Herpes Virus, Oral Clinical Signs and QoL: Systematic Review of Recent Data
Abstract
1. Introduction
1.1. Background
1.2. Aim
2. Materials and Methods
2.1. Application Protocol and Website Recording Data
2.2. Target Questions
- What is the oral manifestation of a herpes virus infection, and what about new therapies?
- Is there an implication in Quality of Life (QoL) and psychological profile?
2.3. Search Strategy
2.4. Collection Data
2.5. Manuscript Selections
2.6. Research Classifications
2.7. Exclusion and Inclusion Criteria
- Herpes virus infection oral manifestations and therapies;
- QoL and psychological implications of this infection;
- Herpes virus clinical signs and therapy.
- Patients with other specific disease as osteoporosis, immunologic disorders, uncontrolled diabetes mellitus, or other surgical risk related systemic conditions;
- Not enough information regarding the topic;
- Animal or in vitro studies;
- Articles published prior to 1 February 2009;
- No access to the title and abstract.
2.8. Strategy for Collecting Data
2.9. Record of the Extracted and Collected Data Extraction
- Author (Year)—authors and year;
- Sample Size—sample size number;
- Sample Groups—type of groups;
- Posology—therapy posology;
- Bias risk—type of study and induced risk of bias;
- Statistical results—statistical results.
2.10. Risk of Bias Assessment
- Selection bias;
- Performance bias and detection bias;
- Attrition bias;
- Reporting bias;
- Examiner blinding, examiner calibration, standardized follow-up description, standardized residual graft measurement, standardized radiographic assessment.
2.11. Herpes Virus Oral and Systemical Implications
2.11.1. Disease Definition
2.11.2. Disease Clinics
2.11.3. Diagnosis
2.12. Herpes Virus Adopted Therapy
3. Results
4. Discussion
4.1. Literature Review
4.2. Additional Information
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zoccali, R.; Muscatello, M.R.A.; Bruno, A.; Cedro, C.; Campolo, D.; Pandolfo, G.; Meduri, M. The role of defense mechanisms in the modulation of anger experience and expression: Gender differences and influence on self-report measures. Personal. Individ. Differ. 2007, 43, 1426–1436. [Google Scholar] [CrossRef]
- Pantry, S.N.; Medveczky, P.G. Latency, Integration, and Reactivation of Human Herpesvirus-6. Viruses 2017, 9, 194. [Google Scholar] [CrossRef] [PubMed]
- Eisenberg, R.J.; Atanasiu, D.; Cairns, T.M.; Gallagher, J.R.; Krummenacher, C.; Cohen, G.H. Herpes virus fusion and entry: A story with many characters. Viruses 2012, 4, 800–832. [Google Scholar] [PubMed]
- De Silva, S.; Bowers, W.J. Herpes Virus Amplicon Vectors. Viruses 2009, 1, 594–629. [Google Scholar] [CrossRef]
- Dai, X.; Zhou, Z.H. Structure of the herpes simplex virus 1 capsid with associated tegument protein complexes. Science 2018, 360, 7298. [Google Scholar] [CrossRef]
- Schoggins, J.W. IMMUNOLOGY. Viruses carry antiviral cargo. Science 2015, 349, 1166–1167. [Google Scholar] [PubMed]
- Carbone, F.R.; Gebhardt, T. Immunology. A neighborhood watch upholds local immune protection. Science 2014, 346, 40–41. [Google Scholar] [CrossRef]
- Mosca, J.D.; Bednarik, D.P.; Raj, N.B.; Rosen, C.A.; Sodroski, J.G.; Haseltine, W.A.; Pitha, P.M. Herpes simplex virus type-1 can reactivate transcription of latent human immunodeficiency virus. Nature 1987, 325, 67–70. [Google Scholar] [CrossRef]
- Heldwein, E.E. Up close with herpesviruses. Science 2018, 360, 34–35. [Google Scholar] [CrossRef]
- Howard, M.; Brink, N.; Miller, R.; Tedder, R. Association of human herpes virus with pulmonary Kaposi’s sarcoma. Lancet 1995, 346, 712. [Google Scholar] [CrossRef]
- Gagliano, A.; Aricò, I.; Calarese, T.; Condurso, R.; Germanò, E.; Cedro, C.; Spina, E.; Silvestri, R. Restless Leg Syndrome in ADHD children: Levetiracetam as a reasonable therapeutic option. Brain Dev. 2011, 33, 480–486. [Google Scholar] [CrossRef]
- Bruno, A.; Pandolfo, G.; Scimeca, G.; Leonardi, V.; Cedro, C.; Racchiusa, S.; Zoccali, R.A.; Muscatello, M.R.A. Anger in health, benign breast disease and breast cancer: A prospective case-control study. In Vivo 2014, 28, 973–978. [Google Scholar]
- Fiorillo, L.; De Stefano, R.; Cervino, G.; Crimi, S.; Bianchi, A.; Campagna, P.; Herford, A.S.; Laino, L.; Cicciù, M. Oral and Psychological Alterations in Haemophiliac Patients. Biomedicines 2019, 7, 33. [Google Scholar] [CrossRef]
- Cervino, G.; Terranova, A.; Briguglio, F.; De Stefano, R.; Famà, F.; D’Amico, C.; Amoroso, G.; Marino, S.; Gorassini, F.; Mastroieni, R.; et al. Diabetes: Oral health related quality of life and oral alterations. BioMed Res. Int. 2019, 2019, 5907195. [Google Scholar] [CrossRef]
- Savovic, J.; Turner, R.M.; Mawdsley, D.; Jones, H.E.; Beynon, R.; Higgins, J.P.T.; Sterne, J.A.C. Association Between Risk-of-Bias Assessments and Results of Randomized Trials in Cochrane Reviews: The ROBES Meta-Epidemiologic Study. Am. J. Epidemiol. 2018, 187, 1113–1122. [Google Scholar] [CrossRef] [PubMed]
- Mansournia, M.A.; Higgins, J.P.; Sterne, J.A.; Hernan, M.A. Biases in Randomized Trials: A Conversation Between Trialists and Epidemiologists. Epidemiol. (Camb. Mass.) 2017, 28, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A. The Cochrane Collaboration‘s tool for assessing risk of bias in randomised trials. BMJ (Clin. Res.) 2011, 343, d5928. [Google Scholar] [CrossRef]
- Coburn, K.M.; Vevea, J.L. Publication bias as a function of study characteristics. Psychol. Methods 2015, 20, 310–330. [Google Scholar] [CrossRef]
- Laino, L.; Cicciù, M.; Fiorillo, L.; Crimi, S.; Bianchi, A.; Amoroso, G.; Monte, I.P.; Herford, A.S.; Cervino, G. Surgical Risk on Patients with Coagulopathies: Guidelines on Hemophiliac Patients for Oro-Maxillofacial Surgery. Int. J. Environ. Res. Public Health 2019, 16, 1386. [Google Scholar] [CrossRef]
- Cervino, G.; Fiorillo, L.; Laino, L.; Herford, A.S.; Lauritano, F.; Giudice, G.L.; Fama, F.; Santoro, R.; Troiano, G.; Iannello, G.; et al. Oral Health Impact Profile in Celiac Patients: Analysis of Recent Findings in a Literature Review. Gastroenterol. Res. Pract. 2018, 2018, 7848735. [Google Scholar] [CrossRef] [PubMed]
- Sparks Stein, P.; Steffen, M.J.; Smith, C.; Jicha, G.; Ebersole, J.L.; Abner, E.; Dawson, D. Serum antibodies to periodontal pathogens are a risk factor for Alzheimer’s disease. Alzheimer’s Dement. 2012, 8, 196–203. [Google Scholar] [CrossRef]
- Fatahzadeh, M.; Schwartz, R.A. Human herpes simplex virus infections: Epidemiology, pathogenesis, symptomatology, diagnosis, and management. J. Am. Acad. Dermatol. 2007, 57, 737–763. [Google Scholar] [CrossRef] [PubMed]
- Billet, B.; Wynendaele, R.; Vanquathem, N.E. A Novel Minimally Invasive Wireless Technology for Neuromodulation via Percutaneous Intercostal Nerve Stimulation for Post-Herpetic Neuralgia: A Case Report with Short-Term Follow-up. Pain Pract. Off. J. World Inst. Pain 2018, 18, 374–379. [Google Scholar] [CrossRef]
- Cervino, G.; Cicciù, M.; Biondi, A.; Bocchieri, S.; Herford, A.S.; Laino, L.; Fiorillo, L. Antibiotic Prophylaxis on Third Molar Extraction: Systematic Review of Recent Data. Antibiotics 2019, 8, 53. [Google Scholar] [CrossRef]
- Annunziata, G.; Maisto, M.; Schisano, C.; Ciampaglia, R.; Narciso, V.; Tenore, G.C.; Novellino, E. Resveratrol as a Novel Anti-Herpes Simplex Virus Nutraceutical Agent: An Overview. Viruses 2018, 10, 473. [Google Scholar] [CrossRef]
- Polansky, H.; Javaherian, A.; Itzkovitz, E. Clinical Trial of Herbal Treatment Gene-Eden-VIR/Novirin in Oral Herpes. J. Evid. Based Integr. Med. 2018, 23. [Google Scholar] [CrossRef]
- Semprini, A.; Singer, J.; Shortt, N.; Braithwaite, I.; Beasley, R. Protocol for a randomised controlled trial of 90% kanuka honey versus 5% aciclovir for the treatment of herpes simplex labialis in the community setting. BMJ Open 2017, 7, e017766. [Google Scholar] [CrossRef] [PubMed]
- Palli, M.A.; McTavish, H.; Kimball, A.; Horn, T.D. Immunotherapy of Recurrent Herpes Labialis with Squaric Acid. JAMA Dermatol. 2017, 153, 828–829. [Google Scholar] [CrossRef] [PubMed]
- Batavia, A.S.; Secours, R.; Espinosa, P.; Jean Juste, M.A.; Severe, P.; Pape, J.W.; Fitzgerald, D.W. Diagnosis of HIV-Associated Oral Lesions in Relation to Early versus Delayed Antiretroviral Therapy: Results from the CIPRA HT001 Trial. PLoS ONE 2016, 11, e0150656. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Zheng, R.; Jiang, J.; Dickinson, D.; Fu, B.; Chu, T.C.; Lee, L.H.; Pearl, H.; Hsu, S. Topical lipophilic epigallocatechin-3-gallate on herpes labialis: A phase II clinical trial of AverTeaX formula. Oral Surg. Oral Med.Oral Pathol. Oral Radiol. 2015, 120, 717–724. [Google Scholar] [CrossRef]
- You, Y.; Wang, L.; Li, Y.; Wang, Q.; Cao, S.; Tu, Y.; Li, S.; Bai, L.; Lu, J.; Wei, Z.; et al. Multicenter randomized study of inosine pranobex versus acyclovir in the treatment of recurrent herpes labialis and recurrent herpes genitalis in Chinese patients. J. Dermatol. 2015, 42, 596–601. [Google Scholar] [CrossRef]
- Miller, C.S.; Westgate, P.M. Implications of medical screenings of patients arriving for dental treatment: The results of a comprehensive laboratory screening. J. Am. Dent. Assoc. (1939) 2014, 145, 1027–1035. [Google Scholar] [CrossRef]
- Senti, G.; Iannaccone, R.; Graf, N.; Felder, M.; Tay, F.; Kundig, T. A randomized, double-blind, placebo-controlled study to test the efficacy of topical 2-hydroxypropyl-Beta-cyclodextrin in the prophylaxis of recurrent herpes labialis. Dermatol. (Basel Switz.) 2013, 226, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Khemis, A.; Duteil, L.; Coudert, A.C.; Tillet, Y.; Dereure, O.; Ortonne, J.P. Evaluation of the efficacy and safety of a CS20(R) protective barrier gel containing OGT compared with topical aciclovir and placebo on functional and objective symptoms of labial herpes recurrences: A randomized clinical trial. J. Eur. Acad. Dermatol. Venereol. 2012, 26, 1240–1246. [Google Scholar] [CrossRef]
- Skulason, S.; Holbrook, W.P.; Thormar, H.; Gunnarsson, G.B.; Kristmundsdottir, T. A study of the clinical activity of a gel combining monocaprin and doxycycline: A novel treatment for herpes labialis. J. Oral Pathol. Med. Off. Publ. Int. Assoc. Oral Pathol. Am. Acad. Oral Pathol. 2012, 41, 61–67. [Google Scholar] [CrossRef]
- Munoz Sanchez, P.J.; Capote Femenias, J.L.; Diaz Tejeda, A.; Tuner, J. The effect of 670-nm low laser therapy on herpes simplex type 1. Photomed. Laser Surg. 2012, 30, 37–40. [Google Scholar] [CrossRef] [PubMed]
- Busch, R.; Graubaum, H.J.; Gruenwald, J.; Faergemann, J. Therapeutic effect of 1, 5-pentanediol for herpes simplex labialis: A randomized, double-blind, placebo-controlled clinical trial. Adv. Ther. 2009, 26, 719–727. [Google Scholar] [CrossRef] [PubMed]
- Bieber, T.; Chosidow, O.; Bodsworth, N.; Tyring, S.; Hercogova, J.; Bloch, M.; Davis, M.; Lewis, M.; Boutolleau, D.; Attali, P. Efficacy and safety of aciclovir mucoadhesive buccal tablet in immunocompetent patients with labial herpes (LIP Trial): A double-blind, placebo-controlled, self-initiated trial. J. Drugs Dermatol. 2014, 13, 791–798. [Google Scholar]
- Dougal, G.; Lee, S.Y. Evaluation of the efficacy of low-level light therapy using 1072 nm infrared light for the treatment of herpes simplex labialis. Clin. Exp. Dermatol. 2013, 38, 713–718. [Google Scholar] [CrossRef]
- Matarese, G.; Ramaglia, L.; Cicciu, M.; Cordasco, G.; Isola, G. The effects of diode laser therapy as an adjunct to scaling and root planing in the treatment of aggressive periodontitis: A 1-Year Randomized Controlled Clinical Trial. Photomed. Laser Surg. 2017, 35, 702–709. [Google Scholar] [CrossRef] [PubMed]
- Cervino, G.; Fiorillo, L.; Herford, A.S.; Laino, L.; Troiano, G.; Amoroso, G.; Crimi, S.; Matarese, M.; D’Amico, C.; Nastro Siniscalchi, E.; et al. Alginate Materials and Dental Impression Technique: A Current State of the Art and Application to Dental Practice. Mar. Drugs 2018, 17, 18. [Google Scholar] [CrossRef] [PubMed]
- Fama, F.; Cicciu, M.; Sindoni, A.; Nastro-Siniscalchi, E.; Falzea, R.; Cervino, G.; Polito, F.; De Ponte, F.; Gioffre-Florio, M. Maxillofacial and concomitant serious injuries: An eight-year single center experience. Chin. J. Traumatol. 2017, 20, 4–8. [Google Scholar] [CrossRef]
- Rullo, R.; Scalzone, P.; Laino, L.; Russo, A.; Festa, V.M.; Fiorillo, L.; Cicciu, M. Solitary Plasmacytoma of the Mandible: Early Diagnosis and Surgical Management. J. Craniofac. Surg. 2019. [Google Scholar] [CrossRef] [PubMed]
- Oteri, G.; Procopio, R.M.; Cicciù, M. Giant salivary gland calculi (GSGC): Report of two cases. Open Dent. J. 2011, 5, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Poli, P.P.; Beretta, M.; Cicciù, M.; Maiorana, C. Alveolar ridge augmentation with titanium mesh. A retrospective clinical study. Open Dent. J. 2014, 8, 148–158. [Google Scholar] [CrossRef]
- Lombardi, T.; Bernardello, F.; Berton, F.; Porrelli, D.; Rapani, A.; Camurri Piloni, A.; Fiorillo, L.; Di Lenarda, R.; Stacchi, C. Efficacy of Alveolar Ridge Preservation after Maxillary Molar Extraction in Reducing Crestal Bone Resorption and Sinus Pneumatization: A Multicenter Prospective Case-Control Study. BioMed Res. Int. 2018, 2018, 9352130. [Google Scholar] [CrossRef] [PubMed]
- Cervino, G.; Fiorillo, L.; Herford, A.S.; Romeo, U.; Bianchi, A.; Crimi, S.; Amico, C.; De Stefano, R.; Troiano, G.; Santoro, R.; et al. Molecular Biomarkers Related to Oral Carcinoma: Clinical Trial Outcome Evaluation in a Literature Review. Dis. Markers 2019, 2019, 8040361. [Google Scholar] [CrossRef] [PubMed]
- Troiano, G.; Laino, L.; Cicciu, M.; Cervino, G.; Fiorillo, L.; D‘Amico, C.; Zhurakivska, K.; Lo Muzio, L. Comparison of Two Routes of Administration of Dexamethasone to Reduce the Postoperative Sequelae After Third Molar Surgery: A Systematic Review and Meta-Analysis. Open Dent. J. 2018, 12, 181–188. [Google Scholar] [CrossRef]
- Herford, A.S.; Cicciù, M.; Eftimie, L.F.; Miller, M.; Signorino, F.; Famà, F.; Cervino, G.; Lo Giudice, G.; Bramanti, E.; Lauritano, F.; et al. rhBMP-2 applied as support of distraction osteogenesis: A split-mouth histological study over nonhuman primates mandibles. Int. J. Clin. Exp. Med. 2016, 9, 17187–17194. [Google Scholar]
- Bramanti, E.; Matacena, G.; Cecchetti, F.; Arcuri, C.; Cicciù, M. Oral health-related quality of life in partially edentulous patients before and after implant therapy: A 2-year longitudinal study. Oral Implantol. (Rome) 2013, 6, 37–42. [Google Scholar] [CrossRef]
- Lo Giudice, G.; Cutroneo, G.; Centofanti, A.; Artemisia, A.; Bramanti, E.; Militi, A.; Rizzo, G.; Favaloro, A.; Irrera, A.; Lo Giudice, R.; et al. Dentin morphology of root canal surface: A quantitative evaluation based on a scanning electronic microscopy study. BioMed Res. Int. 2015, 2015, 164065. [Google Scholar] [CrossRef] [PubMed]
- Beretta, M.; Cicciù, M.; Bramanti, E.; Maiorana, C. Schneider membrane elevation in presence of sinus septa: Anatomic features and surgical management. Int. J. Dent. 2012, 2012, 261905. [Google Scholar] [CrossRef] [PubMed]
- Muscatello, M.R.A.; Bruno, A.; Carroccio, C.; Cedro, C.; La Torre, D.; Di Rosa, A.E.; Zoccali, R.; Aragona, M.; La Torre, F.; Mattei, A.; et al. Association between burnout and anger in oncology versus ophthalmology health care professionals. Psychol. Rep. 2006, 99, 641–650. [Google Scholar] [CrossRef]
- Cervino, G.; Fiorillo, L.; Spagnuolo, G.; Bramanti, E.; Laino, L.; Lauritano, F.; Cicciu, M. Interface Between MTA and Dental Bonding Agents: Scanning Electron Microscope Evaluation. J. Int. Soc. Prev. Community Dent. 2017, 7, 64–68. [Google Scholar]
- Cicciu, M.; Fiorillo, L.; Herford, A.S.; Crimi, S.; Bianchi, A.; D‘Amico, C.; Laino, L.; Cervino, G. Bioactive Titanium Surfaces: Interactions of Eukaryotic and Prokaryotic Cells of Nano Devices Applied to Dental Practice. Biomedicines 2019, 7, 12. [Google Scholar] [CrossRef] [PubMed]
- Matarese, G.; Ramaglia, L.; Fiorillo, L.; Cervino, G.; Lauritano, F.; Isola, G. Implantology and Periodontal Disease: The Panacea to Problem Solving? Open Dent. J. 2017, 11, 460–465. [Google Scholar] [CrossRef]
- Orion, E.; Wolf, R. Psychologic factors in the development of facial dermatoses. Clin. Dermatol. 2014, 32, 763–766. [Google Scholar] [CrossRef]
| Pathology | Description | Example |
|---|---|---|
| Herpertic gingivostomatitis | Herpetic gingivostomatitis is often the initial presentation that occurs during the first herpes infection. | 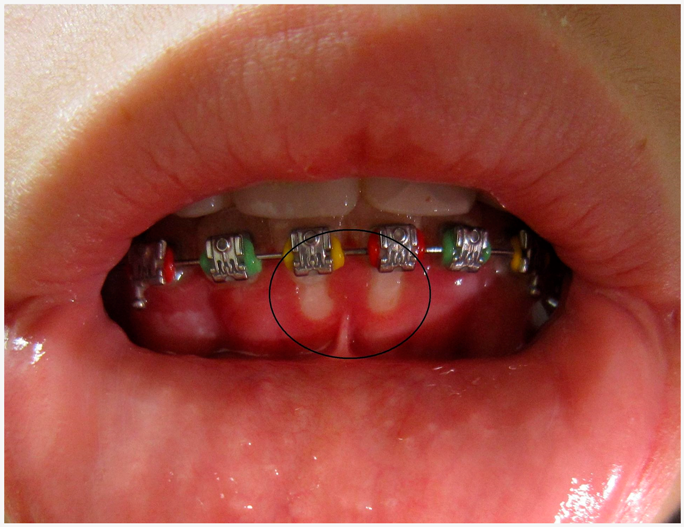 1. Herpetic gingivostomatitis CC BY-SA 3.0 licence, adapted, for concession of James Heilman, MD. |
| Herpes labialis | Infection occurs when the virus comes into contact with the oral mucosa or abraded skin. | 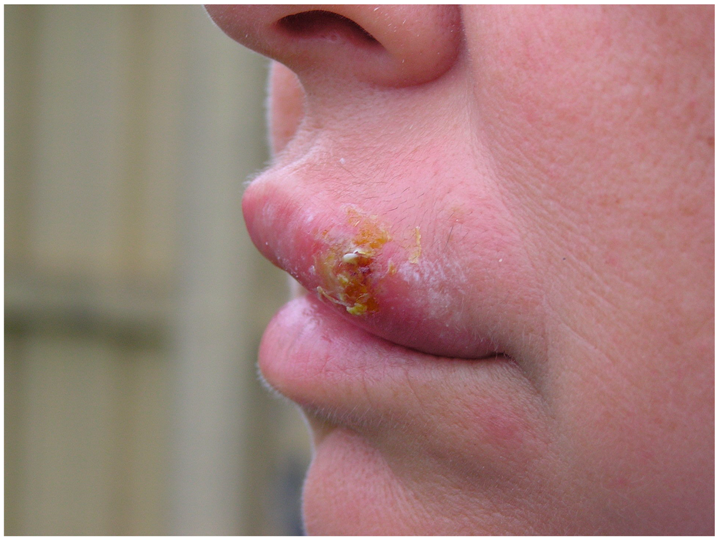 2. Herpes labialis manifestation, public domain photo, adapted for gentle concession of Ben Tillman. |
| Herpes genitalis | When symptomatic, the typical manifestation of HSV-1 or HSV-2 genital infection is characterized by clusters of papules and inflamed vesicles on the outer surface of the genitals that resemble those found in cold sores. | 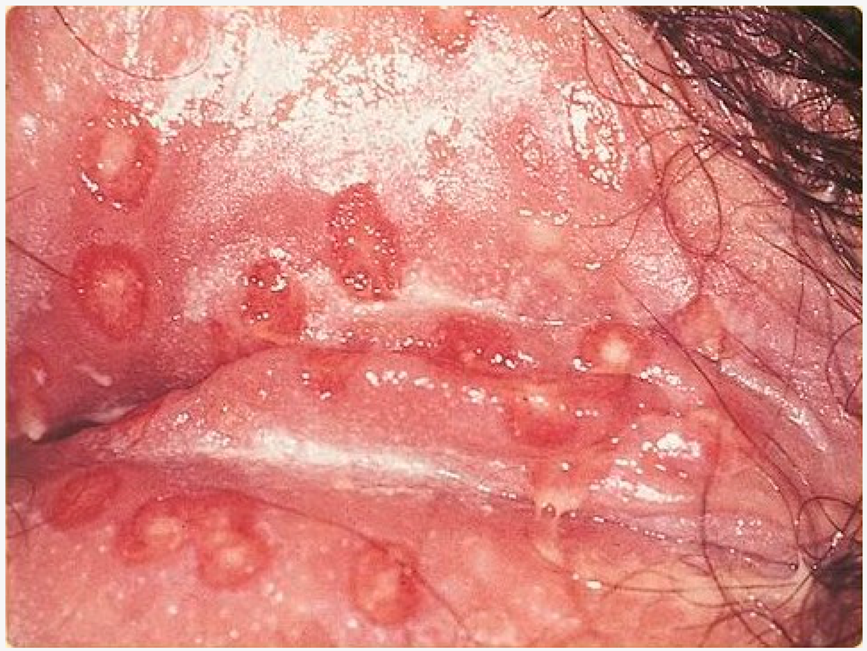 3. Herpes genitalis manifestations, licence CC BY-SA 3.0, adapted for gentle concession of SOA-AIDS Amsterdam. |
| Herpetic paterecleris | The herpetic paterecleris is a painful infection that usually affects the fingers or thumbs. Occasionally, infection occurs on the fingers. People who engage in contact sports such as wrestling, rugby and football sometimes acquire a condition caused by HSV-1 known as gladiatorial herpes that presents itself as ulceration of the skin of the face, ears and neck. Symptoms include fever, headache, sore throat and swollen glands. It affects the eyes or eyelids from time to time. |  4. Herpetic paterecleris, licence CC BY SA 3.0, adapted for gentle concession of James Heilman, MD. |
| Herpetic encephalitis or meningitis | Herpetic brain infection is thought to be due to the transmission of viruses from a peripheral site and the following reactivation of HSV-1, along the axon of the trigeminal nerve, to the brain. HSV is the most common cause of viral encephalitis. When the brain is infected, the virus shows a preference for the temporal lobe. HSV-2 is the most common cause of Mollaret’s meningitis, a type of recurrent viral meningitis. | 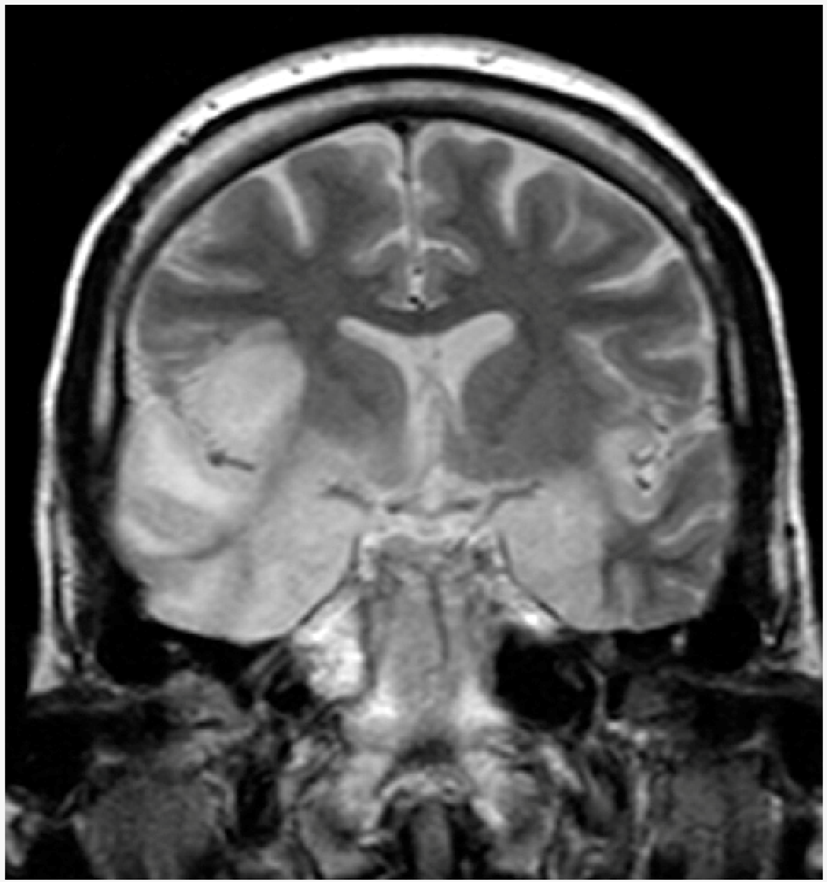 5. Herpes simplex encephalitis, licence CC BY 3.0, adapted with concession of Dr Laughlin Dawes—http://www.radpod.org/2007/03/24/herpes-simplex-encephalitis/. |
| Herpetic esophagytis | Symptoms may include pain when swallowing (odynophagia) and difficulty swallowing (dysphagia). It is often associated with impaired immune function. | 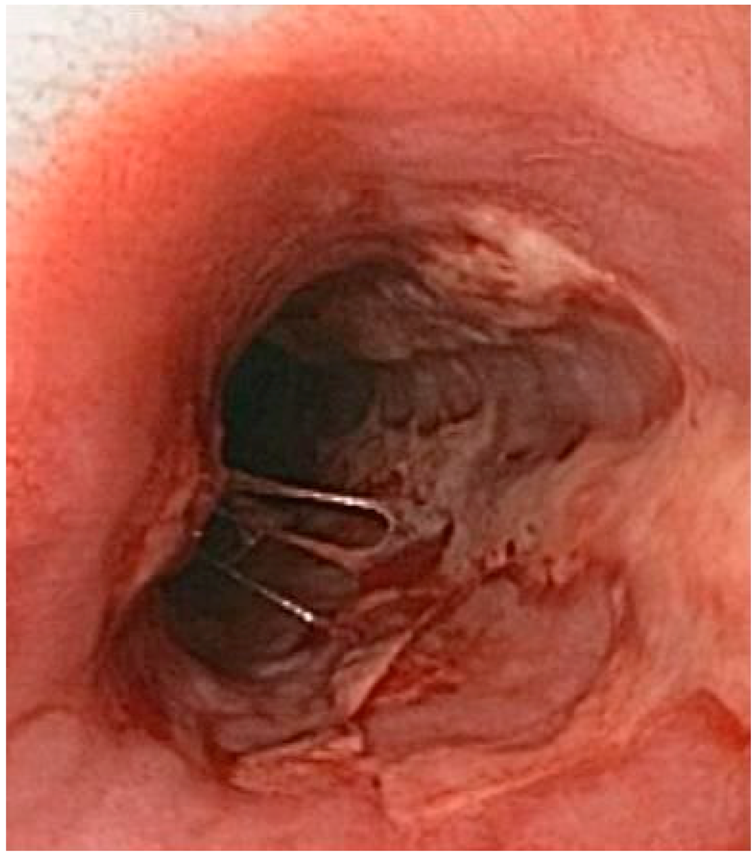 6. Herpetic esophagitis, licence CC BY-SA 3.0, adapted with concession of Donald E. Mansell, MD. |
| Author (Year) | Sample Size | Sample Groups | Posology | Bias Risk | Statistical Results |
|---|---|---|---|---|---|
| Polansky et al. [26] (2018) | 68 |
| 1 to 4 capsules per day over a period of 2 to 36 month | Significant | |
| Semprini et al. [27] (2017) | 950 proposed |
| Kanuka honey with 5% aciclovir cream | Open label, parallel group | Protocol study |
| Palli et al. [28] (2017) | 43 |
| Double blind | p = 0.009 | |
| Batavia et al. [29] (2016) | 816 (HIV infected) |
| Antiretroviral therapy | p < 0.01 | |
| Zhao et al. [30] (2015) | 40 |
| Lipophilic catechins (AverTeaX, Camellix, LLC, Evans, GA, USA) used 6-8 times daily until reduction. | Double blind | p < 0.003 QoL reduction p = 0.016 |
| You et al. [31] (2015) | 144 |
|
| Double blind | Not significative total symptom scores Lower recurrence in Test Group |
| Miller et al. [32] (2014) | 171 |
| p < 0.05 | ||
| Bieber et al. (2014) | 775 |
| Acyclovir tablet 50mg | Double blind | Significative |
| Dougall et al. (2013) | 87 |
| 1072 nm | Double blind | p = 0.01 |
| Senti et al. [33] (2013) | 40 |
| Polyethylene glycol (PEG) formulation containing 20% hydroxypropyl-beta-cyclodextrin (2-HPbetaCD). The gel was applied to the lips twice daily for 6 months. | Double blind | p < 0.003 |
| Khemis et al. [34] (2012) | 106 |
| CS20 (Acura 24 (r)) protective barrier gel | Assessor blinded | p = 0.012 |
| Skulason et al. [35] (2012) | 150 |
|
| Double blind | Healing time p = 0.05 Pain p = 0.0114 |
| Munoz et al. [36] (2012) | 232 |
| 670-nm laser irradiation, 40 mW, 1.6 J, 2.04 J/cm(2), 51 mW/cm | Double blind | No statistical analysis was performed because of large sample size. |
| Busch et al. [37] (2009) | 105 |
| PD gel or placebo gel twice daily to both lips | Double blind | Recurrence time p > 0.05 Pain and clinical efficacy p = 0.001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crimi, S.; Fiorillo, L.; Bianchi, A.; D’Amico, C.; Amoroso, G.; Gorassini, F.; Mastroieni, R.; Marino, S.; Scoglio, C.; Catalano, F.; et al. Herpes Virus, Oral Clinical Signs and QoL: Systematic Review of Recent Data. Viruses 2019, 11, 463. https://doi.org/10.3390/v11050463
Crimi S, Fiorillo L, Bianchi A, D’Amico C, Amoroso G, Gorassini F, Mastroieni R, Marino S, Scoglio C, Catalano F, et al. Herpes Virus, Oral Clinical Signs and QoL: Systematic Review of Recent Data. Viruses. 2019; 11(5):463. https://doi.org/10.3390/v11050463
Chicago/Turabian StyleCrimi, Salvatore, Luca Fiorillo, Alberto Bianchi, Cesare D’Amico, Giulia Amoroso, Francesca Gorassini, Roberta Mastroieni, Stefania Marino, Cristina Scoglio, Francesco Catalano, and et al. 2019. "Herpes Virus, Oral Clinical Signs and QoL: Systematic Review of Recent Data" Viruses 11, no. 5: 463. https://doi.org/10.3390/v11050463
APA StyleCrimi, S., Fiorillo, L., Bianchi, A., D’Amico, C., Amoroso, G., Gorassini, F., Mastroieni, R., Marino, S., Scoglio, C., Catalano, F., Campagna, P., Bocchieri, S., De Stefano, R., Fiorillo, M. T., & Cicciù, M. (2019). Herpes Virus, Oral Clinical Signs and QoL: Systematic Review of Recent Data. Viruses, 11(5), 463. https://doi.org/10.3390/v11050463










