Seasonality Drives Microbial Community Structure, Shaping both Eukaryotic and Prokaryotic Host–Viral Relationships in an Arctic Marine Ecosystem
Abstract
1. Introduction
2. Materials and Methods
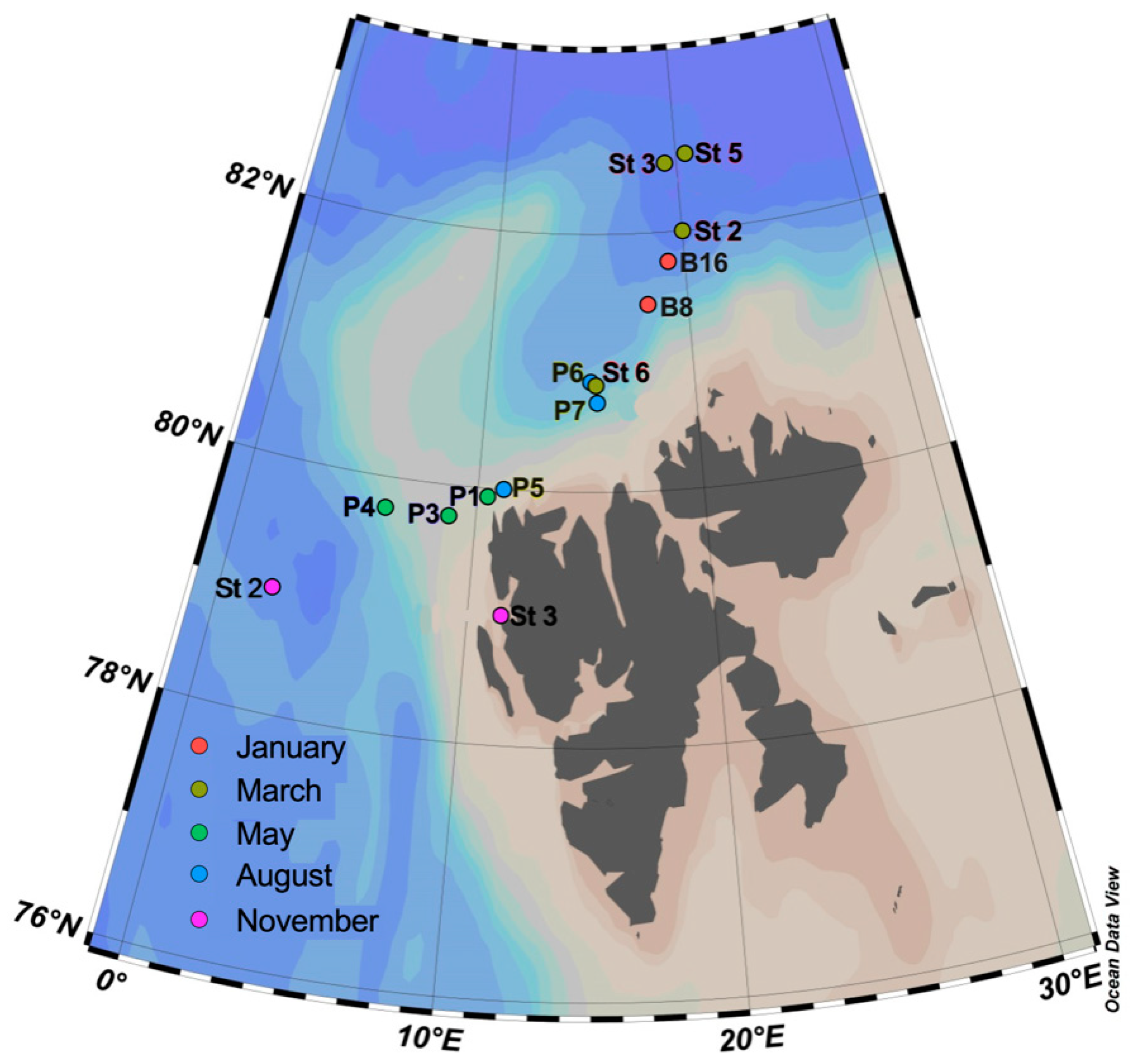
2.1. Sampling and Sample Preparation
Flow Cytometry (FCM)
2.2. DNA Extraction and High-Throughput Sequencing Library Construction
2.3. Sequence Analysis
3. Results
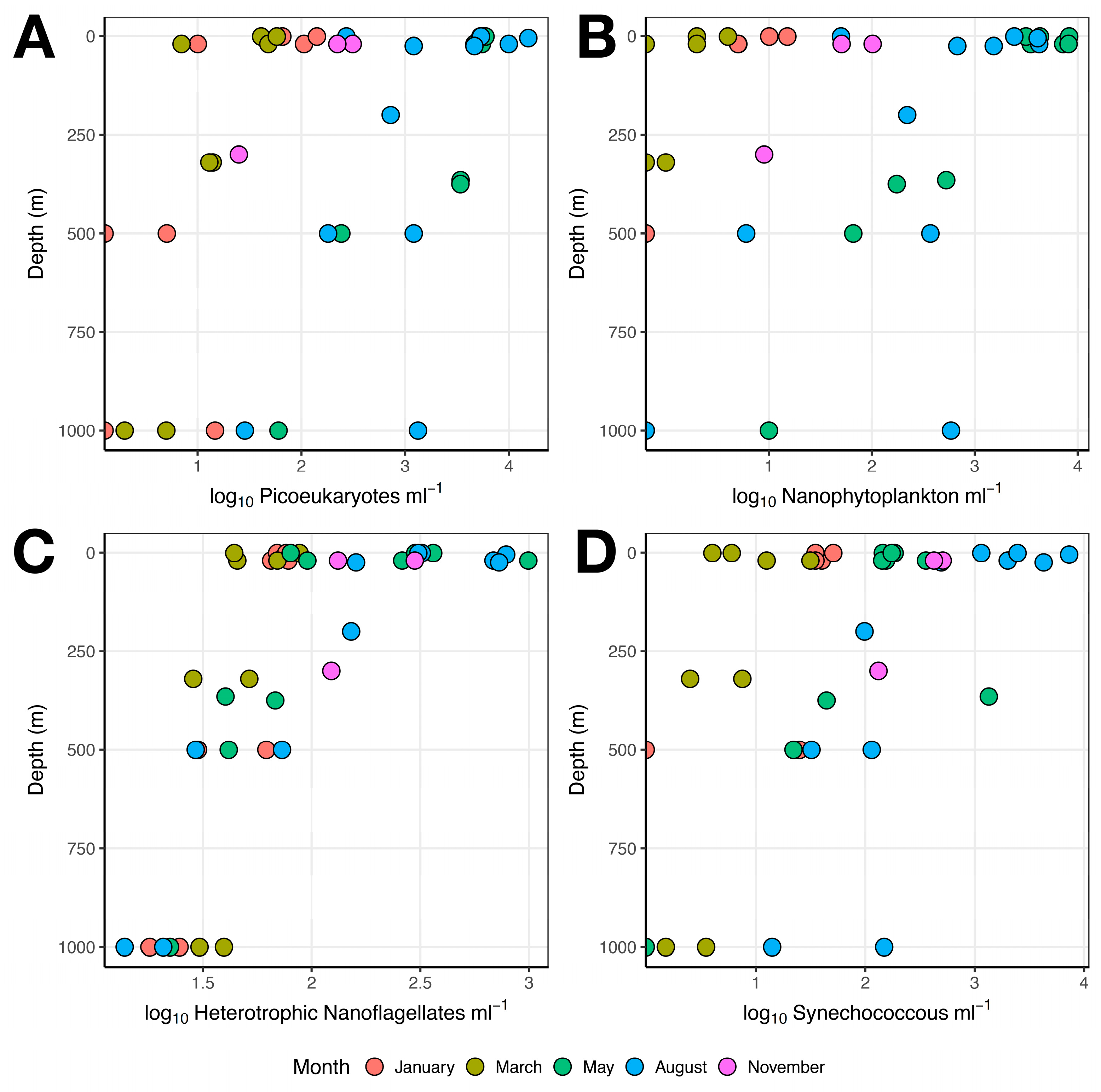
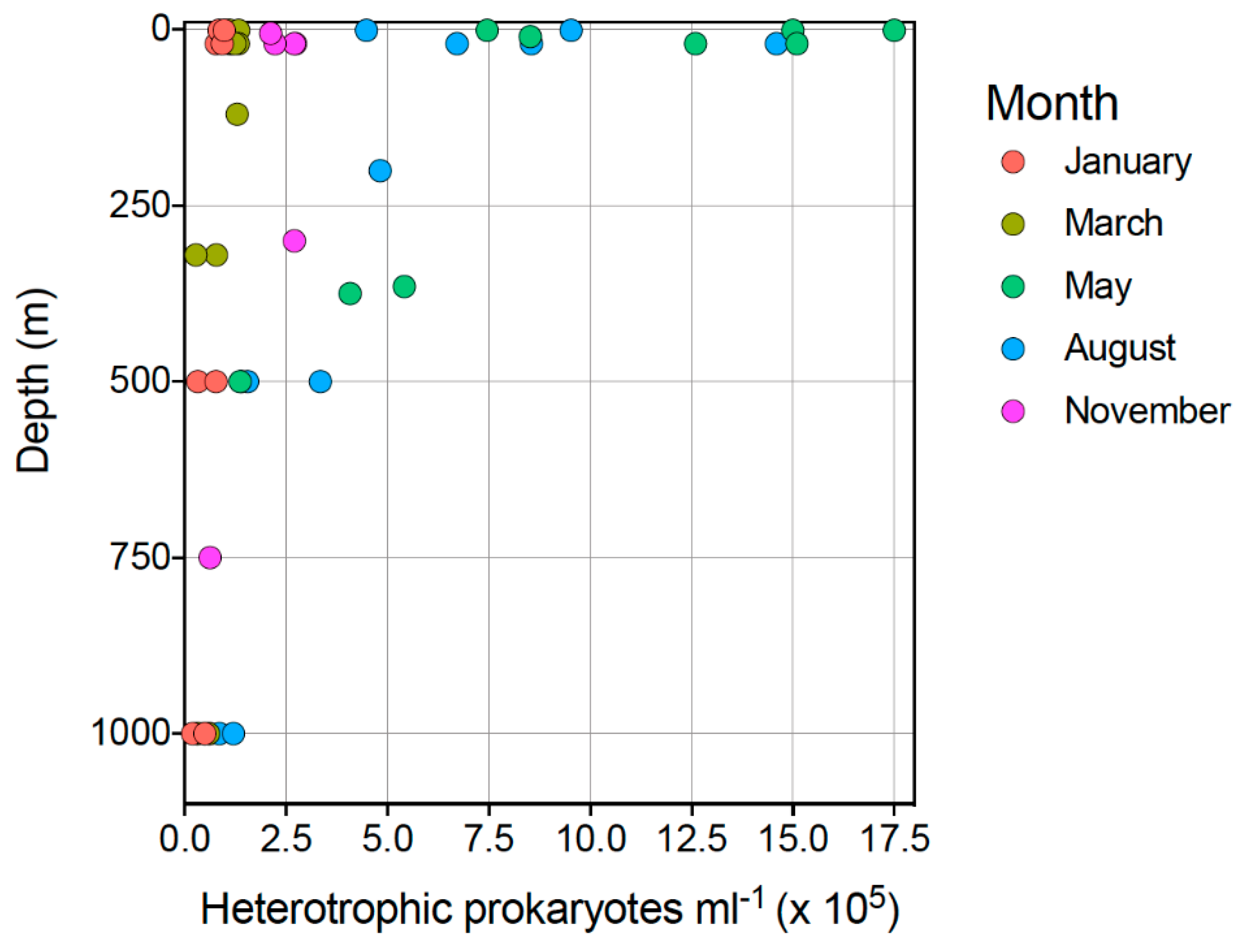
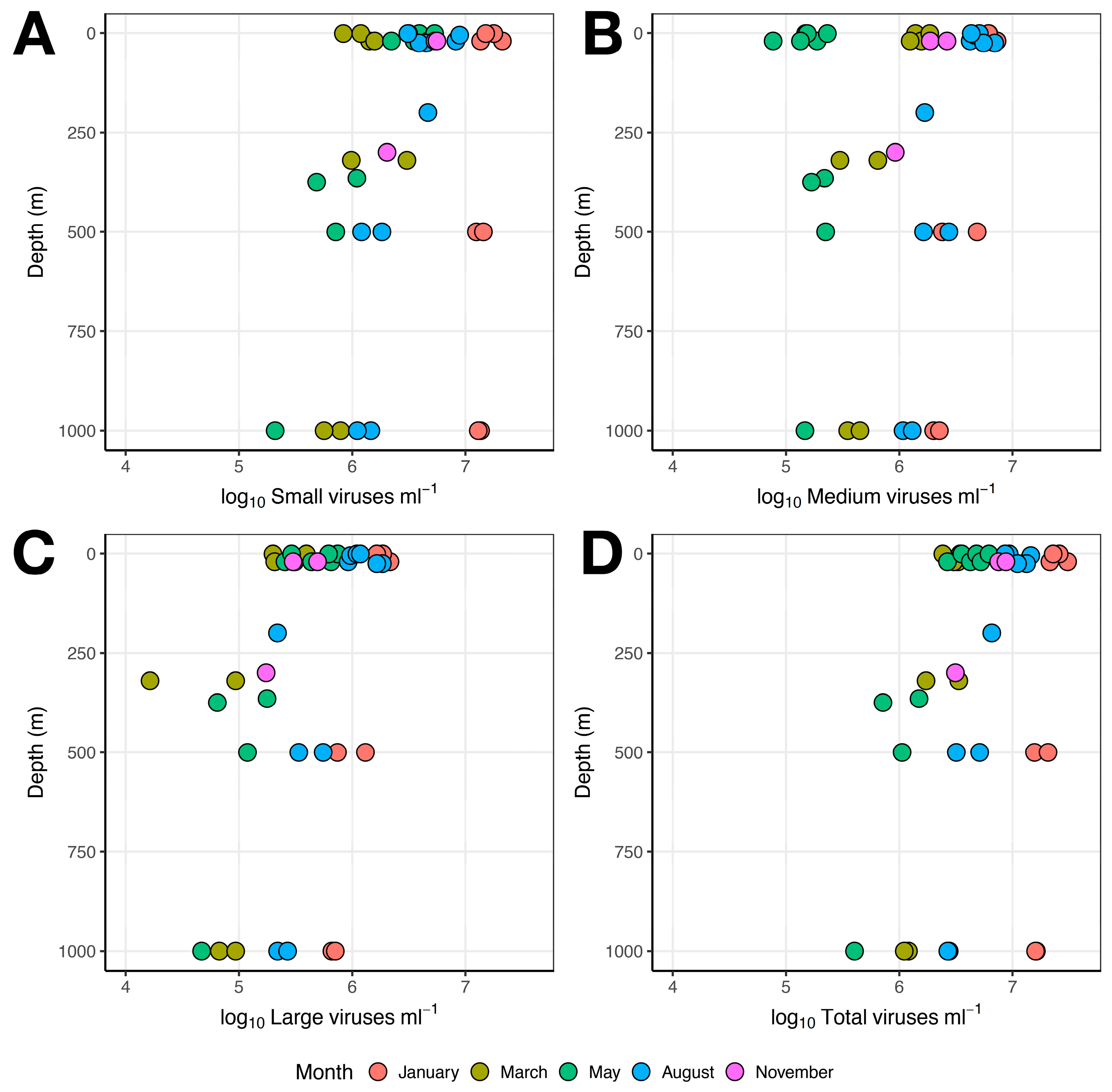
3.1. Microbial Abundance
3.1.1. Single-Celled Eukaryotes and Cyanobacteria
3.1.2. Heterotrophic Prokaryotes (HPs)
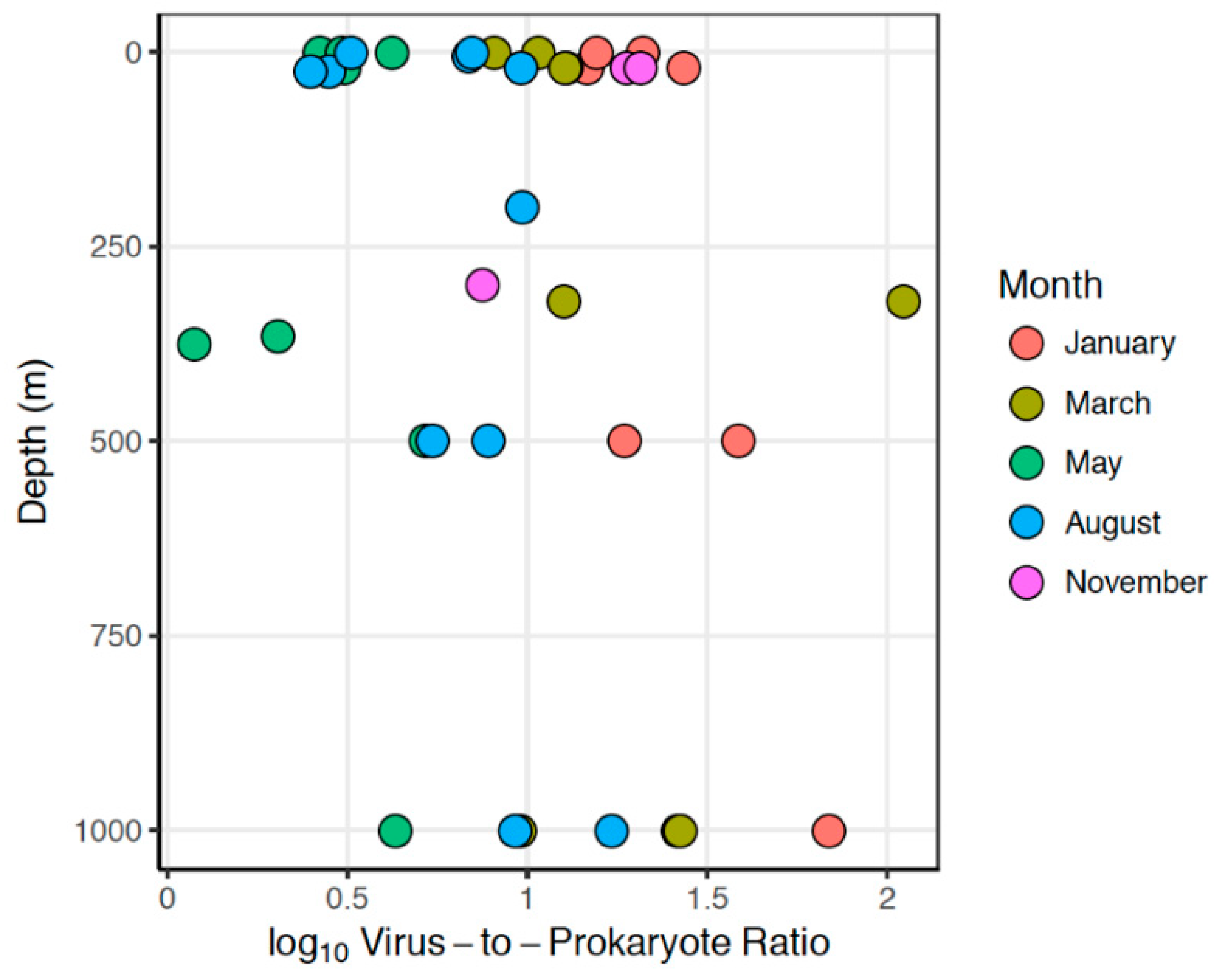
3.1.3. Viruses
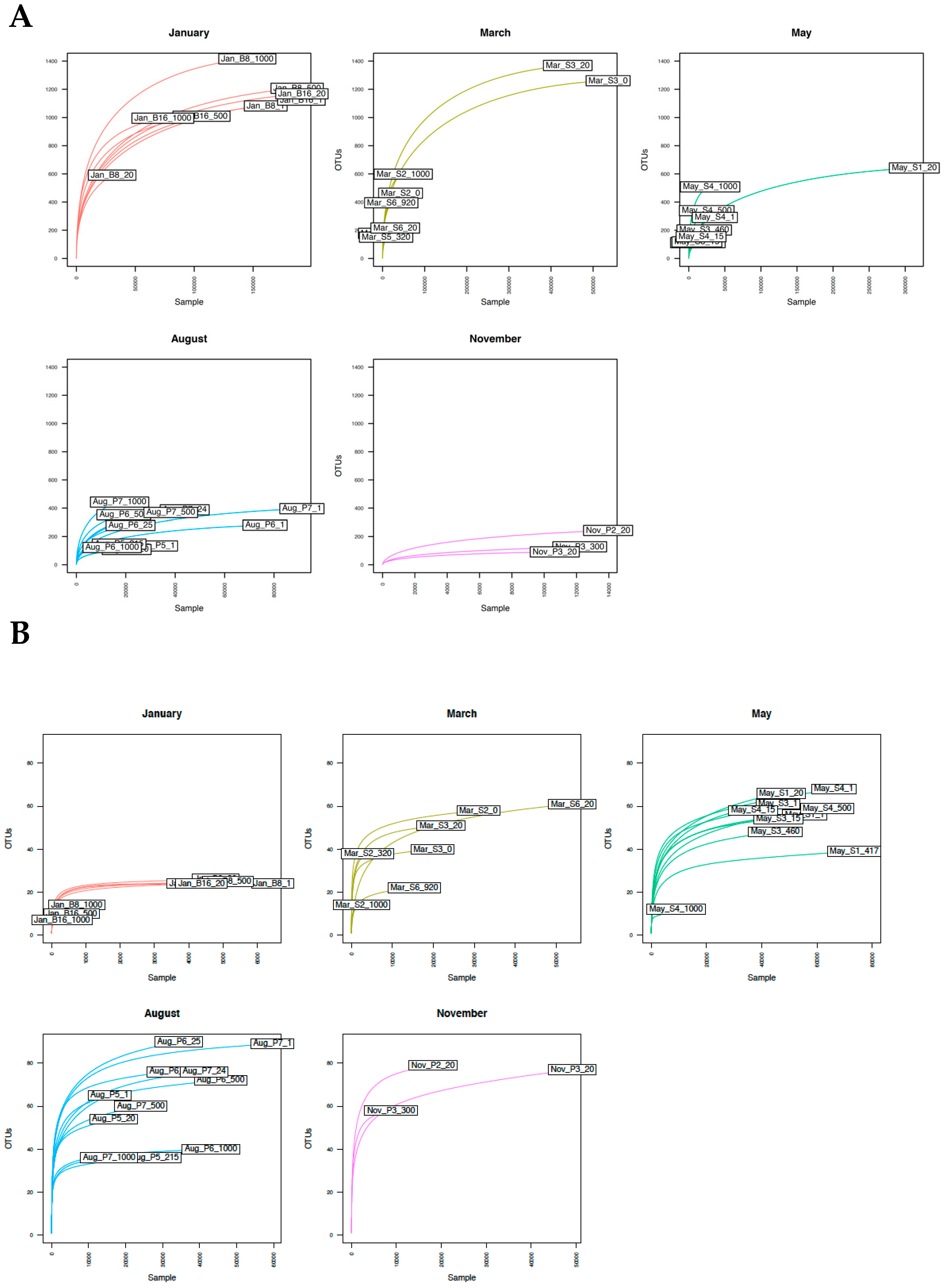
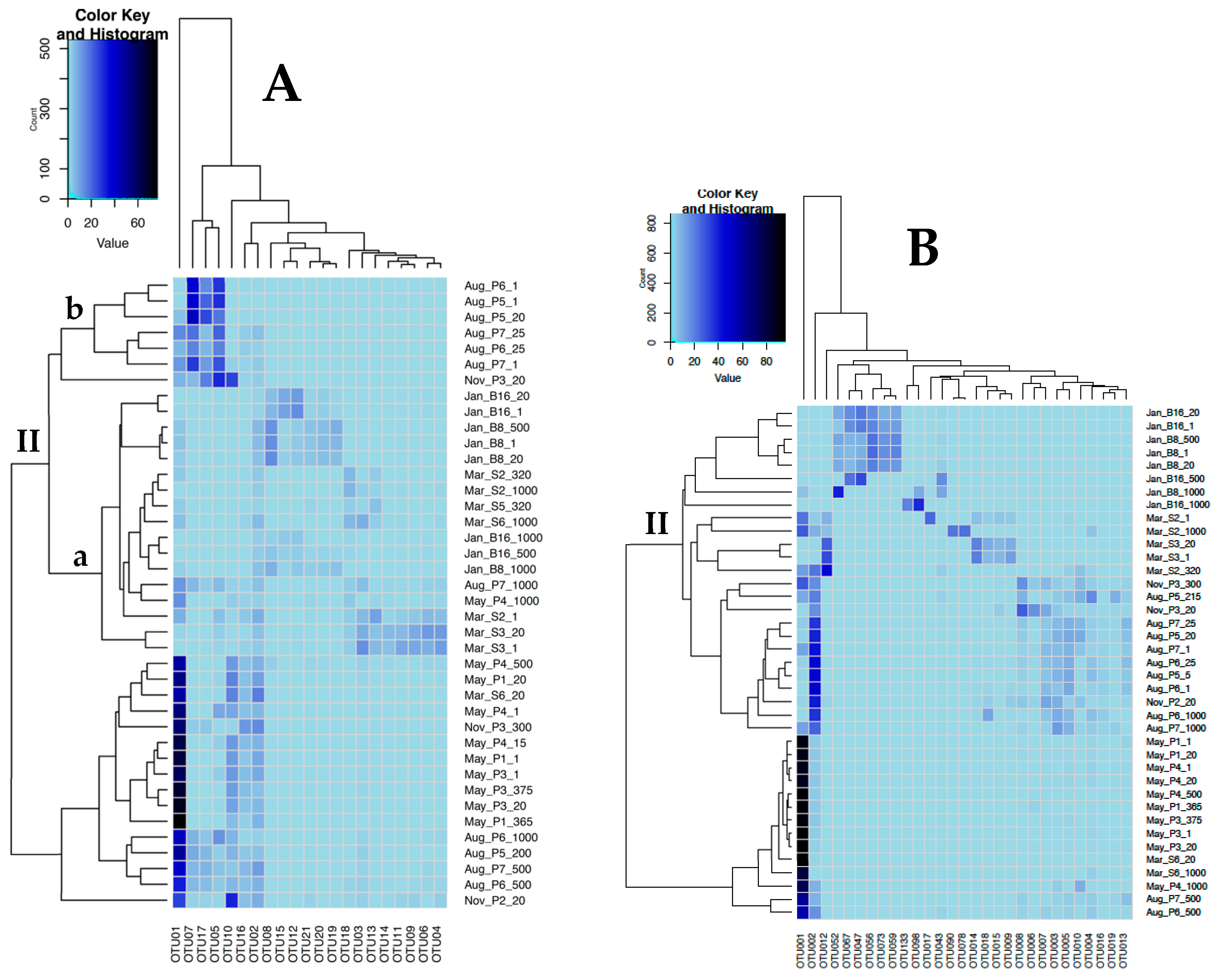
3.2. Sequence Analysis
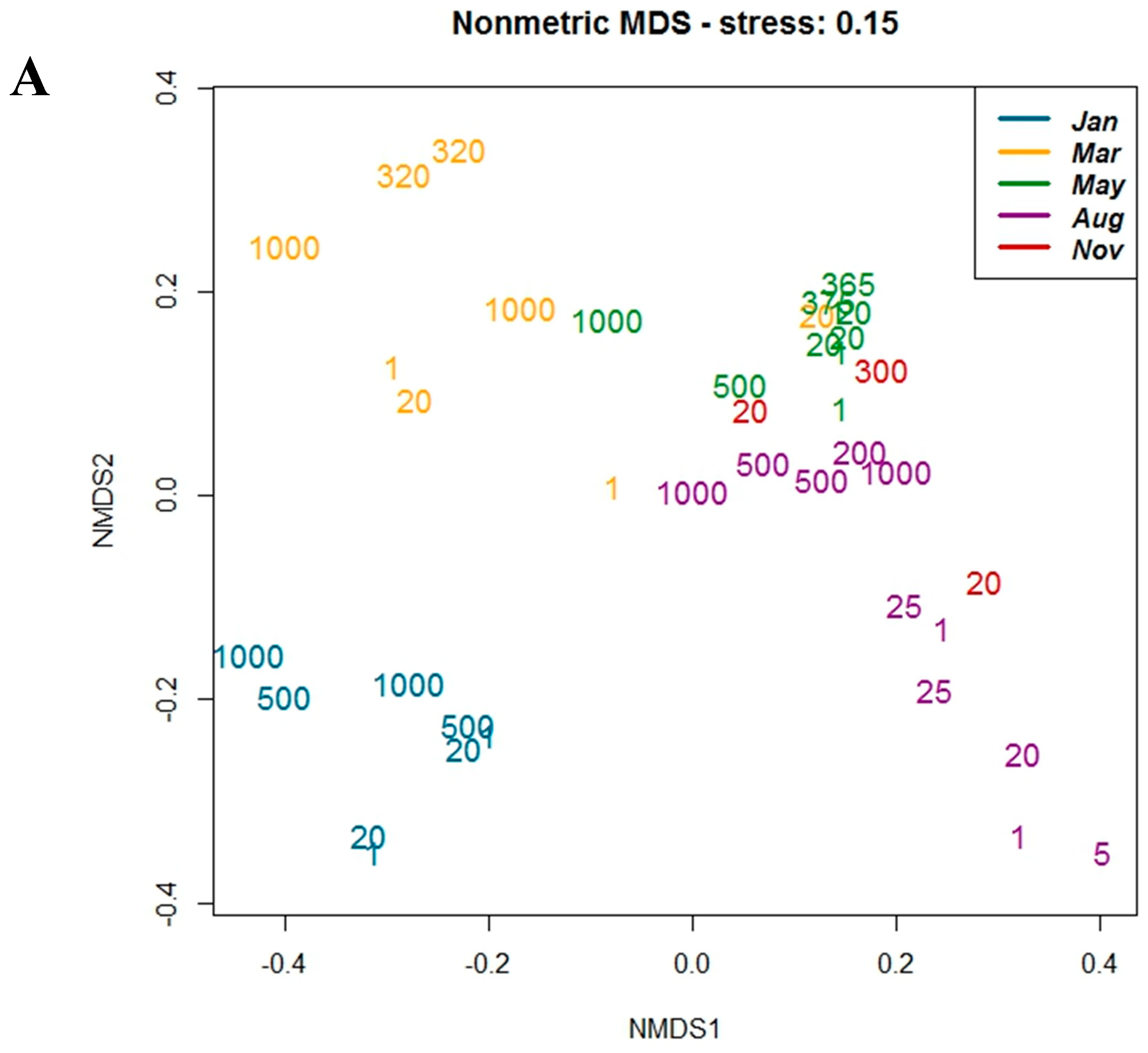
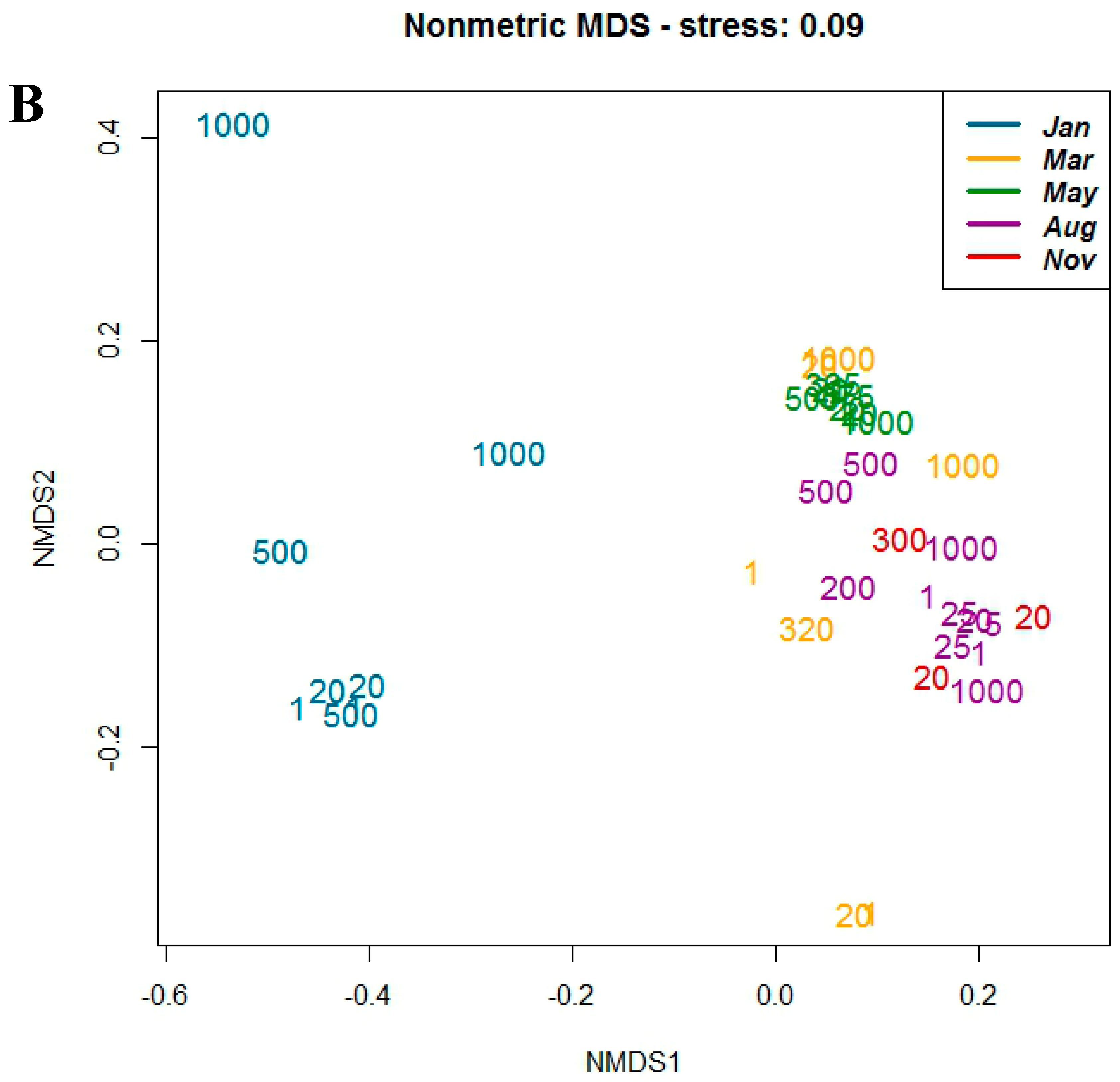
3.3. Correlation with Environmental Parameters
4. Discussion
4.1. Linking Virus and Host Communities
4.2. Seasonality of Virus Assemblages
4.3. Depth- and Winter-Driven Diversification
4.4. Grazing by Microzooplankton
Supplementary Material
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Stroeve, J.; Holland, M.M.; Meier, W.; Scambos, T.; Serreze, M. Arctic sea ice decline: Faster than forecast. Geophys. Res. Lett. 2007, 34. [Google Scholar] [CrossRef]
- Berge, J.; Cottier, F.; Last, K.S.; Varpe, Ø.; Leu, E.; Søreide, J.; Eiane, K.; Falk-Petersen, S.; Willis, K.; Nygård, H.; et al. Diel vertical migration of arctic zooplankton during the polar night. Biol. Lett. 2009, 5, 69–72. [Google Scholar] [CrossRef] [PubMed]
- Berge, J.; Daase, M.; Renaud, P.E.; Ambrose, W.G.; Darnis, G.; Last, K.S.; Leu, E.; Cohen, J.H.; Johnsen, G.; Moline, M.A.; et al. Unexpected levels of biological activity during the polar night offer new perspectives on a warming arctic. Curr. Biol. 2015, 25, 2555–2561. [Google Scholar] [CrossRef] [PubMed]
- Wilson, B.; Müller, O.; Nordmann, E.-L.; Seuthe, L.; Bratbak, G.; Øvreås, L. Changes in marine prokaryote composition with season and depth over an arctic polar year. Front. Mar. Sci. 2017, 4, 95. [Google Scholar] [CrossRef]
- Paulsen, M.L.; Seuthe, L.; Reigstad, M.; Larsen, A.; Cape, M.R.; Vernet, M. Asynchronous accumulation of organic carbon and nitrogen in the atlantic gateway to the Arctic ocean. Front. Mar. Sci. 2018. [Google Scholar] [CrossRef]
- Pedrós-Alió, C.; Potvin, M.; Lovejoy, C. Diversity of planktonic microorganisms in the Arctic ocean. Prog. Oceanogr. 2015, 139, 233–243. [Google Scholar] [CrossRef]
- Bergh, O.; Borsheim, K.Y.; Bratbak, G.; Heldal, M. High abundance of viruses found in aquatic environments. Nature 1989, 340, 467–468. [Google Scholar] [CrossRef]
- Suttle, C. The viriosphere: The greatest biological diversity on earth and driver of global processes. Environ. Microbiol. 2005, 7, 481–482. [Google Scholar] [CrossRef] [PubMed]
- Thingstad, T.F. Elements of a theory for the mechanisms controlling abundance, diversity, and biogeochemical role of lytic bacterial viruses in aquatic systems. Limnol. Oceanogr. 2000, 45, 1320–1328. [Google Scholar] [CrossRef]
- Winter, C.; Smit, A.; Herndl, G.J.; Weinbauer, M.G. Linking bacterial richness with viral abundance and prokaryotic activity. Limnol. Oceanogr. 2005, 50, 968–977. [Google Scholar] [CrossRef]
- Sandaa, R.-A.; Pree, B.; Larsen, A.; Våge, S.; Töpper, B.; Töpper, P.J.; Thyrhaug, R.; Thingstad, F.T. The response of heterotrophic prokaryote and viral communities to labile organic carbon inputs is controlled by the predator food chain structure. Viruses 2017, 9, 238. [Google Scholar] [CrossRef] [PubMed]
- Breitbart, M.; Bonnain, C.; Malki, K.; Sawaya, N.A. Phage puppet masters of the marine microbial realm. Nat. Microbiol. 2018, 3, 754–766. [Google Scholar] [CrossRef] [PubMed]
- Steward, G.F.; Smith, D.C.; Azam, F. Abundance and production of bacteria and viruses in the bering and chukchi seas. Mar. Ecol. Prog. Ser. 1996, 131, 287–300. [Google Scholar] [CrossRef]
- Yager, P.L.; Connelly, T.L.; Mortazavi, B.; Wommack, K.E.; Bano, N.; Bauer, J.E.; Opsahl, S.; Hollibaugh, J.T. Dynamic bacterial and viral response to an algal bloom at subzero temperatures. Limnol. Oceanogr. 2001, 46, 790–801. [Google Scholar] [CrossRef]
- Middelboe, M.; Nielsen, T.G.; Bjornsen, P.K. Viral and bacterial production in the north water: In situ measurements, batch-culture experiments and characterization and distribution of a virus-host system. Deep Sea Res. Part II Top. Stud. Oceanogr. 2002, 49, 5063–5079. [Google Scholar] [CrossRef]
- Paterson, S.; Vogwill, T.; Buckling, A.; Benmayor, R.; Spiers, A.J.; Thomson, N.R.; Quail, M.; Smith, F.; Walker, D.; Libberton, B.; et al. Antagonistic coevolution accelerates molecular evolution. Nature 2010, 464, 275–278. [Google Scholar] [CrossRef] [PubMed]
- Thingstad, F.; Våge, S.; Storesund, J.E.; Sandaa, R.-A.; Giske, J. When a species is the sum of its strains; how strain-specific viruses can control microbial species diversity. Proc. Natl. Acad. Sci. USA 2014, 111, 7813–7818. [Google Scholar] [CrossRef]
- Bratbak, G.; Thingstad, F.; Heldal, M. Viruses and the microbial loop. Microb. Ecol. 1994, 28, 209–221. [Google Scholar] [CrossRef]
- Wilhelm, S.W.; Suttle, C.A. Viruses and nutrient cycles in the sea—Viruses play critical roles in the structure and function of aquatic food webs. BioScience 1999, 49, 781–788. [Google Scholar] [CrossRef]
- Suttle, C.A. Marine viruses—Major players in the global ecosystem. Nat. Rev. Microbiol. 2007, 5, 801–812. [Google Scholar] [CrossRef]
- Guidi, L.; Chaffron, S.; Bittner, L.; Eveillard, D.; Larhlimi, A.; Roux, S.; Darzi, Y.; Audic, S.; Berline, L.; Brum, J.R.; et al. Plankton networks driving carbon export in the oligotrophic ocean. Nature 2016, 532, 465. [Google Scholar] [CrossRef] [PubMed]
- Suttle, C.A. Viruses in the sea. Nature 2005, 437, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Sandaa, R.-A. Burden or benefit? Virus-host interactions in the marine environment. Res. Microbiol. 2008, 159, 374–381. [Google Scholar] [CrossRef] [PubMed]
- Adriaenssens, E.M.; Cowan, D.A. Using signature genes as tools to assess environmental viral ecology and diversity. Appl. Environ. Microbiol. 2014, 80, 4470–4480. [Google Scholar] [CrossRef] [PubMed]
- Pagarete, A.; Chow, C.-E.T.; Johannessen, T.; Fuhrman, J.A.; Thingstad, T.F.; Sandaa, R.A. Strong seasonality and interannual recurrence in marine myovirus communities. Appl. Environ. Microbiol. 2013, 79, 6253–6259. [Google Scholar] [CrossRef] [PubMed]
- Johannessen, T.; Larsen, A.; Bratbak, G.; Pagarete, A.; Edvardsen, B.; Egge, E.; Sandaa, R.-A. Seasonal dynamics of haptophytes and dsdna algal viruses suggest complex virus-host relationship. Viruses 2017, 9, 84. [Google Scholar] [CrossRef] [PubMed]
- Ackermann, H.W.; Krisch, H.M. A catalogue of t4-type bacteriophages. Arch. Virol. 1997, 142, 2329–2345. [Google Scholar] [CrossRef]
- Filee, J.; Tetart, F.; Suttle, C.A.; Krisch, H.M. Marine t4-type bacteriophages, a ubiquitous component of the dark matter of the biosphere. Proc. Nat.l Acad. Sci. USA 2005, 102, 12471–12476. [Google Scholar] [CrossRef]
- Le Moine Bauer, S.; Stensland, A.; Daae, F.L.; Sandaa, R.-A.; Thorseth, I.H.; Steen, I.H.; Dahle, H. Water masses and depth structure prokaryotic and T4-like viral communities around hydrothermal systems of the nordic seas. Front. Microbiol. 2018, 9, 1002. [Google Scholar] [CrossRef]
- Payet, J.P.; Suttle, C.A. To kill or not to kill: The balance between lytic and lysogenic viral infection is driven by trophic status. Limnol. Oceanogr. 2013, 58, 465–474. [Google Scholar] [CrossRef]
- Liu, L.; Cai, L.; Zhang, R. Co-existence of freshwater and marine T4-like myoviruses in a typical subtropical estuary. FEMS Microbiol. Ecol. 2017, 93. [Google Scholar] [CrossRef] [PubMed]
- Wilson, W.H.; Van Etten, J.L.; Allen, M.J. The phycodnaviridae: The story of how tiny giants rule the world. Curr. Top. Microbiol. Immunol. 2009, 328, 1–42. [Google Scholar] [PubMed]
- Schvarcz, C.R.; Steward, G.F. A giant virus infecting green algae encodes key fermentation genes. Virology 2018, 518, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Mihara, T.; Koyano, H.; Hingamp, P.; Grimsley, N.; Goto, S.; Ogata, H. Taxon richness of “megaviridae” exceeds those of bacteria and archaea in the ocean. Microbiol. Environ. 2018, 33, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Hingamp, P.; Grimsley, N.; Acinas, S.G.; Clerissi, C.; Subirana, L.; Poulain, J.; Ferrera, I.; Sarmento, H.; Villar, E.; Lima-Mendez, G.; et al. Exploring nucleo-cytoplasmic large DNA viruses in tara oceans microbial metagenomes. ISME J. 2013, 7, 1678–1695. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, A.; Bratbak, G.; Heldal, M. Isolation and characterization of a virus infecting phaeocystis pouchetii (prymnesiophyceae). J. Phycol. 1996, 32, 923–927. [Google Scholar] [CrossRef]
- Marquardt, M.; Vader, A.; Stübner, E.I.; Reigstad, M.; Gabrielsen, T.M. Strong seasonality of marine microbial eukaryotes in a high-arctic fjord (Isfjorden, in west Spitsbergen, Norway). Appl. Environ. Microbiol. 2016, 82, 1868. [Google Scholar] [CrossRef]
- Lovejoy, C.; Vincent, W.F.; Bonilla, S.; Roy, S.; Martineau, M.-J.; Terrado, R.; Potvin, M.; Massana, R.; Pedrós-Alió, C. Distribution, phylogeny, and growth of cold−adapted picoprasinophytes in Arctic seas. J. Phycol. 2007, 43, 78–89. [Google Scholar] [CrossRef]
- Paulsen, M.L.; Doré, H.; Garczarek, L.; Seuthe, L.; Müller, O.; Sandaa, R.-A.; Bratbak, G.; Larsen, A. Synechococcus in the atlantic gateway to the arctic ocean. Front. Mar. Sci. 2016, 3, 191. [Google Scholar] [CrossRef]
- Marie, D.; Brussaard, C.P.D.; Thyrhaug, R.; Bratbak, G.; Vaulot, D. Enumeration of marine viruses in culture and natural samples by flow cytometry. Appl. Environ. Microbiol. 1999, 65, 45–52. [Google Scholar]
- Zubkov, M.V.; Burkill, P.H.; Topping, J.N. Flow cytometric enumeration of DNA-stained oceanic planktonic protists. J. Plankton Res. 2007, 29, 79–86. [Google Scholar] [CrossRef]
- Larsen, A.; Flaten, G.A.F.; Sandaa, R.A.; Castberg, T.; Thyrhaug, R.; Erga, S.R.; Jacquet, S.; Bratbak, G. Spring phytoplankton bloom dynamics in norwegian coastal waters: Microbial community succession and diversity. Limnol. Oceanogr. 2004, 49, 180–190. [Google Scholar] [CrossRef]
- Larsen, J.B.; Larsen, A.; Thyrhaug, R.; Bratbak, G.; Sandaa, R. Marine viral populations detected during a nutrient induced phytoplankton bloom at elevated pCO2 levels. Biogeosciences 2008, 5, 523–533. [Google Scholar] [CrossRef]
- Christaki, U.; Courties, C.; Massana, R.; Catala, P.; Lebaron, P.; Gasol, J.M.; Zubkov, M.V. Optimized routine flow cytometric enumeration of heterotrophic flagellates using SYBR Green I. Limnol. Oceanogr. Methods 2011, 9, 329–339. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. Qiime allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335. [Google Scholar] [CrossRef] [PubMed]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J.; et al. Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. Search and clustering orders of magnitude faster than blast. Bioinformatics 2010, 26, 2460–2461. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.G.; Friendly, M.; Kindt, R.; Legendre, P.; McGlin, D.; Minchin, P.; O’Hara, R.B.; Simpson, G.; Solymos, P.; et al. Vegan: Community Ecology Package. R package Version 2.4-3. 2017. Available online: https://www.researchgate.net/publication/323265822_vegan_Community_Ecology_Package_R_package_version_24-3_2017_accessed_2016_Jan_1 (accessed on 1 January 2016).
- Wickham, H. Ggplot2: Elegant graphics for data analysis. Springer Publishing Company, Incorporated: New York, NY, USA, 2009. [Google Scholar]
- Warnes, G.R.; Bolker, B.; Bonebakker, L.; Gentleman, R.; Liaw, W.H.A.; Lumley, T.; Maechler, M.; Magnusson, A.; Moeller, S.; Schwartz, M.; et al. Gplots: Various R Programming Tools for Plotting Data. R Package, Version. 3.0.1.; Comprehensive R Archive Network. Available online: https://cran.r-project.org/web/packages/gplots/index.html (accessed on 1 January 2016).
- Kent, W.J. Blattextemdash the blast-like alignment tool. Genome Res. 2002, 12, 656–664. [Google Scholar] [CrossRef]
- Edgar, R.C. Muscle: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. Mega6: Molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef]
- Jones, D.T.; Taylor, W.R.; Thornton, J.M. The rapid generation of mutation data matrices from protein sequences. Comput. Appl. Biosci. 1992, 8, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Seuthe, L.; Töpper, B.; Reigstad, M.; Thyrhaug, R.; Vaquer-Sunyer, R. Microbial communities and processes in ice-covered arctic waters of the northwestern fram strait (75 to 80° N) during the vernal pre-bloom phase. Aquat. Microb. Ecol. 2011, 64, 253–266. [Google Scholar] [CrossRef]
- Wassmann, P.; Reigstad, M. Future arctic ocean seasonal ice zones and implications for pelagic-benthic coupling. Oceanography 2011, 24, 221–231. [Google Scholar] [CrossRef]
- Wilson, W.H.; Gilg, I.C.; Moniruzzaman, M.; Field, E.K.; Koren, S.; LeCleir, G.R.; Martinez Martinez, J.; Poulton, N.J.; Swan, B.K.; Stepanauskas, R.; et al. Genomic exploration of individual giant ocean viruses. ISME J. 2017, 11, 1736. [Google Scholar] [CrossRef] [PubMed]
- Brussaard, C.P.D.; Bratbak, G.; Baudoux, A.-C.; Ruardij, P. Phaeocystis and its interaction with viruses. Biogeochemistry 2007, 83, 201–215. [Google Scholar] [CrossRef]
- Bidle, K.D.; Falkowski, P.G. Cell death in planktonic, photosynthetic microorganisms. Nat. Rev. Microbiol. 2004, 2, 643. [Google Scholar] [CrossRef]
- Egge, E.; John, U.; Vaulot, D.; Lund-Paulsen, M.; Larsen, A.; Edvardsen, B. Marine protist diversity and dynamics throughout the arctic year. In prep.
- Joli, N.; Monier, A.; Logares, R.; Lovejoy, C. Seasonal patterns in arctic prasinophytes and inferred ecology of Bathycoccus unveiled in an arctic winter metagenome. ISME J. 2017, 11, 1372–1385. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, C.J.M.; Flintrop, C.M.; Rost, B. The arctic picoeukaryote Micromonas pusilla benefits synergistically from warming and ocean acidification. Biogeosciences 2018, 15, 4353–4365. [Google Scholar] [CrossRef]
- Thingstad, T.F.; Lignell, R. Theoretical models for the control of bacterial growth rate, abundance, diversity and carbon demand. Aquat. Microb. Ecol. 1997, 13, 19–27. [Google Scholar] [CrossRef]
- Zwirglmaier, K.; Jardillier, L.; Ostrowski, M.; Mazard, S.; Garczarek, L.; Vaulot, D.; Not, F.; Massana, R.; Ulloa, O.; Scanlan, D.J. Global phylogeography of marine Synechococcus and Prochlorococcus reveals a distinct partitioning of lineages among oceanic biomes. Environ. Microbiol. 2007, 10, 147–161. [Google Scholar] [CrossRef] [PubMed]
- Cottrell, M.T.; Kirchman, D.L. Photoheterotrophic microbes in the arctic ocean in summer and winter. Appl. Environ. Microbiol. 2009, 75, 4958–4966. [Google Scholar] [CrossRef] [PubMed]
- Sandaa, R.A.; Larsen, A. Seasonal variations in viral-host populations in norwegian coastal waters: Focusing on the cyanophage community infecting marine synechococcus species. Appl. Environ. Microbiol. 2006, 72, 4610–4618. [Google Scholar] [CrossRef] [PubMed]
- Ray, J.L.; Töpper, B.; An, S.; Silyakova, A.; Spindelböck, J.; Thyrhaug, R.; DuBow, M.S.; Thingstad, T.F.; Sandaa, R.-A. Effect of increased pco2 on bacterial assemblage shifts in response to glucose addition in fram strait seawater mesocosms. FEMS Microbiol. Ecol. 2012, 82, 713–723. [Google Scholar] [CrossRef] [PubMed]
- Tarutani, K.; Nagasaki, K.; Yamaguchi, M. Viral impacts on total abundance and clonal composition of the harmful bloom-forming phytoplankton heterosigma akashiwo. Appl. Environ. Microbiol. 2000, 66, 4916–4920. [Google Scholar] [CrossRef] [PubMed]
- Kimura, S.; Sako, Y.; Yoshida, T. Rapid microcystis cyanophage gene diversification revealed by long- and short-term genetic analyses of the tail sheath gene in a natural pond. Appl. Environ. Microbiol. 2013, 79, 2789–2795. [Google Scholar] [CrossRef] [PubMed]
- Berge, J.; Renaud, P.E.; Darnis, G.; Cottier, F.; Last, K.; Gabrielsen, T.M.; Johnsen, G.; Seuthe, L.; Weslawski, J.M.; Leu, E.; et al. In the dark: A review of ecosystem processes during the arctic polar night. Prog. Oceanogr. 2015, 139, 258–271. [Google Scholar] [CrossRef]
- Darnis, G.; Robert, D.; Pomerleau, C.; Link, H.; Archambault, P.; Nelson, R.J.; Geoffroy, M.; Tremblay, J.-É.; Lovejoy, C.; Ferguson, S.H.; et al. Current state and trends in canadian arctic marine ecosystems: Ii. Heterotrophic food web, pelagic-benthic coupling, and biodiversity. Clim. Chang. 2012, 115, 179–205. [Google Scholar] [CrossRef]
- Stoecker, D.K.; Hansen, P.J.; Caron, D.A.; Mitra, A. Mixotrophy in the marine plankton. Annu. Rev. Mar. Sci. 2017, 9, 311–335. [Google Scholar] [CrossRef]
- De Corte, D.; Sintes, E.; Yokokawa, T.; Herndl, G.J. Changes in viral and bacterial communities during the ice-melting season in the coastal arctic (Kongsfjorden, Ny-Ålesund). Environ. Microbiol. 2011, 13, 1827–1841. [Google Scholar] [CrossRef]
- Long, A.M.; Short, S.M. Seasonal determinations of algal virus decay rates reveal overwintering in a temperate freshwater pond. ISME J. 2016, 10, 1602. [Google Scholar] [CrossRef]
- Williamson, K. Soil phage ecology: Abundance, distribution, and interactions with bacterial hosts. In Biocommunication in Soil Microorganisms; Günther, W., Ed.; Springer: Berlin/Heidelberg, Germany, 2011; Volume 23, pp. 113–136. [Google Scholar]
- Paulsen, M.L.; Riisgaard, K.; St. John, M.; Thingstad, T.F.; Nielsen, T.G. Heterotrophic nanoflagelate grazing faciliates subarctic Atlantic spring bloom development. Aquat. Microb. Ecol. 2017, 78, 161–176. [Google Scholar] [CrossRef]
- Brum, J.R.; Hurwitz, B.L.; Schofield, O.; Ducklow, H.W.; Sullivan, M.B. Seasonal time bombs: Dominant temperate viruses affect southern ocean microbial dynamics. ISME J. 2016, 10, 437–449. [Google Scholar] [CrossRef] [PubMed]
- Boras, J.A.; Sala, M.M.; Vázquez-Domínguez, E.; Weinbauer, M.G.; Vaqué, D. Annual changes of bacterial mortality due to viruses and protists in an oligotrophic coastal environment (nw mediterranean). Environ. Microbiol. 2009, 11, 1181–1193. [Google Scholar] [CrossRef] [PubMed]
- Williamson, S.J.; Houchin, L.A.; McDaniel, L.; Paul, J.H. Seasonal variation in lysogeny as depicted by prophage induction in tampa bay, florida. Appl. Environ. Microbiol. 2002, 68, 4307–4314. [Google Scholar] [CrossRef] [PubMed]
- Knowles, B.; Silveira, C.B.; Bailey, B.A.; Barott, K.; Cantu, V.A.; Cobián-Güemes, A.G.; Coutinho, F.H.; Dinsdale, E.A.; Felts, B.; Furby, K.A.; et al. Lytic to temperate switching of viral communities. Nature 2016, 531, 466. [Google Scholar] [CrossRef] [PubMed]
- Stasko, A.; Bluhm, B.; Michel, C.; Archambault, P.; Majewski, A.; Reist, J.; Swanson, H.; Power, M. Benthic–pelagic trophic coupling in an arctic marine food web along vertical water mass and organic matter gradients. Mar. Ecol. Prog. Ser. 2018, 594. [Google Scholar] [CrossRef]
- Mari, X.; Kerros, M.-E.; Weinbauer, M.G. Virus attachment to transparent exopolymeric particles along trophic gradients in the southwestern lagoon of new caledonia. Appl. Environ. Microbiol. 2007, 73, 5245–5252. [Google Scholar] [CrossRef]
- Lawrence, J.E.; Suttle, C. Effect of viral infection on sinking rates of Heterosigma akashiwo and its implications for bloom termination. Aquat. Microb. Ecool. 2004, 37, 1–7. [Google Scholar] [CrossRef]
- Nissimov, J.I.; Vandzura, R.; Johns, C.T.; Natale, F.; Haramaty, L.; Bidle, K.D. Dynamics of transparent exopolymer particle production and aggregation during viral infection of the coccolithophore, Emiliania huxleyi. Environ. Microbiol. 2018, 20, 2880–2897. [Google Scholar] [CrossRef]
- Fuhrman, J.A. Marine viruses and their biogeochemical and ecological effects. Nature 1999, 399, 541. [Google Scholar] [CrossRef]
- Wells, L.E.; Deming, J.W. Significance of bacterivory and viral lysis in bottom waters of franklin bay, canadian arctic, during winter. Aquat. Microb. Ecol. 2006, 43, 209–221. [Google Scholar] [CrossRef]
- Anderson, M.R.; Richard, B.R. Seasonal patterns in grazing mortality of bacterioplankton in polar oceans: A bipolar comparison. Aquat. Microb. Ecol. 2001, 25, 195–206. [Google Scholar] [CrossRef]
- Meshram, A.R.; Vader, A.; Kristiansen, S.; Gabrielsen, T.M. Microbial eukaryotes in an arctic under-ice spring bloom north of svalbard. Front. Microbiol. 2017, 8, 1099. [Google Scholar] [CrossRef] [PubMed]
- Moreira, D.; López-García, P. The rise and fall of picobiliphytes: How assumed autotrophs turned out to be heterotrophs. BioEssays 2014, 36, 468–474. [Google Scholar] [CrossRef] [PubMed]
- Lovejoy, C. Changing views of arctic protists (marine microbial eukaryotes) in a changing arctic. Acta Protozool. 2014, 53, 91–100. [Google Scholar]
 ). The evolutionary history was inferred using the maximum likelihood method based on the JTT matrix-based model [54] with 100 bootstraps. Branch lengths indicate the number of amino acid substitutions per site. Abbreviations: CroV = Cafeteria roenbergensis virus; Moumou = Moumouvirus goulette; Mimi = Mimivirus; Mega = Megavirus chiliensis; AaV = Aureococcus anophagefferens virus; PoV = Pyramimonas orientalis virus; PkV = Prymnesium kappa virus; HeV = Haptolina ericina virus; HhV = Haptolina hirta virus; CeV = Chrysochromulina ericina virus; PgV = Phaeocystis globosa virus; PpV = Phaeocystis pouchetii virus; PBCV = Paramecium bursaria chlorella virus; MpV = Micromonas pusilla virus; OsV = Ostreococcus sp. virus; OlV = Ostreococcus lucimarinus virus; BpV = Bathycoccus prasinos virus. Scale bar represents 0.2 substitutions per site.
). The evolutionary history was inferred using the maximum likelihood method based on the JTT matrix-based model [54] with 100 bootstraps. Branch lengths indicate the number of amino acid substitutions per site. Abbreviations: CroV = Cafeteria roenbergensis virus; Moumou = Moumouvirus goulette; Mimi = Mimivirus; Mega = Megavirus chiliensis; AaV = Aureococcus anophagefferens virus; PoV = Pyramimonas orientalis virus; PkV = Prymnesium kappa virus; HeV = Haptolina ericina virus; HhV = Haptolina hirta virus; CeV = Chrysochromulina ericina virus; PgV = Phaeocystis globosa virus; PpV = Phaeocystis pouchetii virus; PBCV = Paramecium bursaria chlorella virus; MpV = Micromonas pusilla virus; OsV = Ostreococcus sp. virus; OlV = Ostreococcus lucimarinus virus; BpV = Bathycoccus prasinos virus. Scale bar represents 0.2 substitutions per site.
 ). The evolutionary history was inferred using the maximum likelihood method based on the JTT matrix-based model [54] with 100 bootstraps. Branch lengths indicate the number of amino acid substitutions per site. Abbreviations: CroV = Cafeteria roenbergensis virus; Moumou = Moumouvirus goulette; Mimi = Mimivirus; Mega = Megavirus chiliensis; AaV = Aureococcus anophagefferens virus; PoV = Pyramimonas orientalis virus; PkV = Prymnesium kappa virus; HeV = Haptolina ericina virus; HhV = Haptolina hirta virus; CeV = Chrysochromulina ericina virus; PgV = Phaeocystis globosa virus; PpV = Phaeocystis pouchetii virus; PBCV = Paramecium bursaria chlorella virus; MpV = Micromonas pusilla virus; OsV = Ostreococcus sp. virus; OlV = Ostreococcus lucimarinus virus; BpV = Bathycoccus prasinos virus. Scale bar represents 0.2 substitutions per site.
). The evolutionary history was inferred using the maximum likelihood method based on the JTT matrix-based model [54] with 100 bootstraps. Branch lengths indicate the number of amino acid substitutions per site. Abbreviations: CroV = Cafeteria roenbergensis virus; Moumou = Moumouvirus goulette; Mimi = Mimivirus; Mega = Megavirus chiliensis; AaV = Aureococcus anophagefferens virus; PoV = Pyramimonas orientalis virus; PkV = Prymnesium kappa virus; HeV = Haptolina ericina virus; HhV = Haptolina hirta virus; CeV = Chrysochromulina ericina virus; PgV = Phaeocystis globosa virus; PpV = Phaeocystis pouchetii virus; PBCV = Paramecium bursaria chlorella virus; MpV = Micromonas pusilla virus; OsV = Ostreococcus sp. virus; OlV = Ostreococcus lucimarinus virus; BpV = Bathycoccus prasinos virus. Scale bar represents 0.2 substitutions per site.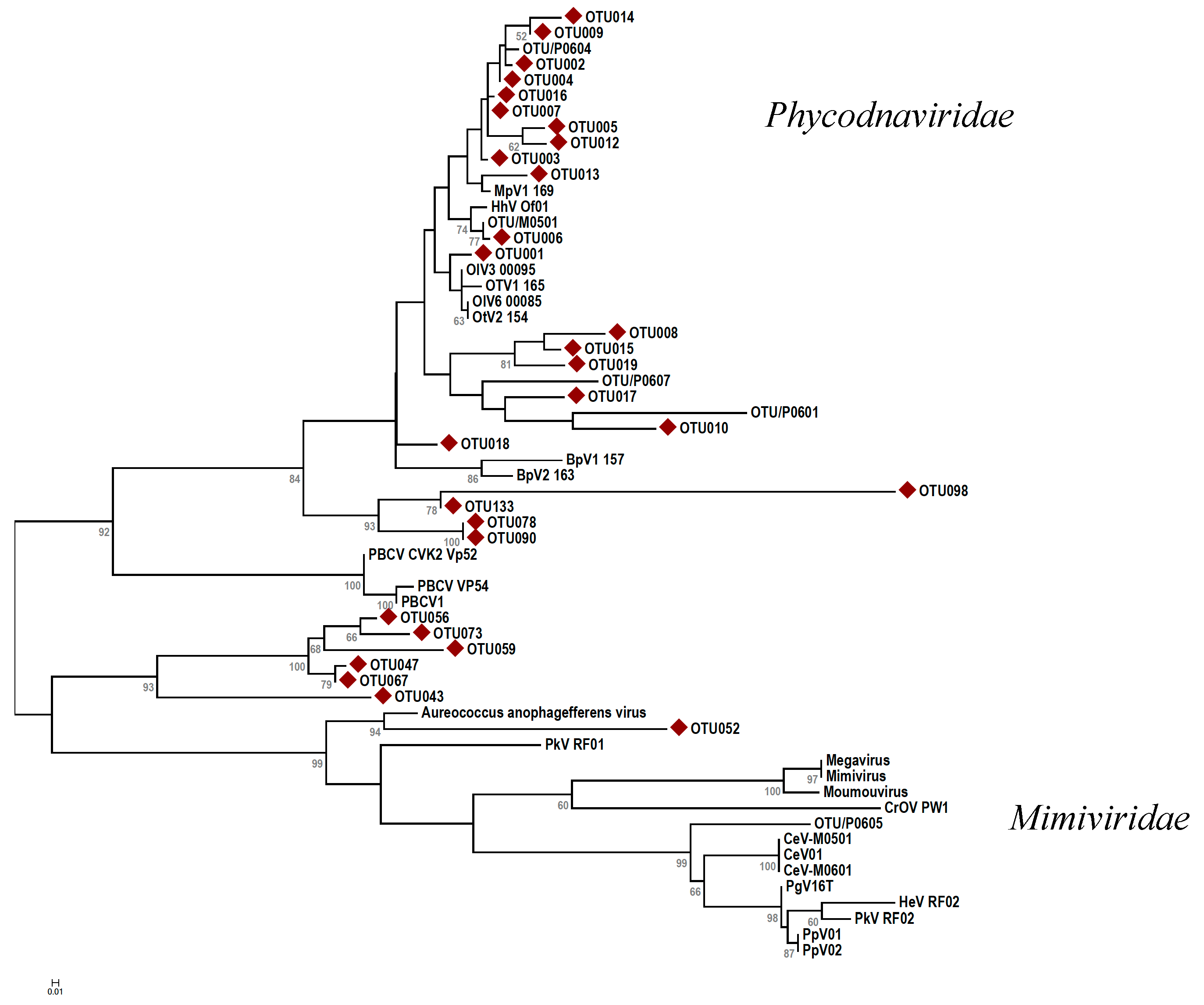
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sandaa, R.-A.; E. Storesund, J.; Olesin, E.; Lund Paulsen, M.; Larsen, A.; Bratbak, G.; Ray, J.L. Seasonality Drives Microbial Community Structure, Shaping both Eukaryotic and Prokaryotic Host–Viral Relationships in an Arctic Marine Ecosystem. Viruses 2018, 10, 715. https://doi.org/10.3390/v10120715
Sandaa R-A, E. Storesund J, Olesin E, Lund Paulsen M, Larsen A, Bratbak G, Ray JL. Seasonality Drives Microbial Community Structure, Shaping both Eukaryotic and Prokaryotic Host–Viral Relationships in an Arctic Marine Ecosystem. Viruses. 2018; 10(12):715. https://doi.org/10.3390/v10120715
Chicago/Turabian StyleSandaa, Ruth-Anne, Julia E. Storesund, Emily Olesin, Maria Lund Paulsen, Aud Larsen, Gunnar Bratbak, and Jessica Louise Ray. 2018. "Seasonality Drives Microbial Community Structure, Shaping both Eukaryotic and Prokaryotic Host–Viral Relationships in an Arctic Marine Ecosystem" Viruses 10, no. 12: 715. https://doi.org/10.3390/v10120715
APA StyleSandaa, R.-A., E. Storesund, J., Olesin, E., Lund Paulsen, M., Larsen, A., Bratbak, G., & Ray, J. L. (2018). Seasonality Drives Microbial Community Structure, Shaping both Eukaryotic and Prokaryotic Host–Viral Relationships in an Arctic Marine Ecosystem. Viruses, 10(12), 715. https://doi.org/10.3390/v10120715




