Electrodeposited Co and Ni Hexacyanoferrates: Insights into Structure and Morphology
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. Material Characterization
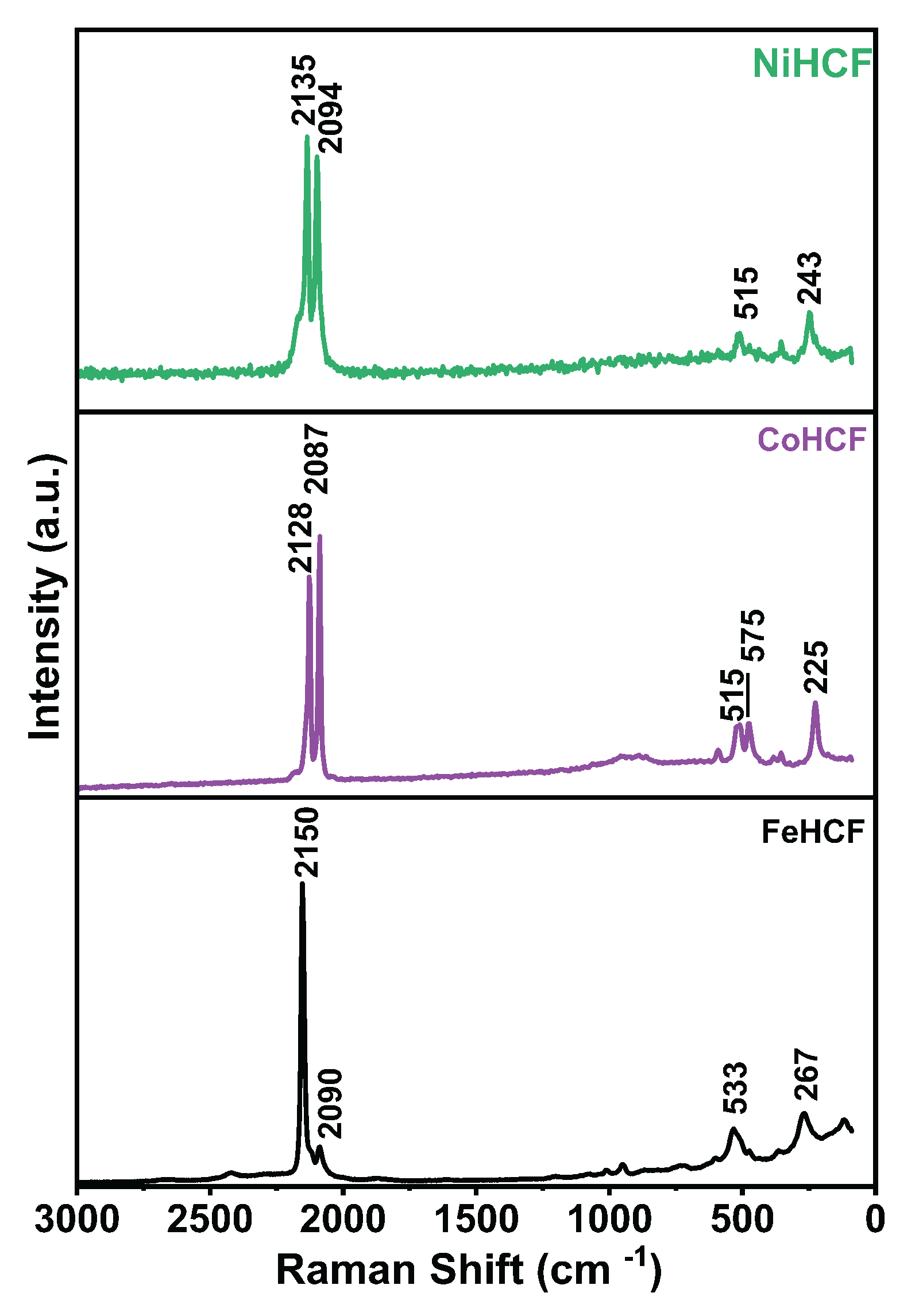
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PB | Prussian blue |
| PBA | Prussian blue analog |
| HCF | Hexacyanoferrate |
| CV | Cyclic voltammetry |
| EDX | energy-dispersive X-ray |
| XRD | X-Ray diffraction |
| FIB | Focused ion beam |
| TEM | Transmission electron microscopy |
| EELS | Electron energy loss spectroscopy |
| STEM | Scanning transmission electron microscopy |
| HRTEM | High-resolution transmission electron microscopy |
| BFTEM | Bright-field transmission electron microscopy |
| HAADF | High angle annular dark field |
| FFT | Fast Fourier transform |
| XPS | X-ray photoelectron spectroscopy |
References
- Neff, V.D. Electrochemical Oxidation and Reduction of Thin Films of Prussian Blue. J. Electrochem. Soc. 1978, 125, 886–887. [Google Scholar] [CrossRef]
- Goda, E.S.; Lee, S.; Sohail, M.; Yoon, K.R. Prussian blue and its analogues as advanced supercapacitor electrodes. J. Energy Chem. 2020, 50, 206–229. [Google Scholar] [CrossRef]
- Molina-Maldonado, P.; Ruíz-Guerrero, R.; Hernández-Fuentes, C. Role of the Outer Metal of Double Metal Cyanides on the Catalytic Efficiency in Styrene Oxidation. Catalysts 2019, 9, 905. [Google Scholar] [CrossRef]
- Ulusoy Ghobadi, T.G.; Ozbay, E.; Karadas, F. How to Build Prussian Blue Based Water Oxidation Catalytic Assemblies: Common Trends and Strategies. Chem.–A Eur. J. 2021, 27, 3638–3649. [Google Scholar] [CrossRef]
- Chu, Z.; Liu, Y.; Jin, W. Recent progress in Prussian blue films: Methods used to control regular nanostructures for electrochemical biosensing applications. Biosens. Bioelectron. 2017, 96, 17–25. [Google Scholar] [CrossRef]
- Isfahani, V.B.; Memarian, N.; Dizaji, H.R.; Arab, A.; Silva, M. The physical and electrochromic properties of Prussian Blue thin films electrodeposited on ITO electrodes. Electrochim. Acta 2019, 304, 282–291. [Google Scholar] [CrossRef]
- Kraft, A. Too blue to be good? A critical overview on the electrochromic properties and applications of Prussian blue. Sol. Energy Mater. Sol. Cells 2024, 278, 113195. [Google Scholar] [CrossRef]
- Krap, C.; Balmaseda, J.; Zamora, B.; Reguera, E. Hydrogen storage in the iron series of porous Prussian blue analogues. Int. J. Hydrogen Energy 2010, 35, 10381–10386. [Google Scholar] [CrossRef]
- Hong, P.; Xu, C.; Yan, C.; Dong, Y.; Zhao, H.; Lei, Y. Prussian Blue and Its Analogues for Commercializing Fast-Charging Sodium/Potassium-Ion Batteries. ACS Energy Lett. 2025, 10, 750–778. [Google Scholar] [CrossRef]
- Keggin, J.F.; Miles, F.D. Structures and Formulæ of the Prussian Blues and Related Compounds. Nature 1936, 137, 577–578. [Google Scholar] [CrossRef]
- Wei, Y.; Zheng, M.; Zhu, W.; Pang, H. Hollow structures Prussian blue, its analogs, and their derivatives: Synthesis and electrochemical energy-related applications. Carbon Neutraliz. 2023, 2, 271–299. [Google Scholar] [CrossRef]
- Singh, B.; Indra, A. Prussian blue- and Prussian blue analogue-derived materials: Progress and prospects for electrochemical energy conversion. Mater. Today Energy 2020, 16, 100404. [Google Scholar] [CrossRef]
- Agrisuelas, J.; García-Jareño, J.; Moreno-Guerrero, C.; Roig, A.; Vicente, F. Identification of electroactive sites in Prussian Yellow films. Electrochim. Acta 2013, 113, 825–833. [Google Scholar] [CrossRef]
- Huang, Y.; Mu, W.; Bi, X.; Hou, Z.; Lei, X.; Wang, Q.; Luo, S. Progress in defect engineering of high-performance Prussian blue analogues as cathode materials for sodium-ion batteries. J. Energy Storage 2025, 111, 115435. [Google Scholar] [CrossRef]
- Liu, Y.; Chu, W.; Xu, Y.; Yuan, Z.; Liu, J.; Zhao, H.; Liu, Q.; Zhang, W. The effect of coprecipitation and heating temperature on structural evolution and electrochemical performance of iron-based prussian blue analogs. J. Solid State Electrochem. 2024, 28, 4105–4118. [Google Scholar] [CrossRef]
- Zhang, R.; Liu, Y.; Liu, H.; Zhong, Y.; Zhang, Y.; Wu, Z.; Wang, X. Y-tube assisted coprecipitation synthesis of iron-based Prussian blue analogues cathode materials for sodium-ion batteries. RSC Adv. 2024, 14, 12096–12106. [Google Scholar] [CrossRef]
- Wang, Q.; Yuan, R.; Yang, M.; Gao, W.; Jiao, S.; Tian, D.; Jiao, H.; Yu, H.; Sun, D. Exploring coating electrodeposition protocols from a cross-electrolyte and cross-metal perspective. J. Mater. Sci. Technol. 2025, 226, 122–134. [Google Scholar] [CrossRef]
- Sreekumar, R.; Nair, A.S. Recent trends and developments in two-dimensional materials based electrodeposited nickel nanocomposite coatings. FlatChem 2022, 36, 100434. [Google Scholar] [CrossRef]
- Itaya, K.; Akahoshi, H.; Toshima, S. Electrochemistry of Prussian Blue Modified Electrodes: An Electrochemical Preparation Method. J. Electrochem. Soc. 1982, 129, 1498–1500. [Google Scholar] [CrossRef]
- Avila, L.B.; Müller, C.K.; Hildebrand, D.; Faita, F.L.; Baggio, B.F.; Cid, C.C.P.; Pasa, A.A. Resistive Switching in Electrodeposited Prussian Blue Layers. Materials 2020, 13, 5618. [Google Scholar] [CrossRef] [PubMed]
- Faita, F.; Avila, L.; Silva, J.; Boratto, M.; Cid, C.P.; Graeff, C.; Gomes, M.; Müller, C.; Pasa, A. Abnormal resistive switching in electrodeposited Prussian White thin films. J. Alloys Compd. 2022, 896, 162971. [Google Scholar] [CrossRef]
- Baggio, B.F.; Vicente, C.; Pelegrini, S.; Plá Cid, C.C.; Brandt, I.S.; Tumelero, M.A.; Pasa, A.A. Morphology and Structure of Electrodeposited Prussian Blue and Prussian White Thin Films. Materials 2019, 12, 1103. [Google Scholar] [CrossRef]
- Agnihotry, S.; Singh, P.; Joshi, A.G.; Singh, D.; Sood, K.; Shivaprasad, S. Electrodeposited Prussian blue films: Annealing effect. Electrochim. Acta 2006, 51, 4291–4301. [Google Scholar] [CrossRef]
- Husmann, S.; Orth, E.S.; Zarbin, A.J. A multi-technique approach towards the mechanistic investigation of the electrodeposition of Prussian blue over carbon nanotubes film. Electrochim. Acta 2019, 312, 380–391. [Google Scholar] [CrossRef]
- Liu, S.; Li, H.; Jiang, M.; Li, P. Platinum hexacyanoferrate: A novel Prussian Blue analogue with stable electroactive properties. J. Electroanal. Chem. 1997, 426, 27–30. [Google Scholar] [CrossRef]
- Kulesza, P.J.; Malik, M.A.; Schmidt, R.; Smolinska, A.; Miecznikowski, K.; Zamponi, S.; Czerwinski, A.; Berrettoni, M.; Marassi, R. Electrochemical preparation and characterization of electrodes modified with mixed hexacyanoferrates of nickel and palladium. J. Electroanal. Chem. 2000, 487, 57–65. [Google Scholar] [CrossRef]
- Chen, S.M. Preparation, characterization, and electrocatalytic oxidation properties of iron, cobalt, nickel, and indium hexacyanoferrate. J. Electroanal. Chem. 2002, 521, 29–52. [Google Scholar] [CrossRef]
- Wang, Y.; Rui, Y.; Li, F.; Li, M. Electrodeposition of nickel hexacyanoferrate/layered double hydroxide hybrid film on the gold electrode and its application in the electroanalysis of ascorbic acid. Electrochim. Acta 2014, 117, 398–404. [Google Scholar] [CrossRef]
- Pohlitz, M.; Müller, C.K. Study of Growth and Properties of Electrodeposited Sodium Iron Hexacyanoferrate Films. Materials 2022, 15, 7491. [Google Scholar] [CrossRef]
- Kahl, F.; Gerheim, V.; Linck, M.; Müller, H.; Schillinger, R.; Uhlemann, S. Test and characterization of a new post-column imaging energy filter. In Advances in Imaging and Electron Physics Including Proceedings CPO-10; Elsevier: Amsterdam, The Netherlands, 2019; pp. 35–70. [Google Scholar] [CrossRef]
- Kamlesh; Aggarwal, P.; Mudgal, M.; Srivastava, A.K.; Raizada, P.; Singh, P.; Paul, A.; Singh, A. Electrochemistry of Nickelocene-Ferrocene Organometallic Complexes for Electrodeposition of Nickel–Iron–Based Nanostructured Film under Ambient Conditions for Oxygen Evolution Reaction. ACS Appl. Nano Mater. 2024, 7, 24455–24468. [Google Scholar] [CrossRef]
- Diao, T.; Lin, Q.; Dawson, G. Experimental Electrochemical Potentials of Nickel Complexes. Synlett 2021, 32, 1606–1620. [Google Scholar] [CrossRef]
- Phadke, S.; Mysyk, R.; Anouti, M. Effect of cation (Li+, Na+, K+, Rb+, Cs+) in aqueous electrolyte on the electrochemical redox of Prussian blue analogue (PBA) cathodes. J. Energy Chem. 2020, 40, 31–38. [Google Scholar] [CrossRef]
- Li, Z.; Jia, C.; Wan, Z.; Cao, J.; Shi, J.; Xue, J.; Liu, X.; Wu, H.; Xiao, C.; Li, C.; et al. Boosting mechanical durability under high humidity by bioinspired multisite polymer for high-efficiency flexible perovskite solar cells. Nat. Commun. 2025, 16, 1771. [Google Scholar] [CrossRef]
- Malik, M.A.; Kulesza, P.J.; Marassi, R.; Nobili, F.; Miecznikowski, K.; Zamponi, S. Countercation intercalation and kinetics of charge transport during redox reactions of nickel hexacyanoferrate. Electrochim. Acta 2004, 49, 4253–4258. [Google Scholar] [CrossRef]
- Huff, M. Review Paper: Residual Stresses in Deposited Thin-Film Material Layers for Micro- and Nano-Systems Manufacturing. Micromachines 2022, 13, 2084. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.; Wang, Z.; Fu, Q.; Zhou, W.; Mo, Y.; Tu, J.; Wang, Z.; Gao, P.; Fan, C.; Liu, J. Dual-salt assisted synergistic synthesis of Prussian white cathode towards high-capacity and long cycle potassium ion battery. J. Energy Chem. 2023, 83, 16–23. [Google Scholar] [CrossRef]
- Zi, S.; Wang, Z.; Ke, J.; Mo, Y.; Wang, K.; Chen, S.; Tang, R.; Li, Y.; Gao, P.; Liu, J. FeHS vacancies in a Prussian white cathode leads to enhanced FeLS activity and electrode kinetics for boosted K+ storage. J. Mater. Chem. A 2024, 12, 10403–10411. [Google Scholar] [CrossRef]
- Zhou, A.; Cheng, W.; Wang, W.; Zhao, Q.; Xie, J.; Zhang, W.; Gao, H.; Xue, L.; Li, J. Hexacyanoferrate-Type Prussian Blue Analogs: Principles and Advances Toward High-Performance Sodium and Potassium Ion Batteries. Adv. Energy Mater. 2020, 11, 2000943. [Google Scholar] [CrossRef]
- Xiao, Y.; Xiao, J.; Zhao, H.; Li, J.; Zhang, G.; Zhang, D.; Guo, X.; Gao, H.; Wang, Y.; Chen, J.; et al. Prussian Blue Analogues for Sodium-Ion Battery Cathodes: A Review of Mechanistic Insights, Current Challenges, and Future Pathways. Small 2024, 20, 2401957. [Google Scholar] [CrossRef]
- Cattermull, J.; Roth, N.; Cassidy, S.J.; Pasta, M.; Goodwin, A.L. K-Ion Slides in Prussian Blue Analogues. J. Am. Chem. Soc. 2023, 145, 24249–24259. [Google Scholar] [CrossRef]
- Lee, J.H.; Bae, J.G.; Kim, M.S.; Heo, J.Y.; Lee, H.J.; Lee, J.H. Effect of the Interaction between Transition Metal Redox Center and Cyanide Ligand on Structural Evolution in Prussian White Cathodes. ACS Nano 2024, 18, 1995–2005. [Google Scholar] [CrossRef] [PubMed]
- Steen, W.A.; Jeerage, K.M.; Schwartz, D.T. Raman Spectroscopy of Redox Activity in Cathodically Electrodeposited Nickel Hexacyanoferrate Thin Films. Appl. Spectrosc. 2002, 56, 1021–1029. [Google Scholar] [CrossRef]
- Adak, S.; Daemen, L.L.; Hartl, M.; Williams, D.; Summerhill, J.; Nakotte, H. Thermal expansion in 3d-metal Prussian Blue Analogs—A survey study. J. Solid State Chem. 2011, 184, 2854–2861. [Google Scholar] [CrossRef]
- Le, L.H.T.; Nguyen, S.A.; Nguyen, T.D.; Le, V.C.T.; Van Cao, H.; Nguyen, N.B.; Le, T.P.T. Prussian Blue Analogues of A2[Fe(CN)6] (A: Cu2+, Co2+, and Ni2+) and Their Composition-Dependent Sorption Performances towards Cs+, Sr2+, and Co2+. J. Nanomater. 2021, 2021, 5533620. [Google Scholar] [CrossRef]
- Ojwang, D.O.; Svensson, M.; Njel, C.; Mogensen, R.; Menon, A.S.; Ericsson, T.; Häggström, L.; Maibach, J.; Brant, W.R. Moisture-Driven Degradation Pathways in Prussian White Cathode Material for Sodium-Ion Batteries. ACS Appl. Mater. Interfaces 2021, 13, 10054–10063. [Google Scholar] [CrossRef]
- Rahman, M.A.; Cai, Z.; Moushumy, Z.M.; Tagawa, R.; Hidaka, Y.; Nakano, C.; Islam, M.S.; Sekine, Y.; Nishina, Y.; Ida, S.; et al. Engineering Zeolitic-Imidazolate-Framework-Derived Mo-Doped Cobalt Phosphide for Efficient OER Catalysts. ACS Omega 2024, 9, 36114–36121. [Google Scholar] [CrossRef]
- Deabate, S.; Fourgeot, F.; Henn, F. X-ray diffraction and micro-Raman spectroscopy analysis of new nickel hydroxide obtained by electrodialysis. J. Power Sources 2000, 87, 125–136. [Google Scholar] [CrossRef]
- Penche, G.; González-Marcos, M.P.; González-Velasco, J.R. Transition Metal Hexacyanoferrate(II) Complexes as Catalysts in the Ring-Opening Copolymerization of CO2 and Propylene Oxide. Top. Catal. 2022, 65, 1541–1555. [Google Scholar] [CrossRef]
- Moretti, G.; Gervais, C. Raman spectroscopy of the photosensitive pigment Prussian blue. J. Raman Spectrosc. 2018, 49, 1198–1204. [Google Scholar] [CrossRef]
- Elsayed, A.A.; Othman, A.M.; Sabry, Y.M.; Marty, F.; Omran, H.; Khalil, D.; Liu, A.Q.; Bourouina, T. Substrate Signal Inhibition in Raman Analysis of Microplastic Particles. ACS Omega 2023, 8, 9854–9860. [Google Scholar] [CrossRef]
- Vančo, Ľ.; Kotlár, M.; Vretenár, V.; Kadlečíková, M.; Vojs, M.; Vogrinčič, P. Correlated reflectance and Raman spectroscopy in substrates with coherent transparent layers. Surfaces Interfaces 2022, 34, 102309. [Google Scholar] [CrossRef]
- Sada, K.; Nanda, S.; Khani, H.; Manthiram, A. Deciphering the local structure of Prussian blue analogue cathodes with Raman spectroscopy for sodium-ion batteries. J. Mater. Chem. A 2025, 13, 22903–22914. [Google Scholar] [CrossRef]
- Gerber, S.J.; Erasmus, E. Electronic effects of metal hexacyanoferrates: An XPS and FTIR study. Mater. Chem. Phys. 2018, 203, 73–81. [Google Scholar] [CrossRef]
- Zhu, Y.; Chen, P.; Zhou, Y.; Nie, W.; Xu, Y. The pursuit of optimal sodium ions prestoring in potassium–sodium–cobalt hexacyanoferrate: Toward high discharge performance supercapacitors. J. Mater. Sci. 2018, 53, 12467–12476. [Google Scholar] [CrossRef]
- Park, J.S.; Yang, S.; Kang, Y.C. Prussian blue analogue nanocubes with hollow interior and porous walls encapsulated within reduced graphene oxide nanosheets and their sodium-ion storage performances. Chem. Eng. J. 2020, 393, 124606. [Google Scholar] [CrossRef]
- Yang, J.; Hou, W.; Ye, L.; Hou, G.; Yan, C.; Zhang, Y. Vanadium Hexacyanoferrate Prussian Blue Analogs for Aqueous Proton Storage: Excellent Electrochemical Properties and Mechanism Insights. Small 2023, 20, 2305386. [Google Scholar] [CrossRef]
- Zhang, Y.; Jin, F.; Liu, H.; Jin, Z. In-situ XPS monitoring bimetallic Prussian blue analogs and metal oxides heterojunction for photocatalytic hydrogen production. Fuel 2024, 361, 130655. [Google Scholar] [CrossRef]
- Martins, J.B.; Husmann, S.; da Veiga, A.G.; Zarbin, A.J.G.; Rocco, M.L.M. Probing the Electronic Structure of Prussian Blue and Analog Films by Photoemission and Electron Energy Loss Spectroscopies. ChemPhysChem 2024, 25, e202300590. [Google Scholar] [CrossRef]
- Cheng, L.; Luo, Y.; Wang, H.; Zhou, Z.; Yang, M.; Li, C.; Zheng, Y.; Li, M.; Wang, L.; Sun, K. Optimized synthesis and electrochemical behaviors of Prussian blue analogues cathodes for potassium-ion batteries. Mater. Rep. Energy 2025, 5, 100331. [Google Scholar] [CrossRef]









| Material | C (%) | N (%) | O (%) | Fe (%) | Co (%) | Ni (%) | K (%) |
|---|---|---|---|---|---|---|---|
| FeHCF | 41.3 | 35.8 | 6.4 | 12.6 | — | — | 3.8 |
| CoHCF | 38.0 | 34.2 | 5.6 | 5.6 | 6.0 | — | 10.3 |
| NiHCF | 43.7 | 38.1 | 1.8 | 4.4 | — | 5.0 | 7.0 |
| Material | C (%) | N (%) | O (%) | Fe (%) | Co (%) | Ni (%) | K (%) |
|---|---|---|---|---|---|---|---|
| FeHCF | 42.4 | 28.8 | 8.8 | 12.3 | — | — | 14.9 |
| CoHCF | 33.6 | 18.7 | 15.4 | 7.3 | 6.8 | — | 18.3 |
| NiHCF | 39.1 | 21.7 | 21.3 | 4.2 | — | 3.3 | 10.5 |
| Peak | FeHCF | CoHCF | NiHCF |
|---|---|---|---|
| Fe2+ 2p3/2 | 710.3 | 709.3 | 708.7 |
| Fe2+ 2p1/2 | 722.9 | 722.3 | 721.8 |
| Fe3+ 2p3/2 | 712.8 | 713.2 | 711.5 |
| Fe3+ 2p1/2 | 726.0 | 726.5 | 724.7 |
| Co2+ 2p3/2 | 779.9 | ||
| Co2+ 2p1/2 | 795.0 | ||
| Ni2+ 2p3/2 | 854.6 | ||
| Ni2+ 2p1/2 | 872.0 | ||
| K+ 2p3/2 | 293.2 | 295.6 | 295.0 |
| K+ 2p1/2 | 299.1 | 297.6 | 297.3 |
| K 2s | 380.0 | 379.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia, L.d.O.; Pohlitz, M.; Kalady, M.F.; Röder, F.; Lubk, A.; Wolf, D.; Müller, C.K. Electrodeposited Co and Ni Hexacyanoferrates: Insights into Structure and Morphology. Materials 2025, 18, 5547. https://doi.org/10.3390/ma18245547
Garcia LdO, Pohlitz M, Kalady MF, Röder F, Lubk A, Wolf D, Müller CK. Electrodeposited Co and Ni Hexacyanoferrates: Insights into Structure and Morphology. Materials. 2025; 18(24):5547. https://doi.org/10.3390/ma18245547
Chicago/Turabian StyleGarcia, Larissa de O., Michael Pohlitz, Mohammed F. Kalady, Falk Röder, Axel Lubk, Daniel Wolf, and Christian K. Müller. 2025. "Electrodeposited Co and Ni Hexacyanoferrates: Insights into Structure and Morphology" Materials 18, no. 24: 5547. https://doi.org/10.3390/ma18245547
APA StyleGarcia, L. d. O., Pohlitz, M., Kalady, M. F., Röder, F., Lubk, A., Wolf, D., & Müller, C. K. (2025). Electrodeposited Co and Ni Hexacyanoferrates: Insights into Structure and Morphology. Materials, 18(24), 5547. https://doi.org/10.3390/ma18245547






