A Review of Hantavirus Research in Indonesia: Prevalence in Humans and Rodents, and the Discovery of Serang Virus
Abstract
1. Introduction
2. Methods
2.1. Estimating the Prevalence of Previous Hantavirus Infection (PHI) and Identifying Acute Hantavirus Infection (AHI)
2.2. Review of Published AHIs and PHIs in Humans and Serological and Molecular Evidence of HTV Infections in Rodents
3. Results
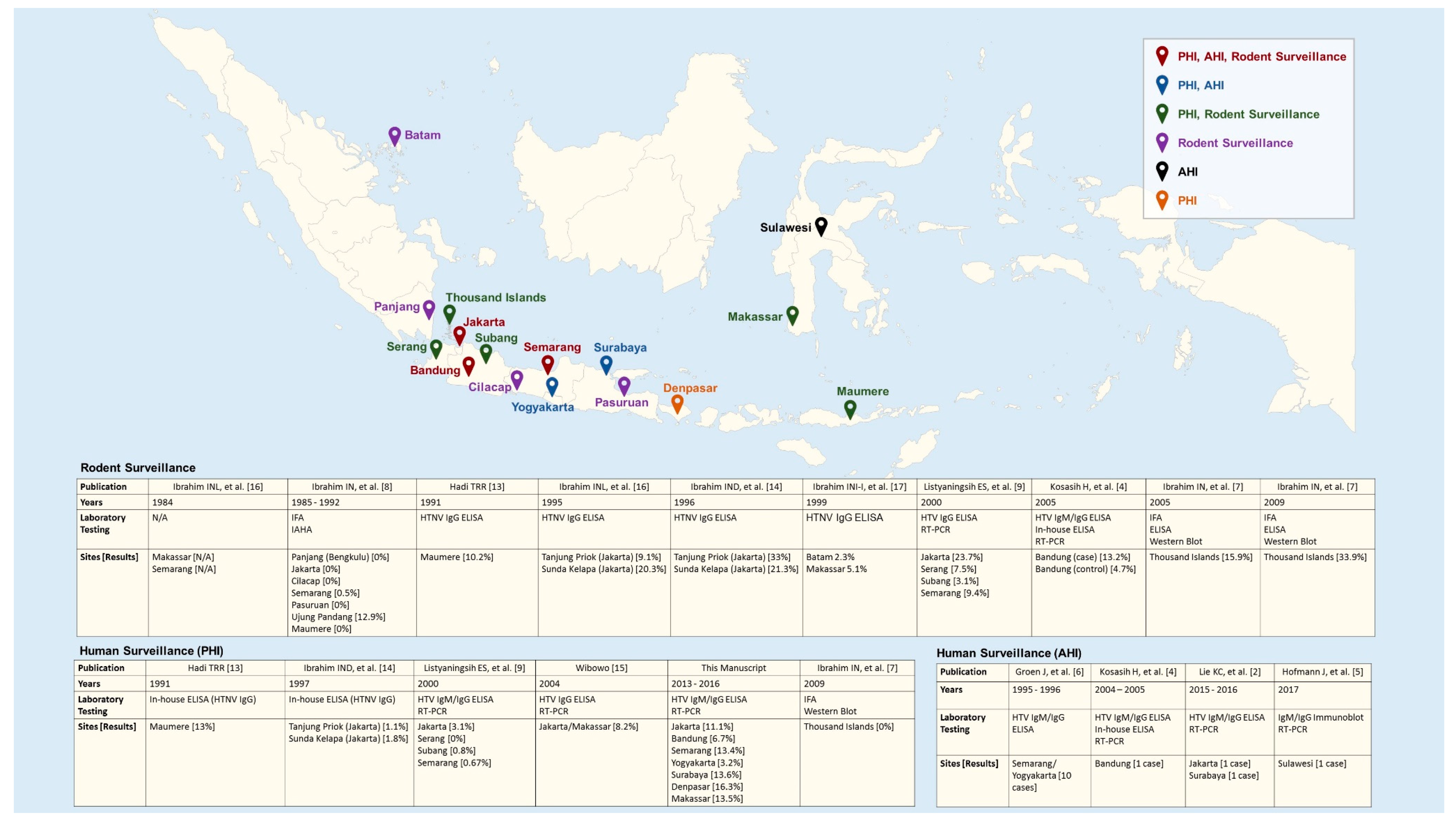
3.1. Prevalence of PHI in Humans
3.2. Acute Hantavirus Infections
3.3. Serological Surveillance in Rodents
3.4. Molecular Surveillance in Rodents
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- International Committee on Taxonomy of Viruses. Available online: https://talk.ictvonline.org/taxonomy/ (accessed on 25 July 2019).
- Milholland, M.T.; Castro-Arellano, I.; Suzan, G.; Garcia-Pena, G.E.; Lee, T.E., Jr.; Rohde, R.E.; Alonso Aguirre, A.; Mills, J.N. Global diversity and distribution of hantaviruses and their hosts. Ecohealth 2018, 15, 163–208. [Google Scholar] [CrossRef] [PubMed]
- Avšič-Županc, T.; Saksida, A.; Korva, M. Hantavirus infections. Clin. Microbiol. Infect. 2019, 21, e6–e16. [Google Scholar] [CrossRef] [PubMed]
- Lie, K.C.; Aziz, M.H.; Kosasih, H.; Neal, A.; Halim, C.L.; Wulan, W.N.; Karyana, M.; Hadi, U. Case report: Two confirmed cases of human seoul virus infections in Indonesia. BMC Infect. Dis. 2018, 18, 578. [Google Scholar] [CrossRef] [PubMed]
- Suharti, C.; van Gorp, E.C.; Dolmans, W.M.; Groen, J.; Hadisaputro, S.; Djokomoeljanto, R.J.; OA, D.M.E.; van der Meer, J.W. Hanta virus infection during dengue virus infection outbreak in Indonesia. Acta Med. Indones 2009, 41, 75–80. [Google Scholar] [PubMed]
- Kosasih, H.; Ibrahim, I.N.; Wicaksana, R.; Alisjahbana, B.; Hoo, Y.; Yo, I.H.; Antonjaya, U.; Widjaja, S.; Winoto, I.; Williams, M.; et al. Evidence of human hantavirus infection and zoonotic investigation of hantavirus prevalence in rodents in western Java, Indonesia. Vector Borne Zoonotic Dis. 2011, 11, 709–713. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, J.; Weiss, S.; Kuhns, M.; Zinke, A.; Heinsberger, H.; Kruger, D.H. Importation of human seoul virus infection to Germany from Indonesia. Emerg. Infect. Dis. 2018, 24, 1099–1102. [Google Scholar] [CrossRef] [PubMed]
- Groen, J.; Suharti, C.; Koraka, P.; van Gorp, E.C.; Sutaryo, J.; Lundkvist, A.; Osterhaus, A.D. Serological evidence of human hantavirus infections in Indonesia. Infection 2002, 30, 326–327. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, I.N.; Shimizu, K.; Yoshimatsu, K.; Yunianto, A.; Salwati, E.; Yasuda, S.P.; Koma, T.; Endo, R.; Arikawa, J. Epidemiology of hantavirus infection in thousand islands regency of Jakarta, Indonesia. J. Vet. Med. Sci. 2013, 75, 1003–1008. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, I.N.; Sudomo, M.; Morita, C.; Uemura, S.; Muramatsu, Y.; Ueno, H.; Kitamura, T. Seroepidemiological survey of wild rats for seoul virus in Indonesia. Jpn J. Med. Sci. Biol. 1996, 49, 69–74. [Google Scholar] [CrossRef][Green Version]
- Listyaningsih, E.S.; Kosasih, H.; Ibrahim, I.N.; Widjaja, S.; Tan, R.; Winoto, I.; Porter, K.R.; Beckett, C.; Blair, P.J. The Finding of Puumala and Seoul Hantaviruses in Rattus sp within Java, Indonesia; ASTMH: Washington, DC, USA, 2005. [Google Scholar]
- Mulyono, A.R.P.; Dimas, B.W. Dual infection with leptospira and hantavirus at rattus norvegicus in Maumere, Flores, Nusa Tenggara Timur. J. Litbang Pengendali. Penyakit Bersumber Binatang Banjrnegara 2017, 13. [Google Scholar] [CrossRef]
- Klempa, B.; Fichet-Calvet, E.; Lecompte, E.; Auste, B.; Aniskin, V.; Meisel, H.; Denys, C.; Koivogui, L.; ter Meulen, J.; Kruger, D.H. Hantavirus in african wood mouse, guinea. Emerg. Infect. Dis. 2006, 12, 838–840. [Google Scholar] [CrossRef] [PubMed]
- Dekonenko, A.; Ibrahim, M.S.; Schmaljohn, C.S. A colorimetric pcr-enzyme immunoassay to identify hantaviruses. Clin. Diagn. Virol. 1997, 8, 113–121. [Google Scholar] [CrossRef]
- Hadi, T.R. Laporan Penelitian Penyakit Virus Hantaan Bersumber Tikus di Pelabuhan Maumere, Flores; Pusat Penelitian Ekologi Kesehatan, B: Jakarta, Indonesia, 1992. [Google Scholar]
- Ibrahim, I.N.; Kasnodihardjo, K.; Lestari, E.W.; Kursino, E.; Wijono, W. Laporan Akhir Penelitian Ekologi Penyakit Bersumber Rodensia (Tikut, Mencit) dan Insectivore (Cucurut) di Kota Pelabuhan Laut di Indonesia; Pusat Penelitian Ekologi Kesehatan, B, Badan Penelitian dan Pengembangan Kesehatan Jakarta Kementerian Kesehatan: Jakarta, Indonesia, 1997. [Google Scholar]
- Wibowo, W. Epidemiologi hantavirus di Indonesia. Bull. Penelit. Kesehat. 2010, 38. [Google Scholar] [CrossRef]
- Ibrahim, I.N.; Lestari, E.W.; Kursino, E.; Wijono, W. Laporan Akhir Penelitian Ekologi Penyakit Bersumber Rodensia (Tikus, Mencit) dan Cecurut di Kota Pelabuhan Laut di Indonesia; Pusat Penelitian Ekologi Kesehatan, B, Badan Penelitian dan Pengembangan Kesehatan Jakarta: Jakarta, Indonesia, 1996. [Google Scholar]
- Ibrahim, I.N.; Idram, N.S.I.; Erlina, S.; Kursino, E.; Sumarno, S. Laporan Penelitian Infeksi Hantavirus di Beberapa Pelabuhan Laut di Indonesia (Lanjutan Tahap ii: Survei Serologis Infeksi Hantavirus Pada Manusia dan Hewan Reservoir); Kesehatan, P.P.E., Ed.; Badan Penelitian dan Pengembangan Kesehatan Jakarta: Jakarta, Indonesia, 2002. [Google Scholar]
- Plyusnina, A.; Ibrahim, I.N.; Plyusnin, A. A newly recognized hantavirus in the asian house rat (rattus tanezumi) in Indonesia. J. Gen. Virol 2009, 90, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Lederer, S.; Lattwein, E.; Hanke, M.; Sonnenberg, K.; Stoecker, W.; Lundkvist, A.; Vaheri, A.; Vapalahti, O.; Chan, P.K.; Feldmann, H.; et al. Indirect immunofluorescence assay for the simultaneous detection of antibodies against clinically important old and new world hantaviruses. PLoS Negl. Trop. Dis. 2013, 7, e2157. [Google Scholar] [CrossRef] [PubMed]
- Hadi, U.; Alisjahbana, B.; Lokida, D.; Lie, K.C.; Susila, I.M.; Pelupessy, N.M.; Kosasih, H.; Wibawa, T.; Tjitra, E.; Karyana, M.; et al. Underdiagnoses of Rodent-Borne Diseases in Patients Hospitalized with Acute Fever in Indonesia; ASTMH: New Orleans, LA, USA, 2018. [Google Scholar]
- Bi, Z.; Formenty, P.B.; Roth, C.E. Hantavirus infection: A review and global update. J. Infect. Dev. Ctries 2008, 2, 3–23. [Google Scholar] [CrossRef] [PubMed]
- Kruger, D.H.; Schonrich, G.; Klempa, B. Human pathogenic hantaviruses and prevention of infection. Hum. Vaccin. 2011, 7, 685–693. [Google Scholar] [CrossRef] [PubMed]
- Hart, C.A.; Bennett, M. Hantavirus infections: Epidemiology and pathogenesis. Microbes Infect. 1999, 1, 1229–1237. [Google Scholar] [CrossRef]
- Chaloemwong, J.; Tantiworawit, A.; Rattanathammethee, T.; Hantrakool, S.; Chai-Adisaksopha, C.; Rattarittamrong, E.; Norasetthada, L. Useful clinical features and hematological parameters for the diagnosis of dengue infection in patients with acute febrile illness: A retrospective study. BMC Hematol. 2018, 18, 20. [Google Scholar] [CrossRef]
- Zhang, Y.Z.; Zou, Y.; Yan, Y.Z.; Hu, G.W.; Yao, L.S.; Du, Z.S.; Jin, L.Z.; Liu, Y.Y.; Li, M.H.; Chen, H.X.; et al. Detection of phylogenetically distinct puumala-like viruses from red-grey vole clethrionomys rufocanus in China. J. Med. Virol. 2007, 79, 1208–1218. [Google Scholar] [CrossRef]
- Kang, H.J.; Kosoy, M.Y.; Shrestha, S.K.; Shrestha, M.P.; Pavlin, J.A.; Gibbons, R.V.; Yanagihara, R. Short report: Genetic diversity of thottapalayam virus, a hantavirus harbored by the asian house shrew (suncus murinus) in Nepal. Am. J. Trop. Med. Hyg. 2011, 85, 540–545. [Google Scholar] [CrossRef] [PubMed]
| Tested/ Subjects | Positive (%) | Pediatric Pos/Tested (%) | Adults Pos/Tested (%) | Male Pos/Tested (%) | Female Pos/Tested (%) | |
|---|---|---|---|---|---|---|
| Bandung | 45/269 | 3 (6.7) | 1/12 (8.3) | 2/33 (6.1) | 2/27 (7.4) | 1/18 (5.6) |
| Jakarta | 45/156 | 5 (11.1) | 2/25 (8) | 3/20 (15) | 3/25 (12) | 2/20 (10) |
| Semarang | 67/257 | 9 (13.4) | 0/7 (0) | 9/60 (15) | 6/35 (17.1) | 3/32 (9.4) |
| Yogyakarta | 31/169 | 1 (3.2) | 0/10 (0) | 1/21 (4.8) | 1/20 (5) | 0/11 (0) |
| Surabaya | 44/221 | 6 (13.6) | 0/9 (0) | 6/35 (17.1) | 6/23 (26) | 0/21 (0) |
| Denpasar | 43/213 | 7 (16.3) | 0/7 (0) | 7/36 (19.4) | 6/23 (26) | 1/20 (5) |
| Makassar | 52/201 | 7 (13.5) | 2/13 (15.4) | 5/39 (12.8) | 5/33 (15.2) | 2/19 (10.5) |
| Total | 327/1486 | 38 (11.6) | 5/83 (6) | 33/244 (13.5) | 29/186 (15.6) | 9/131 (6.9) |
| Groen (2002), Suharti (2009) | Kosasih (2011) | Hofmann (2018) | Lie (2018) | |
|---|---|---|---|---|
| City/island | Semarang, Yogyakarta (Central Java) | Bandung | Sulawesi | Surabaya, Jakarta |
| Year(s) | 1995–1996 | 2004–2005 | 2017 | 2015–2016 |
| Number of cases | 10 | 1 | 1 | 2 |
| Methods of confirmation | HTV IgM and IgG ELISA and IFA (SEOV, HTN, PUU), Immunoblotting | HTV IgM and IgG ELISA, in-house ELISA RT-PCR (negative) | IgM, IgG Immunoblot (DOBV, HTNV, SEOV), RT-PCR (positive) | HTV IgM and IgG ELISA RT-PCR (positive) |
| Evidence of rodent infection | N/A | Yes | N/A | N/A |
| Demography * | ||||
| Age | Mean: 17.6 (13-N/A) 90% under 20 years old | 25 | 70 | 55; 27 |
| Gender (male:female) | 2:8 | male | male | 1:1 |
| Clinical ** | ||||
| Constitutional symptoms | 100% (Fever, headache) 80% (Weakness, joint/muscle pain, nausea/vomiting, epigastric pain) 20% (Retro-orbital pain) | Fever, headache, nausea, vomiting, abdominal discomfort, loss of appetite, malaise | Fever, severe diarrhea, thoracic/back pain, bronchopulmonary symptom | Patient 1. Fever, abdominal/epigastric pain, nausea, vomiting, retro-orbital pain. Patient 2. Fever, anorexia, headache, nausea, vomiting, arthralgia |
| Signs | 40% Hepatomegaly 20% (Lymphadenopathy, epistaxis/gum bleeding, hematemesis/melena) | Patient 1. Lethargy Patient 2. Decrease of consciousness, jaundice. | ||
| rash | 40% Exanthema 80% Petechiae | |||
| Hematology (reference range) | Median (range) | |||
| Hb (mg/dL) ** | 12 (10.6–13.7) | N/A | 17.6 | 13.2; 16.5 |
| Hct (40–50%) ** | 37.7 (34–41.7) | 50 | N/A | 38.8; 50.6 |
| Leukocytes (3500–9000 cells/dL) * | Normal leukocytes (50%) 5300 (3600–6300) | 9600 | 11.7–16.8 | 3850; 5790 |
| Platelet (150,000–450,000 cells/dL) * | Normal Thrombocytes (50%) 155,000 (24,000–267,000) | 38,000 | 66,000 | 50,000; 24,700 |
| Lymphocyte (23.1–49.9%) | N/A | N/A | N/A | 8.4; 16 |
| Chemistries ** | ||||
| BUN (10–23 mg/dL) | N/A | 25 | 240 | 10; 20.3 |
| Creatinine (0.5–1.2 mg/dL) | 0.8 (0.6–0.93) | 0.9 | 5.5 | 0.57; 1.16 |
| Protein (6.3–8.2 g/dL) | N/A | 5.8 | N/A | N/A |
| Albumin (3.5–5.2 g/dL) | N/A | 3.3 | N/A | N/A |
| Bilirubin (<1.4 mg/dL) | 0.53 (0.46–1.32) | N/A | N/A | 0.97; 4.68 |
| SGOT (<38 U/L) | 15 (3–56) | 130 | 234 | 216; 3900 |
| SGPT (<41 U/L) | 17 (6–27) | 76 | 173 | 116; 891 |
| GGT (<60 U/L) | N/A | N/A | 159 | N/A |
| CRP (<5 mg/dL) | N/A | N/A | 54.4 | N/A; 41.1 |
| Procalcitonin (<0.5 ng/dL) | N/A | N/A | N/A | N/A; 5.98 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lukman, N.; Kosasih, H.; Ibrahim, I.N.; Pradana, A.A.; Neal, A.; Karyana, M. A Review of Hantavirus Research in Indonesia: Prevalence in Humans and Rodents, and the Discovery of Serang Virus. Viruses 2019, 11, 698. https://doi.org/10.3390/v11080698
Lukman N, Kosasih H, Ibrahim IN, Pradana AA, Neal A, Karyana M. A Review of Hantavirus Research in Indonesia: Prevalence in Humans and Rodents, and the Discovery of Serang Virus. Viruses. 2019; 11(8):698. https://doi.org/10.3390/v11080698
Chicago/Turabian StyleLukman, Nurhayati, Herman Kosasih, Ima Nurisa Ibrahim, Antonius Arditya Pradana, Aaron Neal, and Muhammad Karyana. 2019. "A Review of Hantavirus Research in Indonesia: Prevalence in Humans and Rodents, and the Discovery of Serang Virus" Viruses 11, no. 8: 698. https://doi.org/10.3390/v11080698
APA StyleLukman, N., Kosasih, H., Ibrahim, I. N., Pradana, A. A., Neal, A., & Karyana, M. (2019). A Review of Hantavirus Research in Indonesia: Prevalence in Humans and Rodents, and the Discovery of Serang Virus. Viruses, 11(8), 698. https://doi.org/10.3390/v11080698





