Characterization of Equine Parvovirus in Thoroughbred Breeding Horses from Germany
Abstract
1. Introduction
2. Materials and Methods
2.1. Serum Sample Collection
2.2. Detection of EqPV-H DNA
2.3. Detection of Anti-EqPV-H Antibodies
2.4. Data Collection and Study Design
2.5. Sequencing and Phylogeny
3. Results
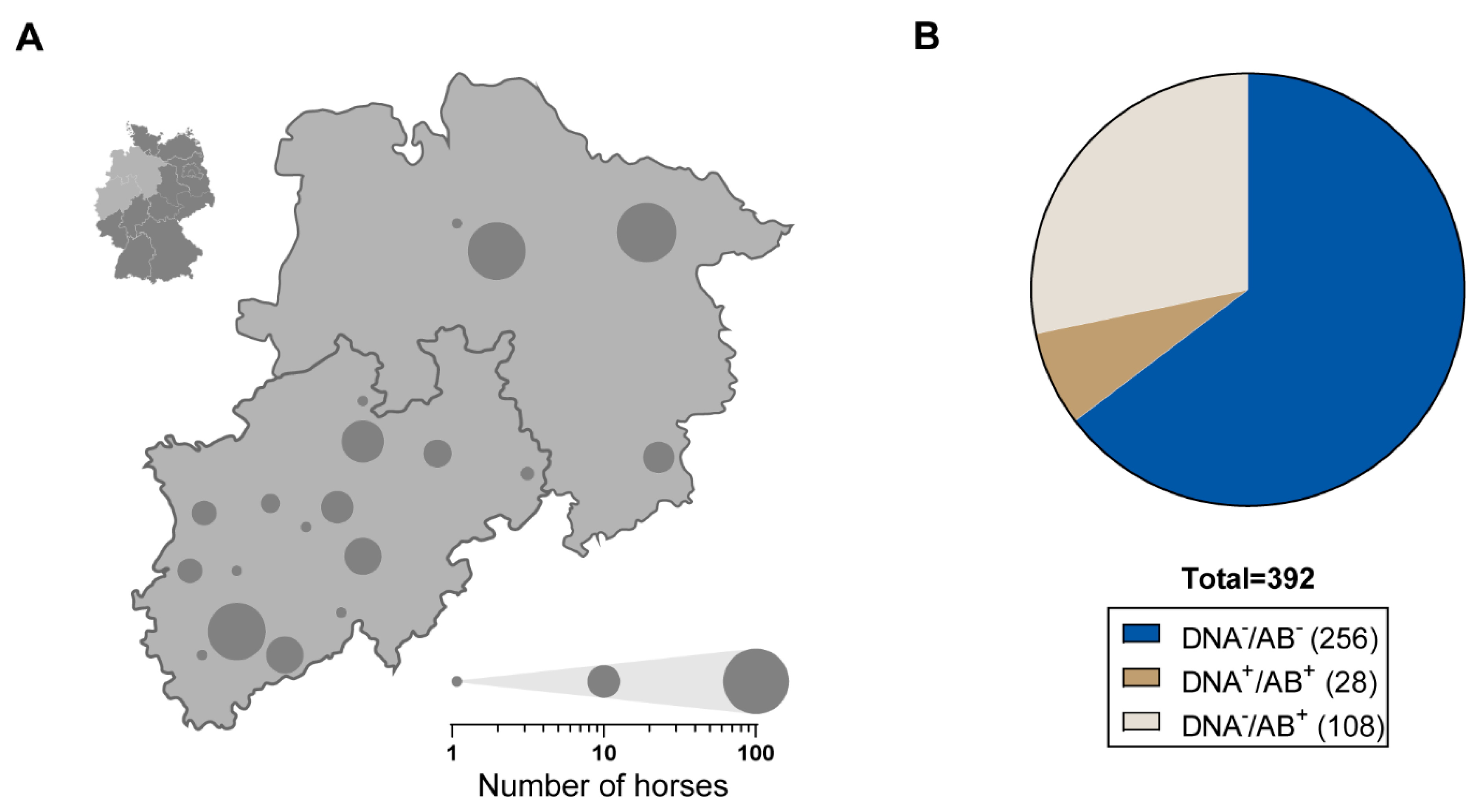
3.1. Frequent Occurrence of EqPV-H among German Thoroughbreds
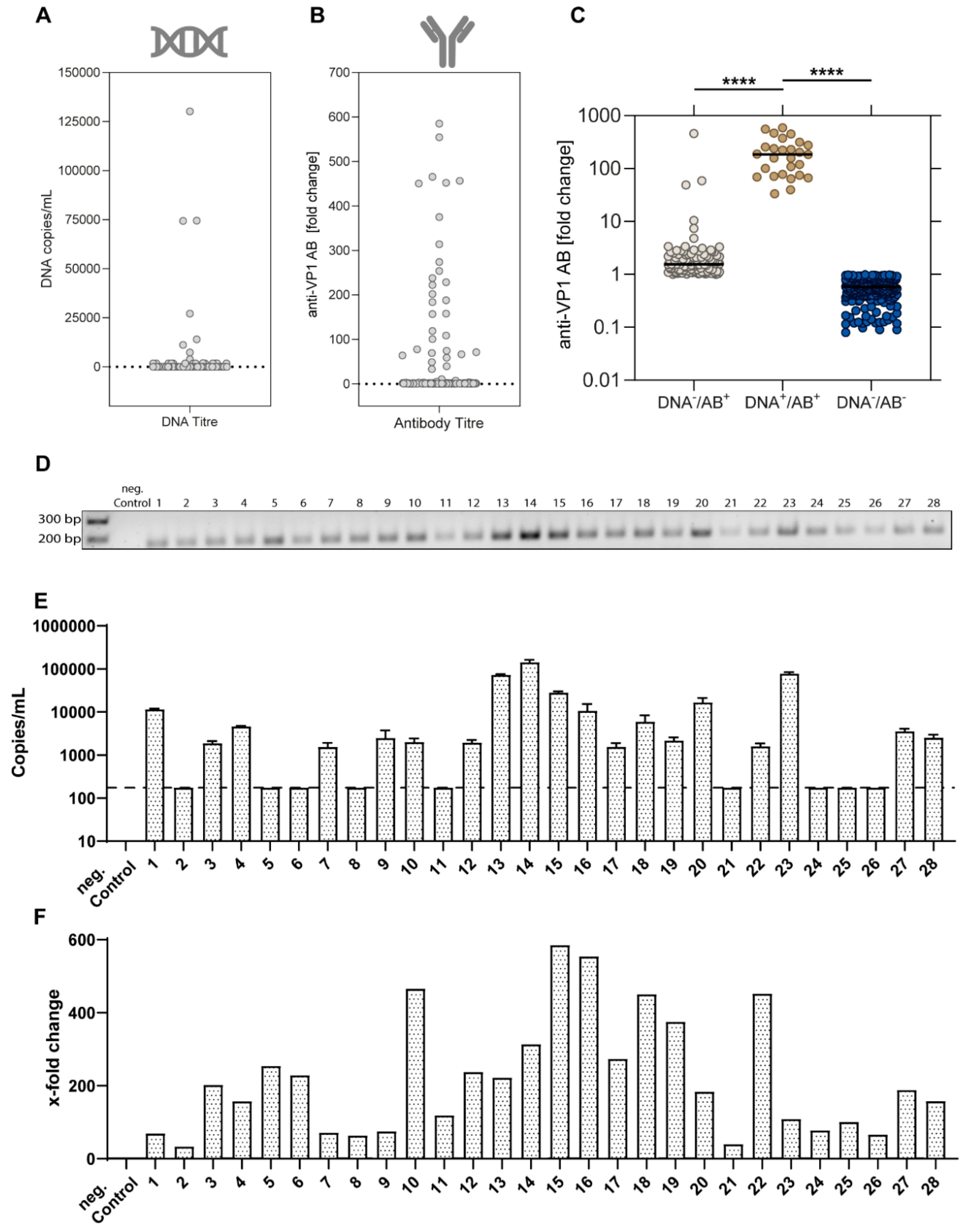
3.2. Viral Characteristics of EqPV-H-Positive Horses
3.3. Age and Breeding History are Potential Risk Factors of EqPV-H Infection in Thoroughbreds
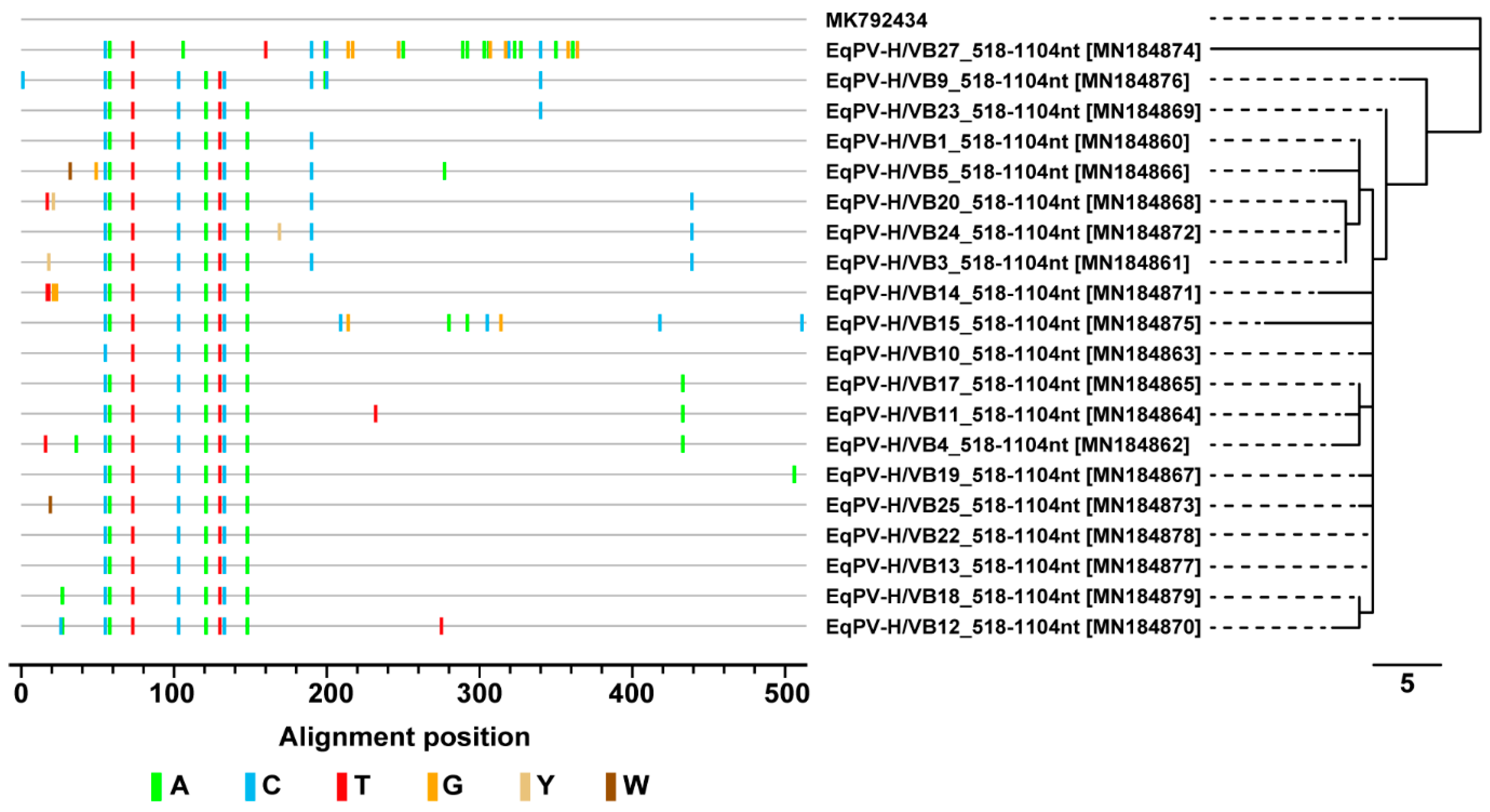
3.4. Sequence and Phylogenetic Analysis of EqPV-H Detected in Thoroughbreds
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sturgeon, B. Theiler’s disease. Vet. Rec. 2017, 180, 14–15. [Google Scholar] [CrossRef] [PubMed]
- Theiler, A. Acute Liver-Atrophy and Parenchymatous Hepatitis in Horses. In 5th and 6th Repts. of the Director of Veterinary Research; Dept. of Agriculture, Union of South Africa: Pretoria, South Africa, 1918; pp. 7–164. [Google Scholar]
- Panciera, R.J. Serum hepatitis in the horse. J. Am. Vet. Med. Assoc. 1969, 155, 408–410. [Google Scholar] [PubMed]
- Guglick, M.A.; MacAllister, C.G.; Ely, R.W.; Edwards, W.C. Hepatic disease associated with administration of tetanus antitoxin in eight horses. J. Am. Vet. Med. Assoc. 1995, 206, 1737–1740. [Google Scholar] [PubMed]
- Aleman, M.; Nieto, J.E.; Carr, E.A.; Carlson, G.P. Serum Hepatitis Associated with Commercial Plasma Transfusion in Horses. J. Vet. Intern. Med. 2005, 19, 120–122. [Google Scholar] [CrossRef] [PubMed]
- Chandriani, S.; Skewes-Cox, P.; Zhong, W.; Ganem, D.E.; Divers, T.J.; van Blaricum, A.J.; Tennant, B.C.; Kistler, A.L. Identification of a previously undescribed divergent virus from the Flaviviridae family in an outbreak of equine serum hepatitis. Proc. Natl. Acad. Sci. USA 2013, 110, E1407–E1415. [Google Scholar] [CrossRef]
- Tomlinson, J.E.; Tennant, B.C.; Struzyna, A.; Mrad, D.; Browne, N.; Whelchel, D.; Johnson, P.J.; Jamieson, C.; Löhr, C.V.; Bildfell, R.; et al. Viral testing of 10 cases of Theiler’s disease and 37 in-contact horses in the absence of equine biologic product administration, A prospective study (2014–2018). J. Vet. Intern. Med. 2019, 33, 258–265. [Google Scholar] [CrossRef]
- Tomlinson, J.E.; van de Walle, G.R.; Divers, T.J. What Do We Know About Hepatitis Viruses in Horses? The Veterinary clinics of North America. Equine Pract. 2019, 35, 351–362. [Google Scholar] [CrossRef]
- Cotmore, S.F.; Agbandje-McKenna, M.; Chiorini, J.A.; Mukha, D.V.; Pintel, D.J.; Qiu, J.; Soderlund-Venermo, M.; Tattersall, P.; Tijssen, P.; Gatherer, D.; et al. The family Parvoviridae. Arch. Virol. 2014, 159, 1239–1247. [Google Scholar] [CrossRef]
- Palinski, R.M.; Mitra, N.; Hause, B.M. Discovery of a novel Parvovirinae virus, porcine parvovirus 7, by metagenomic sequencing of porcine rectal swabs. Virus Genes 2016, 52, 564–567. [Google Scholar] [CrossRef]
- Lu, G.; Sun, L.; Ou, J.; Xu, H.; Wu, L.; Li, S. Identification and genetic characterization of a novel parvovirus associated with serum hepatitis in horses in China. Emerg. Microbes Infect. 2018, 7, 170. [Google Scholar] [CrossRef]
- Wong, F.C.; Spearman, J.G.; Smolenski, M.A.; Loewen, P.C. Equine Parvovirus: Initial Isolation and Partial Characterization. Can. J. Comp. Med. 1985, 49, 50–54. [Google Scholar] [PubMed]
- Li, L.; Giannitti, F.; Low, J.; Keyes, C.; Ullmann, L.S.; Deng, X.; Aleman, M.; Pesavento, P.A.; Pusterla, N.; Delwart, E. Exploring the virome of diseased horses. J. Gen. Virol. 2015, 96, 2721–2733. [Google Scholar] [CrossRef] [PubMed]
- Tomlinson, J.E.; Kapoor, A.; Kumar, A.; Tennant, B.C.; Laverack, M.A.; Beard, L.; Delph, K.; Davis, E.; Schott Ii, H.; Lascola, K.; et al. Viral testing of 18 consecutive cases of equine serum hepatitis, A prospective study (2014–2018). J. Vet. Intern. Med. 2019, 33, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Divers, T.J.; Tennant, B.C.; Kumar, A.; McDonough, S.; Cullen, J.; Bhuva, N.; Jain, K.; Chauhan, L.S.; Scheel, T.K.H.; Lipkin, W.I.; et al. New Parvovirus Associated with Serum Hepatitis in Horses after Inoculation of Common Biological Product. Emerg. Infect. Dis. 2018, 24, 303–310. [Google Scholar] [CrossRef]
- Divers, T.J.; Tomlinson, J.E. Theiler’s disease. Equine Vet. Educ. 2019, 19, 120. [Google Scholar] [CrossRef]
- Meister, T.L.; Tegtmeyer, B.; Postel, A.; Cavalleri, J.-M.V.; Todt, D.; Stang, A.; Steinmann, E. Equine Parvovirus-Hepatitis Frequently Detectable in Commercial Equine Serum Pools. Viruses 2019, 11, 461. [Google Scholar] [CrossRef]
- Reichert, C.; Campe, A.; Walter, S.; Pfaender, S.; Welsch, K.; Ruddat, I.; Sieme, H.; Feige, K.; Steinmann, E.; Cavalleri, J.M.V. Frequent occurrence of nonprimate hepacivirus infections in Thoroughbred breeding horses—A cross-sectional study for the occurrence of infections and potential risk factors. Vet. Microbiol. 2017, 203, 315–322. [Google Scholar] [CrossRef]
- Burbelo, P.D.; Ching, K.H.; Klimavicz, C.M.; Iadarola, M.J. Antibody profiling by Luciferase Immunoprecipitation Systems (LIPS). J. Vis. Exp. JoVE 2009, e1549. [Google Scholar] [CrossRef]
- Burbelo, P.D.; Dubovi, E.J.; Simmonds, P.; Medina, J.L.; Henriquez, J.A.; Mishra, N.; Wagner, J.; Tokarz, R.; Cullen, J.M.; Iadarola, M.J.; et al. Serology-enabled discovery of genetically diverse hepaciviruses in a new host. J. Virol. 2012, 86, 6171–6178. [Google Scholar] [CrossRef]
- Pfaender, S.; Cavalleri, J.M.V.; Walter, S.; Doerrbecker, J.; Campana, B.; Brown, R.J.P.; Burbelo, P.D.; Postel, A.; Hahn, K.; Anggakusuma Riebesehl, N.; et al. Clinical course of infection and viral tissue tropism of hepatitis C virus-like nonprimate hepaciviruses in horses. Hepatol. (Baltim. Md.) 2015, 61, 447–459. [Google Scholar] [CrossRef]
- Keele, B.F.; Giorgi, E.E.; Salazar-Gonzalez, J.F.; Decker, J.M.; Pham, K.T.; Salazar, M.G.; Sun, C.; Grayson, T.; Wang, S.; Li, H.; et al. Identification and characterization of transmitted and early founder virus envelopes in primary HIV-1 infection. Proc. Natl. Acad. Sci. USA 2008, 105, 7552–7557. [Google Scholar] [CrossRef] [PubMed]
- Grandin, T. Assessment of stress during handling and transport. J. Anim. Sci. 1997, 75, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.L.; Jones, J.H.; Hornof, W.J.; Miles, J.A.; Longworth, K.E.; Willits, N.H. Effects of road transport on indices of stress in horses. Equine Vet. J. 1996, 28, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.; Biau, S.; Möstl, E.; Becker-Birck, M.; Morillon, B.; Aurich, J.; Faure, J.-M.; Aurich, C. Changes in cortisol release and heart rate variability in sport horses during long-distance road transport. Domest. Anim. Endocrinol. 2010, 38, 179–189. [Google Scholar] [CrossRef]
- Alexander, S.L.; Irvine, C.H. The effect of social stress on adrenal axis activity in horses: The importance of monitoring corticosteroid-binding globulin capacity. J. Endocrinol. 1998, 157, 425–432. [Google Scholar] [CrossRef]
- Chucri, T.M.; Monteiro, J.M.; Lima, A.R.; Salvadori, M.L.B.; Kfoury, J.R.; Miglino, M.A. A review of immune transfer by the placenta. J. Reprod. Immunol. 2010, 87, 14–20. [Google Scholar] [CrossRef]
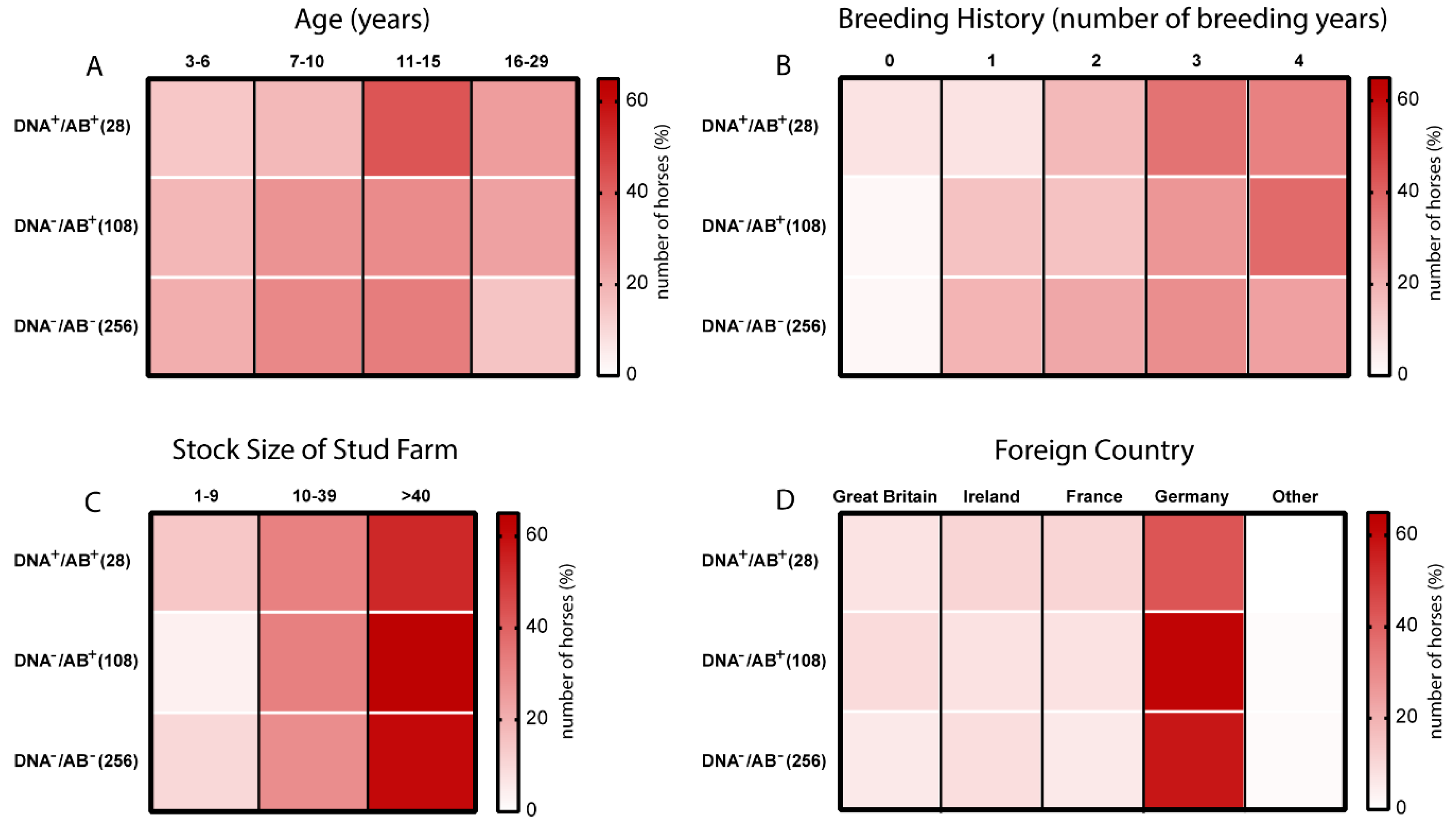
| Variables (n) | State of EqPV-H Infection | |||||
|---|---|---|---|---|---|---|
| DNA−/AB+ (108) | DNA+/AB+ (27) * | DNA−/AB− (256) | ||||
| n | % | n | % | n | % | |
| GENDER ** | ||||||
| Mare (380) | 106 | 98.15 | 26 | 92.86 | 248 | 96.88 |
| Stallion (10) | 2 | 1.85 | 8 | 3.12 | ||
| AGE | ||||||
| 3–6 years (78) | 20 | 18.52 | 4 | 14.29 | 54 | 21.09 |
| 7–10 years (113) | 30 | 27.78 | 5 | 17.86 | 78 | 30.47 |
| 11–15 years (129) | 32 | 29.6 | 12 | 42.86 | 85 | 33.20 |
| 16–29 years (72) | 26 | 24.07 | 7 | 25 | 39 | 15.23 |
| NUMBER OF BREEDING YEARS | ||||||
| Not covered (9) | 2 | 1.85 | 2 | 7.14 | 5 | 1.95 |
| 1 breeding year (68) | 17 | 15.74 | 2 | 7.14 | 49 | 19.14 |
| 2 breeding year (80) | 17 | 15.74 | 5 | 17.86 | 58 | 22.66 |
| 3 breeding year (113) | 29 | 26.85 | 10 | 35.71 | 74 | 28.91 |
| 4 breeding year (112) | 41 | 37.96 | 9 | 32.14 | 62 | 24.22 |
| STOCK SIZE | ||||||
| 1–9 horses (34) | 4 | 3.70 | 4 | 14.29 | 26 | 10.16 |
| 10–39 horses (119) | 35 | 32.41 | 9 | 32.14 | 75 | 29.30 |
| >40 horses (239) | 69 | 63.89 | 15 | 53.57 | 155 | 60.55 |
| FOREIGN COUNTRY ** | ||||||
| Great Britain (27) | 10 | 9.26 | 2 | 7.14 | 15 | 5.86 |
| Ireland (33) | 8 | 7.41 | 3 | 10.71 | 22 | 8.59 |
| France (26) | 8 | 7.41 | 3 | 10.71 | 15 | 5.856 |
| Germany (227) | 67 | 62.04 | 12 | 42.86 | 148 | 57.81 |
| Other (4) | 1 | 0.93 | 0 | 0 | 3 | 1.172 |
| EqHV-COINFECTION ** | ||||||
| RNA−/AB+ (196) | 60 | 55.57 | 12 | 44.45 | 124 | 48.44 |
| RNA+/AB+ (41) | 8 | 7.41 | 4 | 14.82 | 29 | 11.33 |
| RNA−/AB− (152) | 40 | 37.04 | 10 | 37.04 | 102 | 39.84 |
| Sample ID | EqPV Sequence Name | NCBI Accession Number |
|---|---|---|
| 1 | EqPV-H/VB1_518-1104nt | MN184860 |
| 3 | EqPV-H/VB3_518-1104nt | MN184861 |
| 4 | EqPV-H/VB4_518-1104nt | MN184862 |
| 10 | EqPV-H/VB10_518-1104nt | MN184863 |
| 11 | EqPV-H/VB11_518-1104nt | MN184864 |
| 17 | EqPV-H/VB17_518-1104nt | MN184865 |
| 5 | EqPV-H/VB5_518-1104nt | MN184866 |
| 19 | EqPV-H/VB19_518-1104nt | MN184867 |
| 20 | EqPV-H/VB20_518-1104nt | MN184868 |
| 23 | EqPV-H/VB23_518-1104nt | MN184869 |
| 12 | EqPV-H/VB12_518-1104nt | MN184870 |
| 14 | EqPV-H/VB14_518-1104nt | MN184871 |
| 24 | EqPV-H/VB24_518-1104nt | MN184872 |
| 25 | EqPV-H/VB25_518-1104nt | MN184873 |
| 27 | EqPV-H/VB27_518-1104nt | MN184874 |
| 15 | EqPV-H/VB15_518-1104nt | MN184875 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meister, T.L.; Tegtmeyer, B.; Brüggemann, Y.; Sieme, H.; Feige, K.; Todt, D.; Stang, A.; Cavalleri, J.-M.V.; Steinmann, E. Characterization of Equine Parvovirus in Thoroughbred Breeding Horses from Germany. Viruses 2019, 11, 965. https://doi.org/10.3390/v11100965
Meister TL, Tegtmeyer B, Brüggemann Y, Sieme H, Feige K, Todt D, Stang A, Cavalleri J-MV, Steinmann E. Characterization of Equine Parvovirus in Thoroughbred Breeding Horses from Germany. Viruses. 2019; 11(10):965. https://doi.org/10.3390/v11100965
Chicago/Turabian StyleMeister, Toni Luise, Birthe Tegtmeyer, Yannick Brüggemann, Harald Sieme, Karsten Feige, Daniel Todt, Alexander Stang, Jessika-M.V. Cavalleri, and Eike Steinmann. 2019. "Characterization of Equine Parvovirus in Thoroughbred Breeding Horses from Germany" Viruses 11, no. 10: 965. https://doi.org/10.3390/v11100965
APA StyleMeister, T. L., Tegtmeyer, B., Brüggemann, Y., Sieme, H., Feige, K., Todt, D., Stang, A., Cavalleri, J.-M. V., & Steinmann, E. (2019). Characterization of Equine Parvovirus in Thoroughbred Breeding Horses from Germany. Viruses, 11(10), 965. https://doi.org/10.3390/v11100965






