Magnesium Silicate Coatings Were Prepared by Micro-Arc Oxidation on the Surface of Magnesium Alloys Through the Synergistic Effect of SiO32−/F−
Abstract
1. Introduction
2. Experimental Section
2.1. Materials and Reagents
2.2. Experimental Techniques
3. Results and Discussion
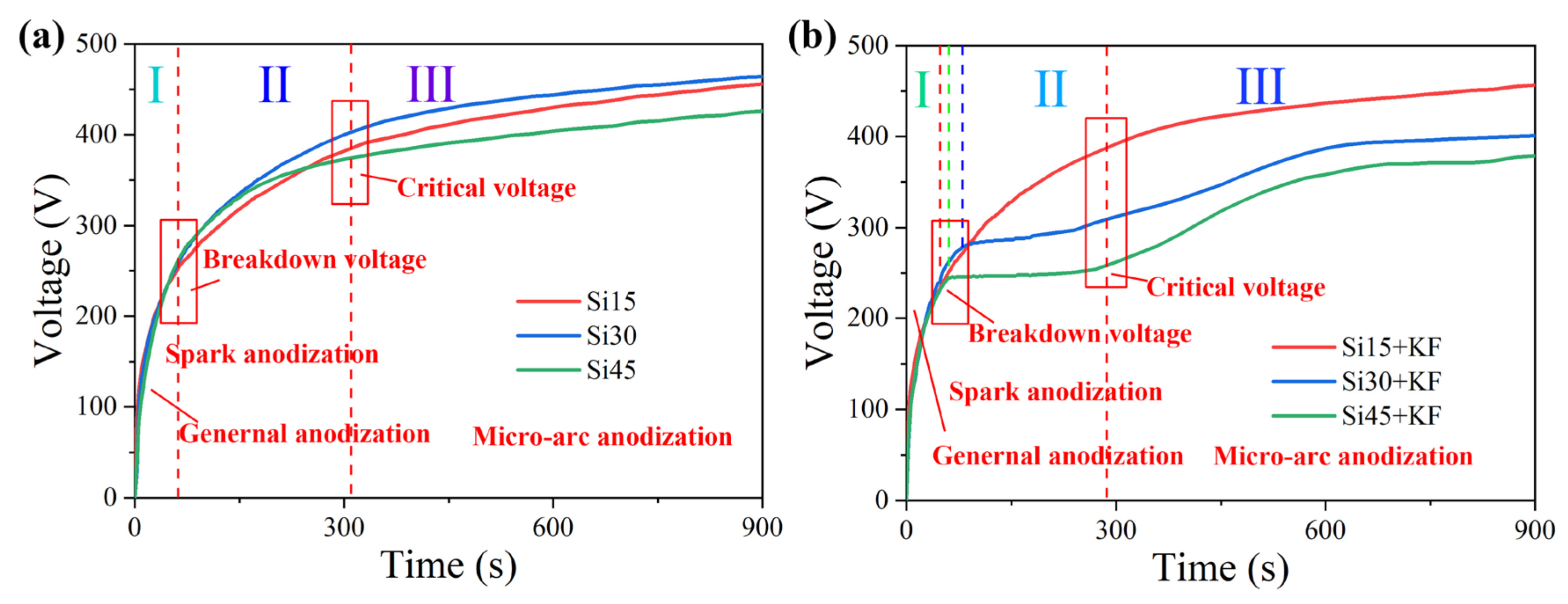
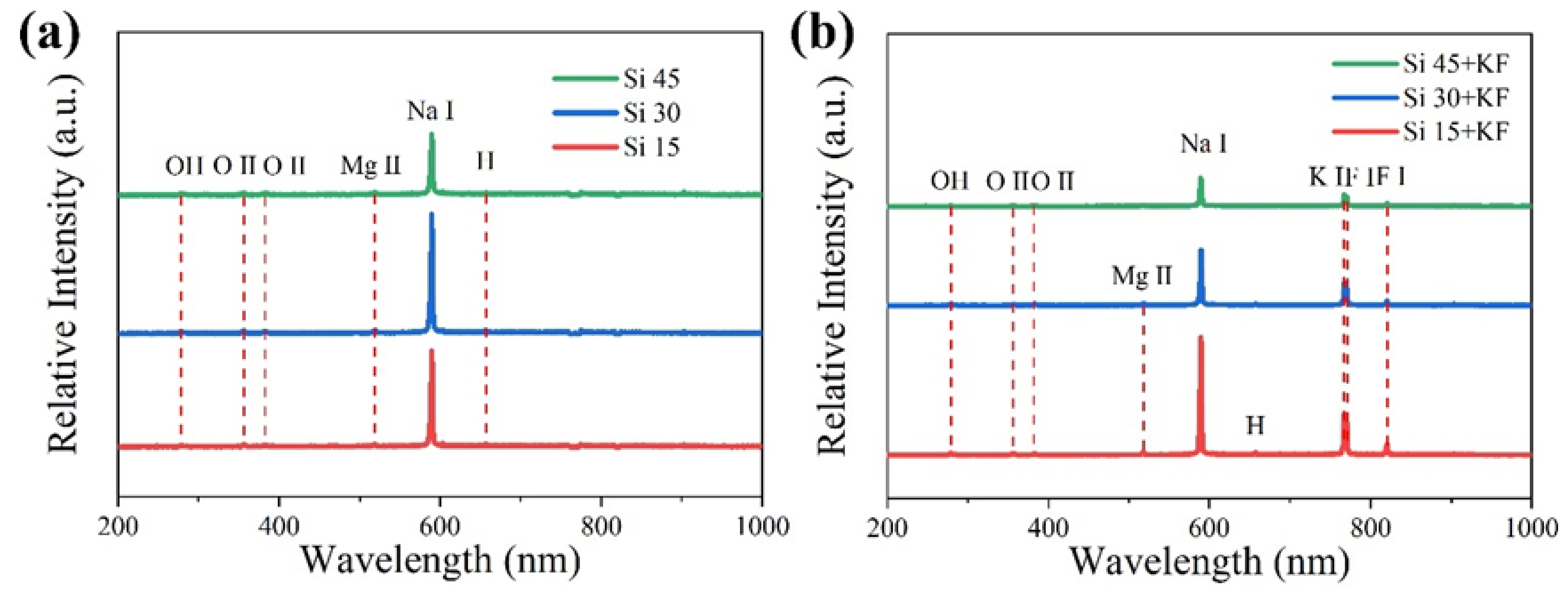
3.1. The Influence of SiO3 2−/F− on the Micro-Arc Oxidation Discharge Characteristics of Magnesium Alloy Surfaces
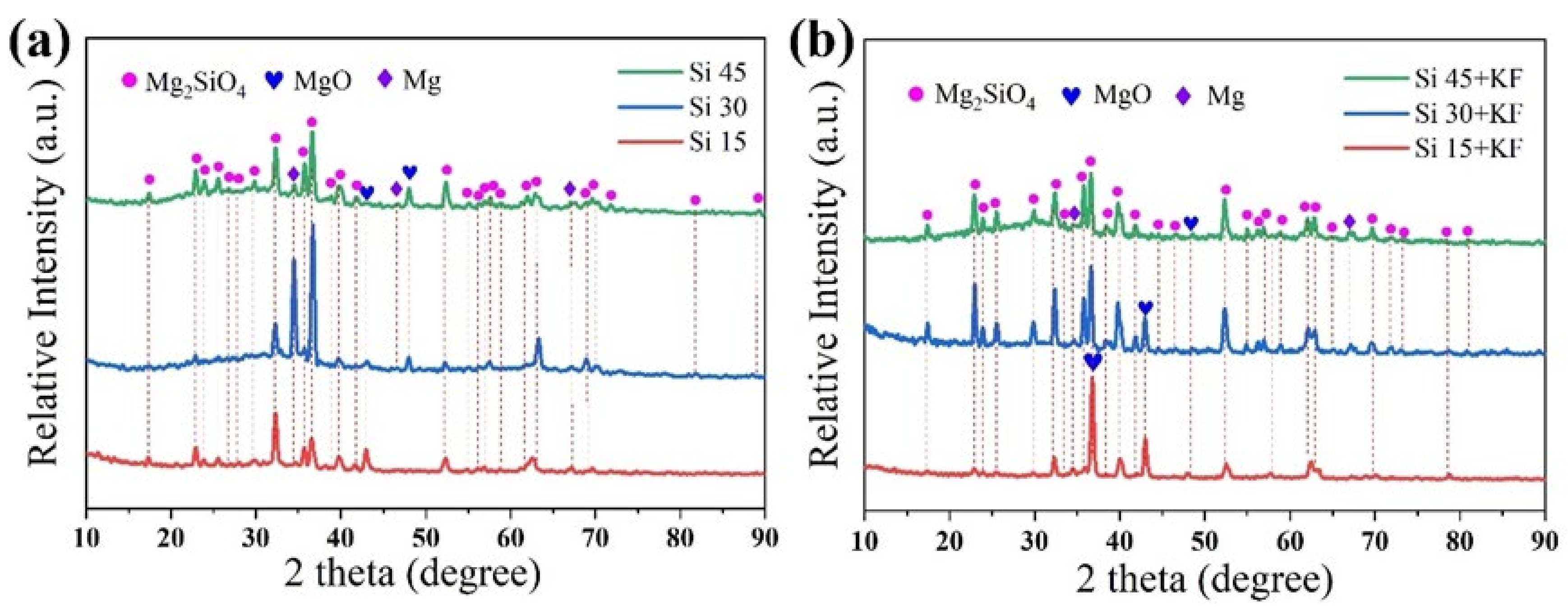
3.2. Effects of SiO32−/F− on the Phase Composition and Microstructure of Micro-Arc Oxidation Coatings on Magnesium Alloys
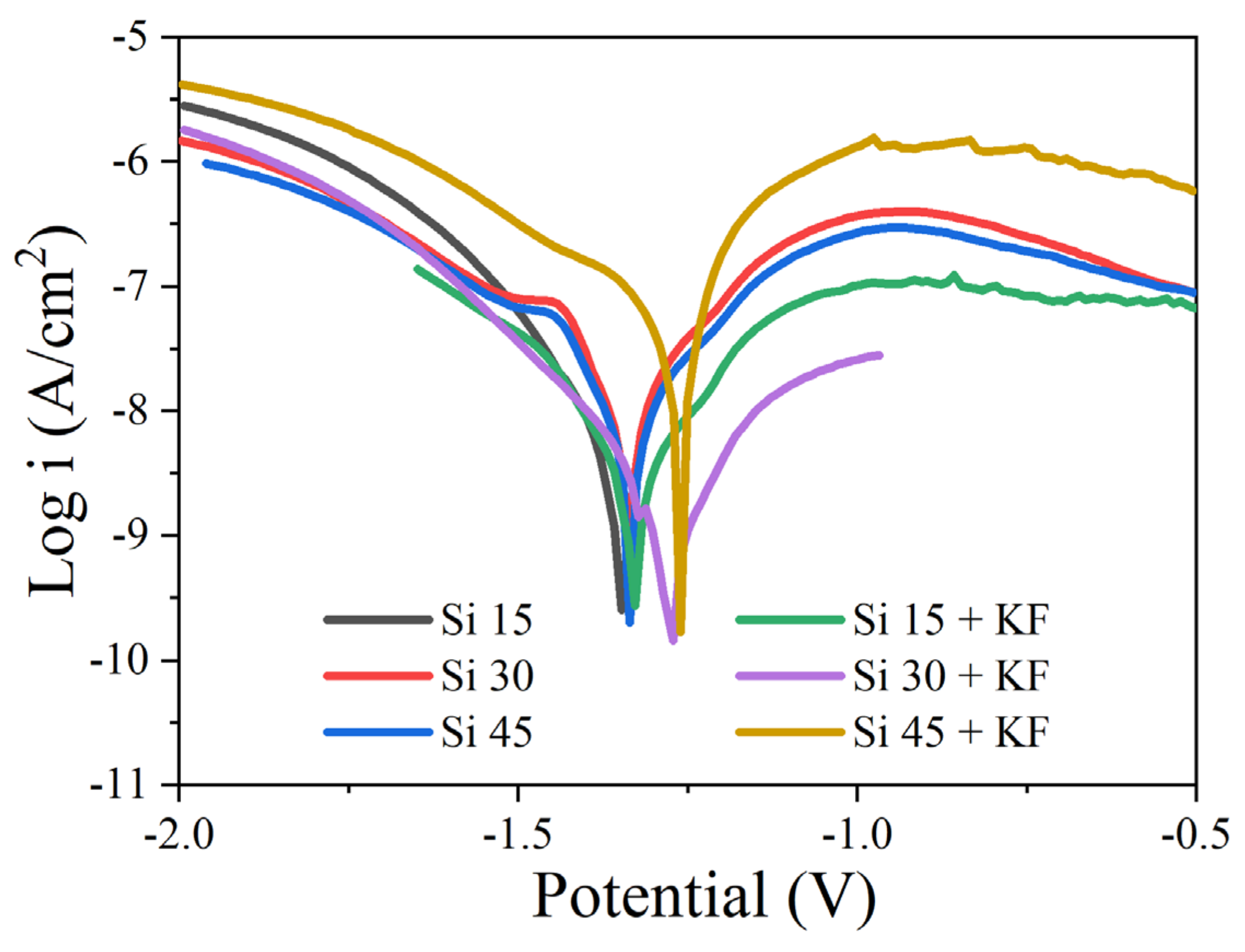
3.3. Influence of SiO32−/F− on the Corrosion Protection Performance of Micro-Arc Oxidation Coatings on Magnesium Alloys
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Richards, A.M.; Coleman, N.W.; Knight, T.A.; Belkoff, S.M.; Mears, S.C. Bone Density and Cortical Thickness in Normal, Osteopenic, and Osteoporotic Sacra. J. Osteoporos. 2010, 2010, 504078. [Google Scholar] [CrossRef]
- Wu, W.; Chen, L.; Zhang, D.; Xia, Y.; Huang, X. Research Progress on Micro-arc Oxidation of Mag-nesium-Lithium Alloys. Electroplat. Finish. 2023, 45, 71–79. [Google Scholar] [CrossRef]
- Song, G.-L.; Atrens, A. Recently Deepened Insights Regarding Mg Corrosion and Advanced Engineering Applications of Mg Alloys. J. Magnes. Alloy. 2023, 11, 3948–3991. [Google Scholar] [CrossRef]
- Sun, L.; Ma, Y.; Dong, H.; An, L.; Wang, S. Role of Sodium Silicate in the Formation of Micro-arc Oxi-dation Coatings on Magnesium Alloys. Chin. J. Rare Met. 2020, 44, 378–386. [Google Scholar] [CrossRef]
- Yang, W.; Liu, Z.; Huang, H. Galvanic Corrosion Behavior between AZ91D Magnesium Alloy and Copper in Distilled Water. Corros. Sci. 2021, 188, 109562. [Google Scholar] [CrossRef]
- Jiang, J.; Geng, X.; Zhang, X. Stress Corrosion Cracking of Magnesium Alloys: A Review. J. Magnes. Alloy. 2023, 11, 1906–1930. [Google Scholar] [CrossRef]
- Luo, Y.; Huang, J.; Yu, X.; Xie, Y.; Zhang, J.; Yu, S.; Fan, D.; Liu, J. Surface Microstructure and Corrosion Characterization of AZ31 Magnesium Alloys Fabricated by Laser Surface-Modification. J. Alloys Compd. 2024, 994, 174708. [Google Scholar] [CrossRef]
- Cui, B.; Wang, J.; Zhang, X.; Feng, Z.; Yang, G.; Yue, Y.; Li, J. Effects of Heat Treatment on the Microstructure and Corrosion Behavior of SLM AZ91D Magnesium Alloy. Surf. Technol. 2021, 50, 323–329+365. [Google Scholar] [CrossRef]
- Su, L.X. Influence of Heat Treatment Process on the Properties of Die-Cast Magnesium Alloy AM50. Master’s Thesis, Changchun University of Technology, Changchun, China, 2016. [Google Scholar]
- Zhang, C.; Qi, M.; Peng, P.; Long, S.; Wan, X.; Wang, Q.; Yang, Q.; Dai, Q.; Wu, L. Induced Growth Orientation Deviation of Mg(OH)2 by Heat Treatment to Enhance Corrosion Resistance of Mg-Sc Alloy. Corros. Sci. 2025, 257, 113271. [Google Scholar] [CrossRef]
- Shen, G.; Lyu, S.; Si, B.; Yu, Q.; Zheng, H.; Wang, X.; Chen, M. First-Principles Study of the Corrosion of Mg Alloys and Mg-Based Composites in Cl-Containing Electrolyte. Appl. Surf. Sci. 2025, 709, 163811. [Google Scholar] [CrossRef]
- Hwang, I.J.; Hwang, D.Y.; Ko, Y.G.; Shin, D.H. Correlation between Current Frequency and Electrochemical Properties of Mg Alloy Coated by Micro Arc Oxidation. Surf. Coat. Technol. 2012, 206, 3360–3365. [Google Scholar] [CrossRef]
- Zhu, H.; Wu, C.; Xue, L.; Yang, L.; Liu, Y.; Wang, D.; Liang, Y.; Peng, Z. Effect of Laser Cleaning on the Growth and Properties of Micro-Arc Oxidation Layers of AZ31 Magnesium Alloy. Surf. Coat. Technol. 2024, 488, 131051. [Google Scholar] [CrossRef]
- Ma, X.; Jin, S.; Wu, R.; Ji, Q.; Hou, L.; Krit, B.; Betsofen, S. Influence Alloying Elements of Al and Y in MgLi Alloy on the Corrosion Behavior and Wear Resistance of Microarc Oxidation Coatings. Surf. Coat. Technol. 2022, 432, 128042. [Google Scholar] [CrossRef]
- Tang, Y.; Zhao, X.; Jiang, K.; Chen, J.; Zuo, Y. The Influences of Duty Cycle on the Bonding Strength of AZ31B Magnesium Alloy by Microarc Oxidation Treatment. Surf. Coat. Technol. 2010, 205, 1789–1792. [Google Scholar] [CrossRef]
- Wang, J.; Dou, J.; Wang, Z.; Hu, C.; Liu, J.; Yu, H.; Chen, C. Corrosion Resistance and Biodegradability of Micro-Arc Oxidation Coatings with the Variable Sodium Fluoride Concentration on ZM21 Magnesium Alloys. J. Alloys Compd. 2023, 962, 171172. [Google Scholar] [CrossRef]
- Kazanski, B.; Kossenko, A.; Zinigrad, M.; Lugovskoy, A. Fluoride Ions as Modifiers of the Oxide Layer Produced by Plasma Electrolytic Oxidation on AZ91D Magnesium Alloy. Appl. Surf. Sci. 2013, 287, 461–466. [Google Scholar] [CrossRef]
- Thanaa, T.T.; Fattah-alhosseini, A.; Alkaseem, M.; Kaseem, M. Improving the Surface Properties of Mg Based-Plasma Electrolytic Oxidation (PEO) Coatings under the Fluoride Electrolytes: A Review. Inorg. Chem. Commun. 2024, 170, 113163. [Google Scholar] [CrossRef]
- Jia, Y.; Tian, H. Research Progress on Micro-arc Oxidation Technology of Magnesium Alloys. Shandong Chem. Ind. 2025, 54, 89–91. [Google Scholar] [CrossRef]
- Li, Z.; Wang, Y.; Wang, B.; Yuan, M.; Xun, M.; Zhang, H. Progress in the Study of Micro-Arc Oxidation Film Layers on Biomedical Metal Surfaces. Corros. Rev. 2025, 43, 175–187. [Google Scholar] [CrossRef]
- Chen, X.; Xie, W.; Tang, S.; Zhang, M.; Song, H.; Ran, Q.; Zhang, D. Study on the Structure and Corrosion Resistance of MoS2/MgO Micro-Arc Oxidation Composite Ceramic Coating on the Surface of ZK60 Magnesium Alloy. Anti-Corros. Methods Mater. 2024, 71, 213–222. [Google Scholar] [CrossRef]
- Yang, S.; Sun, R.; Chen, K. Self-Healing Performance and Corrosion Resistance of Phytic Acid/Cerium Composite Coating on Microarc-Oxidized Magnesium Alloy. Chem. Eng. J. 2022, 428, 131198. [Google Scholar] [CrossRef]
- Khan, M.A.; Safira, A.R.; Aadil, M.; Kaseem, M. Development of Anti-Corrosive Coating on AZ31 Mg Alloy Modified by MOF/LDH/PEO Hybrids. J. Magnes. Alloys 2024, 12, 586–607. [Google Scholar] [CrossRef]
- Asoh, H.; Asakura, K.; Hashimoto, H. Effect of Alcohol Addition on the Structure and Corrosion Resistance of Plasma Electrolytic Oxidation Films Formed on AZ31B Magnesium Alloy. RSC Adv. 2020, 10, 9026–9036. [Google Scholar] [CrossRef] [PubMed]
- Kaseem, M.; Hussain, T.; Rehman, Z.U.; Ko, Y.G. Stabilization of AZ31 Mg Alloy in Sea Water via Dual Incorporation of MgO and WO3 during Micro-Arc Oxidation. J. Alloys Compd. 2021, 853, 157036. [Google Scholar] [CrossRef]
- Zhang, Y.; Guo, Y.; Zhou, P.; Zhang, T.; Wang, F.; Chen, L. A Plasma Electrolytic Oxidation Coating on High-Purity Mg with Excellent Long-Term Degradation Performance and Biocompatibility. Surf. Coat. Technol. 2024, 493, 131236. [Google Scholar] [CrossRef]
- Tang, Q.; Li, X.; Liu, G.; Zhai, D.; Shen, J. Influence of Co-Doping on Soft Spark Micro-Arc Oxidation on ZrO2-AZ91D Magnesium Alloy. Metall. Mater. Trans. A-Phys. Metall. Mater. Sci. 2024, 55, 745–760. [Google Scholar] [CrossRef]
- Mori, Y.; Koshi, A.; Liao, J.; Asoh, H.; Ono, S. Characteristics and Corrosion Resistance of Plasma Electrolytic Oxidation Coatings on AZ31B Mg Alloy Formed in Phosphate–Silicate Mixture Electrolytes. Corros. Sci. 2014, 88, 254–262. [Google Scholar] [CrossRef]
- Sun, L.; Ma, Y.; An, L.; Wang, X.; Gao, W. Comparative Study on the Electrochemical Response of Corrosion Resistance of Micro-arc Oxidation Coatings with Different Thicknesses under High and Low Voltages. Surf. Technol. 2021, 50, 366–374. [Google Scholar] [CrossRef]
- Wu, T.; Zhang, K. Corrosion and Protection of Magnesium Alloys: Recent Advances and Future Perspectives. Coatings 2023, 13, 1533. [Google Scholar] [CrossRef]
- Liu, Y.; Li, L.; Lu, G.; Yu, S. Preparation of Biomimetic Hydrophobic Coatings on AZ91D Magnesium Alloy Surface. Sci. China Technol. Sci. 2010, 53, 2972–2975. [Google Scholar] [CrossRef]
- Matthew, J.A.D. Crc handbook of chemistry and physics–weast. RC Nat. 1988, 331, 127. [Google Scholar] [CrossRef]





| Element | Al | Zn | Si | Mn | Fe | Cu | Mg |
|---|---|---|---|---|---|---|---|
| Contents (%) | 3.12 | 1.04 | 0.006 | 0.44 | 0.001 | 0.001 | bal. |
| Specimens | Electrolyte |
|---|---|
| Si 15 | 15 g/L Na2SiO3·9H2O + 5 g/L NaOH |
| Si 30 | 30 g/L Na2SiO3·9H2O + 5 g/L NaOH |
| Si 45 | 45 g/L Na2SiO3·9H2O + 5 g/L NaOH |
| Si 15 + KF | 15 g/L Na2SiO3·9H2O + 5 g/L NaOH + 0.129 mol/L KF·2H2O |
| Si 30 + KF | 30 g/L Na2SiO3·9H2O + 5 g/L NaOH + 0.129 mol/L KF·2H2O |
| Si 45 + KF | 45 g/L Na2SiO3·9H2O + 5 g/L NaOH + 0.129 mol/L KF·2H2O |
| Specimens | pH | Conductivity (mS/cm) |
|---|---|---|
| Si 15 | 12.8 | 40.6 |
| Si 30 | 13.02 | 52.4 |
| Si 45 | 13.18 | 63.1 |
| Si 15+KF | 13.11 | 50.9 |
| Si 30+KF | 13.31 | 59.3 |
| Si 45+KF | 13.39 | 70.5 |
| Specimens | Average Size (μm) | Area (%) |
|---|---|---|
| Si 15 | 1.487 | 8.732 |
| Si 30 | 2.14 | 8.632 |
| Si 45 | 3.817 | 8.616 |
| Si 15 + KF | 1.418 | 8.053 |
| Si 30 + KF | 1.209 | 7.83 |
| Si 45 + KF | 3.529 | 7.438 |
| Specimens | Content of Elements (wt.%) | |||||
|---|---|---|---|---|---|---|
| Mg | O | Si | F | Na | K | |
| Si 15 + KF | 38.19 | 41.94 | 13.04 | 5.26 | 1.28 | 0.29 |
| Si 30 + KF | 29.90 | 43.48 | 18.48 | 4.02 | 3.13 | 0.99 |
| Si 45 + KF | 22.99 | 44.25 | 22.92 | 1.05 | 6.67 | 2.13 |
| Specimens | Ecorr (mV vs. Ag/AgCl) | icorr (A/cm2) |
|---|---|---|
| Si 15 | −1357 | 5.13 × 10−8 |
| Si 30 | −1336 | 4.76 × 10−8 |
| Si 45 | −1328 | 3.62 × 10−8 |
| Si 15 + KF | −1327 | 8.91 × 10−9 |
| Si 30 + KF | −1276 | 3.89 × 10−9 |
| Si 45 + KF | −1268 | 9.12 × 10−8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Y.; Zhang, S.; Li, H.; Liu, C.; Wang, H.; Ma, L. Magnesium Silicate Coatings Were Prepared by Micro-Arc Oxidation on the Surface of Magnesium Alloys Through the Synergistic Effect of SiO32−/F−. Materials 2025, 18, 4760. https://doi.org/10.3390/ma18204760
Zhang Y, Zhang S, Li H, Liu C, Wang H, Ma L. Magnesium Silicate Coatings Were Prepared by Micro-Arc Oxidation on the Surface of Magnesium Alloys Through the Synergistic Effect of SiO32−/F−. Materials. 2025; 18(20):4760. https://doi.org/10.3390/ma18204760
Chicago/Turabian StyleZhang, Yuru, Shudong Zhang, Hongtao Li, Cancan Liu, Hao Wang, and Li Ma. 2025. "Magnesium Silicate Coatings Were Prepared by Micro-Arc Oxidation on the Surface of Magnesium Alloys Through the Synergistic Effect of SiO32−/F−" Materials 18, no. 20: 4760. https://doi.org/10.3390/ma18204760
APA StyleZhang, Y., Zhang, S., Li, H., Liu, C., Wang, H., & Ma, L. (2025). Magnesium Silicate Coatings Were Prepared by Micro-Arc Oxidation on the Surface of Magnesium Alloys Through the Synergistic Effect of SiO32−/F−. Materials, 18(20), 4760. https://doi.org/10.3390/ma18204760






