Equine Rhinitis A Virus Infection in Thoroughbred Racehorses—A Putative Role in Poor Performance?
Abstract
1. Introduction
2. Materials and Methods
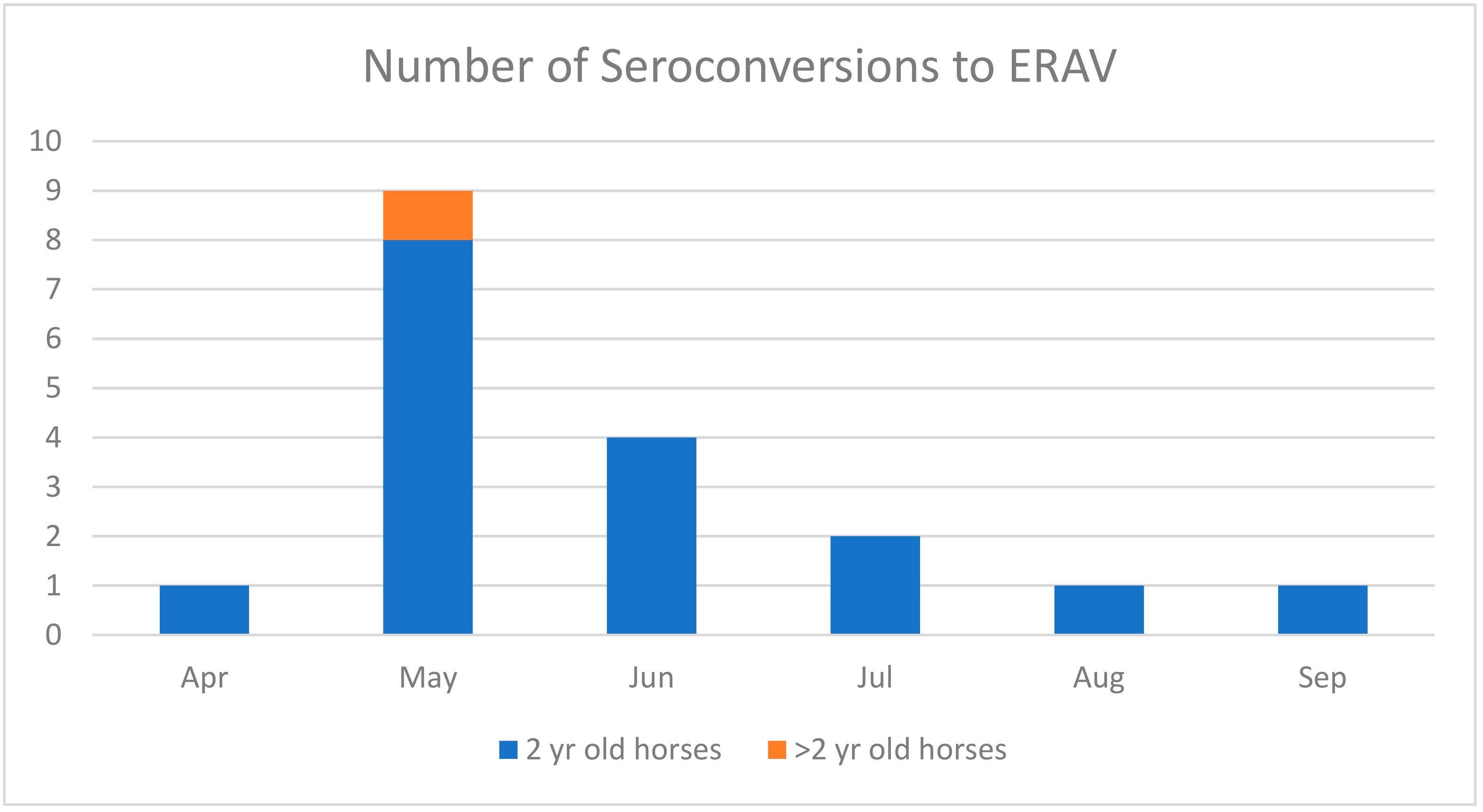
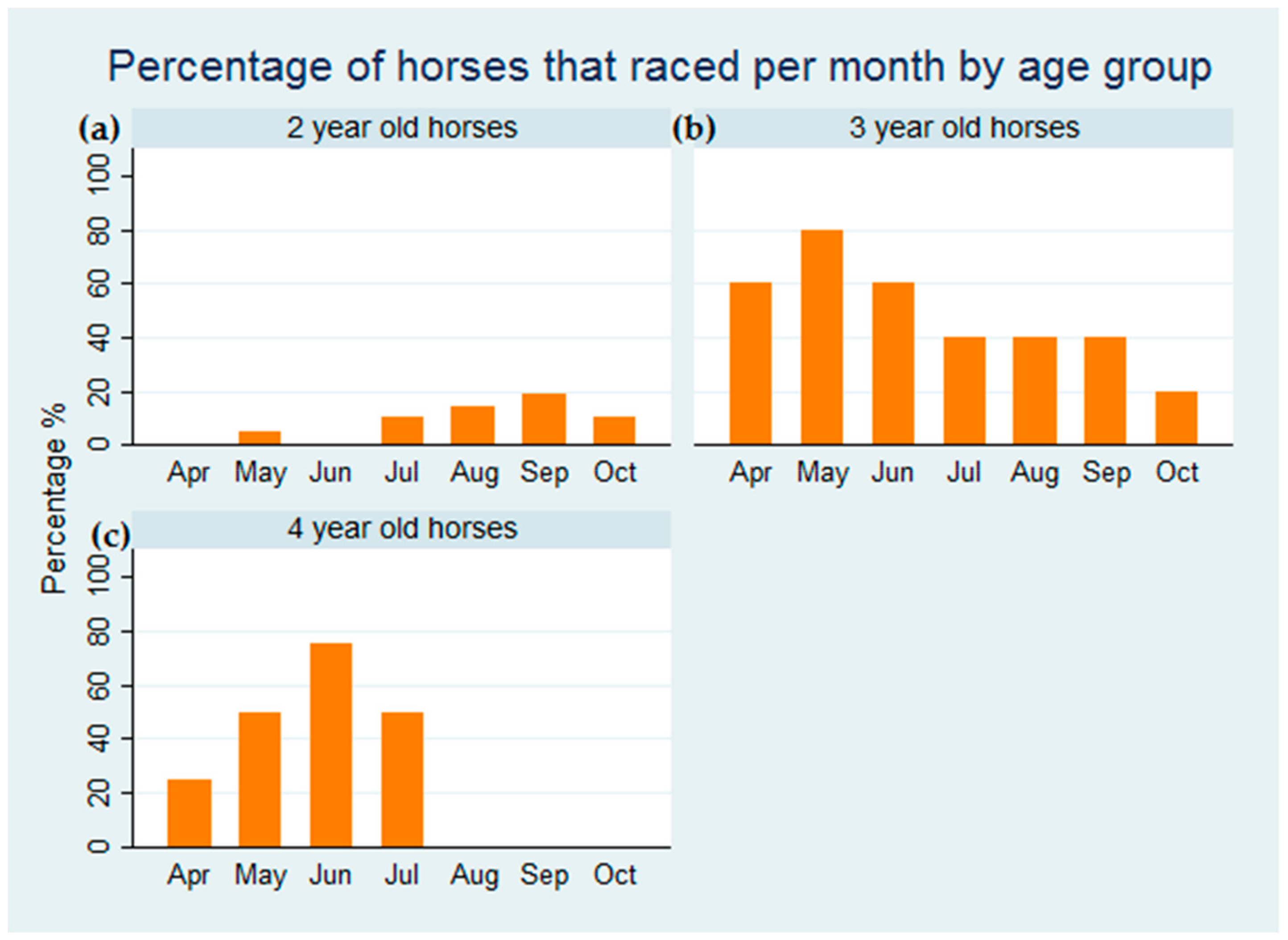
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wilsher, S.; Allen, W.R.; Wood, J.L. Factors associated with failure of thoroughbred horses to train and race. Equine Vet. J. 2006, 38, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Cullinane, A.; Newton, J.R. Equine influenza-A global perspective. Vet. Microbiol. 2013, 167, 205–214. [Google Scholar] [CrossRef] [PubMed]
- Newton, J.R.; Daly, J.M.; Spencer, L.; Mumford, J.A. Description of the outbreak of equine influenza (H3N8) in the United Kingdom in 2003, during which recently vaccinated horses in Newmarket developed respiratory disease. Vet. Rec. 2006, 158, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Gildea, S.; Arkins, S.; Cullinane, A. Management and environmental factors involved in equine influenza outbreaks in Ireland 2007-2010. Equine Vet. J. 2011, 43, 608–617. [Google Scholar] [CrossRef] [PubMed]
- Gildea, S.; Fitzpatrick, D.A.; Cullinane, A. Epidemiological and virological investigations of equine influenza outbreaks in Ireland (2010-2012). Influenza Other Respir. Viruses 2013, 7 (Suppl. 4), 61–72. [Google Scholar] [CrossRef]
- Willoughby, R.; Ecker, G.; McKee, S.; Riddolls, L.; Vernaillen, C.; Dubovi, E.; Lein, D.; Mahony, J.B.; Chernesky, M.; Nagy, E.; et al. The effects of equine rhinovirus, influenza virus and herpesvirus infection on tracheal clearance rate in horses. Can. J. Vet. Res. 1992, 56, 115–121. [Google Scholar]
- Allen, G.P.; Kydd, J.; Slater, J.D.; Smith, K.C. Equid herpesvirus 1 and equid herpesvirus 4 infections. In Infectious Diseases on Livestock, 2nd ed.; Coetzer, J.A.W., Tustin, R.C., Eds.; Oxford Press: Cape Town, South Africa, 2004; pp. 829–859. [Google Scholar]
- Pusterla, N.; Kass, P.H.; Mapes, S.; Johnson, C.; Barnett, D.C.; Vaala, W.; Gutierrez, C.; McDaniel, R.; Whitehead, B.; Manning, J. Surveillance programme for important equine infectious respiratory pathogens in the USA. Vet. Rec. 2011, 169, 12. [Google Scholar] [CrossRef]
- Burgess, B.A.; Tokateloff, N.; Manning, S.; Lohmann, K.; Lunn, D.P.; Hussey, S.B.; Morley, P.S. Nasal shedding of equine herpesvirus-1 from horses in an outbreak of equine herpes myeloencephalopathy in Western Canada. J. Vet. Intern. Med. 2012, 26, 384–392. [Google Scholar] [CrossRef]
- Carman, S.; Rosendal, S.; Huber, L.; Gyles, C.; McKee, S.; Willoughby, R.A.; Dubovi, E.; Thorsen, J.; Lein, D. Infectious agents in acute respiratory disease in horses in Ontario. J. Vet. Diagn. Invest. 1997, 9, 17–23. [Google Scholar] [CrossRef]
- Li, F.; Drummer, H.E.; Ficorilli, N.; Studdert, M.J.; Crabb, B.S. Identification of noncytopathic equine rhinovirus 1 as a cause of acute febrile respiratory disease in horses. J. Clin. Microbiol. 1997, 35, 937–943. [Google Scholar]
- Plummer, G. An equine respiratory virus with enterovirus properties. Nature 1962, 195, 519–520. [Google Scholar] [CrossRef] [PubMed]
- Studdert, M.; Gleeson, L. Isolation and characterisation of an equine rhinovirus. Zentralbl Veterinarmed B 1978, 25, 225–237. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Mendez, A.; Viel, L.; Hewson, J.; Doig, P.; Carman, S.; Chambers, T.; Tiwari, A.; Dewey, C. Surveillance of equine respiratory viruses in Ontario. Can. J. Vet. Res. 2010, 74, 271–278. [Google Scholar] [PubMed]
- Ditchfield, J.; Macpherson, L. Properties and classification of 2 new rhinoviruses recovered from horses in Toronto. Cornell. Vet. 1965, 55, 181–189. [Google Scholar]
- Sugiura, T.; Matsumura, T.; Imagawa, H.; Fukunaga, Y. A seven year serological study of viral agents causing respiratory infection with pyrexia among racehorses in Japan. In Equine Infectious Diseases V; Powell, D.G., Ed.; University of Kentucky Publications: Lexington, NY, USA, 1988; pp. 258–261. [Google Scholar]
- McCollum, W.; Timoney, P. Studies on the seroprevalence and frequency of equine rhinovirus-I and-II infection in normal horse urine. In Equine Infectious Diseases VI; Plowright, W., Rossdale, P.D., Wade, J.F., Eds.; R&W Publications: Newmarket, UK, 1992; pp. 83–87. [Google Scholar]
- Black, W.D.; Wilcox, R.S.; Stevenson, R.A.; Hartley, C.A.; Ficorilli, N.P.; Gilkerson, J.R.; Studdert, M.J. Prevalence of serum neutralising antibody to equine rhinitis A virus (ERAV), equine rhinitis B virus 1 (ERBV1) and ERBV2. Vet. Microbiol. 2007, 119, 65–71. [Google Scholar] [CrossRef]
- Klaey, M.; Sanchez-Higgins, M.; Leadon, D.P.; Cullinane, A.; Straub, R.; Gerber, H. Field case study of equine rhinovirus 1 infection: clinical signs and clinicopathology. Equine Vet. J. 1998, 30, 267–269. [Google Scholar] [CrossRef]
- Powell, D.G.; Burrows, R.; Spooner, P.; Mumford, J.; Thomson, G. A study of infectious respiratory disease among horses in Great Britain, 1971-1976. In Equine Infectious Diseases IV; Bryans, J.T., Gerber, H., Eds.; Veterinary Publications: Princeton, NJ, USA, 1978; pp. 451–459. [Google Scholar]
- Burrell, M.H.; Wood, J.L.; Whitwell, K.E.; Chanter, N.; Mackintosh, M.E.; Mumford, J.A. Respiratory disease in thoroughbred horses in training: the relationships between disease and viruses, bacteria and environment. Vet. Rec. 1996, 139, 308–313. [Google Scholar] [CrossRef]
- Cardwell, J.; Smith, K.; Wood, J.; Newton, J. A longitudinal study of respiratory infections in British National Hunt racehorses. Vet. Rec. 2013, 172, 637. [Google Scholar] [CrossRef]
- Wood, J.L.; Newton, J.R.; Chanter, N.; Mumford, J.A. Association between respiratory disease and bacterial and viral infections in British racehorses. J. Clin. Microbiol. 2005, 43, 120–126. [Google Scholar] [CrossRef]
- Thomson, G.R.; Mumford, J.A.; Campbell, J.; Griffiths, L.; Clapham, P. Serological detection of equid herpesvirus 1 infections of the respiratory tract. Equine Vet. J. 1976, 8, 58–65. [Google Scholar] [CrossRef]
- Cullinane, A.; Weld, J.; Osborne, M.; Nelly, M.; McBride, C.; Walsh, C. Field studies on equine influenza vaccination regimes in thoroughbred foals and yearlings. Vet. J. 2001, 161, 174–185. [Google Scholar] [CrossRef] [PubMed]
- Hofer, B.; Steck, F.; Gerber, H.; Löhrer, J.; Nicolet, J.; Paccaud, M. An Investigation of the Etiology of Viral Respiratory Disease in a Remount Depot. In Equine Infectious Diseases III; Bryans, J.T., Gerber, H., Eds.; Karger: Basel, Switzerland, 1973; pp. 527–545. [Google Scholar]
- Burrows, R. Equine rhinovirus and adenovirus infections. In Proceedings of the 24th Annual Convention of the American Association of Equine Practitioners, St. Louis, MO, USA, 1978; pp. 299–306. [Google Scholar]
- Plummer, G. Studies on an equine respiratory virus. Vet. Rec. 1962, 74, 967–970. [Google Scholar]
- Diaz-Mendez, A.; Hewson, J.; Shewen, P.; Nagy, E.; Viel, L. Characteristics of respiratory tract disease in horses inoculated with equine rhinitis A virus. Am. J. Vet. Res. 2014, 75, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Quinlivan, M.; Maxwell, G.; Lyons, P.; Arkins, S.; Cullinane, A. Real-time RT-PCR for the detection and quantitative analysis of equine rhinitis viruses. Equine Vet. J. 2010, 42, 98–104. [Google Scholar] [CrossRef] [PubMed]
- Lynch, S.E.; Gilkerson, J.R.; Symes, S.J.; Huang, J.A.; Hartley, C.A. Persistence and chronic urinary shedding of the aphthovirus equine rhinitis A virus. Comp. Immunol. Microbiol. Infect. Dis. 2013, 36, 95–103. [Google Scholar] [CrossRef] [PubMed]
- De Boer, G.F.; Osterhaus, A.D.; van Oirschot, J.T.; Wemmenhove, R. Prevalence of antibodies to equine viruses in the Netherlands. Tijdschr. Diergeneeskd. 1979, 104, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Burrows, R.; Goodridge, D. Observations of picornavirus, adenovirus, and equine herpesvirus infections in the Pirbright pony herd. In Equine Infectious Diseases IV; Bryans, J.T., Gerber, H., Eds.; Veterinary Publications: Princeton, NJ, USA, 1978; pp. 155–164. [Google Scholar]
- Burrows, R. Equine rhinoviruses. In Proceedings of the Equine Infectious Diseases II, Paris, France, 16–18 June 1969; Bryans, J.T., Gerber, H., Eds.; Karger: Basel, Switzerland, 1970; pp. 154–164. [Google Scholar]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Back, H.; Weld, J.; Walsh, C.; Cullinane, A. Equine Rhinitis A Virus Infection in Thoroughbred Racehorses—A Putative Role in Poor Performance? Viruses 2019, 11, 963. https://doi.org/10.3390/v11100963
Back H, Weld J, Walsh C, Cullinane A. Equine Rhinitis A Virus Infection in Thoroughbred Racehorses—A Putative Role in Poor Performance? Viruses. 2019; 11(10):963. https://doi.org/10.3390/v11100963
Chicago/Turabian StyleBack, Helena, John Weld, Cathal Walsh, and Ann Cullinane. 2019. "Equine Rhinitis A Virus Infection in Thoroughbred Racehorses—A Putative Role in Poor Performance?" Viruses 11, no. 10: 963. https://doi.org/10.3390/v11100963
APA StyleBack, H., Weld, J., Walsh, C., & Cullinane, A. (2019). Equine Rhinitis A Virus Infection in Thoroughbred Racehorses—A Putative Role in Poor Performance? Viruses, 11(10), 963. https://doi.org/10.3390/v11100963




