Burdock-Derived Composites Based on Biogenic Gold, Silver Chloride and Zinc Oxide Particles as Green Multifunctional Platforms for Biomedical Applications and Environmental Protection
Abstract
1. Introduction
- (i)
- In the first step, biogenic monocomponent particles were prepared. For this purpose, the burdock aqueous extract was used as a precursor for “green” particles of gold, silver chloride, and zinc oxide (AuNPs, AgClNPs, and ZnO, respectively).
- (ii)
- The developed monocomponent particles were further used as building blocks to achieve bi- (AuZnO and AgClZnO) and tricomponent (AuAgClZnO) particles.
- (iii)
- The complex characterization of AuAgClZnO composite as compared to AuNPs, AgClNPs, ZnO, AuZnO, and AgClZnO.
2. Materials and Methods
2.1. Preparation of AgClNPs and AuNPs by Using the Burdock Extract
2.2. Preparation of Composites Based on ZnO, AgClNPs, AuNPs, and a Mixture of AuNPs-AgClNPs in Burdock Extract
- (i)
- HAuCl4,aq + EB (phyto-compounds) → AuNPs;
- (ii)
- AgNO3,aq + EB (phyto-compounds) → AgClNPs;
- (iii)
- Zn(NO3)2 + 2NaOH → Zn(OH)2↓ + 2NaNO3;
- (iv)
- EB/AuNPs/AgClNPs/(AuNPs + AgClNPs) + Zn(OH)2ZnO/AuZnO/AgClZnO/AuAgClZnO + H2O.
- (i)
- Zn(NO3)2 + 2NaOH → Zn(OH)2↓ + 2NaNO3;
- (ii)
- EB + Zn(OH)2.
2.3. Physicochemical and Biological Characterization of “Green” Developed Composites
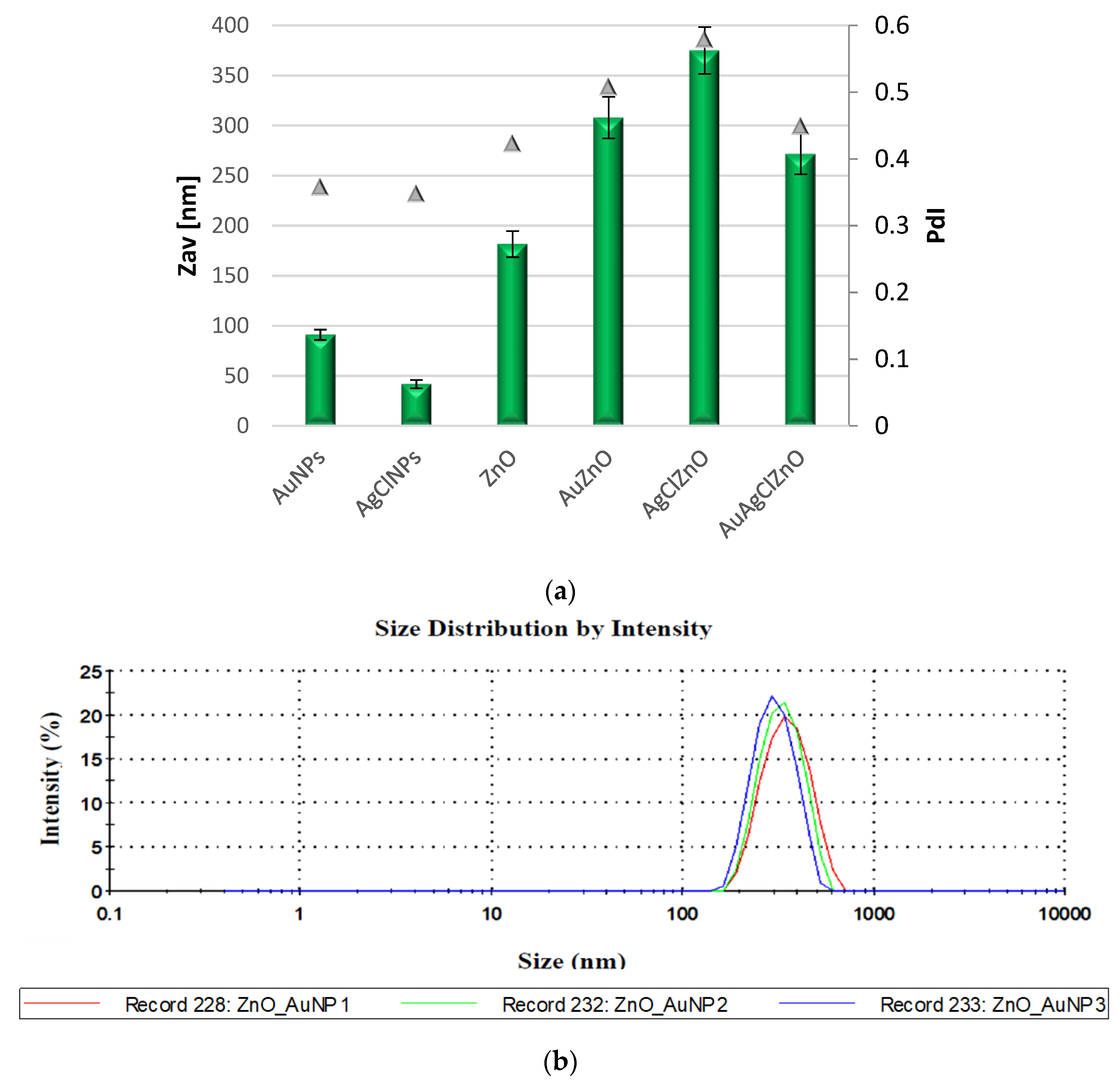
2.4. Evaluation of Particle Size Distribution
2.5. Electrokinetic Potential Analysis
2.6. Biological Characterization of Developed Materials
2.6.1. In Vitro Antioxidant Activity Analysis
2.6.2. In Vitro Antibacterial Activity Analysis
3. Results and Discussion
3.1. Optical Characterization of Phytoderived Materials
3.2. Evaluation of Zeta Potential of the Phytometallic Particles
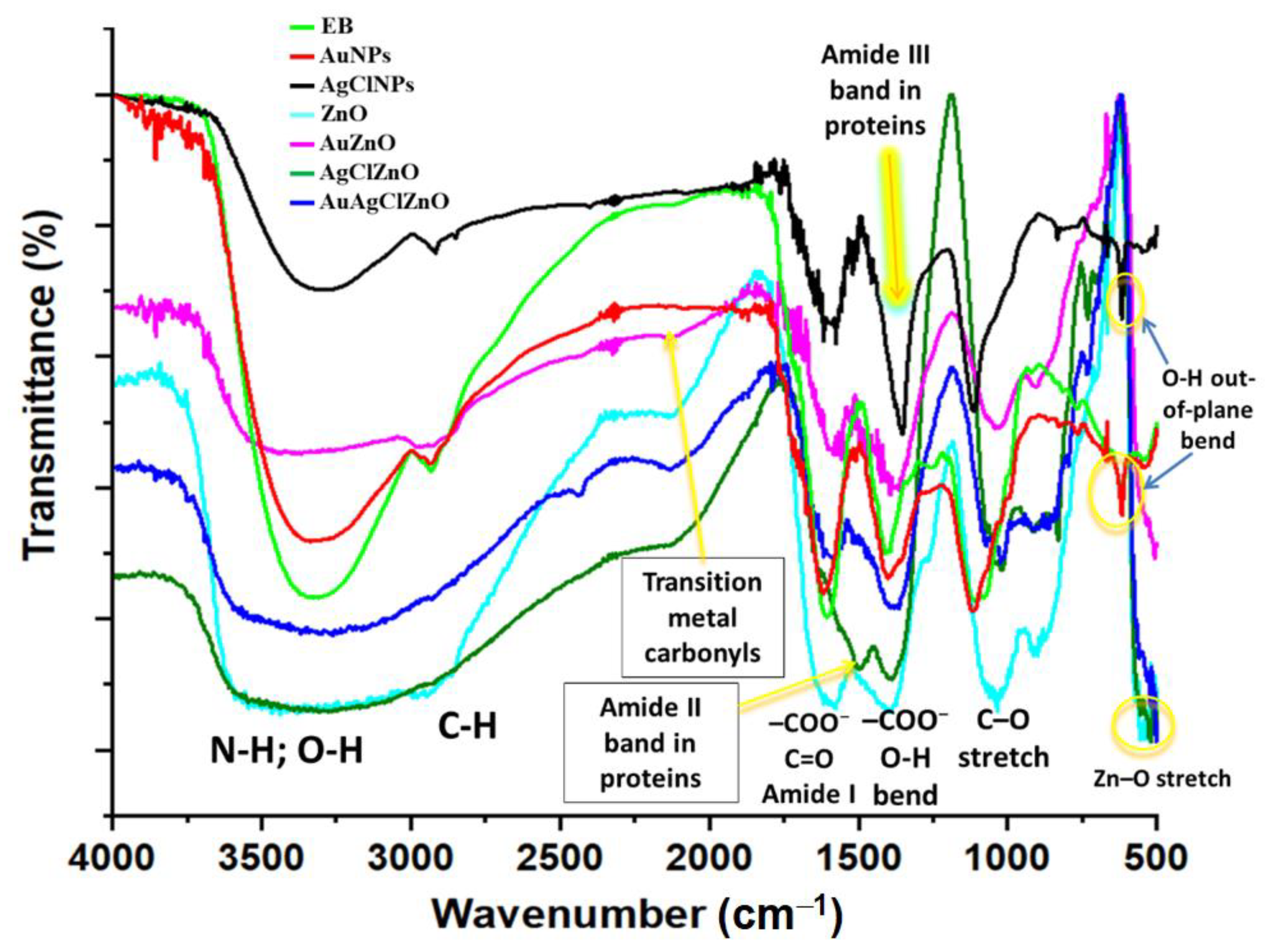
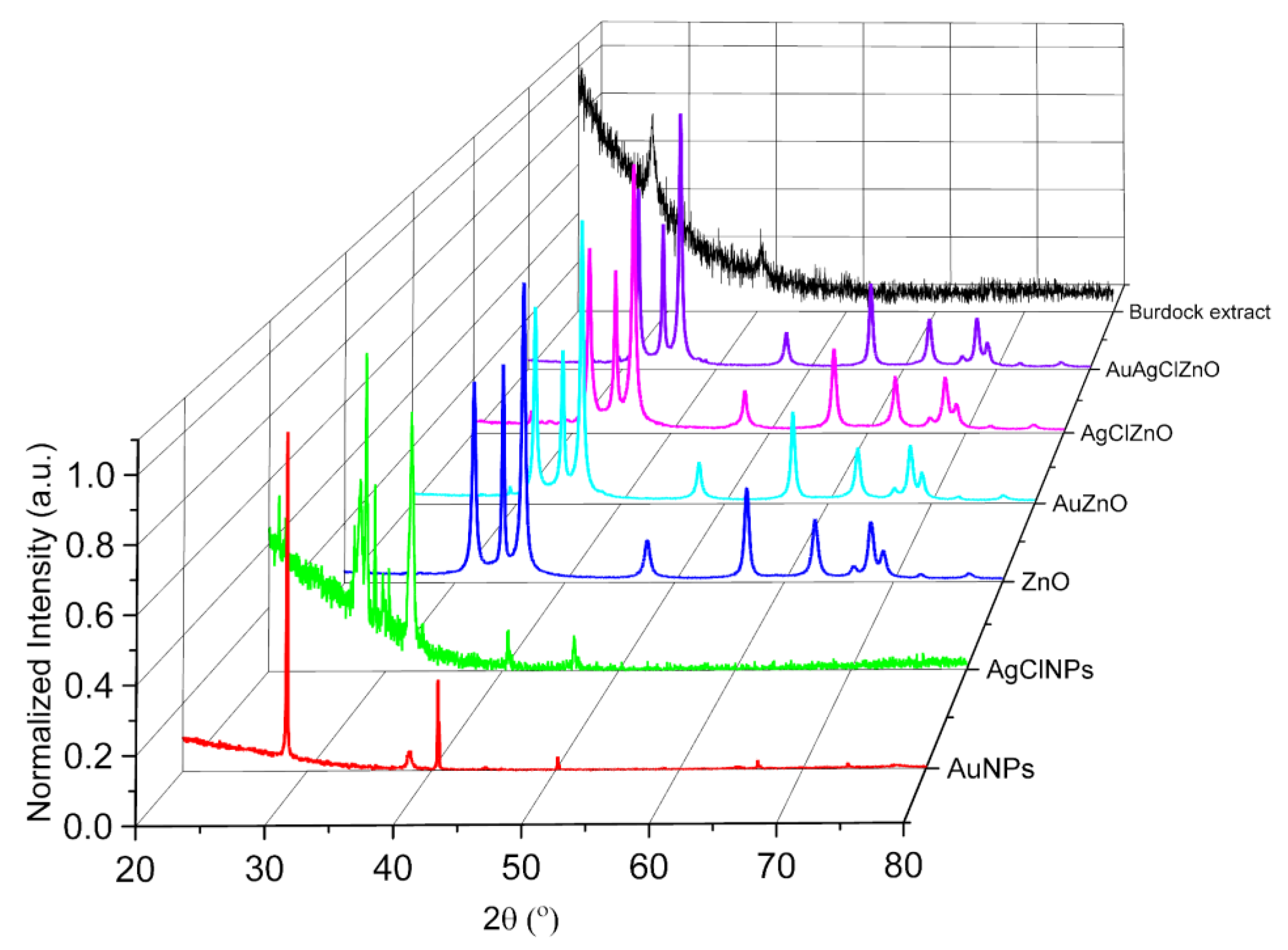
3.3. Structural Characterization
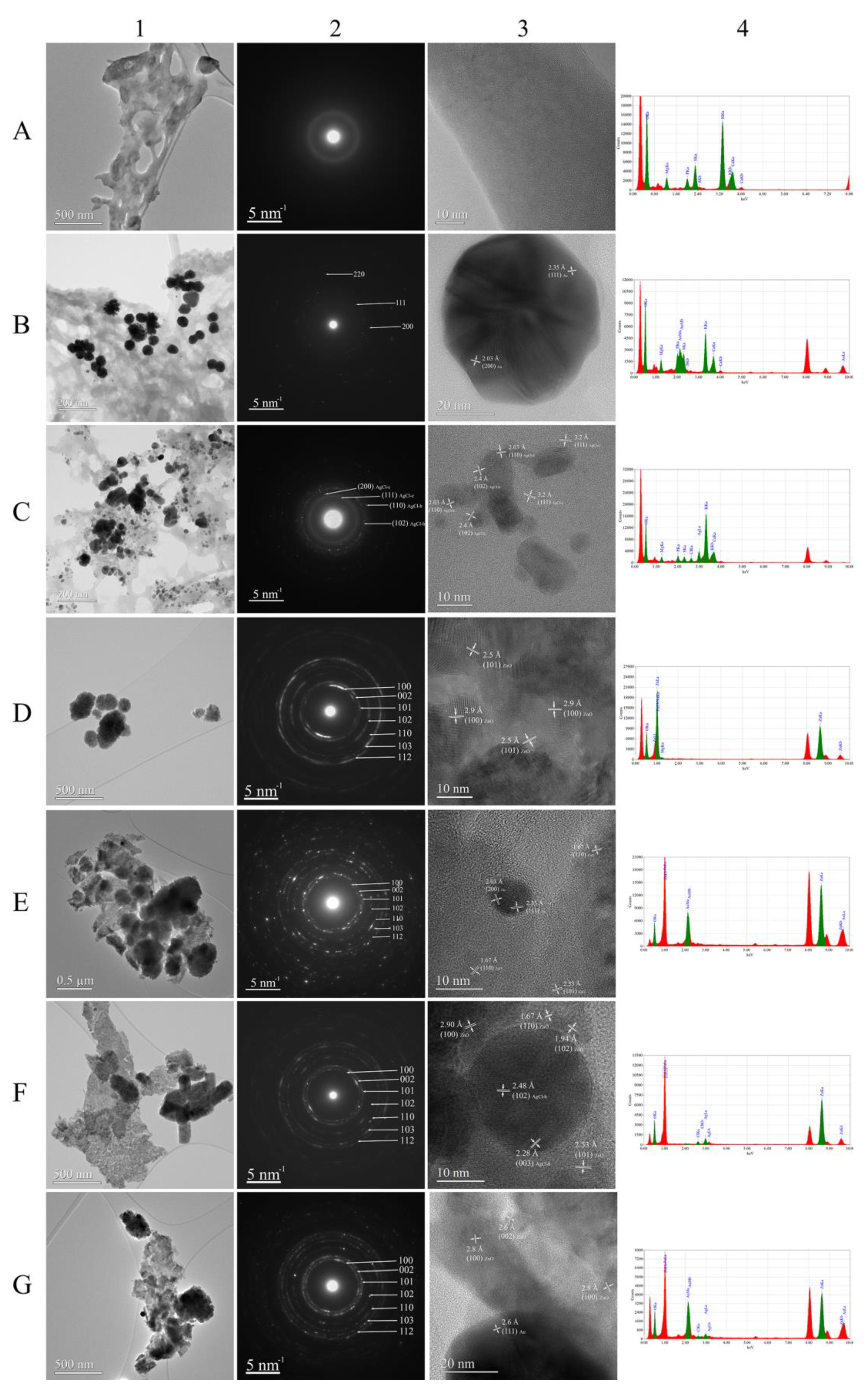
3.4. Size and Morphological Characterization of Phytmaterials
3.5. Morphological and Compositional Characterization
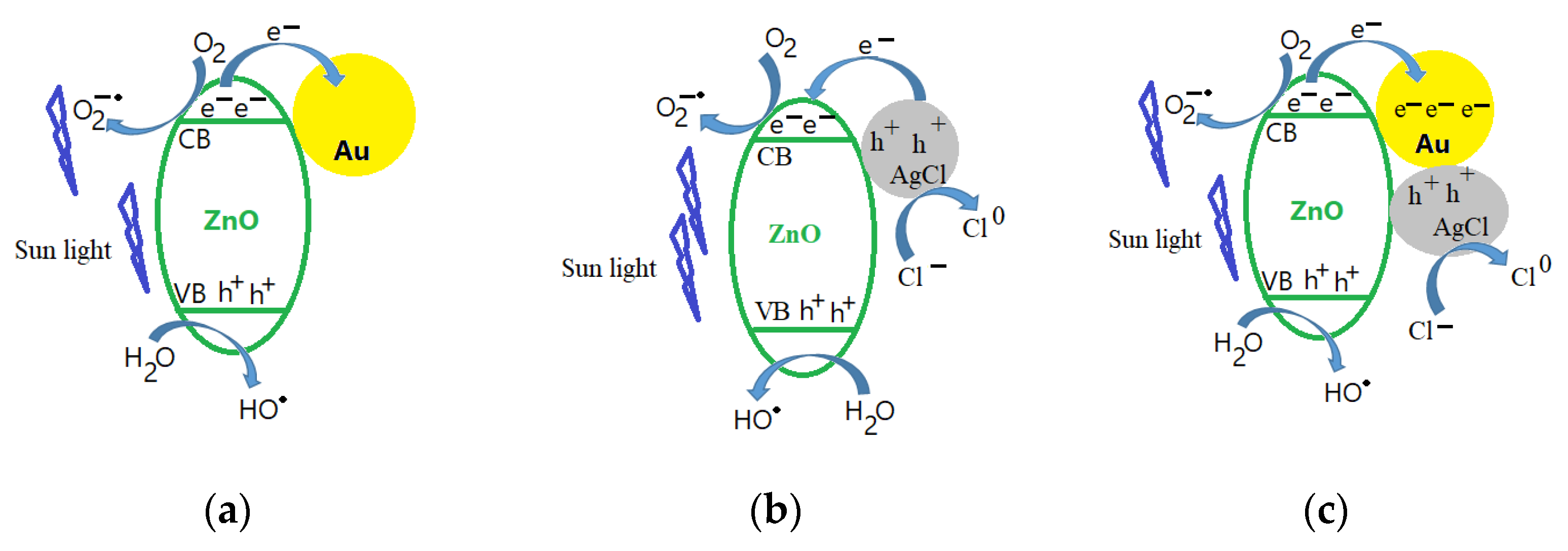
3.6. The Photocatalytic Properties of Obtained ZnO-Based Materials
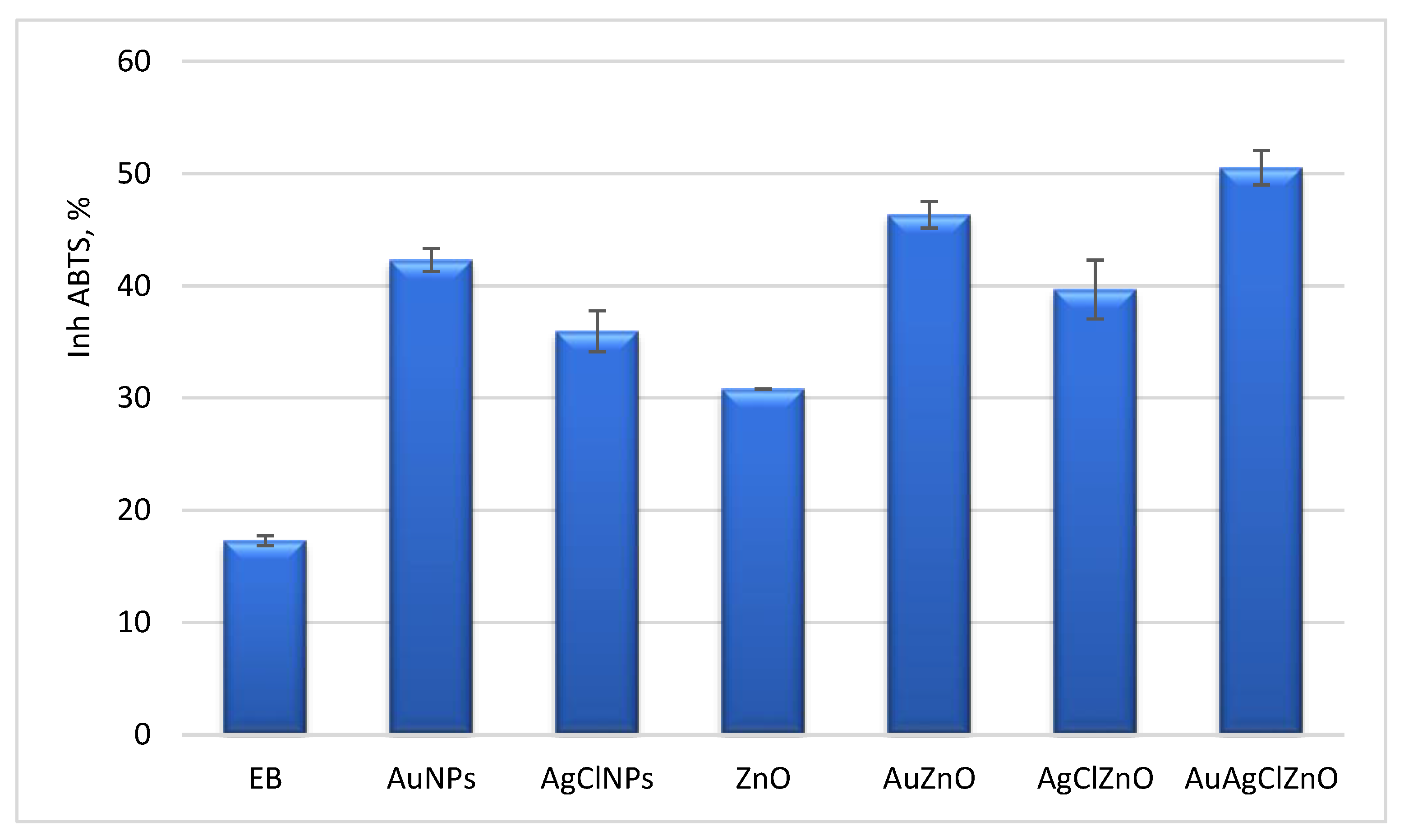
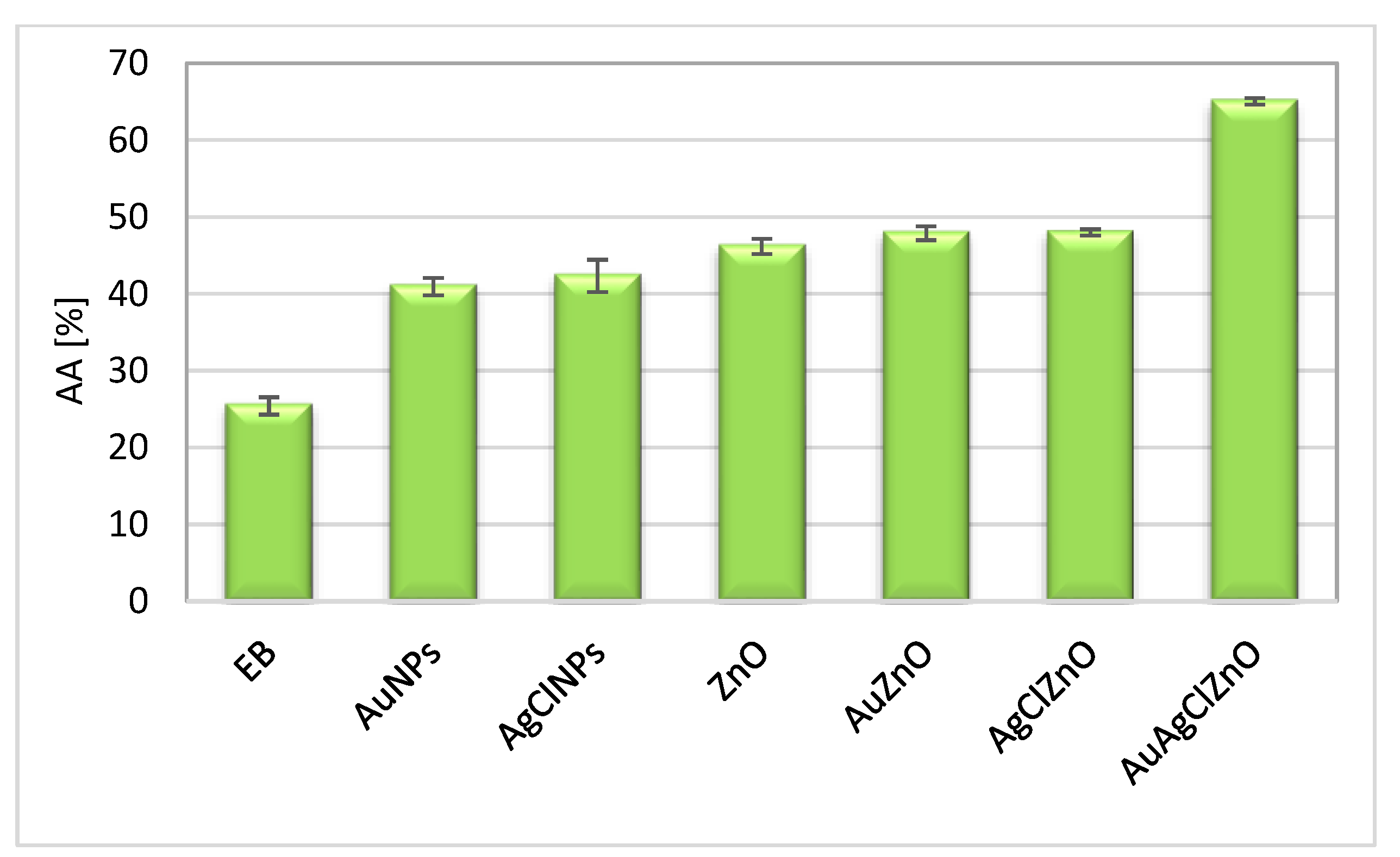
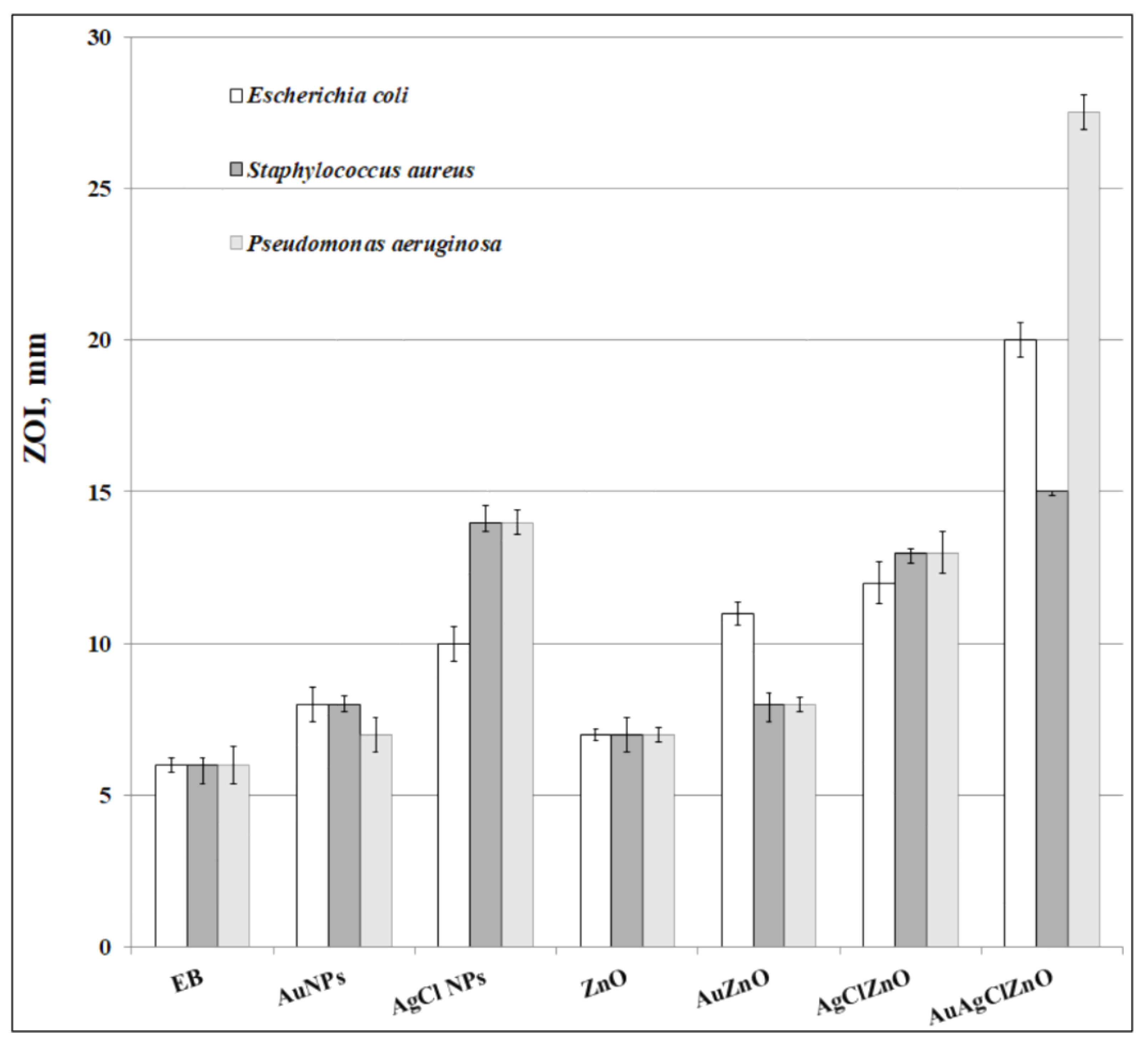
3.7. Evaluation of Antioxidant and Antibacterial Properties of Phytoderived Materials
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nadaf, S.J.; Jadhav, N.R.; Naikwadi, H.S.; Savekar, P.L.; Sapkal, I.D.; Kambli, M.M.; Desai, I.A. Green Synthesis of Gold and Silver Nanoparticles: Updates on Research, Patents, and Future Prospects. OpenNano 2022, 8, 100076. [Google Scholar] [CrossRef]
- Katas, H.; Moden, N.Z.; Lim, C.S.; Celesistinus, T.; Chan, J.Y.; Ganasan, P.; Abdalla, S.S.I. Biosynthesis and potential applications of silver and gold nanoparticles and their chitosan-based nanocomposites in nanomedicine. J. Nanotechnol. 2018, 2018, 4290705. [Google Scholar] [CrossRef]
- Mody, V.V.; Siwale, R.; Singh, A.; Mody, H.R. Introduction to metallic nanoparticles. J. Pharm. Bioallied Sci. 2010, 2, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, D.J.; Duran, A.; Cabral, A.D.; Fonseca, F.L.A.; Bueno, R.F.; Rosa, D.S. Questioning ZnO, Ag, and Ag/ZnO nanoparticles as antimicrobial agents for textiles: Do they guarantee total protection against bacteria and SARS-CoV-2? J. Photochem. Photobiol. B-Biol. 2022, 234, 112538. [Google Scholar] [CrossRef]
- Koga, H.; Umemura, Y.; Kitaoka, T. Design of catalyst layers by using paper-like fiber/metal nanocatalyst composites for efficient NOX reduction. Compos. Part B 2011, 42, 1108–1113. [Google Scholar] [CrossRef]
- Pragathiswaran, C.; Thulasi, G.; Al-Ansari, M.M.; Al-Humaid, L.A.; Saravanan, M. Experimental investigation and electrochemical characterization of titanium coated nanocomposite materials for biomedical applications. J. Mol. Struct. 2021, 1231, 129932. [Google Scholar] [CrossRef]
- Koga, H.; Umemura, Y.; Tomoda, A.; Suzuki, R.; Kitaoka, T. In situ synthesis of platinum nanocatalysts on a microstructured paperlike matrix for the catalytic purification of exhaust gases. ChemSusChem 2010, 3, 604–608. [Google Scholar] [CrossRef]
- Majee, S.; Karlsson, M.C.F.; Wojcik, P.J.; Sawatdee, A.; Mulla, M.Y.; ul Hassan Alvi, N.; Dyreklev, P.; Beni, V.; Nilsson, D. Low temperature chemical sintering of inkjet-printed Zn nanoparticles for highly conductive flexible electronic components. npj Flex. Electron. 2021, 5, 14. [Google Scholar] [CrossRef]
- Hong, G.-B.; Wang, J.-F.; Chuang, K.-J.; Cheng, H.-Y.; Chang, K.-C.; Ma, C.-M. Preparing Copper Nanoparticles and Flexible Copper Conductive Sheets. Nanomaterials 2022, 12, 360. [Google Scholar] [CrossRef]
- Andritsos, K.; Theodorakos, I.; Zacharatos, F.; Kabla, A.; Melamed, S.; de la Vega, F.; Porte, Y.; Too, P.; Zergioti, I. Conformal laser printing and laser sintering of Ag nanoparticle inks: A digital approach for the additive manufacturing of micro-conductive patterns on patterned flexible substrates. Virtual Phys. Prototyp. 2023, 18, e2138462. [Google Scholar] [CrossRef]
- Matsukawa, M.; Wang, K.-H.; Imura, Y.; Kawai, T. Au Nanoparticle Monolayer Nanosheets as Flexible Transparent Conductive Electrodes. ACS Appl. Nano Mater. 2021, 4, 10845–10851. [Google Scholar] [CrossRef]
- Wali, L.A.; Dheyab, A.B.; Alwan, A.M. Study the influence of shell thickness in bimetallic Ag core&Au shell configurations integrated in bare Si PN junction solar cells. Mater. Sci. Eng. B-Solid State Mater. Adv. Technol. 2023, 288, 116210. [Google Scholar]
- Jayarambabu, N.; Velupla, S.; Akshaykranth, A.; Anitha, N.; Rao, T.V. Bambusa arundinacea leaves extract-derived Ag NPs: Evaluation of the photocatalytic, antioxidant, antibacterial, and anticancer activities. Appl. Phys. A-Mater. Sci. Process. 2023, 129, 13. [Google Scholar] [CrossRef]
- Zhang, S.; Jiang, W.; Liu, G.; Liu, S.; Chen, H.; Lyu, G.; Yang, G.; Liu, Y.; Ni, Y. Preparation of ultrafine and highly loaded silver nanoparticle composites and their highly efficient applications as reductive catalysts and antibacterial agents. J. Colloid Interface Sci. 2023, 629, 766–777. [Google Scholar] [CrossRef] [PubMed]
- Kaloti, M.; Kumar, A. Sustainable Catalytic Activity of Ag-Coated Chitosan-Capped γ-Fe2O3 Superparamagnetic Binary Nanohybrids (Ag-γ-Fe2O3@CS) for the Reduction of Environmentally Hazardous Dyes—A Kinetic Study of the Operating Mechanism Analyzing Methyl Orange Reduction. ACS Omega 2018, 3, 1529–1545. [Google Scholar] [CrossRef] [PubMed]
- Campelo, J.; Luna, D.; Luque, R.; Marinas, J.; Romero, A. Sustainable Preparation of Supported Metal Nanoparticles and Their Applications in Catalysis. ChemSusChem 2009, 2, 18–45. [Google Scholar] [CrossRef] [PubMed]
- Manias, E. Stiffer by design. Nat. Mater. 2007, 6, 9–11. [Google Scholar] [CrossRef] [PubMed]
- Naseem, T.; Waseem, M. A comprehensive review on the role of some important nanocomposites for antimicrobial and wastewater applications. Int. J. Environ. Sci. Technol. 2022, 19, 2221–2246. [Google Scholar] [CrossRef]
- Yaqoob, A.A.; Ahmad, H.; Parveen, T.; Ahmad, A.; Oves, M.; Ismail, I.M.I.; Qari, H.A.; Umar, K.; Mohamad Ibrahim, M.N. Recent advances in metal decorated nanomaterials and their various biological applications: A review. Front. Chem. 2020, 8, 341. [Google Scholar] [CrossRef]
- Patra, S.; Mukherjee, S.; Barui, A.K.; Ganguly, A.; Sreedhar, B.; Patra, C.R. Green synthesis, characterization of gold and silver nanoparticles and their potential application for cancer therapeutics. Mater. Sci. Eng. C 2015, 53, 298–309. [Google Scholar] [CrossRef]
- Barbinta-Patrascu, M.E.; Ungureanu, C.; Iordache, S.M.; Bunghez, I.R.; Badea, N.; Rau, I. Green silver nanobioarchitectures with amplified antioxidant and antimicrobial properties. J. Mater. Chem. B 2014, 2, 3221–3231. [Google Scholar] [CrossRef]
- Barbinta-Patrascu, M.E.; Badea, N.; Ungureanu, C.; Constantin, M.; Pirvu, C.; Rau, I. Silver-based biohybrids ‘‘green” synthesized from Chelidonium majus L. Opt. Mater. 2016, 56, 94–99. [Google Scholar] [CrossRef]
- Rad, S.S.; Sani, A.M.; Mohseni, S. Biosynthesis, characterization and antimicrobial activities of zinc oxide nanoparticles from leaf extract of Mentha pulegium (L.). Microb. Pathog. 2019, 131, 239–245. [Google Scholar] [CrossRef]
- Yu, N.; Peng, H.; Qiu, L.; Wang, R.; Jiang, C.; Cai, T.; Sun, Y.; Li, Y.; Xiong, H. New pectin-induced green fabrication of Ag@AgCl/ZnO nanocomposites for visible-light triggered antibacterial activity. Int. J. Biol. Macromol. 2019, 141, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Dobrucka, R.; Romaniuk-Drapala, A.; Kaczmarek, M. Biologically synthesized of Au/Pt/ZnO nanoparticles using Arctium lappa extract and cytotoxic activity against leukemia. Biomed. Microdevices 2020, 22, 72. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, A.K.; Verma, N.; Kaushal, P. Role of Biogenic Capping Agents in the Synthesis of Metallic Nanoparticles and Evaluation of Their Therapeutic Potential. Front. Nanotechnol. 2022, 3, 801620. [Google Scholar] [CrossRef]
- Carneiro, R.d.S.; Canuto, M.R.; Ribeiro, L.K.; Ferreira, D.C.L.; Assunção, A.F.C.; Costa, C.A.C.B.; de Freitas, J.D.; Rai, M.; Cavalcante, L.S.; Alves, W.d.S.; et al. Novel Antibacterial Efficacy of ZnO Nanocrystals/Ag Nanoparticles Loaded with Extract of Ximenia americana L. (Stem Bark) for Wound Healing. S. Afr. J. Bot. 2022, 151, 18–32. [Google Scholar] [CrossRef]
- Selvan, D.S.A.; Murugesan, S.; Shobana, S.; Lakshmi, B.; Veena, V.; Rahiman, A.K. In vitro cytotoxicity efficacy of phytosynthesized ag/ZnO nanocomposites using Murraya koenigii and Zingiber officinale extracts. Mater. Chem. Phys. 2021, 272, 124903. [Google Scholar] [CrossRef]
- Gavade, N.L.; Kadam, A.N.; Babar, S.B.; Gophane, A.D.; Garadkar, K.M.; Lee, S.-W. Biogenic synthesis of gold-anchored ZnO nanorods as photocatalyst for sunlight-induced degradation of dye effluent and its toxicity assessment. Ceram. Int. 2020, 46, 11317–11327. [Google Scholar] [CrossRef]
- Anjum, S.; Nawaz, K.; Ahmad, B.; Hano, C.; Abbasi, B.H. Green synthesis of biocompatible core–shell (Au–Ag) and hybrid (Au–ZnO and Ag–ZnO) bimetallic nanoparticles and evaluation of their potential antibacterial, antidiabetic, antiglycation and anticancer activities. RSC Adv. 2022, 12, 23845. [Google Scholar] [CrossRef]
- Mahmoudi, B.; Soleimani, F.; Keshtkar, H.; Ali Nasseri, M.; Kazemnejadi, M. Green synthesis of trimetallic oxide nanoparticles and their use as an efficient catalyst for the green synthesis of quinoline and spirooxindoles: Antibacterial, cytotoxicity and anti-colon cancer effects. Inorg. Chem. Commun. 2021, 133, 108923. [Google Scholar] [CrossRef]
- Das, P.; Karankar, V.S. New avenues of controlling microbial infections through anti-microbial and anti-biofilm potentials of green mono-and multi-metallic nanoparticles: A review. J. Microbiol. Methods 2019, 167, 105766. [Google Scholar] [CrossRef] [PubMed]
- Barbinta-Patrascu, M.E.; Badea, N.; Ungureanu, C.; Besliu, D.; Antohe, S. Bioactive phyto-nanosilver particles “green” synthesized from Clary Sage, Burdock, Southernwood and Asparagus. Rom. Rep. Phys. 2020, 72, 606. [Google Scholar]
- Ferracane, R.; Graziani, G.; Gallo, M.; Fogliano, V.; Ritieni, A. Metabolic profile of the bioactive compounds of burdock (Arctium lappa) seeds, roots and leaves. J. Pharm. Biomed. Anal. 2010, 51, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Lacatusu, I.; Badea, G.; Popescu, M.; Bordei, N.; Istrati, D.; Moldovan, L.; Seciu, A.M.; Pandeli, M.I.; Rasit, I.; Badea, N. Marigold extract, azelaic acid and black caraway oil into lipid nanocarriers provides a strong anti-inflammatory effect in vivo. Ind. Crop Prod. 2017, 109, 141–150. [Google Scholar] [CrossRef]
- The European Committee on Antimicrobial Susceptibility Testing. Clinical Breakpoints—Bacteria (v 10.0). 2020. Available online: https://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/Breakpoint_tables/v_10.0_Breakpoint_Tables.pdf (accessed on 10 November 2020).
- Kowalska-Krochmal, B.; Dudek-Wicher, R. The Minimum Inhibitory Concentration of Antibiotics: Methods, Interpretation, Clinical Relevance. Pathogens 2021, 10, 165. [Google Scholar] [CrossRef]
- NCCLS M7 A6; Methods for Dilution Antimicrobial Susceptibility. Test for Bacteria that Grow Aerobically. NCCLS: Wayne, PA, USA, 2003.
- CLSI M02-A11; Performance Standards for Antimicrobial Disk Susceptibility Tests. Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2012.
- CLSI. Method for Antifungal Disk Diffusion Susceptibility Testing of Yeasts, Approved Guideline; CLSI document M44-A; CLSI: Wayne, PA, USA, 2004. [Google Scholar]
- Zgura, I.; Enculescu, M.; Istrate, C.; Negrea, R.; Bacalum, M.; Nedelcu, L.; Barbinta-Patrascu, M.E. Performant composite materials based on oxide semiconductors and metallic nanoparticles generated from cloves and mandarin peel extracts. Nanomaterials 2020, 10, 2146. [Google Scholar] [CrossRef]
- Rahman, F.; Majed Patwary, M.A.; Bakar Siddique, M.A.; Bashar, M.S.; Haque, M.A.; Akter, B.; Rashid, R.; Haque, M.A.; Royhan Uddin, A.K.M. Green synthesis of zinc oxide nanoparticles using Cocos nucifera leaf extract: Characterization, antimicrobial, antioxidant and photocatalytic activity. R. Soc. Open Sci. 2022, 9, 220858. [Google Scholar] [CrossRef]
- Zgura, I.; Preda, N.; Enculescu, M.; Diamandescu, L.; Negrila, C.; Bacalum, M.; Ungureanu, C.; Barbinta-Patrascu, M.E. Cytotoxicity, Antioxidant, Antibacterial, and Photocatalytic Activities of ZnO–CdS Powders. Materials 2020, 13, 182. [Google Scholar] [CrossRef]
- Vijayakumar, S.; Vaseeharan, B.; Malaikozhundan, B.; Shobiya, M. Laurus nobilis leaf extract mediated green synthesis of ZnO nanoparticles: Characterization and biomedical applications. Biomed. Pharmacother. 2016, 84, 1213–1222. [Google Scholar] [CrossRef]
- Lomelí-Rosales, D.A.; Zamudio-Ojeda, A.; Reyes-Maldonado, O.K.; López-Reyes, M.E.; Basulto-Padilla, G.C.; Lopez-Naranjo, E.J.; Zuñiga-Mayo, V.M.; Velázquez-Juárez, G. Green Synthesis of Gold and Silver Nanoparticles Using Leaf Extract of Capsicum chinense Plant. Molecules 2022, 27, 1692. [Google Scholar] [CrossRef] [PubMed]
- Barbinta-Patrascu, M.E.; Chilom, C.; Nichita, C.; Zgura, I.; Iftimie, S.; Antohe, S. Biophysical insights on Jack bean urease in the presence of silver chloride phytonanoparticles generated from Mentha piperita L. leaves. Rom. Rep. Phys. 2022, 74, 605. [Google Scholar]
- Fageria, P.; Gangopadhyay, S.; Pande, S. Synthesis of ZnO/Au and ZnO/Ag nanoparticles and their photocatalytic application using UV and visible light. RSC Adv. 2014, 4, 24962–24972. [Google Scholar] [CrossRef]
- Barbinta-Patrascu, M.E.; Ungureanu, C.; Badea, N.; Iordache, S.M. Optical studies on human hair fibres treated with a natural extract of red tulip flowers. Optoelectron. Adv. Mater. 2022, 16, 458–463. [Google Scholar]
- Coates, J. Interpretation of Infrared Spectra, a Practical Approach. In Encyclopedia of Analytical Chemistry; Meyers, R.A., Ed.; John Wiley & Sons Ltd.: Chichester, UK, 2000; pp. 1–23. [Google Scholar]
- Jankovic, S.; Milisavic, D.; Okolic, T.; Jelic, D. Synthesis of ZnO-Ag nanoparticles by sol-gel method. Contemp. Mater. 2019, 10, 22–27. [Google Scholar] [CrossRef]
- Padalia, H.; Moteriya, P.; Chanda, S. Silver nanoparticles of marigold flower and its synergistic antimicrobial potential. Arab. J. Chem. 2015, 8, 732–741. [Google Scholar] [CrossRef]
- Kora, A.J.; Sashidhar, R.B. Biogenic silver nanoparticles synthesized with rhamnogalacturonan gum: Antibacterial activity, cytotoxicity and its mode of action. Arab. J. Chem. 2018, 11, 313–323. [Google Scholar] [CrossRef]
- Donga, S.; Bhadu, G.R.; Chanda, S. Antimicrobial, antioxidant and anticancer activities of gold nanoparticles green synthesized using Mangifera indica seed aqueous extract. Artif. Cells Nanomed. Biotechnol. 2020, 48, 1315–1325. [Google Scholar] [CrossRef] [PubMed]
- Hameed, S.; Khalil, A.T.; Ali, M.; Numan, M.; Khamlich, S.; Shinwari, Z.K.; Maaza, M. Greener synthesis of ZnO and Ag–ZnO nanoparticles using Silybum marianum for diverse biomedical applications. Nanomedicine 2019, 14, 655–673. [Google Scholar] [CrossRef]
- Selvarajan, E.; Mohanasrinivasan, V. Biosynthesis and characterization of ZnO nanoparticles using Lactobacillus plantarum VITES07. Mater. Lett. 2013, 112, 180–182. [Google Scholar] [CrossRef]
- Taghizadeh, M.; Siyahi, V.; Ashassi-Sorkhabi, H.; Zarrini, G. ZnO, AgCl and AgCl/ZnO nanocomposites incorporated chitosan in the form of hydrogel beads for photocatalytic degradation of MB, E. coli and S. aureus. Int. J. Biol. Macromol. 2020, 147, 1018–1028. [Google Scholar] [CrossRef] [PubMed]
- Zgura, I.; Preda, N.; Socol, G.; Ghica, C.; Ghica, D.; Enculescu, M.; Negut, I.; Nedelcu, L.; Frunza, L.; Ganea, C.P.; et al. Wet chemical synthesis of ZnO-CdS composites and their photocatalytic activity. Mater. Res. Bull. 2018, 99, 174–181. [Google Scholar] [CrossRef]
- Ahmad, M.; Rehman, W.; Khan, M.M.; Qureshi, M.T.; Gul, A.; Haq, S.; Ullah, R.; Rab, A.; Menaa, F. Phytogenic fabrication of ZnO and gold decorated ZnO nanoparticles for photocatalytic degradation of Rhodamine B. J. Environ. Chem. Eng. 2021, 9, 104725. [Google Scholar] [CrossRef]
- Tao, S.; Yang, M.; Chen, H.; Zhao, S.; Chen, G. Continuous Synthesis of Ag/AgCl/ZnO Composites Using Flow Chemistry and Photocatalytic Application. Ind. Eng. Chem. Res. 2018, 57, 3263–3273. [Google Scholar] [CrossRef]
- Huang, Y.; Zhu, D.; Zhang, Q.; Zhang, Y.; Cao, J.; Shen, Z.; Ho, W.; Lee, S.C. Synthesis of a Bi2O2CO3/ZnFe2O4 heterojunction with enhanced photocatalytic activity for visible light irradiation-induced NO removal. Appl. Catal. B Environ. 2018, 234, 70–78. [Google Scholar] [CrossRef]
- Koga, H.; Umemura, Y.; Kitaoka, T. In Situ Synthesis of Bimetallic Hybrid Nanocatalysts on a Paper-Structured Matrix for Catalytic Applications. Catalysts 2011, 1, 69–82. [Google Scholar] [CrossRef]
- Dhatwalia, J.; Kumari, A.; Chauhan, A.; Mansi, K.; Thakur, S.; Saini, R.V.; Guleria, I.; Lal, S.; Kumar, A.; Batoo, K.M.; et al. Rubus ellipticus Sm. Fruit Extract Mediated Zinc Oxide Nanoparticles: A Green Approach for Dye Degradation and Biomedical Applications. Materials 2022, 15, 3470. [Google Scholar] [CrossRef]
- Sharmila, G.; Thirumarimurugan, M.; Muthukumaran, C. Green synthesis of ZnO nanoparticles using Tecoma castanifolia leaf extract: Characterization and evaluation of its antioxidant, bactericidal and anticancer activities. Microchem. J. 2019, 145, 578–587. [Google Scholar] [CrossRef]
- Patra, J.K.; Baek, K.H. Novel green synthesis of gold nanoparticles using Citrullus lanatus rind and investigation of proteasome inhibitory activity, antibacterial, and antioxidant potential. Int. J. Nanomed. 2015, 10, 7253–7264. [Google Scholar]
- Mahmud, Z.H.; Islam, M.S.; Imran, K.M.; Hakim, S.A.I.; Worth, M.; Ahmed, A.; Hossan, S.; Haider, M.; Islam, M.R.; Hossain, F.; et al. Occurrence of Escherichia coli and faecal coliforms in drinking water at source and household point-of-use in Rohingya camps, Bangladesh. Gut Pathog. 2019, 11, 52. [Google Scholar] [CrossRef]
- Hardalo, C.; Edberg, S.C. Pseudomonas aeruginosa: Assessment of risk from drinking water. Crit. Rev. Microbiol. 1997, 23, 47–75. [Google Scholar] [CrossRef] [PubMed]
- Rice, S.A.; van den Akker, B.; Pomati, F.; Roser, D. A risk assessment of Pseudomonas aeruginosa in swimming pools: A review. J. Water Health 2012, 10, 181–196. [Google Scholar] [CrossRef]
- LeChevallier, M.W.; Seidler, R.J. Staphylococcus aureus in rural drinking water. Appl. Enoiron. Microbiol. 1980, 39, 739–742. [Google Scholar] [CrossRef] [PubMed]
- Kristanti, R.A.; Hadibarata, T.; Syafrudin, M.; Yilmaz, M.; Abdullah, S. Microbiological Contaminants in Drinking Water: Current Status and Challenges. Water Air Soil Pollut. 2022, 233, 299. [Google Scholar] [CrossRef]
- Al-Mohaimeed, A.M.; Al-Onazi, W.A.; El-Tohamy, M.F. Multifunctional Eco-Friendly Synthesis of ZnO Nanoparticles in Biomedical Applications. Molecules 2022, 27, 579. [Google Scholar] [CrossRef] [PubMed]
- Jing, Y.; Mu, B.; Zhang, M.; Wang, L.; Zhong, H.; Liu, X.; Wang, A. Zinc-loaded palygorskite nanocomposites for catheter coating with excellent antibacterial and anti-biofilm properties. Colloid Surf. A-Physicochem. Eng. Asp. 2020, 600, 124965. [Google Scholar] [CrossRef]
- Jing, S.; Sadishkumar, S.; Prabhu, N. Biomedical applications of wo3, zno and graphene nanoparticles. Int. J. Mech. Eng. 2021, 6, 972–982. [Google Scholar]
- Mehrvarz, A.; Khalil-Allafi, J.; Khosrowshahi, A.K. Biocompatibility and antibacterial behavior of electrochemically deposited Hydroxyapatite/ZnO porous nanocomposite on NiTi biomedical alloy. Ceram. Int. 2022, 48, 16326–16336. [Google Scholar] [CrossRef]
- Lallo da Silva, B.; Abuçafy, M.P.; Berbel Manaia, E.; Oshiro Junior, J.A.; Chiari-Andréo, B.G.; Pietro, R.C.R.; Chiavacci, L.A. Relationship Between Structure and Antimicrobial Activity of Zinc Oxide Nanoparticles: An Overview. Int. J. Nanomed. 2019, 14, 9395–9410. [Google Scholar] [CrossRef]
- Sathiyaraj, S.; Suriyakala, G.; Gandhi, A.D.; Babujanarthanam, R.; Almaary, K.S.; Chen, T.W.; Kaviyarasu, K. Biosynthesis, characterization, and antibacterial activity of gold nanoparticles. J. Infect. Public Health 2021, 14, 1842–1847. [Google Scholar] [CrossRef] [PubMed]
- Kubasheva, Z.; Sprynskyy, M.; Railean-Plugaru, V.; Pomastowski, P.; Ospanova, A.; Buszewski, B. Synthesis and Antibacterial Activity of (AgCl, Ag)NPs/Diatomite Hybrid Composite. Materials 2020, 13, 3409. [Google Scholar] [CrossRef] [PubMed]
- Sabzevar, A.H.; Hashemitabar, G.R.; Rad, M.; Vatandoost, J. Synthesis and Biological Properties of Silver Chloride Nanoparticles Using Cell-free Extracts of Aeromonas hydrophila and Antibacterial Activity against Drug-Resistant Bacteria. Braz. Arch. Biol. Technol. 2021, 64, e21210010. [Google Scholar] [CrossRef]
- Ji, Y.; Yang, X.; Ji, Z.; Zhu, L.; Ma, N.; Chen, D.; Jia, X.; Tang, J.; Cao, Y. DFT-Calculated IR Spectrum Amide I, II, and III Band Contributions of N-Methylacetamide Fine Components. ACS Omega 2020, 5, 8572–8578. [Google Scholar] [CrossRef] [PubMed]
- Barbinta-Patrascu, M.E.; Nichita, C.; Badea, N.; Ungureanu, C.; Bacalum, M.; Zgura, I.; Iosif, L.; Antohe, S. Biophysical aspects of bio-nanosilver generated from Urtica dioica Leaves and Vitis vinifera fruits’ extracts. Rom. Rep. Phys. 2021, 73, 601. [Google Scholar]



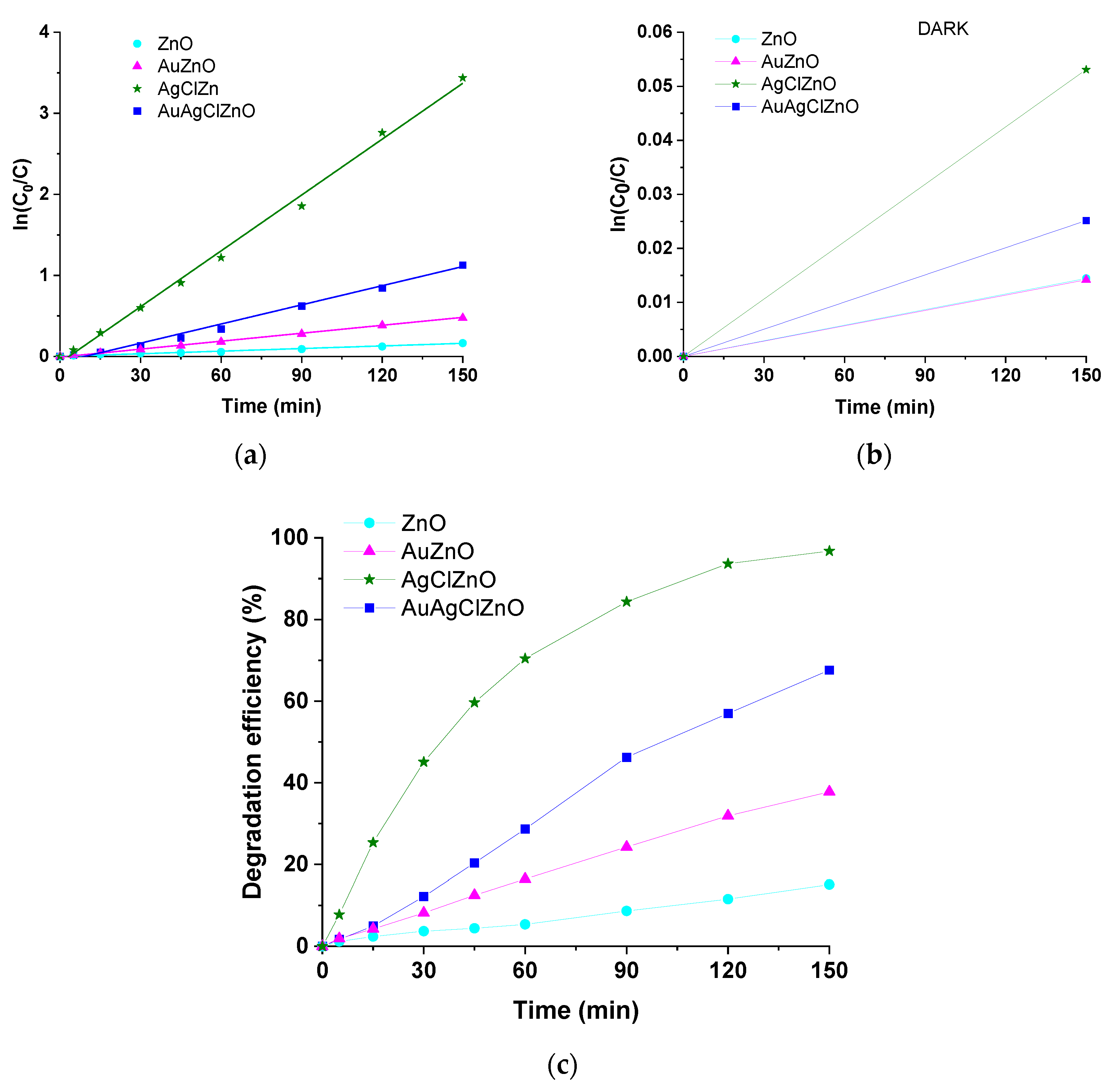
| Sample | k (min−1) | R2 |
|---|---|---|
| ZnO | 1.07 × 10−3 | 0.98984 |
| AuZnO | 3.27 × 10−3 | 0.99813 |
| AgClZnO | 22.97 × 10−3 | 0.99899 |
| AuAgClZnO | 7.88 × 10−3 | 0.99113 |
| Microorganism | Concentration of AuNPs Used (µg/mL) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 400 | 200 | 100 | 50 | 25 | 12.5 | 6.25 | 3.125 | 1.56 | 0.78 | 0.39 | 0.195 | |
| Escherichia coli ATCC 8738 | S | S | R | R | R | R | R | R | R | R | R | R |
| Staphylococcus aureus ATCC BAA 1026 | S | R | R | R | R | R | R | R | R | R | R | R |
| Pseudomonas aeruginosa ATCC 15442 | S | S | R | R | R | R | R | R | R | R | R | R |
| Concentration of AgClNPs used (µg/mL) | ||||||||||||
| Escherichia coli ATCC 8738 | S | S | S | R | R | R | R | R | R | R | R | R |
| Staphylococcus aureus ATCC BAA 1026 | S | S | S | R | R | R | R | R | R | R | R | R |
| Pseudomonas aeruginosa ATCC 15442 | S | S | S | R | R | R | R | R | R | R | R | R |
| Concentration of ZnO particles used (µg/mL) | ||||||||||||
| Escherichia coli ATCC 8738 | S | S | R | R | R | R | R | R | R | R | R | R |
| Staphylococcus aureus ATCC BAA 1026 | S | R | R | R | R | R | R | R | R | R | R | R |
| Pseudomonas aeruginosa ATCC 15442 | S | S | R | R | R | R | R | R | R | R | R | R |
| Concentration of AuZnO used (µg/mL) | ||||||||||||
| Escherichia coli ATCC 8738 | S | S | S | R | R | R | R | R | R | R | R | R |
| Staphylococcus aureus ATCC BAA 1026 | S | S | R | R | R | R | R | R | R | R | R | R |
| Pseudomonas aeruginosa ATCC 15442 | S | S | R | R | R | R | R | R | R | R | R | R |
| Concentration of AgClZnO used (µg/mL) | ||||||||||||
| Escherichia coli ATCC 8738 | S | S | S | R | R | R | R | R | R | R | R | R |
| Staphylococcus aureus ATCC BAA 1026 | S | S | S | S | R | R | R | R | R | R | R | R |
| Pseudomonas aeruginosa ATCC 15442 | S | S | S | R | R | R | R | R | R | R | R | R |
| Concentration of AuAgClZnO used (µg/mL) | ||||||||||||
| Escherichia coli ATCC 8738 | S | S | S | S | S | R | R | R | R | R | R | R |
| Staphylococcus aureus ATCC BAA 1026 | S | S | S | S | R | R | R | R | R | R | R | R |
| Pseudomonas aeruginosa ATCC 15442 | S | S | S | S | S | R | R | R | R | R | R | R |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zgura, I.; Badea, N.; Enculescu, M.; Maraloiu, V.-A.; Ungureanu, C.; Barbinta-Patrascu, M.-E. Burdock-Derived Composites Based on Biogenic Gold, Silver Chloride and Zinc Oxide Particles as Green Multifunctional Platforms for Biomedical Applications and Environmental Protection. Materials 2023, 16, 1153. https://doi.org/10.3390/ma16031153
Zgura I, Badea N, Enculescu M, Maraloiu V-A, Ungureanu C, Barbinta-Patrascu M-E. Burdock-Derived Composites Based on Biogenic Gold, Silver Chloride and Zinc Oxide Particles as Green Multifunctional Platforms for Biomedical Applications and Environmental Protection. Materials. 2023; 16(3):1153. https://doi.org/10.3390/ma16031153
Chicago/Turabian StyleZgura, Irina, Nicoleta Badea, Monica Enculescu, Valentin-Adrian Maraloiu, Camelia Ungureanu, and Marcela-Elisabeta Barbinta-Patrascu. 2023. "Burdock-Derived Composites Based on Biogenic Gold, Silver Chloride and Zinc Oxide Particles as Green Multifunctional Platforms for Biomedical Applications and Environmental Protection" Materials 16, no. 3: 1153. https://doi.org/10.3390/ma16031153
APA StyleZgura, I., Badea, N., Enculescu, M., Maraloiu, V.-A., Ungureanu, C., & Barbinta-Patrascu, M.-E. (2023). Burdock-Derived Composites Based on Biogenic Gold, Silver Chloride and Zinc Oxide Particles as Green Multifunctional Platforms for Biomedical Applications and Environmental Protection. Materials, 16(3), 1153. https://doi.org/10.3390/ma16031153








