Radial Growth Behavior of Pines on Romanian Degraded Lands
Abstract
1. Introduction
2. Materials and Methods
2.1. Erosion Background
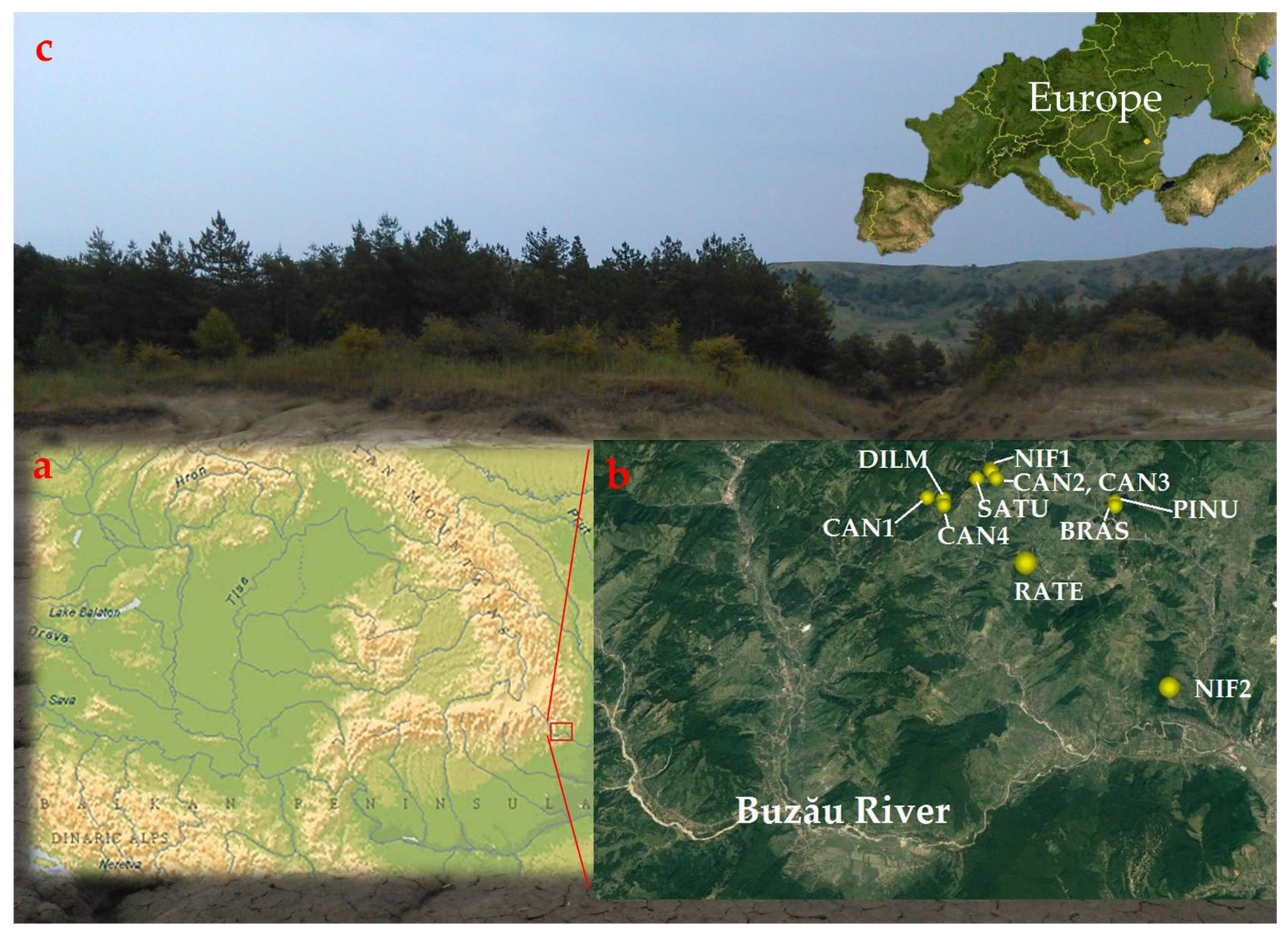
2.2. Study Stands
2.3. Sampling Design and Data Compilation
2.4. Dendroecological Study
2.5. Statistical Analyses
3. Results
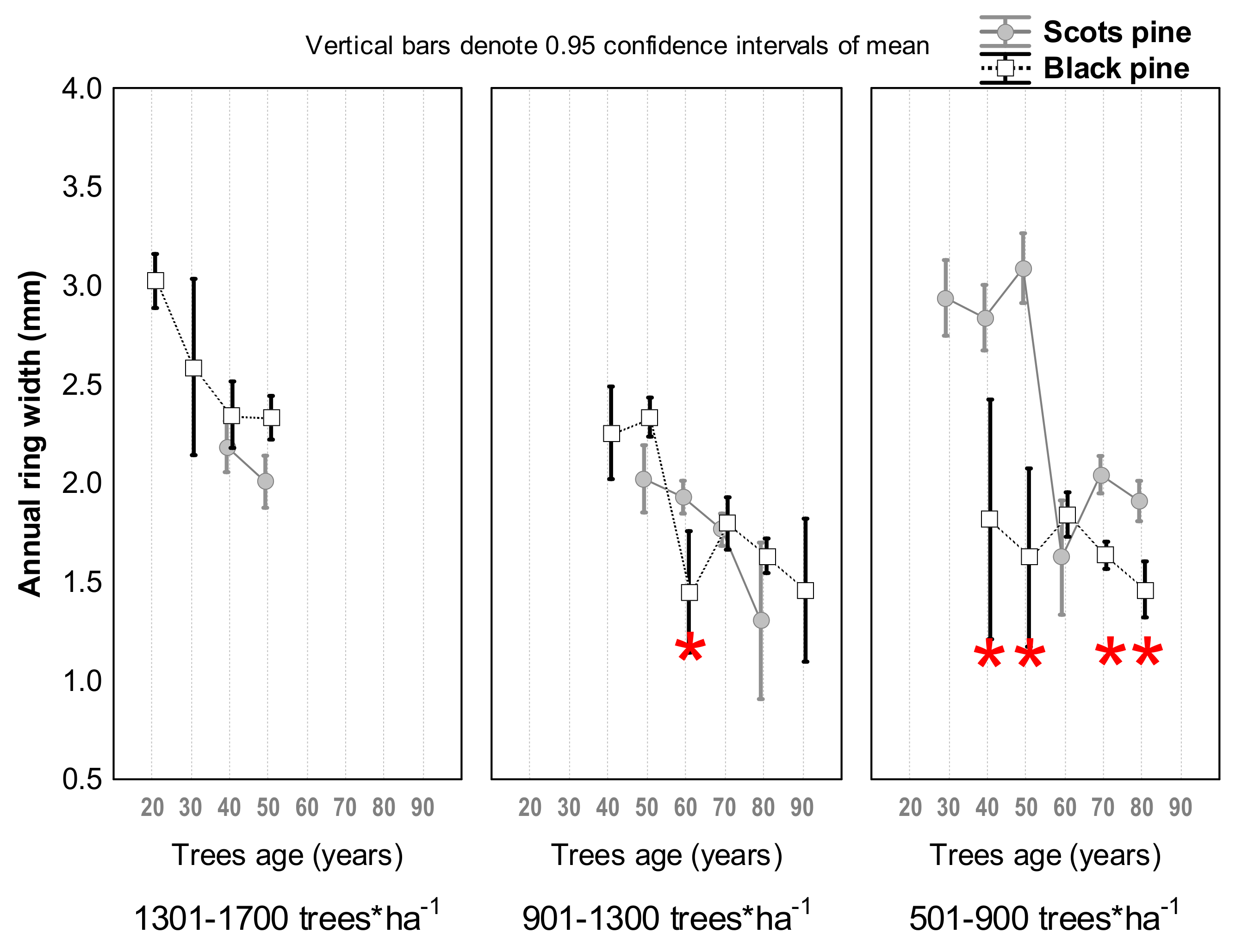
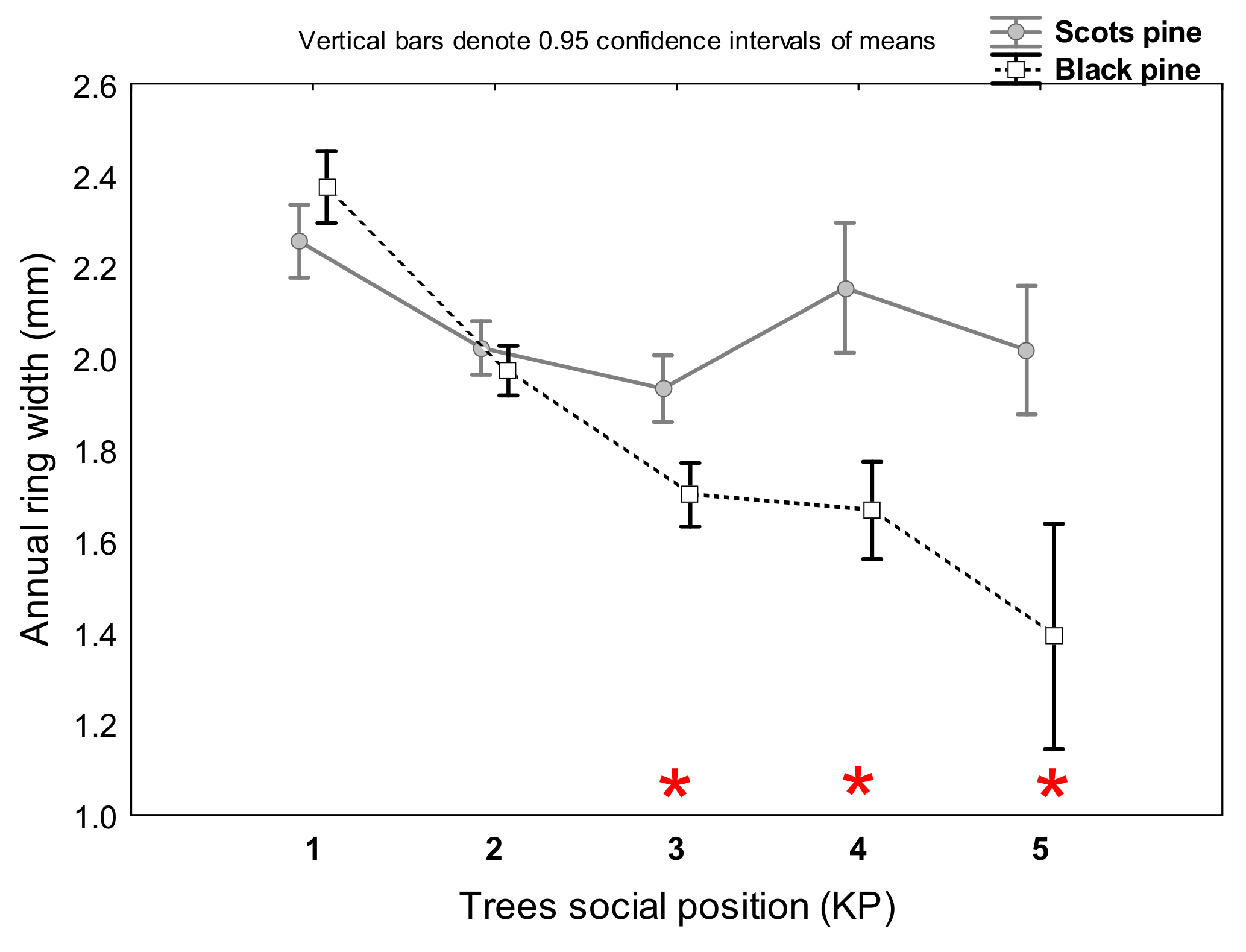
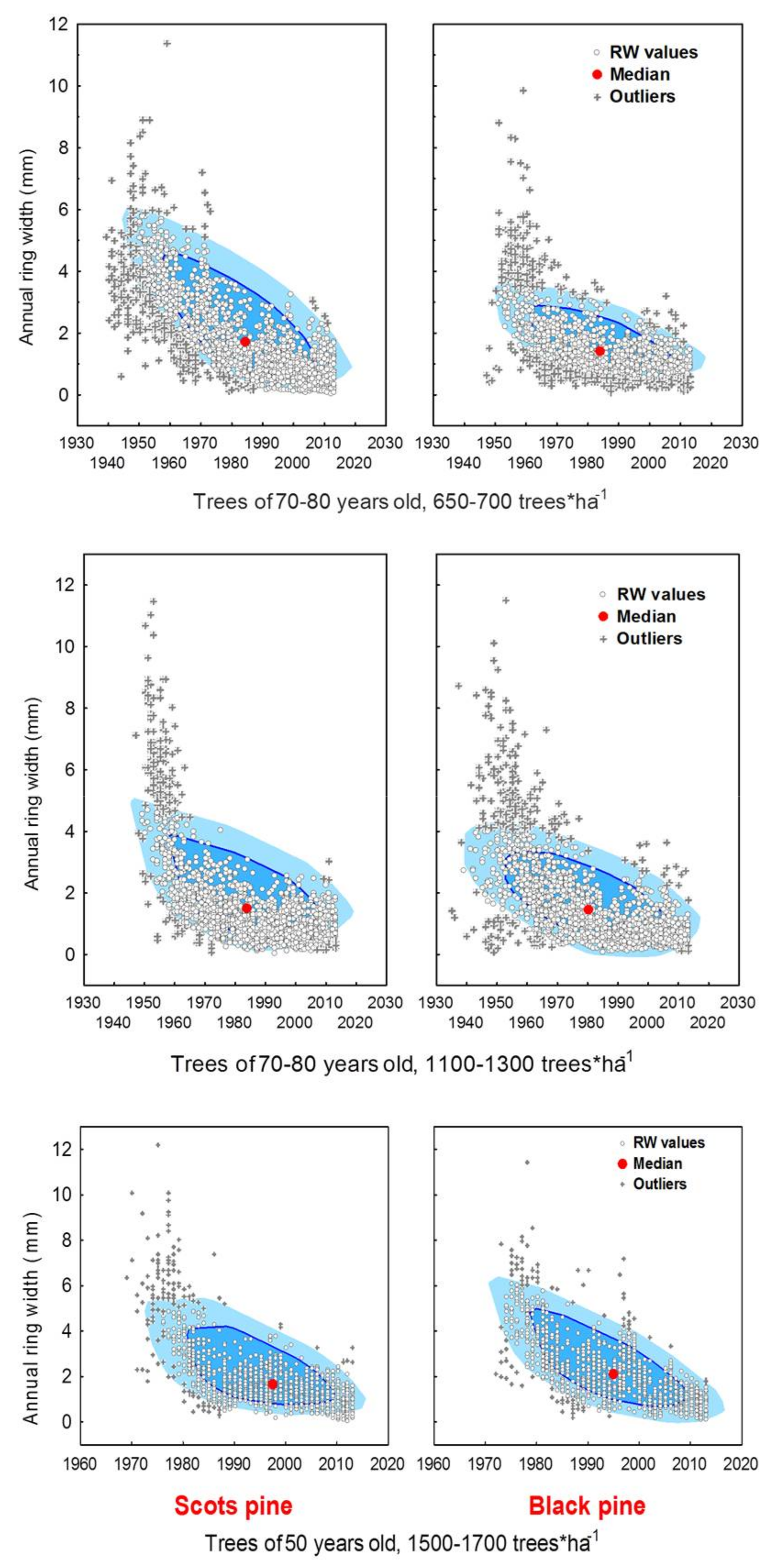
3.1. Differences in Tree Growth between the Two Pine Species
3.2. Climate Share in Radial Growth
3.3. Logging Effects
4. Discussion
4.1. Scots Pine vs. Black Pine in Terms of Radial Growth and Survival
4.2. Age-Dependency against Climate-Related Trends in Growth
4.3. The Management of the Ecological Reconstruction on Land Degraded by Pluvial Erosion
4.4. Trees Response to Management
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Roman-Amat, B. Les programmes d’amélioration: Résultats actuels et escomptés: Pin sylvestre (Pinus sylvestris L). Rev. For. Fr. 1986, 38, 132–134. [Google Scholar] [CrossRef]
- Portefaix, C.; Roman-Amat, B. Les programmes d’amélioration: Résultats actuels et escomptés: Pins laricio de Corse et de Calabre (Pinus nigra Arb. ssp. laricio Poiret 1804) (Breeding programmes: Current and anticipated results: Corsican and Calabrian pine). Rev. For. Fr. 1986, 38, 129–131. [Google Scholar]
- Schulze, E.-D.; Beck, E.; Müller-Hohenstein, K. Plant Ecology; Springer: Berlin/Heidelberg, Germany, 2005; p. 702. ISBN 9783540208334. [Google Scholar]
- Vorob’ev, V.N. Pine Forests: Utilization of Their Products; Science Publishers: Enfield, CT, USA, 2007; p. 271. ISBN 9781578085941. [Google Scholar]
- Critchfield, W.B.; Little, E.L. Geographic Distribution of the Pines of the World; U.S. Department of Agriculture, Forest Service: Washington, DC, USA, 1966; Volume 19, p. 116.
- Nys, C.; Bonischot, R.; Gelhaye, D. Réponse d’un peuplement de pin laricio de corse (Pinus nigra espèce laricio) à la fertilisation en Sologne (The response to fertilizers of a Corsican pine (Pinus nigra var. laricio) stand in Sologne). Rev. For. Fr. 1985, 37, 482–486. [Google Scholar]
- Schultz, R.P. Loblolly—The pine for the twenty-first century. New For. 1999, 17, 71–88. [Google Scholar] [CrossRef]
- Baumann, K.; Schneider, B.U.; Marschner, P.; Hüttl, R.F. Seedling biomass and element content of Pinus sylvestris and Pinus nigra grown in sandy substrates with lignite. Geoderma 2006, 136, 573–578. [Google Scholar] [CrossRef]
- Jolyet, A. Lemnul de pin negru (Pinus austriaca). Rev. Păd 1908, 23, 53–55. [Google Scholar]
- Traci, C. Împădurirea Terenurilor Degradate (Afforestation of Degraded Lands); Ceres Publishing House: Bucharest, Romania, 1985; p. 282. [Google Scholar]
- Sanchez, L.G.; Prada, M.A. Los pinos como especies basicas de la restauracion forestal en el medio mediterraneo. Ecología 1993, 7, 113–126. [Google Scholar]
- Madrigal, A. Problématica de la ordenación de masas artificiales en España. Cuad. Soc. Esp. Cienc. For. 1998, 6, 13–20. [Google Scholar]
- Cortina, J.; Amat, B.; Castillo, V.; Fuentes, D.; Maestre, F.T.; Padilla, F.M.; Rojo, L. The restoration of vegetation cover in the semi-arid Iberian southeast. J. Arid Environ. 2011, 75, 1377–1384. [Google Scholar] [CrossRef]
- Kuznetsova, T.; Tilk, M.; Pärn, H.; Lukjanova, A.; Mandre, M. Growth, aboveground biomass, and nutrient concentrationof young Scots pine and longepole pine in oil shale post-mining landscapes in Estonia. Environ. Monit. Assess. 2011, 183, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Ganatsas, P.; Tsitsoni, T.; Tsakaldimi, M.; Zagas, T. Reforestation of degraded Kermes oak shrublands with planted pines: Effects on vegetation cover, species diversity and community structure. New For. 2012, 43, 1–11. [Google Scholar] [CrossRef]
- Ciortuz, I.; Păcurar, V. Ameliorații Silvice; LuxLibris Publishing House: Brașov, Romania, 2004; p. 231. ISBN 9739458130. [Google Scholar]
- Giurgiu, V. Considerații asupra stării pădurilor României I: Declinul suprafeței pădurilor și marginalizarea împădurilor. Rev. Păd 2010, 125, 3–16. [Google Scholar]
- Untaru, E.; Constandache, C.; Nistor, S. Starea actuală și proiecții pentru viitor în privința reconstrucției ecologice prin împădurire a terenurilor degradate din România. (I). Rev. Păd 2012, 127, 28–34. [Google Scholar]
- Constandache, C. Ameliorarea și Refacerea Pinetelor Necorespunzătoare sub Raport Productiv și Protectiv Instalate pe Terenurile Degradate Din Bazinul Hidrografic al Râului Putna. Ph.D. Thesis, Transilvania University of Brașov, Brașov, Romania, 2003. [Google Scholar]
- Traci, C.; Untaru, E. Comportarea și Efectul Ameliorativ și de Consolidare a Culturilor Forestiere de pe Terenurile Degradate din Perimetrele Experimentale; Forest Research and Management Institute Research Paper; Forest Research and Management Institute: Bucharest, Romania, 1986. [Google Scholar]
- Traci, C.; Mușat, I. Folosirea pinului negru și a pinului silvestru la împăduririle terenurilor degradate. Rev. Păd 1955, 70, 211–217. [Google Scholar]
- Bouriaud, O.; Popa, I. Comparative dendroclimatic study of Scots pine, Norway spruce and silver fir in the Vrancea Range, Eastern Carpathians Mountains. Trees 2009, 23, 95–106. [Google Scholar] [CrossRef]
- Sander, D.H. Height-Age Curves for Austrian Pine in Windbreaks on Loess Soils of Nebraska; USDA Forest Service Research Note RM; Rocky Mountain Forest and Range Experiment Station: Fort Collins, CO, USA, 1963; p. 13. [Google Scholar]
- Amodei, T.; Guibal, F.; Fady, B. Relationship between climate and radial growth in black pine (Pinus nigra Arnold ssp. salzmanni (Dunal) Franco) from the south of France. Ann. For. Sci. 2013, 70, 41–47. [Google Scholar]
- Martín-Benito, D.; Cherubini, P.; del Río, M.; Cañellas, I. Growth response to climate and drought in Pinus nigra Arn. trees of different crown classes. Trees 2008, 22, 363–373. [Google Scholar] [CrossRef]
- Ruiz-Labourdette, D.; Génova, M.; Schmitz, M.F. Summer rainfall variability in European Mediterranean mountains from the sixteenth to the twentieth century reconstructed from tree rings. Int. J. Biometeorol. 2014, 58, 1627–1639. [Google Scholar] [CrossRef] [PubMed]
- Sangüesa-Barreda, G.; Camarero, J.J.; Linares, J.C.; Hernández, R.; Oliva, J.; Gazol, A.; González de Andrés, E.; Montes, F.; García-Martín, A.; de la Riva, J. Papel de los factores bióticos y las sequías en el decaimiento del bosque: Aportaciones desde la dendroecología (Role of biotic factors and droughts in the forest decline: Contributions from dendroecology). Rev. Ecosist. 2015, 24, 15–23. [Google Scholar] [CrossRef]
- Rigling, A.; Bräker, O.; Schneider, G.; Schweingruber, F. Intra-annual tree-ring parameters indicating differences in drought stress of Pinus sylvestris forests within the Erico-Pinion in the Valais (Switzerland). Plant Ecol. 2002, 163, 105–121. [Google Scholar] [CrossRef]
- Orwig, D.A.; Abrams, M.D. Variation in radial growth responses to drought among species, site and canopy strata. Trees 1997, 11, 474–484. [Google Scholar] [CrossRef]
- Simionescu, A.; Traci, C.; Frațian, A.; Popescu, T.; Mărcioiu, A. Despre uscarea unor arborete de pin și măsuri de prevenire a extinderii acestui fenomen. Rev. Păd 1963, 78, 442–446. [Google Scholar]
- Ceuca, G.; Constantinescu, N.; Drocan, R.; Georgescu, C.C.; Nițu, G.; Tomescu, A. Studiu privind condițiile de vegetație ale arboretelor de pin cu fenomene de uscare. Ann. For. Res. 1957, 18, 204–249. [Google Scholar]
- Untaru, E. Premise privind împădurirea terenurilor degradate în condițiile schimbărilor climatice generate de încălzirea globală. Rev. Păd 2010, 125, 20–25. [Google Scholar]
- Lebourgeois, F. Climatic signals in earlywood, latewood and total ring width of Corsican pine from western France. Ann. For. Sci. 2000, 57, 155–164. [Google Scholar] [CrossRef]
- Waring, R.H. Characteristics of trees predisposed to die. BioScience 1987, 37, 569–574. [Google Scholar] [CrossRef]
- Moreno-Gutiérrez, C.; Battipaglia, G.; Cherubini, P.; Saurer, M.; Nicolás, E.; Contreras, S.; Querejeta, J. Stand structure modulates the long-term vulnerability of Pinus halepensis to climatic drought in a semiarid Mediterranean ecosystem. Plant Cell Environ. 2012, 35, 1026–1039. [Google Scholar] [CrossRef] [PubMed]
- Schweingruber, F.H. Wood Structure and Environment, 1st ed.; Springer: Berlin/Heidelberg, Germany, 2007; p. 279. ISBN 978-3-540-48548-3. [Google Scholar]
- Bednarz, B.; Kochanowski, D. Radial growth response of Scots pine (Pinus sylvestris L.), black pine (Pinus nigra Arnold), and black alder (Alnus glutinosa (L.) Gaertn.) to the nun moth (Lymantria monacha L.) outbreak in the SŁowiński National Park and the Damnica Forest District (Northern Poland). Electron. J. Pol. Agric. Univ. 2010, 13, 1–12. [Google Scholar]
- Cook, E.R.; Zedaker, S.M. The dendroecology of red spruce decline. In Ecology and Decline of Red Spruce in the Eastern United States; Eagar, C., Adams, M.B., Eds.; Springer: New York, NY, USA, 1992; pp. 192–234. ISBN 9780387977867. [Google Scholar]
- Wodzicki, T.J. Natural factors affecting wood structure. Wood Sci. Technol. 2001, 35, 5–26. [Google Scholar] [CrossRef]
- Montoro Girona, M.; Rossi, S.; Lussier, J.-M.; Walsh, D.; Morin, H. Understanding tree growth responses after partial cuttings: A new approach. PLoS ONE 2017, 12, e0172653. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, T.P.; Sullivan, D.S. Acceleration of old-growth structural attributes in lodgepole pine forest: Tree growth and stand structure 25 years after thinning. For. Ecol. Manag. 2016, 365, 96–106. [Google Scholar] [CrossRef]
- Mäkinen, H.; Hynynen, J. Wood density and tracheid properties of Scots pine: Responses to repeated fertilization and timing of the first commercial thinning. Forestry 2014, 87, 437–447. [Google Scholar] [CrossRef]
- Missanjo, E.; Kamanga-Thole, G. Effect of first thinning and pruning on the individual growth of Pinus patula tree species. J. For. Res. 2015, 26, 827. [Google Scholar] [CrossRef]
- Goudiaby, V.; Brais, S.; Berninger, F.; Schneider, R. Vertical patterns in specific volume increment along stems of dominant jack pine (Pinus banksiana) and black spruce (Picea mariana) after thinning. Can. J. For. Res. 2012, 42, 733–748. [Google Scholar] [CrossRef]
- Oktaba, J.; Paschalis, P.; Staniszewski, P. Selected indicators of pine and spruce wood technical quality from the forest being under the impact of industrial pollution. Folia For. Pol. Ser. A For. 2002, 44, 77–86. [Google Scholar]
- Valor, T.; Piqué, M.; López, B.C.; González-Olabarria, J.R. Influence of tree size, reduced competition, and climate on the growth response of Pinus nigra Arn. salzmannii after fire. Ann. For. Sci. 2013, 70, 503–513. [Google Scholar]
- Tikhonova, I.V.; Tarakanov, V.V.; Knorre, A.A. Contributions of genotypic and meteorological factors to variation of annual tree increment in clonal Scots pine plantations. Russ. J. Ecol. 2012, 43, 179–184. [Google Scholar] [CrossRef]
- Dobbertin, M. Tree growth as indicator of tree vitality and of tree reaction to environmental stress: A review. Eur. J. For. Res. 2005, 124, 319–333. [Google Scholar] [CrossRef]
- Hâruța, O.; Fodor, E.; Teușdea, A. Boli complexe la Pinus nigra Arnold în defileul Crișului Repede (Complex diseases in Pinus nigra Arnold situated along Crisul Repede river gorge). Ann. For. Res. 2007, 50, 169–184. [Google Scholar]
- Maestre, F.T.; Cortina, J. Are Pinus halepensis plantations useful as a restoration tool in semiarid Mediterranean areas? For. Ecol. Manag. 2004, 198, 303–317. [Google Scholar] [CrossRef]
- Constandache, C. Cercetări privind regenerarea sub masiv și introducerea la adăpostul masivului a unor specii autohtone valoroase, în arborete apropiate de exploatabilitate, de pe terenuri degradate. Ann. For. Res. 2004, 47, 63–81. [Google Scholar]
- Barbero, M.; Loisel, L.; Médail, F.; Quézel, P. Signification biogéographique et biodiversité des forêts du bassin méditerranéen. Bocconea 2001, 13, 11–25. [Google Scholar]
- Vallauri, D.R.; Aronson, J.; Barbero, M. An analysis of forest restoration 120 years after reforestation on badlands in the southwestern Alps. Restor. Ecol. 2002, 10, 16–26. [Google Scholar] [CrossRef]
- Constandache, C.; Untaru, E.; Ivan, V. Cercetări privind refacerea-ameliorarea arboretelor necorespunzătoare de pe terenuri degradate din Vrancea (Research regarding the reconstruction of destructed stand on claimed lands in Vrancea County). Ann. For. Res. 2001, 44, 168–173. [Google Scholar]
- Greavu, M. Cercetări Privind Împădurirea Terenurilor Erodate, Ravenate și Stâncoase Din Podișul Dobrogei de Nord. Ph.D. Thesis, Transilvania University of Brașov, Brașov, Romania, 2003. [Google Scholar]
- Niță, M.D.; Clinciu, I.; Popa, B. Evaluation of stream bed dynamics from vidas torrential valley using terrestrial measurements and gis techniques. Environ. Eng. Manag. J. 2015, 15, 1387–1395. [Google Scholar]
- Dresner, S.; Ekins, P.; McGeevor, K.; Tomei, J. Forests and climate change—Global understandings and possible responses. In Forestry and Climate Change; Freer-Smith, P.H., Broadmeadow, M.S.J., Lynch, J.M., Eds.; CAB International: Wallingford, UK, 2007; pp. 38–48. ISBN 9781845932954. [Google Scholar]
- Spencer, T.; Douglas, I.; Greer, T.; Sinun, W. Vegetation and fluvial geomorphic processes in South-east Asian tropical rainforests. In Vegetation and Erosion: Processes and Environments; Thornes, J.B., Ed.; John Wiley: Chichester, UK, 1990; pp. 451–469. ISBN 0471926302. [Google Scholar]
- Velcea, V.A.; Savu, A. Geografia Carpaților și Subcarpaților; Didactică si Pedagogică Publishing House: Bucharest, Romania, 1982; p. 300. [Google Scholar]
- Lefter, R. Studiul terenurilor degradate din Podișul Moldovei și ameliorarea lor prin culturi forestiere (The study of the Moldavian Plateau eroded lands and their improvement by means of the forest crops). Rev. Păd 1966, 81, 570–576. [Google Scholar]
- Martı́nez-Vilalta, J.; Piñol, J. Drought-induced mortality and hydraulic architecture in pine populations of the NE Iberian Peninsula. For. Ecol. Manag. 2002, 161, 247–256. [Google Scholar] [CrossRef]
- Martínez-Vilalta, J.; Piñol, J.; Sala, A.M. The hydraulic architecture of Pinaceae—A review. Plant Ecol. 2004, 171, 3–13. [Google Scholar] [CrossRef]
- Rundel, P.W.; Yoder, B.J. Ecophysiology of Pinus. In Ecology and Biogeography of Pinus; Richardson, D.M., Ed.; Cambridge University Press: Cambridge, UK, 1998; pp. 296–323. ISBN 978-0521789103. [Google Scholar]
- Johnson, A.H.; Cook, E.R.; Siccama, T.G. Climate and red spruce growth and decline in the northern Appalachians. Proc. Natl. Acad. Sci. USA 1988, 85, 5369–5373. [Google Scholar] [CrossRef] [PubMed]
- Dinulică, F. Lemnul de Compresiune la Brad (Compression Wood from Silver Fir); Ceres Publishing House: Bucharest, Romania, 2012; p. 296. ISBN 978-973-40-0981-7. [Google Scholar]
- Badea, L.; Băcăuanu, V.; Posea, G. Relieful României. In Geografia României I: Geografia Fizică; Badea, L., Gâștescu, P., Velcea, V., Eds.; Romanian Academy Publishing House: Bucharest, Romania, 1983; pp. 64–194. [Google Scholar]
- Țîștea, D.; Stoenescu, Ș.M.; Dissescu, C.; Donciu, C.; Topor, N.; Fetov, V. Date climatologice. In Clima Republicii Populare Române; Institutul Meteorologic: Bucharest, Romania, 1961; Volume 2, pp. 242–249. [Google Scholar]
- Bogdan, O.; Țîștea, D. Clima României. In Geografia României; Badea, L., Gâștescu, P., Velcea, V., Eds.; Romanian Academy Publishing House: Bucharest, Romania, 1983; Volume 1, pp. 195–292. [Google Scholar]
- Food and Agriculture Organization (FAO). World Reference Base for Soils Resources; World Soil Research Report No. 84; FAO: Rome, Italy, 1998; Available online: http://www.fao.org/docrep/W8594E/W8594E00.htm (accessed on 2 April 2017).
- Dincă, L.; Spârchez, G.; Dincă, M. Romanian’s forest soil GIS map and database and their ecological implications. Carpath. J. Earth Environ. 2014, 9, 133–142. [Google Scholar]
- Spârchez, G.; Dincă, L.; Marin, G.; Dincă, M.; Enescu, E.R. Variation of eutric cambisols’ chemical properties based on altitudinal and geomorphological zoning. Environ. Eng. Manag. J. 2017, 16, 2911–2918. [Google Scholar]
- Dincă, L.; Dincă, M.; Vasile, D.; Spârchez, G.; Holonec, L. Calculating organic carbon stock from forest soils. Not. Bot. Hort Agrobot. 2015, 43, 568–575. [Google Scholar] [CrossRef][Green Version]
- Silvestru-Grigore, C.V.; Enescu, R.E.; Spârchez, G. Specificul ecologic al stațiunilor plantate cu pin pe terenuri degradate din Subcarpații Buzăului. Rev. Păd 2015, 130, 98–107. [Google Scholar]
- Kraft, G. Beiträge zur Lehre von den Durchforstungen, Schlagstellungen und Lichtungshieben (Contributions to the Theory of Thinnings, Distance between Trees and Natural Regeneration of Stands); Klindworth’s Verlag: Hannover, Germany, 1884; p. 147. [Google Scholar]
- Guay, R.; Gagnon, R.; Morin, H. A new automatic and interactive tree ring measurement system based on a line scan camera. For. Chron. 1992, 68, 138–141. [Google Scholar] [CrossRef]
- WinDENDRO. WinDENDROTM2006 for Tree-Ring Analysis, Manual of Exploitation; Régent Instruments Inc.: Québec City, QC, Canada, 2007; p. 133. [Google Scholar]
- Pilcher, J.R. Sample preparation, cross-dating and measurement. In Methods of Dendrochronology; Cook, E.R., Kairiukstis, L.A., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1989; pp. 40–51. ISBN 0-7923-0586-8. [Google Scholar]
- Kerhoulas, L.P.; Kane, J.M. Sensitivity of ring growth and carbon allocation to climatic variation vary within ponderosa pine trees. Tree Physiol. 2011, 32, 14–32. [Google Scholar] [CrossRef] [PubMed]
- Cook, E.R.; Peters, K. The smoothing spline: A new approach to standardizing forest interior tree-ring width series for dendroclimatic studies. Tree-Ring Bull. 1981, 41, 45–53. [Google Scholar]
- Fritts, H.C.; Guiot, J.; Gordon, G.A. Verification. In Methods of Dendrochronology; Cook, E.R., Kairiukstis, L.A., Eds.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1989; pp. 178–185. ISBN 0-7923-0586-8. [Google Scholar]
- Cropper, J.P. Tree-ring skeleton plotting by computer. Tree-Ring Bull. 1979, 39, 47–54. [Google Scholar]
- Schweingruber, F.H.; Eckstein, D.; Serre-Bachet, F.; Bräker, O.U. Identification, presentation and interpretation of the event years and pointer years in dendrochronology. Dendrochronologia 1990, 8, 9–38. [Google Scholar]
- Tsoumis, G.; Panagiotidis, N. Effect of growth conditions on wood quality characteristics of Black pine (Pinus nigra Arn.). Wood Sci. Technol. 1980, 14, 301–310. [Google Scholar] [CrossRef]
- Dytham, C. Choosing and Using Statistics: A Biologist’s Guide, 3rd ed.; Wiley-Blackwell: Oxford, UK, 2011; pp. 171–172. ISBN 978-1-4051-9838-7. [Google Scholar]
- Box, G.E.P.; Jenkins, G.M.; Reinsel, G.R. Time Series Analysis: Forecasting and Control; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2008; pp. 474–489. ISBN 978-0-470-27284-8. [Google Scholar]
- Bruchwald, A.; Kliczkowska, A. Kształtowani się bonitacji dla drzewostanów sosnowych Polski (Characteristics of pine stand quality class in Poland). Prace Instytutu Badawczego Leśnictwa 1997, 836/842, 63–73. [Google Scholar]
- Crecente-Campo, F.; Pommerening, A.; Rodríguez-Soalleiro, R. Impacts of thinning on structure, growth and risk of crown fire in a Pinus sylvestris L. plantation in northern Spain. For. Ecol. Manag. 2009, 257, 1945–1954. [Google Scholar] [CrossRef]
- Sławski, M. Ilościowa charakterystyka zróżnicowania struktury borów sosnowych różnego wieku (Quantitative characteristic of structure in Scots pine stands of various age). Sylwan 2012, 156, 349–359. [Google Scholar]
- Condés, S.; del Río, M. Climate modifies tree interactions in terms of basal area growth and mortality in monospecific and mixed Fagus sylvatica and Pinus sylvestris forests. Eur. J. For. Res. 2015, 134, 1095–1108. [Google Scholar] [CrossRef]
- Newton, P.F. Simulating the potential effects of a changing climate on black spruce and jack pine plantation productivity by site quality and locale through model adaptation. Forests 2016, 7, 223. [Google Scholar] [CrossRef]
- Riofrío, J.; Río, M.; del Bravo, F. Mixing effects on growth efficiency in mixed pine forests. Forestry 2017, 90, 381–392. [Google Scholar] [CrossRef]
- Strimbu, V.C.; Bokalo, M.; Comeau, P.G. Deterministic Models of Growth and Mortality for Jack Pine in Boreal Forests of Western Canada. Forests 2017, 8, 410. [Google Scholar] [CrossRef]
- Xenakis, G.; Ray, D.; Mencuccini, M. Effects of climate and site characteristics on Scots pine growth. Eur. J. For. Res. 2012, 131, 427–439. [Google Scholar] [CrossRef]
- Redmond, M.D.; Kelsey, K.C.; Urza, A.K.; Barger, N.N. Interacting effects of climate and landscape physiography on piñon pine growth using an individual-based approach. Ecosphere 2017, 8, e01681. [Google Scholar] [CrossRef]
- Šeho, M.; Kohnle, U.; Albrecht, A.; Lenk, E. Wachstumsanalysen von vier Schwarzkiefer-Provenienzen (Pinus nigra) auf trockenen Standorten in Baden-Württemberg (Growth analyses of four provenances of European Black Pine (Pinus nigra) growing on dry sites in southwest Germany (Baden-Wuerttemberg)). Allg. Forst. Jagdztg. 2010, 181, 104–116. [Google Scholar]
- Eilmann, B.; Rigling, A. Tree-growth analyses to estimate tree species’ drought tolerance. Tree Physiol. 2012, 32, 178–187. [Google Scholar] [CrossRef] [PubMed]
- Martin-Benito, D.; Anchukaitis, K.J.; Evans, M.N.; del Río, M.; Beeckman, H.; Cañellas, I. Effects of Drought on Xylem Anatomy and Water-Use Efficiency of Two Co-Occurring Pine Species. Forests 2017, 8, 332. [Google Scholar] [CrossRef]
- Öner, N.; Eren, F. The comparisons between root collar diameter and height growth of black pine (Pinus nigra Arnold.) and Scots pine (Pinus sylvestris L.) seedlings in Bolu forest nursery. J. Appl. Biol. Sci. 2008, 2, 7–12. [Google Scholar]
- Haralamb, A. Specii de tranziție în lucrările de fixarea terenurilor degradate. Rev. Păd 1935, 48, 319–326. [Google Scholar]
- Poljanšek, S.; Levanič, T.; Ballian, D.; Jalkanen, R. Tree growth and needle dynamics of P. nigra and P. sylvestris and their response to climate and fire disturbances. Trees 2015, 29, 683–694. [Google Scholar]
- Rundel, P.W.; Richardson, D.M. Pines. In Encyclopedia of Forest Sciences; Burley, J., Evans, J., Youngquist, J.A., Eds.; Elsevier Ltd.: Oxford, UK, 2004; pp. 1430–1441. ISBN 0-12-145160-7. [Google Scholar]
- Lévesque, M.; Rigling, A.; Brang, P. Réponse à la sécheresse de conifères indigènes et exotiques: Une étude dendroécologique (Drought response of native and non-native conifers: A dendroecological study). Schweiz. Z. Forstwes. 2015, 166, 372–379. [Google Scholar] [CrossRef]
- Feichtinger, L.M.; Eilmann, B.; Buchmann, N.; Rigling, A. Growth adjustments of conifers to drought and to century-long irrigation. For. Ecol. Manag. 2014, 334, 96–105. [Google Scholar] [CrossRef]
- Lévesque, M.; Siegwolf, R.; Saurer, M.; Eilmann, B.; Rigling, A. Increased water-use efficiency does not lead to enhanced tree growth under xeric and mesic conditions. New Phytol. 2014, 203, 94–109. [Google Scholar] [CrossRef] [PubMed]
- Leo, M.; Oberhuber, W.; Schuster, R.; Grams, T.E.; Matyssek, R.; Wieser, G. Evaluating the effect of plant water availability on inner alpine coniferous trees based on sap flow measurements. Eur. J. For. Res. 2014, 133, 691–698. [Google Scholar] [CrossRef]
- Yakoto, A.; Takahara, K.; Akashi, K. Water stress. In Physiology and Molecular Biology of Stress Tolerance in Plants; Madhava Rao, K.V., Raghavendra, A.S., Reddy, K.J., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; pp. 15–39. ISBN 978-1-4020-4225-6. [Google Scholar]
- Wieser, G.; Leo, M.; Oberhuber, W. Transpiration and canopy conductance in an inner alpine Scots pine (Pinus sylvestris L.) forest. Flora 2014, 209, 491–498. [Google Scholar] [CrossRef] [PubMed]
- Petrucco, L.; Nardini, A.; Von Arx, G.; Saurer, M.; Cherubini, P.; Tognetti, R. Isotope signals and anatomical features in tree rings suggest a role for hydraulic strategies in diffuse drought-induced die-back of Pinus nigra. Tree Physiol. 2017, 37, 523–535. [Google Scholar] [PubMed]
- Șofletea, N.; Curtu, L. Dendrologie (Dendrology); Transilvania University Press: Brașov, Romania, 2007; pp. 68–79. [Google Scholar]
- Silvestru-Grigore, C.V.; Spârchez, G.; Dinulică, F. Starea de sănătate a arboretelor de pin instalate pe terenuri degradate din Subcarpații Buzăului (The health condition of pine stands installed on degraded lands in Buzau under Carpahians). Rev. Păd 2016, 131, 7–18. [Google Scholar]
- Pazdrowski, W. Technological value of Scots pine (Pinus sylvestris L.) wood depending on the quality of tree stems in final crops (in Polish). Roczniki Akad. Rol. Pozn. 1988, 170, 72. [Google Scholar]
- Kärenlampi, P.P.; Riekkinen, M. Pine heartwood formation as a maturation phenomenon. J. Wood Sci. 2002, 48, 467–472. [Google Scholar] [CrossRef]
- Evans, J. The productivity of second and third rotations of pine in the Usutu Forest. Swazil. Commonw. For. Rev. 1986, 65, 205–214. [Google Scholar]
- Sheppard, K.R. Plantation Silviculture; Martinus Nijhoff Publishers: Dordrecht, The Netherlands, 1986; p. 322. [Google Scholar]
- Zobel, B.J.; Sprague, J.R. Juvenile Wood in Forest Trees; Springer: Berlin, Germany, 1998; p. 300. ISBN 978-3-642-72128-1. [Google Scholar]
- Zobel, B.J.; Jett, J.B. Genetics of Wood Production; Springer: Berlin/Heidelberg, Germany; New York, NY, USA, 1995; p. 337. [Google Scholar]
- Keenan, R.J.; van Dijk, A.I.J.M. Planted forests and water in perspective. For. Ecol. Manag. 2007, 251, 1–9. [Google Scholar]
- Génova, M.; Moya, P. Dendroecological analysis of relict pine forests in the centre of the Iberian Peninsula. Biodivers. Conserv. 2012, 21, 2949–2965. [Google Scholar] [CrossRef]
- Pärn, H. Hariliku männi puistute radiaalkasvu ja kliimategurite vaheliste seoste ajalisest varieeruvusest (Temporal variability in the relationships between the radial growth of Scots pine stands and the climate). Metsanduslikud Uurim. 2004, 40, 65–79. [Google Scholar]
- Barčić, D.; Španjol, Ž.; Rosavec, R. Utjecaj na stanište i razvoj šumskih kultura crnoga bora (Pinus nigra J. F. Arnold) na krškom submediteranskom području (Impact on site and development of black pine (Pinus nigra J.F. Arnold) forest cultures in the submediterannean karst area). Croat. J. For. Eng. 2011, 32, 131–139. [Google Scholar]
- Li, Y.T.; Lo, Y.H.; Lin, Y.C.; Guan, B.T.; Blanco, J.A.; You, C.H. Bringing the natives back: Identifying and alleviating establishment limitations of native hardwood species in a conifer plantation. Forests 2018, 9, 4. [Google Scholar] [CrossRef]
- Lebreton, P.; Choisy, J.P. Avifaune et altérations forestières. III. Incidences avifaunistiques des aménagements forestières: Substitution Quercus/Pinus en milieu subméditerranéen. Bul. D′écologie 1991, 22, 213–220. [Google Scholar]
- Birsan, M.-V.; Dumitrescu, A.; Micu, D.M.; Cheval, S. Changes in annual temperature extremes in the Carpathians since AD 1961. Nat. Hazards 2014, 74, 1899–1910. [Google Scholar] [CrossRef]
- Rey, F.; Berger, F. Management of Austrian black pine on marly lands for sustainable protection against erosion (Southern Alps, France). New For. 2006, 31, 535–545. [Google Scholar] [CrossRef]
- Burger, J.A. Management effects on growth, production and sustainability of managed forest ecosystems: Past trends and future directions. For. Ecol. Manag. 2009, 258, 2335–2346. [Google Scholar] [CrossRef]
- Abrams, M.D.; Orwig, D.A. Structure, radial growth dynamics and recent climatic variations of a 320-year-old Pinus rigida rock outcrop community. Oecologia 1995, 101, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Greavu, M.; Untaru, E.; Filat, M. Cercetări privind îngrijirea și conducerea arboretelor instalate pe terenuri degradate. Ann. For. Res. 1995, 43, 31–38. [Google Scholar]
- Pérez-de-Lis, G.; García-González, I.; Rozas, V.; Arévalo, J.R. Effects of thinning intensity on radial growth patterns and temperature sensitivity in Pinus canariensis afforestations on Tenerife Islands, Spain. Ann. For. Sci. 2011, 68, 1093–1104. [Google Scholar] [CrossRef]
- Primicia, I.; Artázcoz, R.; Imbert, J.; Puertas, F.; Traver, M.; Castillo, F. Influence of thinning intensity and canopy type on Scots pine stand and growth dynamics in a mixed managed forest. For. Syst. 2016, 25, e057. [Google Scholar] [CrossRef]
- Hereş, A.M.; Martínez-Vilalta, J.; Claramunt López, B. Growth patterns in relation to drought-induced mortality at two Scots pine (Pinus sylvestris L.) sites in NE Iberian Peninsula. Trees 2012, 26, 621–630. [Google Scholar] [CrossRef]
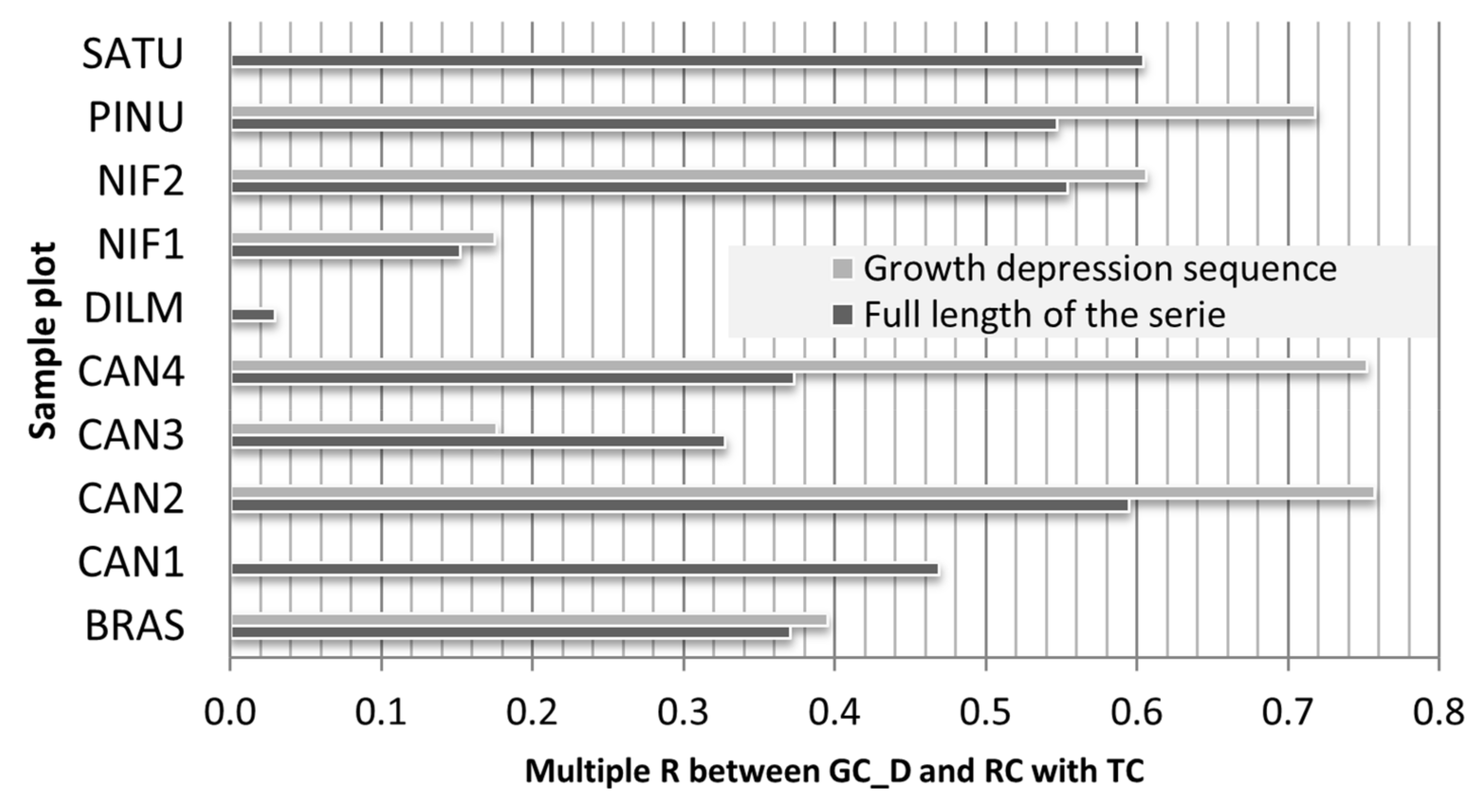
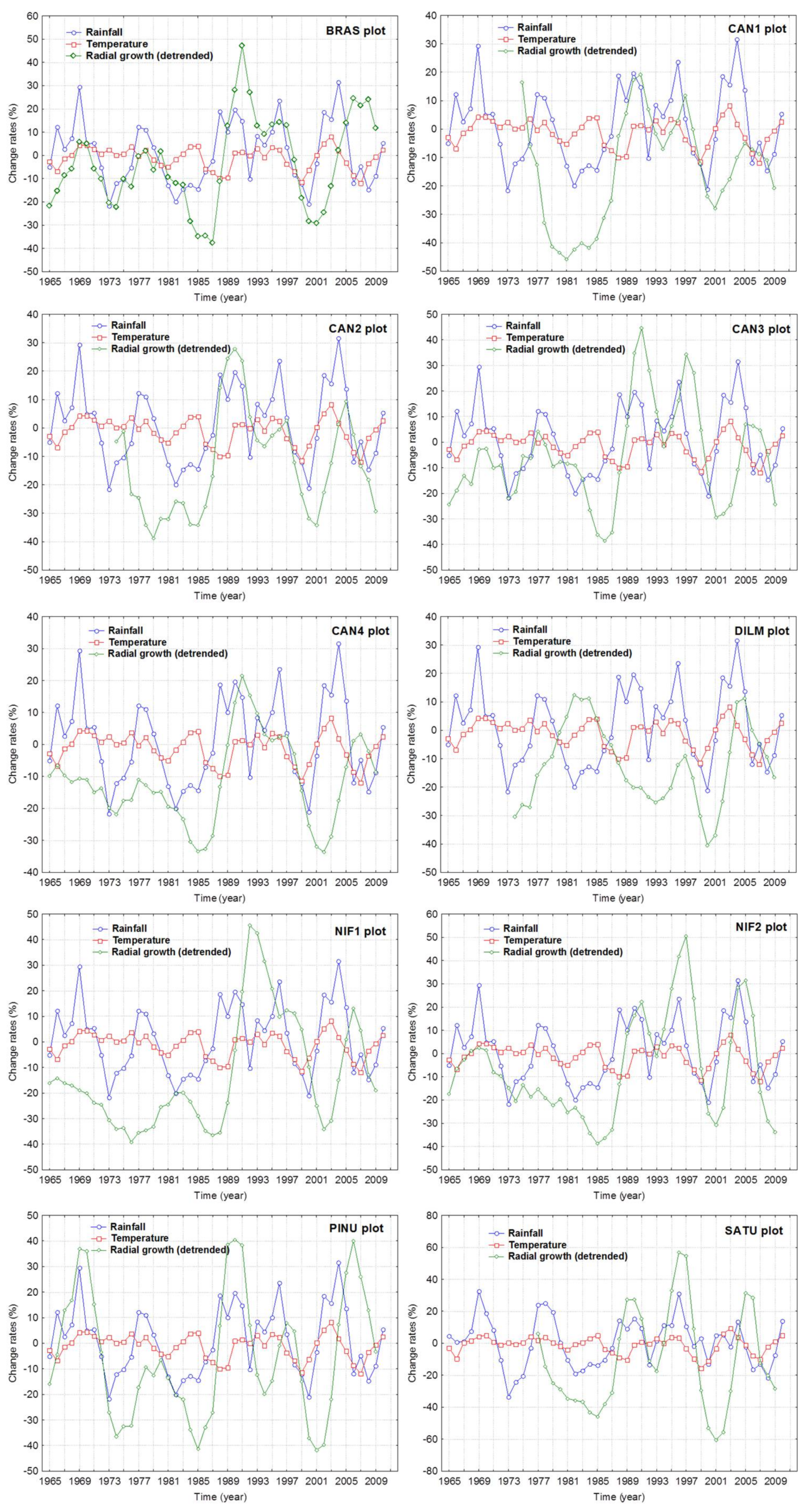
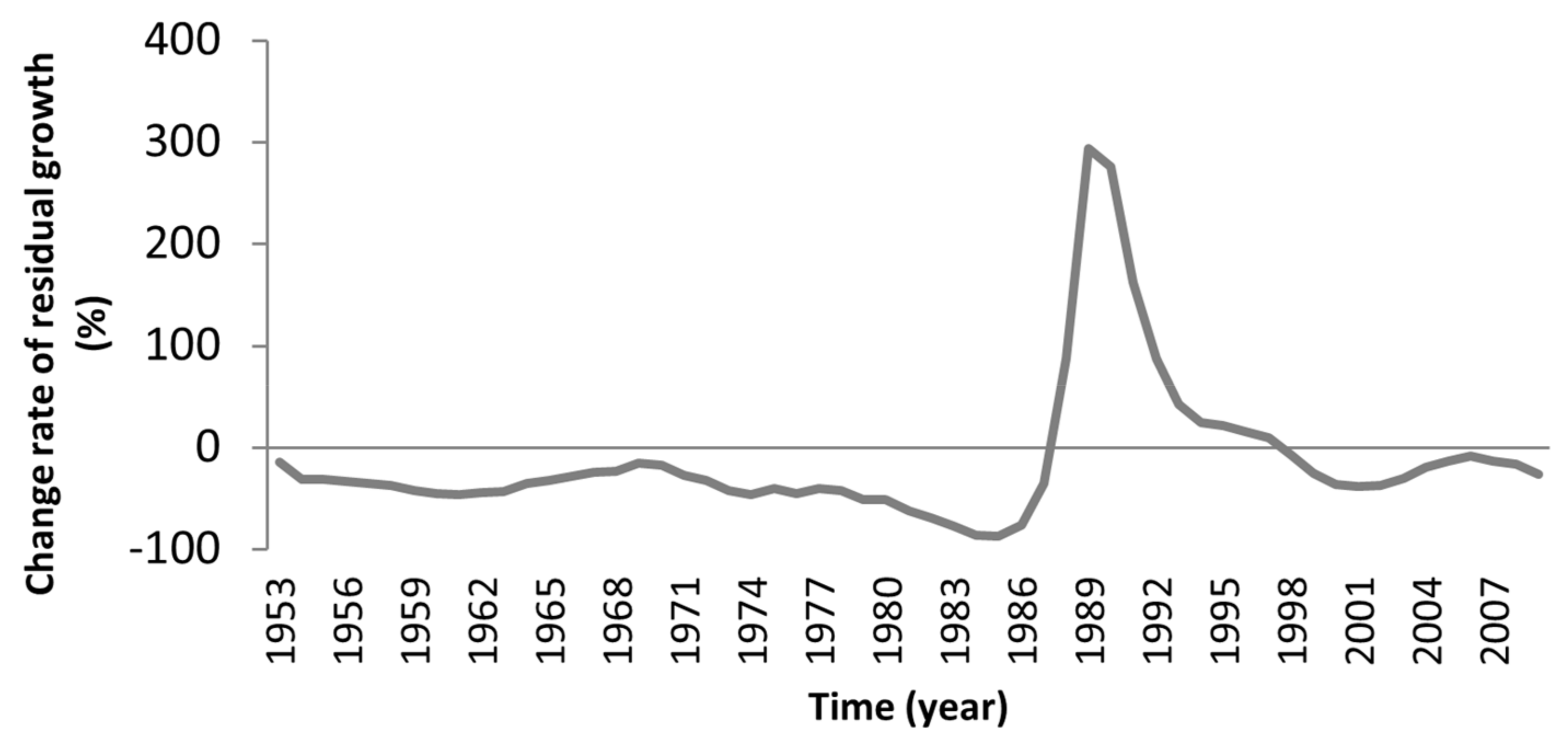
| Features | Site Plot | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| RATE | SATU | CAN1 | DILM | CAN2 | NIF1 | NIF2 | PINU | BRAS | CAN3 | CAN4 | |
| Altitude (m a.s.l.) | 300 | 295 | 370 | 700 | 325 | 305 | 320 | 665 | 505 | 325 | 340 |
| Exposition | W | NW | N | SE | W | N | N | W | N | NW | W |
| Slope (°) | 15 | 25 | 21 | 20 | 40 | 10 | 10 | 18 | 25 | 40 | 30 |
| Soil substratum | marl | marl | marl | chalky sand stone | marl | clayey marl | marl | sands | loess | marl | marl |
| Soil type * | Cmeu | Cmeu | Cmeu | Cmeu-li | Cmeu | Cmeu | Cmeu | Cmeu-ll | Cmeu | Cmeu | Cmeu |
| Soil physiological thickness (cm) | 42 | 46 | 48 | 32 | 45 | 45 | 54 | 50 | 47 | 47 | 48 |
| Nitrogen in horizon A (%) | 0.28 | 0.26 | 0.22 | 0.18 | 0.30 | 0.47 | 0.36 | 0.20 | 0.15 | 0.21 | 0.14 |
| Carbon in horizon A (%) | 2.95 | 3.76 | 2.05 | 2.37 | 3.52 | 7.03 | 5.08 | 2.78 | 1.80 | 3.21 | 1.63 |
| Base saturation (%) | 57.36 | 55.76 | 56.76 | 53.92 | 58.53 | 39.22 | 53.65 | 54.07 | 54.68 | 58.85 | 56.95 |
| Cation exchange capacity in horizon A (0.01 meq g−1) | 16.98 | 17.36 | 18.13 | 16.95 | 17.17 | 23.15 | 16.17 | 16.72 | 16.68 | 17.23 | 17.33 |
| The year of plantation establishment | 1992 | 1962 | 1962 | 1957 | 1952 | 1948 | 1935 | 1935 | 1947 | 1937 | 1937 |
| Stand composition (%) ** | 90BP 10HD | 100BP | 70SP 20BP 10HD | 100SP | 70BP 20SP 10HD | 90SP 10HD | 100BP | 90SP 10BP | 70SP 30BP | 80BP 20SP | 100BP |
| Stand density (trees·ha−1) | 1840 | 1680 | 1520 | 740 | 1100 | 1160 | 1080 | 680 | 1280 | 640 | 780 |
| The management of plantations | |||||||||||
| Stand age at intervention | - | 31 | 32 | 39 | 43 | 25 | - | - | 42 | 59 | 59 |
| Harvest intensity (% of removal) | - | 3.2 | 3.8 | 2.2 | 7.0 | 6.4 | - | - | 21.1 | 1.5 | 3.2 |
| Dependent Variables | Median | Total Variance between Rings | Predictors (Fixed Effects) | |||||
|---|---|---|---|---|---|---|---|---|
| Tree Age | Stand Density | Specie | Tree Social Position | |||||
| Covariates | ||||||||
| Tree Age | Stand Density | Tree Age | Stand Density | |||||
| p Values from F Test | ||||||||
| Annual ring width, mm | 1.50 | 2.65 | <0.01 | <0.001 | 0.04 | 0.04 | <0.001 | <0.001 |
| Annual range of ring width, mm | 6.61 | 3.78 | 0.30 | <0.001 | <0.01 | <0.01 | 0.05 | 0.08 |
| Annual average of the last 10 years ring width, mm | 0.97 | 0.34 | <0.01 | <0.001 | 0.53 | 0.57 | <0.001 | <0.001 |
| Stand Composition | No of Trees·ha−1 (Mean ± Standard Deviation) |
|---|---|
| Scots pine stands | 1193 ± 228 |
| Black pine stands | 1109 ± 468 |
| Pine mixtures | 1364 ± 344 |
| The Managed Plots | The Growth Changes after the Intervention | |||
|---|---|---|---|---|
| Change Rate of Undetrended Ring Width (%) | Change Rate of Residual Serie of Ring Width (%) | Change Rate of Air Temperature (%) | Change Rate of Rainfall (%) | |
| BRAS | +18.93 | +294.23 | −9.60 | +10.46 |
| CAN1 | −7.03 | −25.61 | −0.90 | +4.51 |
| CAN2 | −2.69 | −15.98 | +3.49 | +10.16 |
| CAN3 | +16.47 | 7.72 | +2.46 | +23.56 |
| CAN4 | +2.37 | +86.64 | +2.46 | +23.56 |
| DILM | −12.18 | −18.86 | +2.46 | +23.56 |
| NIF1 | −30.62 | −36.74 | +2.43 | −21.63 |
| SATU | −17.36 | −26.65 | +3.00 | +8.49 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silvestru-Grigore, C.V.; Dinulică, F.; Spârchez, G.; Hălălișan, A.F.; Dincă, L.C.; Enescu, R.E.; Crișan, V.E. Radial Growth Behavior of Pines on Romanian Degraded Lands. Forests 2018, 9, 213. https://doi.org/10.3390/f9040213
Silvestru-Grigore CV, Dinulică F, Spârchez G, Hălălișan AF, Dincă LC, Enescu RE, Crișan VE. Radial Growth Behavior of Pines on Romanian Degraded Lands. Forests. 2018; 9(4):213. https://doi.org/10.3390/f9040213
Chicago/Turabian StyleSilvestru-Grigore, Ciprian Valentin, Florin Dinulică, Gheorghe Spârchez, Aureliu Florin Hălălișan, Lucian Constantin Dincă, Raluca Elena Enescu, and Vlad Emil Crișan. 2018. "Radial Growth Behavior of Pines on Romanian Degraded Lands" Forests 9, no. 4: 213. https://doi.org/10.3390/f9040213
APA StyleSilvestru-Grigore, C. V., Dinulică, F., Spârchez, G., Hălălișan, A. F., Dincă, L. C., Enescu, R. E., & Crișan, V. E. (2018). Radial Growth Behavior of Pines on Romanian Degraded Lands. Forests, 9(4), 213. https://doi.org/10.3390/f9040213







