From Lesions to Viral Clones: Biological and Molecular Diversity amongst Autochthonous Brazilian Vaccinia Virus
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethics Approval
2.2. Cells and Viruses
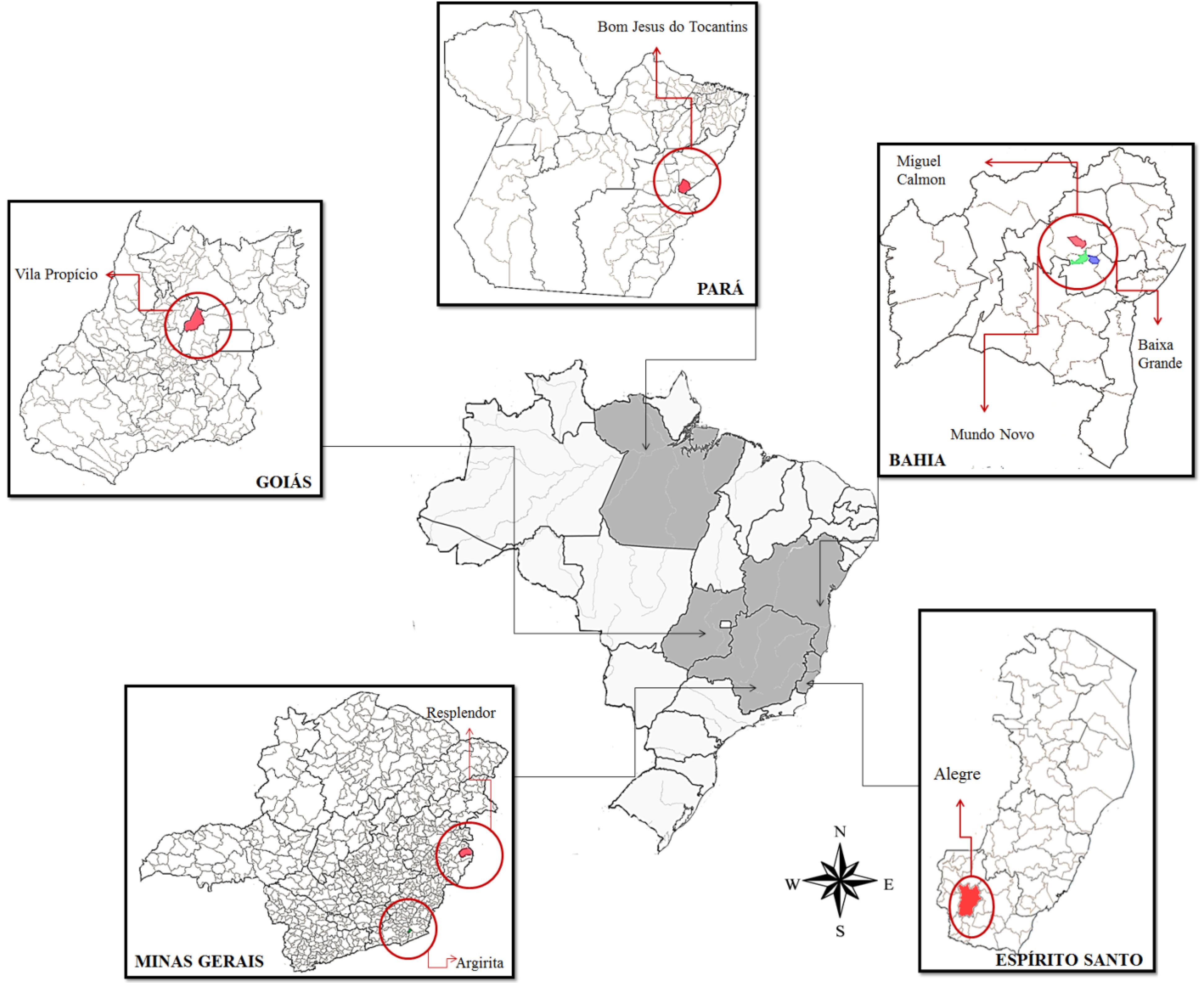
2.3. Clinical Samples
| VACV-BR isolates (abbreviation) | Isolated clones | Source | Isolation state | Isolation year |
|---|---|---|---|---|
| VACV-BABV | VACV-BABV | Bos taurus | Bahia | 2011 |
| Clone 1 to Clone 5 | ||||
| VACV-BAB2V | VACV-BAB2V | Bos taurus | Bahia | 2011 |
| Clone 1 to Clone 5 | ||||
| VACV-BAHV | VACV- BAHV | Homo sapiens | Bahia | 2011 |
| Clone 1 to Clone 5 | ||||
| VACV-GOBV | VACV-GOBV | Bos taurus | Goiás | 2011 |
| Clone 1 to Clone 5 | ||||
| VACV-GOB2V | VACV-GOB2V | Bos taurus | Goiás | 2011 |
| Clone 1 to Clone 5 | ||||
| VACV-ESBV | VACV-ESBV | Bos taurus | Espírito Santo | 2008 |
| Clone 1 to Clone 5 | ||||
| VACV-MGBV | VACV-MGBV | Bos taurus | Minas Gerais | 2005 |
| Clone 1 to Clone 4 | ||||
| VACV-MGHV | VACV-MGHV | Homo sapiens | Minas Gerais | 2011 |
| Clone 1 to Clone 5 | ||||
| VACV-PABV | VACV-PABV | Bos taurus | Pará | 2011 |
| Clone 1 to Clone 5 | ||||
| VACV-PAB2V | VACV-PAB2V | Bos taurus | Pará | 2010 |
| Clone 1 to Clone 4 |
2.4. Sample Processing and Virus Isolation
2.5. Viral Clone Plaque Purification
2.6. Biological Assays
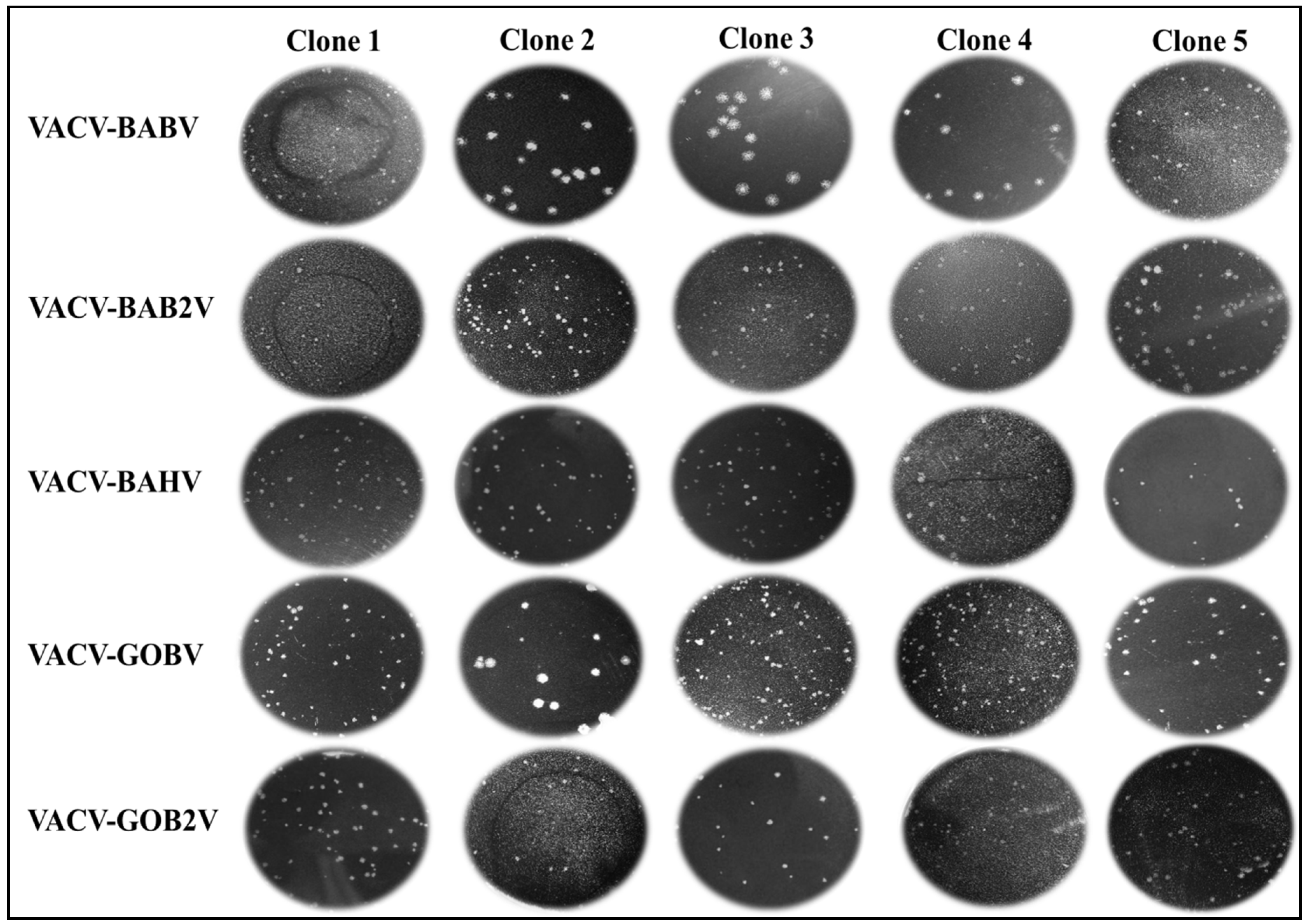
2.6.1. Plaque Phenotype
2.6.2. Comet Phenotype Assay
2.6.3. Growth Curve Assays
2.7. Virulence in BALB/c Mice
2.8. Molecular Assays
Amplification of A56R, A26L and C23L Genes and Phylogenetic Analyses
3. Results
3.1. Viral Clone Isolation and Biological Assays

3.2. Virulence of VACV-Clones in Balb/c Mice

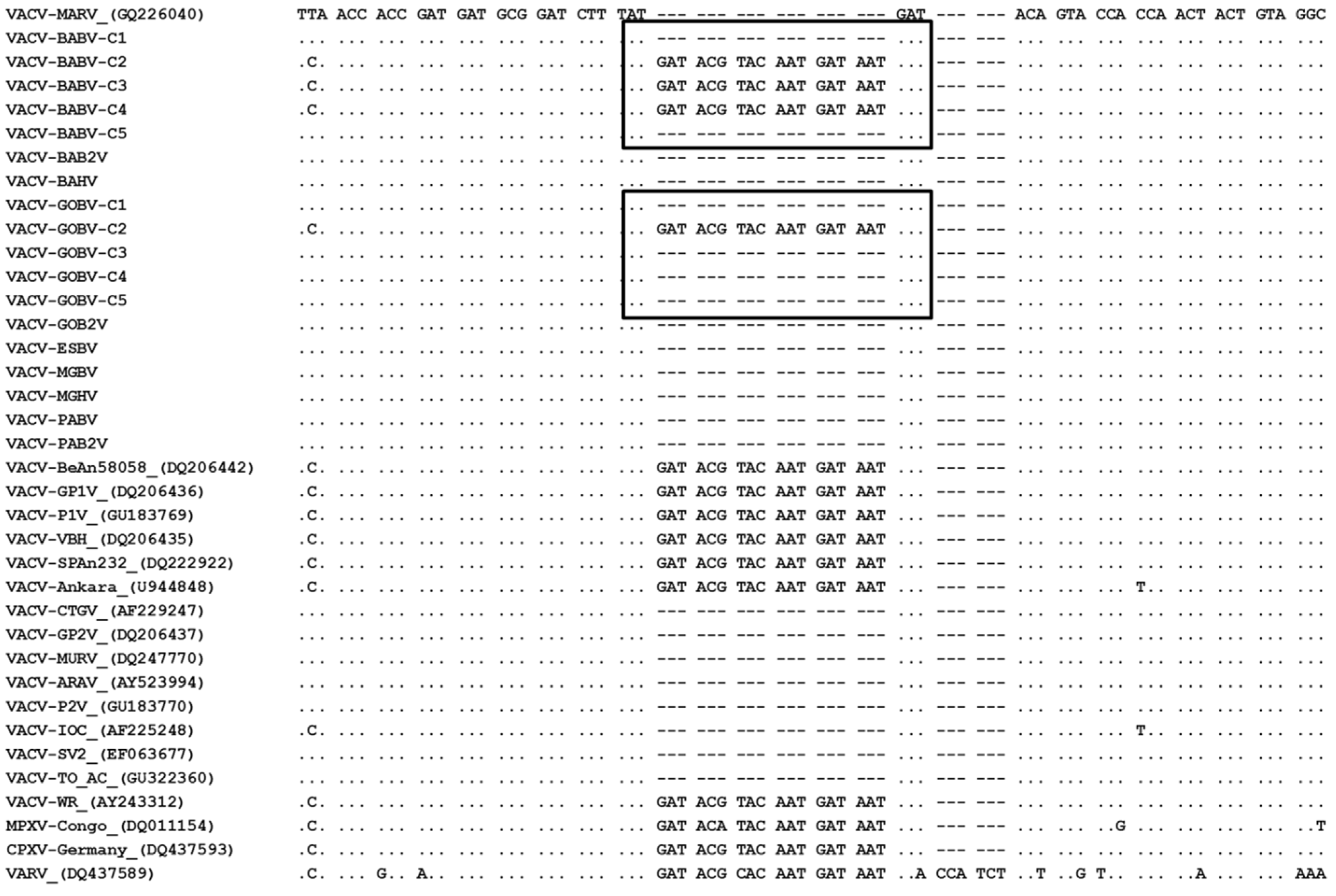
3.3. Phylogeny
| Clones | Genes | Clones | Genes | ||||
|---|---|---|---|---|---|---|---|
| A56R | A26L | C23L | A56R | A26L | C23L | ||
| BABV-Clone 1 | Group 1 | Group 1 | Group 1 | GOB2V-Clone 1 | Group 1 | Group 1 | Group 1 |
| BABV-Clone 2 | Group 2 | Group 1 | Group 2 | GOB2V-Clone 2 | Group 1 | Group 1 | Group 1 |
| BABV-Clone 3 | Group 2 | Group 2 | Group 2 | GOB2V-Clone 3 | Group 1 | Group 1 | Group 1 |
| BABV-Clone 4 | Group 2 | Group 2 | Group 2 | GOB2V-Clone 4 | Group 1 | Group 1 | Group 1 |
| BABV-Clone 5 | Group 1 | Group 1 | Group 1 | GOB2V-Clone 5 | Group 1 | Group 1 | Group 1 |
| BAB2V-Clone 1 | Group 1 | Group 1 | Group 1 | ESBV-Clone 1 | Group 1 | Group 1 | Group 1 |
| BAB2V-Clone 2 | Group 1 | Group 1 | Group 1 | ESBV-Clone 2 | Group 1 | Group 1 | Group 1 |
| BAB2V-Clone 3 | Group 1 | Group 1 | Group 1 | ESBV-Clone 3 | Group 1 | Group 1 | Group 1 |
| BAB2V-Clone 4 | Group 1 | Group 1 | Group 1 | ESBV-Clone 4 | Group 1 | Group 1 | Group 1 |
| BAB2V-Clone 5 | Group 1 | Group 1 | Group 1 | ESBV-Clone 5 | Group 1 | Group 1 | Group 1 |
| BAHV-Clone 1 | Group 1 | Group 1 | Group 1 | MGHV-Clone 1 | Group 1 | Group 1 | Group 1 |
| BAHV-Clone 2 | Group 1 | Group 1 | Group 1 | MGHV-Clone 2 | Group 1 | Group 1 | Group 2 |
| BAHV-Clone 3 | Group 1 | Group 1 | Group 1 | MGHV-Clone 3 | Group 1 | Group 1 | Group 1 |
| BAHV-Clone 4 | Group 1 | Group 1 | Group 1 | MGHV-Clone 4 | Group 1 | Group 1 | Group 1 |
| BAHV-Clone 5 | Group 1 | Group 1 | Group 1 | MGHV-Clone 5 | Group 1 | Group 1 | Group 1 |
| GOBV-Clone 1 | Group 1 | Group 1 | Group 1 | PABV-Clone 1 | Group 1 | Group 1 | Group 1 |
| GOBV-Clone 2 | Group 2 | Group 2 | Group 2 | PABV-Clone 2 | Group 1 | Group 1 | Group 1 |
| GOBV-Clone 3 | Group 1 | Group 1 | Group 1 | PABV-Clone 3 | Group 1 | Group 1 | Group 1 |
| GOBV-Clone 4 | Group 1 | Group 1 | Group 1 | PABV-Clone 4 | Group 1 | Group 1 | Group 1 |
| GOBV-Clone 5 | Group 1 | Group 1 | Group 1 | PABV-Clone 5 | Group 1 | Group 1 | Group 1 |
| MGBV-Clone 1 | Group 1 | Group 1 | Group 1 | PAB2V-Clone 1 | Group 1 | Group 1 | Group 1 |
| MGBV-Clone 2 | Group 1 | Group 1 | Group 1 | PAB2V-Clone 2 | Group 1 | Group 1 | Group 1 |
| MGBV-Clone 3 | Group 1 | Group 1 | Group 1 | PAB2V-Clone 3 | Group 1 | Group 1 | Group 1 |
| MGBV-Clone 4 | Group 1 | Group 1 | Group 1 | PAB2V-Clone 4 | Group 1 | Group 1 | Group 1 |

4. Discussion
Supplementary Files
Supplementary File 1Supplementary File 2Acknowledgments
Author Contributions
Conflicts of Interest
References and Notes
- Fenner, F.; Henderson, D.A.; Arita, I.; Jezek, A.; Ladnyi, I.D. Smallpox and Its Eradication; World Health Organization Press: Geneva, Switzerland, 1988. [Google Scholar]
- Chen, H.; Chuai, X.; Deng, Y.; Wen, B.; Wang, W.; Xiong, S.; Ruan, L.; Tan, W. Optimisation of prime-boost immunization in mice using novel protein-based and recombinant vaccinia (Tiantan)-based HBV vaccine. PLoS One 2012, 7, e43730. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Zhang, K.; Tan, W.; Wang, Y.; Chen, H.; Wu, X.; Ruan, L. A recombinant DNA and vaccinia virus prime-boost regimen induces potent long-term T-cell responses to HCV in BALB/c mice. Vaccine 2009, 27, 2085–2088. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Du, S.; Li, C.; Wang, Y.; Wang, M.; Li, Y.; Yin, R.; Li, X.; Ren, D.; Qin, Y.; et al. Immunogenicity analysis following human immunodeficiency virus recombinant DNA and recombinant vaccinia virus Tian Tan prime-boost immunization. Sci. China Life Sci. 2013, 56, 531–540. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, M.G.; Carroll, D.S.; Karem, K.L. Factors affecting the likelihood of monkeypox’s emergence and spread in the post-smallpox era. Curr. Opin. Virol. 2012, 2, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Pahlitzsch, R.; Hammarin, A.L.; Widell, A. A case of facial cellulitis and necrotizing lymphadenitis due to cowpox virus infection. Clin. Infect. Dis. 2006, 43, 737–742. [Google Scholar] [CrossRef] [PubMed]
- Karem, K.L.; Reynolds, M.; Braden, Z.; Lou, G.; Bernard, N.; Patton, J.; Damon, I.K. Characterization of acute-phase humoral immunity to monkeypox: Use ofimmunoglobulin M enzyme-linked immunosorbent assay for detection of monkeypox infection during the 2003 North American outbreak. Clin. Diagn Lab. Immunol. 2005, 12, 867–872. [Google Scholar] [PubMed]
- Rimoin, A.W.; Mulembakani, P.M.; Johnston, S.C.; Lloyd Smith, J.O.; Kisalu, N.K.; Kinkela, T.L.; Blumberg, S.; Thomassen, H.A.; Pike, B.L.; Fair, J.N.; et al. Major increase in human monkeypox incidence 30 years after smallpox vaccination campaigns cease in the Democratic Republic of Congo. Proc. Natl. Acad. Sci. USA 2010, 107, 16262–16267. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Hosamani, M.; Balamurugan, V.; Bhanuprakash, V.; Rasool, T.J.; Yadav, M.P. Buffalopox: An emerging and re-emerging zoonosis. Anim. Health Res. Rev. 2007, 8, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Trindade, G.S.; Guedes, M.I.; Drumond, B.P.; Mota, B.E.; Abrahao, J.S.; Lobato, Z.I.; Gomes, J.A.; Corrêa-Oliveira, R.; Nogueira, M.L.; Kroon, E.G.; et al. Zoonotic vaccinia virus: Clinical and immunological characteristics in a naturally infected patient. Clin. Infect. Dis. 2009, 48, 37–40. [Google Scholar] [CrossRef]
- Kroon, E.G.; Mota, B.E.; Abrahão, J.S.; da Fonseca, F.G.; de Souza Trindade, G. Zoonotic Brazilian Vaccinia virus: from field to therapy. Antivir. Res. 2011, 92, 150–163. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, D.B.; Assis, F.L.; Ferreira, P.C.; Bonjardim, C.A.; de Souza Trindade, G.; Kroon, E.G.; Abrahão, J.S. Short Report: Group 1 Vaccinia virus Zoonotic Outbreak in Maranhão State, Brazil. Am. J. Trop. Med. Hyg. 2013, 89, 1142–1145. [Google Scholar] [CrossRef] [PubMed]
- Kalthoff, D.; Bock, W.I.; Hühn, F.; Beer, M.; Hoffmann, B. Fatal cowpox virus infection in cotton-top tamarins (Saguinus oedipus) in Germany. Vector Borne Zoonotic Dis. 2014, 14, 303–305. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.T.; Travassos, C.E.; Ferreira, J.M.; Abrahao, J.S.; Rocha, E.S.; Ferreira, F.V.; dos Santos, J.R.; Bonjardim, C.A.; Ferreira, P.C.; Kroon, E.G. Natural human infections with Vaccinia virus during bovine vaccinia outbreaks. J. Clin. Virol. 2009, 44, 308–313. [Google Scholar] [CrossRef] [PubMed]
- Megid, J.; Appolinário, C.M.; Langoni, H.; Pituco, E.M.; Okuda, L.H. Vaccinia virus in humans and cattle in southwest region of Sao Paulo state, Brazil. Am. J. Trop. Med. Hyg. 2008, 79, 647–665. [Google Scholar] [PubMed]
- Damon, I.K. Poxviruses. In Fields Virology, 6th ed.; Knipe, D.M., Howley, P.M., Eds.; Lippincott, Williams and Wilkins: Philadelphia, PA, USA, 2014; Volume 2, p. 2160. [Google Scholar]
- Batista, V.H.; Scremin, J.; Aguiar, L.M.; Schatzmayr, H.G. Vulvar infection and possible human-to-human transmission of bovine poxvirus disease. Virus Rev. Res. 2009, 14, 1–10. [Google Scholar]
- Oliveira, G.P.; Fernandes, A.T.; Assis, F.L.; Alves, P.A.; Franco Luiz, A.P.M.; Figueiredo, L.B.; Costa de Almeida, C.M.; Travassos, C.E.P.F.; Trindade, G.S.; Abrahão, J.S.; et al. Intrafamilial transmission of Vaccinia virus during a bovine Vaccinia outbreak in Brazil: A new insight in viral transmission chain. Am. J. Trop. Med. Hyg. 2014, 90, 1021–1023. [Google Scholar] [CrossRef] [PubMed]
- Lopes, O.S.; Lacerda, J.P.; Fonseca, I.E.; Castro, D.P.; Forattini, O.P.; Rabello, E.X. CotiaVirus: A New Agent Isolated from Sentinel Mice in Sao Paulo, Brazil. Am. J. Trop. Med. Hyg. 1965, 14, 156–157. [Google Scholar] [PubMed]
- Fonseca, F.G.; Lanna, M.C.; Campos, M.A.; Kitajima, E.W.; Peres, J.N.; Golgher, R.R.; Ferreira, P.C.; Kroon, E.G. Morphological and molecular characterization of the poxvirus BeAn 58058. Arch. Virol. 1998, 143, 1171–1186. [Google Scholar] [CrossRef] [PubMed]
- Fonseca, F.G.; Trindade, G.S.; Silva, R.L.; Bonjardim, C.A.; Ferreira, P.C.; Kroon, E.G. Characterization of a vaccinia-like virus isolated in a Brazilian forest. J. Gen. Virol. 2002, 83, 223–228. [Google Scholar] [PubMed]
- Damaso, C.R.; Esposito, J.J.; Condit, R.C.; Moussatche, N. An emergent poxvirus from humans and cattle in Rio de Janeiro State: Cantagalo virus may derive from Brazilian smallpox vaccine. Virology 2000, 277, 439–449. [Google Scholar] [CrossRef] [PubMed]
- Trindade, G.S.; da Fonseca, F.G.; Marques, J.T.; Diniz, S.; Leite, J.A.; de Bodt, S.; van der Peer, Y.; Bonjardim, C.A.; Ferreira, P.C.; Kroon, E.G. Belo Horizonte virus: a vaccinia-like virus lacking the A-type inclusion body gene isolated from infected mice. J. Gen. Virol. 2004, 85, 2015–2021. [Google Scholar] [CrossRef] [PubMed]
- Assis, F.L.; Borges, I.A.; Ferreira, P.C.; Bonjardim, C.A.; Trindade, G.S.; Lobato, Z.I.; Guedes, M.I.; Mesquita, V.; Kroon, E.G.; Abrahão, J.S. Group 2 vaccinia virus, Brazil. Emerg. Infect. Dis. 2012, 18, 2035–2038. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, J.M.; Drumond, B.P.; Guedes, M.I.; Pascoal-Xavier, M.A.; Almeida-Leite, C.M.; Arantes, R.M.; Mota, B.E.; Abrahão, J.S.; Alves, P.A.; Oliveira, F.M.; et al. Virulence in Murine Model Shows the Existence of Two Distinct Populations of Brazilian Vaccinia virus Strains. PLoS One 2008, 3, e3043. [Google Scholar] [CrossRef] [PubMed]
- Trindade, G.S.; Lobato, Z.I.; Drumond, B.P.; Leite, J.A.; Trigueiro, R.C.; Guedes, M.I.; Fonseca, F.G.; Santos, J.R.; Bonjardim, C.A.; Ferreira, P.C.; Kroon, E.G. Shortreport: Isolation of two vaccinia virus strains froma single bovine vaccinia outbreak in rural area fromBrazil: Implications on the emergence of zoonotic orthopoxviruses. Am. J. Trop. Med. Hyg. 2006, 75, 486–490. [Google Scholar] [PubMed]
- Drumond, B.P.; Leite, J.A.; da Fonseca, F.G.; Bonjardim, C.A.; Ferreira, P.C.; Kroon, E.G. Brazilian Vaccinia virus strains are genetically divergent and differ from the Lister vaccine strain. Microbes Infect. 2008, 10, 185–197. [Google Scholar] [CrossRef] [PubMed]
- Campos, R.K.; Brum, M.C.; Nogueira, C.E.; Drumond, B.P.; Alves, P.A.; Siqueira-Lima, L.; Assis, F.L.; Trindade, G.S.; Bonjardim, C.A.; Ferreira, P.C.; et al. Assessing the variability of Brazilian Vaccinia virus isolates from a horse exanthematic lesion: Coinfection with distinct viruses. Arch. Virol. 2011, 156, 275–283. [Google Scholar] [CrossRef] [PubMed]
- Leite, J.A.; Drumond, B.P.; de Souza Trindade, G.; Bonjardim, C.A.; Ferreira, P.C.; Kroon, E.G. Brazilian Vaccinia virus strains show genetic polymorphism at the ati gene. Virus Genes 2007, 35, 531–539. [Google Scholar] [CrossRef] [PubMed]
- Assis, F.L.; Almeida, G.M.; Oliveira, D.B.; Franco-Luiz, A.P.; Campos, R.K.; Guedes, M.I.; Fonseca, F.G.; Trindade, G.S.; Drumond, B.P.; Kroon, E.G.; et al. Characterization of a new Vaccinia virus isolate reveals the C23L gene as a putative genetic marker for autochthonous Group 1 Brazilian Vaccinia virus. PLoS One 2012, 11, e50413. [Google Scholar] [CrossRef]
- Trindade, G.S.; da Fonseca, F.G.; Marques, J.T.; Nogueira, M.L.; Mendes, L.C.; Borges, A.S.; Peiró, J.R.; Pituco, E.M.; Bonjardim, C.A.; Ferreira, P.C.; et al. Araçatuba virus: A vaccinialike virus associated with infection in humans and cattle. Emerg Infect. Dis. 2003, 9, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Abrahão, J.S.; Guedes, M.I.; Trindade, G.S.; Fonseca, F.G.; Campos, R.K.; Mota, B.F.; Lobato, Z.I.; Fernandes, A.T.; Rodrigues, G.O.; Lima, L.S.; et al. One more piece in the VACV ecological puzzle: Could peridomestic rodents be the link between wildlife and bovine vaccinia outbreaks in Brazil? PLoS One 2009, 19, e7428. [Google Scholar] [CrossRef]
- Reed, K.D.; Melski, J.W.; Graham, M.B.; Regnery, R.L.; Sotir, M.J.; Wegner, M.V.; Kazmierczak, J.J.; Stratman, E.J.; Li, Y.; Fairley, J.A.; et al. The detection of monkeypox in humans in the Western Hemisphere. N. Engl. J. Med. 2004, 350, 342–350. [Google Scholar] [CrossRef] [PubMed]
- Gubser, C.; Hue, S.; Kellam, P.; Smith, G.L. Poxvirus genomes: A phylogenetic analysis. J. Gen. Virol. 2004, 85, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Li, G.; Liszewski, M.K.; Atkinson, J.P.; Jahrling, P.B.; Feng, Z.; Schriewer, J.; Buck, C.; Wang, C.; Lefkowitz, E.J.; et al. Virulence differences between monkeypox virus isolates from West Africa and the Congo basin. Virology 2005, 340, 46–63. [Google Scholar] [CrossRef] [PubMed]
- Esposito, J.J.; Sammons, S.A.; Frace, A.M.; Osborne, J.D.; Olsen-Rasmussen, M.; Zhang, M.; Govil, D.; Damon, I.K.; Kline, R.; Laker, M.; et al. Genome sequence diversity and clues to the evolution of variola (smallpox) virus. Science 2006, 313, 807–812. [Google Scholar] [CrossRef] [PubMed]
- Emerson, G.L.; Li, Y.; Frace, M.A.; Olsen-Rasmussen, M.A.; Khristova, M.L.; Govil, D.; Sammons, S.A.; Regnery, R.L.; Karem, K.L.; Damon, I.K.; et al. The phylogenetics and ecology of the orthopoxviruses endemic to North America. PLoS One 2009, 4, e7666. [Google Scholar] [CrossRef] [PubMed]
- Gubser, C.; Hue, S.; Kellam, P.; Smith, G.L.; Hendrickson, R.C.; Wang, C.; Hatcher, E.L.; Lefkowitz, E.J. Orthopoxvirus genome evolution: The role of gene loss. Viruses 2010, 2, 1933–1967. [Google Scholar] [CrossRef] [PubMed]
- Kugelman, J.R.; Johnston, S.C.; Mulembakani, P.M.; Kisalu, N.; Lee, M.S.; Koroleva, G.; McCarthy, S.E.; Gestole, M.C.; Wolfe, N.D.; Fair, J.N.; et al. Genomic variability of monkeypox virus among humans, Democratic Republic of the Congo. Emerg. Infect. Dis. 2014, 20, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Okeke, M.I.; Okoli, A.S.; Nilssen, Ø.; Moens, U.; Tryland, M.; Bøhn, T.; Traavik, T. Molecular characterization and phylogenetics of Fennoscandian cowpox virus isolates based on the p4c and atip genes. Virol. J. 2014, 11, 119–120. [Google Scholar] [CrossRef] [PubMed]
- Osborne, J.D.; Da Silva, M.; Frace, A.M.; Sammons, S.A.; Olsen-Rasmussen, M.; Upton, C.; Buller, R.M.; Chen, N.; Feng, Z.; Roper, R.L.; et al. Genomic differences of Vaccinia virus clones from Dryvax smallpox vaccine: The Dryvax-like ACAM2000 and the mouse neurovirulent Clone-3. Vaccine 2007, 25, 8807–8832. [Google Scholar] [CrossRef] [PubMed]
- Garcel, A.; Perino, J.; Crance, J.M.; Drillien, R.; Garin, D.; Favier, A.L. Phenotypic and genetic diversity ofthe traditional Lister smallpox vaccine. Vaccine 2009, 27, 708–717. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, S.; Sakiyama, T.; Hasegawa, H.; Saijo, M.; Maeda, A.; Kurane, I.; Maeno, G.; Kimura, J.; Hirama, C.; Yoshida, T.; et al. An attenuated LC16m8 smallpox vaccine: Analysis of fullgenome sequence and induction of immune protection. J. Virol. 2005, 79, 11873–11891. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.; Upton, C.; Hazes, B.; Evans, D.H. Genomic analysis of the vaccinia virus strain variants found in Dryvax vaccine. J. Virol. 2011, 85, 13049–13060. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.; Liang, M.; Evans, D.H. Genomic analysis of vaccinia virus strain TianTan provides new insights into the evolution and evolutionary relationships between Orthopoxviruses. Virology 2013, 442, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Fenner, F.; Comben, B.M. Genetic studies with mammalian poxviruses. I.Demonstration of recombination between two strains of vaccina virus. Virology 1958, 5, 530–548. [Google Scholar] [CrossRef] [PubMed]
- Bedson, H.S.; Dumbell, K.R. Hybrids Derived from the Viruses of Variola Major and Cowpox. J. Hyg. 1964, 62, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.; Evans, D.H. Genome scale patterns of recombination between coinfecting vaccinia viruses. J. Virol. 2014, 88, 5277–5286. [Google Scholar] [CrossRef] [PubMed]
- Campos, M.A.S.; Kroon, E.G. Critical period for reversible block of vaccinia virus replication. Rev. Braz. Microbiol. 1993, 24, 104–110. [Google Scholar]
- Joklik, W.K. The purification of four strains of poxvirus. Virology 1962, 18, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Sambrook, J.; Russell, D.W. Molecular Cloning: A Laboratory Manual, 3rd ed.; Nolan, C., Ford, N., Eds.; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2001. [Google Scholar]
- Trindade, G.S.; Li, Y.; Olson, V.A.; Emerson, G.; Regnery, R.L.; da Fonseca, F.G.; Kroon, E.G.; Damon, I.K. Real-time PCR assay to identify variants of Vaccinia virus: Implications for the diagnosis of bovine vaccinia in Brazil. J. Virol. Methods 2008, 152, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Ropp, S.L.; Jin, Q.; Knight, J.C.; Massung, R.F.; Esposito, J.J. PCR strategy for identification and differentiation of small pox and other Orthopoxviruses. J. Clin. Microbiol. 1995, 33, 2069–2076. [Google Scholar] [PubMed]
- Trindade, G.S.; Emerson, G.L.; Carroll, D.S.; Kroon, E.G.; Damon, I.K. Brazilian Vaccinia Viruses and Their Origins. Emerg. Infect. Dis. 2007, 13, 965–972. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira, G.; Assis, F.; Almeida, G.; Albarnaz, J.; Lima, M.; Andrade, A.C.; Calixto, R.; Oliveira, C.; Diomedes Neto, J.; Trindade, G.; et al. From Lesions to Viral Clones: Biological and Molecular Diversity amongst Autochthonous Brazilian Vaccinia Virus. Viruses 2015, 7, 1218-1237. https://doi.org/10.3390/v7031218
Oliveira G, Assis F, Almeida G, Albarnaz J, Lima M, Andrade AC, Calixto R, Oliveira C, Diomedes Neto J, Trindade G, et al. From Lesions to Viral Clones: Biological and Molecular Diversity amongst Autochthonous Brazilian Vaccinia Virus. Viruses. 2015; 7(3):1218-1237. https://doi.org/10.3390/v7031218
Chicago/Turabian StyleOliveira, Graziele, Felipe Assis, Gabriel Almeida, Jonas Albarnaz, Maurício Lima, Ana Cláudia Andrade, Rafael Calixto, Cairo Oliveira, José Diomedes Neto, Giliane Trindade, and et al. 2015. "From Lesions to Viral Clones: Biological and Molecular Diversity amongst Autochthonous Brazilian Vaccinia Virus" Viruses 7, no. 3: 1218-1237. https://doi.org/10.3390/v7031218
APA StyleOliveira, G., Assis, F., Almeida, G., Albarnaz, J., Lima, M., Andrade, A. C., Calixto, R., Oliveira, C., Diomedes Neto, J., Trindade, G., Ferreira, P. C., Kroon, E. G., & Abrahão, J. (2015). From Lesions to Viral Clones: Biological and Molecular Diversity amongst Autochthonous Brazilian Vaccinia Virus. Viruses, 7(3), 1218-1237. https://doi.org/10.3390/v7031218








