Exposure to Hantavirus is a Risk Factor Associated with Kidney Diseases in Sri Lanka: A Cross Sectional Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Selection of Diagnostic Method for CKDu Patient Sera
2.1.1. Virus and Cells
2.1.2. Indirect Immunofluorescent Antibody Assay
2.1.3. Preparation of an rNP-Based IFA Antigen
2.1.4. Mouse Monoclonal Antibodies and Its IFA Profiles
2.1.5. ELISA by Using Recombinant NPs
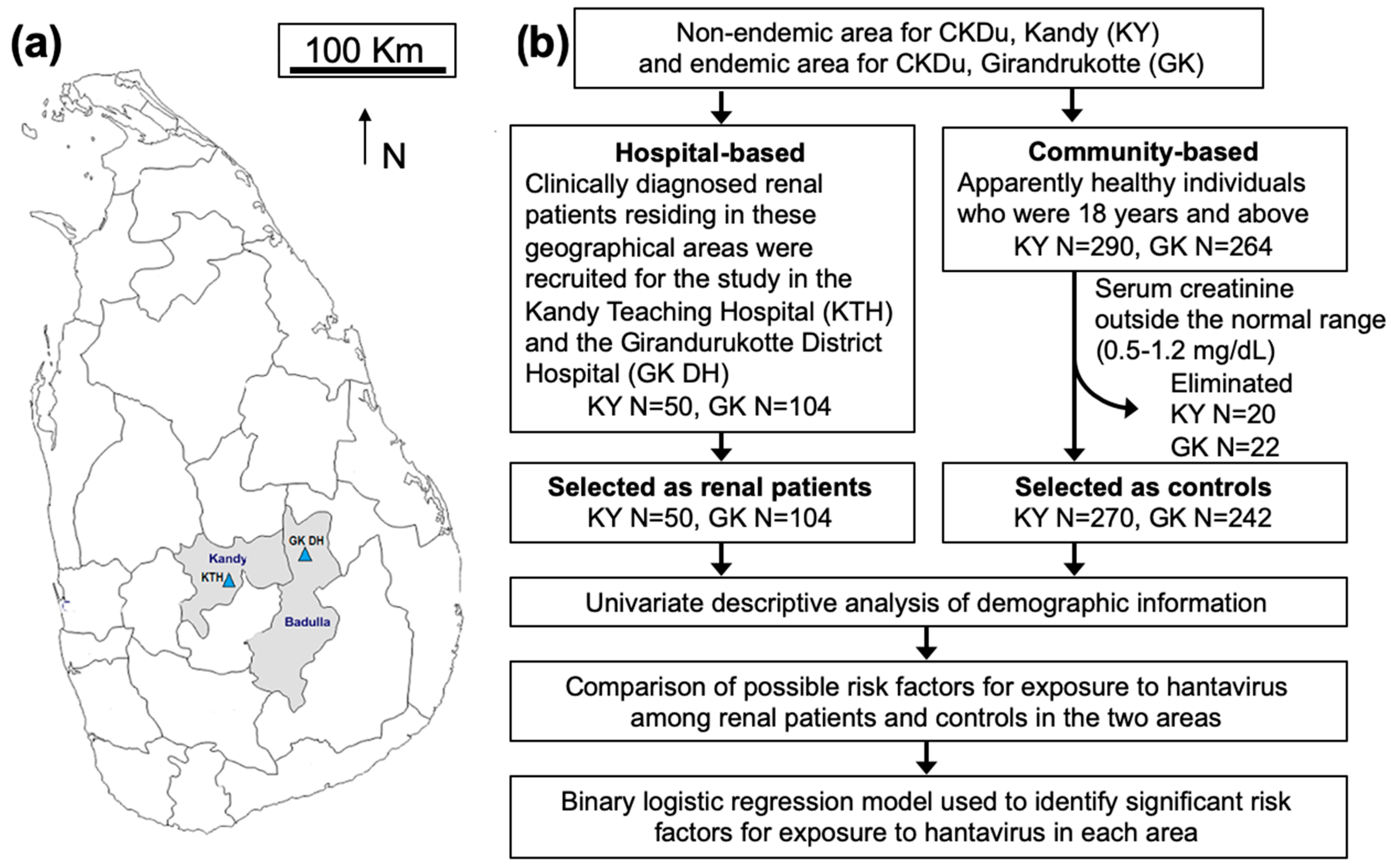
2.2. Study Design, Study Population and Sample Size Calculation for the Cross-Sectional Study
2.2.1. Hospital-Based Study
2.2.2. Community-Based Study
2.3. Ethical Approval
2.4. Collection of Data and Blood Samples
2.5. Statistical Analysis
2.5.1. Univariate Descriptive Analysis
2.5.2. Comparison of Possible Risk Factors for Exposure to Hantavirus among Seropositive Individuals in the Two Areas
2.5.3. Binary Logistic Regression Model
3. Results
3.1. Characteristics of Anti-Hantavirus Antibodies in Sera Obtained in Sri Lanka
3.1.1. Reactivities in IFA
3.1.2. Reactivities in ELISA
3.2. Demographic Characteristics and Serological Results of the Study Subjects
3.3. Comparison of Possible Risk Factors for Exposure to Hantavirus
3.4. Binary Logistic Regression Models
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, H.; Naghave, M.; Allen, C.; Barber, R.M.; Bhutta, Z.A.; Carter, A.; Casey, D.C.; Charlson, F.J.; Chen, A.Z.; Coates, M.M.; et al. Global, regional, and national life expectancy, all-cause mortality, and cause-specific mortality for 249 causes of death, 1980–2015: A systematic analysis for the global burden of disease study 2015. Lancet 2016, 388, 1459–1544. [Google Scholar] [CrossRef]
- Athuraliya, N.T.; Abeysekera, T.D.; Amerasinghe, P.H.; Kumarasiri, R.; Bandara, P.; Karunaratne, U.; Milton, A.H.; Jones, A.L. Uncertain etiologies of proteinuric-chronic kidney disease in rural Sri Lanka. Kidney Int. 2011, 80, 1212–1221. [Google Scholar] [CrossRef] [PubMed]
- Weaver, V.M.; Fadrowski, J.J.; Jaar, B.G. Global dimensions of chronic kidney disease of unknown etiology (CKDU): A modern era environmental and/or occupational nephropathy? BMC Nephrol. 2015, 16, 145. [Google Scholar] [CrossRef] [PubMed]
- Rajapakse, S.; Shivanthan, M.C.; Selvarajah, M. Chronic kidney disease of unknown etiology in Sri Lanka. Int. J. Occup. Environ. Health 2016, 22, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Plyusnin, A.; Vapalahti, O.; Lundkvist, A. Hantaviruses: Genome structure, expression and evolution. J. Gen. Virol. 1996, 77, 2677–2687. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, R.C.; Guterres, A.; Fernandes, J.; D’Andrea, P.S.; Bonvicino, C.R.; de Lemos, E.R. Hantavirus reservoirs: Current status with an emphasis on data from Brazil. Viruses 2014, 6, 1929–1973. [Google Scholar] [CrossRef]
- Gamage, C.D.; Yoshimatsu, K.; Sarathkumara, Y.D.; Thiviyaaluxmi, K.; Nanayakkara, N.; Arikawa, J. Serological evidence of hantavirus infection in girandurukotte, an area endemic for chronic kidney disease of unknown aetiology (CKDU) in Sri Lanka. Int. J. Infect. Dis. 2017, 57, 77–78. [Google Scholar] [CrossRef] [PubMed]
- Yoshimatsu, K.; Gamage, C.D.; Sarathkumara, Y.D.; Kulendiran, T.; Muthusinghe, D.S.; Nanayakkara, N.; Gunarathne, L.; Shimizu, K.; Tsuda, Y.; Arikawa, J. Thailand orthohantavirus infection in patients with chronic kidney disease of unknown aetiology in Sri Lanka. Arch. Virol. 2019, 164, 267–271. [Google Scholar] [CrossRef]
- Elwell, M.R.; Ward, G.S.; Tingpalapong, M.; LeDuc, J.W. Serologic evidence of hantaan-like virus in rodents and man in Thailand. Southeast Asian J. Trop. Med. Public Health 1985, 16, 349–354. [Google Scholar]
- Pattamadilok, S.; Lee, B.H.; Kumperasart, S.; Yoshimatsu, K.; Okumura, M.; Nakamura, I.; Araki, K.; Khoprasert, Y.; Dangsupa, P.; Panlar, P.; et al. Geographical distribution of hantaviruses in Thailand and potential human health significance of Thailand virus. Am. J. Trop. Med. Hyg. 2006, 75, 994–1002. [Google Scholar] [CrossRef]
- Suputthamongkol, Y.; Nitatpattana, N.; Chayakulkeeree, M.; Palabodeewat, S.; Yoksan, S.; Gonzalez, J.P. Hantavirus infection in thailand: First clinical case report. Southeast Asian J. Trop. Med. Public Health 2005, 36, 217–220. [Google Scholar] [PubMed]
- Gamage, C.D.; Yasuda, S.P.; Nishio, S.; Kularatne, S.A.; Weerakoon, K.; Rajapakse, J.; Nwafor-Okoli, C.; Lee, R.B.; Obayashi, Y.; Yoshimatsu, K.; et al. Serological evidence of Thailand virus-related hantavirus infection among suspected leptospirosis patients in Kandy, Sri Lanka. Jpn. J. Infect. Dis. 2011, 64, 72–75. [Google Scholar] [PubMed]
- Araki, K.; Yoshimatsu, K.; Ogino, M.; Ebihara, H.; Lundkvist, A.; Kariwa, H.; Takashima, I.; Arikawa, J. Truncated hantavirus nucleocapsid proteins for serotyping hantaan, seoul, and dobrava hantavirus infections. J. Clin. Microbiol. 2001, 39, 2397–2404. [Google Scholar] [CrossRef] [PubMed]
- Yoshimatsu, K.; Arikawa, J.; Kariwa, H. Application of a recombinant baculovirus expressing hantavirus nucleocapsid protein as a diagnostic antigen in ifa test: Cross reactivities among 3 serotypes of hantavirus which causes hemorrhagic fever with renal syndrome (HFRS). J. Vet. Med. Sci. 1993, 55, 1047–1050. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lokupathirage, S.M.W.; Muthusinghe, D.S.; Shimizu, K.; Nishigami, K.; Noda, K.; Tsuda, Y.; Sarathkumara, Y.D.; Gunewardana, S.; Arikawa, J.; Gamage, C.D.; et al. Serological evidence of thailand orthohantavirus or antigenically related virus infection among rodents in a chronic kidney disease of unknown etiology (CKDU) endemic area, girandurukotte, Sri Lanka. Vector-borne zoonotic dis. Vector Borne Zoonotic Dis. 2019, in press. [Google Scholar] [CrossRef] [PubMed]
- Yoshimatsu, K.; Arikawa, J.; Tamura, M.; Yoshida, R.; Lundkvist, A.; Niklasson, B.; Kariwa, H.; Azuma, I. Characterization of the nucleocapsid protein of hantaan virus strain 76-118 using monoclonal antibodies. J. Gen. Virol. 1996, 77, 695–704. [Google Scholar] [CrossRef] [PubMed]
- Arikawa, J.; Schmaljohn, A.L.; Dalrymple, J.M.; Schmaljohn, C.S. Characterization of hantaan virus envelope glycoprotein antigenic determinants defined by monoclonal antibodies. J. Gen. Virol. 1989, 70, 615–624. [Google Scholar] [CrossRef] [PubMed]
- Koma, T.; Yoshimatsu, K.; Pini, N.; Safronetz, D.; Taruishi, M.; Levis, S.; Endo, R.; Shimizu, K.; Yasuda, S.P.; Ebihara, H.; et al. Truncated hantavirus nucleocapsid proteins for serotyping sin nombre, andes, and laguna negra hantavirus infections in humans and rodents. J. Clin. Microbiol. 2010, 48, 1635–1642. [Google Scholar] [CrossRef]
- Yoshimatsu, K.; Arikawa, J. Serological diagnosis with recombinant n antigen for hantavirus infection. Virus Res. 2014, 187, 77–83. [Google Scholar] [CrossRef]
- Takakura, A.; Goto, K.; Itoh, T.; Yoshimatsu, K.; Takashima, I.; Arikawa, J. Establishment of an enzyme-linked immunosorbent assay for detection of hantavirus antibody of rats using a recombinant of nucleocapsid protein expressed in escherichia coli. Exp. Anim. 2003, 52, 25–30. [Google Scholar] [CrossRef]
- Amada, T.; Yoshimatsu, K.; Yasuda, S.P.; Shimizu, K.; Koma, T.; Hayashimoto, N.; Gamage, C.D.; Nishio, S.; Takakura, A.; Arikawa, J. Rapid, whole blood diagnostic test for detecting anti-hantavirus antibody in rats. J. Virol. Methods 2013, 193, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Palihawadana, P. Research Programme for Chronic Kidney Disease of Unknown Aetiology in Sri Lanka; Weekly Epidemiological Report Sri Lanka: Peradeniya, Sri Lanka, 2009; Volume 36, pp. 1–4. [Google Scholar]
- World Health Organization. Case Definition of Chronic Kidney Disease of Unknown Etiology (CKDU). Available online: http://www.searo.who.int/srilanka/documents/designing_a_step-wise_approach_to_estimate_the_burden_and_to_understand_the_etiology_of_ckdu_in_srl.pdf (accessed on 27 July 2019).
- Wijkstrom, J.; Jayasumana, C.; Dassanayake, R.; Priyawardane, N.; Godakanda, N.; Siribaddana, S.; Ring, A.; Hultenby, K.; Soderberg, M.; Elinder, C.G.; et al. Morphological and clinical findings in sri lankan patients with chronic kidney disease of unknown cause (CKDU): Similarities and differences with mesoamerican nephropathy. PLoS ONE 2018, 13, e0193056. [Google Scholar] [CrossRef] [PubMed]
- Gamage, C.D.; Sarathkumara, Y.D. Chronic kidney disease of uncertain etiology in Sri Lanka: Are leptospirosis and hantaviral infection likely causes? Med. Hypotheses 2016, 91, 16–19. [Google Scholar] [CrossRef] [PubMed]
- Amada, T.; Yoshimatsu, K.; Koma, T.; Shimizu, K.; Gamage, C.D.; Shiokawa, K.; Nishio, S.; Ahlm, C.; Arikawa, J. Development of an immunochromatography strip test based on truncated nucleocapsid antigens of three representative hantaviruses. Virol. J. 2014, 11, 87. [Google Scholar] [CrossRef]
- Munoz-Zanzi, C.; Saavedra, F.; Otth, C.; Domancich, L.; Hott, M.; Padula, P. Serological evidence of hantavirus infection in apparently healthy people from rural and slum communities in southern Chile. Viruses 2015, 7, 2006–2013. [Google Scholar] [CrossRef] [PubMed]
- Limongi, J.E.; da Costa, F.C.; Pinto, R.M.; de Oliveira, R.C.; Bragagnolo, C.; Lemos, E.R.; de Paula, M.B.; Pajuaba Neto, A.A.; Ferreira, M.S. Cross-sectional survey of hantavirus infection, Brazil. Emerg. Infect. Dis. 2009, 15, 1981–1983. [Google Scholar] [CrossRef] [PubMed]
- Ayala, H.M.; Herath, S.; Kubota, K.; Kawakami, T.; Nagasawa, S.; Motoyama, A.; Weragoda, S.K.; Tushara Chaminda, G.G.; Yatigammana, S.K. Potential risk of drinking water to human health in Sri Lanka. Environ. Forensics 2017, 18, 241–250. [Google Scholar]
- Sayanthooran, S.; Gunerathne, L.; Abeysekera, T.D.J.; Magana-Arachchi, D.N. Transcriptome analysis supports viral infection and fluoride toxicity as contributors to chronic kidney disease of unknown etiology (CKDU) in Sri Lanka. Int. Urol. Nephrol. 2018, 50, 1667–1677. [Google Scholar] [CrossRef]
- Agampodi, S.B.; Amarasinghe, G.S.; Naotunna, P.; Jayasumana, C.S.; Siribaddana, S.H. Early renal damage among children living in the region of highest burden of chronic kidney disease of unknown etiology (CKDU) in Sri Lanka. BMC Nephrol. 2018, 19, 115. [Google Scholar] [CrossRef]
- Meier, M.; Kramer, J.; Jabs, W.J.; Nolte, C.; Hofmann, J.; Kruger, D.H.; Lehnert, H.; Nitschke, M. Proteinuria and the clinical course of dobrava-belgrade hantavirus infection. Nephron Extra 2018, 8, 1–10. [Google Scholar] [CrossRef]
- Martynova, E.V.; Maksudova, A.N.; Shakirova, V.G.; Abdulkhakov, S.R.; Khaertynova, I.M.; Anokhin, V.A.; Ivanova, V.V.; Abiola, I.M.; Garanina, E.E.; Tazetdinova, L.G.; et al. Urinary clusterin is upregulated in nephropathia epidemica. Dis. Mark. 2018, 2018, 8658507. [Google Scholar] [CrossRef] [PubMed]
- Nanayakkara, S.; Senevirathna, S.T.; Karunaratne, U.; Chandrajith, R.; Harada, K.H.; Hitomi, T.; Watanabe, T.; Abeysekera, T.; Aturaliya, T.N.; Koizumi, A. Evidence of tubular damage in the very early stage of chronic kidney disease of uncertain etiology in the north central province of sri lanka: A cross-sectional study. Environ. Health Prev. Med. 2012, 17, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Nanayakkara, S.; Komiya, T.; Ratnatunga, N.; Senevirathna, S.T.; Harada, K.H.; Hitomi, T.; Gobe, G.; Muso, E.; Abeysekera, T.; Koizumi, A. Tubulointerstitial damage as the major pathological lesion in endemic chronic kidney disease among farmers in north central province of Sri Lanka. Environ. Health Prev. Med. 2012, 17, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Adams, K.; Jameson, L.; Meigh, R.; Brooks, T. Hantavirus: An infectious cause of acute kidney injury in the UK. BMJ Case Rep. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Miettinen, M.H.; Makela, S.M.; Ala-Houhala, I.O.; Huhtala, H.S.; Koobi, T.; Vaheri, A.I.; Pasternack, A.I.; Porsti, I.H.; Mustonen, J.T. Tubular proteinuria and glomerular filtration 6 years after puumala hantavirus-induced acute interstitial nephritis. Nephron Clin. Prac. 2009, 112, c115–c120. [Google Scholar] [CrossRef] [PubMed]
| Area | Status | Gender | Number | Age Mean (SD) | Serum Antibody Positive to Hantavirus (%) | Occupation Paddy Farming |
|---|---|---|---|---|---|---|
| Girandurukotte | Renal patients | Male | 70 | 58 (12.3) | 42 (60.0%) | 65 |
| Female | 34 | 60 (10.2) | 10 (29.4%) | 27 | ||
| Total | 104 | 59 (11.7) | 52 (50.0%) | 92 | ||
| Community | Male | 98 | 47 (14.8) | 25 (25.5%) | 68 | |
| Female | 144 | 45 (14.0) | 17 (11.8%) | 44 | ||
| Total | 242 | 46 (14.3) | 42 (17.4%) | 112 | ||
| Kandy | Renal patients | Male | 28 | 54 (12.6) | 7 (25.0%) | 2 |
| Female | 22 | 54 (11.1) | 2 (9.1%) | 1 | ||
| Total | 50 | 54 (11.8) | 9 (18.0%) | 3 | ||
| Community | Male | 126 | 48 (14.5) | 10 (7.9%) | 10 | |
| Female | 144 | 46 (13.5) | 9 (6.3%) | 3 | ||
| Total | 270 | 47 (14.0) | 19 (7.0%) | 13 |
| Kandy | Girandurukotte | |||||||
|---|---|---|---|---|---|---|---|---|
| Category | Anti-Hantavirus Antibody | χ2 | p-Value | Anti-Hantavirus Antibody | χ2 | p-Value | ||
| Positive | Negative | Positive | Negative | |||||
| Age | ||||||||
| Age category of ≥ 40 years | ||||||||
| Renal patients | 8 | 38 | 3.85 | 0.050 | 48 | 47 | 21.89 | <0.001 |
| Control | 14 | 166 | 32 | 116 | ||||
| Age category of < 40 years | ||||||||
| Renal patients | 1 | 3 | 2.42 | 0.235 | 4 | 5 | 7.99 | 0.005 |
| Control | 5 | 85 | 10 | 84 | ||||
| Gender | ||||||||
| Female | ||||||||
| Renal patients | 2 | 20 | 0.25 | 0.642 | 10 | 24 | 6.53 | 0.011 |
| Community | 9 | 135 | 17 | 126 | ||||
| Male | ||||||||
| Renal patients | 7 | 21 | 6.79 | 0.010 | 42 | 28 | 20.3 | <0.001 |
| Community | 10 | 116 | 25 | 73 | ||||
| Occupation | ||||||||
| Paddy farming | ||||||||
| Renal patients | 3 | 0 | 11.08 | 0.007 | 47 | 45 | 18.3 | <0.001 |
| Community | 1 | 12 | 25 | 87 | ||||
| Other occupations | ||||||||
| Renal patients | 6 | 41 | 1.81 | 0.178 | 5 | 7 | 18.3 | 0.009 |
| Community | 18 | 239 | 17 | 113 | ||||
| Storing crop at house | ||||||||
| Yes | ||||||||
| Renal patients | 2 | 4 | 1.44 | 0.260 | 48 | 49 | 29.32 | <0.001 |
| Community | 4 | 26 | 28 | 131 | ||||
| No | ||||||||
| Renal patients | 7 | 37 | 4.85 | 0.028 | 4 | 3 | 6.55 | 0.027 |
| Community | 15 | 225 | 14 | 69 | ||||
| Rats seen at home or surroundings | ||||||||
| Yes | ||||||||
| Renal patients | 8 | 25 | 12.58 | <0.001 | 45 | 39 | 38.42 | <0.001 |
| Community | 12 | 194 | 34 | 163 | ||||
| No | ||||||||
| Renal patients | 1 | 16 | 0.39 | 0.535 | 7 | 13 | 2.31 | 0.128 |
| Community | 7 | 57 | 8 | 37 | ||||
| Predicted Risk Factors | Kandy | Girandurukotte | ||||
|---|---|---|---|---|---|---|
| χ2 | p-Value | OR (95% CI) | χ2 | p-Value | OR (95% CI) | |
| Status | ||||||
| Renal patient | 4.08 | 0.044 | 2.64 (1.07 to 6.54) | 18.83 | <0.001 | 3.66 (2.01 to 6.64) |
| Age | ||||||
| Age ≥ 40 years | 0.01 | 0.935 | 1.04 (0.38 to 2.86) | 3.62 | 0.057 | 1.92 (0.96 to 3.82) |
| Gender | ||||||
| Male | 0.70 | 0.402 | 1.43 (0.62 to 3.31) | 12.88 | <0.001 | 2.79 (1.58 to 4.95) |
| Occupation | ||||||
| Farmer | 1.02 | 0.313 | 2.18 (0.49 to 9.63) | 0 | 0.985 | 0.99 (0.45 to 2.18) |
| Engaged in agriculture-related activities | ||||||
| Yes | 2.76 | 0.097 | 2.53 (0.88 to 7.27) | 1.41 | 0.235 | 1.82 (0.67 to 4.91) |
| Store crops at house | ||||||
| Yes | 0.02 | 0.897 | 0.92 (0.24 to 3.47) | 1.30 | 0.255 | 0.65 (0.32 to 1.36) |
| Sighting rodents at home or surrounding | ||||||
| Yes | 0.06 | 0.446 | 0.67 (0.25 to 1.85) | 2.21 | 0.137 | 1.86 (0.81 to 4.24) |
| Presence of rodent excreta at home or surrounding | ||||||
| Yes | 0 | 0.982 | 1.01 (0.40 to 2.54) | 1.72 | 0.19 | 0.65 (0.34 to 1.24) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sarathkumara, Y.D.; Gamage, C.D.; Lokupathirage, S.; Muthusinghe, D.S.; Nanayakkara, N.; Gunarathne, L.; Shimizu, K.; Tsuda, Y.; Arikawa, J.; Yoshimatsu, K. Exposure to Hantavirus is a Risk Factor Associated with Kidney Diseases in Sri Lanka: A Cross Sectional Study. Viruses 2019, 11, 700. https://doi.org/10.3390/v11080700
Sarathkumara YD, Gamage CD, Lokupathirage S, Muthusinghe DS, Nanayakkara N, Gunarathne L, Shimizu K, Tsuda Y, Arikawa J, Yoshimatsu K. Exposure to Hantavirus is a Risk Factor Associated with Kidney Diseases in Sri Lanka: A Cross Sectional Study. Viruses. 2019; 11(8):700. https://doi.org/10.3390/v11080700
Chicago/Turabian StyleSarathkumara, Yomani D., Chandika D. Gamage, Sithumini Lokupathirage, Devinda S. Muthusinghe, Nishantha Nanayakkara, Lishanthe Gunarathne, Kenta Shimizu, Yoshimi Tsuda, Jiro Arikawa, and Kumiko Yoshimatsu. 2019. "Exposure to Hantavirus is a Risk Factor Associated with Kidney Diseases in Sri Lanka: A Cross Sectional Study" Viruses 11, no. 8: 700. https://doi.org/10.3390/v11080700
APA StyleSarathkumara, Y. D., Gamage, C. D., Lokupathirage, S., Muthusinghe, D. S., Nanayakkara, N., Gunarathne, L., Shimizu, K., Tsuda, Y., Arikawa, J., & Yoshimatsu, K. (2019). Exposure to Hantavirus is a Risk Factor Associated with Kidney Diseases in Sri Lanka: A Cross Sectional Study. Viruses, 11(8), 700. https://doi.org/10.3390/v11080700





