Corrosion Behaviour in CO2 Pipeline Transport: A Review of the Impact of Condensates and Impurities
Highlights
- Role of the principal CCTS strategy and effect on the resulting flux
- The role of water in the CO2 flux and Top of the line corrosion
- Role of the different impurities on the corrosion behaviour
Abstract
1. Introduction
1.1. Energy Transition: The CO2 Impact
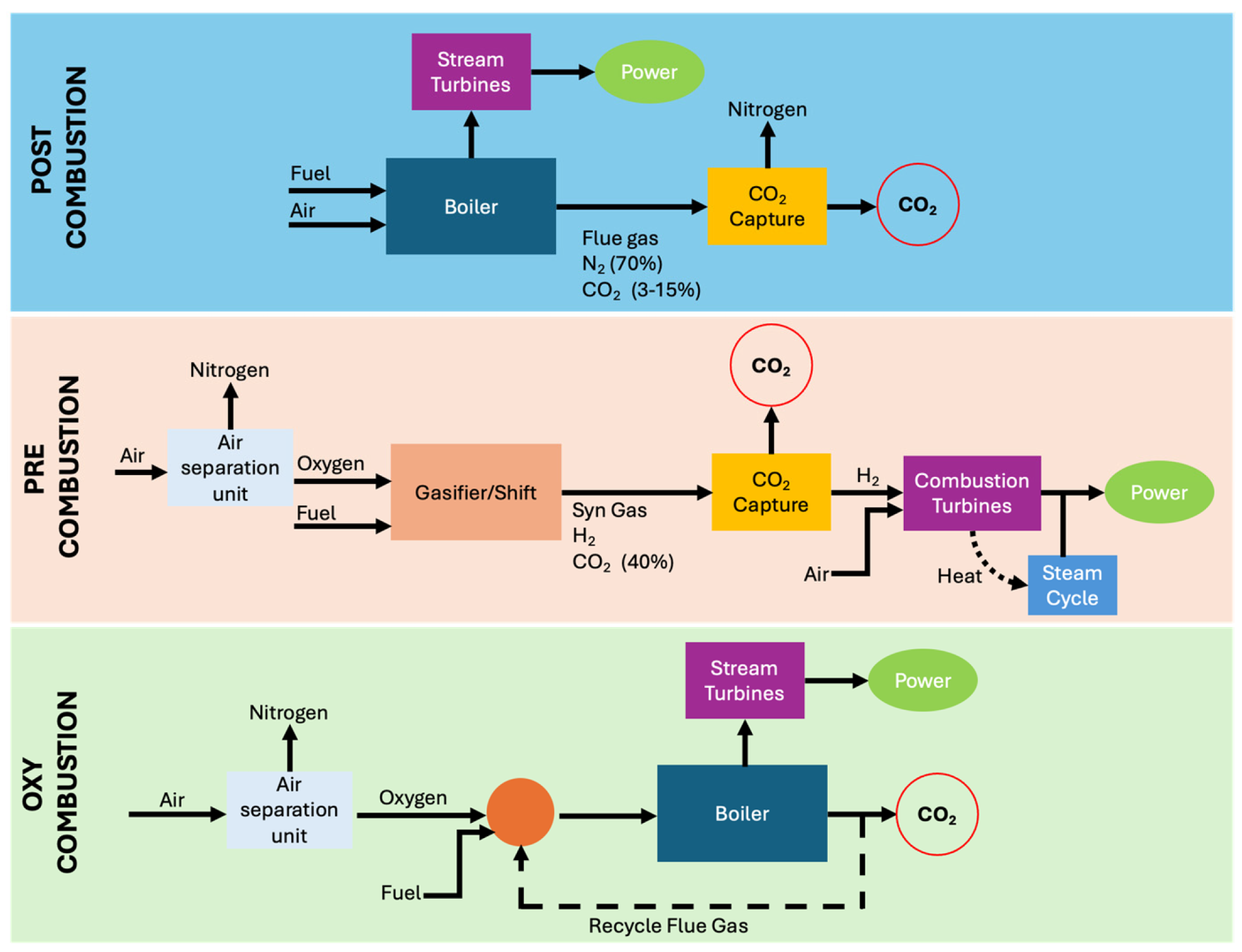
1.2. CO2 Capture Techniques
- Pre-combustion: This method involves removing CO2 from fossil fuels upstream of the combustion process, resulting in a hydrogen-rich gas that can be used as a clean and versatile fuel in power generation plants or for alternative uses (transportation, basic chemicals, etc.). From an energy standpoint, the associated penalty is generally low, both because the process deals with limited flow rates and because it can operate under pressure, a condition that facilitates CO2 capture and reduces the energy cost of regenerating the sorbent agents [6,7].
- Post-combustion: This approach involves removing CO2 from flue gases after the combustion of the fuel source, e.g., extracting CO2 from the exhaust gases of power plants and industrial facilities. Post-combustion systems are the most technologically mature, thanks to the experience gained in the oil and gas sector and small-scale gas treatment applications. They are best suited for retrofitting existing plants, provided there is sufficient space available, given the large volumes involved. The main disadvantages are the high capital costs associated with the need to treat large gas volumes as well as significant energy penalties due to the regeneration phase [6,7].
- Oxy-fuel combustion: This third approach, among the most promising today, involves combusting the fuel source with nearly pure oxygen instead of air. This results in a flue gas stream rich in CO2 and free of nitrogen. After passing through pollutant removal units and a condensation section to remove water vapour, the stream can be sent to storage. This process already finds applications in the steel and glass industries and is currently being explored in power generation at the global level [6,7].
2. Working Fluid Transported
2.1. Target Composition Criteria Within the Current Regulatory Framework
- Large volumes of O2, N2, H2, Ar, and H2O (in the form of vapour or condensate);
- ○
- Particulates/dust that need to be mechanically separated (and which are generally absent during transport);
- ○
- Traces of components that may act as catalyst poisons or be toxic to downstream process microorganisms, or even for human consumption. For example, acidic components such as H2S, SO2, SO2, HCl, HF, COS, CS2, CH3SH, HCN, NO, NO2/NO3, and Cl2; basic components such as NH3 and amines, whose presence is due to the flue gas purification process (amine-based processes being among the most common); combustible components such as CO, CH4, and organic substances; metallic components such as mercury, heavy metals (Ni, Cr, etc.), and alkali and alkaline earth metals (Na, K, Ca, Ba), which occur in the form of aerosols rather than particulates; volatile organic compounds such as aromatic hydrocarbons, olefins, aldehydes/organic acids, dioxins/furans, oils/greases, etc.
- The maintenance of stable conditions; this may involve adjusting the injection capacity of the system (pipeline and well geometry, reservoir conditions, ambient temperature, compression and reservoir conditions, ambient temperature, compression and pumping equipment, etc.)
- The identification of flow conditions that can reduce hydraulic capacity (hydrate formation) and compromise system integrity (erosion, hydrate formation, corrosion potential, etc.)
- Maintaining temperature within an acceptable range; pressure reductions resulting from normal operations may lead to temperature drops due to evaporation of liquid CO2, affecting heat transfer and pipelines.
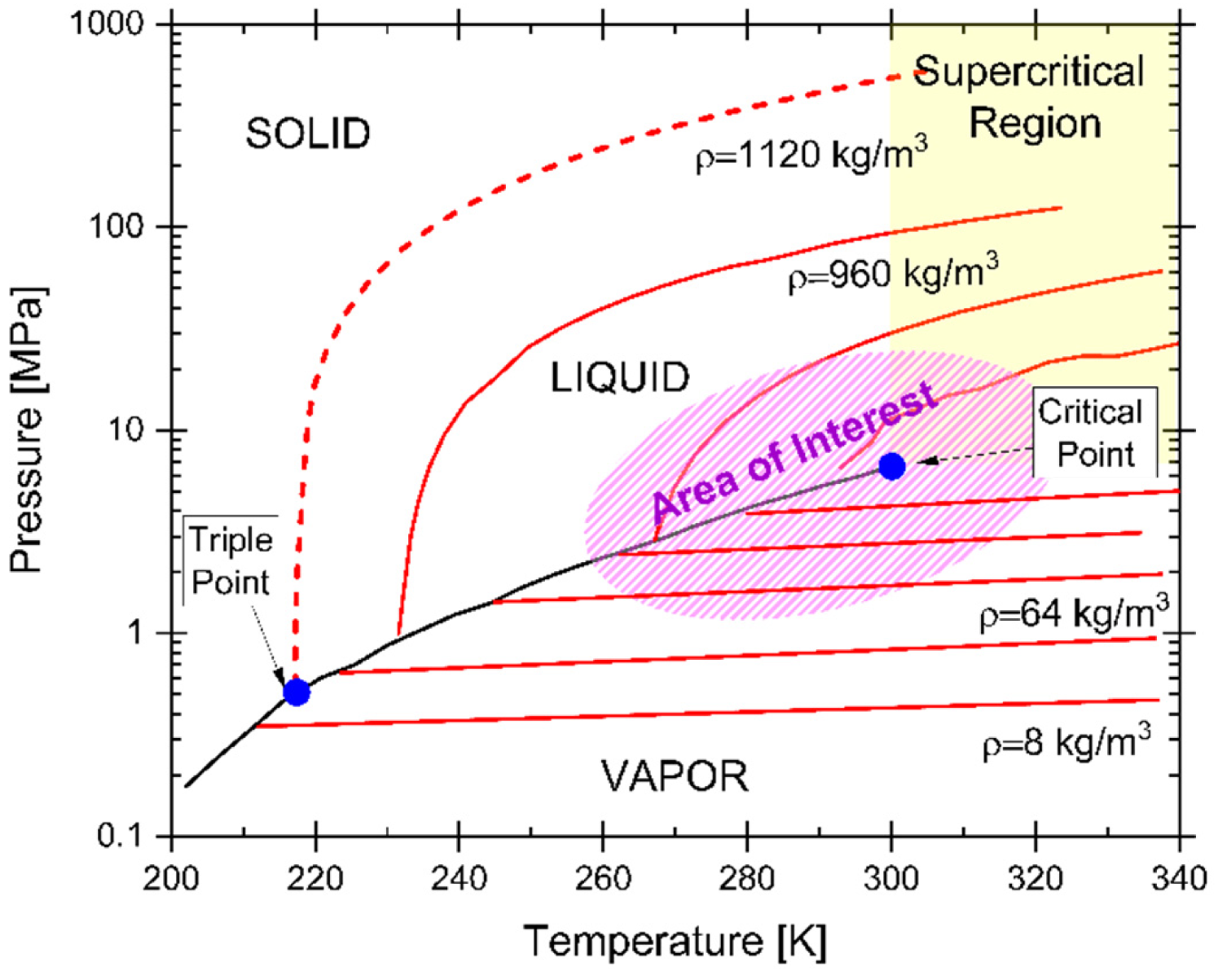
2.2. Phase Transitions and CO2 Phase Diagram
2.3. Effects of Impurities on the Physical Properties of the Fluid and on Facilities
2.3.1. Variation of the Critical Point: Canadian Case Study on Impurities
2.3.2. Phase Diagram Variation: The Role of Water
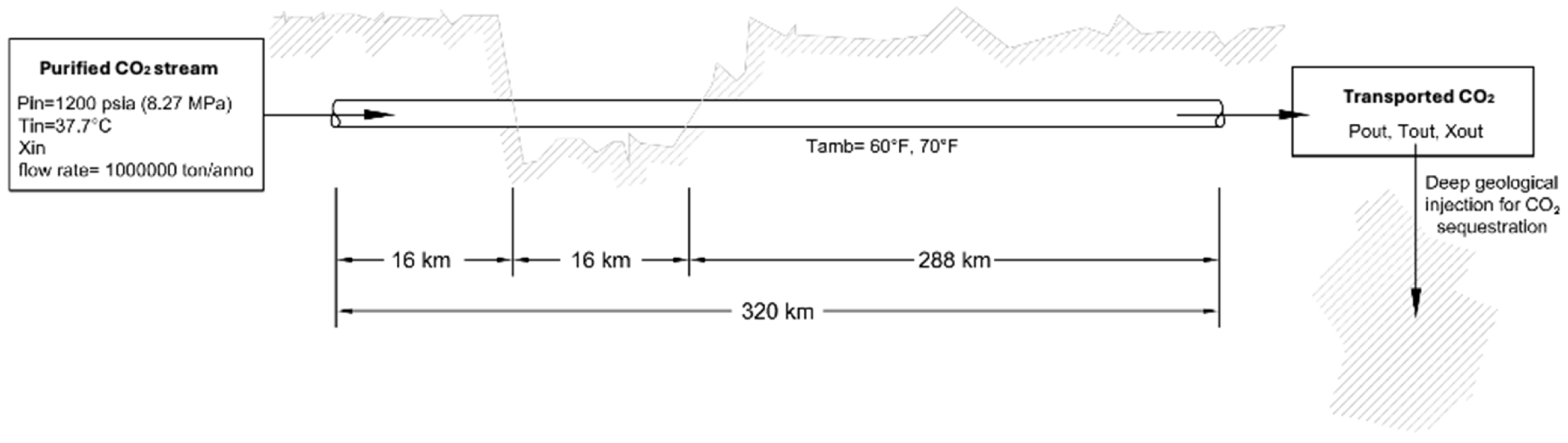
2.3.3. Variation of Physical Properties of CO2 Mixtures Induced by Pipeline Transport: Pipeline Model
3. Phenomena of Corrosion Associated with CO2 Transport
3.1. The Role of Water in the Corrosion of Transported CO2 Mixtures
- Dissolution of CO2
- Hydration of CO2
- Dissociation of carbonic acid
3.1.1. Dry CO2 Flow (Absence of Water)
3.1.2. CO2 Flow in the Presence of Water
Mixtures of CO2 with Small Quantities of Water (Below Solubility Limit)
- Carbon steels and Cr-containing stainless steels show negligible or undetectable corrosion rates (<0.001 mm/year) in the absence of water
- Carbon steels incur mild corrosion (<0.1 mm/year) when water content in supercritical CO2 remains below saturation (undersaturated conditions)
- Carbon steels suffer corrosion rates exceeding 0.1 mm/year, (and even reaching 1 mm/year) when in water-saturated supercritical CO2.
Mixtures of CO2 with Saturated Aqueous Phase
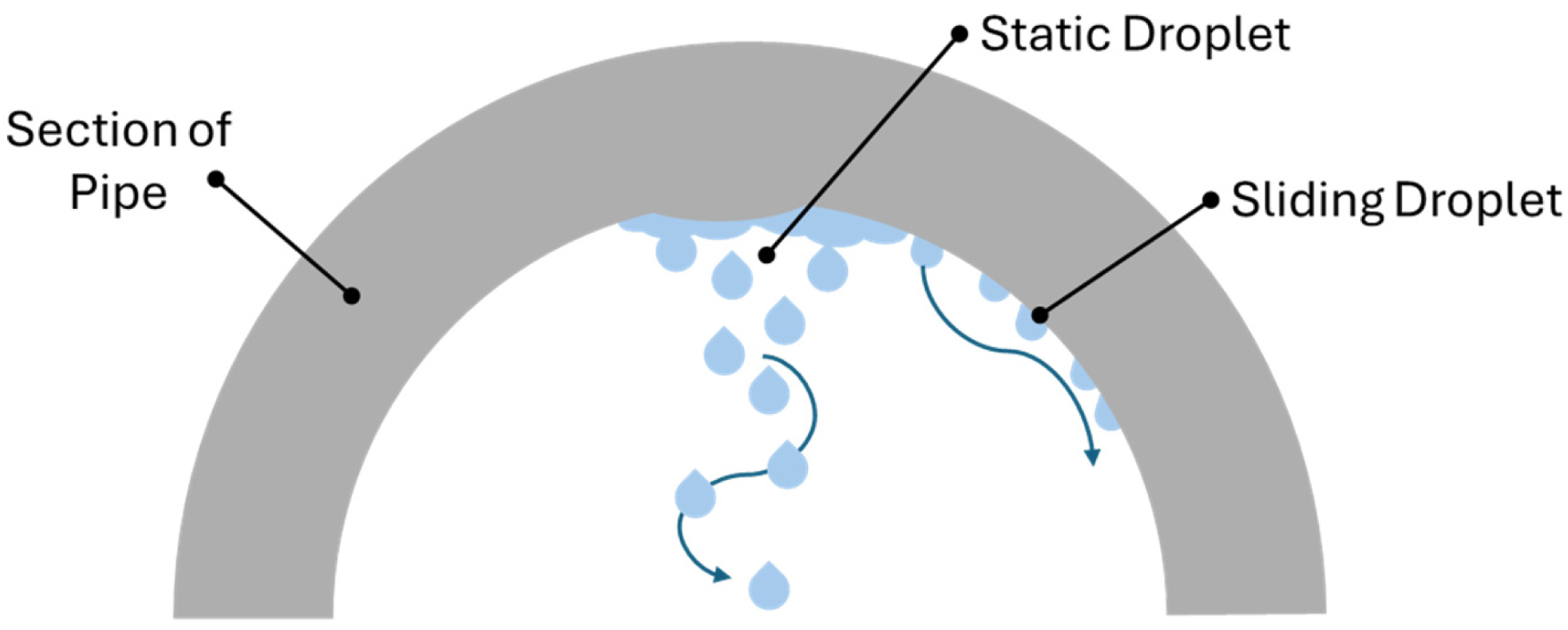
3.2. Corrosion Mechanism: Top of the Line Corrosion (TLC)
- pitting corrosion, when attack initiates at small defects in the passive corrosion-product layer where the base metal interfaces with the environment
- mesa corrosion, when steel is not corroded uniformly but presents surface pits often wide and flat-bottomed, surrounded by corrosion products [84].
3.2.1. Factors Influencing TLC Mechanisms
- Pipeline walls separating a hot internal environment from a colder external one (e.g., seawater or soil)
- High internal condensation rates
- High internal temperatures
- High CO2 partial pressures
- High water-vapour loads.
Gas Temperature
CO2 Partial Pressure
Water Condensation Rate
- Gas temperature
- Surface sub-cooling
- Incondensable gas concentration
- Gas velocity
- System pressure
- Pipeline inner diameter.
3.2.2. TLC Mechanisms in CO2-Dominant Environments
- FeCO3 saturation forms a highly protective scale during early stages, limiting Fe ion release and metal dissolution. Yet continuous renewal of condensate (lacking Fe ions) sustains corrosion.
- Equilibrium is reached only when FeCO3 saturation approaches unity; the Fe ion input from steel matches the dilution by fresh condensate.
- High condensation rates promote rapid solution renewal at walls, hindering stable protective film development and maintaining high corrosion rates; conversely low condensate mass flux supports protective scale formation, yielding low but non-zero corrosion rates.
- Temperature strongly affects FeCO3 deposition kinetics. At low temperatures (40 °C), the corrosion rate is steady but moderate due to inhibited protective film formation. At temperatures > 70 °C, protective film quickly grows but forms surface cracks. Defects in the coating foster localized TLC attacks, which are eventually limited over time: the electrolyte within fissures promotes film growth, partially occluding defects. Localized attack rates can reach 5–10 mm/year depending on the condensation rate and environmental aggressiveness.
3.3. Role of Other Impurities on Corrosion Behaviour
3.3.1. Oxygen (O2)
3.3.2. Nitrogen Dioxide (NO2) and Sulphur Dioxide (SO2)
- O2, SO2, and NO2 can accelerate the corrosion of carbon steels in CO2/H2O environments. Among the three impurities at the same concentration, NO2 has the most significant effect on the corrosion rate of carbon steel, followed by SO2 and O2
- For SO2, corrosion can occur in the CO2/SO2/H2O system even when the water content is well below the solubility limit of CO2 in water. The corrosion rate increases with the concentration of SO2.
3.3.3. Effects of Real Impurity Mixtures
Effect of Mixed Impurities
Amine Treatment and Effects on Corrosion Rates
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Masson-Delmotte, V.; Zhai, P.; Pörtner, H.-O.; Roberts, D.; Skea, J.; Shukla, P.R.; Pirani, A.; Moufouma-Okia, W.; Péan, C.; Pidcock, R.; et al. Global Warming of 1.5 °C: IPCC Special Report on Impacts of Global Warming of 1.5 °C; Cambridge University Press: Cambridge, UK, 2022. [Google Scholar]
- Barns, D.W.; Edmonds, J.A.; Reilly, J.M. Use of the Edmonds-Reilly Model to Model Energy-Related Greenhouse Gas Emission; EEA: Copenhagen, Denmark, 2011. [Google Scholar]
- Onyebuchi, V.E.; Kolios, A.; Hanak, D.P.; Biliyok, C.; Manovic, V. A systematic review of key challenges of CO2 transport via pipelines. Renew. Sustain. Energy Rev. 2018, 81, 2563–2583. [Google Scholar] [CrossRef]
- Hausfather, Z. An assessment of current policy scenarios over the 21st century and the reduced plausibility of high-emissions pathways. Dialogues Clim. Change 2025, 2, 26–32. [Google Scholar] [CrossRef]
- Haussfather, Z. Global CO2 Emissions Have Been Flat for a Decade, New Data Reveals; Carbon Brief: London, UK, 2021. [Google Scholar]
- Raganati, F.; Ammendola, P. CO2 Post-combustion Capture: A Critical Review of Current Technologies and Future Directions. Energy Fuels 2024, 38, 13858–13905. [Google Scholar] [CrossRef]
- ENEA. Tecnologie per la cattura del CO2 Speciale sulle Tecnologie di Carbon Capture and Storage. ENEA Magazine. Available online: https://www.eai.enea.it/archivio/speciale-sulle-tecnologie-di-carbon-capture-and-storage.html (accessed on 22 March 2026).
- ISO 27913:2024; Carbon Dioxide Capture, Transportation and Geological Storage—Pipeline Transportation Systems. ISO: Geneva, Switzerland, 2024.
- Span, R.; Wagner, W. A New Equation of State for Carbon Dioxide Covering the Fluid Region from the Triple-Point Temperature to 1100 K at Pressures up to 800 MPa. J. Phys. Chem. Ref. Data 1996, 25, 1509–1596. [Google Scholar] [CrossRef]
- Estrada-Alexanders, A.F.; Trusler, J.P.M. Speed of sound in carbon dioxide at temperatures between (220 and 450) K and pressures up to 14 MPa. J. Chem. Thermodyn. 1998, 30, 1589–1601. [Google Scholar] [CrossRef]
- Gil, L.; Otín, S.F.; Embid, J.M.; Gallardo, M.A.; Blanco, S.; Artal, M.; Velasco, I. Experimental setup to measure critical properties of pure and binary mixtures and their densities at different pressures and temperatures. Determination of the precision and uncertainty in the results. J. Supercrit. Fluids 2008, 44, 123–138. [Google Scholar] [CrossRef]
- Mantilla, I.D.; Cristancho, D.E.; Ejaz, S.; Hall, K.R.; Atilhan, M.; Iglesias-Silva, G.A. P-p-T data for carbon dioxide from (310 to 450) K up to 160 MPa. J. Chem. Eng. Data 2010, 55, 4611–4613. [Google Scholar] [CrossRef]
- Masłowski, M.; Labus, K.; Czupski, M.; Ptak, S. Corrosion Resistance of Well Steel in a Supercritical Carbon Dioxide Environment in Geothermal Systems Utilizing Depleted Hydrocarbon Reservoirs. Energies 2025, 18, 6239. [Google Scholar] [CrossRef]
- Sun, H.; Wang, H.; Zeng, Y.; Liu, J. Corrosion challenges in supercritical CO2 transportation, storage, and utilization—A review. Renew. Sustain. Energy Rev. 2023, 179, 113292. [Google Scholar] [CrossRef]
- Wang, Z.M.; Song, G.L.; Zhang, J. Corrosion Control in CO2 Enhanced Oil Recovery from a Perspective of Multiphase Fluids. Front. Mater. 2019, 6, 272. [Google Scholar] [CrossRef]
- Cole, I.S.; Corrigan, P.; Sim, S.; Birbilis, N. Corrosion of pipelines used for CO2 transport in CCS: Is it a real problem? Int. J. Greenh. Gas Control 2011, 5, 749–756. [Google Scholar] [CrossRef]
- Eldevik, F.; Graver, B.; Torbergsen, L.E.; Saugerud, O.T. Development of a Guideline for Safe, Reliable and Cost Efficient Transmission of CO2 in Pipelines. Energy Procedia 2009, 1, 1579–1585. [Google Scholar] [CrossRef]
- Jung, W.; Nicot, J.-P. Impurities in CO2-Rich Mixtures Impact CO2 Pipeline Design: Implications for Calculating CO2 Transport Capacity. In Proceedings of the SPE International Conference on CO2 Capture, Storage, and Utilization, New Orleans, LA, USA, 10–12 November 2010. [Google Scholar]
- Antonio, L.; Giuseppe, D.; Paolo, T.; Carlo Maria, S. CCTS (Carbon Capture Transportation & Storage) Transportation Issues. In Proceedings of the Twenty-First International Offshore and Polar Engineering Conference, Maui, HI, USA, 19–24 June 2011; p. ISOPE-I-11-162. [Google Scholar]
- Koornneef, J.; Ramírez, A.; Turkenburg, W.; Faaij, A. The environmental impact and risk assessment of CO2 capture, transport and storage—An evaluation of the knowledge base. Prog. Energy Combust. Sci. 2012, 38, 62–86. [Google Scholar] [CrossRef]
- Dooley, J.J.; Dahowski, R.T.; Davidson, C.L. Comparing Existing Pipeline Networks with the Potential Scale of Future U.S. CO2 Pipeline Networks. Energy Procedia 2009, 1, 1595–1602. [Google Scholar] [CrossRef]
- Johnsen, K.; Helle, K.; Røneid, S.; Holt, H. DNV recommended practice: Design and operation of CO2 pipelines. Energy Procedia 2011, 4, 3032–3039. [Google Scholar] [CrossRef]
- Wang, J.; Ryan, D.; Anthony, E.J.; Wildgust, N.; Aiken, T. Effects of impurities on CO2 transport, injection and storage. Energy Procedia 2011, 4, 3071–3078. [Google Scholar] [CrossRef]
- Ben, W.; Julia, R.; Martin, D. The Effect of Impurities on a Simplified CCS Network. In Proceedings of the PSIG Annual Meeting, Prague, Czech Republic, 16–19 April 2013. [Google Scholar]
- Verma, S.; Oakes, C.S.; Chugunov, N.; Ramakrishnan, T.S. Effect of contaminants on the thermodynamic properties of CO2-rich fluids and ramifications in the design of surface and injection facilities for geologic CO2 sequestration. Energy Procedia 2011, 4, 2340–2347. [Google Scholar] [CrossRef]
- Løvseth, S.W.; Skaugen, G.; Stang, H.G.J.; Jakobsen, J.P.; Wilhelmsen, Ø.; Span, R.; Wegge, R. CO2 mix Project: Experimental Determination of Thermo-physical Properties of CO2-rich Mixtures. Energy Procedia 2013, 37, 7841–7849. [Google Scholar] [CrossRef]
- Joana, S.; Joris, M.; Evangelos, T. Technical and Economic Characteristics of a CO2 Transmission Pipeline Infrastructure; Publications Office: Luxembourg, 2011. [Google Scholar]
- Jia, L.; Tan, Y.; Wang, C.; Anthony, E.J. Experimental study of oxy-fuel combustion and sulfur capture in mini-CFBC. Energy Fuels 2007, 21, 3160–3164. [Google Scholar] [CrossRef]
- Wang, J.; Anthony, E.J. Clean combustion of solid fuels. Appl. Energy 2008, 85, 73–79. [Google Scholar] [CrossRef]
- IEA; GHG. Impact of Impurities on CO2 Capture, Transport and Storage; International Energy Agency Greenhouse Gas R&D Programme: Cheltenham, UK, 2004. [Google Scholar]
- Dall’Acqua, D.; Terenzi, A.; Leporini, M.; D’aLessandro, V.; Giacchetta, G.; Marchetti, B. A new tool for modelling the decompression behaviour of CO2 with impurities using the Peng-Robinson equation of state. Appl. Energy 2017, 206, 1432–1445. [Google Scholar] [CrossRef]
- Wei, L.; Zhang, Y.; Pang, X.; Gao, K. Corrosion behaviors of steels under supercritical CO2 conditions. Corros. Rev. 2015, 33, 151–174. [Google Scholar] [CrossRef]
- Longhi, J. Phase equilibria in the system CO2-H2O I: New equilibrium relations at low temperatures. Geochim. Cosmochim. Acta 2005, 69, 529–539. [Google Scholar] [CrossRef]
- Hu, J.; Duan, Z.; Zhu, C.; Chou, I.M. PVTx properties of the CO2-H2O and CO2-H2O-NaCl systems below 647 K: Assessment of experimental data and thermodynamic models. Chem. Geol. 2007, 238, 249–267. [Google Scholar] [CrossRef]
- Duan, Z.; Hu, J.; Li, D.; Mao, S. Densities of the CO2-H2O and CO2-H2O-NaCl systems up to 647 K and 100 MPa. Energy Fuels 2008, 22, 1666–1674. [Google Scholar] [CrossRef]
- Diamond, L.W. Review of the systematics of CO-H2O fluid inclusions. Lithos 2001, 55, 69–99. [Google Scholar] [CrossRef]
- Chapoy, A.; Mohammadi, A.H.; Chareton, A.; Tohidi, B.; Richon, D. Measurement and Modeling of Gas Solubility and Literature Review of the Properties for the Carbon Dioxide-Water System. Ind. Eng. Chem. Res. 2004, 43, 1794–1802. [Google Scholar] [CrossRef]
- Diamond, L.W.; Akinfiev, N.N. Solubility of CO2 in water from −1.5 to 100 °C and from 0.1 to 100 MPa: Evaluation of literature data and thermodynamic modelling. Fluid Phase Equilibria 2003, 208, 265–290. [Google Scholar] [CrossRef]
- Paulus, M.E.; Penoncello, S.G. Correlation for the carbon dioxide and water mixture based on the Lemmon-Jacobsen mixture model and the Peng-Robinson equation of state. Int. J. Thermophys. 2006, 27, 1373–1386. [Google Scholar] [CrossRef]
- Novitskiy, A.A.; Perez, E.; Wu, W.; Ke, J.; Poliakoff, M. A New continuous method for performing rapid phase equilibrium measurements on binary mixtures containing CO2 or H2O at high pressures and temperatures. J. Chem. Eng. Data 2009, 54, 1580–1584. [Google Scholar] [CrossRef]
- Campos, C.E.P.S.; Villardi, H.G.D.A.; Pessoa, F.L.P.; Uller, A.M.C. Solubility of carbon dioxide in water and hexadecane: Experimental measurement and thermodynamic modeling. J. Chem. Eng. Data 2009, 54, 2881–2886. [Google Scholar] [CrossRef]
- Kunz, O.; Wagner, W. The GERG-2008 wide-range equation of state for natural gases and other mixtures: An expansion of GERG-2004. J. Chem. Eng. Data 2012, 57, 3032–3091. [Google Scholar] [CrossRef]
- Pint, B.A.; Lehmusto, J.; Lance, M.J.; Keiser, J.R. Effect of pressure and impurities on oxidation in supercritical CO2. Mater. Corros. 2019, 70, 1400–1409. [Google Scholar] [CrossRef]
- Gong, Y.; Young, D.J.; Kontis, P.; Chiu, Y.; Larsson, H.; Shin, A.; Pearson, J.; Moody, M.; Reed, R. On the breakaway oxidation of Fe9Cr1Mo steel in high pressure CO2. Acta Mater. 2017, 130, 361–374. [Google Scholar] [CrossRef]
- Gong, Y.; Gill, S.P.A.; Yan, S.; Higginson, R.; Sumner, J.; Simms, N.J.; Larsson, H.; Shin, A.; Pearson, J.M.; Young, D.J.; et al. Assessment of corrosive attack of Fe9Cr1Mo alloys in pressurised CO2 for prediction of breakaway oxidation. Corros. Sci. 2023, 222, 111385. [Google Scholar] [CrossRef]
- Vitse, F.; Nesic, S.; Gunaltun, Y.; de Torreben, D.L.; Duchet-Suchaux, P. Mechanistic Model for the Prediction of Top-of-the-Line Corrosion Risk. Corrosion 2003, 59, 1075–1084. [Google Scholar]
- Nesic, S.; Postlethwaite, J.; Olsen, S. An Electrochemical Model for Prediction of Corrosion of Mild Steel in Aqueous Carbon Dioxide Solutions. Corrosion 1996, 52, 280–294. [Google Scholar] [CrossRef]
- Islam, M.M.; Pojtanabuntoeng, T.; Gubner, R.; Kinsella, B. Electrochemical investigation into the dynamic mechanism of CO2 corrosion product film formation on the carbon steel under the water-condensation condition. Electrochim. Acta 2021, 390, 138880. [Google Scholar] [CrossRef]
- Al-Moubaraki, A.H.; Obot, I.B.; Al-Moubaraki, A.H.; Obot, I.B. Top of the line corrosion: Causes, mechanisms, and mitigation using corrosion inhibitors. Arab. J. Chem. 2020, 14, 103116. [Google Scholar] [CrossRef]
- Singer, M. Study of the Localized Nature of Top of the Line Corrosion in sweet environment. Corrosion 2017, 73, 1030–1055. [Google Scholar] [CrossRef]
- Wang, M.; Tan, M.Y.; Zhu, Y.; Huang, Y.; Xu, Y. Probing top-of-the-line corrosion using coupled multi-electrode array in conjunction with local electrochemical measurement. npj Mater. Degrad. 2023, 7, 16. [Google Scholar] [CrossRef]
- Schremp, F.W.; Erson, G.R.R. Effect of Supercritical Carbon Dioxide (CO2) on Construction Materials. Soc. Pet. Eng. J. 1975, 15, 227–233. [Google Scholar] [CrossRef]
- Eslami, M.; Farah Santos, B.A.; Young, D.; Serenário, M.E.D.; Gjertsen, S.; Singer, M. Influence of High CO2 Partial Pressure on Top-of-the-Line Corrosion. In Proceedings of the AMPP Annual Conference and Expo 2024 Association for Materials Protection and Performance, New Orleans, LA, USA, 3–7 March 2024; pp. 1–15. [Google Scholar]
- West, J. Design and operation of a supercritical CO2 pipelinecompression system: Sacroc Unit, Scurry County, Texas. In Proceedings of the SPE Permian Basin Oil Recovery Conference, Midland, TX, USA, 11–12 March 1974; p. 4804. [Google Scholar]
- Russick, E.M.; Poulter, G.A.; Adkins, C.L.J.; Sorensen, N.R. Corrosive Effects of Supercritical Carbon Dioxide and Cosolvents on Metals. J. Supercrit. Fluids 1996, 9, 43–50. [Google Scholar] [CrossRef]
- Zhang, Y.C.; Gao, K.W.; Schmitt, G. Water effect on steel corrosion under supercritical CO2 conditions. In Proceedings of the Corrosion Association for Materials Protection and Performance, Houston, TX, USA, 13–17 March 2011; p. 11378. [Google Scholar]
- Propp, W.A.; Carleson, T.E.; Wai, C.M.; Taylor, P.R.; Daehling, K.W.; Huang, S.; Abdel-Latif, M. Corrosion in Supercritical Fluids; Idaho National Engineering Laboratory: Washington, DC, USA, 1996. [Google Scholar]
- Gill, T. CO2 pipeline: Description and 12 years of operation. In Proceedings of the Pipeline Engineering Symposium, New York, NY, USA, 17–21 February 1985; p. 59. [Google Scholar]
- Newton, L.E., Jr. SACROC CO2 project-corrosion problems and solutions. In Proceedings of the Corrosion 1984; NACE: New Orleans, LA, USA, 1984; Volume 23, pp. 17–23. [Google Scholar]
- Collier, J.; Papavinasam, S.; Li, J.; Shi, C.; Liu, P.; Gravel, J.-P. Effect of Impurities on the Corrosion Performance of Steels in Supercritical Carbon Dioxide: Optimization of Experimental Procedure. In Proceedings of the Corrosion 2013; NACE: Orlando, FL, USA, 2013. [Google Scholar]
- Beck, J.; Lvov, S.; Fedkin, M.; Ziomek-Moroz, M.; Holcomb, G.; Tylczak, J.; Alman, D. Electrochemical system to study corrosion of metals in supercritical CO2 fluids. In Proceedings of the CORROSION 2011 Association for Materials Protection and Performance; NACE International: Houston, TX, USA, 2011; p. 11380. [Google Scholar]
- Gale, J.; Davison, J. Transmission of CO2-safety and economic considerations. Energy 2004, 29, 1319–1328. [Google Scholar] [CrossRef]
- Choi, Y.S.; Nesic, S. Effect of water content on the corrosion behavior of carbon steel in supercritical CO2 phase with impurities. In Proceedings of the Corrosion 2011 Association for Materials Protection and Performance; NACE International: Houston, TX, USA, 2011. [Google Scholar]
- Farelas, F.; Choi, Y.; Nešić, S. Corrosion behavior of API X65 carbon steel under supercritical and liquid carbon dioxide phases in the presence of water and sulfur dioxide. Corros. Sci. 2013, 243–250. [Google Scholar] [CrossRef]
- Dugstad, A.; Morland, B.; Clausen, S. Corrosion of transport pipelines for CO2—Effect of water ingress. Energy Procedia 2011, 4, 3063–3070. [Google Scholar] [CrossRef]
- Dugstad, A.; Clausen, S.; Morland, B. Transport of dense phase CO2 in C-steel pipelines-when is corrosion an issue? In Proceedings of the Corrosion 2011 Association for Materials Protection and Performance; NACE International: Houston, TX, USA, 2011. [Google Scholar]
- Hua, Y.; Barker, R.; Neville, A. Effect of temperature on the critical water content for general and localised corrosion of X65 carbon steel in the transport of supercritical CO2. Int. J. Greenh. Gas Control 2014, 31, 48–60. [Google Scholar] [CrossRef]
- Newton, L.E., Jr.; McClay, R.A. Corrosion and operational problems, CO2 project. In Proceedings of the SPE Permian Basin Oil and Gas Recovery Conference, Midland, TX, USA, 10–11 March 1977. [Google Scholar]
- Ayello, F.; Evans, K.J.; Sridhar, N.; Thodla, R. Effect of Liquid Impurities on Corrosion of Carbon Steel in Supercritical CO2. In Proceedings of the International Pipeline Conference, Calgary, AB, Canada, 27 September–1 October 2010; pp. 111–123. [Google Scholar]
- Dugstad, A.; Halseid, M.; Morland, B. Effect of SO2 and NO2 on corrosion and solid formation in dense phase CO2 pipelines. Energy Procedia 2013, 37, 2877–2887. [Google Scholar] [CrossRef]
- Sim, S.; Bocher, F.; Cole, I.S.; Chen, X.-B.; Birbilis, N. Investigating the effect of water content in supercritical CO2 as relevant to the corrosion of Carbon capture and storage pipelines. Corrosion 2014, 70, 185–195. [Google Scholar] [CrossRef]
- Thodla, R.; Francois, A.; Sridhar, N. Materials performance in supercritical CO2 environments. In Proceedings of the Corrosion 2009 Association for Materials Protection and Performance; NACE International: Houston, TX, USA, 2009. [Google Scholar]
- Dugstad, A.; Halseid, M.; Morland, B.; Clausen, S. Dense phase CO2 transport—When is corrosion a threat? In Proceedings of the Northern Area Western Conference, Vancouver, BC, Canada, 12 November 2011. [Google Scholar]
- Cabrini, M.; Lorenzi, S.; Pastore, T.; Radaelli, M. Corrosion rate of high CO2 pressure pipeline steel for carbon capture transport and storage. La Metal. Ital. 2014, 6, 21–27. [Google Scholar]
- Choi, Y.S.; Nesic, S. Corrosion Behavior of Carbon Steel in Supercritical CO2-Water Environments. In Proceedings of the CORROSION 2009 Association for Materials Protection and Performance; NACE International: Houston, TX, USA, 2010. [Google Scholar]
- Choi, Y.S.; Nesic, S.; Young, D. Effect of impurities on the corrosion behavior of CO2 transmission pipeline steel in supercritical CO2-water environments. Environ. Sci. Technol. 2010, 44, 9233–9238. [Google Scholar] [CrossRef]
- Ayello, F.; Evans, K.; Thodla, R.; Sridhar, N. Effect of impurities on corrosion of steel in supercritical CO2. In Proceedings of the CORROSION 2010 Association for Materials Protection and Performance; NACE International: Houston, TX, USA, 2010. [Google Scholar]
- Seiersten, M. Material selection for separation, transportation and disposal of CO2. In Proceedings of the Corrosion Association for Materials Protection and Performance; NACE International: Houston, TX, USA, 2001; pp. 1–10. [Google Scholar]
- Nyborg, R. Overview of CO2 Corrosion Models for Wells and Pipelines. In Proceedings of the NACE International Annual Conference, Denver, Colorado, CO, USA, 7–11 April 2002. [Google Scholar]
- NORSOK M-506:2005; CO2 Corrosion Rate Calculation Model. Norwegian Technological Standards Institute: Lysaker, Norway, 2005.
- Lorenzi, S.; Cabrini, M.; Carugo, F.; Pastore, T. Studio della corrosione in condense sature di CO2. La Metall. Ital. 2022, 52–56. [Google Scholar]
- Sala, S.; Marina, C.; Tommaso, P. Studio Della Corrosione ad Alte Pressioni Parziali di CO2. Master’s Thesis, Univeristy of Bergamo, Bergamo, Italy, 2020. [Google Scholar]
- Alami, H.E.; Augustin, C.; Orlans, B.; Servier, J.J. Carbon capture and storage projects: Material integrity for CO2 injection and storage. In Proceedings of the EuroCorr, Stockholm, Sweden, 4–8 September 2011; p. 4741. [Google Scholar]
- Belarbi, Z.; Vu, T.N.; Farelas, F.; Young, D.; Singer, M.; Nesic, S. Thiols as volatile corrosion inhibitors for top-of-the-line corrosion. Corros. NACE 2017, 73, 892–899. [Google Scholar] [CrossRef] [PubMed]
- Rafefi, A. Development and evaluation of thin film electrical resistance sensors for monitoring CO2 top of the line corrosion. Sens. Actuators B Chem. 2021, 346, 130492. [Google Scholar] [CrossRef]
- Rohsenow, W.M.; Hartnett, J.R.; Cho, Y.I. (Eds.) Handbook of Heat Transfer; Mcgraw-Hill: New York, NY, USA, 1998. [Google Scholar]
- Gunaltun, Y.M.; Larrey, D. Water condensation rate critical. Oil Gas J. 2000, 98, 58. [Google Scholar]
- Ruhl, A.S.; Kranzmann, A. Corrosion in supercritical CO 2 by diffusion of flue gas acids and water. J. Supercrit. Fluids 2012, 68, 81–86. [Google Scholar] [CrossRef]
- Choi, Y.S.; Hassani, S.; Vu, T.N.; Nešić, S.; Abas, A.Z.B. Effect of H2S on the corrosion behavior of pipeline steels in supercritical and liquid CO2 environments. Corrosion 2016, 72, 999–1009. [Google Scholar] [CrossRef]
- Hashizume, S.; Kobayashi, N.; Trillo, E. Corrosion performance of CRAs in water containing chloride ions under supercritical CO2. In Proceedings of the Corrosion; NACE International: Houston, TX, USA, 2013; p. 2264. [Google Scholar]
- Xiang, Y.; Wang, Z.; Li, Z.; Ni, W.D. Effect of temperature on corrosion behaviour of X70 steel in high pressure CO2/SO2/O2/H2O environments. Corros. Eng. Sci. Technol. 2013, 48, 121–129. [Google Scholar] [CrossRef]
- Xiang, Y.; Wang, Z.; Li, Z.; Ni, W.D. Effect of Exposure Time on the Corrosion Rates of X70 Steel in Supercritical CO2/SO2/O2/H2O Environments. Corrosion 2012, 69, 251–258. [Google Scholar] [CrossRef]
- Hua, Y.; Barker, R.; Neville, A. Understanding the Influence of SO2 and O2 on the Corrosion of Carbon Steel in Water-Saturated Supercritical CO2. Corrosion 2014, 71, 667–683. [Google Scholar] [CrossRef]
- Barker, R.; Hua, Y.; Neville, A. Internal corrosion of carbon steel pipelines for dense-phase CO2 transport in carbon capture and storage (CCS)—A review. Int. Mater. Rev. 2017, 62, 1–31. [Google Scholar] [CrossRef]
- Hua, Y.; Shamsa, A.; Barker, R.; Neville, A. Protectiveness, morphology and composition of corrosion products formed on carbon steel in the presence of Cl−, Ca2+ and Mg2+ in high pressure CO2 environments. Appl. Surf. Sci. 2018, 455, 667–682. [Google Scholar] [CrossRef]
- Xu, M.; Zhang, Q.; Yang, X.; Wang, Z.; Liu, J.; Li, Z. Impact of surface roughness and humidity on X70 steel corrosion in supercritical CO2 mixture with SO2, H2O, and O2. J. Supercrit. Fluids 2016, 107, 286–297. [Google Scholar] [CrossRef]
- Xu, M.; Zhang, Q.; Wang, Z.; Liu, J.; Li, Z. Effect of High-Concentration O2 on Corrosion Behavior of X70 Steel in Water-Containing Supercritical CO2 with SO2. Corrosion 2016, 73, 290–302. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Sun, C.; Wang, Y. Effects of O2 and SO2 on Water Chemistry Characteristics and Corrosion Behavior of X70 Pipeline Steel in Supercritical CO2 Transport System. Ind. Eng. Chem. Res. 2018, 57, 2365–2375. [Google Scholar] [CrossRef]
- Sun, C.; Liu, J.; Sun, J.; Lin, X.; Wang, Y. Probing the initial corrosion behavior of X65 steel in CCUS-EOR environments with impure supercritical CO2 fluids. Corros. Sci. 2021, 189, 109585. [Google Scholar] [CrossRef]
- Sun, C.; Wang, Y.; Sun, J.; Lin, X.; Li, X.; Liu, H.; Cheng, X. Effect of impurity on the corrosion behavior of X65 steel in water-saturated supercritical CO2 system. J. Supercrit. Fluids 2016, 116, 70–82. [Google Scholar] [CrossRef]
- Yevtushenko, O.; Bäßler, R. Water impact on corrosion resistance of pipeline steels in circulating supercritical CO2 with SO2-and NO2-impurities. In Proceedings of the Corrosion; NACE International: Houston, TX, USA, 2014; p. 3838. [Google Scholar]
- Wei, L.; Pang, X.; Gao, K. Corrosion of low alloy steel and stainless steel in supercritical CO2/H2O/H2S systems. Corros. Sci. 2016, 111, 637–648. [Google Scholar] [CrossRef]
- Bäßler, R. Corrosion stability of piping steels in a circulating supercritical impure CO2 environment. In Proceedings of the Kolloquium Uni Potsdam, Potsdam, Germany, 26–28 October 2016. [Google Scholar]
- Yevtushenko, O.; Bettge, D.; Bohraus, S.; Bäßler, R.; Pfennig, A.; Kranzmann, A. Corrosion behavior of steels for CO2 injection. Process Saf. Environ. Prot. 2014, 92, 108–118. [Google Scholar] [CrossRef]
- Paschke, B.; Kather, A. Corrosion of Pipeline and Compressor Materials Due to Impurities in Separated CO2 from Fossil-Fuelled Power Plants. Energy Procedia 2012, 23, 207–215. [Google Scholar] [CrossRef]
- Annesini, M.C.; Augelletti, R.; De Filippis, P.; Scarsella, M.; Verdone, N. Sviluppo di un Processo di Separazione Della CO2 dal Biogas Mediante Assorbimento con Soluzioni Amminiche in Solvente Organico; ENEA: Rome, Italy, 2013. [Google Scholar]
- Sun, C.; Sun, J.; Wang, Y.; Lin, X.; Li, X.; Cheng, X.; Liu, H. Synergistic effect of O2, H2S and SO2 impurities on the corrosion behavior of X65 steel in water-saturated supercritical CO2 system. Corros. Sci. 2016, 107, 193–203. [Google Scholar] [CrossRef]
- Xiang, Y.; Xu, M.; Choi, Y.S. State-of-the-art overview of pipeline steel corrosion in impure dense CO2 for CCS transportation: Mechanisms and models. Corros. Eng. Sci. Technol. 2017, 52, 485–509. [Google Scholar] [CrossRef]
- Sibhat, M.; Zhu, Q.; Tsegay, G.; Yao, G.; Yin, G.; Zhou, Y.; Zhao, J. Enhancement technologies of ammonia-based carbon capture: A review of developments and challenges. Int. J. Greenh. Gas Control 2024, 136, 104196. [Google Scholar] [CrossRef]
- Kongshaug, K.O.; Seiersten, M. Baseline experiments for the modelling of corrosion at high CO2 pressure. In Proceedings of the CORROSION; NACE International: Houston, TX, USA, 2004; p. 046300. [Google Scholar]





| Component | Hazard(s) in a CCU Context | Units | Limit |
|---|---|---|---|
| CO2 | Asphyxiation, and can act as a toxicant at high concentrations | mol% | >95.0 |
| N2 a | Enhances the potential for ductile fracture Occupies store pore space inefficiently | mol% | ≤4.0 |
| H2 a,b,c | Enhances the potential for ductile fracture and hydrogen induced crack propagation Affects the size of the multi-phase zone | mol% | ≤1.0 |
| Ar a | Occupies store pore space inefficiently, enhanced potential for running ductile fractures | mol% | ≤4.0 |
| CO a | Health and safety: toxic gas | mol% | ≤0.2 |
| Methane a | Occupies store pore space inefficiently | mol% | ≤4.0 |
| Ethane a | Occupies store pore space inefficiently | mol% | ≤4.0 |
| Propane and other aliphatic hydrocarbons d | Liquid drop-out is possible | mol% | ≤0.15 in total |
| H2O | Enables corrosion of carbon steel | ppm mol | ≤50 |
| O2 b,e | Enables oxidation of carbon steel Enhances bacterial growth in storage strata Other chemical reactions (e.g., with NOx, SOx, H2S) | ppm mol | ≤10 |
| NOx (NO, NO2) f | Degradation of store caprock Takes place in the production of nitric and sulfuric acid | ppm mol | ≤10 |
| SOx (SO, SO2, SO3) g | Degradation of store caprock Reactions with NO2 can produce sulfuric acid | ppm mol | ≤10 |
| H2S h | Health and safety: toxic gas with foul odour | ppm mol | ≤5 |
| COS | Health and safety: toxic gas with foul odour | ppm mol | ≤100 |
| CS2 | Health and safety: toxic gas with foul odour | ppm mol | ≤20 |
| NH3 | Can react to form solid ammonium carbamate and other ammonium salts | ppm mol | ≤10 |
| BTEX i | Health and safety: toxic | ppm mol | ≤15 in total |
| Methanol | Can introduce a liquid corrosive phase | ppm mol | ≤350 |
| Solid particulates j,k | Can reduce store permeability. Damage to compressor components | mg/Nm3 | ≤1 in total |
| Toxic metal j | Health and safety: toxic | mg/Nm3 | ≤0,15 |
| VOCs l | Health and safety: toxic | mg/Nm3 | ≤48 in total |
| Acid forming compounds m | Enables corrosion of carbon steel | mg/Nm3 | ≤150 in total |
| Amines n,o | Can introduce a liquid corrosive phase | ppb mol | ≤100 in total |
| Glycols p | Enables aqueous corrosion of carbon steel | — | |
| Nitrosamines and nitramines q | Health and safety: bio-toxic | μg/Nm3 | ≤3 in total |
| Naphthalene | Health and safety: toxic | ppb mol | ≤100 |
| Dioxins and furans r | Health and safety: toxic | ng/Nm3 | ≤0.02 in total |
| Component | Notes | Units | Limit |
|---|---|---|---|
| CO2 | Dry basis | mol% | >95.0 |
| N2 | Total non-condensables to be <5 mol% | mol% | a,b |
| H2 | mol% | ≤1 | |
| Ar | mol% | a,b | |
| CO | mol% | ≤0.7 | |
| Methane | mol% | ||
| Ethane | mol% | ||
| Propane and other aliphatic hydrocarbons | Total hydrocarbons to be <5 mol% and a dew point of product with respect to hydrocarbons to be <−20 °C. | mol% | ≤1 |
| H2O | The limit for water may be higher (e.g., 630 ppm mol) if the CO2 stream contains very low levels of O2, NOx, and SOx (e.g., geological CO2).b | ppm mol | ≤100 |
| O2 | ppm mol | ≤10 | |
| NOx (NO, NO2) | ppm mol | ≤1.5 | |
| SOx (SO, SO2, SO3) | ppm mol | ≤1 | |
| H2S | ppm mol | ≤55 | |
| Total sulphur | ppm mol | ≤50 | |
| Solid particulates | ppm wt | ≤1 | |
| Mercury | ng/L | ≤5 | |
| Amines | ppm wt | ≤1 | |
| Glycols | Must not be present in a liquid state at the temperature and pressure conditions of the pipeline. | ppm mol | ≤50 |
| Compressor lube oil carry-over | ppm wt | ≤50 ppmw | |
| Liquids | CO2 stream shall be free of liquids at delivery conditions and shall not produce condensed liquids in the pipeline at pipeline temperature and pressure. | ||
| No. | Source and Type of Plant | Transported Mixture | Reference |
|---|---|---|---|
| 1 | CO2 stream from oxy-fuel combustion in a fluidized bed combustor at the CanmetENERGY pilot plant | 5.2 vol% O2, 221 ppm CO, 1431 ppm SO2, 243 ppm NO | [28] |
| 2 | CO2 stream from a zero-emission process proposed by CanmetENERGY | 1.05% CO, 1.7% SO2, 0.32% H2, 690 ppm H2S | [29] |
| 3 | CO2 stream from the Cansolv® absorption system | 2.9% SO2 | [30], as reported in a previous IEA GHG report |
| 4 | Predicted CO2 stream from a pre-combustion capture plant | 1 vol% H2, 0.9 vol% N2, 300 ppm Ar, 100 ppm H2S, COS and other impurities | Composition data provided by IEA GHG [30] |
| 5 | Predicted CO2 stream from an oxy-fuel combustion plant | 5.8 vol% N2, 4.7 vol% O2, 4.47 vol% Ar, 100 ppm NOx, 50 ppm SO2, 20 ppm SO2, 50 ppm CO | Composition data provided by IEA GHG [30] |
| N° | P (bar) | T (°C) | H2O (ppmv) | Material | t (h) | Flow | Corrosion Rate (mm/year) | Refs. |
|---|---|---|---|---|---|---|---|---|
| 1 | 80 | 40 | 244 | C-steel | 168 | Static | 0.08 | [72] |
| 11 | 80 | 50 | Sat | X65 | 14, 24, 48 | Static | 0.024 ≈ 0.1 | [71] |
| 26 | 79.6–82 | 35 | 10 g | SS: 304 L, 3161 C-Steel: X42, X60 | 120 | 100 rpm | SS: 0.0005–0.0008 C Steel: 0.007 | [61] |
| 29 | 80 | 50 | 650 (Undersat) | X65 | 24 | Static | No corrosion | [64] |
| 30 | 80 | 50 | 3310 (Sat) | X65 | 24 | Static | 0.38 | [64,65,76] |
| 31 | 80 | 50 | 10 g (Sat.) | X65 | 24 | Static | 0.4–1 | [65] |
| 32 | 75.8 | 40 | 244 | C Steel | 5 | Static | 1.2 | [73,74,77] |
| 35 | 79 | 31 | 244 | C steel | 5 | Static | 1.1 | [73] |
| 37 | 95 182 | 50–130 | 100 g (Sat.) | C steel | 96 | 995 | 0.014–0.043 | [63] |
| 39 | 100 | 20 | 1220 | X65 | 720 | Static | No corrosion | [78] |
| 45 | 100 | 25 | 488 e 1222 | X65 | 336 | 3 rpm | 0 | [67] |
| 46 | 125 | 80 | 1.5 g | 38Mn6/C75 | 96 | 995 | 0.0036 | [63] |
| 49 | 123–146 | 25–60 | Saturated | X65 | 48–400 | 180 rpm | 0.01–0.1 | [75] |
| 50 | 240 | 50 | 40 g (Sat.) | 24 | Static | Not given | [56] |
| Environment | Corrosion Rate (mm/years) | Refs. |
|---|---|---|
| H2O-rich phases | 19.2 | [63,64,65,75] |
| 10.6 | [75] | |
| 5–15 | [32] | |
| 0.6 | [78] | |
| 1.7 | [78] | |
| 5–30 | [81,82] | |
| CO2-rich phases | 0.38 | [75] |
| 0.013–0.043 | [32] | |
| 0.04 | [78] | |
| 0.01–0.1 | [81,82] |
| N° | P | T | H2O | O2 | Steel | t | Flow | Corrosion Rate | Refs. |
|---|---|---|---|---|---|---|---|---|---|
| (bar) | (°C) | (ppmv) | (ppmv) | (h) | (rpm) | (mm/anno) | |||
| 1 | 75.8 | 40 | 2440 | 100 | 5 | Static | [74] | ||
| 2 | 80 | 50 | Saturated (10 g) | 0 | X65 | 24 | static | 0.38 | [65] |
| 2% (1.6 bar) | 0.6 | ||||||||
| 4% (3.3 bar) | 1 | ||||||||
| 6% (5.1 bar) | 0.9 | ||||||||
| 3 | 80 | 50 | 650 | 3.3 bar | X65 | 24 | Static | NO corrosion | [64] |
| 2000 | 4% | NO corrosion | |||||||
| 3000 | ≤0.01 | ||||||||
| 4 | 79.6–82 | 35 | Saturated (100 g) | 0 | 304 L | 120 | 100 | 0.002 | [61] |
| 316 L | 0.001 | ||||||||
| X42 | 0.014 | ||||||||
| X60 | |||||||||
| 5 | 94.8–103 | 49 | Saturated (100 g) | 3 v% | 304 L | 120 | 100 | 0.003 | [61] |
| 316 L | 0.004 | ||||||||
| X42 | 0.099 | ||||||||
| X60 | 0.093 | ||||||||
| 6 | 100 | 60 | Saturated | Yes | X42 | 120 | Static | 0.008 | [89] |
| (1 mL 55.6 mmol) | (−1000 ppm) | (−1 mg) No corrosion | [89] | ||||||
| 7 | 100 | 20 | 1220 | 488 | x65 | 720 | Static | No corrosion | [70] |
| 8 | 100 | 10 | 50 v% | 0 | X65 | 312 | Static | 0.5 | [70] |
| 20 (Saturated) | 0.336 | 0.8 | |||||||
| 50 | 0 | 336 | 0.5 | ||||||
| 50 | 0 | 336 | 2.7 | ||||||
| 10 | 200 | 312 | 1.2 | ||||||
| 20 | 100 | 336 | 1.3 | ||||||
| 50 | 200 | 432 | 0.6 (pit corrosion rate 17) | ||||||
| 9 | 150 | 80 | Saturated | 1000 ppm | 288 | 120 | 0.2–0.9 | [90] | |
| 10 | 150 | 100 | 5% NaCl | 0.045 bar | Type 420 | 720 | static | 0.08 Localized | [88] |
| 0.45 bar | 0.25 Localized | ||||||||
| 11 | 300 | 100 | 5% NaCl | 0.045 bar | Type 420 | 720 | static | 0.07 Negligible localized | [88] |
| 0.45 bar | 0.34 Localized | ||||||||
| 12 | 150 | 100 | 5% NaCl | 0.0045 bar | M-SS | 720 | Static | 0.01 localized | [88] |
| 0.45 bar | 0.04 Localized | ||||||||
| 13 | 300 | 100 | 5% NaCl | 0.045 bar | M-SS | 720 | Statie | 0.02 No Localized | [88] |
| 0.45 bar | 0.07 localized | ||||||||
| 14 | 100 | 12 | 1000 | X65 | 48–96 | 0–3 m/s | 5 to10 | [70] | |
| 13 | 0 | 3 to 5 | |||||||
| 15 | 80 | 50 | 400 ml Water phase | 4%; 0 | X65 | 24 | Static | 19.3; 19.2 | [76] |
| 120 | 14.1; 10.6 | ||||||||
| 16 | 80 | 50,400 mL Water phase | 4% | 3Cr | Static | 0.01 | [64,65] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Gritti, L.; Coffetti, D.; Nani, L.; Lorenzi, S.; Cabrini, M. Corrosion Behaviour in CO2 Pipeline Transport: A Review of the Impact of Condensates and Impurities. Materials 2026, 19, 2048. https://doi.org/10.3390/ma19102048
Gritti L, Coffetti D, Nani L, Lorenzi S, Cabrini M. Corrosion Behaviour in CO2 Pipeline Transport: A Review of the Impact of Condensates and Impurities. Materials. 2026; 19(10):2048. https://doi.org/10.3390/ma19102048
Chicago/Turabian StyleGritti, Luca, Denny Coffetti, Lorenzo Nani, Sergio Lorenzi, and Marina Cabrini. 2026. "Corrosion Behaviour in CO2 Pipeline Transport: A Review of the Impact of Condensates and Impurities" Materials 19, no. 10: 2048. https://doi.org/10.3390/ma19102048
APA StyleGritti, L., Coffetti, D., Nani, L., Lorenzi, S., & Cabrini, M. (2026). Corrosion Behaviour in CO2 Pipeline Transport: A Review of the Impact of Condensates and Impurities. Materials, 19(10), 2048. https://doi.org/10.3390/ma19102048












