Preparation and Characterization of Bacterial Cellulose/Carboxymethyl Cellulose Composite Films
Highlights
- In situ CMC incorporation densified the BC nanofiber network without changing cellulose I.
- Citric acid post-treatment possibly promoted ester-linkage-assisted network stabilization and contributed to improved integrated properties.
- BC/CMC-3 favored retention, strength, and barrier performance, whereas BC/CMC-4 favored re-hydration.
- CMC dosage can be used to tailor BC films for moisture-management-related applications.
- The strategy provides a simple route for regulating water management and mechanics simultaneously.
- The XNH6-based system shows promise for high-value BC composite material development.
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
2.2. Instruments
2.3. Experimental Procedures
2.3.1. Screening and Identification of BC-Producing Strains
2.3.2. Cultivation of XNH6 and Purification of BC
2.3.3. Preparation of BC/CMC Composite Films
2.3.4. Citric Acid Post-Treatment of BC/CMC Composite Films
2.4. Characterization of BC/CMC Composite Films
2.4.1. FTIR
2.4.2. XPS
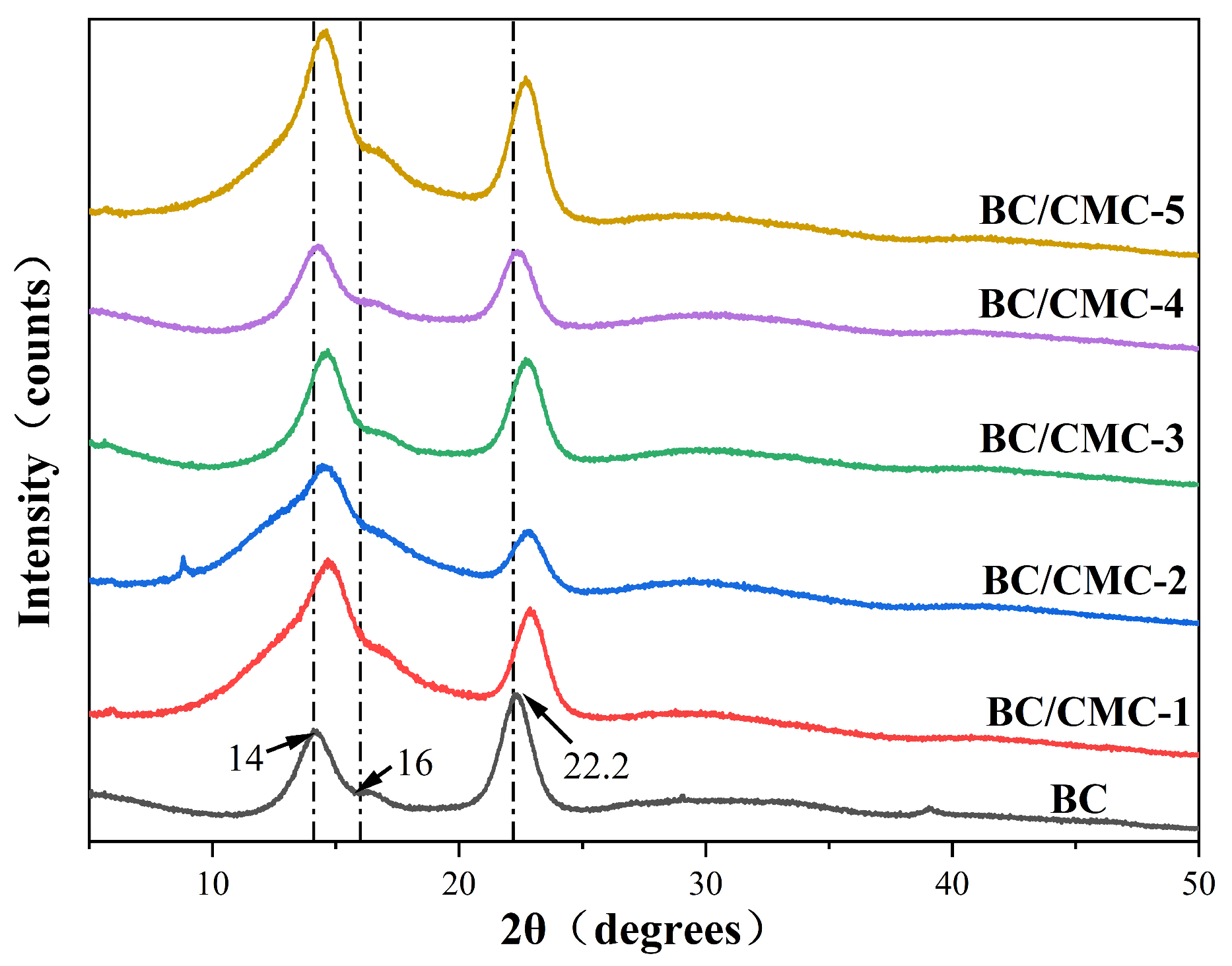
2.4.3. XRD
2.4.4. SEM
2.4.5. Water Retention and Re-Hydration Measurements
2.4.6. Water Vapor Transmission Rate (WVTR)
2.4.7. Water Contact Angle
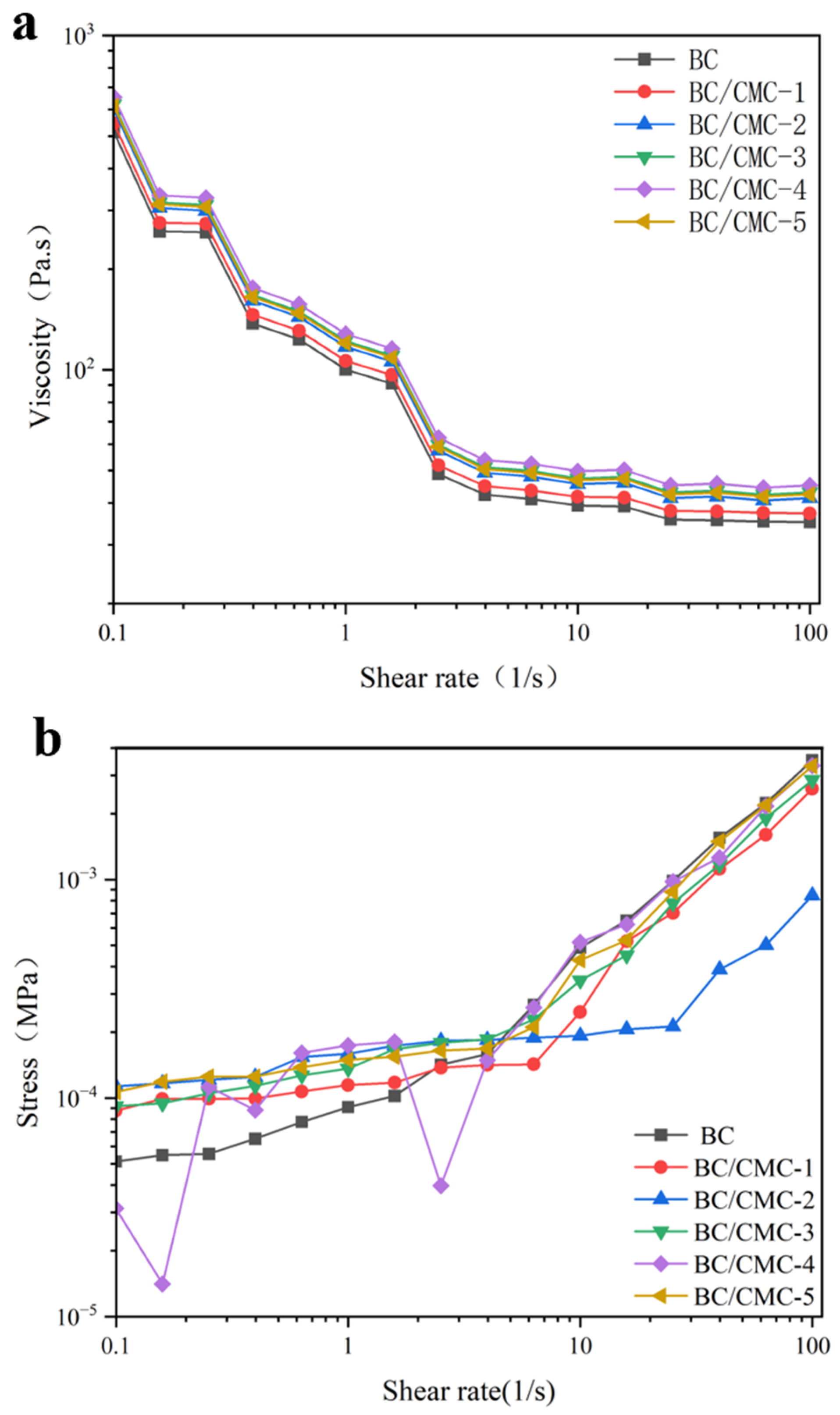
2.4.8. Rheological Measurements
2.4.9. Mechanical Properties
2.5. Statistical Analysis
3. Results
3.1. Isolation and Identification of the BC-Producing Strain
3.2. Structural and Functional Characterization of BC/CMC Composite Films
3.2.1. FTIR Analysis
3.2.2. XPS Analysis
3.2.3. XRD Analysis
3.2.4. SEM Analysis
3.2.5. Water Retention and Re-Hydration Properties
3.2.6. Water Vapor Transmission Rate
3.2.7. Water Contact Angle Analysis
3.2.8. Rheological Properties
3.2.9. Mechanical Properties Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| FTIR | Fourier transform infrared spectroscopy |
| XPS | X-ray photoelectron spectrometer |
| XRD | X-Ray diffraction |
| SEM | Scanning electron microscope |
| HS | Hestrin-Schramm |
References
- Liu, M.; Shen, X.; Guo, F.; Jiang, W.; Zhang, W.; Xin, F.; Jiang, Y.; Jiang, M. Unraveling the biosynthesis and regulation mechanism of bacterial cellulose: Challenges and strategies toward industrial production. Carbohydr. Polym. 2026, 377, 124891. [Google Scholar] [CrossRef]
- Navya, P.V.; Gayathri, V.; Samanta, D.; Sampath, S. Bacterial cellulose: A promising biopolymer with interesting properties and applications. Int. J. Biol. Macromol. 2022, 220, 435–461. [Google Scholar] [CrossRef]
- Gregory, D.A.; Tripathi, L.; Fricker, A.T.R.; Asare, E.; Orlando, I.; Raghavendran, V.; Roy, I. Bacterial cellulose: A smart biomaterial with diverse applications. Mater. Sci. Eng. R-Rep. 2021, 145, 100623. [Google Scholar] [CrossRef]
- Lu, Q.M.; Yu, X.J.; Yagoub, A.A.; Wahia, H.; Zhou, C.S. Application and challenge of nanocellulose in the food industry. Food Biosci. 2021, 43, 101285. [Google Scholar] [CrossRef]
- Campano, C.; Rivero-Buceta, V.; Hernandez-Arriaga, A.M.; Manoli, M.T.; Prieto, M.A. Pushing the limits of bacterial cellulose for biomedicine: A review. Int. J. Biol. Macromol. 2025, 323, 146701. [Google Scholar] [CrossRef]
- Pan, X.; Li, J.; Ma, N.; Ma, X.; Gao, M. Bacterial cellulose hydrogel for sensors. Chem. Eng. J. 2023, 461, 142062. [Google Scholar] [CrossRef]
- Chen, K.; Li, Y.; Du, Z.; Hu, S.; Huang, J.; Shi, Z.; Su, B.; Yang, G. CoFe2O4 embedded bacterial cellulose for flexible, biodegradable, and self-powered electromagnetic sensor. Nano Energy 2022, 102, 107740. [Google Scholar] [CrossRef]
- Santos, M.R.D.; Durval, I.J.B.; Medeiros, A.D.M.; Silva Júnior, C.; Converti, A.; Costa, A.F.S.; Sarubbo, L.A. Biotechnology in Food Packaging Using Bacterial Cellulose. Foods 2024, 13, 3327. [Google Scholar] [CrossRef]
- Zhang, Y.; Sun, D.-W.; Xu, L. Bacterial cellulose-driven sustainable food packaging innovations: Biosynthesis, functionalization, and applications. Trends Food Sci. Technol. 2025, 163, 105167. [Google Scholar] [CrossRef]
- Chen, S.; Tang, H.; Fan, X.; Li, B.; Wang, Y.; Zhou, W.; Jiang, X.; Dong, X.; Wang, Y.; Zhao, P.; et al. Bio-orthogonal functionalization of bacterial cellulose combining metabolic glycoengineering and click chemistry. Nat. Commun. 2026, 17, 2304. [Google Scholar] [CrossRef] [PubMed]
- Turganova, R.; Tuleyeva, R.; Belkozhayev, A.; Gizatullina, N.; Yelemessova, G.; Taubatyrova, A.; Mussalimova, M.; Shynykul, Z.; Toleutay, G. Bacterial Cellulose for Sustainable Food Packaging: Production Pathways, Structural Design, and Functional Modification Strategies. Polymers 2025, 17, 3165. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Li, C.; Tang, Y. Constructing bacterial cellulose and its composites: Regulating treatments towards applications. Cellulose 2024, 31, 7793–7817. [Google Scholar] [CrossRef]
- Kumaravel, A.; Lin, S.-P.; Santoso, S.P.; Shanmugasundaram, S.; Hsu, H.-Y.; Hsieh, C.-W.; Chou, Y.-C.; Lin, H.-W.; Cheng, K.-C. Unlocking the potential of bacterial cellulose: Synthesis, functionalization, and industrial impact. Int. J. Biol. Macromol. 2025, 311, 143951. [Google Scholar] [CrossRef]
- Liu, X.; Sun, T.; Tao, Q.; Bai, X.; Zhao, Q. Review on bacterial cellulose in sustainable food systems: Structures, modifications, and emerging applications. Food Biosci. 2025, 72, 107534. [Google Scholar] [CrossRef]
- Revin, V.V.; Liyaskina, E.V.; Parchaykina, M.V.; Kuzmenko, T.P.; Kurgaeva, I.V.; Revin, V.D.; Ullah, M.W. Bacterial Cellulose-Based Polymer Nanocomposites: A Review. Polymers 2022, 14, 4670. [Google Scholar] [CrossRef]
- Busuioc, C.; Isopencu, G.O.; Deleanu, I.-M. Bacterial Cellulose–Polyvinyl Alcohol Based Complex Composites for Controlled Drug Release. Appl. Sci. 2023, 13, 1015. [Google Scholar] [CrossRef]
- Rzhepakovsky, I.; Piskov, S.; Avanesyan, S.; Sizonenko, M.; Timchenko, L.; Anfinogenova, O.; Nagdalian, A.; Blinov, A.; Denisova, E.; Kochergin, S.; et al. Composite of bacterial cellulose and gelatin: A versatile biocompatible scaffold for tissue engineering. Int. J. Biol. Macromol. 2024, 256, 128369. [Google Scholar] [CrossRef] [PubMed]
- Liu, K.; Wang, Y.; Liu, W.; Zheng, C.; Xu, T.; Du, H.; Yuan, Z.; Si, C. Bacterial cellulose/chitosan composite materials for biomedical applications. Chem. Eng. J. 2024, 494, 153014. [Google Scholar] [CrossRef]
- Munhoz, L.L.S.; Guillens, L.C.; Alves, B.C.; Nascimento, M.; Meneguin, A.B.; Carbinatto, F.M.; Arruda, G.; Barud, H.D.S.; Aro, A.; Casagrande, L.R.; et al. Bacterial nanocellulose/calcium alginate hydrogel for the treatment of burns. Acta Cirúrgica Bras. 2024, 39, e393324. [Google Scholar] [CrossRef]
- Pasaribu, K.M.; Mahendra, I.P.; Karina, M.; Masruchin, N.; Sholeha, N.A.; Gea, S.; Gupta, A.; Johnston, B.; Radecka, I. A review: Current trends and future perspectives of bacterial nanocellulose-based wound dressings. Int. J. Biol. Macromol. 2024, 279, 135602. [Google Scholar] [CrossRef] [PubMed]
- Campebell, R.C.; Oliveira, A.B.; Fagundes, J.L.A.; Fortes, B.N.A.; Veado, H.C.; Macedo, I.L.; Dallago, B.S.L.; Barud, H.S.; Adorno, J.; Salvador, P.A.V.; et al. Evaluation of Bacterial Cellulose/Alginate-Based Hydrogel and Frog Skin Dressings in Equine Skin Wound Healing. Gels 2025, 11, 107. [Google Scholar] [CrossRef]
- Isopencu, G.; Deleanu, I.; Busuioc, C.; Oprea, O.; Surdu, V.-A.; Bacalum, M.; Stoica, R.; Stoica-Guzun, A. Bacterial Cellulose—Carboxymethylcellulose Composite Loaded with Turmeric Extract for Antimicrobial Wound Dressing Applications. Int. J. Mol. Sci. 2023, 24, 1719. [Google Scholar] [CrossRef] [PubMed]
- Siripongpreda, T.; Somchob, B.; Rodthongkum, N.; Hoven, V.P. Bacterial cellulose-based re-swellable hydrogel: Facile preparation and its potential application as colorimetric sensor of sweat pH and glucose. Carbohydr. Polym. 2021, 256, 117506. [Google Scholar] [CrossRef] [PubMed]
- Mirshafiei, M.; Afshar, A.K.; Yazdian, F.; Rashedi, H.; Rahdar, A.; Aboudzadeh, M.A. Bacterial cellulose: A sustainable nanostructured polymer for biosensor development. RSC Sustain. 2026, 4, 178–206. [Google Scholar] [CrossRef]
- Lormaneenopparat, P.; Yukird, J.; Rodthongkum, N.; Hoven, V.P. Bacterial cellulose composite hydrogel for pre-concentration and mass spectrometric detection of thiol-containing biomarker. Int. J. Biol. Macromol. 2023, 253, 126855. [Google Scholar] [CrossRef] [PubMed]
- Hwang, D.; Kang, D.; Sung, K.; Shin, S. A nanofibrous bacterial cellulose-carboxymethyl cellulose composite with high wet strength and active ester-mediated stable tissue adhesion in dynamic environments. Mater. Horiz. 2026. [Google Scholar] [CrossRef]
- Nongnual, T.; Butprom, N.; Boonsang, S.; Kaewpirom, S. Citric acid crosslinked carboxymethyl cellulose edible films: A case study on preserving freshness in bananas. Int. J. Biol. Macromol. 2024, 267, 131135. [Google Scholar] [CrossRef]
- Wang, L.; Zheng, H.H.; Wang, W.H.; Deng, K.W.; Tian, H.M. Physicochemical properties of bacterial cellulose from a strain of Komagataeibacter intermedius and analytical studies on its application. Int. J. Biol. Macromol. 2024, 283, 137472. [Google Scholar] [CrossRef]
- Lettieri, R.; Caravella, A.; Quintarelli, G.; D’Ottavi, C.; Licoccia, S.; Gatto, E. Lignin-Containing Cellulose Acetate Films from Grapevine Waste: A Sustainable Path to Compostable Bioplastics. ACS Sustain. Chem. Eng. 2025, 13, 16178–16191. [Google Scholar] [CrossRef]
- Frone, A.N.; Panaitescu, D.M.; Chiulan, I.; Nicolae, C.A.; Casarica, A.; Gabor, A.R.; Trusca, R.; Damian, C.M.; Purcar, V.; Alexandrescu, E.; et al. Surface Treatment of Bacterial Cellulose in Mild, Eco-Friendly Conditions. Coatings 2018, 8, 221. [Google Scholar] [CrossRef]
- Yu, C.; Sun, H.; Yao, L.; Weng, Y. NaOH/Urea-Compatible Chitosan/Carboxymethylcellulose Films: Orthogonal Optimization of Packaging Properties. Molecules 2025, 30, 2279. [Google Scholar] [CrossRef]
- Patel, A.; Parmar, M.; Patel, P.; Patel, B.; Pandya, A.; Patel, R.; Gosai, H. Tailoring bacterial cellulose composites derived from Komagateibacter saccharivorans APPK1: Functional enhancements for biomedical applications. Biotechnol. Sustain. Mater. 2026, 3, 2. [Google Scholar] [CrossRef]
- Wang, L.; Zhao, M.; Tian, H.; Zheng, X.; Gu, Y. Mechanistic study on the effect of exopolysaccharides produced by Streptomyces entericaulis XR1 on water holding capacity and texture of fermented yoghurt. Int. J. Biol. Macromol. 2026, 341, 150302. [Google Scholar] [CrossRef]
- Jantrawut, P.; Bunrueangtha, J.; Suerthong, J.; Kantrong, N. Fabrication and Characterization of Low Methoxyl Pectin/Gelatin/Carboxymethyl Cellulose Absorbent Hydrogel Film for Wound Dressing Applications. Materials 2019, 12, 1628. [Google Scholar] [CrossRef]
- Mir, I.S.; Riaz, A.; Fréchette, J.; Roy, J.S.; McElhinney, J.; Pu, S.; Balakrishnan, H.K.; Greener, J.; Dumée, L.F.; Messaddeq, Y. Bacterial cellulose-graphene oxide composite membranes with enhanced fouling resistance for bio-effluents management. npj Clean Water 2024, 7, 111. [Google Scholar] [CrossRef]
- Insua, I.; Etzold, O.; Calafel, I.; Aguirresarobe, R.; Calderón, M.; Fernández, M. Rheological Insight into the 3D Printability of Carboxymethyl Cellulose-Based Hydrogels. Gels 2025, 11, 259. [Google Scholar] [CrossRef]
- Saadi, M.; Cui, Y.; Bhakta, S.P.; Hassan, S.; Harikrishnan, V.; Siqueira, I.R.; Pasquali, M.; Bennett, M.; Ajayan, P.M.; Rahman, M.M. Flow-induced 2D nanomaterials intercalated aligned bacterial cellulose. Nat. Commun. 2025, 16, 5825. [Google Scholar] [CrossRef] [PubMed]
- Pratinthong, K.; Panyathip, R.; Thanakkasaranee, S.; Jantanasakulwong, K.; Tongdeesoontorn, W.; Noiwan, D.; Karbowiak, T.; Rachtanapun, C.; Rachtanapun, P. Sustainable Durian Rind Carboxymethyl Cellulose/Poly(vinyl) Alcohol Hydrogels Synthesis for Enhancing Crosslinking and Release Kinetics Efficiency. Gels 2025, 11, 728. [Google Scholar] [CrossRef]
- Dima, S.O.; Panaitescu, D.M.; Orban, C.; Ghiurea, M.; Doncea, S.M.; Fierascu, R.C.; Nistor, C.L.; Alexandrescu, E.; Nicolae, C.A.; Trică, B.; et al. Bacterial Nanocellulose from Side-Streams of Kombucha Beverages Production: Preparation and Physical-Chemical Properties. Polymers 2017, 9, 374. [Google Scholar] [CrossRef]
- Martins, D.; Dourado, F.; Gama, M. Effect of ionic strength, pH and temperature on the behaviour of re-dispersed BC:CMC—A comparative study with xanthan gum. Food Hydrocoll. 2023, 135, 108163. [Google Scholar] [CrossRef]
- Deleanu, I.M.; Busuioc, C.; Deleanu, M.; Stoica-Guzun, A.; Rotaru, M.; Ștefan, V.A.; Isopencu, G. Antimicrobial Carboxymethyl Cellulose-Bacterial Cellulose Composites Loaded with Green Synthesized ZnO and Ag Nanoparticles for Food Packaging. Int. J. Mol. Sci. 2024, 25, 12890. [Google Scholar] [CrossRef] [PubMed]
- Kunjalukkal Padmanabhan, S.; Lamanna, L.; Friuli, M.; Sannino, A.; Demitri, C.; Licciulli, A. Carboxymethylcellulose-Based Hydrogel Obtained from Bacterial Cellulose. Molecules 2023, 28, 829. [Google Scholar] [CrossRef]
- Luz, E.P.C.G.; Chaves, P.H.S.; Vieira, L.d.A.P.; Ribeiro, S.F.; Borges, M.d.F.; Andrade, F.K.; Muniz, C.R.; Infantes-Molina, A.; Rodríguez-Castellón, E.; Rosa, M.d.F.; et al. In vitro degradability and bioactivity of oxidized bacterial cellulose-hydroxyapatite composites. Carbohydr. Polym. 2020, 237, 116174. [Google Scholar] [CrossRef]
- Kraisuriyawong, P.; Kornsuthisopon, C.; Pavasant, P.; Rattanapisit, K.; Phoolcharoen, W.; Hoven, V.P. Functionalizable bacterial cellulose composite membrane for guided tissue regeneration. Int. J. Biol. Macromol. 2024, 268, 131655. [Google Scholar] [CrossRef]
- Du, M.; Xiao, Z.; Luo, Y. Sustainable strategies on cultivation, modification and rehydration of bacterial cellulose to overcome hornification for industrial applications: A review. Carbohydr. Polym. Technol. Appl. 2025, 9, 100736. [Google Scholar] [CrossRef]
- Du, M.; Zhu, H.; DiMauro, S.; Qin, Y.; Xiao, Z.; Luo, Y. Rehydratable dry bacterial cellulose formulation and the role of sodium alginate in mitigating hornification. Int. J. Biol. Macromol. 2025, 304, 140795. [Google Scholar] [CrossRef] [PubMed]
- Beghetto, V.; Conca, S.; Santandrea, D. Carboxymethyl Cellulose-Based Films for Sustainable Food Packaging: Modification Strategies and Structure–Property Relationships. Polymers 2026, 18, 552. [Google Scholar] [CrossRef]
- Yildirim-Yalcin, M.; Tornuk, F.; Toker, O.S. Recent advances in the improvement of carboxymethyl cellulose-based edible films. Trends Food Sci. Technol. 2022, 129, 179–193. [Google Scholar] [CrossRef]
- Hickman, T.J.; Tao, L.; Stingelin, N.; Meredith, J.C. Low-water-permeability foils based on bio-renewable cellulose derivatives. RSC Sustain. 2024, 2, 3451–3455. [Google Scholar] [CrossRef]












| Sample | CMC Concentration in HS Medium | Culture Volume | Inoculum | Fermentation Condition | Citric Acid Treatment | Final Film |
|---|---|---|---|---|---|---|
| BC | 0 g/L | 20 mL | 10% (v/v) | 30 °C, 6 d, static | untreated | Pure BC film |
| BC/CMC-1 | 0.2 g/L | 20 mL | 10% (v/v) | 30 °C, 6 d, static | 10% CA, 140 °C, 15 min | BC/CMC composite film |
| BC/CMC-2 | 0.4 g/L | 20 mL | 10% (v/v) | 30 °C, 6 d, static | 10% CA, 140 °C, 15 min | BC/CMC composite film |
| BC/CMC-3 | 0.6 g/L | 20 mL | 10% (v/v) | 30 °C, 6 d, static | 10% CA, 140 °C, 15 min | BC/CMC composite film |
| BC/CMC-4 | 0.8 g/L | 20 mL | 10% (v/v) | 30 °C, 6 d, static | 10% CA, 140 °C, 15 min | BC/CMC composite film |
| BC/CMC-5 | 1.0 g/L | 20 mL | 10% (v/v) | 30 °C, 6 d, static | 10% CA, 140 °C, 15 min | BC/CMC composite film |
| Sample | C/at. % | O/at. % | O/C | C–C/C–H/% | C–O/C–O–C/% | O–C–O/C=O/% | O–C=O/% |
|---|---|---|---|---|---|---|---|
| BC | 75.45 | 22.01 | 0.292 | 38.71 | 47.15 | 12.12 | 2 |
| BC/CMC-3 | 63.59 | 32.63 | 0.513 | 23.3 | 57.18 | 16.73 | 2.79 |
| BC/CMC-4 | 63.93 | 33.47 | 0.524 | 23.38 | 55.39 | 19.73 | 1.49 |
| Sample | 2θ (°) | Crystallinity (%) | ||
|---|---|---|---|---|
| (101) | (110) | (200) | ||
| BC | 14.00 | 16.00 | 22.20 | 87.49 |
| BC/CMC-1 | 14.72 | 16.61 | 22.85 | 81.59 |
| BC/CMC-2 | 14.53 | 16.55 | 22.80 | 80.92 |
| BC/CMC-3 | 14.62 | 16.72 | 22.76 | 77.69 |
| BC/CMC-4 | 14.34 | 16.37 | 22.43 | 82.57 |
| BC/CMC-5 | 14.58 | 16.57 | 22.66 | 80.67 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Wang, L.; Li, W.; Lu, Y.; Xu, S. Preparation and Characterization of Bacterial Cellulose/Carboxymethyl Cellulose Composite Films. Materials 2026, 19, 2038. https://doi.org/10.3390/ma19102038
Wang L, Li W, Lu Y, Xu S. Preparation and Characterization of Bacterial Cellulose/Carboxymethyl Cellulose Composite Films. Materials. 2026; 19(10):2038. https://doi.org/10.3390/ma19102038
Chicago/Turabian StyleWang, Liang, Wendi Li, Yunfa Lu, and Sai Xu. 2026. "Preparation and Characterization of Bacterial Cellulose/Carboxymethyl Cellulose Composite Films" Materials 19, no. 10: 2038. https://doi.org/10.3390/ma19102038
APA StyleWang, L., Li, W., Lu, Y., & Xu, S. (2026). Preparation and Characterization of Bacterial Cellulose/Carboxymethyl Cellulose Composite Films. Materials, 19(10), 2038. https://doi.org/10.3390/ma19102038





