Evolution and Interspecies Transmission of Canine Distemper Virus—An Outlook of the Diverse Evolutionary Landscapes of a Multi-Host Virus
Abstract
1. Introduction
2. Disease in Domestic Dogs and Wildlife
3. Reservoirs vs Viral Adaptation: Two Different Transmission Sceneries
3.1. The Role of Reservoirs in CDV Epidemiology
3.2. Role of Asymptomatic Animals in CDV Transmission
3.3. Viral Adaptation: the Role of Specialist vs Generalist Strains
4. CDV and Measles: Complementary Infection Models
Genetic Variability of CDV
5. Evolutionary Rates and Molecular Clock of CDV
6. Pathogenicity Predictions
7. H Gene vs. Complete Genome
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- MacLachlan, N.; Dubovi, E.; Fenner, F. Paramyxoviridae. In Fenner’s Veterinary Virology, 4th ed.; Academic Press: Boston, MA, USA, 2011; pp. 299–325. [Google Scholar]
- King, A.M.Q.; Lefkowitz, E.J.; Mushegian, A.R.; Adams, M.J.; Dutilh, B.E.; Gorbalenya, A.E.; Harrach, B.; Harrison, R.L.; Junglen, S.; Knowles, N.J.; et al. Changes to taxonomy and the international code of virus classification and nomenclature ratified by the international committee on taxonomy of viruses (2018). Arch. Virol. 2018, 163, 2601–2631. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Gutierrez, M.; Ruiz-Saenz, J. Diversity of susceptible hosts in canine distemper virus infection: A systematic review and data synthesis. BMC Vet. Res. 2016, 12, 78. [Google Scholar] [CrossRef] [PubMed]
- Loots, A.K.; Mitchell, E.; Dalton, D.L.; Kotze, A.; Venter, E.H. Advances in canine distemper virus pathogenesis research: A wildlife perspective. J. Gen. Virol. 2017, 98, 311–321. [Google Scholar] [CrossRef] [PubMed]
- Anderson, D.E.; Von Messling, V. Region between the canine distemper virus M and F genes modulates virulence by controlling fusion protein expression. J. Virol. 2008, 82, 10510–10518. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, N.D.; Dunavan, C.P.; Diamond, J. Origins of major human infectious diseases. Nature 2007, 447, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Schuhmann, K.M.; Pfaller, C.K.; Conzelmann, K.K. The measles virus V protein binds to p65 (Rela) to suppress NF-kappaB activity. J. Virol. 2011, 85, 3162–3171. [Google Scholar] [CrossRef] [PubMed]
- Chinnakannan, S.K.; Nanda, S.K.; Baron, M.D. Morbillivirus v proteins exhibit multiple mechanisms to block type 1 and type 2 interferon signalling pathways. PLoS ONE 2013, 8, e57063. [Google Scholar] [CrossRef]
- Rothlisberger, A.; Wiener, D.; Schweizer, M.; Peterhans, E.; Zurbriggen, A.; Plattet, P. Two domains of the v protein of virulent canine distemper virus selectively inhibit stat1 and stat2 nuclear import. J. Virol. 2010, 84, 6328–6343. [Google Scholar] [CrossRef]
- Svitek, N.; Gerhauser, I.; Goncalves, C.; Grabski, E.; Döring, M.; Kalinke, U.; Anderson, D.E.; Cattaneo, R.; Von Messling, V. Morbillivirus control of the interferon response: Relevance of stat2 and mda5 but not stat1 for canine distemper virus virulence in ferrets. J. Virol. 2013, 88, 2941–2950. [Google Scholar] [CrossRef]
- Pomeroy, L.W.; Bjornstad, O.N.; Holmes, E.C. The evolutionary and epidemiological dynamics of the paramyxoviridae. J. Mol. Evol. 2008, 66, 98–106. [Google Scholar] [CrossRef]
- Sarkar, J.; Balamurugan, V.; Sen, A.; Saravanan, P.; Sahay, B.; Rajak, K.K.; Rasool, T.J.; Bhanuprakash, V.; Singh, R.K. Sequence analysis of morbillivirus CD150 receptor-signaling lymphocyte activation molecule (SLAM) of different animal species. Virus Genes 2009, 39, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Tatsuo, H.; Yanagi, Y. The morbillivirus receptor SLAM (CD150). Microbiol. Immunol. 2002, 46, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Noyce, R.S.; Bondre, D.G.; Ha, M.N.; Lin, L.-T.; Sisson, G.; Tsao, M.-S.; Richardson, C.D. Tumor cell marker PVRL4 (nectin 4) is an epithelial cell receptor for measles virus. PLoS Pathog. 2011, 7, e1002240. [Google Scholar] [CrossRef] [PubMed]
- Schultz, R.D.; Thiel, B.; Mukhtar, E.; Sharp, P.; Larson, L.J. Age and long-term protective immunity in dogs and cats. J. Comp. Pathol. 2010, 142, S102–S108. [Google Scholar] [CrossRef] [PubMed]
- Bolt, G.; Jensen, T.D.; Gottschalck, E.; Arctander, P.; Appel, M.J.; Buckland, R.; Blixenkrone-Moller, M. Genetic diversity of the attachment (H) protein gene of current field isolates of canine distemper virus. J. Gen. Virol. 1997, 78, 367–372. [Google Scholar] [CrossRef] [PubMed]
- Russell, C.J.; Jardetzky, T.S.; Lamb, R.A. Membrane fusion machines of paramyxoviruses: Capture of intermediates of fusion. EMBO J. 2001, 20, 4024–4034. [Google Scholar] [CrossRef] [PubMed]
- Bringolf, F.; Herren, M.; Wyss, M.; Vidondo, B.; Langedijk, J.P.; Zurbriggen, A.; Plattet, P. Dimerization efficiency of canine distemper virus matrix protein regulates membrane-budding activity. J. Virol. 2017, 91. [Google Scholar] [CrossRef] [PubMed]
- Sarute, N.; Calderon, M.G.; Perez, R.; La Torre, J.; Hernandez, M.; Francia, L.; Panzera, Y. The fusion protein signal-peptide-coding region of canine distemper virus: A useful tool for phylogenetic reconstruction and lineage identification. PLoS ONE 2013, 8, e63595. [Google Scholar] [CrossRef]
- Elia, G.; Camero, M.; Losurdo, M.; Lucente, M.S.; Larocca, V.; Martella, V.; Decaro, N.; Buonavoglia, C. Virological and serological findings in dogs with naturally occurring distemper. J. Virol. Methods 2015, 213, 127–130. [Google Scholar] [CrossRef]
- De Vries, R.D.; Ludlow, M.; de Jong, A.; Rennick, L.J.; Verburgh, R.J.; van Amerongen, G.; van Riel, D.; van Run, P.; Herfst, S.; Kuiken, T.; et al. Delineating morbillivirus entry, dissemination and airborne transmission by studying in vivo competition of multicolor canine distemper viruses in ferrets. PLoS Pathog. 2017, 13, e1006371. [Google Scholar] [CrossRef]
- Summers, B.A.; Greisen, H.A.; Appel, M.J. Early events in canine distemper demyelinating encephalomyelitis. Acta Neuropathol. 1979, 46, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Alves, L.; Khosravi, M.; Avila, M.; Ader-Ebert, N.; Bringolf, F.; Zurbriggen, A.; Vandevelde, M.; Plattet, P. SLAM- and nectin-4-independent noncytolytic spread of canine distemper virus in astrocytes. J. Virol. 2015, 89, 5724–5733. [Google Scholar] [CrossRef] [PubMed]
- Rudd, P.A.; Cattaneo, R.; Von Messling, V. Canine distemper virus uses both the anterograde and the hematogenous pathway for neuroinvasion. J. Virol. 2006, 80, 9361–9370. [Google Scholar] [CrossRef] [PubMed]
- Von Messling, V.; Springfeld, C.; Devaux, P.; Cattaneo, R. A ferret model of canine distemper virus virulence and immunosuppression. J. Virol. 2003, 77, 12579–12591. [Google Scholar] [CrossRef] [PubMed]
- Koutinas, A.F.; Baumgartner, W.; Tontis, D.; Polizopoulou, Z.; Saridomichelakis, M.N.; Lekkas, S. Histopathology and immunohistochemistry of canine distemper virus-induced footpad hyperkeratosis (hard pad disease) in dogs with natural canine distemper. Vet. Pathol. 2004, 41, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Lunardi, M.; Darold, G.M.; Amude, A.M.; Headley, S.A.; Sonne, L.; Yamauchi, K.C.I.; Boabaid, F.M.; Alfieri, A.F.; Alfieri, A.A. Canine distemper virus active infection in order pilosa, family myrmecophagidae, species tamandua tetradactyla. Vet. Microbiol. 2018, 220, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Feng, N.; Yu, Y.; Wang, T.; Wilker, P.; Wang, J.; Li, Y.; Sun, Z.; Gao, Y.; Xia, X. Fatal canine distemper virus infection of giant pandas in China. Sci. Rep. 2016, 6, 27518. [Google Scholar] [CrossRef] [PubMed]
- Gordon, C.H.; Banyard, A.C.; Hussein, A.; Laurenson, M.K.; Malcolm, J.R.; Marino, J.; Regassa, F.; Stewart, A.M.; Fooks, A.R.; Sillero-Zubiri, C. Canine distemper in endangered ethiopian wolves. Emerg. Infect. Dis. 2015, 21, 824–832. [Google Scholar] [CrossRef]
- Seimon, T.A.; Miquelle, D.G.; Chang, T.Y.; Newton, A.L.; Korotkova, I.; Ivanchuk, G.; Lyubchenko, E.; Tupikov, A.; Slabe, E.; McAloose, D. Canine distemper virus: An emerging disease in wild endangered amur tigers (Panthera tigris altaica). MBio 2013, 4. [Google Scholar] [CrossRef]
- Terio, K.A.; Craft, M.E. Canine distemper virus (CDV) in another big cat: Should cdv be renamed carnivore distemper virus? MBio 2013, 4, e00702–e00713. [Google Scholar] [CrossRef]
- Qiu, W.; Zheng, Y.; Zhang, S.; Fan, Q.; Liu, H.; Zhang, F.; Wang, W.; Liao, G.; Hu, R. Canine distemper outbreak in rhesus monkeys, China. Emerg. Infect. Dis. 2011, 17, 1541–1543. [Google Scholar] [CrossRef]
- Nikolin, V.M.; Olarte-Castillo, X.A.; Osterrieder, N.; Hofer, H.; Dubovi, E.; Mazzoni, C.J.; Brunner, E.; Goller, K.V.; Fyumagwa, R.D.; Moehlman, P.D.; et al. Canine distemper virus in the serengeti ecosystem: Molecular adaptation to different carnivore species. Mol. Ecol. 2017, 26, 2111–2130. [Google Scholar] [CrossRef]
- Haas, L.; Hofer, H.; East, M.; Wohlsein, P.; Liess, B.; Barrett, T. Canine distemper virus infection in Serengeti spotted hyenas. Vet. Microbiol. 1996, 49, 147–152. [Google Scholar] [CrossRef]
- Harder, T.C.; Kenter, M.; Appel, M.J.; Roelke-Parker, M.E.; Barrett, T.; Osterhaus, A.D. Phylogenetic evidence of canine distemper virus in serengeti’s lions. Vaccine 1995, 13, 521–523. [Google Scholar] [CrossRef]
- Roelke-Parker, M.E.; Munson, L.; Packer, C.; Kock, R.; Cleaveland, S.; Carpenter, M.; O’Brien, S.J.; Pospischil, A.; Hofmann-Lehmann, R.; Lutz, H.; et al. A canine distemper virus epidemic in serengeti lions (Panthera leo). Nature 1996, 379, 441–445. [Google Scholar] [CrossRef]
- Lednicky, J.A.; Dubach, J.; Kinsel, M.J.; Meehan, T.P.; Bocchetta, M.; Hungerford, L.L.; Sarich, N.A.; Witecki, K.E.; Braid, M.D.; Pedrak, C.; et al. Genetically distant american canine distemper virus lineages have recently caused epizootics with somewhat different characteristics in raccoons living around a large suburban zoo in the USA. Virol. J. 2004, 1, 2. [Google Scholar] [CrossRef]
- Davidson, W.R.; Appel, M.J.; Doster, G.L.; Baker, O.E.; Brown, J.F. Diseases and parasites of red foxes, gray foxes, and coyotes from commercial sources selling to fox-chasing enclosures. J. Wildl. Dis. 1992, 28, 581–589. [Google Scholar] [CrossRef]
- Williams, E.S.; Thorne, E.T.; Appel, M.J.; Belitsky, D.W. Canine distemper in black-footed ferrets (Mustela nigripes) from wyoming. J. Wildl. Dis. 1988, 24, 385–398. [Google Scholar] [CrossRef]
- Denzin, N.; Herwig, V.; van der Grinten, E. Occurrence and geographical distribution of canine distemper virus infection in red foxes (vulpes vulpes) of saxony-anhalt, germany. Vet. Microbiol. 2013, 162, 214–218. [Google Scholar] [CrossRef]
- Lopez-Pena, M.; Quiroga, M.I.; Vazquez, S.; Nieto, J.M. Detection of canine distemper viral antigen in foxes (vulpes vulpes) in northwestern spain. J. Wildl. Dis. 1994, 30, 95–98. [Google Scholar] [CrossRef]
- Martella, V.; Bianchi, A.; Bertoletti, I.; Pedrotti, L.; Gugiatti, A.; Catella, A.; Cordioli, P.; Lucente, M.S.; Elia, G.; Buonavoglia, C. Canine distemper epizootic among red foxes, Italy, 2009. Emerg. Infect. Dis. 2010, 16, 2007. [Google Scholar] [CrossRef]
- Avendano, R.; Barrueta, F.; Soto-Fournier, S.; Chavarria, M.; Monge, O.; Gutierrez-Espeleta, G.A.; Chaves, A. Canine Distemper Virus in Wild Felids of Costa Rica. J. Wildl. Dis. 2016, 52, 373–377. [Google Scholar] [CrossRef]
- Megid, J.; de Souza, V.A.; Teixeira, C.R.; Cortez, A.; Amorin, R.L.; Heinemman, M.B.; Cagnini, D.Q.; Richtzenhain, L.J. Canine distemper virus in a crab-eating fox (Cerdocyon thous) in brazil: Case report and phylogenetic analyses. J. Wildl. Dis. 2009, 45, 527–530. [Google Scholar] [CrossRef]
- Nava, A.F.; Cullen, L., Jr.; Sana, D.A.; Nardi, M.S.; Filho, J.D.; Lima, T.F.; Abreu, K.C.; Ferreira, F. First evidence of canine distemper in Brazilian free-ranging felids. Ecohealth 2008, 5, 513–518. [Google Scholar] [CrossRef]
- Furtado, M.M.; Hayashi, E.M.; Allendorf, S.D.; Coelho, C.J.; de Almeida Jacomo, A.T.; Megid, J.; Ramos Filho, J.D.; Silveira, L.; Torres, N.M.; Ferreira Neto, J.S. Exposure of free-ranging wild carnivores and domestic dogs to canine distemper virus and parvovirus in the cerrado of central Brazil. Ecohealth 2016, 13, 549–557. [Google Scholar] [CrossRef]
- Woodroffe, R.; Cleaveland, S.; Courtenay, O.; Laurenson, M.K.; Artois, A. Infectious disease. In The Biology and Conservation of Wild Canids; Macdonald, D.W., Sillero-Zubiri, C., Eds.; Oxford University Press: Oxford, UK, 2004; pp. 123–142. [Google Scholar]
- Beineke, A.; Puff, C.; Seehusen, F.; Baumgartner, W. Pathogenesis and immunopathology of systemic and nervous canine distemper. Vet. Immunol. Immunopathol. 2009, 127, 1–18. [Google Scholar] [CrossRef]
- Viana, M.; Cleaveland, S.; Matthiopoulos, J.; Halliday, J.; Packer, C.; Craft, M.E.; Hampson, K.; Czupryna, A.; Dobson, A.P.; Dubovi, E.J.; et al. Dynamics of a morbillivirus at the domestic-wildlife interface: Canine distemper virus in domestic dogs and lions. Proc. Nat. Acad. Sci. USA 2015, 112, 1464–1469. [Google Scholar] [CrossRef]
- McCarthy, A.J.; Shaw, M.A.; Goodman, S.J. Pathogen evolution and disease emergence in carnivores. Proc. Biol. Sci. 2007, 274, 3165–3174. [Google Scholar] [CrossRef]
- Ohishi, K.; Ando, A.; Suzuki, R.; Takishita, K.; Kawato, M.; Katsumata, E.; Ohtsu, D.; Okutsu, K.; Tokutake, K.; Miyahara, H. Host–virus specificity of morbilliviruses predicted by structural modeling of the marine mammal SLAM, a receptor. Comp. Immunol. Microbiol. Infect. Dis. 2010, 33, 227–241. [Google Scholar] [CrossRef]
- Kapil, S.; Allison, R.W.; Johnston, L.; Murray, B.L.; Holland, S.; Meinkoth, J.; Johnson, B. Canine distemper virus strains circulating among north american dogs. Clin. Vaccine Immunol. 2008, 15, 707–712. [Google Scholar] [CrossRef]
- Kapil, S.; Yeary, T.J. Canine distemper spillover in domestic dogs from urban wildlife. Vet. Clin. North Am. Small Anim. Pract 2011, 41, 1069–1086. [Google Scholar] [CrossRef]
- Panzera, Y.; Sarute, N.; Iraola, G.; Hernandez, M.; Perez, R. Molecular phylogeography of canine distemper virus: Geographic origin and global spreading. Mol. Phylogenet. Evol. 2015, 92, 147–154. [Google Scholar] [CrossRef]
- Haydon, D.T.; Cleaveland, S.; Taylor, L.H.; Laurenson, M.K. Identifying reservoirs of infection: A conceptual and practical challenge. Emerg. Infect. Dis. 2002, 8, 1468–1473. [Google Scholar]
- Viana, M.; Mancy, R.; Biek, R.; Cleaveland, S.; Cross, P.C.; Lloyd-Smith, J.O.; Haydon, D.T. Assembling evidence for identifying reservoirs of infection. Trends Ecol. Evol. 2014, 29, 270–279. [Google Scholar] [CrossRef]
- Riley, M.C.; Wilkes, R.P. Sequencing of emerging canine distemper virus strain reveals new distinct genetic lineage in the united states associated with disease in wildlife and domestic canine populations. Virol. J. 2015, 12, 219. [Google Scholar] [CrossRef]
- Goller, K.V.; Fyumagwa, R.D.; Nikolin, V.; East, M.L.; Kilewo, M.; Speck, S.; Muller, T.; Matzke, M.; Wibbelt, G. Fatal canine distemper infection in a pack of African wild dogs in the Serengeti ecosystem, Tanzania. Vet. Microbiol. 2010, 146, 245–252. [Google Scholar] [CrossRef]
- Harder, T.C.; Kenter, M.; Vos, H.; Siebelink, K.; Huisman, W.; van Amerongen, G.; Orvell, C.; Barrett, T.; Appel, M.J.; Osterhaus, A.D. Canine distemper virus from diseased large felids: Biological properties and phylogenetic relationships. J. Gen. Virol. 1996, 77, 397–405. [Google Scholar] [CrossRef]
- Martella, V.; Elia, G.; Buonavoglia, C. Canine distemper virus. Vet. Clin. N. Am. Small Anim. Pract. 2008, 38, 787–797. [Google Scholar] [CrossRef]
- Budaszewski Rda, F.; Pinto, L.D.; Weber, M.N.; Caldart, E.T.; Alves, C.D.; Martella, V.; Ikuta, N.; Lunge, V.R.; Canal, C.W. Genotyping of canine distemper virus strains circulating in Brazil from 2008 to 2012. Virus Res. 2014, 180, 76–83. [Google Scholar] [CrossRef]
- Willi, B.; Spiri, A.M.; Meli, M.L.; Grimm, F.; Beatrice, L.; Riond, B.; Bley, T.; Jordi, R.; Dennler, M.; Hofmann-Lehmann, R. Clinical and molecular investigation of a canine distemper outbreak and vector-borne infections in a group of rescue dogs imported from Hungary to Switzerland. BMC Vet. Res. 2015, 11, 154. [Google Scholar] [CrossRef]
- Pope, J.P.; Miller, D.L.; Riley, M.C.; Anis, E.; Wilkes, R.P. Characterization of a novel canine distemper virus causing disease in wildlife. J. Vet. Diagn. Investig. 2016, 28, 506–513. [Google Scholar] [CrossRef]
- Appel, M.J.; Summers, B.A. Pathogenicity of morbilliviruses for terrestrial carnivores. Vet. Microbiol. 1995, 44, 187–191. [Google Scholar] [CrossRef]
- Cleaveland, S.; Mlengeya, T.; Kaare, M.; Haydon, D.; Lembo, T.; Laurenson, M.K.; Packer, C. The conservation relevance of epidemiological research into carnivore viral diseases in the serengeti. Conserv. Boil. 2007, 21, 612–622. [Google Scholar] [CrossRef]
- Harrison, T.M.; Mazet, J.K.; Holekamp, K.E.; Dubovi, E.; Engh, A.L.; Nelson, K.; Van Horn, R.C.; Munson, L. Antibodies to canine and feline viruses in spotted hyenas (Crocuta crocuta) in the Masai Mara National Reserve. J. Wildl. Dis. 2004, 40, 1–10. [Google Scholar] [CrossRef]
- Munson, L.; Terio, K.A.; Kock, R.; Mlengeya, T.; Roelke, M.E.; Dubovi, E.; Summers, B.; Sinclair, A.R.; Packer, C. Climate extremes promote fatal co-infections during canine distemper epidemics in African lions. PLoS ONE 2008, 3, e2545. [Google Scholar] [CrossRef]
- Dobson, A.; Foufopoulos, J. Emerging infectious pathogens of wildlife. Philos. Trans. R Soc. Lond. B Biol. Sci. 2001, 356, 1001–1012. [Google Scholar] [CrossRef]
- Leendertz, F.H.; Pauli, G.; Maetz-Rensing, K.; Boardman, W.; Nunn, C.; Ellerbrok, H.; Jensen, S.A.; Junglen, S.; Christophe, B. Pathogens as drivers of population declines: The importance of systematic monitoring in great apes and other threatened mammals. Boil. Conserv. 2006, 131, 325–337. [Google Scholar] [CrossRef]
- Gaskin, J.M. Canine distemper virus in domesticated cats and pigs. Adv. Enzym. Relat. Areas Mol. Biol. 1974, 40, 803–806. [Google Scholar]
- Van Heerden, M. An investigation into the health status and diseases of wild dogs (Lycaon pictus) in the Kruger National Park. J. S. Afr. Vet. Assoc. 1995, 66, 18–27. [Google Scholar]
- Maia, O.B.; Gouveia, A.M. Serologic response of maned wolves (Chrysocyon brachyurus) to canine and canine parvovirus vaccination distemper virus. J. Zoo Wildl. Med. 2001, 32, 78–80. [Google Scholar]
- Coke, R.L.; Backues, K.A.; Hoover, J.P.; Saliki, J.T.; Ritchey, J.W.; West, G.D. Serologic responses after vaccination of fennec foxes (Vulpes zerda) and meerkats (Suricata suricatta) with a live, canarypox-vectored canine distemper virus vaccine. J. Zoo Wildl. Med. 2005, 36, 326–330. [Google Scholar] [CrossRef]
- Halbrooks, R.D.; Swango, L.J.; Schnurrenberger, P.R.; Mitchell, F.E.; Hill, E.P. Response of gray foxes to modified live-virus canine distemper vaccines. J. Am. Vet. Med. Assoc. 1981, 179, 1170–1174. [Google Scholar]
- Kazacos, K.R.; Thacker, H.L.; Shivaprasad, H.L.; Burger, P.P. Vaccination-induced distemper in Kinkajous. J. Am. Vet. Med. Assoc. 1981, 179, 1166–1169. [Google Scholar]
- Thomas-Baker, B. Vaccination-induced distemper in maned wolves, vaccination-induced corneal opacity in a maned wolf. Proc. Am. Assoc. Zoo Vet. 1985, 53, 192–197. [Google Scholar]
- Bush, M.; Montali, R.J.; Brownstein, D.; James, A.E., Jr.; Appel, M.J. Vaccine-induced canine distemper in a lesser panda. J. Am. Vet. Med. Assoc. 1976, 169, 959–960. [Google Scholar]
- Carpenter, J.W.; Appel, M.J.; Erickson, R.C.; Novilla, M.N. Fatal vaccine-induced canine distemper virus infection in black-footed ferrets. J. Am. Vet. Med. Assoc. 1976, 169, 961–964. [Google Scholar]
- Martella, V.; Blixenkrone-Moller, M.; Elia, G.; Lucente, M.S.; Cirone, F.; Decaro, N.; Nielsen, L.; Banyai, K.; Carmichael, L.E.; Buonavoglia, C. Lights and shades on an historical vaccine canine distemper virus, the rockborn strain. Vaccine 2011, 29, 1222–1227. [Google Scholar] [CrossRef]
- Anis, E.; Newell, T.K.; Dyer, N.; Wilkes, R.P. Phylogenetic analysis of the wild-type strains of canine distemper virus circulating in the United States. Virol. J. 2018, 15, 118. [Google Scholar] [CrossRef]
- Iwatsuki, K.; Tokiyoshi, S.; Hirayama, N.; Nakamura, K.; Ohashi, K.; Wakasa, C.; Mikami, T.; Kai, C. Antigenic differences in the h proteins of canine distemper viruses. Vet. Microbiol. 2000, 71, 281–286. [Google Scholar] [CrossRef]
- Sadler, R.A.; Ramsay, E.; McAloose, D.; Rush, R.; Wilkes, R.P. Evaluation of two canine distemper virus vaccines in captive tigers (Panthera tigris). J. Zoo Wildl. Med. 2016, 47, 558–563. [Google Scholar] [CrossRef]
- Burrows, R.; Hofer, H.; East, M.L. Demography, extinction and intervention in a small population: The case of the Serengeti wild dogs. Proc. Biol. Sci. 1994, 256, 281–292. [Google Scholar]
- Burrows, R.; Hofer, H.; East, M.L. Population dynamics, intervention and survival in African wild dogs (Lycaon pictus). Proc. Biol. Sci. 1995, 262, 235–245. [Google Scholar]
- McCormick, A. Canine distemper in African cape hunting dogs (Lycaon pictus): Possibly vaccine induced. J. Zoo Anim. Med. 1983, 14, 66–71. [Google Scholar] [CrossRef]
- Pepin, K.M.; Lass, S.; Pulliam, J.R.; Read, A.F.; Lloyd-Smith, J.O. Identifying genetic markers of adaptation for surveillance of viral host jumps. Nat. Rev. Microbiol. 2010, 8, 802–813. [Google Scholar] [CrossRef]
- Pulliam, J.R. Viral host jumps: Moving toward a predictive framework. Ecohealth 2008, 5, 80–91. [Google Scholar] [CrossRef]
- Taylor, L.H.; Latham, S.M.; Woolhouse, M.E. Risk factors for human disease emergence. Philos. Trans. R Soc. Lond B Biol. Sci. 2001, 356, 983–989. [Google Scholar] [CrossRef]
- Woolhouse, M.E.; Haydon, D.T.; Antia, R. Emerging pathogens: The epidemiology and evolution of species jumps. Trends Ecol. Evol. 2005, 20, 238–244. [Google Scholar] [CrossRef]
- Domingo, E.; Holland, J.J. Rna virus mutations and fitness for survival. Annu. Rev. Microbiol. 1997, 51, 151–178. [Google Scholar] [CrossRef]
- Domingo, E.; Sheldon, J.; Perales, C. Viral quasispecies evolution. Microbiol. Mol. Biol. Rev. 2012, 76, 159–216. [Google Scholar] [CrossRef]
- Janzen, D.H. When is it coevolution. Evolution 1980, 34, 611–612. [Google Scholar] [CrossRef]
- Nikolin, V.M.; Osterrieder, K.; von Messling, V.; Hofer, H.; Anderson, D.; Dubovi, E.; Brunner, E.; East, M.L. Antagonistic pleiotropy and fitness trade-offs reveal specialist and generalist traits in strains of canine distemper virus. PLoS ONE 2012, 7, e50955. [Google Scholar] [CrossRef]
- Nikolin, V.M.; Wibbelt, G.; Michler, F.U.; Wolf, P.; East, M.L. Susceptibility of carnivore hosts to strains of canine distemper virus from distinct genetic lineages. Vet. Microbiol. 2012, 156, 45–53. [Google Scholar] [CrossRef]
- Lauring, A.S.; Frydman, J.; Andino, R. The role of mutational robustness in RNA virus evolution. Nat. Rev. Microbiol. 2013, 11, 327–336. [Google Scholar] [CrossRef]
- Holmes, E.C. The evolutionary genetics of emerging viruses. Annu. Rev. Ecol. Evol. Syst. 2009, 40, 353–372. [Google Scholar] [CrossRef]
- Elena, S.F.; Agudelo-Romero, P.; Lalic, J. The evolution of viruses in multi-host fitness landscapes. Open Virol. J. 2009, 3, 1–6. [Google Scholar] [CrossRef]
- Kassen, R. The experimental evolution of specialists, generalists, and the maintenance of diversity. J. Evol. Boil. 2002, 15, 173–190. [Google Scholar] [CrossRef]
- Elena, S.F.; Lenski, R.E. Evolution experiments with microorganisms: The dynamics and genetic bases of adaptation. Nat. Rev. Genet. 2003, 4, 457–469. [Google Scholar] [CrossRef]
- Da Fontoura Budaszewski, R.; von Messling, V. Morbillivirus experimental animal models: Measles virus pathogenesis insights from canine distemper virus. Viruses 2016, 8, 274. [Google Scholar] [CrossRef]
- Rendon-Marin, S.; da Fontoura Budaszewski, R.; Canal, C.W.; Ruiz-Saenz, J. Tropism and molecular pathogenesis of canine distemper virus. Virol. J. 2019, 16, 30. [Google Scholar] [CrossRef]
- Lin, L.T.; Richardson, C.D. The host cell receptors for measles virus and their interaction with the viral hemagglutinin (h) protein. Viruses 2016, 8, 250. [Google Scholar] [CrossRef]
- Noyce, R.S.; Delpeut, S.; Richardson, C.D. Dog nectin-4 is an epithelial cell receptor for canine distemper virus that facilitates virus entry and syncytia formation. Virology 2013, 436, 210–220. [Google Scholar] [CrossRef] [PubMed]
- Tatsuo, H.; Ono, N.; Yanagi, Y. Morbilliviruses use signaling lymphocyte activation molecules (CD150) as cellular receptors. J. Virol. 2001, 75, 5842–5850. [Google Scholar] [CrossRef] [PubMed]
- Ohishi, K.; Suzuki, R.; Maeda, T.; Tsuda, M.; Abe, E.; Yoshida, T.; Endo, Y.; Okamura, M.; Nagamine, T.; Yamamoto, H.; et al. Recent host range expansion of canine distemper virus and variation in its receptor, the signaling lymphocyte activation molecule, in carnivores. J. Wildl. Dis. 2014, 50, 596–606. [Google Scholar] [CrossRef] [PubMed]
- Bieringer, M.; Han, J.W.; Kendl, S.; Khosravi, M.; Plattet, P.; Schneider-Schaulies, J. Experimental adaptation of wild-type canine distemper virus (CDV) to the human entry receptor CD150. PLoS ONE 2013, 8, e57488. [Google Scholar] [CrossRef] [PubMed]
- Delpeut, S.; Noyce, R.S.; Richardson, C.D. The v domain of dog pvrl4 (nectin-4) mediates canine distemper virus entry and virus cell-to-cell spread. Virology 2014, 454–455, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Noyce, R.S.; Richardson, C.D. Nectin 4 is the epithelial cell receptor for measles virus. Trends Microbiol. 2012, 20, 429–439. [Google Scholar] [CrossRef] [PubMed]
- Pratakpiriya, W.; Ping Teh, A.P.; Radtanakatikanon, A.; Pirarat, N.; Thi Lan, N.; Takeda, M.; Techangamsuwan, S.; Yamaguchi, R. Expression of canine distemper virus receptor nectin-4 in the central nervous system of dogs. Sci. Rep. 2017, 7, 349. [Google Scholar] [CrossRef] [PubMed]
- Di Guardo, G.; Giacominelli-Stuffler, R.; Mazzariol, S. Commentary: Slam-and nectin-4-independent noncytolytic spread of canine distemper virus in astrocytes. Front. Microbiol. 2016, 7, 2011. [Google Scholar] [CrossRef] [PubMed]
- Sakai, K.; Nagata, N.; Ami, Y.; Seki, F.; Suzaki, Y.; Iwata-Yoshikawa, N.; Suzuki, T.; Fukushi, S.; Mizutani, T.; Yoshikawa, T.; et al. Lethal canine distemper virus outbreak in cynomolgus monkeys in Japan in 2008. J. Virol. 2013, 87, 1105–1114. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Li, A.; Ye, H.; Shi, Y.; Hu, Z.; Zeng, L. Natural infection with canine distemper virus in hand-feeding rhesus monkeys in China. Vet. Microbiol. 2010, 141, 374–378. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, Y.; Ochikubo, F.; Matsubara, Y.; Tsuruoka, H.; Ishii, M.; Shirota, K.; Nomura, Y.; Sugiyama, M.; Yamanouchi, K. Natural infection with canine distemper virus in a japanese monkey (Macaca fuscata). Vet. Microbiol. 1989, 20, 193–205. [Google Scholar] [CrossRef]
- Sakai, K.; Yoshikawa, T.; Seki, F.; Fukushi, S.; Tahara, M.; Nagata, N.; Ami, Y.; Mizutani, T.; Kurane, I.; Yamaguchi, R.; et al. Canine distemper virus associated with a lethal outbreak in monkeys can readily adapt to use human receptors. J. Virol. 2013, 87, 7170–7175. [Google Scholar] [CrossRef] [PubMed]
- Otsuki, N.; Nakatsu, Y.; Kubota, T.; Sekizuka, T.; Seki, F.; Sakai, K.; Kuroda, M.; Yamaguchi, R.; Takeda, M. The v protein of canine distemper virus is required for virus replication in human epithelial cells. PLoS ONE 2013, 8, e82343. [Google Scholar] [CrossRef] [PubMed]
- Otsuki, N.; Sekizuka, T.; Seki, F.; Sakai, K.; Kubota, T.; Nakatsu, Y.; Chen, S.; Fukuhara, H.; Maenaka, K.; Yamaguchi, R. Canine distemper virus with the intact c protein has the potential to replicate in human epithelial cells by using human nectin4 as a receptor. Virology 2013, 435, 485–492. [Google Scholar] [CrossRef] [PubMed]
- Pfeffermann, K.; Dörr, M.; Zirkel, F.; von Messling, V. Morbillivirus pathogenesis and virus–host interactions. Adv. Virus Res. 2018, 100, 75–98. [Google Scholar] [PubMed]
- Rohowsky-Kochan, C.; Dowling, P.C.; Cook, S.D. Canine distemper virus-specific antibodies in multiple sclerosis. Neurology 1995, 45, 1554–1560. [Google Scholar] [CrossRef] [PubMed]
- De Vries, P.; Uytdehaag, F.G.; Osterhaus, A.D. Canine distemper virus (CDV) immune-stimulating complexes (iscoms), but not measles virus iscoms, protect dogs against CDV infection. J. Gen. Virol 1988, 69, 2071–2083. [Google Scholar] [CrossRef] [PubMed]
- Appel, M.J.; Shek, W.R.; Shesberadaran, H.; Norrby, E. Measles virus and inactivated canine distemper virus induce incomplete immunity to canine distemper. Arch. Virol. 1984, 82, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Baker, J.A.; Sheffy, B.E.; Robson, D.S.; Gilmartin, J. Response to measles virus by puppies with maternally transferred distemper antibodies. Cornell Vet. 1966, 56, 588–594. [Google Scholar]
- Brown, A.L.; Vitamvas, J.A.; Merry, D.L., Jr.; Beckenhauer, W.H. Immune response of pups to modified live-virus canine distemper-measles vaccine. Am. J. Vet. Res. 1972, 33, 1447–1456. [Google Scholar]
- Sheshberadaran, H.; Norrby, E.; McCullough, K.C.; Carpenter, W.C.; Örvell, C. The antigenic relationship between measles, canine distemper and rinderpest viruses studied with monoclonal antibodies. J. Gen. Virol. 1986, 67, 1381–1392. [Google Scholar] [CrossRef] [PubMed]
- Rouxel, R.N.; Svitek, N.; von Messling, V. A chimeric measles virus with canine distemper envelope protects ferrets from lethal distemper challenge. Vaccine 2009, 27, 4961–4966. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wallace, O.L.; Domi, A.; Wright, K.J.; Driscoll, J.; Anzala, O.; Sanders, E.J.; Kamali, A.; Karita, E.; Allen, S. Canine distemper virus neutralization activity is low in human serum and it is sensitive to an amino acid substitution in the hemagglutinin protein. Virology 2015, 482, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Fischer, C.D.B.; Graf, T.; Ikuta, N.; Lehmann, F.K.M.; Passos, D.T.; Makiejczuk, A.; Silveira, M.A.T., Jr.; Fonseca, A.S.K.; Canal, C.W.; Lunge, V.R. Phylogenetic analysis of canine distemper virus in south america clade 1 reveals unique molecular signatures of the local epidemic. Infect. Genet. Evol. 2016, 41, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Sattler, U.; Khosravi, M.; Avila, M.; Pilo, P.; Langedijk, J.P.; Ader-Ebert, N.; Alves, L.A.; Plattet, P.; Origgi, F.C. Identification of amino acid substitutions with compensational effects in the attachment protein of canine distemper virus. J. Virol. 2014, 88, 8057–8064. [Google Scholar] [CrossRef] [PubMed]
- Blixenkrone-Møller, M.; Svansson, V.; Appel, M.; Krogsrud, J.; Have, P.; Örvell, C. Antigenic relationships between field isolates of morbilliviruses from different carnivores. Arch. Virol. 1992, 123, 279–294. [Google Scholar] [CrossRef] [PubMed]
- Espinal, M.A.; Diaz, F.J.; Ruiz-Saenz, J. Phylogenetic evidence of a new canine distemper virus lineage among domestic dogs in colombia, south america. Vet. Microbiol. 2014, 172, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Haas, L.; Martens, W.; Greiser-Wilke, I.; Mamaev, L.; Butina, T.; Maack, D.; Barrett, T. Analysis of the haemagglutinin gene of current wild-type canine distemper virus isolates from germany. Virus Res. 1997, 48, 165–171. [Google Scholar] [CrossRef]
- Panzera, Y.; Calderon, M.G.; Sarute, N.; Guasco, S.; Cardeillac, A.; Bonilla, B.; Hernandez, M.; Francia, L.; Bedo, G.; La Torre, J.; et al. Evidence of two co-circulating genetic lineages of canine distemper virus in South America. Virus Res. 2012, 163, 401–404. [Google Scholar] [CrossRef]
- Zhao, J.J.; Yan, X.J.; Chai, X.L.; Martella, V.; Luo, G.L.; Zhang, H.L.; Gao, H.; Liu, Y.X.; Bai, X.; Zhang, L.; et al. Phylogenetic analysis of the haemagglutinin gene of canine distemper virus strains detected from breeding foxes, raccoon dogs and minks in China. Vet. Microbiol. 2010, 140, 34–42. [Google Scholar] [CrossRef]
- Radtanakatikanon, A.; Keawcharoen, J.; Charoenvisal, N.T.; Poovorawan, Y.; Prompetchara, E.; Yamaguchi, R.; Techangamsuwan, S. Genotypic lineages and restriction fragment length polymorphism of canine distemper virus isolates in Thailand. Vet. Microbiol. 2013, 166, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Martella, V.; Cirone, F.; Elia, G.; Lorusso, E.; Decaro, N.; Campolo, M.; Desario, C.; Lucente, M.S.; Bellacicco, A.L.; Blixenkrone-Moller, M.; et al. Heterogeneity within the hemagglutinin genes of canine distemper virus (CDV) strains detected in Italy. Vet. Microbiol. 2006, 116, 301–309. [Google Scholar] [CrossRef]
- Liao, P.; Guo, L.; Wen, Y.; Yang, Y.; Cheng, S. Phylogenetic features of hemagglutin gene in canine distemper virus strains from different genetic lineages. Int. J. Clin. Exp. Med. 2015, 8, 6607–6612. [Google Scholar]
- Rentería-Solís, Z.; Förster, C.; Aue, A.; Wittstatt, U.; Wibbelt, G.; König, M. Canine distemper outbreak in raccoons suggests pathogen interspecies transmission amongst alien and native carnivores in urban areas from germany. Vet. Microbiol. 2014, 174, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhang, H.; Bai, X.; Martella, V.; Hu, B.; Sun, Y.; Zhu, C.; Zhang, L.; Liu, H.; Xu, S.; et al. Emergence of canine distemper virus strains with two amino acid substitutions in the haemagglutinin protein, detected from vaccinated carnivores in north-eastern china in 2012–2013. Vet. J. 2014, 200, 191–194. [Google Scholar] [CrossRef]
- Feng, N.; Liu, Y.; Wang, J.; Xu, W.; Li, T.; Wang, T.; Wang, L.; Yu, Y.; Wang, H.; Zhao, Y. Canine distemper virus isolated from a monkey efficiently replicates on vero cells expressing non-human primate slam receptors but not human slam receptor. BMC Vet. Res. 2016, 12, 160. [Google Scholar] [CrossRef]
- Ke, G.M.; Ho, C.H.; Chiang, M.J.; Sanno-Duanda, B.; Chung, C.S.; Lin, M.Y.; Shi, Y.Y.; Yang, M.H.; Tyan, Y.C.; Liao, P.C.; et al. Phylodynamic analysis of the canine distemper virus hemagglutinin gene. BMC Vet. Res. 2015, 11, 164. [Google Scholar] [CrossRef]
- Drummond, A.J.; Suchard, M.A.; Xie, D.; Rambaut, A. Bayesian phylogenetics with beauti and the beast 1.7. Mol. Biol. Evol. 2012, 29, 1969–1973. [Google Scholar] [CrossRef]
- Uhl, E.W.; Kelderhouse, C.; Buikstra, J.; Blick, J.P.; Bolon, B.; Hogan, R.J. New world origin of canine distemper: Interdisciplinary insights. Int. J. Paleopathol. 2019, 24, 266–278. [Google Scholar] [CrossRef]
- Beaty, S.M.; Lee, B. Constraints on the genetic and antigenic variability of measles virus. Viruses 2016, 8, 109. [Google Scholar] [CrossRef]
- Yuan, C.; Liu, W.; Wang, Y.; Hou, J.; Zhang, L.; Wang, G. Homologous recombination is a force in the evolution of canine distemper virus. PLoS ONE 2017, 12, e0175416. [Google Scholar] [CrossRef] [PubMed]
- Maganga, G.D.; Labouba, I.; Ngoubangoye, B.; Nkili-Meyong, A.A.; Obame Ondo, D.; Leroy, E.M.; Berthet, N. Molecular characterization of complete genome of a canine distemper virus associated with fatal infection in dogs in gabon, central africa. Virus Res. 2018, 247, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Li, T.; Liu, Y.; Gao, Y.; Yang, S.; Feng, N.; Sun, H.; Wang, S.; Wang, L.; Bu, Z.; et al. Genetic characterization of an isolate of canine distemper virus from a tibetan mastiff in china. Virus Genes 2014, 49, 45–57. [Google Scholar] [CrossRef] [PubMed]
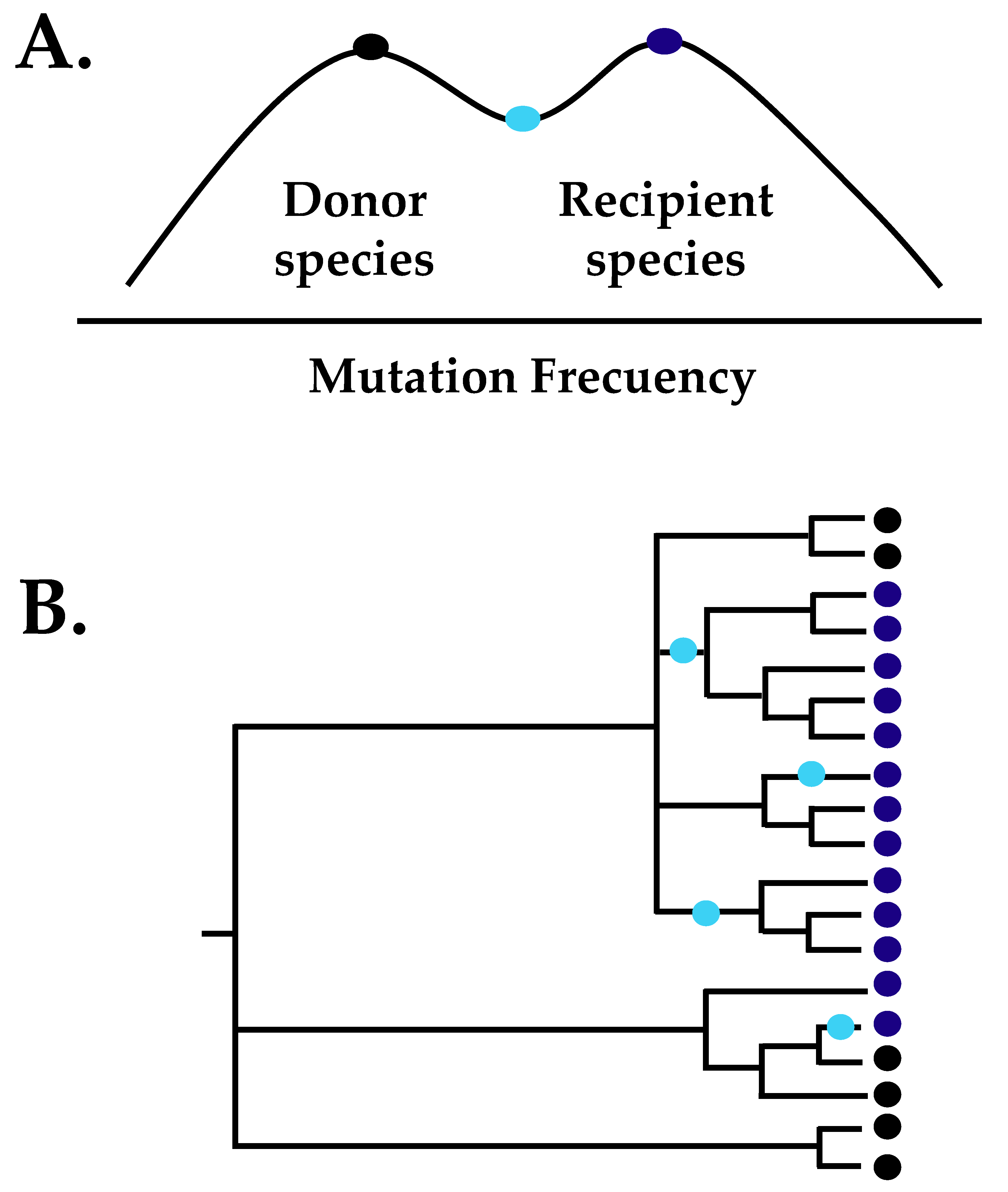
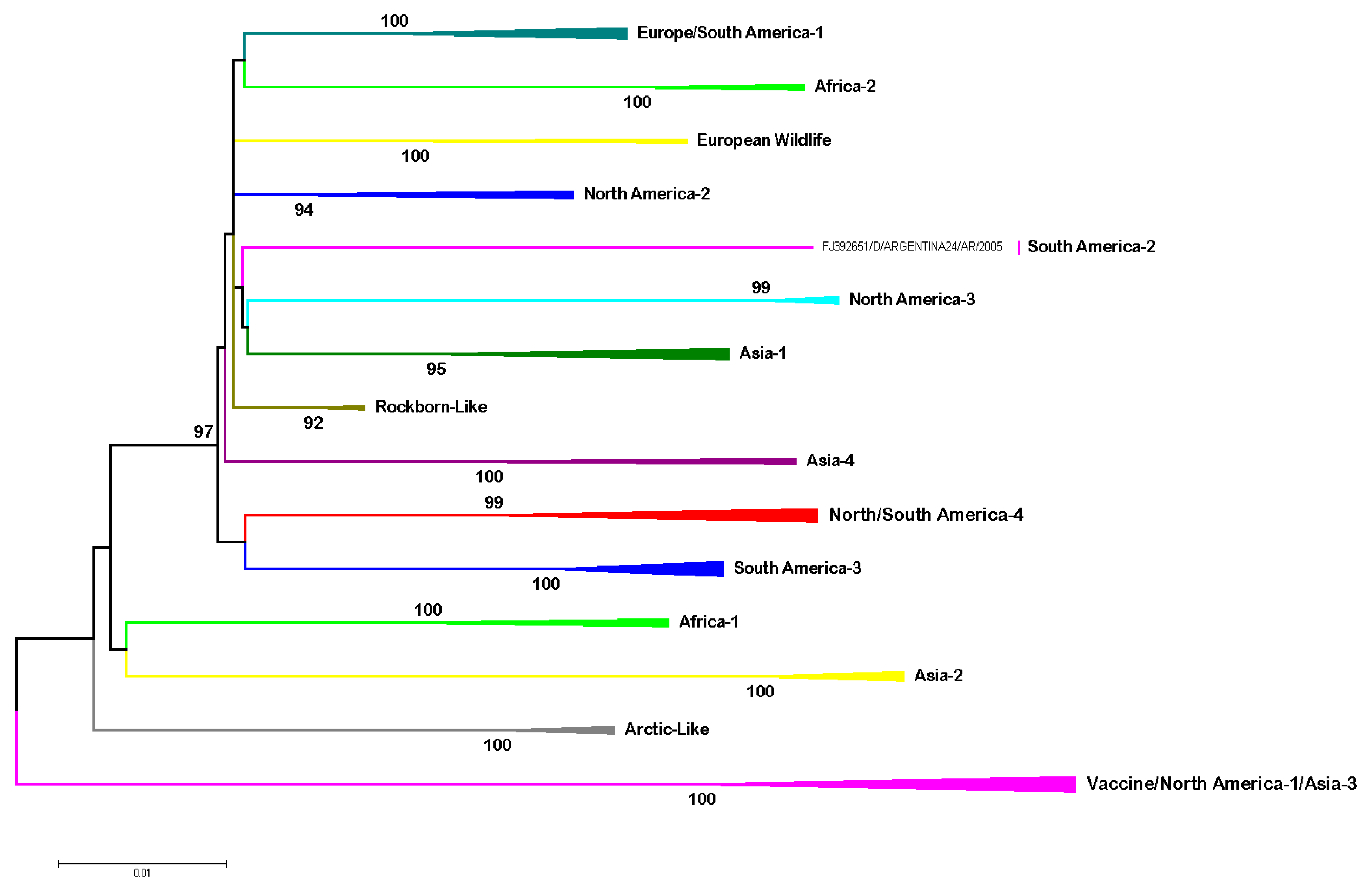
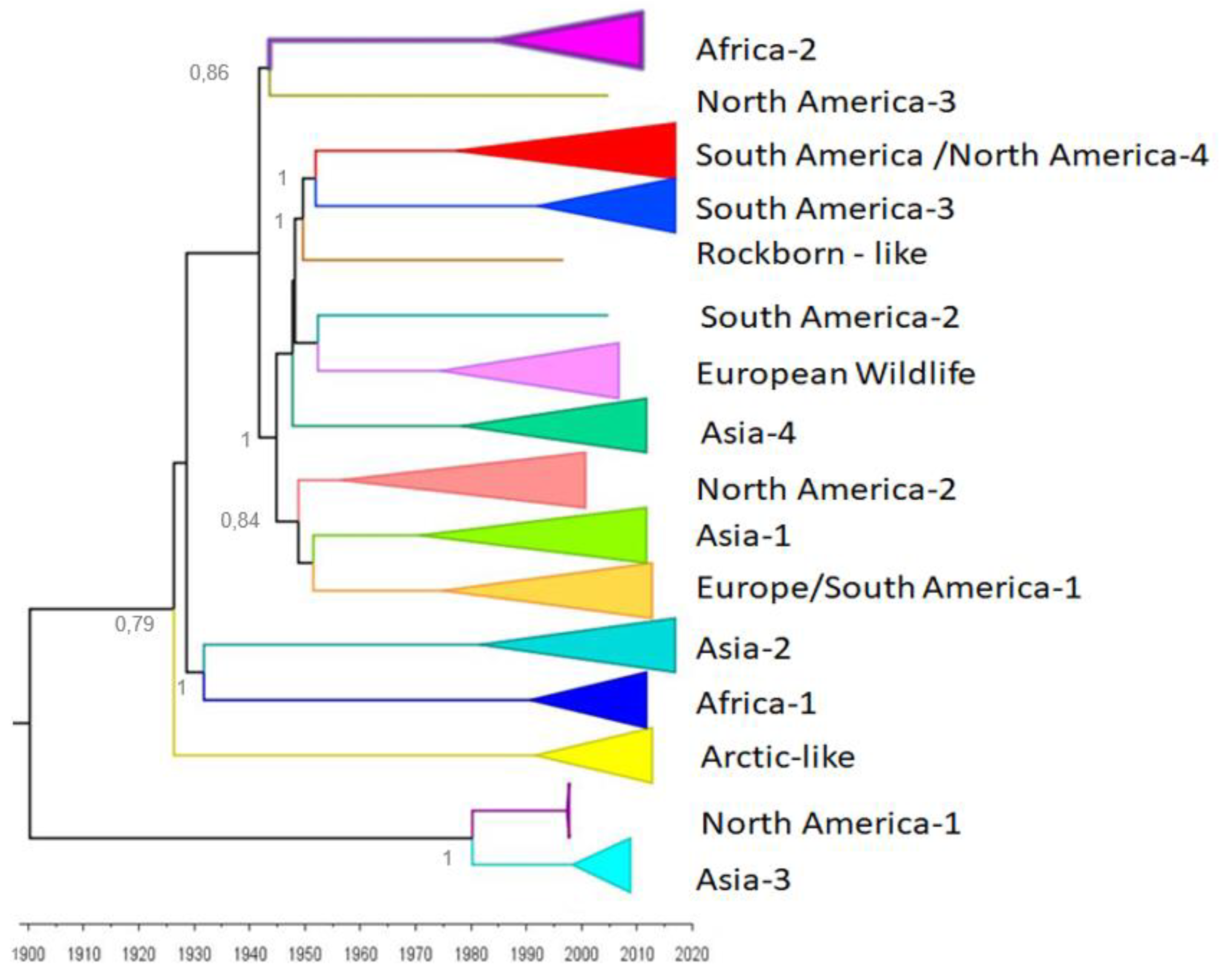
| CDV | Author | |||
|---|---|---|---|---|
| V Gene | F Gene | P Gene | H Gene | |
| Y267C | C116Y | M267V | M548T | Otsuki et al. [115] |
| D540G | Bieringer et al. [106] | |||
| H Gene | |||||
| 530 | 549 | Animal | Lineage | Author | |
| G | Y | Dogs | Europe, Asia 1, America 2, Arctic-like, Asia 2 | Nikolin et al. [94], Liao et al. [135] | |
| G | Y | Wild canids | Europe, Asia 1, | Nikolin et al. [94], Liao et al. [135] | |
| G | Y | Wild species (other than canids) | Europe, Asia 1, | Nikolin et al. [93], Rentería-Solís et al. [136], Liao et al. [135] | |
| G | H | Wild canids | Europe, Asia 1, | Nikolin et al. [93], Panzera et al. [54], Liao et al. [135] | |
| G | H | Wild species (other than canids) | Europe Asia 1, America 2 | Nikolin et al. [93], Panzera et al. [54], Liao et al. [135] | |
| G | H | Dogs | America 2, Europe, South America 1 | Nikolin et al. [93], Liao et al. [135], Fischer et al. [126] | |
| D | Y | Dogs | European Wildlife, South America 2 | Nikolin et al. [93], Liao et al. [135] | |
| D | Y | Wild species (other than canids) | European Wildlife, Africa 2 | Nikolin et al. [33] | |
| D | Y | Rockborn-like, Candur, China, Sweden | Vaccine | Nikolin et al. [93], Liao et al. [135] | |
| N | H | Wild canids | European Wildlife | Nikolin et al. [93] | |
| N | H | Dogs | European Wildlife, America 1 | Liao et al. [135] | |
| N | H | Wild species (other than canids) | America 1 | Rentería-Solís et al. [136], Liao et al. [135] | |
| D | H | Wild species (other than canids) | European Wildlife, Africa 2 | Nikolin et al. [93], Rentería-Solís et al. [136], Liao et al. [135] | |
| C | H | Wild species (other than canids) | European Wildlife | Nikolin et al. [93], | |
| N | Y | Dogs | Arctic-like, Africa 1, America 1 | Nikolin et al. [93], Liao et al. [135] | |
| N | Y | Wild species (other than canids) | America 1 | Nikolin et al. [93], Liao et al. [135] | |
| N | Y | Wild canids | Arctic-like | Liao et al. [135] | |
| E | Y | Dogs | Asia 2 | Nikolin et al. [93], Liao et al. [135] | |
| E | H | Wild species (other than canids) | Asia 2 | Rentería-Solís et al. [136] | |
| R | Y | Wild canids | Asia 2 | Nikolin et al. [93] | |
| R | Y | Dogs | Asia 2 | Liao et al. [135] | |
| R | H | Wild species (other than canids) | America 2 | Nikolin et al. [93], Liao et al. [135] | |
| R | H | Wild species (other than canids) | America 2 | Rentería-Solís et al. [136], | |
| S | H | Vaccines | Nikolin et al. [93], Liao et al. [135] | ||
| S | L | Vaccines | Liao et al. [135] | ||
| S | Y | Dogs | Arctic-like | Liao et al. [135] | |
| A | Y | Dogs | Asia 1 | Liao et al. [135] | |
| V | H | Wild species (other than canids) | European Wildlife | Nikolin et al. [93], Liao et al. [135] | |
| K | Y | Dogs | Asia 2 | Liao et al. [135] | |
| Other Positions Reported in the H Gene | |||||
| 542 | 549 | Animal | Lineage | Author | |
| F | Y | Wild Species (Other Than Canids) | Asia 1 | Zhao et al. [137] | |
| Animal | Lineage | Author | |||
| 530 | 549 | 580 | |||
| G | H | Q | Dogs | South America 1 | Fischer et al. [126] |
| 276 | 392 | 542 | |||
| V | R | F | Wild species (other than canids) | Asia 1 | Feng et al. [138] |
| 530 | 519 | 549 | |||
| N | R | Y | Dogs | South America 3 | Espinal et al. [129] |
| Other Positions Reported in the H and V Genes | |||||
| GENE H | GENE V | Animal | Lineage | Author | |
| 519 | 149 | 134 | |||
| I | H | S | Wild species (other than canids) | Africa 2 | Nikolin et al. [33] |
| R | Y | G | Wild canids | Africa 2 | |
| R | H | G | Wild canids | Africa 2 | |
| R | Y | G | Dogs | Africa 2 | |
| I | H | Wild species (other than canids) | America 2 | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duque-Valencia, J.; Sarute, N.; Olarte-Castillo, X.A.; Ruíz-Sáenz, J. Evolution and Interspecies Transmission of Canine Distemper Virus—An Outlook of the Diverse Evolutionary Landscapes of a Multi-Host Virus. Viruses 2019, 11, 582. https://doi.org/10.3390/v11070582
Duque-Valencia J, Sarute N, Olarte-Castillo XA, Ruíz-Sáenz J. Evolution and Interspecies Transmission of Canine Distemper Virus—An Outlook of the Diverse Evolutionary Landscapes of a Multi-Host Virus. Viruses. 2019; 11(7):582. https://doi.org/10.3390/v11070582
Chicago/Turabian StyleDuque-Valencia, July, Nicolás Sarute, Ximena A. Olarte-Castillo, and Julián Ruíz-Sáenz. 2019. "Evolution and Interspecies Transmission of Canine Distemper Virus—An Outlook of the Diverse Evolutionary Landscapes of a Multi-Host Virus" Viruses 11, no. 7: 582. https://doi.org/10.3390/v11070582
APA StyleDuque-Valencia, J., Sarute, N., Olarte-Castillo, X. A., & Ruíz-Sáenz, J. (2019). Evolution and Interspecies Transmission of Canine Distemper Virus—An Outlook of the Diverse Evolutionary Landscapes of a Multi-Host Virus. Viruses, 11(7), 582. https://doi.org/10.3390/v11070582






