The Impact of Lipid Corona on Rifampicin Intramacrophagic Transport Using Inhaled Solid Lipid Nanoparticles Surface-Decorated with a Mannosylated Surfactant
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. SLNas Preparation
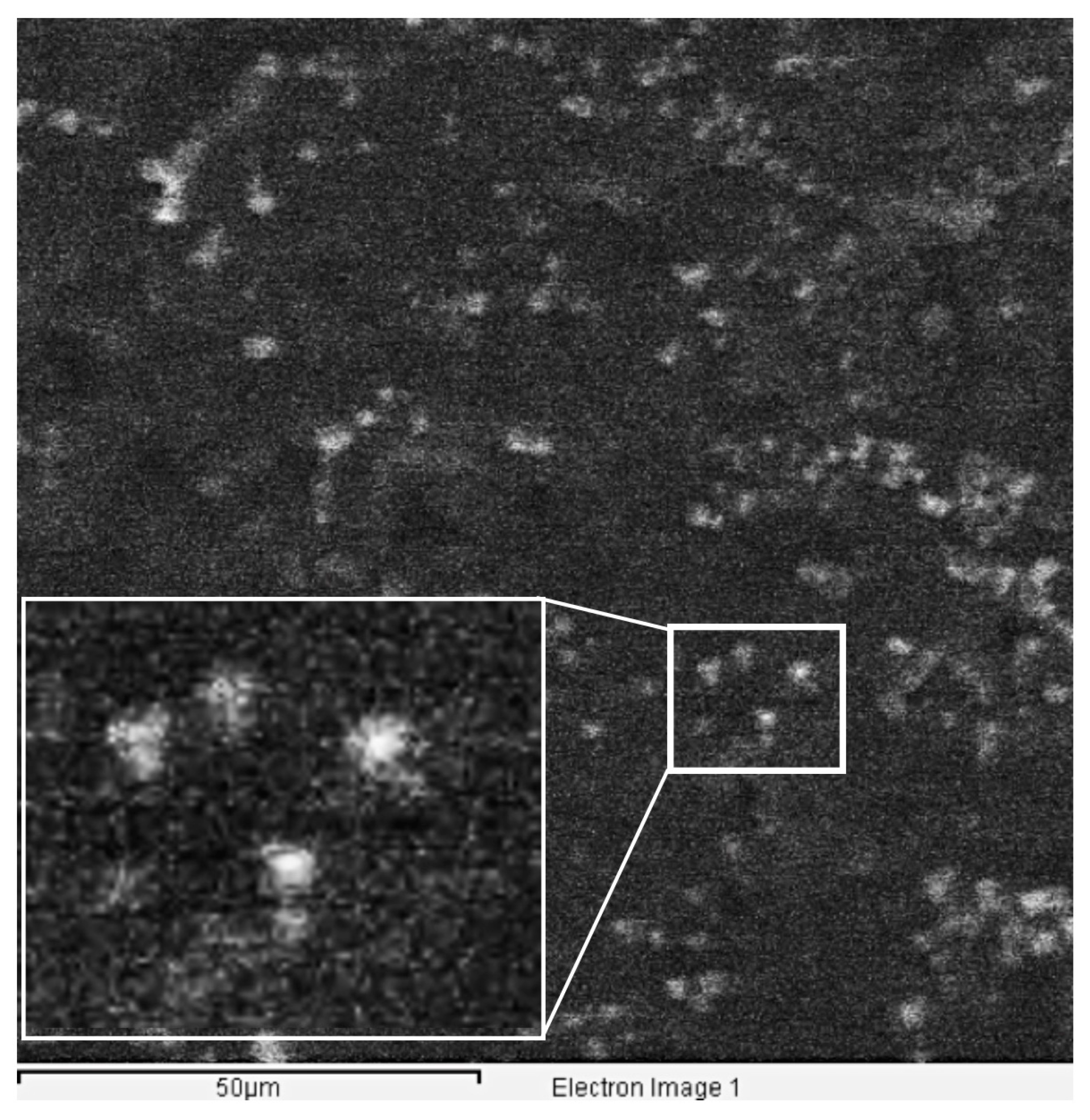
2.2.2. Morphology, Size, and Z-Potential
2.2.3. Density, Carr’s Index, and Specific Surface Area
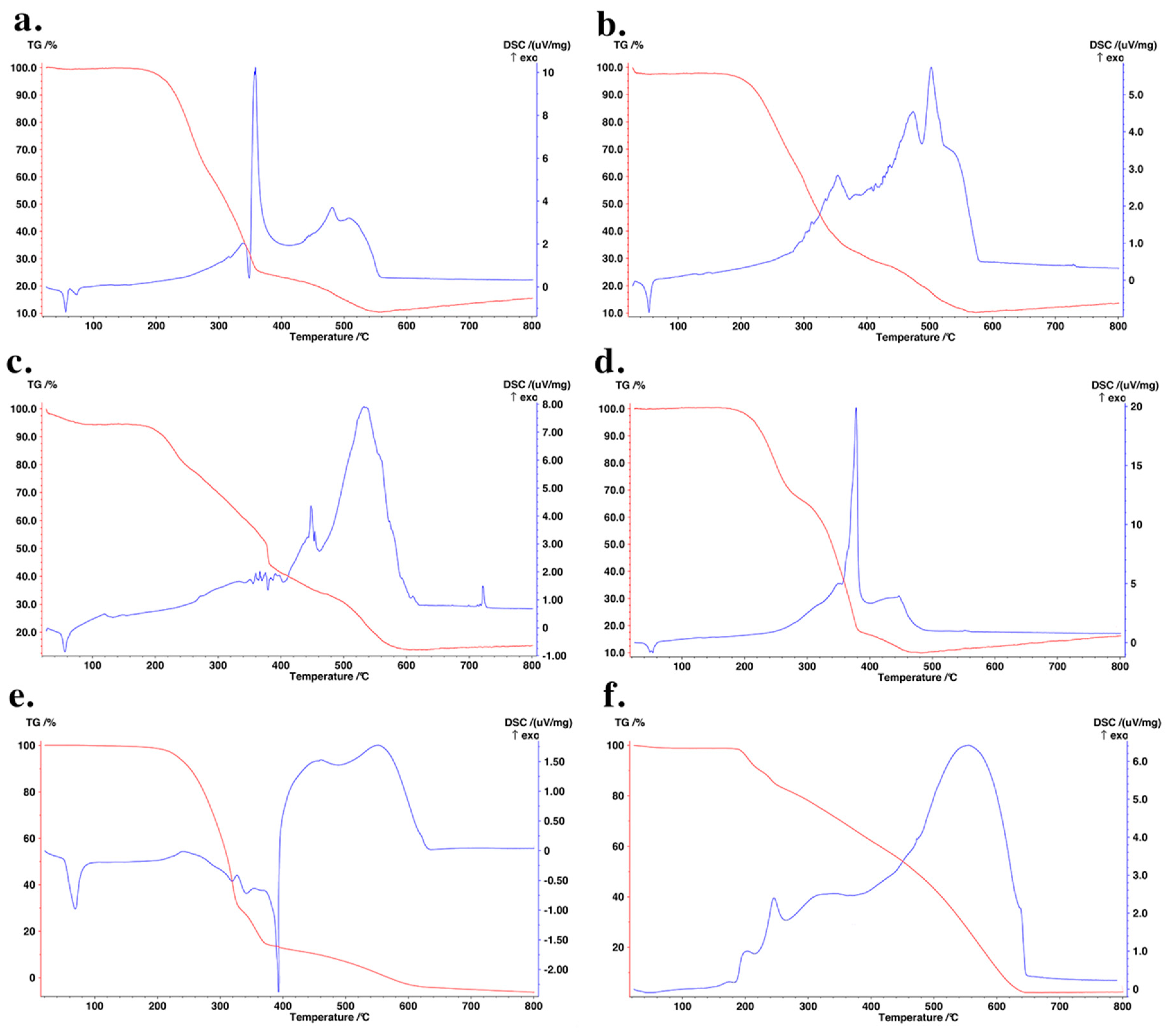
2.2.4. Thermal Analysis
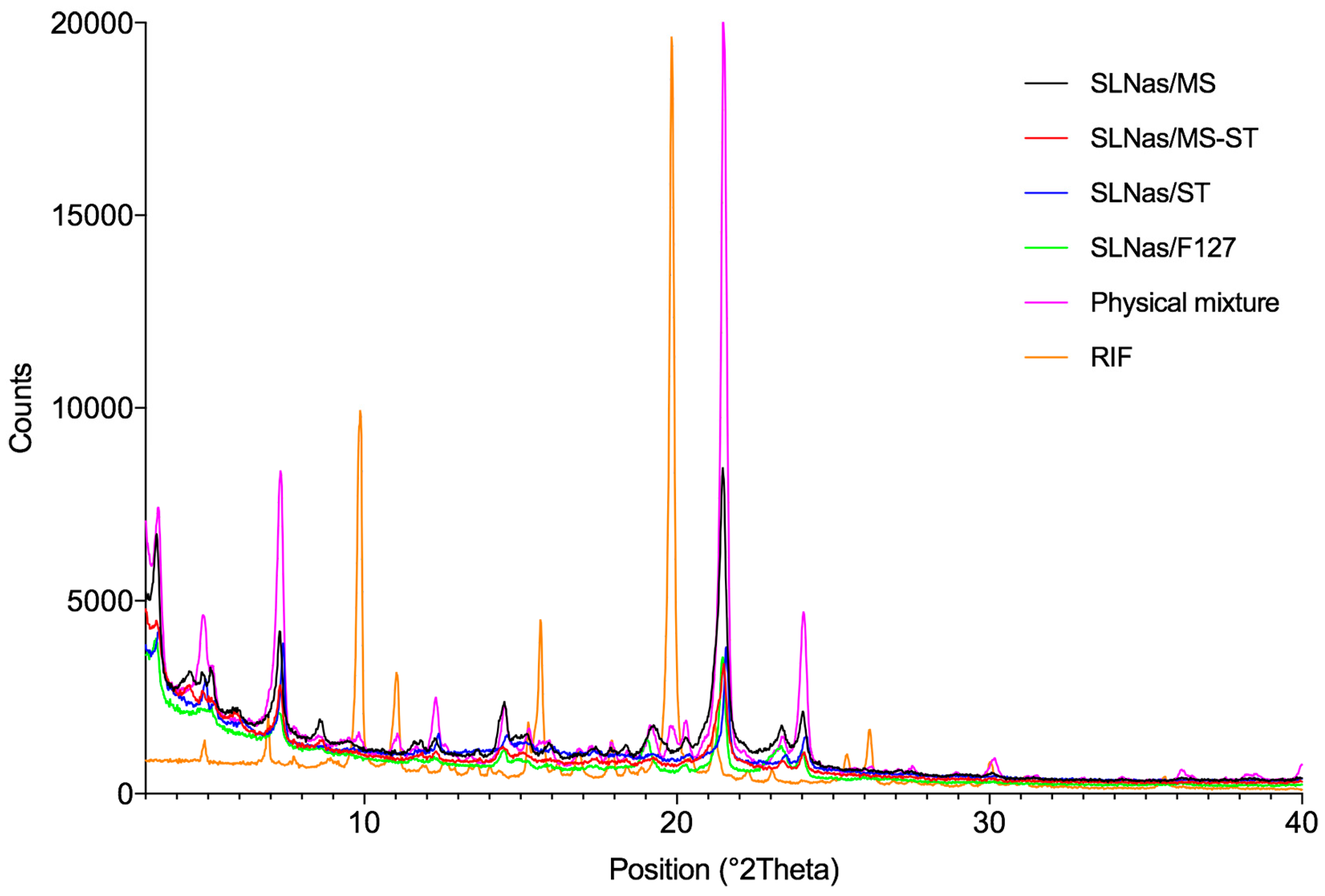
2.2.5. X-Ray Powder Diffraction
2.2.6. X-ray Photoelectron Spectroscopy
2.2.7. Wettability
2.2.8. Drug Loading and Encapsulation Efficiency
2.2.9. Aerodynamic Diameter
2.2.10. Respirability
2.2.11. Lipid Corona Formation
2.2.12. In Vitro Drug Release
2.2.13. Cell Culture Assay
2.2.14. Cytotoxicity Test
2.2.15. Intracellular RIF Determination
2.2.16. MR Inhibition Study
2.2.17. Intracellular RIF Determination after Lipid Corona Formation
2.2.18. Statistical Analysis
3. Results and Discussion
3.1. SLNas Physico-Chemical Properties
3.2. Lipid Corona Formation upon SLNas Contact with Pulmonary Surfactant
3.3. Effect of Pulmonary Surfactant on Drug Release and Macrophage Activity
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lee, W.-H.; Loo, C.-Y.; Traini, D.; Young, P.M. Nano- and micro-based inhaled drug delivery systems for targeting alveolar macrophages. Expert Opin. Drug Deliv. 2015, 12, 1009–1026. [Google Scholar] [CrossRef] [PubMed]
- de Boer, A.H.; Hagedoorn, P.; Hoppentocht, M.; Buttini, F.; Grasmeijer, F.; Frijlink, H.W. Dry powder inhalation: Past, present and future. Expert Opin. Drug Deliv. 2016, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Mehta, P. Dry Powder Inhalers: A Focus on Advancements in Novel Drug Delivery Systems. J. Drug Deliv. 2016, 2016, 8290963. [Google Scholar] [CrossRef] [PubMed]
- Hickey, A.J.; Mansour, H.M. Inhalation Aerosols: Physical and Biological Basis for Therapy, 3rd ed.; CRC Press: Boca Raton, FL, USA, 2019. [Google Scholar]
- Maretti, E.; Costantino, L.; Buttini, F.; Rustichelli, C.; Leo, E.; Truzzi, E.; Iannuccelli, V. Newly synthesized surfactants for surface mannosylation of respirable SLN assemblies to target macrophages in tuberculosis therapy. Drug Deliv. Transl. Res. 2019, 9, 298–310. [Google Scholar] [CrossRef] [PubMed]
- Patel, B.; Gupta, N.; Ahsan, F. Particle engineering to enhance or lessen particle uptake by alveolar macrophages and to influence the therapeutic outcome. Eur. J. Pharm. Biopharm. 2015, 89, 163–174. [Google Scholar] [CrossRef]
- Azad, A.K.; Rajaram, M.V.S.; Schlesinger, L.S. Exploitation of the Macrophage Mannose Receptor (CD206) in Infectious Disease Diagnostics and Therapeutics. J. Cytol. Mol. Biol. 2014, 1, 1154844789. [Google Scholar]
- Costa, A.; Sarmento, B.; Seabra, V. Mannose-functionalized solid lipid nanoparticles are effective in targeting alveolar macrophages. Eur. J. Pharm. Sci. 2018, 114, 103–113. [Google Scholar] [CrossRef]
- Notter, R.H. Lung Surfactants: Basic Science and Clinical Applications, 1th ed.; CRC Press: Boca Raton, FL, USA, 2000. [Google Scholar]
- Griese, M. Pulmonary surfactant in health and human lung diseases: State of the art. Eur. Respir. J. 1999, 13, 1455–1476. [Google Scholar] [CrossRef]
- Hamm, H.; Kroegel, C.; Hohlfeld, J. Surfactant: A review of its functions and relevance in adult respiratory disorders. Respir. Med. 1996, 90, 251–270. [Google Scholar] [CrossRef]
- Glasser, J.R.; Mallampalli, R.K. Surfactant and its role in the pathobiology of pulmonary infection. Microbes Infect. 2012, 14, 17–25. [Google Scholar] [CrossRef]
- Guagliardo, R.; Pérez-Gil, J.; De Smedt, S.; Raemdonck, K. Pulmonary surfactant and drug delivery: Focusing on the role of surfactant proteins. J. Control. Release 2018, 291, 116–126. [Google Scholar] [CrossRef] [PubMed]
- Schleh, C.; Hohlfeld, J.M. Interaction of nanoparticles with the pulmonary surfactant system. Inhal. Toxicol. 2009, 21, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Gasser, M.; Wick, P.; Clift, M.J.D.; Blank, F.; Diener, L.; Yan, B.; Gehr, P.; Krug, H.F.; Rothen-Rutishauser, B. Pulmonary surfactant coating of multi-walled carbon nanotubes (MWCNTs) influences their oxidative and pro-inflammatory potential in vitro. Part. Fibre Toxicol. 2012, 9, 17. [Google Scholar] [CrossRef] [PubMed]
- Vranic, S.; Garcia-Verdugo, I.; Darnis, C.; Sallenave, J.-M.; Boggetto, N.; Marano, F.; Boland, S.; Baeza-Squiban, A. Internalization of SiO2 nanoparticles by alveolar macrophages and lung epithelial cells and its modulation by the lung surfactant substitute Curosurf®. Environ. Sci. Pollut. Res. 2013, 20, 2761–2770. [Google Scholar] [CrossRef] [PubMed]
- Konduru, N.V.; Molina, R.M.; Swami, A.; Damiani, F.; Pyrgiotakis, G.; Lin, P.; Andreozzi, P.; Donaghey, T.C.; Demokritou, P.; Krol, S.; et al. Protein corona: Implications for nanoparticle interactions with pulmonary cells. Part. Fibre Toxicol. 2017, 14, 42. [Google Scholar] [CrossRef] [PubMed]
- Mousseau, F.; Le Borgne, R.; Seyrek, E.; Berret, J.-F. Biophysicochemical Interaction of a Clinical Pulmonary Surfactant with Nanoalumina. Langmuir 2015, 31, 7346–7354. [Google Scholar] [CrossRef] [PubMed]
- Raesch, S.S.; Tenzer, S.; Storck, W.; Rurainski, A.; Selzer, D.; Ruge, C.A.; Perez-Gil, J.; Schaefer, U.F.; Lehr, C.-M. Proteomic and Lipidomic Analysis of Nanoparticle Corona upon Contact with Lung Surfactant Reveals Differences in Protein, but Not Lipid Composition. ACS Nano 2015, 9, 11872–11885. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo, A.; Cruz, A.; Pérez-Gil, J. Pulmonary surfactant and nanocarriers: Toxicity versus combined nanomedical applications. Biochim. Biophys. Acta Biomembr. 2017, 1859, 1740–1748. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.; Bai, X.; Hu, G.; Zuo, Y.Y. Unveiling the Molecular Structure of Pulmonary Surfactant Corona on Nanoparticles. ACS Nano 2017, 11, 6832–6842. [Google Scholar] [CrossRef]
- Wan, F.; Nylander, T.; Foged, C.; Yang, M.; Baldursdottir, S.G.; Nielsen, H.M. Qualitative and quantitative analysis of the biophysical interaction of inhaled nanoparticles with pulmonary surfactant by using quartz crystal microbalance with dissipation monitoring. J. Colloid Interface Sci. 2019, 545, 162–171. [Google Scholar] [CrossRef]
- Kharazian, B.; Hadipour, N.L.; Ejtehadi, M.R. Understanding the nanoparticle–protein corona complexes using computational and experimental methods. Int. J. Biochem. Cell Biol. 2016, 75, 162–174. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.H.; Lee, B.-J. Protein corona: A new approach for nanomedicine design. Int. J. Nanomed. 2017, 12, 3137–3151. [Google Scholar] [CrossRef] [PubMed]
- Mornet, S.; Portier, J.; Duguet, E. A method for synthesis and functionalization of ultrasmall superparamagnetic covalent carriers based on maghemite and dextran. J. Magn. Magn. Mater. 2005, 293, 127–134. [Google Scholar] [CrossRef]
- Kasper, J.Y.; Feiden, L.; Hermanns, M.I.; Bantz, C.; Maskos, M.; Unger, R.E.; Kirkpatrick, C.J. Pulmonary surfactant augments cytotoxicity of silica nanoparticles: Studies on an in vitro air–blood barrier model. Beilstein J. Nanotechnol. 2015, 6, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Behzadi, S.; Serpooshan, V.; Sakhtianchi, R.; Müller, B.; Landfester, K.; Crespy, D.; Mahmoudi, M. Protein corona change the drug release profile of nanocarriers: The “overlooked” factor at the nanobio interface. Colloids Surf. B 2014, 123, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Evora, C.; Soriano, I.; Rogers, R.A.; Shakesheff, K.M.; Hanes, J.; Langer, R. Relating the phagocytosis of microparticles by alveolar macrophages to surface chemistry: The effect of 1,2-dipalmitoylphosphatidylcholine. J. Control. Release 1998, 51, 143–152. [Google Scholar] [CrossRef]
- Lesniak, A.; Fenaroli, F.; Monopoli, M.P.; Åberg, C.; Dawson, K.A.; Salvati, A. Effects of the presence or absence of a protein corona on silica nanoparticle uptake and impact on cells. ACS Nano 2012, 6, 5845–5857. [Google Scholar] [CrossRef] [PubMed]
- Stringer, B.; Imrich, A.; Kobzik, L. Lung Epithelial Cell (A549) Interaction with Unopsonized Environmental Particulates: Quantitation of Particle-Specific Binding and IL-8 Production. Exp. Lung Res. 1996, 22, 495–508. [Google Scholar] [CrossRef] [PubMed]
- Theodorou, I.G.; Ruenraroengsak, P.; Gow, A.; Schwander, S.; Zhang, J.J.; Chung, K.F.; Tetley, T.D.; Ryan, M.P.; Porter, A.E. Effect of pulmonary surfactant on the dissolution, stability and uptake of zinc oxide nanowires by human respiratory epithelial cells. Nanotoxicology 2016, 10, 1351–1362. [Google Scholar] [CrossRef]
- Ruge, C.A.; Schaefer, U.F.; Herrmann, J.; Kirch, J.; Cañadas, O.; Echaide, M.; Pérez-Gil, J.; Casals, C.; Müller, R.; Lehr, C.-M. The interplay of lung surfactant proteins and lipids assimilates the macrophage clearance of nanoparticles. PLoS ONE 2012, 7, e40775. [Google Scholar] [CrossRef]
- Maretti, E.; Rossi, T.; Bondi, M.; Croce, M.A.; Hanuskova, M.; Leo, E.; Sacchetti, F.; Iannuccelli, V. Inhaled Solid Lipid Microparticles to target alveolar macrophages for tuberculosis. Int. J. Pharm. 2014, 462, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Maretti, E.; Rustichelli, C.; Romagnoli, M.; Balducci, A.G.; Buttini, F.; Sacchetti, F.; Leo, E.; Iannuccelli, V. Solid Lipid Nanoparticle assemblies (SLNas) for an anti-TB inhalation treatment—A Design of Experiments approach to investigate the influence of pre-freezing conditions on the powder respirability. Int. J. Pharm. 2016, 511, 669–679. [Google Scholar] [CrossRef] [PubMed]
- Marques, M.R.C.; Loebenberg, R.; Almukainzi, M. Simulated Biological Fluids with Possible Application in Dissolution Testing. Dissolut. Technol. 2011, 18, 15–28. [Google Scholar] [CrossRef]
- European Pharmacopoeia Bulk density and tapped density of powders. 2.9.34. In European Pharmacopoeia; Council of Europe: Strasbourg, France, 2017.
- Briggs, D.; Seah, M.P. Practical Surface Analysis: Auger and X-ray Photoelectron Spectroscopy, 2nd ed.; John Wiley & Son Ltd.: New York, NY, USA, 1990. [Google Scholar]
- Goldstein, J.; Newbury, D.E.; Echlin, P.; Joy, D.C.; Romig, A.D., Jr.; Lyman, C.E.; Fiori, C.; Lifshin, E. Scanning Electron Microscopy and X-ray Microanalysis: A Text for Biologists, Materials Scientists, and Geologists, 2nd ed.; Springer: New York, NY, USA, 1992; ISBN 978-1-4612-7653-1. [Google Scholar]
- Pick, N.; Cameron, S.; Arad, D.; Av-Gay, Y. Screening of compounds toxicity against human Monocytic cell line-THP-1 by flow cytometry. Biol. Proced. 2004, 6, 220–225. [Google Scholar] [CrossRef] [PubMed]
- Ghadiri, M.; Young, P.M.; Traini, D. Strategies to Enhance Drug Absorption via Nasal and Pulmonary Routes. Pharmaceutics 2019, 11, 113. [Google Scholar] [CrossRef] [PubMed]
- Peng, T.; Lin, S.; Niu, B.; Wang, X.; Huang, Y.; Zhang, X.; Li, G.; Pan, X.; Wu, C. Influence of physical properties of carrier on the performance of dry powder inhalers. Acta Pharm. Sin. B 2016, 6, 308–318. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.M.; Martin, G.P.; Marriott, C.; Pritchard, J. The influence of carrier morphology on drug delivery by dry powder inhalers. Int. J. Pharm. 2000, 200, 93–106. [Google Scholar] [CrossRef]
- Ikegami, K.; Kawashima, Y.; Takeuchi, H.; Yamamoto, H.; Isshiki, N.; Momose, D.; Ouchi, K. Improved Inhalation Behavior of Steroid KSR-592 in Vitro with Jethaler® by Polymorphic Transformation to Needle-Like Crystals (β-Form). Pharm. Res. 2002, 19, 1439–1445. [Google Scholar] [CrossRef] [PubMed]
- Chew, N.Y.K.; Tang, P.; Chan, H.-K.; Raper, J.A. How much particle surface corrugation is sufficient to improve aerosol performance of powders? Pharm. Res. 2005, 22, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Son, Y.-J.; McConville, J.T. A new respirable form of rifampicin. Eur. J. Pharm. Biopharm. 2011, 78, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Hamishehkar, H.; Rahimpour, Y.; Javadzadeh, Y. The Role of Carrier in Dry Powder Inhaler. In Recent Advances in Novel Drug Carrier Systems; Sezer, A.D., Ed.; InTechOpen: London, UK, 2012; ISBN 978-953-51-0810-8. [Google Scholar]
- Wang, Y.-B.; Watts, A.B.; Peters, J.I.; Williams, R.O. The impact of pulmonary diseases on the fate of inhaled medicines—A review. Int. J. Pharm. 2014, 461, 112–128. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Liao, J.; Shao, X.; Li, Q.; Lin, Y. The Effect of shape on Cellular Uptake of Gold Nanoparticles in the forms of Stars, Rods, and Triangles. Sci. Rep. 2017, 7, 3827. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharjee, S.; de Haan, L.H.J.; Evers, N.M.; Jiang, X.; Marcelis, A.T.M.; Zuilhof, H.; Rietjens, I.M.C.M.; Alink, G.M. Role of surface charge and oxidative stress in cytotoxicity of organic monolayer-coated silicon nanoparticles towards macrophage NR8383 cells. Part. Fibre Toxicol. 2010, 7, 25. [Google Scholar] [CrossRef] [PubMed]
- Yue, Z.-G.; Wei, W.; Lv, P.-P.; Yue, H.; Wang, L.-Y.; Su, Z.-G.; Ma, G.-H. Surface charge affects cellular uptake and intracellular trafficking of chitosan-based nanoparticles. Biomacromolecules 2011, 12, 2440–2446. [Google Scholar] [CrossRef] [PubMed]
- Edwards, D.A.; Hanes, J.; Caponetti, G.; Hrkach, J.; Ben-Jebria, A.; Eskew, M.L.; Mintzes, J.; Deaver, D.; Lotan, N.; Langer, R. Large porous particles for pulmonary drug delivery. Science 1997, 276, 1868–1871. [Google Scholar] [CrossRef] [PubMed]
- Carr, R.L. Evaluating flow properties of solids. Chem. Eng. 1965, 72, 163–168. [Google Scholar]
- Watling, C.P.; Elliott, J.A.; Scruton, C.; Cameron, R.E. Surface modification of lactose inhalation blends by moisture. Int. J. Pharm. 2010, 391, 29–37. [Google Scholar] [CrossRef]
- Heyder, J.; Gebhart, J.; Rudolf, G.; Schiller, C.F.; Stahlhofen, W. Deposition of particles in the human respiratory tract in the size range 0.005–15 μm. J. Aerosol Sci. 1986, 17, 811–825. [Google Scholar] [CrossRef]
- European Pharmacopoeia Preparations for Inhalation. 2.09.18. In European Pharmacopoeia; Council of Europe: Strasbourg, France, 2017.
- Hoe, S.; Ivey, J.W.; Boraey, M.A.; Shamsaddini-Shahrbabak, A.; Javaheri, E.; Matinkhoo, S.; Finlay, W.H.; Vehring, R. Use of a Fundamental Approach to Spray-Drying Formulation Design to Facilitate the Development of Multi-Component Dry Powder Aerosols for Respiratory Drug Delivery. Pharm. Res. 2013, 31, 449–465. [Google Scholar] [CrossRef]
- Zhu, B.; Young, P.M.; Ong, H.X.; Crapper, J.; Flodin, C.; Qiao, E.L.; Phillips, G.; Traini, D. Tuning aerosol performance using the multibreath Orbital® dry powder inhaler device: Controlling delivery parameters and aerosol performance via modification of puck orifice geometry. J. Pharm. Sci. 2015, 104, 2169–2176. [Google Scholar] [CrossRef]
- Qiu, W.; Sun, X.; Wu, C.; Hjort, K.; Wu, Z. A Contact Angle Study of the Interaction between Embedded Amphiphilic Molecules and the PDMS Matrix in an Aqueous Environment. Micromachines 2014, 5, 515–527. [Google Scholar] [CrossRef]
- Treter, J.; Bonatto, F.; Krug, C.; Soares, G.V.; Baumvol, I.J.R.; Macedo, A.J. Washing-resistant surfactant coated surface is able to inhibit pathogenic bacteria adhesion. Appl. Surf. Sci. 2014, 303, 147–154. [Google Scholar] [CrossRef]
- Hills, B.A. The role of lung surfactant. Br. J. Anaesth. 1990, 65, 13–29. [Google Scholar] [CrossRef] [PubMed]
- Rijt, S.H.; van Bein, T.; Meiners, S. Medical nanoparticles for next generation drug delivery to the lungs. Eur. Respir. J. 2014, 44, 765–774. [Google Scholar] [CrossRef] [PubMed]
- Scalia, S.; Young, P.M.; Traini, D. Solid lipid microparticles as an approach to drug delivery. Expert Opin. Drug Deliv. 2015, 12, 583–599. [Google Scholar] [CrossRef]
- Yan, L.; Cirillo, J.D. Infection of murine macrophage cell lines by Legionella pneumophila. FEMS Microbiol. Lett. 2004, 230, 147–152. [Google Scholar] [CrossRef][Green Version]
- Ragusa, J.; Gonzalez, D.; Li, S.; Noriega, S.; Skotak, M.; Larsen, G. Glucosamine/l-lactide copolymers as potential carriers for the development of a sustained rifampicin release system using Mycobacterium smegmatis as a tuberculosis model. Heliyon 2019, 5, e01539. [Google Scholar] [CrossRef]
- He, P.; Tao, J.; Xue, J.; Chen, Y. Cytotoxicity Property of nano-TiO2 Sol and nano-TiO2 Powder. J. Nanomater. 2011, 2011, 20. [Google Scholar] [CrossRef]
- Rabolli, V.; Thomassen, L.C.J.; Uwambayinema, F.; Martens, J.A.; Lison, D. The cytotoxic activity of amorphous silica nanoparticles is mainly influenced by surface area and not by aggregation. Toxicol. Lett. 2011, 206, 197–203. [Google Scholar] [CrossRef]
- Tan, J.L.; Liu, W.; Nelson, C.M.; Raghavan, S.; Chen, C.S. Simple approach to micropattern cells on common culture substrates by tuning substrate wettability. Tissue Eng. 2004, 10, 865–872. [Google Scholar] [CrossRef]
- Hakkimane, S.S.; Shenoy, V.P.; Gaonkar, S.L.; Bairy, I.; Guru, B.R. Antimycobacterial susceptibility evaluation of rifampicin and isoniazid benz-hydrazone in biodegradable polymeric nanoparticles against Mycobacterium tuberculosis H37Rv strain. Int. J. Nanomed. 2018, 13, 4303–4318. [Google Scholar] [CrossRef]
- Geiser, M. Update on macrophage clearance of inhaled micro- and nanoparticles. J. Aerosol Med. Pulm. Drug Deliv. 2010, 23, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Hussell, T.; Bell, T.J. Alveolar macrophages: Plasticity in a tissue-specific context. Nat. Rev. Immunol. 2014, 14, 81–93. [Google Scholar] [CrossRef] [PubMed]
- D’Addio, S.M.; Baldassano, S.; Shi, L.; Cheung, L.; Adamson, D.H.; Bruzek, M.; Anthony, J.E.; Laskin, D.L.; Sinko, P.J.; Prud’homme, R.K. Optimization of cell receptor-specific targeting through multivalent surface decoration of polymeric nanocarriers. J. Control. Release 2013, 168, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Reading, P.C.; Miller, J.L.; Anders, E.M. Involvement of the mannose receptor in infection of macrophages by influenza virus. J. Virol. 2000, 74, 5190–5197. [Google Scholar] [CrossRef]
- Chono, S.; Kaneko, K.; Yamamoto, E.; Togami, K.; Morimoto, K. Effect of surface-mannose modification on aerosolized liposomal delivery to alveolar macrophages. Drug Dev. Ind. Pharm. 2010, 36, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.; Laurent, S.; Tawil, N.; Yahia, L.; Mahmoudi, M. Nanoparticle and Protein Corona. In Protein-Nanoparticle Interactions: The Bio-Nano Interface; Rahman, M., Laurent, S., Tawil, N., Yahia, L., Mahmoudi, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2013; pp. 21–44. ISBN 978-3-642-37555-2. [Google Scholar]


| Physical Characteristics | SLNas/MS | SLNas/MS-ST | SLNas/ST | SLNas/F127 |
|---|---|---|---|---|
| Circularity | 0.6 ± 0.1 a | 0.83 ± 0.03 b | 0.74 ± 0.07 ab | 0.75 ± 0.06 ab |
| Size (nm) | 740 ± 85 a | 309 ± 30 b | 668 ± 25 a | 408 ± 57 b |
| PDI | 0.60 ± 0.05 | 0.30 ± 0.02 | 0.46 ± 0.05 a | 0.46 ± 0.05 a |
| Z potential (mV) | −35.2 ± 0.1 | −40.5 ± 0.9 | −55 ± 2 | −15 ± 0.1 |
| d(BET) (nm) | 730 ± 10 | 900 ± 20 | 1020 ± 50 a | 1090 ± 20 a |
| ρ true (g/cm3) | 1.147 ± 0.001 | 1.1939 ± 0.0008 | 1.247 ± 0.002 | 1.1791 ± 0.0008 |
| ρ bulk (g/cm3) | 0.048 ± 0.001 | 0.079 ± 0.001 | 0.031 ± 0.000 | 0.147 ± 0.005 |
| ρ tapped (g/cm3) | 0.052 ± 0.001 a | 0.086 ± 0.001 | 0.038 ± 0.000 a | 0.29 ± 0.02 |
| Carr’s Index | 9 ± 2 a | 8 ± 2 a | 17 ± 1 a | 50 ± 8 |
| BET area (m2/g) | 7.2 ± 0.10 | 5.6 ± 0.10 | 4.7 ± 0.20 a | 4.67 ± 0.09 a |
| da (nm) | 210 | 288 | 231 | 676 |
| SLNas Samples | ED (%) | FPF (%) |
|---|---|---|
| SLNas/MS | 87 ± 4 a | 38 ± 5 a |
| SLNas/MS-ST | 84 ± 2 ab | 41 ± 5 a |
| SLNas/ST | 83 ± 1 ab | 53 ± 4 |
| SLNas/F127 | 72 ± 8 b | 11.77 ± 0.01 |
| SLNas Samples | DL (%) | EE (%) |
|---|---|---|
| SLNas/MS | 9.2 ± 0.2 | 36.8 ± 0.9 |
| SLNas/MS-ST | 8.7 ± 0.2 | 34.9 ± 0.9 |
| SLNas/ST | 8.9 ± 0.3 | 36 ± 1 |
| SLNas/F127 | 8.4 ± 0.8 | 34 ± 3 |
| SLNas Samples | C | O | N | S | Na | θ (deg) |
|---|---|---|---|---|---|---|
| SLNas/MS | 71 | 25.5 | 3.3 | 0.0 | 0.0 | 51 ± 5 |
| SLNas/MS-ST | 82 | 15.0 | 2.3 | 0.3 | 0.2 | 32 ± 4 |
| SLNas/ST | 89 | 8.4 | 1.1 | 0.7 | 0.6 | 37 ± 8 |
| SLNas/F127 | 86 | 13.3 | 0.6 | 0.0 | 0.0 | 79 ± 2 |
| SLNas Samples | In Saline Solution | In Saline Solution with Curosurf | Fold Increase in Size | ||
|---|---|---|---|---|---|
| Size (nm) | PDI | Size (nm) | PDI | ||
| SLNas/MS | 962 ± 286 | 0.45 ± 0.20 | 1611 ± 272 | 0.4 ± 0.2 | 1.67 |
| SLNas/MS-ST | 356 ± 64 | 0.29 ± 0.13 | 1839 ± 90 | 0.22 ± 0.01 | 5.17 |
| SLNas/ST | 284 ± 36 | 0.27 ± 0.04 | 884 ± 158 | 0.31 ± 0.02 | 3.11 |
| SLNas/F127 | 551 ± 6 a | 0.68 ± 0.05 | 837 ± 272 a | 0.5 ± 0.3 | 1.52 |
| Samples | C | O | Na | P | S | Cl |
|---|---|---|---|---|---|---|
| Curosurf | 71 ± 2 | 22 ± 2 | 1.1 ± 0.1 | 4.6 ± 0.6 | 0.04 ± 0.04 | 1.6 ± 0.2 |
| SLNas/MS | 85 ± 3 | 8 ± 1 | 3 ± 2 | 0.3 ± 0.1 | 0.03 ± 0.04 | 4 ± 2 |
| SLNas/MS-ST | 83 ± 3 | 8 ± 3 | 4 ± 3 | 0.3 ± 0.2 | 0.01 ± 0.01 | 5 ± 3 |
| SLNas/ST | 91 ± 1 | 8 ± 1 | 0.6 ± 0.2 | 0.26 ± 0.06 | 0.02 ± 0.02 | 0.7 ± 0.2 |
| SLNas/F127 | 94.39 ± 0.05 | 3.94 ± 0.3 | 0.7 ± 0.1 | 0.02 ± 0.01 | 0.01 ± 0.00 | 0.9 ± 0.2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maretti, E.; Rustichelli, C.; Lassinantti Gualtieri, M.; Costantino, L.; Siligardi, C.; Miselli, P.; Buttini, F.; Montecchi, M.; Leo, E.; Truzzi, E.; et al. The Impact of Lipid Corona on Rifampicin Intramacrophagic Transport Using Inhaled Solid Lipid Nanoparticles Surface-Decorated with a Mannosylated Surfactant. Pharmaceutics 2019, 11, 508. https://doi.org/10.3390/pharmaceutics11100508
Maretti E, Rustichelli C, Lassinantti Gualtieri M, Costantino L, Siligardi C, Miselli P, Buttini F, Montecchi M, Leo E, Truzzi E, et al. The Impact of Lipid Corona on Rifampicin Intramacrophagic Transport Using Inhaled Solid Lipid Nanoparticles Surface-Decorated with a Mannosylated Surfactant. Pharmaceutics. 2019; 11(10):508. https://doi.org/10.3390/pharmaceutics11100508
Chicago/Turabian StyleMaretti, Eleonora, Cecilia Rustichelli, Magdalena Lassinantti Gualtieri, Luca Costantino, Cristina Siligardi, Paola Miselli, Francesca Buttini, Monica Montecchi, Eliana Leo, Eleonora Truzzi, and et al. 2019. "The Impact of Lipid Corona on Rifampicin Intramacrophagic Transport Using Inhaled Solid Lipid Nanoparticles Surface-Decorated with a Mannosylated Surfactant" Pharmaceutics 11, no. 10: 508. https://doi.org/10.3390/pharmaceutics11100508
APA StyleMaretti, E., Rustichelli, C., Lassinantti Gualtieri, M., Costantino, L., Siligardi, C., Miselli, P., Buttini, F., Montecchi, M., Leo, E., Truzzi, E., & Iannuccelli, V. (2019). The Impact of Lipid Corona on Rifampicin Intramacrophagic Transport Using Inhaled Solid Lipid Nanoparticles Surface-Decorated with a Mannosylated Surfactant. Pharmaceutics, 11(10), 508. https://doi.org/10.3390/pharmaceutics11100508







