Expression of APOBEC3 Lentiviral Restriction Factors in Cats
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Cells and Culture Conditions
2.3. Viruses
2.4. RNA and DNA Extractions and cDNA Synthesis
2.5. Quantification of Feline APOBEC3 Gene Expression by qPCR
2.6. Assessment of Cytokine and Mitogen Effects on APOBEC3 Gene Expression
2.7. In Vitro FIV Infection
2.8. Assessment of APOBEC3 Gene Expression and FIV Infection in Cat Blood Cell Subtypes
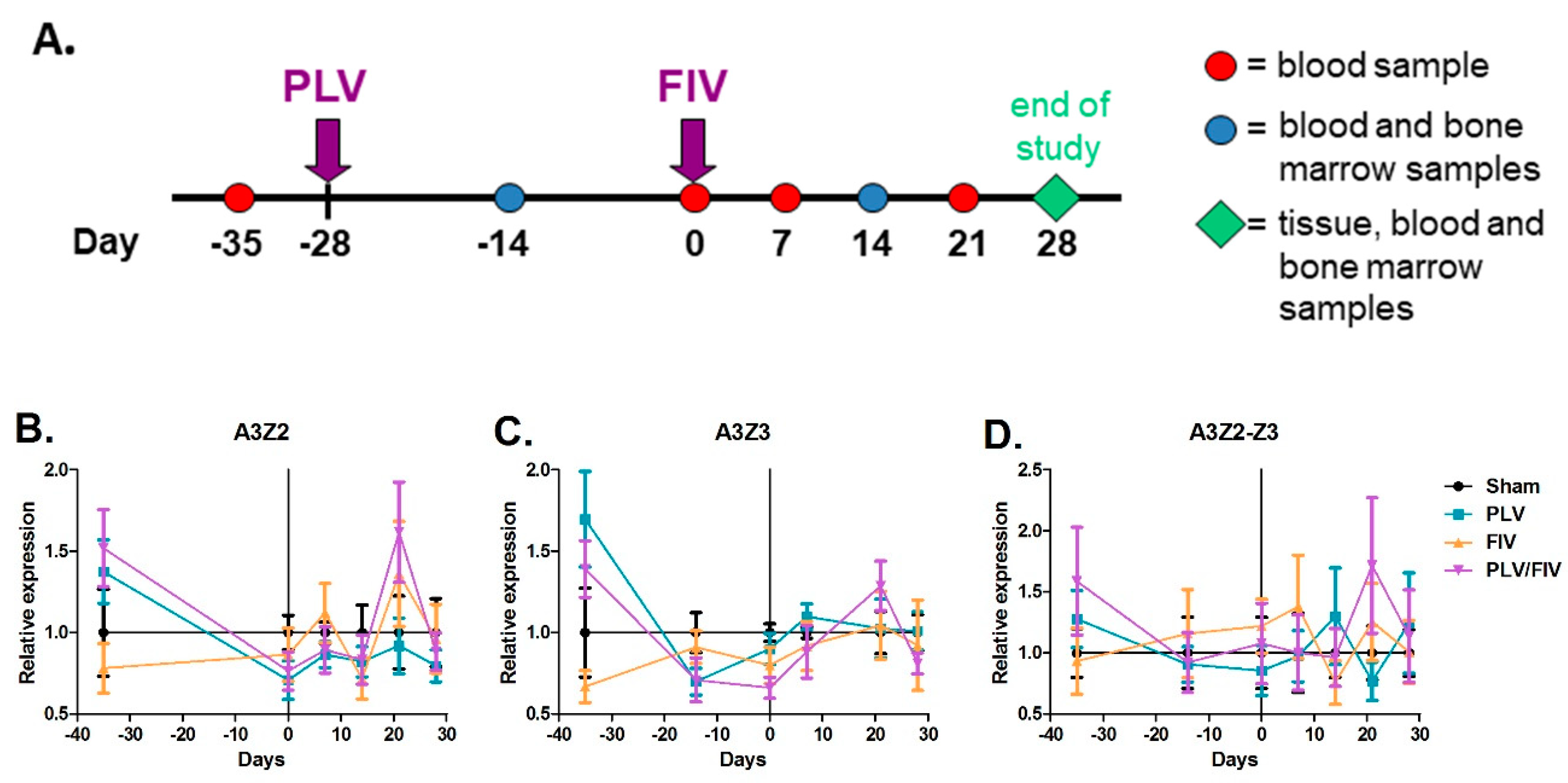
2.9. In Vivo FIV and PLV Co-Infection
2.10. Statistical Analyses
3. Results
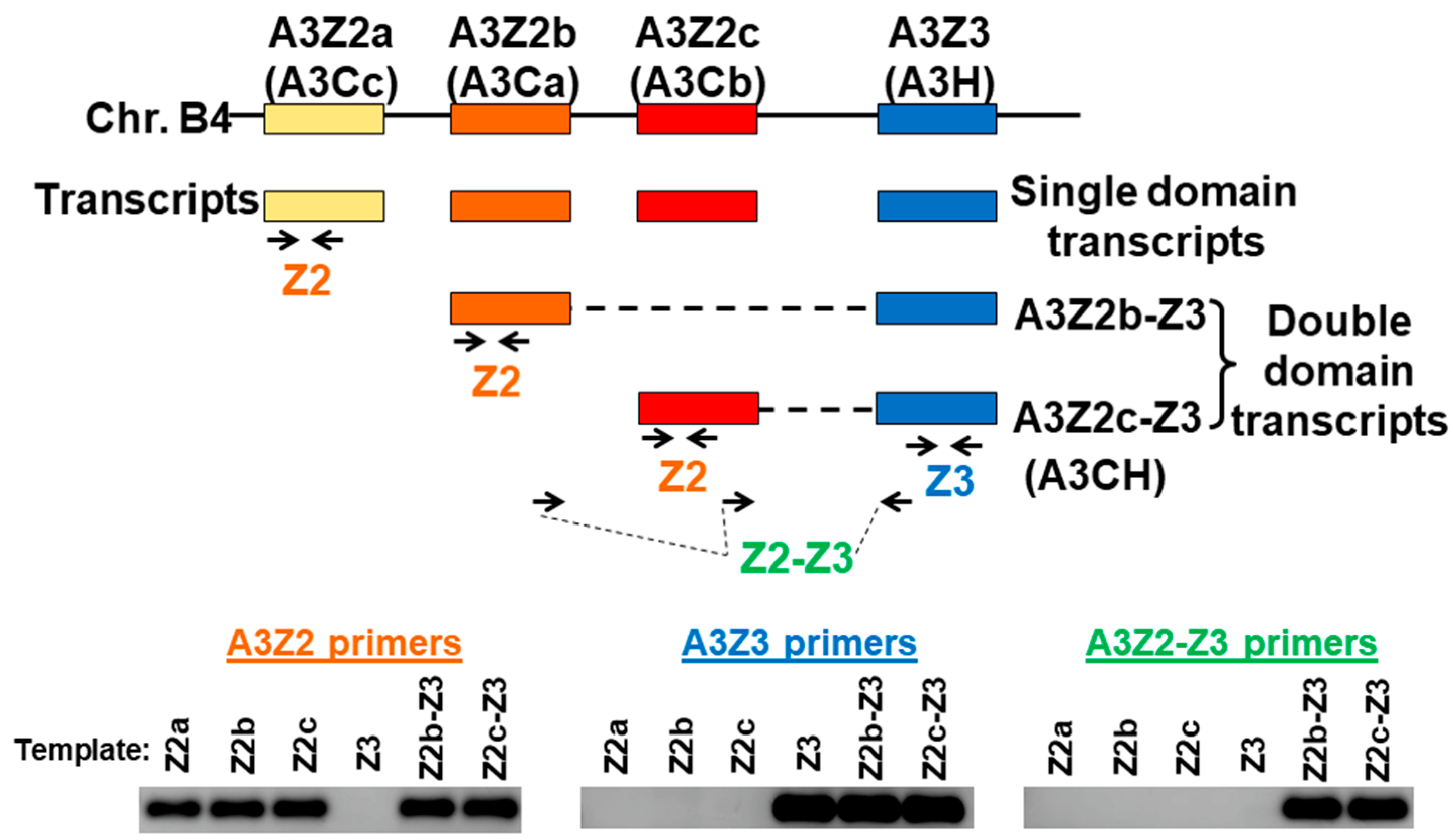
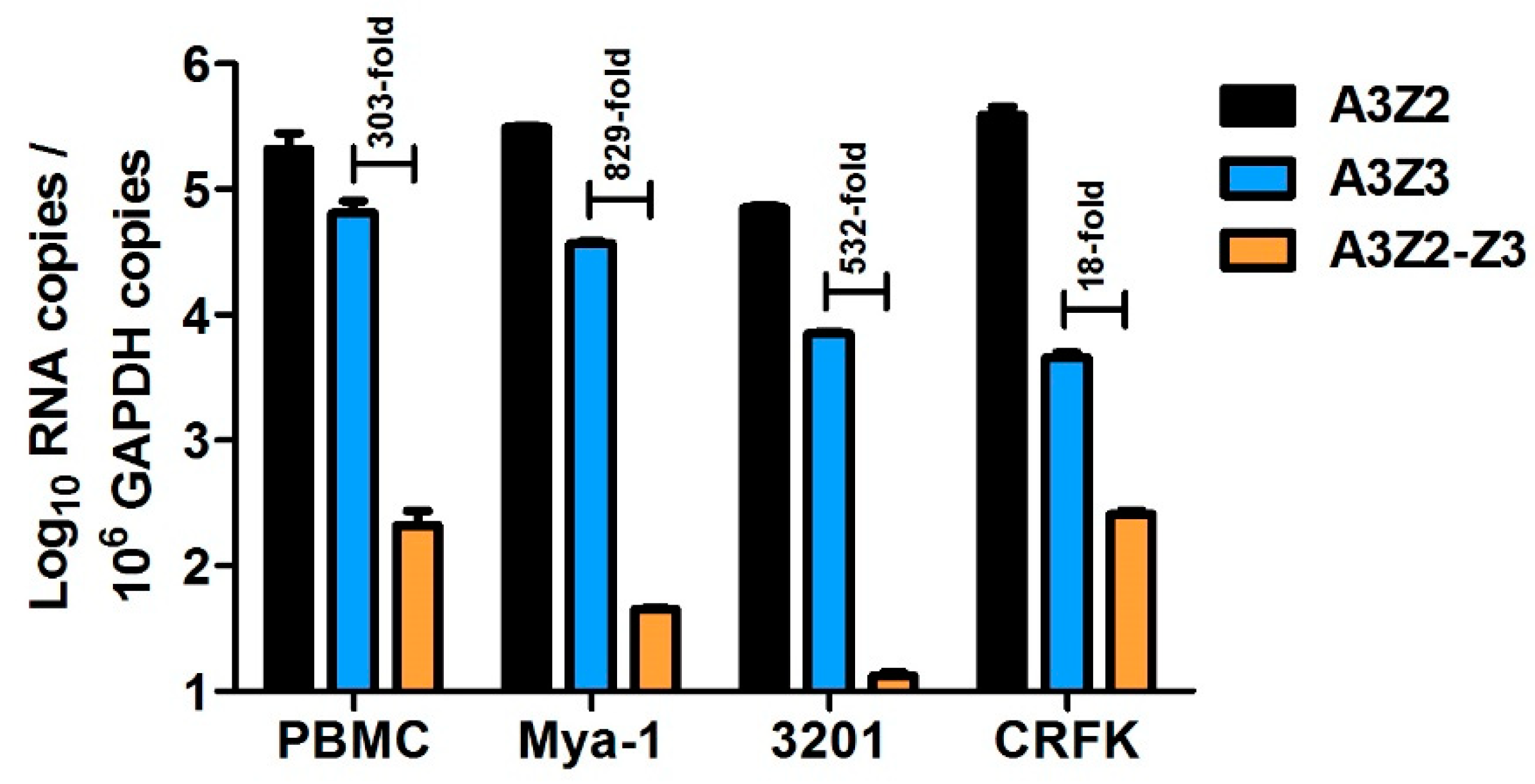
3.1. Expression of APOBEC3 Genes in Cat PBMCs and Cell Lines
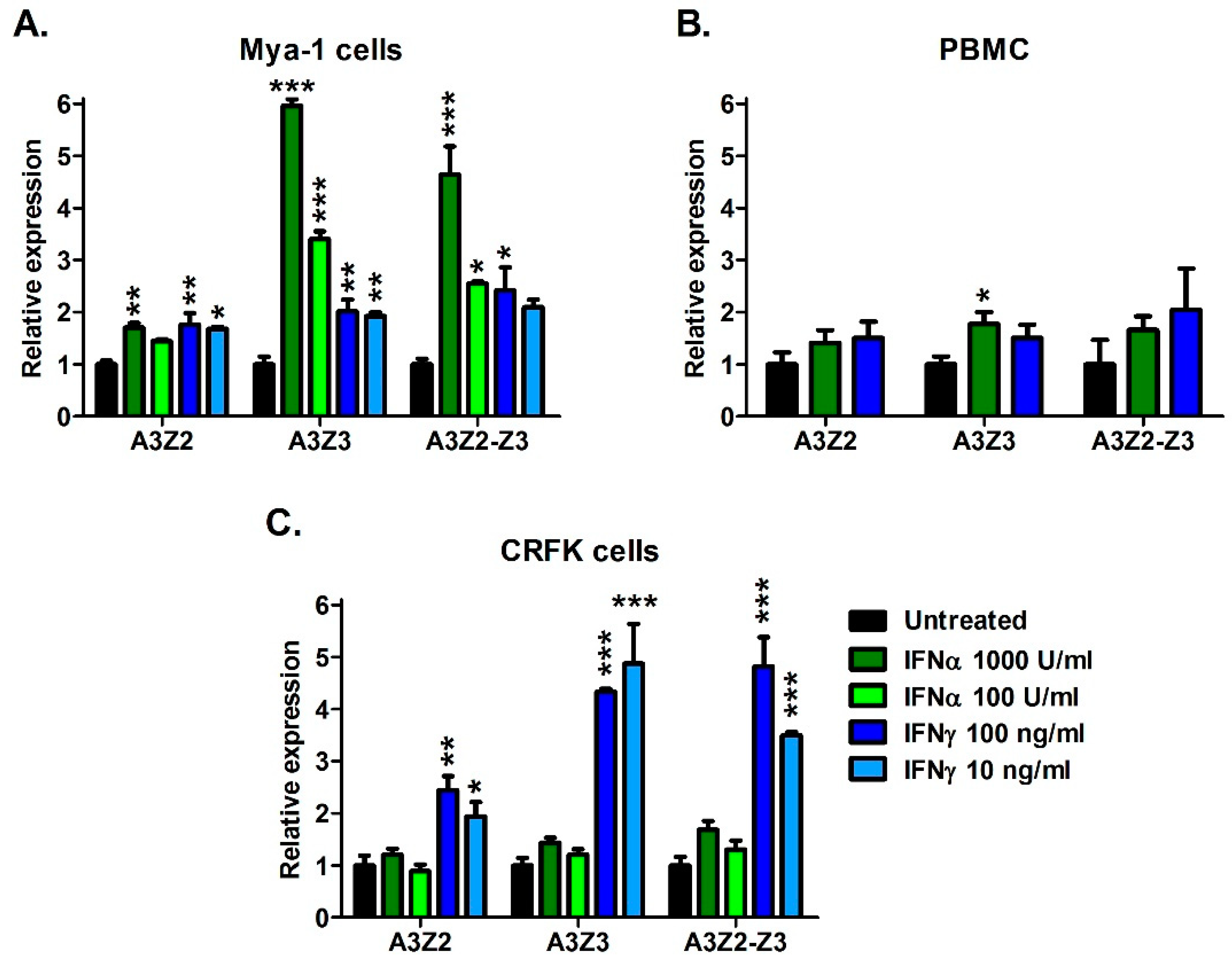
3.2. Cytokines Regulate APOBEC3 Gene Expression in Vitro
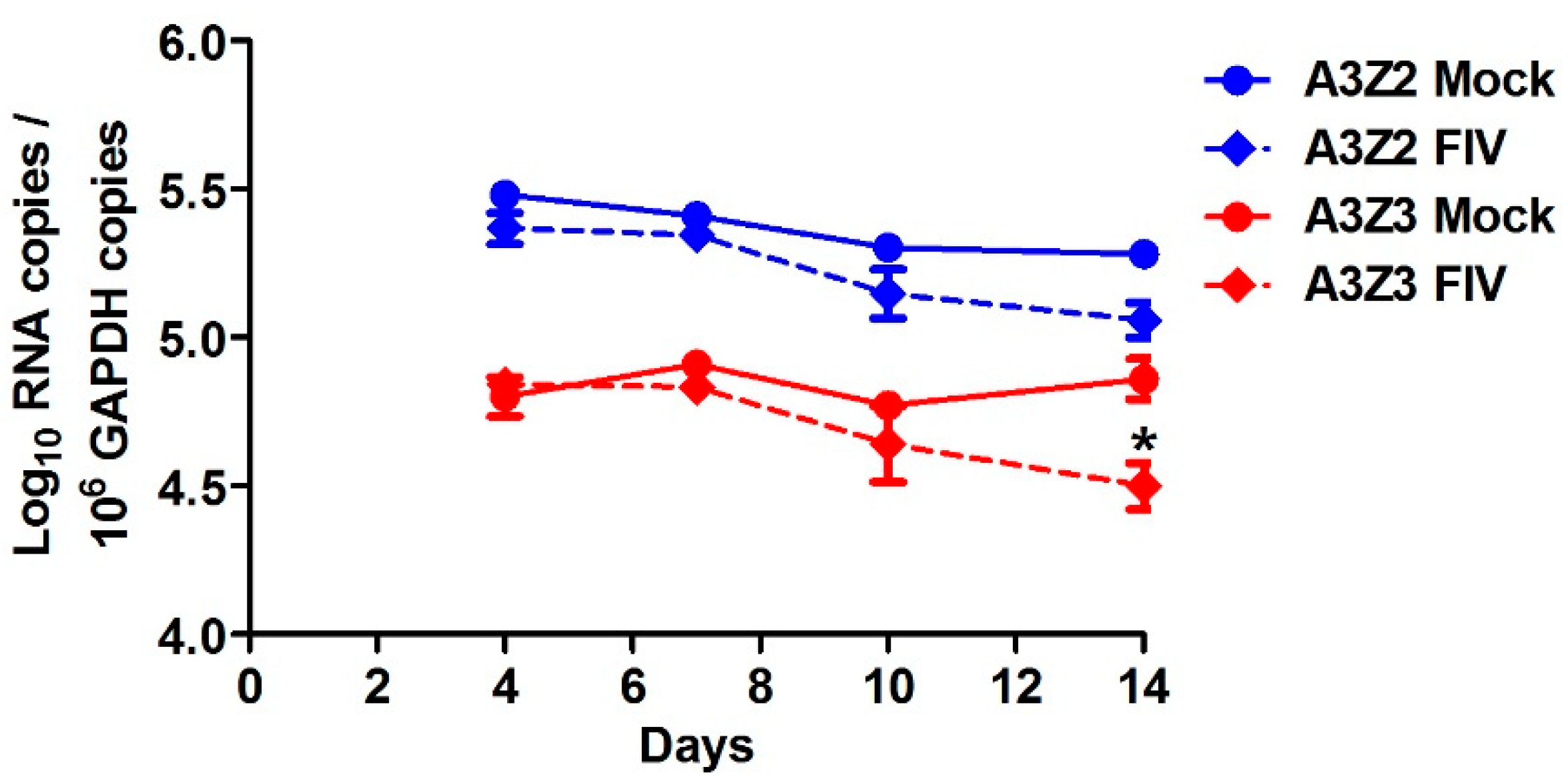
3.3. APOBEC3 Gene Expression in Cat PBMCs Is Not Altered by in Vitro FIV Infection
3.4. Lentivirus Infection Does Not Alter in Vivo APOBEC3 Gene Expression in Cat Blood Cells
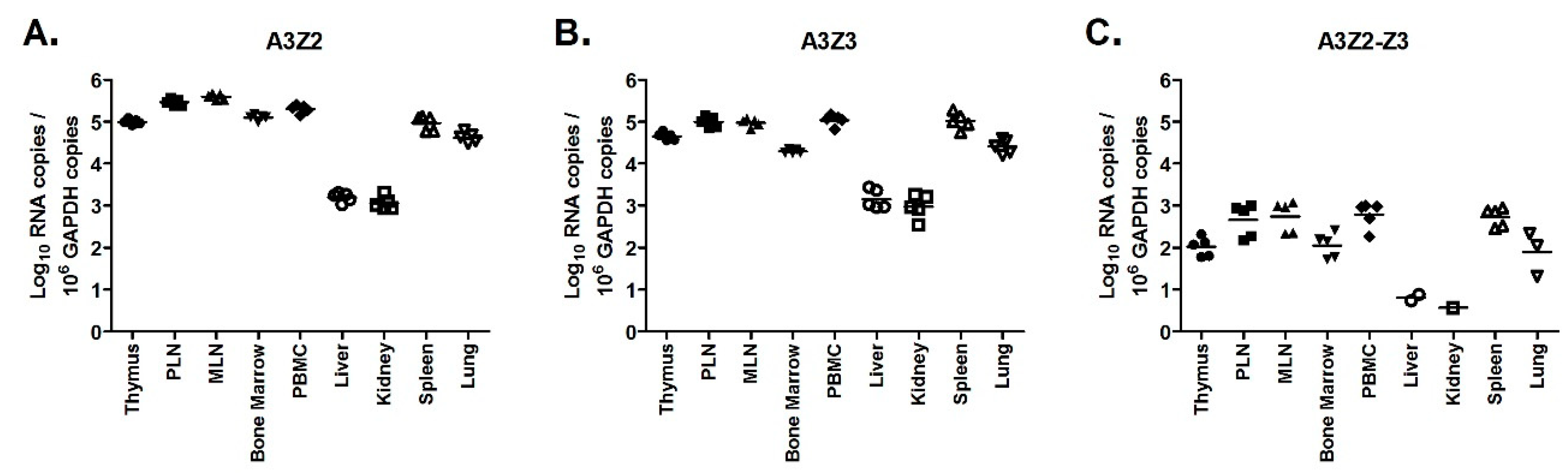
3.5. APOBEC3 Gene Expression Is Higher in Lymphoid Tissues Than Non-Lymphoid Tissues
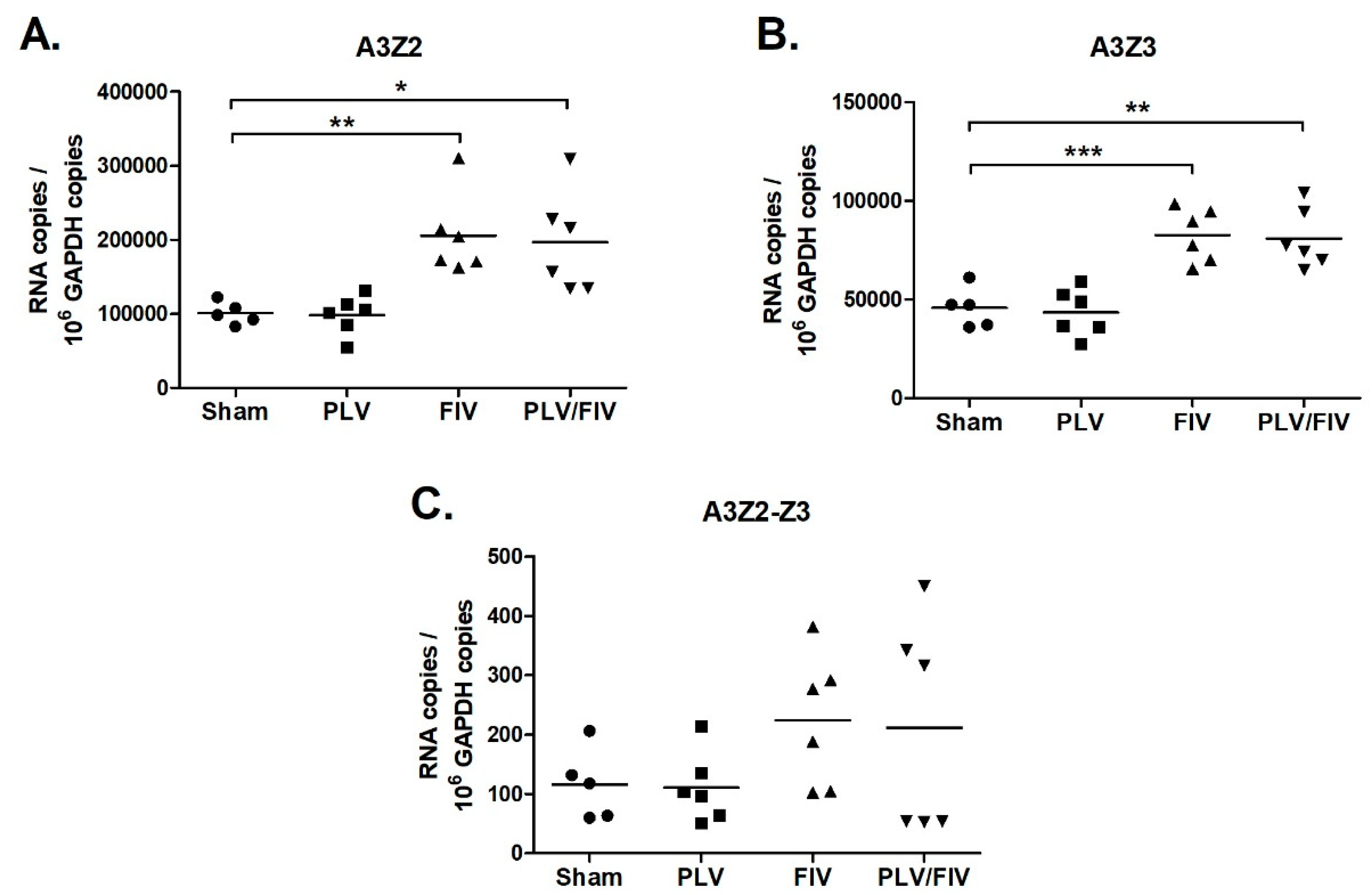
3.6. FIV Infection Upregulates APOBEC3 Gene Expression in Thymus, but Not Other Lymphoid Tissues
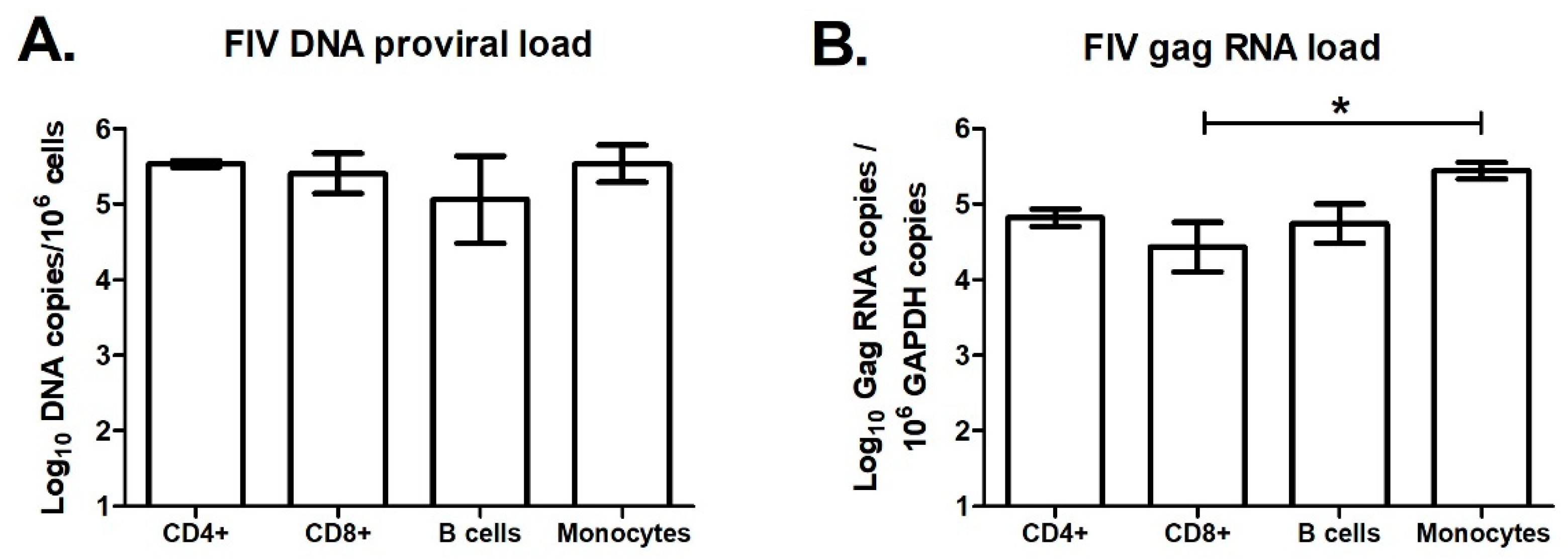
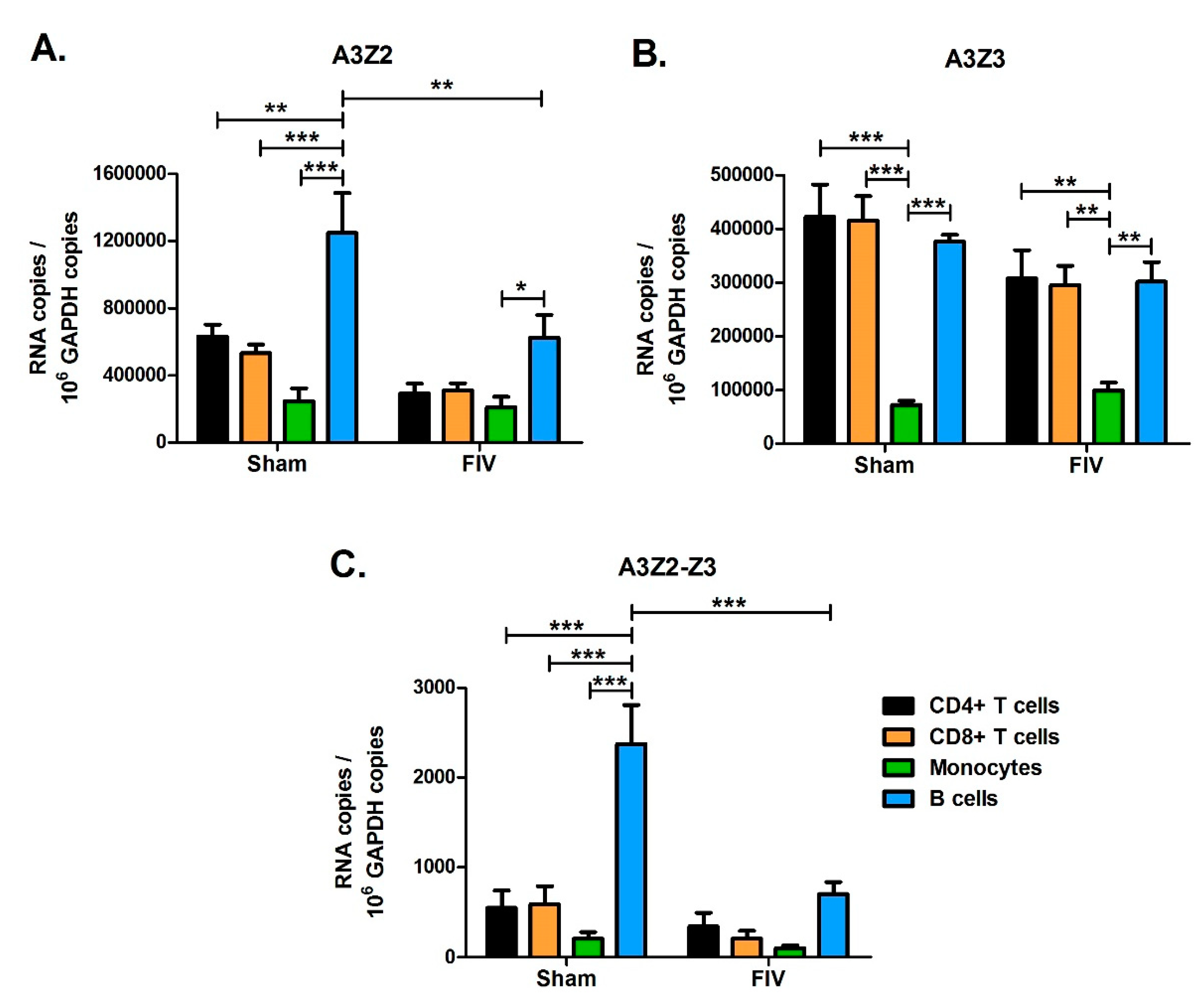
3.7. Effect of FIV Infection on APOBEC3 Expression in Major Blood Cell Sub-Populations
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Harris, R.S.; Bishop, K.N.; Sheehy, A.M.; Craig, H.M.; Petersen-Mahrt, S.K.; Watt, I.N.; Neuberger, M.S.; Malim, M.H. DNA Deamination Mediates Innate Immunity to Retroviral Infection. Cell 2003, 113, 803–809. [Google Scholar] [CrossRef]
- Harris, R.S.; Liddament, M.T. Retroviral restriction by APOBEC proteins. Nat. Rev. Immunol. 2004, 4. [Google Scholar] [CrossRef] [PubMed]
- Mangeat, B.; Turelli, P.; Caron, G.; Friedli, M.; Perrin, L.; Trono, D. Broad antiretroviral defence by human APOBEC3G through lethal editing of nascent reverse transcripts. Nature 2003, 424, 99. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Yang, B.; Pomerantz, R.J.; Zhang, C.; Arunachalam, S.C.; Gao, L. The cytidine deaminase CEM15 induces hypermutation in newly synthesized HIV-1 DNA. Nature 2003, 424, 94. [Google Scholar] [CrossRef] [PubMed]
- Sheehy, A.M.; Gaddis, N.C.; Choi, J.D.; Malim, M.H. Isolation of a human gene that inhibits HIV-1 infection and is suppressed by the viral Vif protein. Nature 2002, 418, 646. [Google Scholar] [CrossRef] [PubMed]
- Bishop, K.N.; Holmes, R.K.; Sheehy, A.M.; Davidson, N.O.; Cho, S.J.; Malim, M.H. Cytidine deamination of retroviral DNA by diverse APOBEC proteins. Curr. Biol. 2004, 14, 1392–1396. [Google Scholar] [CrossRef] [PubMed]
- Holmes, R.K.; Malim, M.H.; Bishop, K.N. APOBEC-mediated viral restriction: Not simply editing? Trends Biochem. Sci. 2007, 32, 118–128. [Google Scholar] [CrossRef] [PubMed]
- Bishop, K.N.; Verma, M.; Kim, E.Y.; Wolinsky, S.M.; Malim, M.H. APOBEC3G inhibits elongation of HIV-1 reverse transcripts. PLoS Pathog. 2008, 4, e1000231. [Google Scholar] [CrossRef]
- Holmes, R.K.; Koning, F.A.; Bishop, K.N.; Malim, M.H. APOBEC3F Can Inhibit the Accumulation of HIV-1 Reverse Transcription Products in the Absence of Hypermutation comparisons with apobec3g. J. Biol. Chem. 2007, 282, 2587–2595. [Google Scholar] [CrossRef] [PubMed]
- Bieniasz, P.D. A multimodal antiretroviral protein. Nat. Microbiol. 2018, 3, 122–123. [Google Scholar] [CrossRef]
- Marin, M.; Rose, K.M.; Kozak, S.L.; Kabat, D. HIV-1 Vif protein binds the editing enzyme APOBEC3G and induces its degradation. Nat. Med. 2003, 9, 1398. [Google Scholar] [CrossRef] [PubMed]
- Mariani, R.; Chen, D.; Schröfelbauer, B.; Navarro, F.; König, R.; Bollman, B. Species-specific exclusion of APOBEC3G from HIV-1 virions by Vif. Cell 2003, 114. [Google Scholar] [CrossRef]
- Sheehy, A.M.; Gaddis, N.C.; Malim, M.H. The antiretroviral enzyme APOBEC3G is degraded by the proteasome in response to HIV-1 Vif. Nat. Med. 2003, 9, 1404–1407. [Google Scholar] [CrossRef] [PubMed]
- Lockridge, K.M.; Himathongkham, S.; Sawai, E.T.; Chienand, M.; Sparger, E.E. The feline immunodeficiency virus vif gene is required for productive infection of feline peripheral blood mononuclear cells and monocyte-derived macrophages. Virology 1999, 261, 25–30. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.; Wang, J.; Nathans, R.S.; Cao, H.; Sharova, N.; Stevenson, M.; Rana, T.M. Synthesis and structure-activity relationship studies of HIV-1 virion infectivity factor (Vif) inhibitors that block viral replication. ChemMedChem. 2012, 7, 1217–1229. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Gu, Q.; de Manuel Montero, M.; Bravo, I.G.; Marques-Bonet, T.; Haussinger, D.; Munk, C. Stably expressed APOBEC3H forms a barrier for cross-species transmission of simian immunodeficiency virus of chimpanzee to humans. PLoS Pathog. 2017, 13, e1006746. [Google Scholar] [CrossRef] [PubMed]
- Bogerd, H.P.; Doehle, B.P.; Wiegand, H.L.; Cullen, B.R. A single amino acid difference in the host APOBEC3G protein controls the primate species specificity of HIV type 1 virion infectivity factor. Proc. Natl. Acad. Sci. USA 2004, 101, 3770–3774. [Google Scholar] [CrossRef] [PubMed]
- Koning, F.A.; Newman, E.N.; Kim, E.Y.; Kunstman, K.J.; Wolinsky, S.M.; Malim, M.H. Defining APOBEC3 expression patterns in human tissues and hematopoietic cell subsets. J. Virol. 2009, 83, 9474–9485. [Google Scholar] [CrossRef] [PubMed]
- Rose, K.M.; Marin, M.; Kozak, S.L.; Kabat, D. Transcriptional regulation of APOBEC3G, a cytidine deaminase that hypermutates human immunodeficiency virus. J. Biol. Chem. 2004, 279, 41744–41749. [Google Scholar] [CrossRef] [PubMed]
- Vetter, M.L.; Johnson, M.E.; Antons, A.K.; Unutmaz, D.; Richard, T. Differences in APOBEC3G expression in CD4+ T helper lymphocyte subtypes modulate HIV-1 infectivity. PLoS Pathog. 2009, 5, e1000292. [Google Scholar] [CrossRef] [PubMed]
- Stopak, K.S.; Chiu, Y.-L.; Kropp, J.; Grant, R.M.; Greene, W.C. Distinct patterns of cytokine regulation of APOBEC3G expression and activity in primary lymphocytes, macrophages, and dendritic cells. J. Biol. Chem. 2007, 282, 3539–3546. [Google Scholar] [CrossRef] [PubMed]
- Refsland, E.W.; Stenglein, M.D.; Shindo, K.; Albin, J.S.; Brown, W.L.; Harris, R.S. Quantitative profiling of the full APOBEC3 mRNA repertoire in lymphocytes and tissues: Implications for HIV-1 restriction. Nucleic Acids Res. 2010, 38, 4274–4284. [Google Scholar] [CrossRef] [PubMed]
- Roberts, S.A.; Lawrence, M.S.; Klimczak, L.J.; Grimm, S.A.; Fargo, D.; Stojanov, P.; Kiezun, A.; Kryukov, G.V.; Carter, S.L.; Saksena, G. An APOBEC cytidine deaminase mutagenesis pattern is widespread in human cancers. Nat. Genet. 2013, 45, 970. [Google Scholar] [CrossRef] [PubMed]
- Malim, M.H. APOBEC proteins and intrinsic resistance to HIV-1 infection. Philos. Trans. R. Soc. Lond B Biol. Sci. 2009, 364, 675–687. [Google Scholar] [CrossRef] [PubMed]
- Goila-Gaur, R.; Strebel, K. HIV-1 Vif, APOBEC, and intrinsic immunity. Retrovirology 2008, 5, 51. [Google Scholar] [CrossRef] [PubMed]
- Peng, G.; Lei, K.J.; Jin, W.; Greenwell-Wild, T.; Wahl, S.M. Induction of APOBEC3 family proteins, a defensive maneuver underlying interferon-induced anti-HIV-1 activity. J. Exp. Med. 2006, 203, 41–46. [Google Scholar] [CrossRef]
- Chen, K.; Huang, J.; Zhang, C.; Huang, S.; Nunnari, G.; Wang, F.X.; Tong, X.; Gao, L.; Nikisher, K.; Zhang, H. Alpha interferon potently enhances the anti-human immunodeficiency virus type 1 activity of APOBEC3G in resting primary CD4 T cells. J. Virol. 2006, 80, 7645–7657. [Google Scholar] [CrossRef] [PubMed]
- Pillai, S.K.; Abdel-Mohsen, M.; Guatelli, J.; Skasko, M.; Monto, A.; Fujimoto, K.; Yukl, S.; Greene, W.C.; Kovari, H.; Rauch, A.; et al. Role of retroviral restriction factors in the interferon-alpha-mediated suppression of HIV-1 in vivo. Proc. Natl. Acad. Sci. USA 2012, 109, 3035–3040. [Google Scholar] [CrossRef]
- Trapp, S.; Derby, N.R.; Singer, R.; Shaw, A.; Williams, V.G.; Turville, S.G.; Bess, J.W.; Lifson, J.D.; Robbiani, M. Double-stranded RNA analog poly (I: C) inhibits human immunodeficiency virus amplification in dendritic cells via type I interferon-mediated activation of APOBEC3G. J. Irology 2009, 83, 884–895. [Google Scholar] [CrossRef]
- Sabbatucci, M.; Covino, D.A.; Purificato, C.; Mallano, A.; Federico, M.; Lu, J.; Rinaldi, A.O.; Pellegrini, M.; Bona, R.; Michelini, Z. Endogenous CCL2 neutralization restricts HIV-1 replication in primary human macrophages by inhibiting viral DNA accumulation. Retrovirology 2015, 12, 4. [Google Scholar] [CrossRef]
- Münk, C.; Willemsen, A.; Bravo, I.G. An ancient history of gene duplications, fusions and losses in the evolution of APOBEC3 mutators in mammals. BMC Evol. Biol. 2012, 12, 71. [Google Scholar] [CrossRef] [PubMed]
- LaRue, R.S.; Jonsson, S.R.; Silverstein, K.A.; Lajoie, M.; Bertrand, D.; El-Mabrouk, N. The artiodactyl APOBEC3 innate immune repertoire shows evidence for a multi-functional domain organization that existed in the ancestor of placental mammals. BMC Mol. Biol. 2008, 9. [Google Scholar] [CrossRef] [PubMed]
- MacGinnitie, A.J.; Anant, S.; Davidson, N.O. Mutagenesis of apobec-1, the catalytic subunit of the mammalian apolipoprotein B mRNA editing enzyme, reveals distinct domains that mediate cytosine nucleoside deaminase, RNA binding, and RNA editing activity. J. Biol. Chem. 1995, 270, 14768–14775. [Google Scholar] [CrossRef] [PubMed]
- LaRue, R.S.; Andrésdóttir, V.; Blanchard, Y.; Conticello, S.G.; Derse, D.; Emerman, M.; Greene, W.C.; Jónsson, S.R.; Landau, N.R.; Löchelt, M. Guidelines for naming nonprimate APOBEC3 genes and proteins. J. Virol. 2009, 83, 494–497. [Google Scholar] [CrossRef] [PubMed]
- Münk, C.; Beck, T.; Zielonka, J.; Hotz-Wagenblatt, A.; Chareza, S.; Battenberg, M. Functions, structure, and read-through alternative splicing of feline APOBEC3 genes. Genome Biol. 2008, 9, R48. [Google Scholar] [CrossRef] [PubMed]
- Chareza, S.; Slavkovic Lukic, D.; Liu, Y.; Rathe, A.M.; Munk, C.; Zabogli, E.; Pistello, M.; Lochelt, M. Molecular and functional interactions of cat APOBEC3 and feline foamy and immunodeficiency virus proteins: Different ways to counteract host-encoded restriction. Virology 2012, 424, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Löchelt, M.; Romen, F.; Bastone, P.; Muckenfuss, H.; Kirchner, N.; Kim, Y.-B.; Truyen, U.; Rösler, U.; Battenberg, M.; Saib, A. The antiretroviral activity of APOBEC3 is inhibited by the foamy virus accessory Bet protein. Proc. Natl. Acad. Sci. USA 2005, 102, 7982–7987. [Google Scholar] [CrossRef] [PubMed]
- Russell, R.A.; Wiegand, H.L.; Moore, M.D.; Schäfer, A.; McClure, M.O.; Cullen, B.R. Foamy virus Bet proteins function as novel inhibitors of the APOBEC3 family of innate antiretroviral defense factors. J. Virol. 2005, 79, 8724–8731. [Google Scholar] [CrossRef]
- Zielonka, J.; Marino, D.; Hofmann, H.; Yuhki, N.; Löchelt, M.; Münk, C. Vif of feline immunodeficiency virus from domestic cats protects against APOBEC3 restriction factors from many felids. J. Virol. 2010, 84. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, W.; Lv, M.; Zuo, T.; Kong, W.; Yu, X. Identification of a Cullin5-ElonginB-ElonginC E3 complex in degradation of feline immunodeficiency virus Vif-mediated feline APOBEC3 proteins. J. Virol. 2011, 85, 12482–12491. [Google Scholar] [CrossRef]
- Zhang, Z.; Gu, Q.; Jaguva Vasudevan, A.A.; Hain, A.; Kloke, B.P.; Hasheminasab, S.; Mulnaes, D.; Sato, K.; Cichutek, K.; Haussinger, D.; et al. Determinants of FIV and HIV Vif sensitivity of feline APOBEC3 restriction factors. Retrovirology 2016, 13, 46. [Google Scholar] [CrossRef] [PubMed]
- Munk, C.; Hechler, T.; Chareza, S.; Lochelt, M. Restriction of feline retroviruses: Lessons from cat APOBEC3 cytidine deaminases and TRIM5alpha proteins. Vet. Immunol. Immunopathol. 2010, 134, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Krupp, A.; McCarthy, K.R.; Ooms, M.; Letko, M.; Morgan, J.S.; Simon, V. APOBEC3G polymorphism as a selective barrier to cross-species transmission and emergence of pathogenic SIV and AIDS in a primate host. PLoS Pathog. 2013, 9. [Google Scholar] [CrossRef] [PubMed]
- Mangeat, B.; Turelli, P.; Liao, S.; Trono, D. A single amino acid determinant governs the species-specific sensitivity of APOBEC3G to Vif action. J. Biol. Chem. 2004, 279, 14481–14483. [Google Scholar] [CrossRef] [PubMed]
- Compton, A.A.; Hirsch, V.M.; Emerman, M. The host restriction factor APOBEC3G and retroviral Vif protein coevolve due to ongoing genetic conflict. Cell Host Microbe 2012, 11, 91–98. [Google Scholar] [CrossRef] [PubMed]
- VandeWoude, S.; Troyer, J.; Poss, M. Restrictions to cross-species transmission of lentiviral infection gleaned from studies of FIV. Vet. Immunol. Immunopathol. 2010, 134. [Google Scholar] [CrossRef] [PubMed]
- LaRue, R.S.; Lengyel, J.; Jonsson, S.R.; Andresdottir, V.; Harris, R.S. Lentiviral Vif degrades the APOBEC3Z3/APOBEC3H protein of its mammalian host and is capable of cross-species activity. J. Virol. 2010, 84. [Google Scholar] [CrossRef]
- Zhang, Z.; Gu, Q.; Marino, D.; Lee, K.-L.; Kong, I.-K.; Häussinger, D.; Münk, C. Feline APOBEC3s, Barriers to Cross-Species Transmission of FIV? Viruses 2018, 10, 186. [Google Scholar] [CrossRef]
- Munk, C.; Zielonka, J.; Constabel, H.; Kloke, B.P.; Rengstl, B.; Battenberg, M.; Bonci, F.; Pistello, M.; Lochelt, M.; Cichutek, K. Multiple restrictions of human immunodeficiency virus type 1 in feline cells. J. Virol. 2007, 81, 7048–7060. [Google Scholar] [CrossRef]
- Simon, J.; Southerling, T.E.; Peterson, J.C.; Meyer, B.E.; Malim, M.H. Complementation of vif-defective human immunodeficiency virus type 1 by primate, but not nonprimate, lentivirus vif genes. J. Virol. 1995, 69, 4166–4172. [Google Scholar]
- Lee, J.; Malmberg, J.L.; Wood, B.A.; Hladky, S.; Troyer, R.; Roelke, M.; Cunningham, M.; McBride, R.; Vickers, W.; Boyce, W. Feline immunodeficiency virus cross-species transmission: Implications for emergence of new lentiviral infections. J. Virol. 2017, 91, e02134-16. [Google Scholar] [CrossRef] [PubMed]
- Franklin, S.P.; Troyer, J.L.; TerWee, J.A.; Lyren, L.M.; Kays, R.W.; Riley, S.P.; Boyce, W.M.; Crooks, K.R.; Vandewoude, S. Variability in assays used for detection of lentiviral infection in bobcats (Lynx rufus), pumas (Puma concolor), and ocelots (Leopardus pardalis). J. Wildlife Dis. 2007, 43, 700–710. [Google Scholar] [CrossRef] [PubMed]
- Poss, M.; Ross, H.A.; Painter, S.L.; Holley, D.C.; Terwee, J.A.; Vandewoude, S.; Rodrigo, A. Feline lentivirus evolution in cross-species infection reveals extensive G-to-A mutation and selection on key residues in the viral polymerase. J. Virol. 2006, 80, 2728–2737. [Google Scholar] [CrossRef] [PubMed]
- TerWee, J.A.; Carlson, J.K.; Sprague, W.S.; Sondgeroth, K.S.; Shropshire, S.B.; Troyer, J.L.; VandeWoude, S. Prevention of immunodeficiency virus induced CD4+ T-cell depletion by prior infection with a non-pathogenic virus. Virology 2008, 377, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Padhi, A.; Ross, H.; Terwee, J.; VandeWoude, S.; Poss, M. Profound differences in virus population genetics correspond to protection from CD4 decline resulting from feline lentivirus coinfection. Viruses 2010, 2, 2663–2680. [Google Scholar] [CrossRef]
- Zheng, X.; Carver, S.; Troyer, R.M.; Terwee, J.A.; VandeWoude, S. Prior virus exposure alters the long-term landscape of viral replication during feline lentiviral infection. Viruses 2011, 3, 1891–1908. [Google Scholar] [CrossRef]
- Sprague, W.S.; Troyer, R.M.; Zheng, X.; Wood, B.A.; Macmillian, M.; Carver, S.; VandeWoude, S. Prior Puma Lentivirus Infection Modifies Early Immune Responses and Attenuates Feline Immunodeficiency Virus Infection in Cats. Viruses 2018, 10, 210. [Google Scholar] [CrossRef]
- Crandell, R.A.; Fabricant, C.G.; Nelson-Rees, W.A. Development, characterization, and viral susceptibility of a feline (Felis catus) renal cell line (CRFK). In Vitro 1973, 9, 176–185. [Google Scholar] [CrossRef]
- Miyazawa, T.; Furuya, T.; Itagaki, S.-I.; Tohya, Y.; Takahashi, E.; Mikami, T. Establishment of a feline T-lymphoblastoid cell line highly sensitive for replication of feline immunodeficiency virus. Arch. Virol. 1989, 108, 131–135. [Google Scholar] [CrossRef]
- Snyder, H.W., Jr.; Hardy, W.D., Jr.; Zuckerman, E.E.; Fleissner, E. Characterisation of a tumour-specific antigen on the surface of feline lymphosarcoma cells. Nature 1978, 275, 656. [Google Scholar] [CrossRef]
- De Rozières, S.; Mathiason, C.K.; Rolston, M.R.; Chatterji, U.; Hoover, E.A.; Elder, J.H. Characterization of a highly pathogenic molecular clone of feline immunodeficiency virus clade C. J. Virol. 2004, 78, 8971–8982. [Google Scholar] [CrossRef] [PubMed]
- Vandewoude, S.; O’brien, S.J.; Langelier, K.; Hardy, W.D.; Slattery, J.P.; Zuckerman, E.E.; Hoover, E.A. Growth of lion and puma lentiviruses in domestic cat cells and comparisons with FIV. Virology 1997, 233, 185–192. [Google Scholar] [CrossRef] [PubMed]
- TerWee, J.A.; Yactor, J.K.; Sondgeroth, K.S.; VandeWoude, S. Puma lentivirus is controlled in domestic cats after mucosal exposure in the absence of conventional indicators of immunity. J. Virol. 2005, 79, 2797–2806. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Troyer, R.M.; Thompson, J.; Elder, J.H.; VandeWoude, S. Accessory genes confer a high replication rate to virulent feline immunodeficiency virus. J. Virol. 2013, 87. [Google Scholar] [CrossRef]
- Leutenegger, C.M.; Mislin, C.N.; Sigrist, B.; Ehrengruber, M.U.; Hofmann-Lehmann, R.; Lutz, H. Quantitative real-time PCR for the measurement of feline cytokine mRNA. Vet. Immunol. Immunopathol. 1999, 71, 291–305. [Google Scholar] [CrossRef]
- Yoshikawa, R.; Nakano, Y.; Yamada, E.; Izumi, T.; Misawa, N.; Koyanagi, Y. Species-specific differences in the ability of feline lentiviral Vif to degrade feline APOBEC3 proteins. Microbiol. Immunol. 2016, 60. [Google Scholar] [CrossRef]
- Konno, Y.; Nagaoka, S.; Kimura, I.; Yamamoto, K.; Kagawa, Y.; Kumata, R.; Aso, H.; Ueda, M.T.; Nakagawa, S.; Kobayashi, T. New World feline APOBEC3 potently controls inter-genus lentiviral transmission. Retrovirology 2018, 15, 31. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Troyer, R.M.; Malmberg, J.L.; Zheng, X.; Miller, C.; MacMillan, M.; Sprague, W.S.; Wood, B.A.; VandeWoude, S. Expression of APOBEC3 Lentiviral Restriction Factors in Cats. Viruses 2019, 11, 831. https://doi.org/10.3390/v11090831
Troyer RM, Malmberg JL, Zheng X, Miller C, MacMillan M, Sprague WS, Wood BA, VandeWoude S. Expression of APOBEC3 Lentiviral Restriction Factors in Cats. Viruses. 2019; 11(9):831. https://doi.org/10.3390/v11090831
Chicago/Turabian StyleTroyer, Ryan M., Jennifer L. Malmberg, Xin Zheng, Craig Miller, Martha MacMillan, Wendy S. Sprague, Britta A. Wood, and Sue VandeWoude. 2019. "Expression of APOBEC3 Lentiviral Restriction Factors in Cats" Viruses 11, no. 9: 831. https://doi.org/10.3390/v11090831
APA StyleTroyer, R. M., Malmberg, J. L., Zheng, X., Miller, C., MacMillan, M., Sprague, W. S., Wood, B. A., & VandeWoude, S. (2019). Expression of APOBEC3 Lentiviral Restriction Factors in Cats. Viruses, 11(9), 831. https://doi.org/10.3390/v11090831






