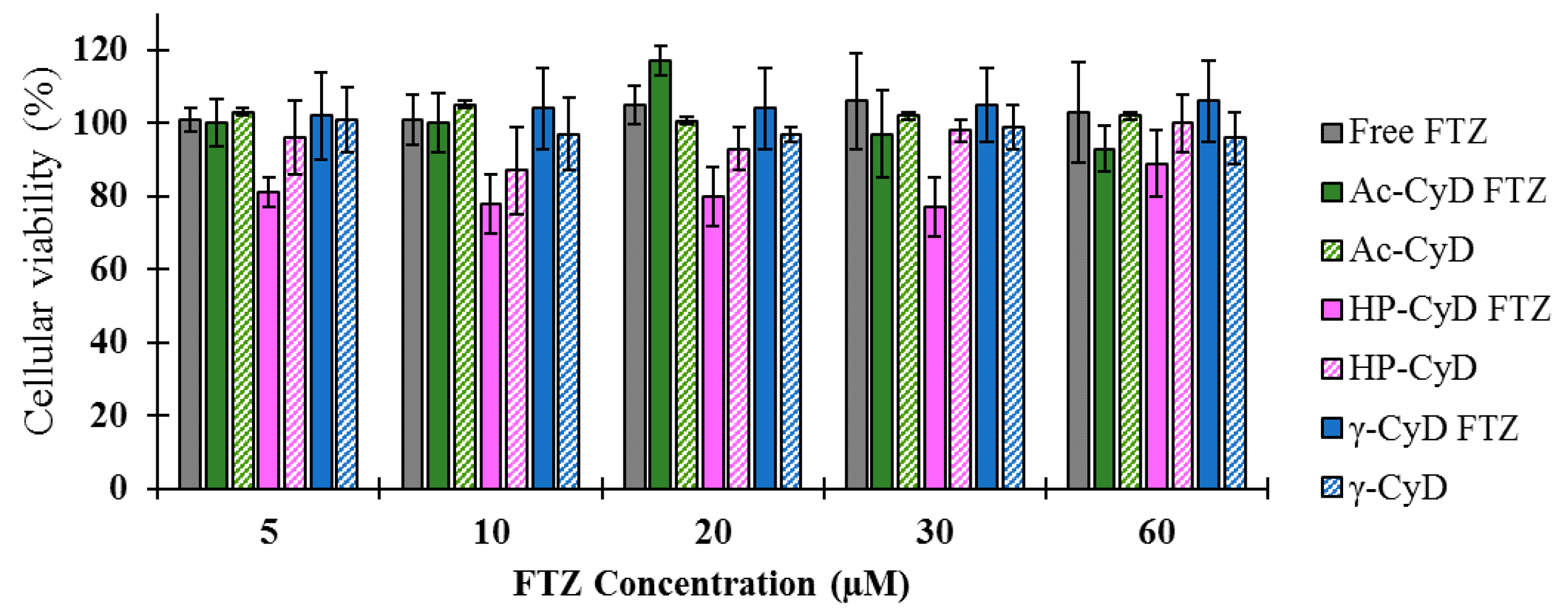
33]. The in vitro tests demonstrated that FTZ in the free form or incorporated in neutral, positive, and negatively charged liposomes did not elicit toxic effects towards the A549 cell line, as cellular viability was always superior to 90% for all tested formulations. Regarding FTZ CyD complexes, and according to the in vitro results, neither FTZ CyD formulations nor empty CyDs or FTZ in the free form resulted in significant reduction on cellular viability of A549 cells. Overall, the in vitro tests demonstrated that both drug delivery systems used for incorporating FTZ are safe to be applied in vivo.
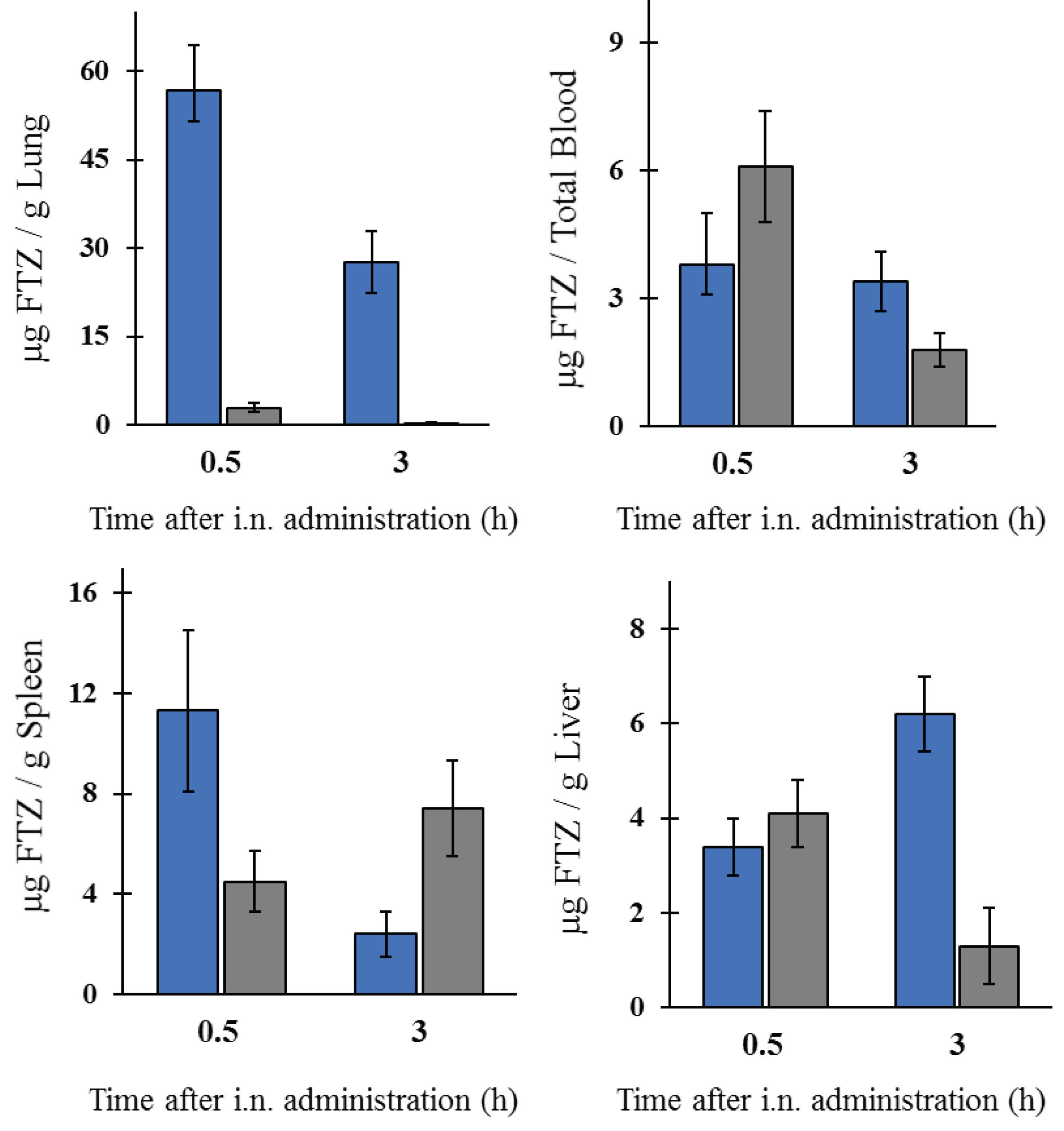
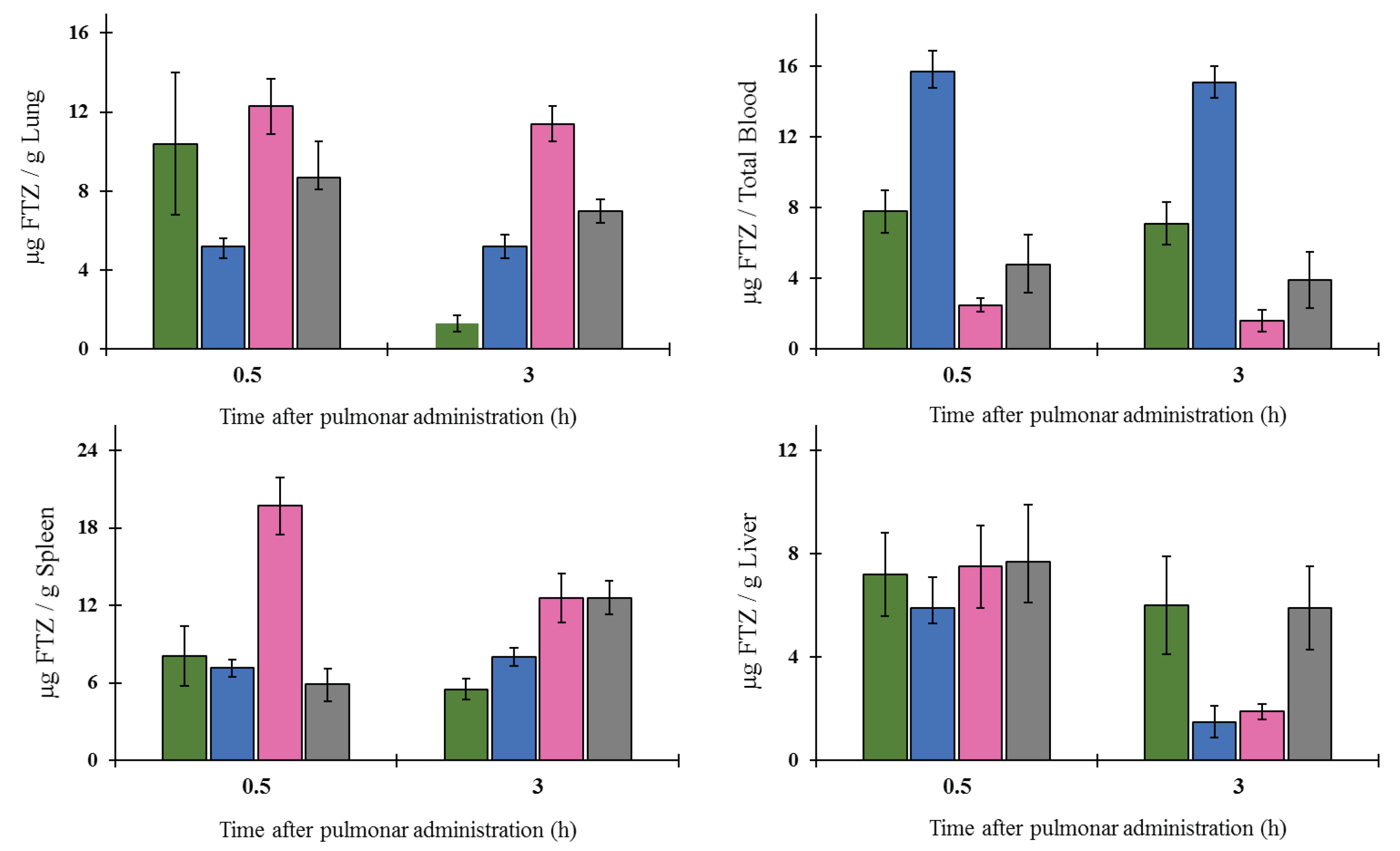
The in vivo biodistribution profile of FTZ in the liposomal form after i.n. administration and in the form of FTZ CyD complexes after pulmonary delivery was assessed and compared with the biodistribution profile of the drug free form. Particular interest was taken on the amount of FTZ accumulated in lungs. The obtained results showed that FTZ incorporated in liposomes resulted in much higher levels of this glucocorticoid in the lungs in comparison with free FTZ: 20 and 30 times, at 0.5 and 3 h after i.n. administration, respectively. Another important result was the determination of FTZ in blood as it represents the amount of drug that has been delivered and absorbed systemically from the lungs [
34]. The amount of FTZ observed for mice that received FTZ in the free form, 30 min after instillation, proves that FTZ reached the lungs. The i.n. administration of FTZ liposomes led to a constant amount of the corticosteroid in the blood for 3 h (around 4 µg), whereas a reduction after the instillation of the free FTZ was observed from 6 to less than 2 µg during the same analyzed time. The results obtained led us to conclude that FTZ in liposomal form was able to promote a depot in the lungs.
In the case of pulmonary administration of FTZ CyD complexes, the design of a nose-only inhalation chamber (
Figure 1) was crucial to perform the biodistribution studies [
19]. The ability of each CyD to deliver FTZ in the organs was evaluated and the results have demonstrated that for FTZ associated to HP-CyD complex, a higher amount of FTZ was obtained in the lung in comparison with the respective free form. This achievement was observed 0.5 and 3 h after animals were exposed to dry powders of FTZ CyD complexes for 5 min. Three hours after exposure, a slight decrease in the FTZ value in the lung was observed compared with the results for the 0.5 h time point. This could be an advantageous outcome as long as a sustained release of the glucocorticoid in the lung is a desirable result. Curiously, the already mentioned study of aerodynamic properties of inhaled particles containing FTZ CyDs indicates FTZ acetyl-γ-CyD complex as having the best characteristics for pulmonary delivery [
17]. The results obtained in vivo presented in this study do not corroborate such early in vitro findings. In fact, in vitro methods could only predict in vivo behavior, but do not replace animal experiments. For this reason, the possibility of testing developed formulations in an animal model of disease is of major relevance, and a reason why a mice model of asthma was established. Mice constitutes the most common species used to model allergic airway inflammation mainly for practical reasons, including relatively low handling costs and reduced amount of formulations needed. However, as mice do not spontaneously develop asthma, an artificial asthmatic reaction had to be induced in the airways. Several mouse models of acute allergic pulmonary inflammation are described in the literature. Different protocols for sensitization, use an allergen, in most of the cases OVA, in combination with an adjuvant, administered by the i.p. route, followed by a challenge of the allergen. In the late steps, the allergen may be inhaled as a nebulized formulation, or administered by intratracheal or i.n. instillation of an aqueous formulation [
35]. In the present work, the sensitization was performed by i.p. injections of OVA in combination with the adjuvant aluminum hydroxide, while the challenge was done using i.n. instillation of OVA in saline, according to Henderson et al. [
24] and Oliveira et al. [
25].
Histological analysis of lung sections of sensitized/challenged mice is a valuable tool to validate the establishment of the pathology. Indeed, in the present work, the visualization of lung sections after hematoxylin and eosin staining confirmed the cellular infiltration with an increased number of eosinophils and bronchioles with hyperplasia of the smooth muscle when compared to naïve mice, these observations being in accordance with the literature [
24,
25,
35].