Sediments from Arctic Tide-Water Glaciers Remove Coastal Marine Viruses and Delay Host Infection
Abstract
1. Introduction
2. Materials and Methods
2.1. Viruses for the Experiments
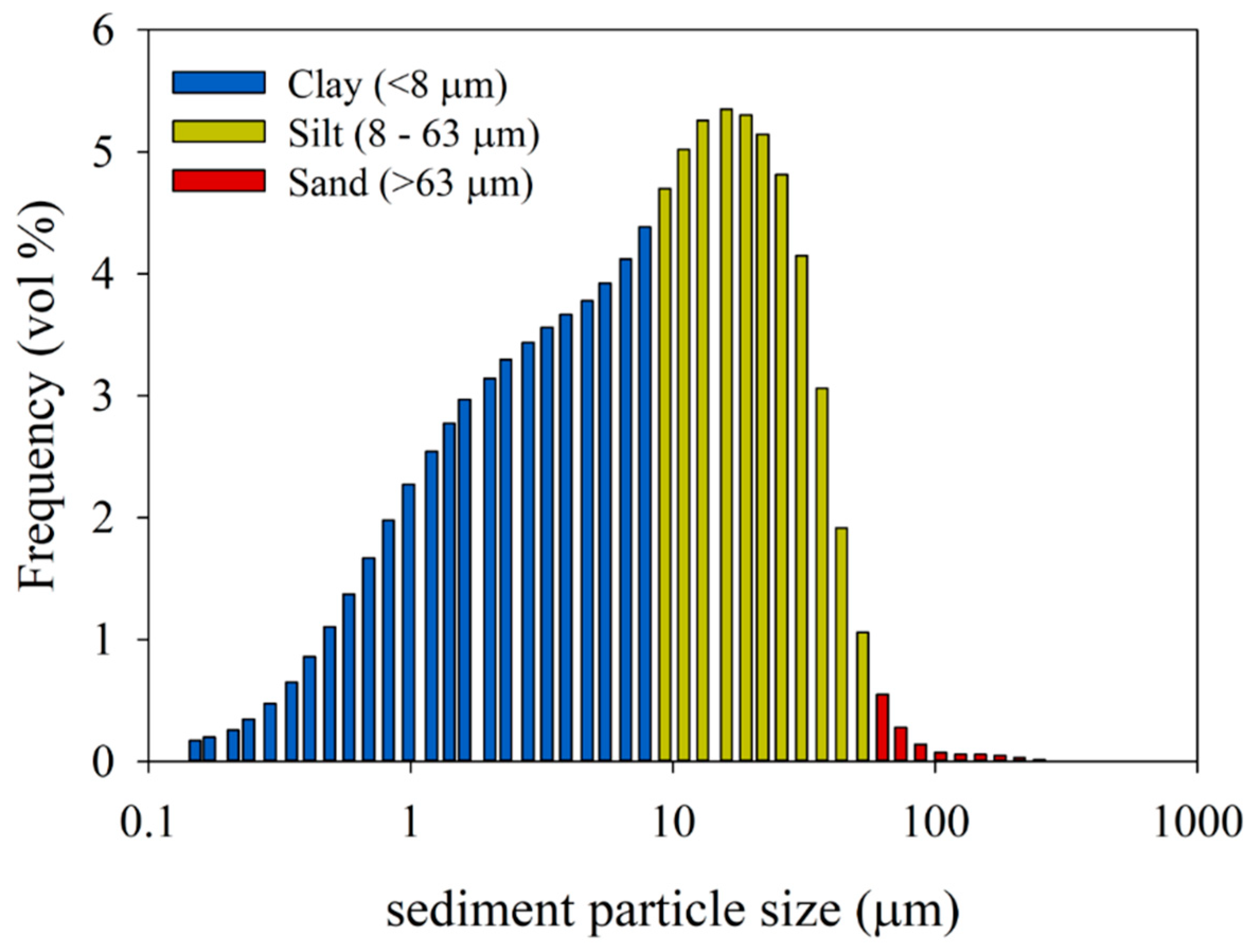
2.2. Sediment Collection for the Experiments
2.3. Adsorption-Settling Experiments
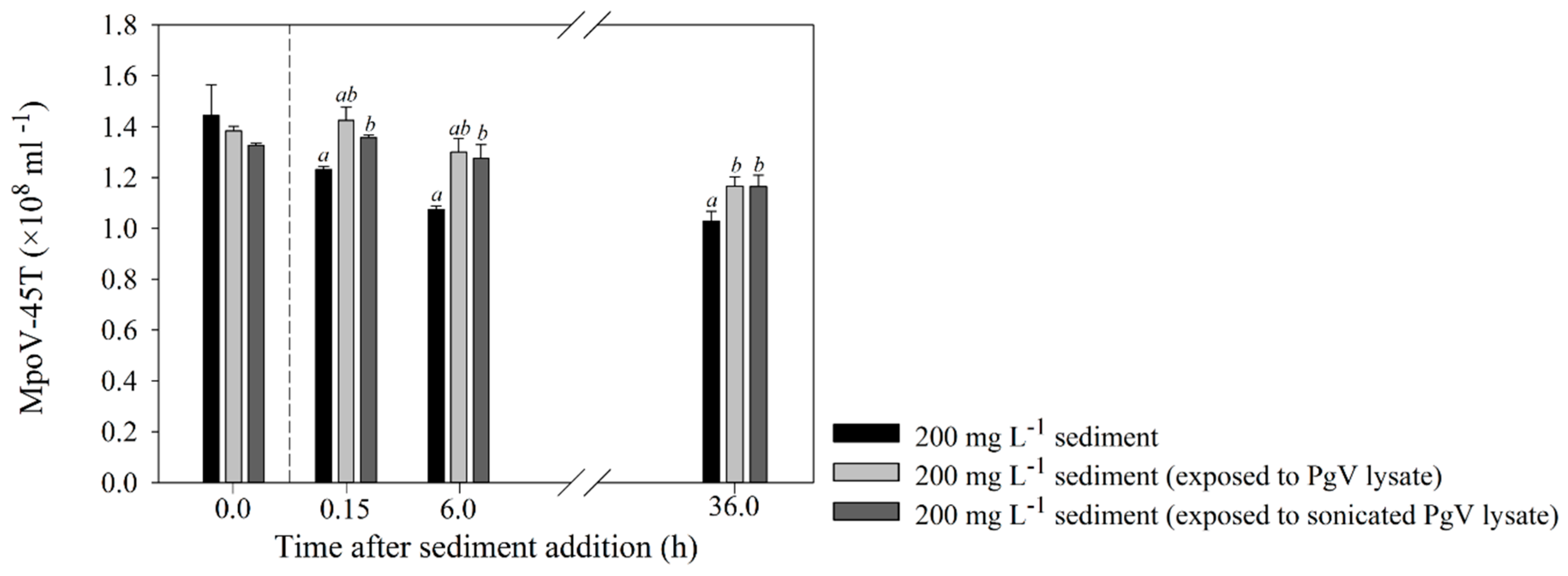
2.4. Effects of Detritus and Other Viruses on the Interaction between Sediment and MpoV-45T
2.5. Determination of Virus Removal Rates
2.6. Virus-Host Infection Experiments
2.7. Sediment as a Transport Vector for Viruses
2.8. Flow Cytometric Quantification of Viral and Phytoplankton Abundances
3. Results
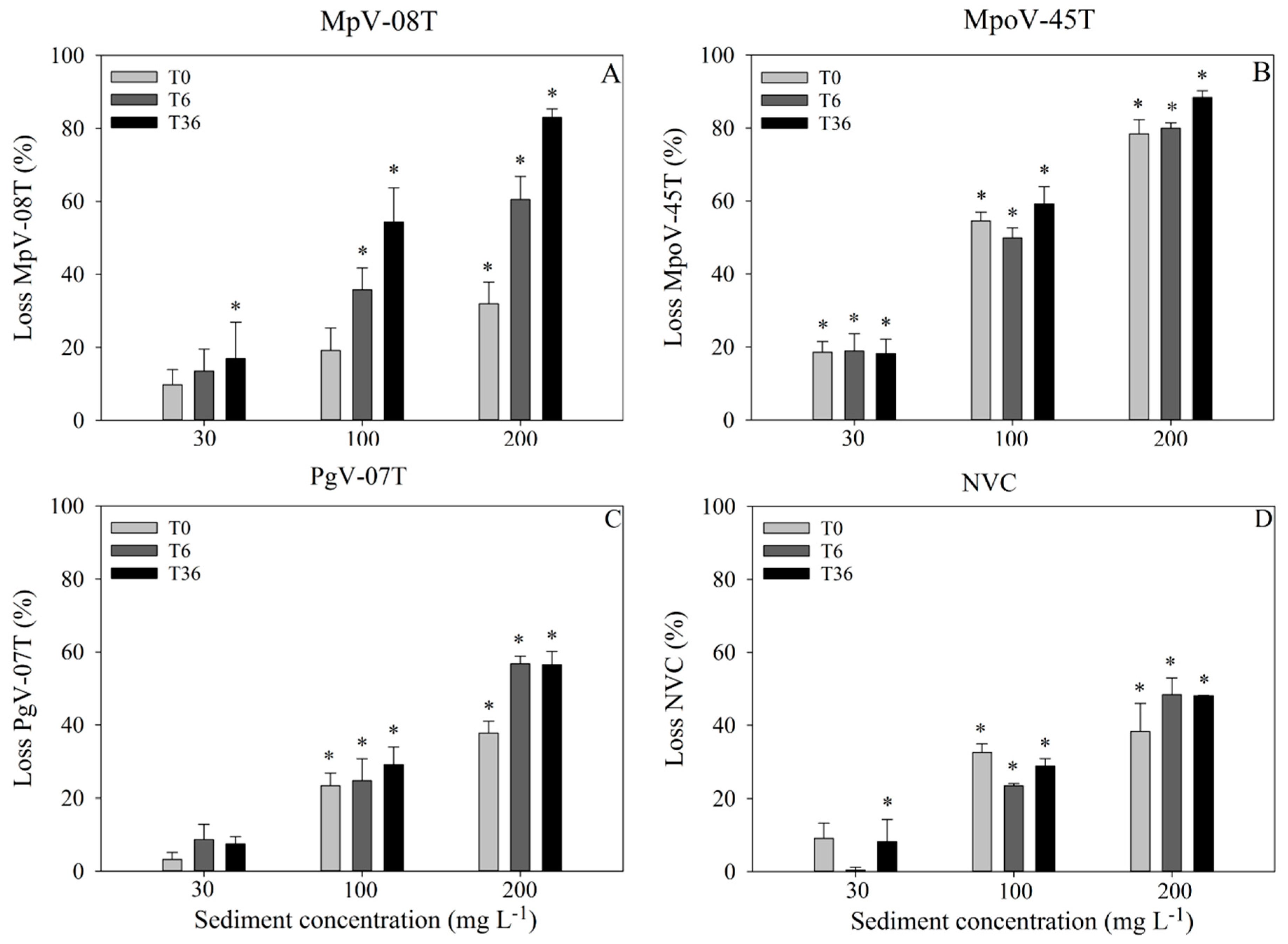
3.1. Adsorption-Settling Experiments
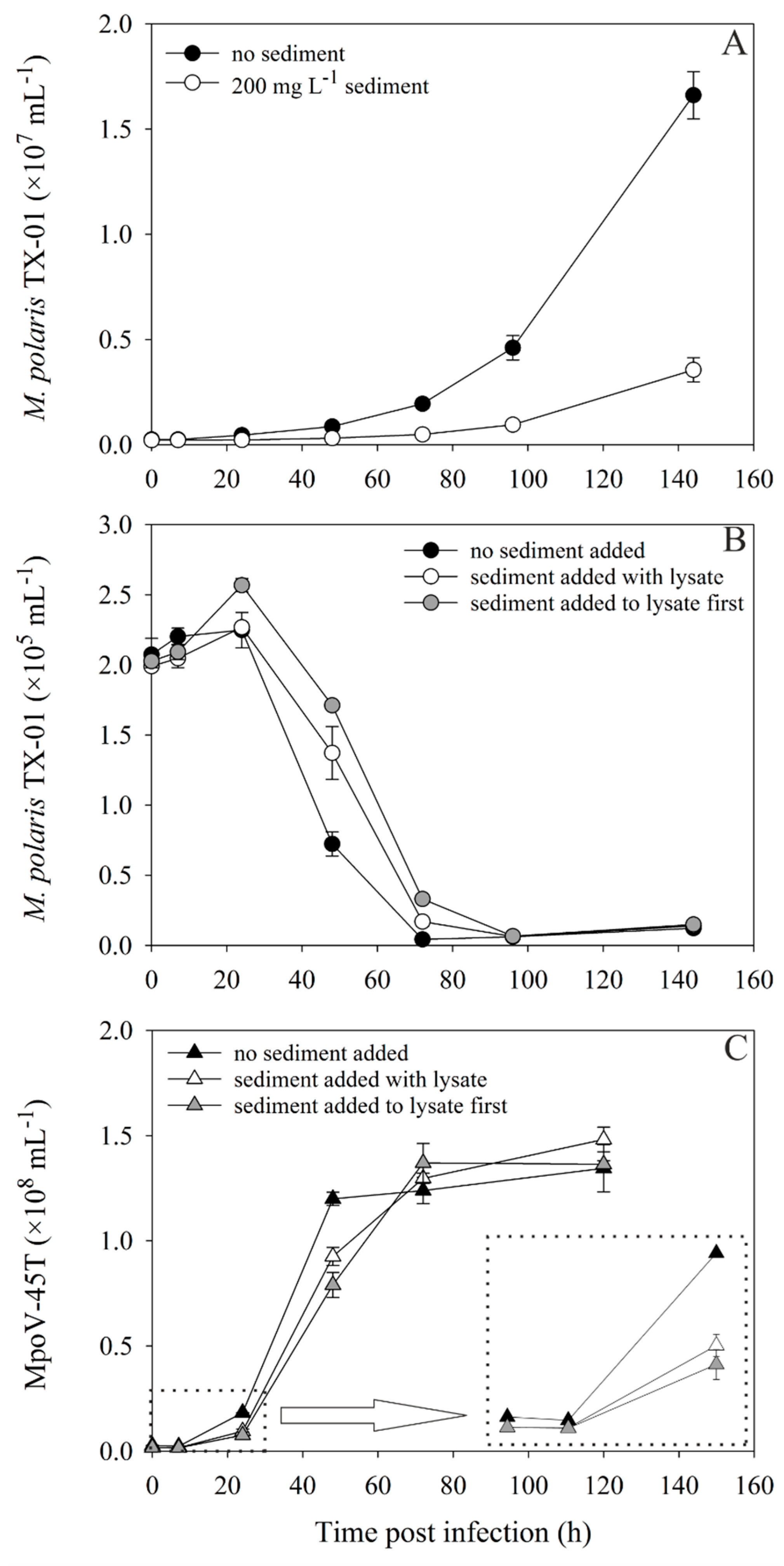
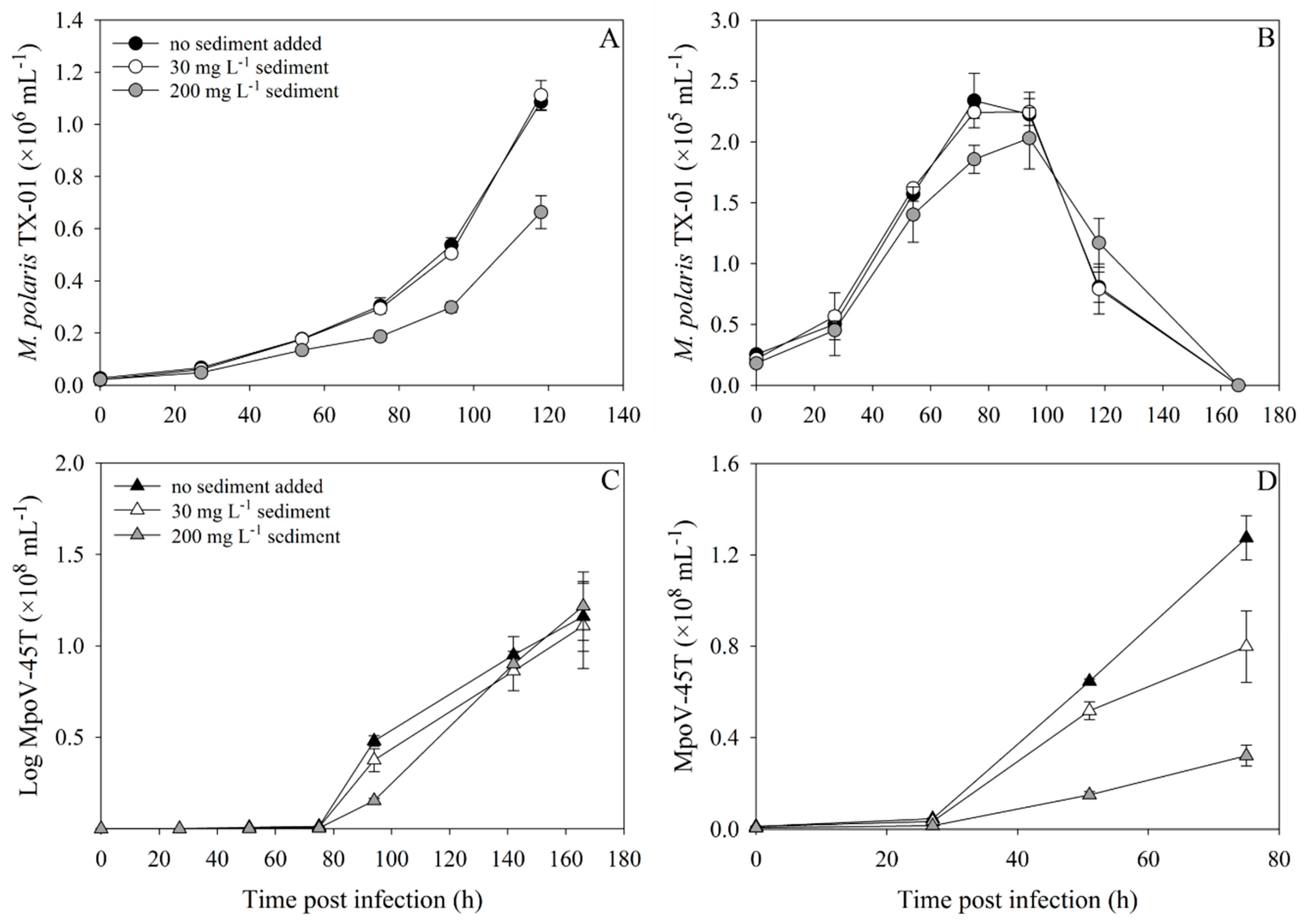
3.2. Virus–Host Interaction Experiments
3.3. Infection of a Host after Desorption from Sediment
4. Discussion
4.1. Adsorption-Settling Experiments
4.2. Effects of Organic Matter
4.3. Virus–Host Interaction
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sommaruga, R. When glaciers and ice sheets melt: Consequences for planktonic organisms. J. Plankton Res. 2015, 37, 509–518. [Google Scholar] [CrossRef] [PubMed]
- Svendsen, H.; Beszczynska-Møller, A.; Hagen, J.O.; Lefauconnier, B.; Tverberg, V.; Gerland, S.; Børre Ørbæk, J.; Bischof, K.; Papucci, C.; Zajaczkowski, M.; et al. The physical environment of Kongsfjorden–Krossfjorden, an Arctic fjord system in Svalbard. Polar Res. 2002, 21, 133–166. [Google Scholar] [CrossRef]
- Arendt, K.E.; Dutz, J.; Jónasdóttir, S.H.; Jung-Madsen, S.; Mortensen, J.; Møller, E.F.; Nielsen, T.G. Effects of suspended sediments on copepods feeding in a glacial influenced sub-Arctic fjord. J. Plankton Res. 2011, 33, 1526–1537. [Google Scholar] [CrossRef]
- Hill, P.R.; Nadeau, O.C. Storm-dominated sedimentation on the inner shelf of the Canadian Beaufort Sea. J. Sediment. Res. 1989, 59, 455–468. [Google Scholar] [CrossRef]
- Luckman, A.; Benn, D.I.; Cottier, F.; Bevan, S.; Nilsen, F.; Inall, M. Calving rates at tidewater glaciers vary strongly with ocean temperature. Nat. Commun. 2015, 6, 8566. [Google Scholar] [CrossRef] [PubMed]
- Murphy, E.J.; Cavanagh, R.D.; Drinkwater, K.F.; Grant, S.M.; Heymans, J.J.; Hofmann, E.E.; Hunt, G.L.; Johnston, N.M. Understanding the structure and functioning of polar pelagic ecosystems to predict the impacts of change. Proc. R. Soc. B 2016, 283, 1646. [Google Scholar] [CrossRef] [PubMed]
- ACIA Chapter 9: Marine Systems. In Arctic Climate Impact Assessment; Cambridge University Press: Cambridge, UK, 2005; pp. 453–538.
- Baudoux, A.C.; Noordeloos, A.A.M.; Veldhuis, M.J.W.; Brussaard, C.P.D. Virally induced mortality of Phaeocystis globosa during two spring blooms in temperate coastal waters. Aquat. Microb. Ecol. 2006, 44, 207–217. [Google Scholar] [CrossRef]
- Evans, C.; Pearce, I.; Brussaard, C.P.D. Viral-mediated lysis of microbes and carbon release in the sub-Antarctic and Polar Frontal zones of the Australian Southern Ocean. Environ. Microbiol. 2009, 11, 2924–2934. [Google Scholar] [CrossRef]
- Gainer, P.J.; Pound, H.L.; Larkin, A.A.; LeCleir, G.R.; DeBruyn, J.M.; Zinser, E.R.; Johnson, Z.I.; Wilhelm, S.W. Contrasting seasonal drivers of virus abundance and production in the North Pacific Ocean. PLoS ONE 2017, 12, e0184371. [Google Scholar] [CrossRef]
- Mojica, K.D.A.; Huisman, J.; Wilhelm, S.W.; Brussaard, C.P.D. Latitudinal variation in virus-induced mortality of phytoplankton across the North Atlantic Ocean. ISME J. 2016, 10, 500–513. [Google Scholar] [CrossRef]
- De Corte, D.; Sintes, E.; Yokokawa, T.; Herndl, G.J. Changes in viral and bacterial communities during the ice-melting season in the coastal Arctic (Kongsfjorden, Ny-Ålesund). Environ. Microbiol. 2011, 13, 1827–1841. [Google Scholar] [CrossRef] [PubMed]
- Lara, E.; Arrieta, J.M.; Garcia-Zarandona, I.; Boras, J.A.; Duarte, C.M.; Agustí, S.; Wassmann, P.F.; Vaqué, D. Experimental evaluation of the warming effect on viral, bacterial and protistan communities in two contrasting Arctic systems. Aquat. Microb. Ecol. 2013, 70, 17–32. [Google Scholar] [CrossRef]
- Maat, D.S.; Biggs, T.; Evans, C.; van Bleijswijk, J.D.L.; van Der Wel, N.N.; Dutilh, B.E.; Brussaard, C.P.D. Characterization and temperature dependence of Arctic Micromonas polaris viruses. Viruses 2017, 9, 134. [Google Scholar] [CrossRef] [PubMed]
- Payet, J.P.; Suttle, C.A. Viral infection of bacteria and phytoplankton in the Arctic Ocean as viewed through the lens of fingerprint analysis. Aquat. Microb. Ecol. 2014, 72, 47–61. [Google Scholar] [CrossRef]
- Brussaard, C.P.D.; Noordeloos, A.A.M.; Witte, H.; Collenteur, M.C.J.; Schulz, K.; Ludwig, A.; Riebesell, U. Arctic microbial community dynamics influenced by elevated CO2 levels. Biogeosciences 2013, 10, 719–731. [Google Scholar] [CrossRef]
- Sheik, A.R.; Brussaard, C.P.; Lavik, G.; Lam, P.; Musat, N.; Krupke, A.; Littmann, S.; Strous, M.; Kuypers, M.M. Responses of the coastal bacterial community to viral infection of the algae Phaeocystis globosa. ISME J. 2014, 8, 212–225. [Google Scholar] [CrossRef]
- Middelboe, M.; Jørgensen, N.O. Viral lysis of bacteria: An important source of dissolved amino acids and cell wall compounds. J. Mar. Biol. Assoc. UK 2006, 86, 605–612. [Google Scholar] [CrossRef]
- Mojica, K.D.A.; Brussaard, C.P.D. Factors affecting virus dynamics and microbial host-virus interactions in marine environments. FEMS Microbiol. Ecol. 2014, 89, 495–515. [Google Scholar] [CrossRef]
- Chattopadhyay, S.; Puls, R.W. Forces dictating colloidal interactions between viruses and soil. Chemosphere 2000, 41, 1279–1286. [Google Scholar] [CrossRef]
- Kapuscinski, R.B.; Mitchell, R. Processes controlling virus inactivation in coastal waters. Water Res. 1980, 14, 363–371. [Google Scholar] [CrossRef]
- Syngouna, V.I.; Chrysikopoulos, C.V. Interaction between viruses and clays in static and dynamic batch systems. Environ. Sci. Technol. 2010, 44, 4539–4544. [Google Scholar] [CrossRef] [PubMed]
- Hewson, I.; Fuhrman, J.A. Viriobenthos production and virioplankton sorptive scavenging by suspended sediment particles in coastal and pelagic waters. Microb. Ecol. 2003, 46, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Katz, A.; Peña, S.; Alimova, A.; Gottlieb, P.; Xu, M.; Block, K.A. Heteroaggregation of an enveloped bacteriophage with colloidal sediments and effect on virus viability. Sci. Total Environ. 2018, 637, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Dowd, S.E.; Pillai, S.D.; Wang, S.; Corapcioglu, M.Y. Delineating the specific influence of virus isoelectric point and size on virus adsorption and transport through sandy soils. Appl. Environ. Microbiol. 1998, 64, 405–410. [Google Scholar] [PubMed]
- LaBelle, R.L.; Gerba, C.P. Influence of estuarine sediment on virus survival under field conditions. Appl. Environ. Microbiol. 1980, 39, 749–755. [Google Scholar] [PubMed]
- Moore, R.S.; Taylor, D.H.; Sturman, L.S.; Reddy, M.M.; Fuhs, G.W. Poliovirus adsorption by 34 minerals and soils. Appl. Environ. Microbiol. 1981, 42, 963–975. [Google Scholar] [PubMed]
- Guillou, N.; Rivier, A.; Chapalain, G.; Gohin, F. The impact of tides and waves on near-surface suspended sediment concentrations in the English Channel. Oceanologia 2017, 59, 28–36. [Google Scholar] [CrossRef]
- Drewes, F.; Peter, H.; Sommaruga, R. Are viruses important in the plankton of highly turbid glacier-fed lakes? Sci. Rep. 2016, 6, 24608. [Google Scholar] [CrossRef]
- Wells, L.E.; Deming, J.W. Effects of temperature, salinity and clay particles on inactivation and decay of cold-active marine Bacteriophage 9A. Aquat. Microb. Ecol. 2006, 45, 31–39. [Google Scholar] [CrossRef]
- Brussaard, C.P.D.; Bratbak, G.; Baudoux, A.C.; Ruardij, P. Phaeocystis and its interaction with viruses. In Phaeocystis, Major Link in the Biogeochemical Cycling of Climate-Relevant Elements; Springer: Dordrecht, The Netherlands, 2007; pp. 201–215. [Google Scholar]
- Cottrell, M.T.; Suttle, C.A. Dynamics of lytic virus infecting the photosynthetic marine picoflagellate Micromonas pusilla. Limnol. Oceanogr. 1995, 40, 730–739. [Google Scholar] [CrossRef]
- Maat, D.S.; Brussaard, C.P.D. Both phosphorus- and nitrogen limitation constrain viral proliferation in marine phytoplankton. Aquat. Microb. Ecol. 2016, 77, 87–97. [Google Scholar] [CrossRef]
- Piedade, G.J.; Wesdorp, E.M.; Montenegro-Borbolla, E.; Maat, D.S.; Brussaard, C.P.D. Influence of irradiance and temperature on the virus MpoV-45T infecting the Arctic picophytoplankter Micromonas polaris. Viruses 2018, 10, 676. [Google Scholar] [CrossRef] [PubMed]
- Martínez Martínez, J.M.; Boere, A.; Gilg, I.; van Lent, J.W.; Witte, H.J.; van Bleijswijk, J.D.; Brussaard, C.P.D. New lipid envelope-containing dsDNA virus isolates infecting Micromonas pusilla reveal a separate phylogenetic group. Aquat. Microb. Ecol. 2015, 74, 17–28. [Google Scholar] [CrossRef]
- Baudoux, A.C.; Brussaard, C.P.D. Characterization of different viruses infecting the marine harmful algal bloom species Phaeocystis globosa. Virology 2005, 341, 80–90. [Google Scholar] [CrossRef] [PubMed]
- Konert, M.; Vandenberghe, J. Comparison of laser grain size analysis with pipette and sieve analysis: A solution for the underestimation of the clay fraction. Sedimentology 1997, 44, 523–535. [Google Scholar] [CrossRef]
- Brussaard, C.P.D. Optimization of procedures for counting viruses by flow cytometry. Appl. Environ. Microbiol. 2004, 70, 1506–1513. [Google Scholar] [CrossRef] [PubMed]
- Marie, D.; Partensky, F.; Vaulot, D.; Brussaard, C. Enumeration of Phytoplankton, Bacteria, and Viruses in Marine Samples. In Current Protocols in Cytometry; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2001; Volume 10, pp. 11.11.1–11.11.15. [Google Scholar]
- Dowdeswell, J.A.; Forsberg, C.F. The size and frequency of icebergs and bergy bits derived from tidewater glaciers in Kongsfjorden, northwest Spitsbergen. Polar Res. 1992, 11, 81–91. [Google Scholar] [CrossRef]
- Murray, A.G.; Jackson, G.A. Viral dynamics: A model of the effects of size, shape, motion and abundance of single-celled planktonic organisms and other particles. Mar. Ecol. Prog. Ser. 1992, 89, 103–116. [Google Scholar] [CrossRef]
- Carlson, G.F., Jr.; Woodard, F.E.; Wentworth, D.F.; Sproul, O.J. Virus inactivation on clay particles in natural waters. J. Water Pollut. Control Fed. 1968, 40, R89–R106. [Google Scholar]
- Stotzky, G.; Schiffenbauer, M.; Lipson, S.M.; Yu, B.H. Surface interactions between viruses and clay minerals and microbes: Mechanisms and implications. Viruses Wastewater Treat. 1981, 199–204. [Google Scholar] [CrossRef]
- Suttle, C.A. Viruses in the sea. Nature 2005, 437, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Schoemann, V.; Becquevort, S.; Stefels, J.; Rousseau, V.; Lancelot, C. Phaeocystis blooms in the global ocean and their controlling mechanisms: A review. J. Sea Res. 2005, 53, 43–66. [Google Scholar] [CrossRef]
- Dam, H.G.; Drapeau, D.T. Coagulation efficiency, organic-matter glues and the dynamics of particles during a phytoplankton bloom in a mesocosm study. Deep Sea Res. Part 2 Top. Stud. Oceanogr. 1995, 42, 111–123. [Google Scholar] [CrossRef]
- Brussaard, C.P.D.; Kuipers, B.; Veldhuis, M.J.W. A mesocosm study of Phaeocystis globosa population dynamics: I. Regulatory role of viruses in bloom control. Harmful Algae 2005, 4, 859–874. [Google Scholar] [CrossRef]
- Davies-Colley, R.J.; Smith, D.G. Turbidity suspended sediment, and water clarity: A review. J. Am. Water Resour. Assoc. 2001, 3, 1085–1101. [Google Scholar] [CrossRef]
- Lind, O.T.; Chrzanowski, T.H.; Dávalos-Lind, L. Clay turbidity and the relative production of bacterioplankton and phytoplankton. Hydrobiologia 1997, 353, 1–18. [Google Scholar] [CrossRef]
- Salter, I.; Böttjer, D.; Christaki, U. The effect of inorganic particle concentration on bacteria–virus–nanoflagellate dynamics. Environ. Microbiol. 2011, 13, 2768–2777. [Google Scholar] [CrossRef]
- Sommaruga, R.; Kandolf, G. Negative consequences of glacial turbidity for the survival of freshwater planktonic heterotrophic flagellates. Sci. Rep. 2014, 4, 4113. [Google Scholar] [CrossRef]
- Wolfe, G.V.; Strom, S.L.; Holmes, J.L.; Radzio, T.; Olson, M.B. Dimethylsulfoniopropionate cleavage by marine phytoplankton in response to mechanical, chemical, or dark stress. J. Phycol. 2002, 38, 948–960. [Google Scholar] [CrossRef]
- Evans, C.; Malin, G.; Wilson, W.H.; Liss, P.S. Infectious titres of Emiliania huxleyi virus 86 are reduced by exposure to millimolar dimethyl sulfide and acrylic acid. Limnol. Oceanogr. 2006, 51, 2468–2471. [Google Scholar] [CrossRef]
- Gadagkar, R.; Gopinathan, K.P. Bacteriophage burst size during multiple infections. J. Biosci. 1980, 2, 253–259. [Google Scholar] [CrossRef]
- Lawrence, J.E.; Chan, A.M.; Suttle, C.A. Viruses causing lysis of the toxic bloom-forming alga Heterosigma akashiwo (Raphidophyceae) are widespread in coastal sediments of British Columbia, Canada. Limnol. Oceanogr. 2002, 47, 545–550. [Google Scholar] [CrossRef]
- Templeton, M.R.; Andrews, R.C.; Hofmann, R. Inactivation of particle-associated viral surrogates by ultraviolet light. Water Res. 2005, 39, 3487–3500. [Google Scholar] [CrossRef] [PubMed]
| Virus Used | Origin | Temp. (°C) | Host | Reference |
|---|---|---|---|---|
| MpV-08T | Temperate | 15 | Micromonas commoda LAC38 1 | [35] |
| MpoV-45T | Arctic | 4 | Micromonas polaris TX-01 | [14] |
| PgV-07T | Temperate | 15 | Phaeocystis globosa G(A) | [36] |
| NVC | Temperate | 15 | Natural community 2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maat, D.S.; Prins, M.A.; Brussaard, C.P.D. Sediments from Arctic Tide-Water Glaciers Remove Coastal Marine Viruses and Delay Host Infection. Viruses 2019, 11, 123. https://doi.org/10.3390/v11020123
Maat DS, Prins MA, Brussaard CPD. Sediments from Arctic Tide-Water Glaciers Remove Coastal Marine Viruses and Delay Host Infection. Viruses. 2019; 11(2):123. https://doi.org/10.3390/v11020123
Chicago/Turabian StyleMaat, Douwe S., Maarten A. Prins, and Corina P. D. Brussaard. 2019. "Sediments from Arctic Tide-Water Glaciers Remove Coastal Marine Viruses and Delay Host Infection" Viruses 11, no. 2: 123. https://doi.org/10.3390/v11020123
APA StyleMaat, D. S., Prins, M. A., & Brussaard, C. P. D. (2019). Sediments from Arctic Tide-Water Glaciers Remove Coastal Marine Viruses and Delay Host Infection. Viruses, 11(2), 123. https://doi.org/10.3390/v11020123




_Brussaard.png)

