Reutilizing Flavonoids from Agricultural By-Products for In Situ Generation and Immobilization of AgNPs on Silk Towards Coloration, Antimicrobial and Anti-UV Functions
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Fabrication of AgNPs@silk
2.3. CCD of Experiment
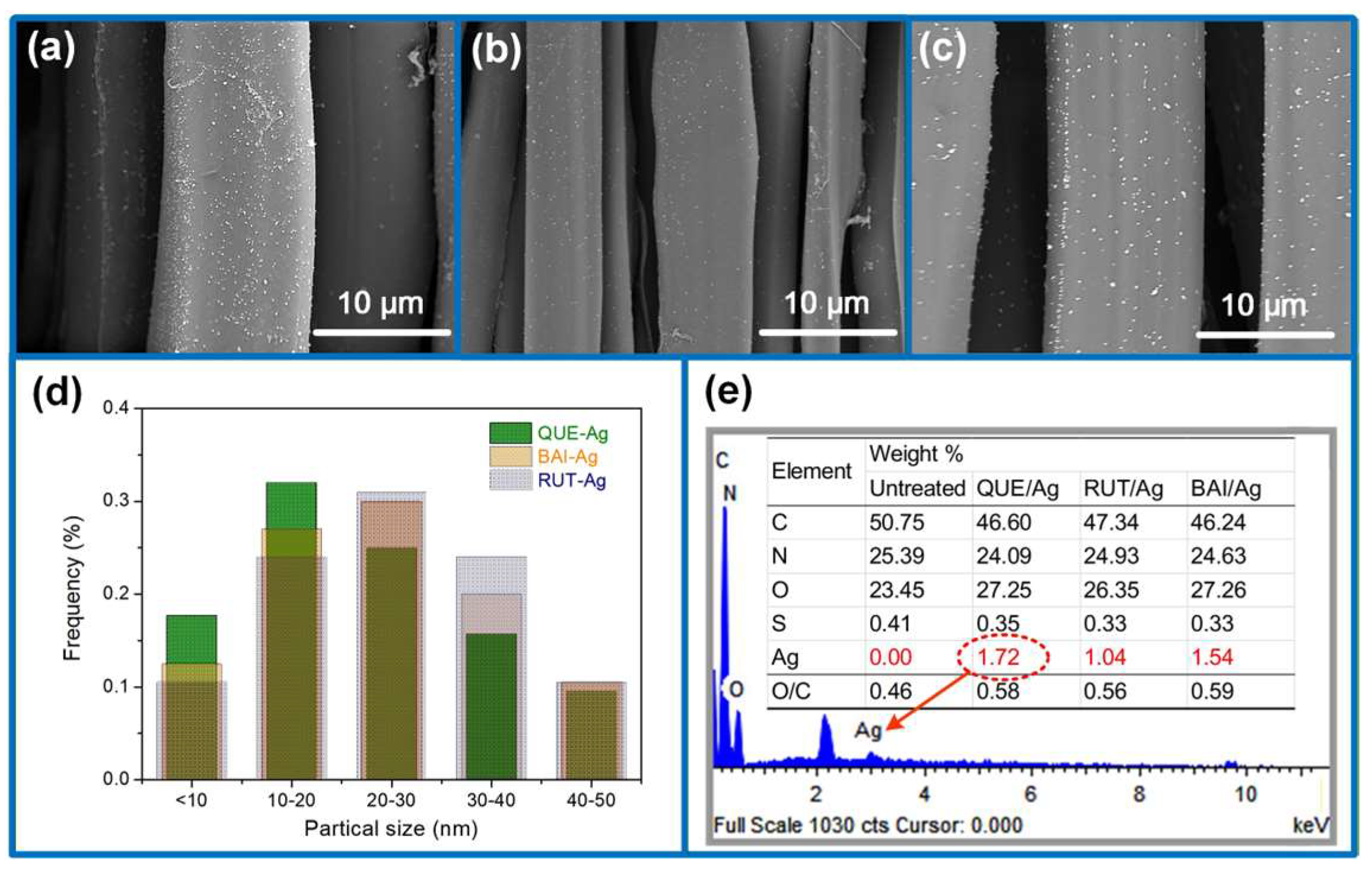
2.4. Characterization of AgNPs@silk
2.5. Functionalities
2.6. Durability
3. Results
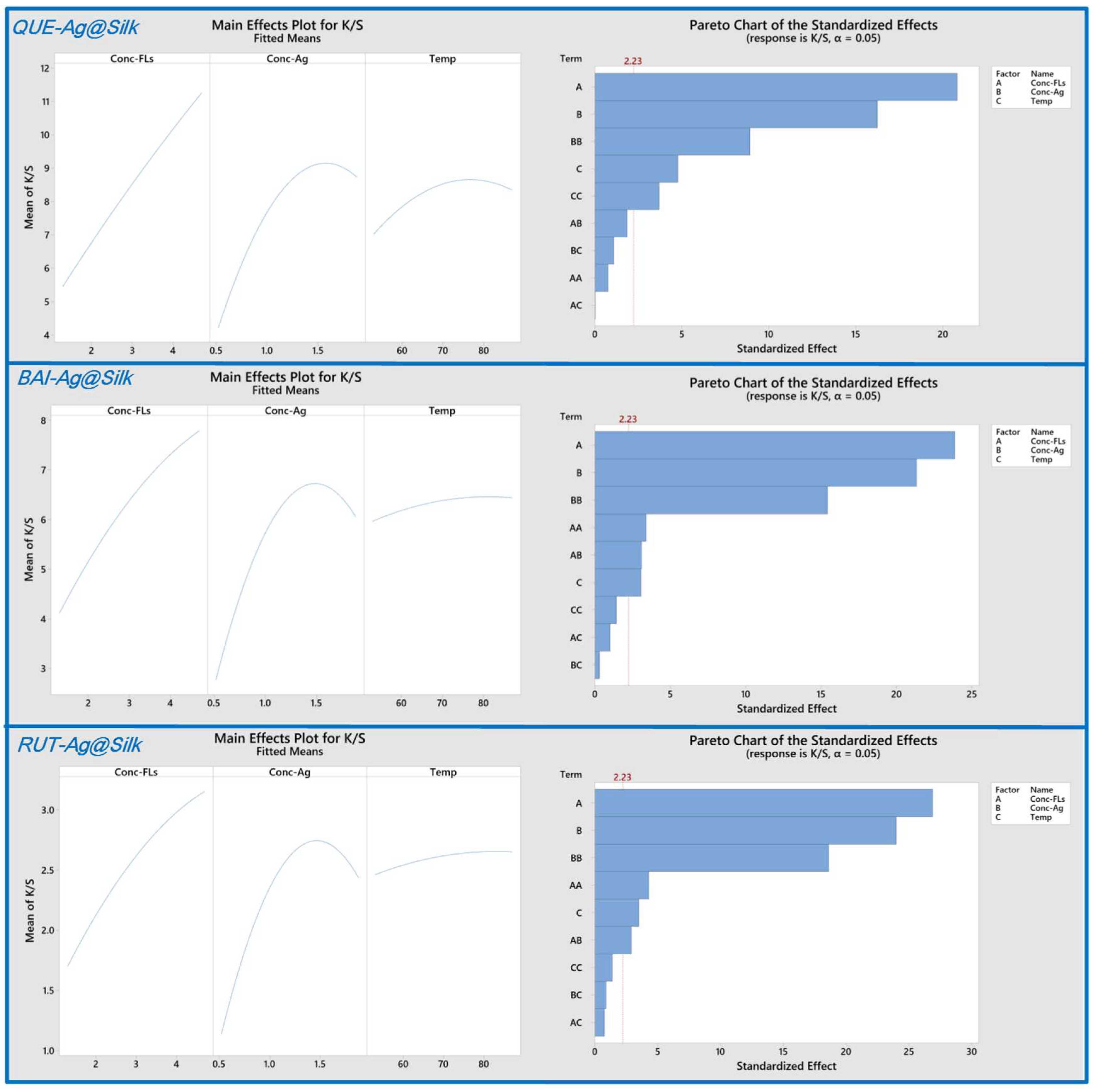
3.1. CCD Experiment
3.2. FTIR and XRD
3.3. Morphological and Tensile Properties
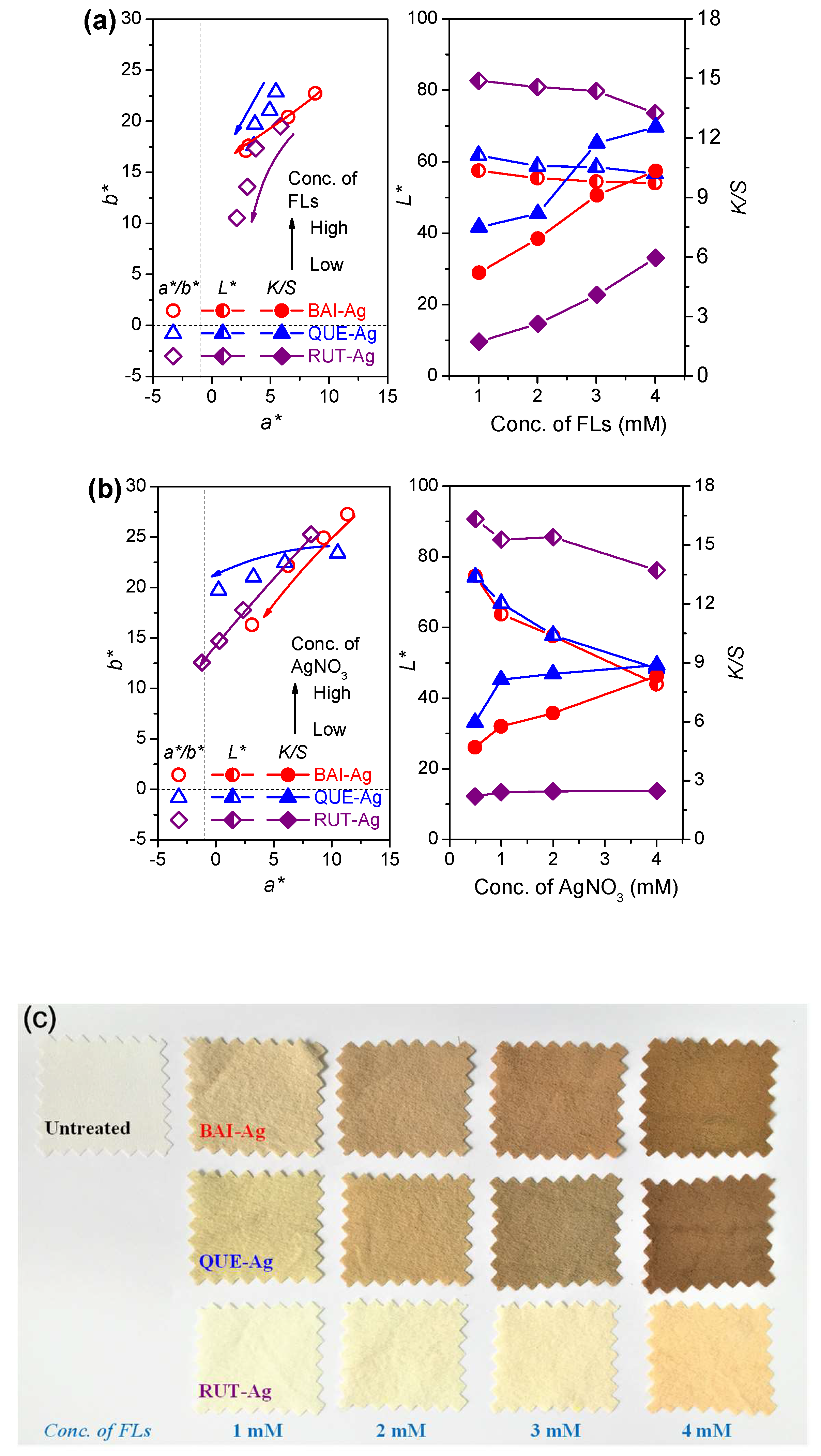
3.4. Color Features
3.5. Functionality
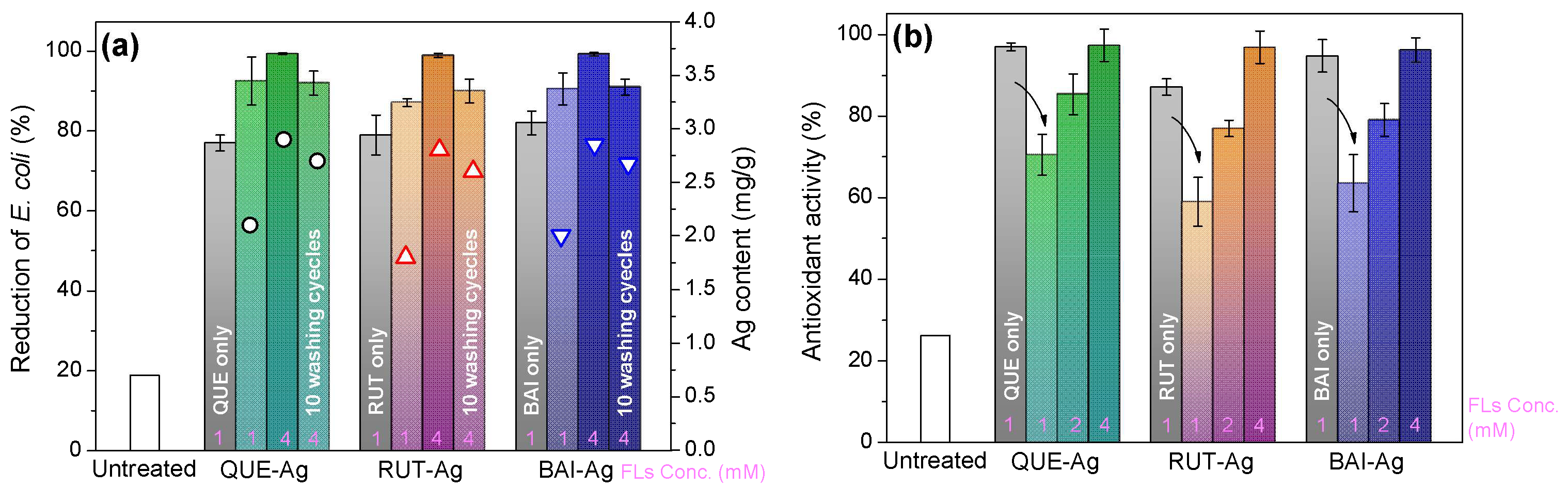
3.5.1. Bioactivity
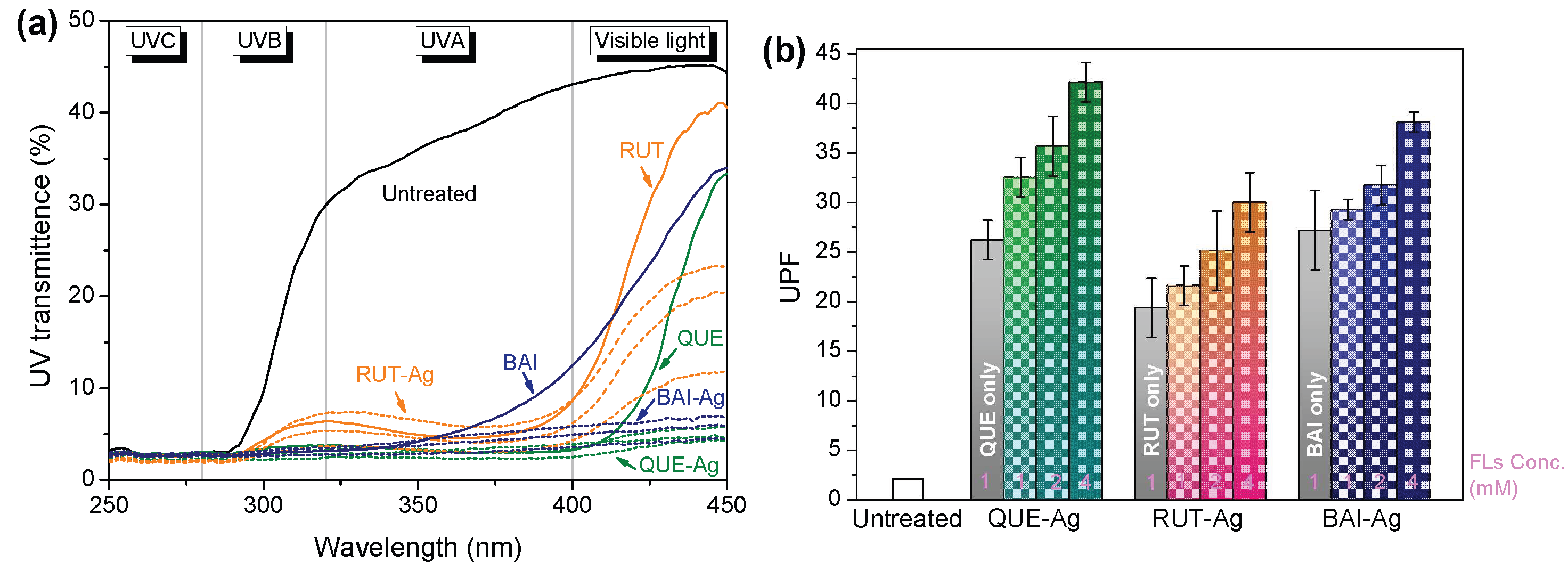
3.5.2. UV Protection
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Shahid, M.; Mohammad, F. Recent advancements in natural dye applications: A review. J. Clean. Prod. 2013, 53, 310–331. [Google Scholar] [CrossRef]
- Haji, A.; Naebe, M. Cleaner dyeing of textiles using plasma treatment and natural dyes: A review. J. Clean. Prod. 2020, 265, 121866. [Google Scholar] [CrossRef]
- Zannat, A.; Uddin, M.N.; Mahmud, S.T.; Mia, R.; Ahmed, T. Natural Dyes and Pigments in functional finishing. In Renewable dyes and pigments; Elsevier: Amsterdam, The Netherlands, 2024; pp. 271–287. [Google Scholar]
- Altıok, E.; Bayçın, D.; Bayraktar, O.; Ülkü, S. Isolation of polyphenols from the extracts of olive leaves (Olea europaea L.) by adsorption on silk fibroin. Sep. Purif. Technol. 2008, 62, 342–348. [Google Scholar] [CrossRef]
- Zhao, Q.; Chen, X.-Y.; Martin, C. Scutellaria baicalensis, the golden herb from the garden of Chinese medicinal plants. Sci. Bull. 2016, 61, 1391–1398. [Google Scholar] [CrossRef] [PubMed]
- Vollmannová, A.; Bojňanská, T.; Musilová, J.; Lidiková, J.; Cifrová, M. Quercetin as one of the most abundant represented biological valuable plant components with remarkable chemoprotective effects—A review. Heliyon 2024, 10, e33342. [Google Scholar] [CrossRef] [PubMed]
- Alam, F.; Mohammadin, K.; Shafique, Z.; Amjad, S.T.; Asad, M.H.H.b. Citrus flavonoids as potential therapeutic agents: A review. Phytother. Res. 2022, 36, 1417–1441. [Google Scholar] [CrossRef] [PubMed]
- Maran, F.N.; Türk, İ.; Akkoyun, B.; Meligy, M.T.F.; Lubiçeva, E.; İkballı, D.; Halvacı, E.; Şen, F. Nanotechnology and its Versatile Applications in Medicine, Environment, Energy, Textiles, Food Industry. Int. J. Boron Sci. Nanotechnol. 2023, 1, 17–48. [Google Scholar]
- Haider, A.; Kang, I.-K. Preparation of silver nanoparticles and their industrial and biomedical applications: A comprehensive review. Adv. Mater. Sci. Eng. 2015, 2015, 165257. [Google Scholar] [CrossRef]
- Raj, A.; Thomas, R.K. Localized surface plasmon resonance (LSPR) applications of gold (Au) and silver (Ag) nanoparticles. In Optical and Molecular Physics; Apple Academic Press: Burlington, ON, Canada, 2021; pp. 43–69. [Google Scholar]
- Mahmud, S.; Pervez, M.N.; Hasan, K.F.; Taher, M.A.; Liu, H.-H. In situ synthesis of green AgNPs on ramie fabric with functional and catalytic properties. Emerg. Mater. Res. 2019, 8, 623–633. [Google Scholar] [CrossRef]
- Montes-Hernandez, G.; Di Girolamo, M.; Sarret, G.; Bureau, S.; Fernandez-Martinez, A.; Lelong, C.; Eymard Vernain, E. In situ formation of silver nanoparticles (Ag-NPs) onto textile fibers. ACS Omega 2021, 6, 1316–1327. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, T.; Sezgin Bozok, S.; Ogulata, R.T. Comparison of in situ and padding method to incorporate Green synthesized AgNPs by using Calendula arvensis into nonwoven fabrics. J. Adhes. Sci. Technol. 2024, 38, 2047–2064. [Google Scholar] [CrossRef]
- Li, Y.; Xie, X.; Wang, C.; Pei, X.; Fang, C.; Yan, R.; Zhong, Y. Antibacterial and UV resistant functions of cotton fabrics: In-situ free radical polymerization of AgNPs using Baicalin. Ind. Crops Prod. 2024, 222, 119818. [Google Scholar] [CrossRef]
- Zhou, Y.; Yang, Z.-Y.; Tang, R.-C. Green and facile fabrication of AgNPs@silk for colorful and multifunctional textiles using baicalin as a natural reductant. J. Clean. Prod. 2018, 170, 940–949. [Google Scholar] [CrossRef]
- Shahid, M.; Zhou, Y.; Cheng, X.-W.; Zar, M.S.; Chen, G.; Tang, R.-C. Ferulic acid promoted in-situ generation of AgNPs@silk as functional colorants. J. Clean. Prod. 2018, 176, 736–744. [Google Scholar] [CrossRef]
- Zhou, Y.; Tang, R.C. Facile and eco-friendly fabrication of AgNPs coated silk for antibacterial and antioxidant textiles using honeysuckle extract. J. Photochem. Photobiol. B Biol. 2018, 178, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Noga, M.; Milan, J.; Frydrych, A.; Jurowski, K. Toxicological Aspects, Safety Assessment, and Green Toxicology of Silver Nanoparticles (AgNPs)-Critical Review: State of the Art. Int. J. Mol. Sci. 2023, 24, 5133. [Google Scholar] [CrossRef] [PubMed]




| Variables | Levels | ||||
|---|---|---|---|---|---|
| −α | −1 | 0 | 1 | α | |
| A: Conc-FLs (% owf) | 1.32 | 2 | 3 | 4 | 4.68 |
| B: Conc-Ag (mmol/L) | 0.53 | 0.8 | 1.2 | 1.6 | 1.87 |
| C: Temp. (°C) | 53.18 | 60 | 70 | 80 | 86.82 |
| Runs | Factors | K/S | ||||
|---|---|---|---|---|---|---|
| Conc-FLs | Conc-Ag | Temp. | QUE | BAI | RUT | |
| 1 | 2 | 0.8 | 60 | 4.16 | 3.36 | 1.40 |
| 2 | 4 | 0.8 | 60 | 6.87 | 5.29 | 2.15 |
| 3 | 2 | 1.6 | 60 | 6.42 | 5.01 | 2.08 |
| 4 | 4 | 1.6 | 60 | 10.24 | 7.49 | 3.02 |
| 5 | 2 | 0.8 | 80 | 5.26 | 3.88 | 1.62 |
| 6 | 4 | 0.8 | 80 | 8.28 | 5.39 | 2.26 |
| 7 | 2 | 1.6 | 80 | 7.37 | 5.28 | 2.18 |
| 8 | 4 | 1.6 | 80 | 10.87 | 7.70 | 3.11 |
| 9 | 1.32 | 1.2 | 70 | 5.36 | 4.15 | 1.69 |
| 10 | 4.68 | 1.2 | 70 | 11.43 | 7.96 | 3.22 |
| 11 | 3 | 0.53 | 70 | 4.23 | 2.88 | 1.16 |
| 12 | 3 | 1.87 | 70 | 8.88 | 6.23 | 2.50 |
| 13 | 3 | 1.2 | 53.18 | 7.37 | 6.06 | 2.51 |
| 14 | 3 | 1.2 | 86.82 | 8.10 | 6.53 | 2.65 |
| 15 | 3 | 1.2 | 70 | 8.92 | 6.34 | 2.62 |
| 16 | 3 | 1.2 | 70 | 8.74 | 6.61 | 2.71 |
| 17 | 3 | 1.2 | 70 | 8.18 | 6.15 | 2.52 |
| 18 | 3 | 1.2 | 70 | 8.20 | 6.35 | 2.61 |
| 19 | 3 | 1.2 | 70 | 8.50 | 6.53 | 2.63 |
| 20 | 3 | 1.2 | 70 | 8.60 | 6.29 | 2.61 |
| Source | DF | Adj SS | Adj MS | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 9 | 74.3126 | 8.2570 | 90.31 | 0.000 |
| Linear | 3 | 65.8738 | 21.9579 | 240.15 | 0.000 |
| Conc-FLs | 1 | 39.6783 | 39.6783 | 433.96 | 0.000 |
| Conc-Ag | 1 | 24.1165 | 24.1165 | 263.76 | 0.000 |
| Temp. | 1 | 2.0790 | 2.0790 | 22.74 | 0.001 |
| Square | 3 | 8.0169 | 2.6723 | 29.23 | 0.000 |
| Conc-FLs×Conc-FLs | 1 | 0.0518 | 0.0518 | 0.57 | 0.469 |
| Conc-Ag×Conc-Ag | 1 | 7.2757 | 7.2757 | 79.57 | 0.000 |
| Temp×Temp | 1 | 1.2465 | 1.2465 | 13.63 | 0.004 |
| 2-Way Interaction | 3 | 0.4219 | 0.1406 | 1.54 | 0.265 |
| Conc-FLs×Conc-Ag | 1 | 0.3139 | 0.3139 | 3.43 | 0.094 |
| Conc-FLs×Temp | 1 | 0.0000 | 0.0000 | 0.00 | 0.986 |
| Conc-Ag×Temp | 1 | 0.1079 | 0.1079 | 1.18 | 0.303 |
| Error | 10 | 0.9143 | 0.0914 | ||
| Lack-of-Fit | 5 | 0.4882 | 0.0976 | 1.15 | 0.442 |
| Pure Error | 5 | 0.4261 | 0.0852 | ||
| Total | 19 | 75.2270 | |||
| Model Summary | |||||
| S | R-sq | R-sq (adj) | R-sq (pred) | ||
| 0.302380 | 98.78% | 97.69% | 94.15% | ||
| Source | DF | Adj SS | Adj MS | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 9 | 35.9738 | 3.9971 | 142.99 | 0.000 |
| Linear | 3 | 28.9191 | 9.6397 | 344.85 | 0.000 |
| Conc-FLs | 1 | 15.9285 | 15.9285 | 569.83 | 0.000 |
| Conc-Ag | 1 | 12.7290 | 12.7290 | 455.37 | 0.000 |
| Temp. | 1 | 0.2616 | 0.2616 | 9.36 | 0.012 |
| Square | 3 | 6.7554 | 2.2518 | 80.56 | 0.000 |
| Conc-FLs×Conc-FLs | 1 | 0.3235 | 0.3235 | 11.57 | 0.007 |
| Conc-Ag×Conc-Ag | 1 | 6.6546 | 6.6546 | 238.06 | 0.000 |
| Temp×Temp | 1 | 0.0568 | 0.0568 | 2.03 | 0.185 |
| 2-Way Interaction | 3 | 0.2993 | 0.0998 | 3.57 | 0.055 |
| Conc-FLs×Conc-Ag | 1 | 0.2681 | 0.2681 | 9.59 | 0.011 |
| Conc-FLs×Temp | 1 | 0.0286 | 0.0286 | 1.02 | 0.335 |
| Conc-Ag×Temp | 1 | 0.0025 | 0.0025 | 0.09 | 0.770 |
| Error | 10 | 0.2795 | 0.0280 | ||
| Lack-of-Fit | 5 | 0.1390 | 0.0278 | 0.99 | 0.504 |
| Pure Error | 5 | 0.1405 | 0.0281 | ||
| Total | 19 | 36.2533 | |||
| Model Summary | |||||
| S | R-sq | R-sq(adj) | R-sq(pred) | ||
| 0.167192 | 99.23% | 98.54% | 96.47% | ||
| Source | DF | Adj SS | Adj MS | F-Value | p-Value |
|---|---|---|---|---|---|
| Model | 9 | 5.78333 | 0.64259 | 185.94 | 0.000 |
| Linear | 3 | 4.53025 | 1.51008 | 436.96 | 0.000 |
| Conc-FLs | 1 | 2.49858 | 2.49858 | 723.00 | 0.000 |
| Conc-Ag | 1 | 1.98923 | 1.98923 | 575.61 | 0.000 |
| Temp. | 1 | 0.04244 | 0.04244 | 12.28 | 0.006 |
| Square | 3 | 1.21923 | 0.40641 | 117.60 | 0.000 |
| Conc-FLs×Conc-FLs | 1 | 0.06344 | 0.06344 | 18.36 | 0.002 |
| Conc-Ag×Conc-Ag | 1 | 1.19691 | 1.19691 | 346.34 | 0.000 |
| Temp×Temp | 1 | 0.00671 | 0.00671 | 1.94 | 0.194 |
| 2-Way Interaction | 3 | 0.03385 | 0.01128 | 3.27 | 0.068 |
| Conc-FLs×Conc-Ag | 1 | 0.02913 | 0.02913 | 8.43 | 0.016 |
| Conc-FLs×Temp | 1 | 0.00200 | 0.00200 | 0.58 | 0.464 |
| Conc-Ag×Temp | 1 | 0.00272 | 0.00272 | 0.79 | 0.396 |
| Error | 10 | 0.03456 | 0.00346 | ||
| Lack-of-Fit | 5 | 0.01795 | 0.00359 | 1.08 | 0.467 |
| Pure Error | 5 | 0.01661 | 0.00332 | ||
| Total | 19 | 5.81789 | |||
| Model Summary | |||||
| S | R-sq | R-sq(adj) | R-sq(pred) | ||
| 0.0587865 | 99.41% | 98.87% | 97.22% | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, W.; Yue, Y.; Zhou, X.; Sun, J.; Chen, L.; Hu, X.; Zhou, Y. Reutilizing Flavonoids from Agricultural By-Products for In Situ Generation and Immobilization of AgNPs on Silk Towards Coloration, Antimicrobial and Anti-UV Functions. Materials 2025, 18, 5409. https://doi.org/10.3390/ma18235409
Chen W, Yue Y, Zhou X, Sun J, Chen L, Hu X, Zhou Y. Reutilizing Flavonoids from Agricultural By-Products for In Situ Generation and Immobilization of AgNPs on Silk Towards Coloration, Antimicrobial and Anti-UV Functions. Materials. 2025; 18(23):5409. https://doi.org/10.3390/ma18235409
Chicago/Turabian StyleChen, Wei, Yijie Yue, Xiaoqi Zhou, Jingyu Sun, Leyang Chen, Xiaoyan Hu, and Yuyang Zhou. 2025. "Reutilizing Flavonoids from Agricultural By-Products for In Situ Generation and Immobilization of AgNPs on Silk Towards Coloration, Antimicrobial and Anti-UV Functions" Materials 18, no. 23: 5409. https://doi.org/10.3390/ma18235409
APA StyleChen, W., Yue, Y., Zhou, X., Sun, J., Chen, L., Hu, X., & Zhou, Y. (2025). Reutilizing Flavonoids from Agricultural By-Products for In Situ Generation and Immobilization of AgNPs on Silk Towards Coloration, Antimicrobial and Anti-UV Functions. Materials, 18(23), 5409. https://doi.org/10.3390/ma18235409







