Study on the Hydration Kinetics Characteristics of Low-Calcium Cementitious Materials Based on Alkali-Activated CWM
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.1.1. CWM
2.1.2. Cement
2.1.3. Alkali-Activator
2.1.4. Experimental Water
2.2. Experimental Mixing Ratio
2.3. Experimental Methods
2.3.1. Laser Particle Size Analysis
2.3.2. X-Ray Diffraction Analysis
2.3.3. Adjustment of the Modulus of Sodium Silicate Solution
- (1)
- Target modulus M1 = 1.0: Use a 10% concentration NaOH solution to directly dilute to the desired concentration. Slowly pour the prepared NaOH solution into the sodium silicate solution with a modulus of M2 = 2.3. The dripping time should be controlled at 18 min. After the dripping is completed, continue to stir for 30–60 min to ensure that Na2O is completely dispersed without local concentration differences. After the stirring is completed, determine the actual modulus by chemical titration and adjust it to the target modulus of 1.0.
- (2)
- Target modulus M3 = 3.3: Use a 25% concentration silica sol colloid solution. Before use, mix it evenly to ensure there is no precipitation. Slowly add the silica sol to the sodium silicate solution, and the dripping time is 28 min. After the dripping is completed, continue to stir for 75 min to ensure that SiO2 is completely dispersed and forms a uniform sodium silicate solution. After the stirring is completed, determine the actual modulus by chemical titration and adjust it to the target modulus.
2.3.4. Water Requirement Test of Normal Consistency for Alkali-Activated CWM-Cement
- (1)
- Preliminary water addition: Based on the fineness of the aggregate and cement, preliminarily estimate the water requirement at 400 mL. Accurately measure the water volume and pour it into the mixing bowl.
- (2)
- Mixing of the paste: Weigh 500 g of mixtures and slowly add it to the water. Immediately start the mixer and stir at a speed of 140 ± 5 r/min for 120 s. Stop stirring for 15 s, and during this time, scrape the paste adhering to the inner wall and the blades into the center of the bowl. Restart the mixer and stir at a speed of 285 ± 10 r/min for 120 s. After mixing is complete, proceed immediately with the test.
- (3)
- Testing with Vicat Apparatus: Place the mixed paste into the Vicat mold. Gently tap the mold 5–10 times with the mold rim, then use a straightedge to strike off the excess paste, making the surface flush with the mold rim. Place the mold on the base of the Vicat Apparatus. Adjust the test needle (diameter 1.13 ± 0.05 mm) so that it just contacts contact the surface of the paste. Suddenly release the test needle and allow it to freely sink into the paste. After 30 s, record the penetration depth.
- (4)
- Judgment criteria: If the penetration depth of the test needle is 6 ± 1 mm, the corresponding water content is defined as the water requirement for standard consistency. If the depth is outside this range, adjust the amount of water, prepare a fresh paste, and repeat the procedure until the criterion is met.
2.3.5. Sample Preparation, Curing and Strength Measurement
2.3.6. Activity Index Calculation of CWM
2.3.7. Hydration Heat Test
2.3.8. TGA Test
3. Results and Discussion
3.1. Activity Index
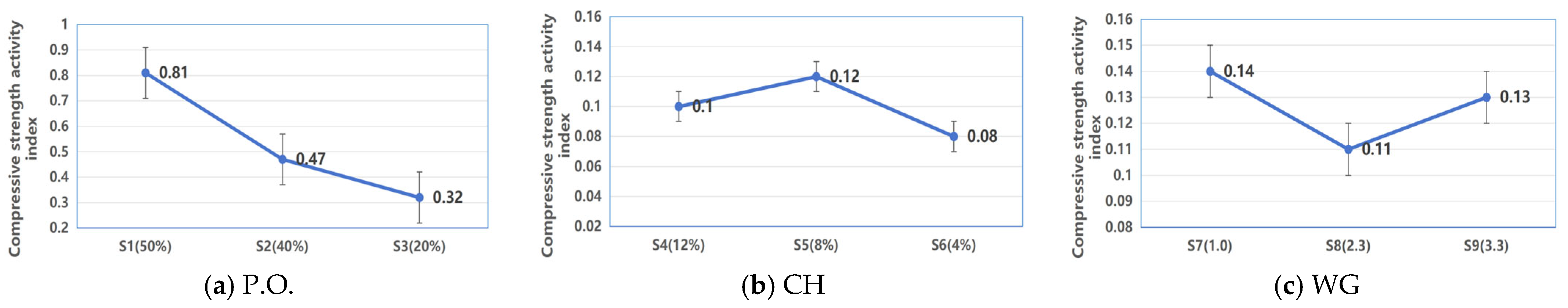
3.1.1. Single Type of Alkali Activating CWM System
- (1)
- P.O.
- (2)
- CH
- (3)
- WG
3.1.2. Double Alkali-Activated CWM System
- (1).
- Composite Activation System of P.O. and CH
- (2).
- Composite Activation System of P.O. and WG
3.1.3. Discussion on the Activation of CWM by Alkali Activators
- (1)
- Performance of single-component alkali activation systems
- (2)
- Analysis of the synergistic effect of composite alkali activation systems
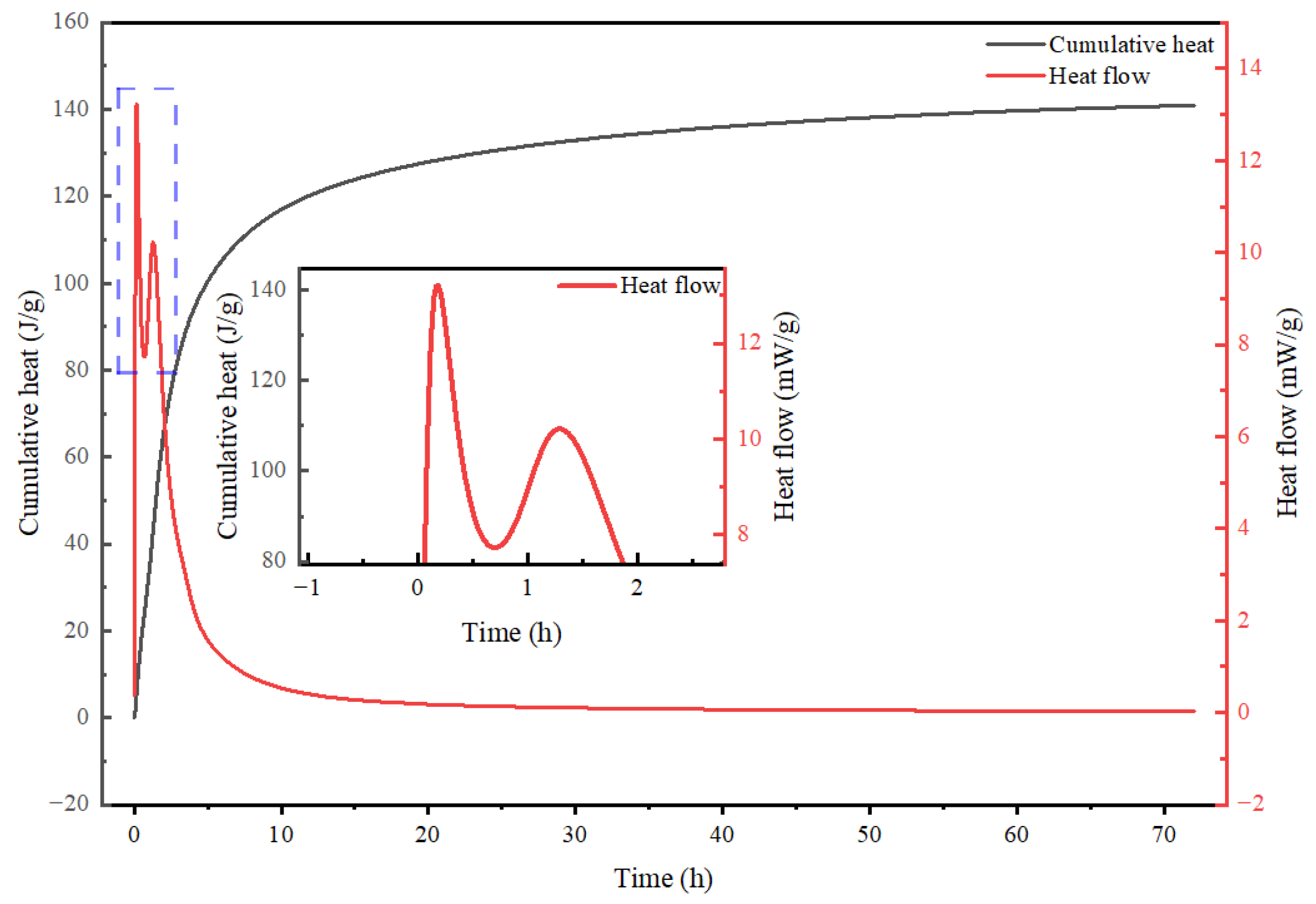
3.2. Hydration Heat Characteristics and Comparative Stage Analysis of Alkali-Activated CWM System
3.2.1. Stage I: Initial Dissolution and Early Gel Nucleation—A Retarded Onset
3.2.2. Stage II: Superimposed Dissolution-Polycondensation—A Unique Thermal Signature
3.2.3. Stage III: Main Polycondensation and Sustained Acceleration—A Prolonged Main Reaction
3.2.4. Stage IV: Deceleration Period—Diffusion Control Governed by Microstructure
3.2.5. Summary of Distinctive Hydration Features
- (1)
- A Retarded Onset (Stage I), due to chemical and physical inhibition mechanisms not found in OPC.
- (2)
- A Unique Superimposition Stage (Stage II), where competing endothermic and exothermic processes create a complex thermal signature absent in OPC’s simple induction period.
- (3)
- A Prolonged Main Acceleration Period (Stage III), driven by sustained alkaline activation, contrasting with OPC’s shorter reaction window limited by reactant depletion.
- (4)
- A Diffusion-Controlled Deceleration (Stage IV), governed by microstructural densification, which shares a common mechanistic endpoint with OPC but is reached via a distinct kinetic pathway.
3.3. Thermogravimetric Analysis of Alkali Activated CWM
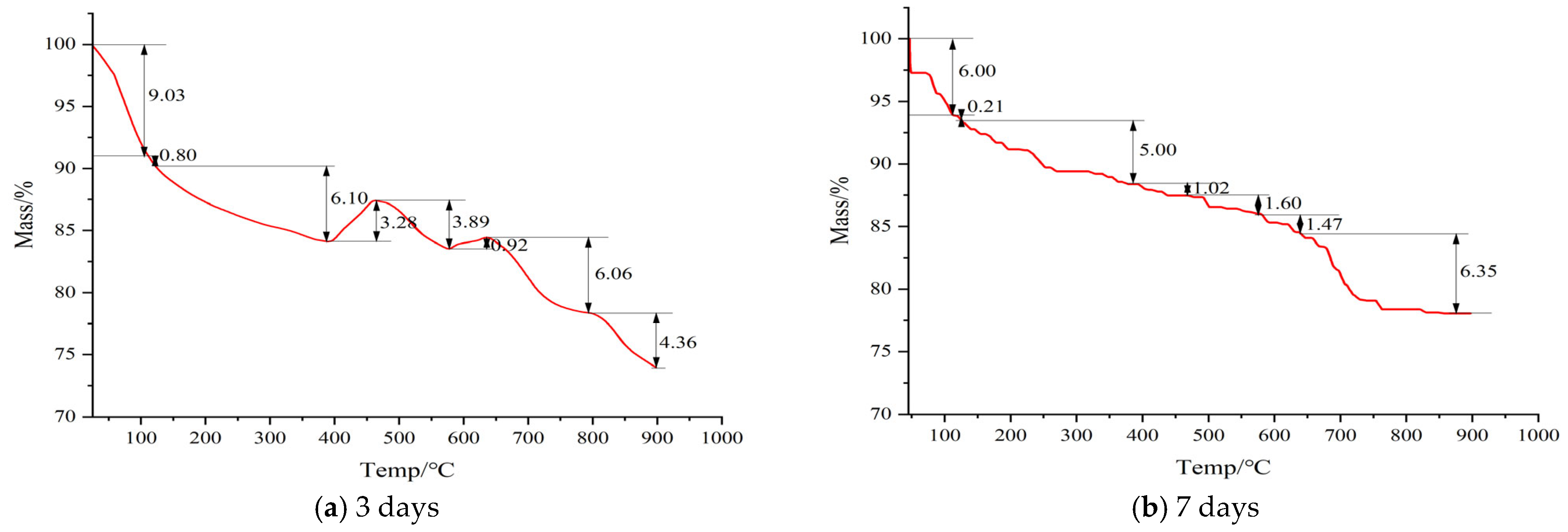
3.3.1. Thermal Decomposition Behavior of the 3-Day Cured Sample
- (1)
- Low-Temperature Mass Loss (<388 °C)
- (2)
- Anomalous Mass Increase (388~465 °C)
- (3)
- Calcium Hydroxide Decomposition (465~574 °C)
- (4)
- Minor Mass increase in the high-temperature range (574~637 °C)
- (5)
- High-temperature decomposition (>637 °C)
3.3.2. Thermal Decomposition Behavior of the 7-Day Cured Sample
- <388 °C: Mass loss of 11.21%, mainly attributed to dehydration of C-S-H gel and aluminate-containing phases (e.g., AFt, AFm) [45].
- 388~574 °C: Mass loss of 2.62%, corresponding to decomposition of residual portlandite (Ca(OH)2). The low mass loss suggests effective consumption of Ca(OH)2 through pozzolanic reactions [44].
- >574 °C: Mass loss of 7.82%, attributed to combined decomposition of C-(A)-S-H gels and carbonate species (e.g., CaCO3) [39].
3.3.3. Thermal Behavior Contrastive Analysis of the 3-Day and 7-Day Cured Sample
- Free Water (<110 °C): Mass loss decreased from −9.03% to −6.00% (+3.03%), confirming conversion of free water to chemically bound water.
- AFt (100–120 °C): Decreased from −0.80% to −0.21% (+0.59%), suggesting transformation of ettringite to more stable phases.
- C-S-H Gel (120–388 °C): Slight decrease from −6.10% to −5.00% (+1.10%), reflecting gel densification.
- Anomalous Region (388–465 °C): A mass gain of +3.28% at 3 days shifted to −1.02% mass loss at 7 days (−4.30%), indicating consumption of reactive Si/Al species and disappearance of high-temperature solid-state reactions.
- Ca(OH)2 (465–574 °C): Decreased from −3.89% to −1.60% (+2.29%), confirming ongoing pozzolanic consumption of portlandite.
- High-Temperature Region (574–637 °C): Mass gain of +0.92% at 3 days shifted to −1.47% mass loss at 7 days (−2.39%), consistent with the depletion of reactive components.
- CaCO3 (>637 °C): Decreased from −10.40% to −6.35% (+4.05%), possibly due to encapsulation by hydration products or carbonate consumption in the alkaline environment.
- (1)
- Alkaline activation: sodium silicate provides a sustained alkaline environment, which promotes the continuous dissolution of silicon and aluminum species from the CWM.
- (2)
- Ion exchange and polycondensation: the exchange among Na+, Ca2+, and other cations facilitates the formation of a dense network of cementitious gels (C-S-H, C-A-S-H, N-A-S-H), leading to the observed improvement in microstructural homogeneity and thermal stability.
4. Hydration Kinetics Characteristics of Alkali-Activated CWM
4.1. Hydrodynamic Model
4.1.1. Model Selection
4.1.2. Determination of Model Parameters
- NG stage: t0 = 0.01 h; n = 1.52144; KNG = 0.0244. NG uses the acceleration period (0.05–0.15 h), with a R-squared value of 0.99955.
- I stage usually occurs during the decline period after the peak, and I uses Time = 0.21667 to 0.5; KI = 0.09464, with a R-squared value of 0.99709.
- D stage: KD = 0.00753, using Time = 20 to 50, with a R-squared value of 0.99987.
4.2. Analysis of Influencing Factors on Hydration Dynamics of Alkali-Activated CWM
- (1)
- Initial Acceleration Period: Dominated by Nucleation and Crystal Growth (NG).
- (2)
- Mid-term Deceleration Period: Controlled by Phase Boundary Reaction (I).
- (3)
- Later Stable Period: Governed by Diffusion (D).
5. Discussion
5.1. Understanding the Hydration Kinetic Parameters of Alkali-Activated CWM
5.2. The Design of Alkaline Activation CWM
5.3. Novelty and Contribution
6. Conclusions and Outlook
6.1. Main Conclusions
- (1)
- Optimized Activator Formulation for Enhanced Reactivity. The activating efficacy on CWM follows the order: composite activator > silicate cement > Ca(OH)2 ≈ sodium silicate alone. A synergistic effect was achieved with a composite activator, leading to the identification of an optimal formulation: 40% CWM, 60% silicate cement, and 8% water glass (modulus 1.0). This optimum aligns with the fundamental principles of balancing activator alkalinity and silicate modulus in alkali-activated system design.
- (2)
- Hydration Process Characteristics Revealed by Thermal Analysis. The thermal decomposition behavior provided critical thermodynamic insights. The anomalous weight gain observed at 3 days indicates an active secondary hydration reaction, attributable to the continuous alkaline dissolution of silico-aluminous phases and subsequent gel formation. By 7 days, the primary hydration is substantially complete, coinciding with a densification of the microstructure. This sequence confirms the high activation efficiency and progressive reaction nature of the composite activator.
- (3)
- Kinetic Mechanism Elucidated by the Krstulović–Dabić (KD) Model. The hydration kinetics of the alkali-activated CWM system were successfully quantified using the KD model. The process is best described as a sequential evolution through three controlling stages: nucleation and growth (NG), phase boundary reaction (I), and diffusion (D). Specifically, the early rapid reaction (before ~0.15 h) is dominated by the NG process, which then transitions to the I process (0.21–0.5 h). The subsequent deceleration (20–50 h) is governed by a combination of I and D processes, before the reaction enters a stable period beyond 50 h where diffusion (D) becomes the sole rate-limiting step. This model quantitatively yielded the reaction rate constants (KNG, KI, KD), the Avrami exponent (n), and the stage transition points (α1, α2), providing a mechanistic explanation for the “early-strength and rapid-hardening” characteristics observed in the composite alkali-activated system at a fundamental kinetic level.
6.2. Future Research Directions
- (1)
- Diversification and Mechanistic Optimization of Activation Systems.
- (2)
- Long-Term Engineering Performance and Durability under Realistic Conditions.
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| CWM | Construction waste micro-powder |
| P.O. | Portland cement |
| CH | Ca(OH)2 |
| WG | Water glass |
References
- Solid Waste and Chemicals Management Technology Center, Ministry of Ecology and Environment. The National Solid Waste Pollution Prevention and Control Information Release Situation Research Report; Solid Waste and Chemicals Management Technology Center: Beijing, China, 2024. [Google Scholar]
- Sarsenbayev, B.; Murtazaev, S.-A.; Salamanova, M.; Kuldeyev, E.; Saidumov, M.; Sarsenbayev, N.; Auyesbek, S.; Sauganova, G.; Abduova, A. Utilization of Anthropogenic and Natural Waste to Produce Construction Raw Materials. Sustainability 2025, 17, 2791. [Google Scholar] [CrossRef]
- He, J.; Wu, J.; Ren, H.; Sang, G.; Dong, Z.; Jin, R. Physical-mechanical properties and solidification mechanism of fluid-solidified muck prepared with whole solid waste and construction muck. Constr. Build. Mater. 2025, 465, 140221. [Google Scholar] [CrossRef]
- Jhatial, A.A.; Nováková, I.; Gjerløw, E. A Review on Emerging Cementitious Materials, Reactivity Evaluation and Treatment Methods. Buildings 2023, 13, 526. [Google Scholar] [CrossRef]
- Rakhimova, N.; Shi, C. Upcycling of concrete wastes as precursors in alkali-activated materials: A review. Constr. Build. Mater. 2024, 436, 136978. [Google Scholar] [CrossRef]
- Ye, W.; Feng, T.; Sun, W.; Gómez-Zamorano, L.Y. Enhancing steel slag cement mortar performance under low-temperature curing through alkali activation: Mechanisms and implications. Front. Mater. 2025, 12, 1576078. [Google Scholar] [CrossRef]
- Rajczyk, K.; Janus, G. Microstructure and properties of geopolymers formed in the alkali activation process of fly ash. Cem. Wapno Beton 2023, 26, 279–293. [Google Scholar] [CrossRef]
- Feng, L.; Yi, S.; Zhao, S.; Zhong, Q.; Ren, F.; Liu, C.; Zhang, Y.; Wang, W.; Xie, N.; Li, Z.; et al. Recycling of Aluminosilicate-Based Solid Wastes through Alkali-Activation: Preparation, Characterization, and Challenges. Buildings 2024, 14, 226. [Google Scholar] [CrossRef]
- Sawadogo, B.; Bao, S.; Otoo, S.L.; Baffoe, E.E.; Leba-Kamanya, C. Preparation of geopolymers from alkaline thermally activated granite waste powder and burnt coal cinder blended with blast furnace slag. Constr. Build. Mater. 2024, 445, 137826. [Google Scholar] [CrossRef]
- Huang, G.; Wang, M.; Liu, Q.; Zhao, S.; Liu, H.; Liu, F.; Feng, L.; Song, J. Simultaneous utilization of mine tailings and steel slag for producing geopolymers: Alkali-hydrothermal activation, workability, strength, and hydration mechanism. Constr. Build. Mater. 2024, 414, 135029. [Google Scholar] [CrossRef]
- Borrachero, M.V.; Payá, J.; Brito, S.; Segura, Y.P.; Soriano, L.; Tashima, M.M.; Monzó, J.M. Reusing Construction and Demolition Waste to Prepare Alkali-Activated Cement. Materials 2022, 15, 3437. [Google Scholar] [CrossRef] [PubMed]
- Haha, M.B.; Le Saout, G.; Winnefeld, F.; Lothenbach, B. Influence of activator type on hydration kinetics, hydrate assemblage and microstructural development of alkali activated blast-furnace slags. Cem. Concr. Res. 2010, 41, 301–310. [Google Scholar] [CrossRef]
- Li, K.; Yang, Z.; Dong, S.; Ning, P.; Ye, D.; Zhang, Y. Hydration heat and kinetics of ternary cement containing ultrafine steel slag and blast-furnace slag at elevated temperatures. Constr. Build. Mater. 2025, 471, 140712. [Google Scholar] [CrossRef]
- Deir, E.; Gebregziabiher, B.S.; Peethamparan, S. Influence of starting material on the early age hydration kinetics, microstructure and composition of binding gel in alkali activated binder systems. Cem. Concr. Compos. 2013, 48, 108–117. [Google Scholar] [CrossRef]
- Li, Z.; Lu, T.; Liang, X.; Dong, H.; Ye, G. Mechanisms of autogenous shrinkage of alkali-activated slag and fly ash pastes. Cem. Concr. Res. 2020, 135, 106107. [Google Scholar] [CrossRef]
- Zhou, S.; Wang, J.; Li, M.; Liu, S. Mitigation of Drying Shrinkage in Cement—CWP Composite Mortar: Effects of CWP Content, W/B and Curing Conditions. Buildings 2026, 16, 418. [Google Scholar] [CrossRef]
- JGJ63-2006; Standard for Water Use in Concrete. China Architecture & Building Press: Beijing, China, 2006.
- GB/T 1346-2024; Test Methods for Standard Consistency Water Content, Setting Time and Soundness of Cement. China Standards Press: Beijing, China, 2024.
- ASTM C109/C109M; Standard Test Method for Compressive Strength of Hydraulic Cement Mortars. ASTM: Conshohocken, PA, USA, 2024.
- ASTM C186; Standard Test Methods for Heat of Hydration of Hydraulic Cement. ASTM: Conshohocken, PA, USA, 1998.
- Li, S.; Chen, D.; Jia, Z.; Li, Y.; Li, P.; Yu, B. Effects of Mud Content on the Setting Time and Mechanical Properties of Alkali-Activated Slag Mortar. Materials 2023, 16, 3355. [Google Scholar] [CrossRef]
- Tang, X.; Zhou, J.; Cao, Y.; Chen, S.; Zhang, H.; Zhang, F. The feasibility of cement-curing agent stabilized materials for industrial construction solid wastes in terms of mechanical properties and microstructure. J. Green Build. 2025, 20, 77–92. [Google Scholar]
- Tang, D.; Yang, C.; Li, X.; Zhu, X.; Yang, K.; Yu, L. Mitigation of efflorescence of alkali-activated slag mortars by incorporating calcium hydroxide. Constr. Build. Mater. 2021, 298, 123873. [Google Scholar] [CrossRef]
- Zhou, S.; Tan, C.; Gao, Y.; Li, Y.; Guo, S. One-part alkali activated slag using Ca(OH)2 and Na2CO3 instead of NaOH as activator: More excellent compressive strength and microstructure. Mater. Res. Express 2021, 8, 085501. [Google Scholar] [CrossRef]
- Xi, X.; Zheng, Y.; Zhuo, J.; Zhang, P.; Golewski, G.L.; Du, C. Influence of water glass modulus and alkali content on the properties of alkali-activated thermally activated recycled cement. Constr. Build. Mater. 2024, 452, 138867. [Google Scholar] [CrossRef]
- Gadouri, H.; Harichane, K.; Ghrici, M. Assessment of sulphates effect on pH and pozzolanic reactions of soil–lime–natural pozzolana mixtures. Int. J. Pavement Eng. 2017, 20, 761–774. [Google Scholar] [CrossRef]
- Bílek, V.; Novotný, R.; Koplík, J.; Kadlec, M.; Kalina, L. Philosophy of rational mixture proportioning of alkali-activated materials validated by the hydration kinetics of alkali-activated slag and its microstructure. Cem. Concr. Res. 2023, 168, 107139. [Google Scholar] [CrossRef]
- Bocullo, V.; Vaičiukynienė, D.; Gečys, R.; Daukšys, M. Effect of Ordinary Portland Cement and Water Glass on the Properties of Alkali Activated Fly Ash Concrete. Minerals 2020, 10, 40. [Google Scholar] [CrossRef]
- Zhang, Z.; Chen, W.; Han, F.; Yan, P. A new hydration kinetics model of composite cementitious materials, Part 2: Physical effect of SCMs. J. Am. Ceram. Soc. 2020, 103, 3880–3895. [Google Scholar] [CrossRef]
- Hinrichs, W.; Odler, I. Investigation of the hydration of Portland blastfurnace slag cement: Hydration kinetics. Adv. Cem. Res. 2012, 2, 9–13. [Google Scholar] [CrossRef]
- Chen, X.; Zhou, J.; Yan, Y. Hydration of ultrafine and ordinary Portland cement at early ages. KSCE J. Civ. Eng. 2014, 18, 1720–1725. [Google Scholar] [CrossRef]
- Toleubayeva, S. Effect of complex additive on exothermic kinetics and hydration stages of cement systems. Int. J. Geomate 2020, 19, 95831. [Google Scholar] [CrossRef]
- Park, S.; Abate, S.Y.; Lee, H.K.; Kim, H.-K. On the quantification of degrees of reaction and hydration of sodium silicate-activated slag cements. Mater. Struct. 2020, 53, 11527. [Google Scholar] [CrossRef]
- Chen, P.; Wang, X.; Zhang, T.; Guo, Y.; Li, K.; Chen, C.; Wu, Z.; Wei, J.; Yu, Q. Effect of ultrafine recycled brick powder on the properties of blended cement: Hydration kinetics, microstructure evolution and properties development. Constr. Build. Mater. 2023, 394, 132239. [Google Scholar] [CrossRef]
- Klein, A.A.; Phillips, A.J. The hydration of Portland cement. J. Frankl. Inst.-Eng. Appl. Math. 2003, 178, 635–639. [Google Scholar] [CrossRef]
- Xiong, X.; He, Y.; Lü, L.; Ma, J.; Wang, F. The Influence of Limestone Powder and Metakaolin Co-Blending on the Hydration Process and Mechanical Properties of Q-Phase Cement. J. Wuhan Univ. Technol.-Mater. Sci. Ed. 2024, 39, 1169–1176. [Google Scholar] [CrossRef]
- Kumar, S.G.K.M.; Kinuthia, J.M.; Oti, J.; Adeleke, B.O. Geopolymer Chemistry and Composition: A Comprehensive Review of Synthesis, Reaction Mechanisms, and Material Properties—Oriented with Sustainable Construction. Materials 2025, 18, 3823. [Google Scholar] [CrossRef]
- Li, X.; Hu, Q.; Robertson, B.; Ley, M.T.; Xiao, X. Direct observation of C3S particles greater than 10 μm during early hydration. Constr. Build. Mater. 2023, 369, 130548. [Google Scholar] [CrossRef]
- Ma, J.; Yu, Z.; Shi, H.; Zhang, Y.; Shen, X. Long-term hydration behavior and pore structure development of cement–limestone binary system. J. Therm. Anal. Calorim. 2020, 143, 843–852. [Google Scholar] [CrossRef]
- Ménétrier, D.; McNamara, D.K.; Jawed, I.; Skalny, J. Early hydration of β-C2S: Surface morphology. Cem. Concr. Res. 2003, 10, 107–110. [Google Scholar] [CrossRef]
- Haas, J.; Nonat, A. From C–S–H to C–A–S–H: Experimental study and thermodynamic modelling. Cem. Concr. Res. 2014, 68, 124–138. [Google Scholar] [CrossRef]
- Kaja, A.M.; Melzer, S.; Brouwers, H.J.H.; Yu, Q. On the optimization of BOF slag hydration kinetics. Cem. Concr. Compos. 2021, 124, 104262. [Google Scholar] [CrossRef]
- Chu, D.C.; Kleib, J.; Amar, M.; Benzerzour, M.; Abriak, N.-E. Determination of the degree of hydration of Portland cement using three different approaches: Scanning electron microscopy (SEM-BSE) and Thermogravimetric analysis (TGA). Case Stud. Constr. Mater. 2021, 15, e00754. [Google Scholar] [CrossRef]
- Deboucha, W.; Leklou, N.; Khelidj, A.; Oudjit, M.N. Hydration development of mineral additives blended cement using thermogravimetric analysis (TGA): Methodology of calculating the degree of hydration. Constr. Build. Mater. 2017, 146, 687–701. [Google Scholar] [CrossRef]
- Odler, I.; Abdul-Maula, S. Possibilities of quantitative determination of the AFt-(ettringite) and AFm-(monosulphate) phases in hydrated cement pastes. Cem. Concr. Res. 2003, 14, 133–141. [Google Scholar] [CrossRef]
- Zhu, X.; Richardson, I.G. Morphology-structural change of C-A-S-H gel in blended cements. Cem. Concr. Res. 2023, 168, 107156. [Google Scholar] [CrossRef]
- Stepkowska, E.T. Simultaneous IR/TG study of calcium carbonate in two aged cement pastes. J. Therm. Anal. Calorim. 2006, 84, 175–180. [Google Scholar] [CrossRef]
- Cheung, J.; Jeknavorian, A.; Roberts, L.; Silva, D. Impact of admixtures on the hydration kinetics of Portland cement. Cem. Concr. Res. 2011, 41, 1289–1309. [Google Scholar] [CrossRef]
- Johnson, W.; Mehl, R. Reaction kinetics in processes of nucleation and growth. Trans. Am. Inst. Min. Metall. Eng. 1939, 135, 416–442. [Google Scholar]
- Avrami, M. Kinetics of phase change. I General theory. J. Chem. Phys. 2005, 7, 1103–1112. [Google Scholar] [CrossRef]
- Avrami, M. Kinetics of phase change. II Transformation-time relations for random distribution of nuclei. J. Chem. Phys. 2005, 8, 212–224. [Google Scholar] [CrossRef]
- Avrami, M. Granulation, phase change, and microstructure kinetics of phase change. III. J. Chem. Phys. 2005, 9, 177–184. [Google Scholar] [CrossRef]
- Scherer, G.W.; Zhang, J.; Thomas, J.J. Nucleation and growth models for hydration of cement. Cem. Concr. Res. 2012, 42, 982–983. [Google Scholar] [CrossRef]
- Cahn, J.W. The kinetics of grain boundary nucleated reactions. Acta Metall. 1956, 4, 449–459. [Google Scholar] [CrossRef]
- Scherer, G.W. Models of confined growth. Cem. Concr. Res. 2012, 42, 1252–1260. [Google Scholar] [CrossRef]
- Krstulović, R.; Dabić, P. A conceptual model of the cement hydration process. Cem. Concr. Res. 2002, 30, 693–698. [Google Scholar] [CrossRef]
- Su, Y.; Zhao, H.; He, X.; Zheng, Z.; Ma, Q.; Ding, J.; Bao, M. The effect of wet-grinding phosphorus slag on the hydration kinetics of Portland cement. Constr. Build. Mater. 2022, 364, 129942. [Google Scholar] [CrossRef]
- He, J.; Yu, S.; Sang, G.; He, J.; Wang, J.; Chen, Z. Properties of Alkali-Activated Slag Cement Activated by Weakly Alkaline Activator. Materials 2023, 16, 3871. [Google Scholar] [CrossRef]
- Zhang, X. Thermodynamic Study on the Hydration of Cement-Based Materials. Ph.D. Thesis, Wuhan University of Technology, Wuhan, China, 2011. [Google Scholar]
- Yan, P.; Zheng, F. Kinetics model for the hydration mechanism of cementitious materials. J. Chin. Ceram. Soc. 2006, 34, 555–559. [Google Scholar]
- Han, F. Study on Hydration Characteristics and Kinetics of Composite Binder. Ph.D. Thesis, China University of Mining & Technology, Beijing, China, 2015. [Google Scholar]
- Xue, C.; Tang, L.; Zhang, Y.; Qiao, H.; Feng, Q.; Su, L. Study on physical and chemical properties of renewable brick power and hydration kinetics of cement paste. J. Compos. Mater. 2025, 11, 1–16. [Google Scholar]
- Chai, C.; Yang, Z.; Li, J.; Zhang, Z.; Wang, Z. Study on the hydration kinetics and microstructure of the sulphate-alkali composite activated slag pastes. Constr. Build. Mater. 2024, 455, 139221. [Google Scholar] [CrossRef]
- Wu, L.; Tian, Y.; Guo, Y.; He, M.; Lei, B. Hydration kinetics model and strength prediction of limestone-fly ash-cement cementitious system. Funct. Mater. 2026, 57, 10–18. [Google Scholar]
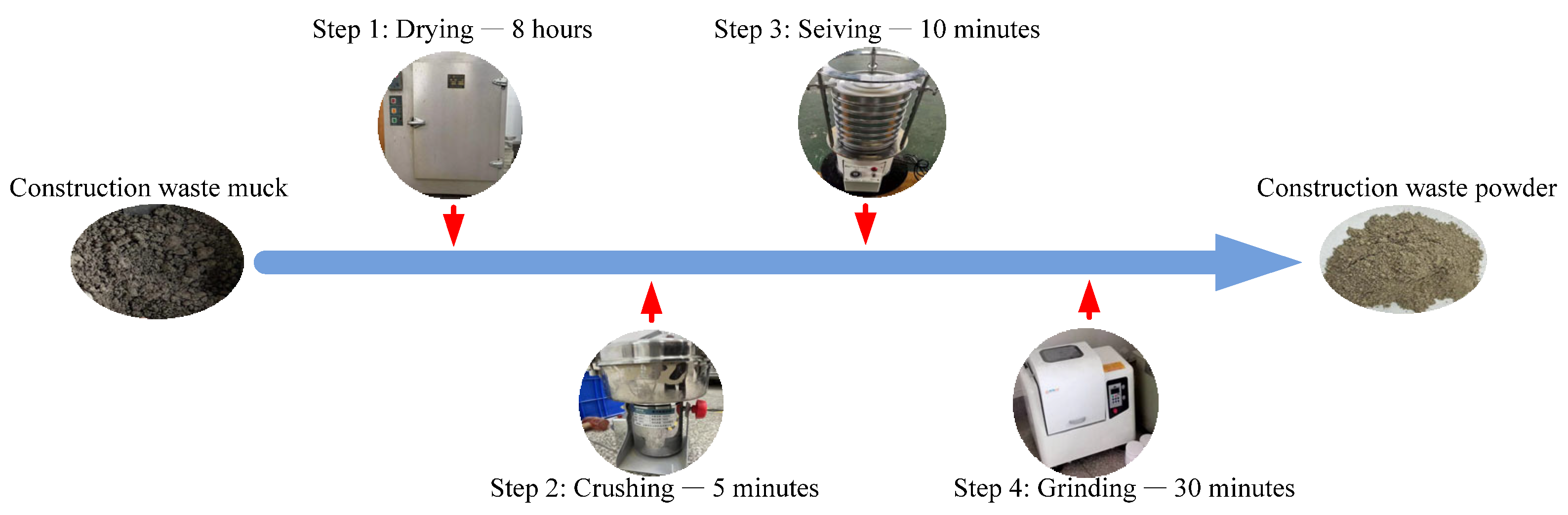




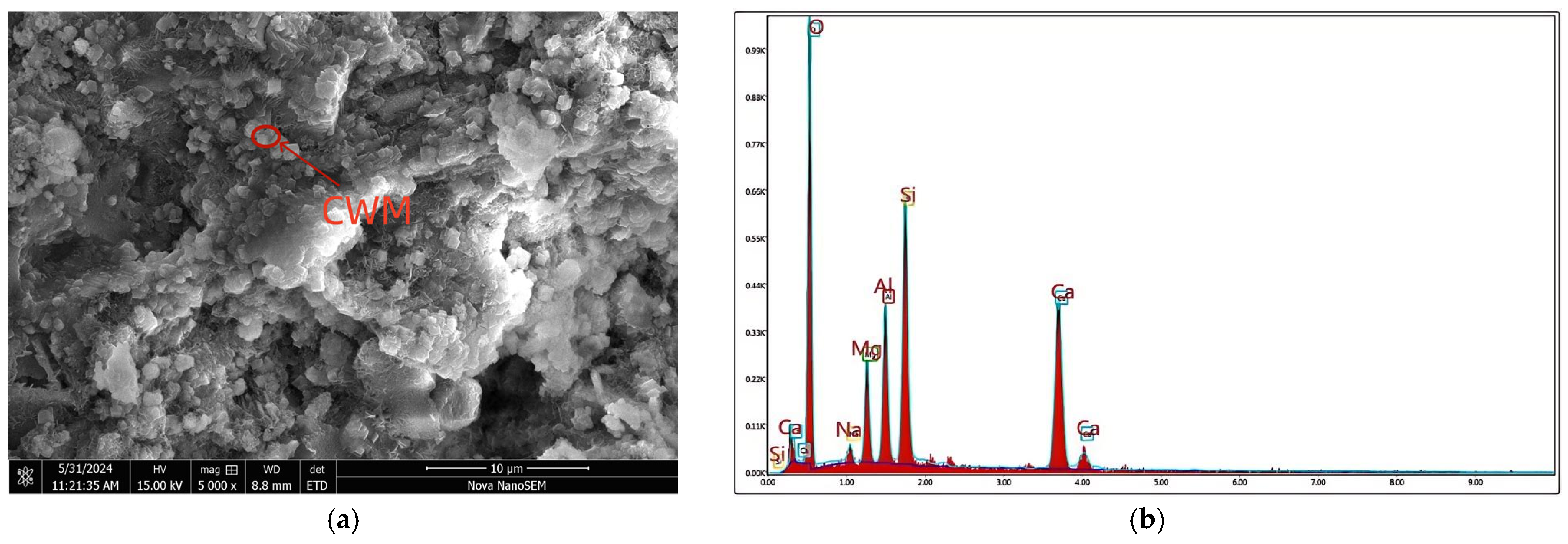
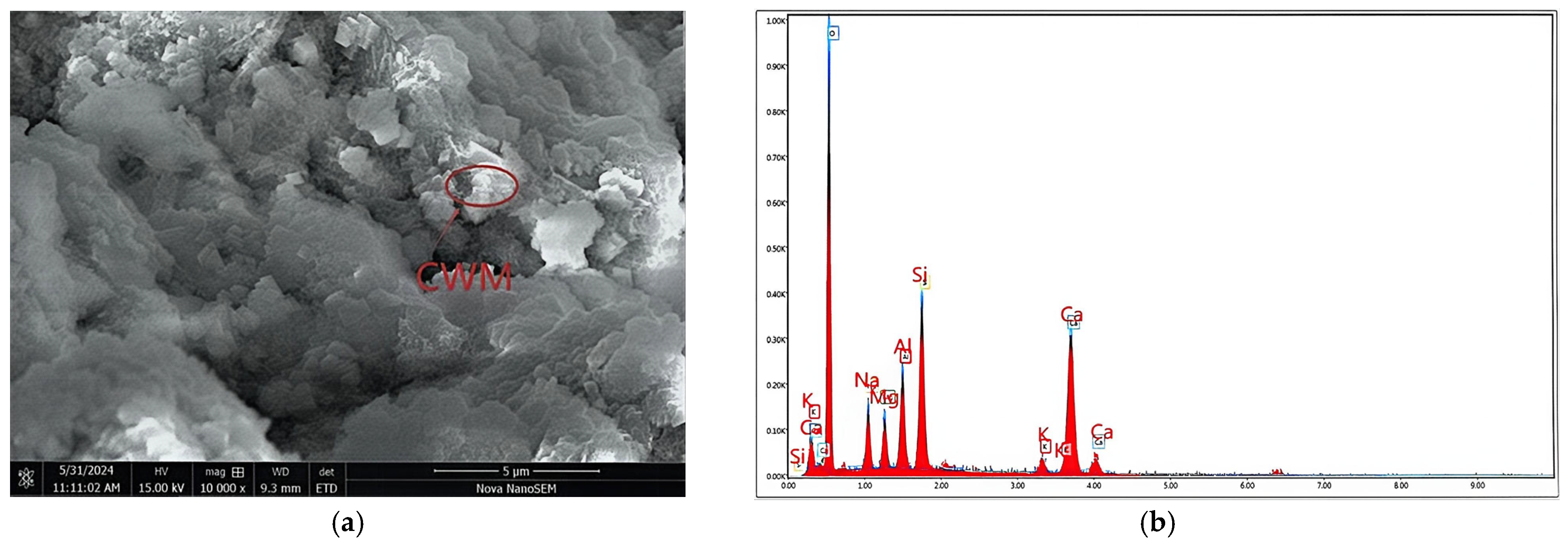
| Compositions | CaO | SiO2 | Al2O3 | Fe2O3 | MgO | SO3 | K2O + Na2O | Loss |
|---|---|---|---|---|---|---|---|---|
| CWM | 12–14 | 60–65 | 10–15 | 3–8 | 1–1.8 | 0.8–1.0 | 0.8–3.2 | 2–4 |
| Cement | 63.81 | 21.60 | 4.35 | 2.95 | 1.76 | 2.06 | 0.67 | 2.80 |
| Particle Characteristics | Specific Surface Area/m2·kg−1 | <1 μm % | 1–3 μm % | 3–10 μm % | 10–20 μm % | 20–30 μm % | 30–40 μm % | >40 μm % |
|---|---|---|---|---|---|---|---|---|
| CWM | 1595 | 5.8 | 20.99 | 38.45 | 18.94 | 8.78 | 3.91 | 3.13 |
| Compositions | SiO2 | CaCO3 | Kaolinite | Illite | Dolomite | Feldspar |
|---|---|---|---|---|---|---|
| CWM | 48–62 | 20–34 | 1–5 | 5–10 | 5–10 | 15–20 |
| Sample | Single Activator | Sample | Two-Component Alkali | |||||
|---|---|---|---|---|---|---|---|---|
| P.O. | CWM | Water | P.O. | CH | CWM | Water | ||
| S0 | 450 | 0 | 129 | S10 | 270 | 24 | 180 | 171 |
| S1 | 360 | 90 | 137 | S11 | 270 | 48 | 180 | 175 |
| S2 | 270 | 180 | 147 | S12 | 270 | 72 | 180 | 178 |
| S3 | 225 | 225 | 151 | Sample | P.O. | WG (Module) | CWM | Water |
| Sample | CH | CWM | Water | S13 | 270 | 14 (1.0) | 180 | 139 |
| S4 | 54 | 450 | 178 | S14 | 270 | 18 (1.0) | 180 | 139 |
| S5 | 36 | 450 | 175 | S15 | 270 | 27 (1.0) | 180 | 139 |
| S6 | 18 | 450 | 171 | S16 | 270 | 14 (2.3) | 180 | 160 |
| Sample | WG (Module) | CWM | Water | S17 | 270 | 18 (2.3) | 180 | 160 |
| S7 | 67.5 (1.0) | 450 | 139 | S18 | 270 | 27 (2.3) | 180 | 160 |
| S8 | 67.5 (2.3) | 450 | 160 | S19 | 270 | 14 (3.3) | 180 | 158 |
| S9 | 67.5 (3.3) | 450 | 158 | S20 | 270 | 18 (3.3) | 180 | 158 |
| S21 | 270 | 27 (3.3) | 180 | 158 | ||||
| Hydration Products | Decomposition Temperature Range (°C) | Mass Loss at 3 Days (%) | Mass Loss at 7 Days (%) | Change (7 d–3 d) |
|---|---|---|---|---|
| free water | <110 °C | −9.03 | −6.00 | +3.03 |
| AFt | 100~120 °C | −0.80 | −0.21 | +0.59 |
| C-S-H gel | 120~388 °C | −6.10 | −5.00 | +1.10 |
| Anomalous Region | 388~465 °C | +3.28 | −1.02 | −4.30 |
| Ca(OH)2 | 465~574 °C | −3.89 | −1.60 | +2.29 |
| High-Temperature Region | 574~637 °C | +0.92 | −1.47 | −2.39 |
| CaCO3 | >637 °C | −10.40 | −6.35 | +4.05 |
| Total Mass Loss | / | −26.02 | −21.65 | +4.37 |
| Model Name | Core Idea | Key Features | Main Limitations |
|---|---|---|---|
| JMAK [49,50,51,52,53] | Hydration is considered a phase transformation process where stable nuclei form randomly in the liquid phase and grow three-dimensionally. | The Avrami equation is used to describe the change in hydration degree over time, assuming random nucleation and constant growth rate. | This model lacks physical significance and cannot describe the mechanism transformation. |
| Boundary Response Model [54] | The reaction rate is controlled by the movement of the phase interface. | Applicable to the description of mid-stage hydration reactions. | An idealized dynamic description. In reality, cement hydration is a complex phenomenon involving multiple interwoven processes. |
| Diffusion-Controlled Model [55] | The growth space of the hydration products is limited by physical or chemical factors rather than unlimited, which directly leads to the transition of the hydration reaction from the acceleration phase to the deceleration phase. | Applicable to the description of the transition from the acceleration phase to the deceleration phase. | For reactions with complex mechanisms, sometimes simple limited growth models fail to capture the entire process accurately. |
| KD Model [56] | The complex cement hydration process is decomposed into a dynamic framework, which consists of three basic control stages: NG, I, and D. | Comprehensively describe the changes in the hydration process of composite cementitious materials. | Many parameters require precise experimental data. |
| n | KNG | KI | KD | α1 | α2 |
|---|---|---|---|---|---|
| 1.52 | 0.0244 | 0.0946 | 0.00753 | 0.016 | 0.433 |
| System | n | KNG | KI | KD | α1 | α2 |
|---|---|---|---|---|---|---|
| Ordinary Portland Cement [59,60] | 1.63–1.92 | 0.0537–0.0578 | 0.012–0.016 | 0.0025–0.0034 | 0.145–0.267 | 0.232–0.332 |
| Slag + Cement [59,61] | 1.42–1.88 | 0.028–0.177 | 0.005–0.018 | 0.0017–0.0080 | 0.070–0.195 | 0.180–0.311 |
| Brick Powder + Cement [62] | 1.71–1.73 | 0.0382–0.0385 | 0.084–0.0085 | 0.0014–0.0015 | 0.111–0.119 | 0.224–0.227 |
| Alkali-Activated Slag [27,63] | 1.40–1.70 | 0.017–0.035 | 0.0025–0.0060 | 0.0003–0.0014 | 0.149–0.253 | 0.218–0.346 |
| Alkali-Activated CWM | 1.52 | 0.0244 | 0.0946 | 0.00753 | 0.016 | 0.433 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zhou, S.; Li, G.; Wang, J.; Zhang, K.; Liu, S. Study on the Hydration Kinetics Characteristics of Low-Calcium Cementitious Materials Based on Alkali-Activated CWM. Materials 2026, 19, 2027. https://doi.org/10.3390/ma19102027
Zhou S, Li G, Wang J, Zhang K, Liu S. Study on the Hydration Kinetics Characteristics of Low-Calcium Cementitious Materials Based on Alkali-Activated CWM. Materials. 2026; 19(10):2027. https://doi.org/10.3390/ma19102027
Chicago/Turabian StyleZhou, Shengbo, Gengfei Li, Jian Wang, Kai Zhang, and Shengjie Liu. 2026. "Study on the Hydration Kinetics Characteristics of Low-Calcium Cementitious Materials Based on Alkali-Activated CWM" Materials 19, no. 10: 2027. https://doi.org/10.3390/ma19102027
APA StyleZhou, S., Li, G., Wang, J., Zhang, K., & Liu, S. (2026). Study on the Hydration Kinetics Characteristics of Low-Calcium Cementitious Materials Based on Alkali-Activated CWM. Materials, 19(10), 2027. https://doi.org/10.3390/ma19102027







