Nitrogen Adsorption Measurement for Pore Structure Characterisation of Cement–Oil Shale Ash Composite Exposed to an Aggressive Salt Environment
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Sorption Isotherms
3.2. Mesopore Volume and Size Analysis
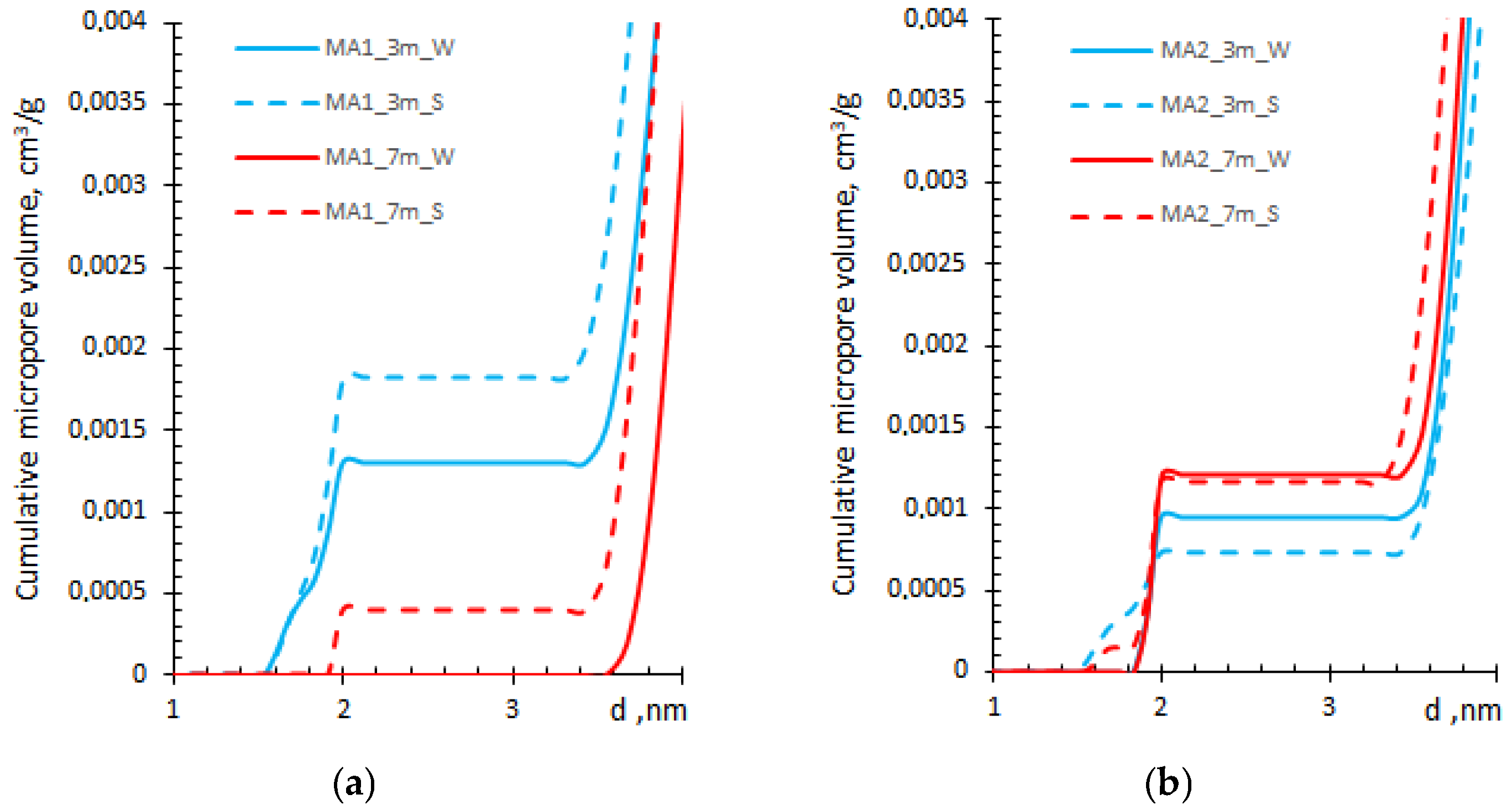
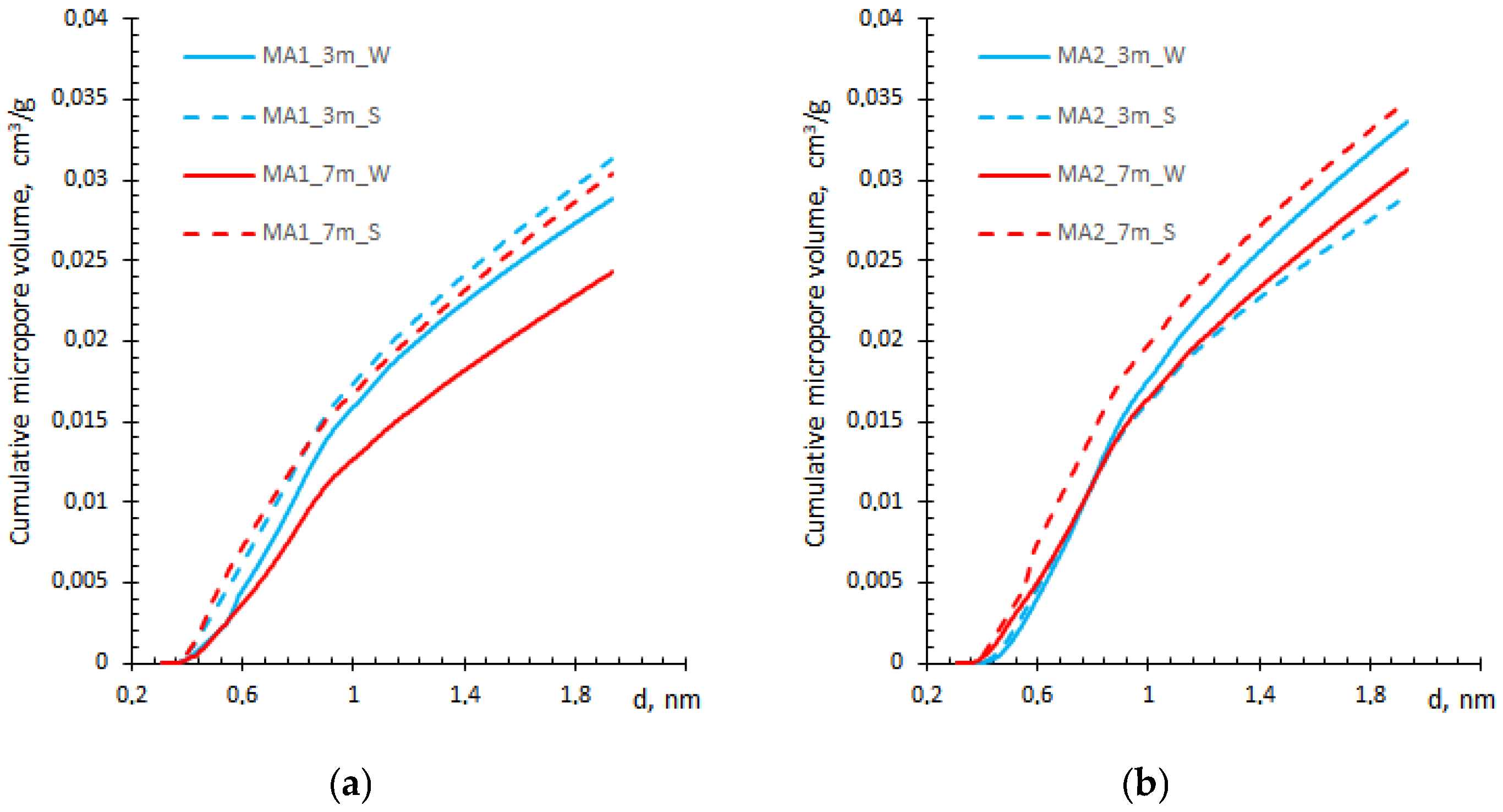
3.3. Micropore Volume and Size Analysis
3.4. Surface Fractal Dimension
4. Concluding Remarks
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jang, J.G.; Kim, G.M.; Kim, H.J.; Lee, H.K. Review on recent advances in CO2 utilization and sequestration technologies in cement-based materials. Constr. Build. Mater. 2016, 127, 762–773. [Google Scholar] [CrossRef]
- Paaver, P.; Järvik, O.; Kirsimäe, K. Design of high volume cfbc fly ash based calcium sulphoaluminate type binder in mixtures with ordinary portland cement. Materials 2021, 14, 5798. [Google Scholar] [CrossRef] [PubMed]
- Koňáková, D.; Pommer, V.; Jerman, M.; Keppert, M.; Černý, R.; Vejmelková, E. Utilization of ceramic powder, calcined shale and sintered mullite as partial replacements of calcium aluminate cement. Constr. Build. Mater. 2022, 326, 126824. [Google Scholar] [CrossRef]
- Liu, J.; Qiu, J.; Wu, P.; Sun, X.; Zhang, S.; Guo, Z. Calcined oil shale residue as a supplementary cementitious material for ordinary Portland cement. Constr. Build. Mater. 2021, 306, 124849. [Google Scholar] [CrossRef]
- Jiang, Z.-L.; Pan, Y.-J.; Lu, J.-F.; Wang, Y.-C. Pore structure characterization of cement paste by different experimental methods and its influence on permeability evaluation. Cem. Concr. Res. 2022, 159, 106892. [Google Scholar] [CrossRef]
- Bullard, J.W.; Jennings, H.M.; Livingston, R.A.; Nonat, A.; Scherer, G.W.; Schweitzer, J.S.; Scrivener, K.L.; Thomas, J.J. Mechanisms of cement hydration. Cem. Concr. Res. 2011, 41, 1208–1223. [Google Scholar] [CrossRef]
- Wenzel, O.; Schwotzer, M.; Müller, E.; Chakravadhanula, V.S.K.; Scherer, T.; Gerdes, A. Investigating the pore structure of the calcium silicate hydrate phase. Mater. Charact. 2017, 133, 133–137. [Google Scholar] [CrossRef]
- Gajewicz-Jaromin, A.M.; McDonald, P.J.; Muller, A.C.A.; Scrivener, K.L. Influence of curing temperature on cement paste microstructure measured by 1H NMR relaxometry. Cem. Concr. Res. 2019, 122, 147–156. [Google Scholar] [CrossRef]
- Kurihara, R.; Maruyama, I. Revisiting Tennis-Jennings method to quantify low-density/high-density calcium silicate hydrates in Portland cement pastes. Cem. Concr. Res. 2022, 156, 106786. [Google Scholar] [CrossRef]
- Sing, K.S.W. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Recommendations 1984). Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Rossetti, A.; Ikumi, T.; Segura, I.; Irassar, E.F. Sulfate performance of blended cements (limestone and illite calcined clay) exposed to aggressive environment after casting. Cem. Concr. Res. 2021, 147, 106495. [Google Scholar] [CrossRef]
- Ragoug, R.; Metalssi, O.O.; Barberon, F.; Torrenti, J.M.; Roussel, N.; Divet, L.; de Lacaillerie, J.-B. Durability of cement pastes exposed to external sulfate attack and leaching: Physical and chemical aspects. Cem. Concr. Res. 2019, 116, 134–145. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, Q.; Li, V.; Su, H.; Gu, C. Durability study on engineered cementitious composites (ECC) under sulfate and chloride environment. Constr. Build. Mater. 2017, 133, 171–181. [Google Scholar] [CrossRef]
- Kalpokaitė-Dičkuvienė, R.; Zabulionis, D. Effect of freeze-thawing and wet-dry cycling in salt solution on strength of biochar-silica fume blended composites. Constr. Build. Mater. 2025, 479, 141496. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, W.; Ge, Y.; Liu, P.; Zhang, A. Analysis of freeze-thaw damage and pore structure deterioration of mortar by low-field NMR. Constr. Build. Mater. 2022, 319, 126097. [Google Scholar] [CrossRef]
- Liu, L.; Wang, X.; Zhou, J.; Chu, H.; Shen, D.; Chen, H.; Qin, S. Investigation of pore structure and mechanical property of cement paste subjected to the coupled action of freezing/thawing and calcium leaching. Cem. Concr. Res. 2018, 109, 133–146. [Google Scholar] [CrossRef]
- Adu-Amankwah, S.; Zajac, M.; Skoček, J.; Němeček, J.; Haha, M.B.; Black, L. Combined influence of carbonation and leaching on freeze-thaw resistance of limestone ternary cement concrete. Constr. Build. Mater. 2021, 307, 125087. [Google Scholar] [CrossRef]
- Liu, X.; Feng, P.; Li, W.; Geng, G.; Huang, J.; Gao, Y.; Mu, S.; Hong, J. Effects of pH on the nano/micro structure of calcium silicate hydrate (C–S–H) under sulfate attack. Cem. Concr. Res. 2021, 140, 106306. [Google Scholar] [CrossRef]
- Babaahmadi, A.; Machner, A.; Kunther, W.; Figueira, J.; Hemstad, P.; De Weerdt, K. Chloride binding in Portland composite cements containing metakaolin and silica fume. Cem. Concr. Res. 2022, 161, 106924. [Google Scholar] [CrossRef]
- Jin, M.; Ma, Y.; Li, W.; Huang, J.; Yan, Y.; Zeng, H.; Lu, C.; Liu, J. Multi-scale investigation on composition-structure of C-(A)-S-H with different Al/Si ratios under attack of decalcification action. Cem. Concr. Res. 2023, 172, 107251. [Google Scholar] [CrossRef]
- Huang, Q.; Wang, Q.; Zhu, X. Contradict mechanism of long-term magnesium and sodium sulfate attacks of nano silica-modified cement mortars—Experimental and thermodynamic modeling. Cem. Concr. Compos. 2024, 147, 105444. [Google Scholar] [CrossRef]
- Aili, A.; Maruyama, I. Review of Several Experimental Methods for Characterization of Micro- and Nano-Scale Pores in Cement-Based Material. Int. J. Concr. Struct. Mater. 2020, 14, 55. [Google Scholar] [CrossRef]
- Schlumberger, C.; Thommes, M. Characterization of Hierarchically Ordered Porous Materials by Physisorption and Mercury Porosimetry—A Tutorial Review. Adv. Mater. Interfaces 2021, 8, 2002181. [Google Scholar] [CrossRef]
- Kurihara, R.; Maruyama, I. Surface area development of Portland cement paste during hydration: Direct comparison with 1H NMR relaxometry and water vapor/nitrogen sorption. Cem. Concr. Res. 2022, 157, 106805. [Google Scholar] [CrossRef]
- Zelenka, T.; Horikawa, T.; Do, D.D. Artifacts and misinterpretations in gas physisorption measurements and characterization of porous solids. Adv. Colloid Interface Sci. 2023, 311, 102831. [Google Scholar] [CrossRef]
- Muller, A.C.A.; Scrivener, K.L.; Gajewicz, A.M.; McDonald, P.J. Use of bench-top NMR to measure the density, composition and desorption isotherm of C–S–H in cement paste. Microporous Mesoporous Mater. 2013, 178, 99–103. [Google Scholar] [CrossRef]
- Zhang, Z.; Scherer, G.W. Evaluation of drying methods by nitrogen adsorption. Cem. Concr. Res. 2019, 120, 13–26. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, G.; Ye, G.; Hu, J. A comparative study on the pore structure of alkali-activated fly ash evaluated by mercury intrusion porosimetry, N2 adsorption and image analysis. J. Mater. Sci. 2018, 53, 5958–5972. [Google Scholar] [CrossRef]
- Wang, J.; Tao, J.; Li, L.; Zhou, C.; Zeng, Q. Thinner fillers, coarser pores? A comparative study of the pore structure alterations of cement composites by graphene oxides and graphene nanoplatelets. Compos. Part A Appl. Sci. Manuf. 2020, 130, 105750. [Google Scholar] [CrossRef]
- Zhu, X.; He, B.; Xie, M.; Zhang, Y.; Jiang, Z. A comprehensive nitrogen adsorption measurement on the pore structure of calcium-silicate-hydrate subjected to cryogenic attack. Measurement 2021, 184, 109941. [Google Scholar] [CrossRef]
- Kalpokaitė-Dičkuvienė, R.; Lukošiūtė, S.I.; Baltušnikas, A.; Čėsnienė, J. Structural observation of cement paste modified with hydrophobic organoclay. Constr. Build. Mater. 2021, 272, 121931. [Google Scholar] [CrossRef]
- Kalpokaitė-Dičkuvienė, R.; Pitak, I.; Baltušnikas, A.; Lukošiūtė, S.I.; Denafas, G.; Čėsnienė, J. Cement substitution by sludge-biomass gasification residue: Synergy with silica fume. Constr. Build. Mater. 2022, 326, 126902. [Google Scholar] [CrossRef]
- Alaloul, W.S.; Al Salaheen, M.; Malkawi, A.B.; Alzubi, K.; Al-Sabaeei, A.M.; Musarat, M.A. Utilizing of oil shale ash as a construction material: A systematic review. Constr. Build. Mater. 2021, 299, 123844. [Google Scholar] [CrossRef]
- Usta, M.C.; Yörük, C.R.; Hain, T.; Paaver, P.; Snellings, R.; Rozov, E.; Gregor, A.; Kuusik, R.; Trikkel, A.; Uibu, M. Evaluation of New Applications of Oil Shale Ashes in Building Materials. Minerals 2020, 10, 765. [Google Scholar] [CrossRef]
- Leben, K.; Mõtlep, R.; Paaver, P.; Konist, A.; Pihu, T.; Paiste, P.; Heinmaa, I.; Nurk, G.; Anthony, E.J.; Kirsimäe, K. Long-term mineral transformation of Ca-rich oil shale ash waste. Sci. Total Environ. 2019, 658, 1404–1415. [Google Scholar] [CrossRef]
- Nov, S.; Cohen, H.; Knop, Y. Treated Oil Shale Ashes as a Substitute for Natural Aggregates. Sand, and Cement in Concrete. Isr. J. Chem. 2020, 60, 638–643. [Google Scholar] [CrossRef]
- Kalpokaitė-Dičkuvienė, R.; Pitak, I.; Baltušnikas, A.; Čėsnienė, J.; Kriūkienė, R.; Lukošiūtė, S.I. Functional and microstructural alterations in hydrated and freeze–thawed cement-oil shale ash composites. Case Stud. Constr. Mater. 2023, 19, e02302. [Google Scholar] [CrossRef]
- Kalpokaitė-Dičkuvienė, R.; Stasiulaitiene, I.; Baltušnikas, A.; Yousef, S. Transforming Waste into Value: The Role of Recovered Carbon Fibre and Oil Shale Ash in Enhancing Cement-Based Structural Composites. Materials 2025, 18, 5636. [Google Scholar] [CrossRef]
- De Weerdt, K.; Justnes, H. The effect of sea water on the phase assemblage of hydrated cement paste. Cem. Concr. Compos. 2015, 55, 215–222. [Google Scholar] [CrossRef]
- Nguyen, H.; Kinnunen, P.; Carvelli, V.; Illikainen, M. Durability of ettringite-based composite reinforced with polypropylene fibers under combined chemical and physical attack. Cem. Concr. Compos. 2019, 102, 157–168. [Google Scholar] [CrossRef]
- Ting, M.Z.Y.; Yi, Y. Durability of cementitious materials in seawater environment: A review on chemical interactions, hardened-state properties and environmental factors. Constr. Build. Mater. 2023, 367, 130224. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Pomonis, P.J.; Tsaousi, E.T. Frenkel-Halsey-Hill equation, dimensionality of adsorption, and pore anisotropy. Langmuir 2009, 25, 9986–9994. [Google Scholar] [CrossRef]
- Yang, J.; Wang, F.; He, X.; Su, Y. Pore structure of affected zone around saturated and large superabsorbent polymers in cement paste. Cem. Concr. Compos. 2019, 97, 54–67. [Google Scholar] [CrossRef]
- Duan, P.; Shui, Z.; Chen, W.; Shen, C. Influence of superplasticizer on composition and pore structure of C–S–H. Constr. Build. Mater. 2013, 44, 87–91. [Google Scholar] [CrossRef]
- Sandoval-Díaz, L.-E.; Aragon-Quiroz, J.-A.; Ruíz-Cardona, Y.-S.; Domínguez-Monterroza, A.-R.; Trujillo, C.-A. Fractal analysis at mesopore scale of modified USY zeolites by nitrogen adsorption: A classical thermodynamic approach. Microporous Mesoporous Mater. 2017, 237, 260–267. [Google Scholar] [CrossRef]
- Yang, Y.; Lu, X.; Wang, Q. Investigation on the co-combustion of low calorific oil shale and its semi-coke by using thermogravimetric analysis. Energy Convers. Manag. 2017, 136, 99–107. [Google Scholar] [CrossRef]





| Notations | Metakaolin | Ash Type | Curing Duration | Curing Medium | |||
|---|---|---|---|---|---|---|---|
| No. 1 | No. 2 | Months (m) | Lime Water | Salt Solution | |||
| MA1_Ref | M | A1 | 1 m | W | |||
| MA2_Ref | M | A2 | 1 m | W | |||
| MA1-3m-W | M | A1 | 3 m | W | |||
| MA2-3m-W | M | A2 | 3 m | W | |||
| MA1-3m-S | M | A1 | 3 m | S | |||
| MA2-3m-S | M | A2 | 3 m | S | |||
| MA1-7m-W | M | A1 | 7 m | W | |||
| MA2-7m-W | M | A2 | 7 m | W | |||
| MA1-7m-S | M | A1 | 7 m | S | |||
| MA2-7m-S | M | A2 | 7 m | S | |||
| Sample | Ref. [38] | Curing Medium—Lime Water | Curing Medium—Salt Solution | ||||
|---|---|---|---|---|---|---|---|
| 3 Months [38] | 7 Months | Change, % | 3 Months | 7 Months | Change, % | ||
| Total specific volume of pores less than 200 nm at p/p0 = 0.99, cm3/g | |||||||
| MA1 | 0.1625 | 0.2126 | 0.1983 | −7% | 0.2010 | 0.2225 | +11% |
| MA2 | 0.1926 | 0.2125 | 0.2163 | +2% | 0.1911 | 0.2112 | +11% |
| Average pore diameter, nm | |||||||
| MA1 | 21.93 | 10.58 | 10.98 | +4% | 9.07 | 10.12 | +12% |
| MA2 | 11.86 | 8.75 | 9.77 | +12% | 9.50 | 8.55 | −10% |
| Specific surface area (BET method) m2/g | |||||||
| MA1 | 29.64 | 80.38 | 72.22 | −10% | 88.67 | 87.96 | −1% |
| MA2 | 64.94 | 97.18 | 88.53 | −9% | 80.46 | 98.82 | +23% |
| Micropore area (DR method), m2/g | |||||||
| MA1 | 24.85 | 63.41 | 50.47 | −20% | 67.44 | 60.70 | −10% |
| MA2 | 55.27 | 66.68 | 67.56 | +1% | 63.74 | 74.87 | +18% |
| Micropore volume (DR method), cm3/g | |||||||
| MA1 | 0.009 | 0.023 | 0.018 | −22% | 0.024 | 0.022 | −8% |
| MA2 | 0.020 | 0.024 | 0.024 | 0% | 0.023 | 0.027 | +17% |
| Samples | NK Method | FHH Method | |||||
|---|---|---|---|---|---|---|---|
| Fractal Dimension (D) | Fractal Dimension (D) | ||||||
| Slope (h) | D = 2 − h | R2 | Slope (h) | D = 3(h + 1) (Equation (1)) | D = 3 + h (Equation (2)) | R2 | |
| 1 month in lime water (reference samples) | |||||||
| MA1 | −0.735 | 2.7353 | 0.984 | −0.567 | h < −0.33 | 2.4328 | 0.998 |
| MA2 | −0.967 | 2.9672 | 0.994 | −0.431 | h < −0.33 | 2.5687 | 0.986 |
| 3 months in lime water | |||||||
| MA1 | −0.973 | 2.9726 | 0.994 | −0.395 | h < −0.33 | 2.6051 | 0.987 |
| MA2 | −1.121 | 3.1211 | 0.992 | −0.371 | h < −0.33 | 2.6288 | 0.977 |
| 7 months in lime water | |||||||
| MA1 | −0.949 | 2.9493 | 0.997 | −0.400 | h < −0.33 | 2.5997 | 0.988 |
| MA2 | −1.007 | 3.0068 | 0.994 | −0.371 | h < −0.33 | 2.6286 | 0.982 |
| 3 months in salt solution | |||||||
| MA1 | −1.050 | 3.0499 | 0.997 | −0.366 | h < −0.33 | 2.6343 | 0.981 |
| MA2 | −1.069 | 3.0692 | 0.994 | −0.385 | h < −0.33 | 2.6146 | 0.980 |
| 7 months in salt solution | |||||||
| MA1 | −1.002 | 3.0023 | 0.997 | −0.381 | h < −0.33 | 2.6149 | 0.981 |
| MA2 | −1.131 | 3.1306 | 0.995 | −0.351 | h < −0.33 | 2.6487 | 0.972 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kalpokaitė-Dičkuvienė, R. Nitrogen Adsorption Measurement for Pore Structure Characterisation of Cement–Oil Shale Ash Composite Exposed to an Aggressive Salt Environment. Materials 2026, 19, 772. https://doi.org/10.3390/ma19040772
Kalpokaitė-Dičkuvienė R. Nitrogen Adsorption Measurement for Pore Structure Characterisation of Cement–Oil Shale Ash Composite Exposed to an Aggressive Salt Environment. Materials. 2026; 19(4):772. https://doi.org/10.3390/ma19040772
Chicago/Turabian StyleKalpokaitė-Dičkuvienė, Regina. 2026. "Nitrogen Adsorption Measurement for Pore Structure Characterisation of Cement–Oil Shale Ash Composite Exposed to an Aggressive Salt Environment" Materials 19, no. 4: 772. https://doi.org/10.3390/ma19040772
APA StyleKalpokaitė-Dičkuvienė, R. (2026). Nitrogen Adsorption Measurement for Pore Structure Characterisation of Cement–Oil Shale Ash Composite Exposed to an Aggressive Salt Environment. Materials, 19(4), 772. https://doi.org/10.3390/ma19040772






