2.2. Procedures and Analysis Data
Compounds were synthesized according to the previously presented procedure [
18].
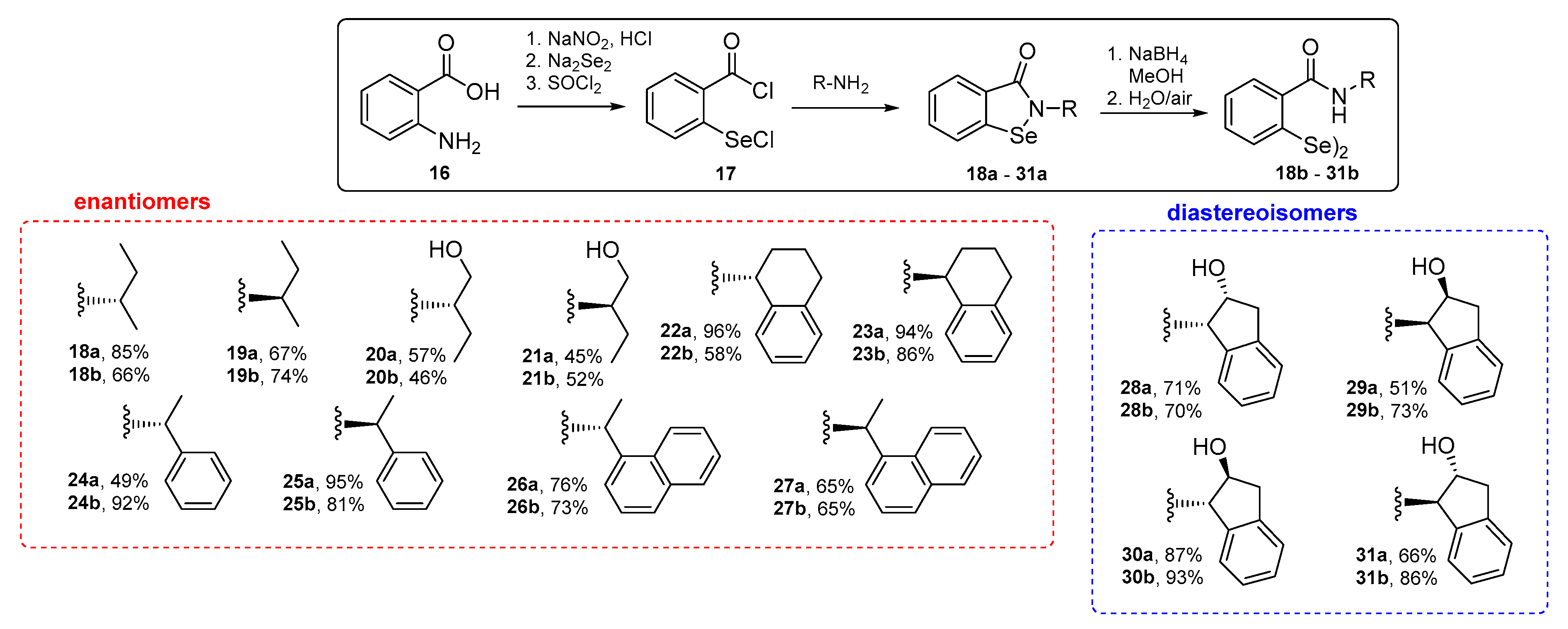
N-[(S)-(+)-sec-butyl]-1,2-benzisoselenazol-3(2H)-one 18a
Yield: 85%; mp 51–52 °C; = +40 (c = 1.12, CHCl3);
1H NMR (700 MHz, DMSO) δ = 0.84 (t, J = 7.0 Hz, 3H), 1.26 (d, J = 7.0 Hz, 3H), 1.58–1.69 (m, 2H), 4.39–4.44 (m, 1H), 7.41–7.44 (m, 1Har), 7.58–7.62 (m, 1Har), 7.79–7.82 (m, 1Har), 8.03–8.06 (m, 1Har) 13C NMR (700 MHz, DMSO) δ = 11.40 (CH3), 21.74 (CH3), 30.57 (CH2), 51.31 (CH), 126.24 (2xCHar), 127.78 (CHar), 129.26 (Car), 131.74 (CHar), 139.22 (Car), 166.67 (C=O) 77Se NMR 700 MHz, DMSO) δ = 804.33 ppm; IR = 2964, 2873, 1738, 1589, 1562, 1443, 1323, 1247, 1217, 1147, 1046, 1020, 788, 737, 697, 591, 509, 416 cm−1. Elemental Anal. Calcd for C11H13NOSe (255.02): C, 51.98; H, 5.15; N, 5.51; Found C, 51.93; H, 5.15; N, 5.49.
N-[(R)-(−)-sec-butyl]-1,2-benzisoselenazol-3(2H)-one 19a
Yield: 67%; mp 50–52 °C; = −38 (c = 1.24, CHCl3);
1H NMR (700 MHz, DMSO) δ = 0.84 (t, J = 7.0 Hz, 3H), 1.26 (d, J = 6.3 Hz, 3H), 1.57–1.69 (m, 2H), 4.39–4.44 (m, 1H), 7.40–7.44 (m, 1Har), 7.58–7.62 (m, 1Har), 7.79–7.82 (m, 1Har), 8.03–8.06 (m, 1Har) 13C NMR (400 MHz, DMSO) δ = 11.42 (CH3), 21.77 (CH3), 30.57 (CH2), 51.26 (CH), 126.22 (CHar), 126.27 (CHar), 127.76 (CHar), 129.28 (Car), 131.71 (CHar), 139.25 (Car), 166.64 (C=O) 77Se NMR (400 MHz, DMSO) δ = 802.56 ppm; IR = 2965, 2873, 1737, 1589, 1563, 1443, 1323, 1246, 1216, 1148, 1047, 1020, 788, 737, 697, 592, 508, 414 cm−1. Elemental Anal. Calcd for C11H13NOSe (255.02): C, 51.98; H, 5.15; N, 5.51; Found C, 51.95; H, 5.16; N, 5.47.
N-[(S)-(+)-1-hydroxy-2-butanyl]-1,2-benzisoselenazol-3(2H)-one 20a
Yield: 45%; mp 108–109 °C; = +47 (c = 0.47, CHCl3);
1H NMR (700 MHz, DMSO) δ = 0.81 (t, J = 7.7 Hz, 3H), 1.53–1.60 (m, 1H), 1.71–1.77 (m, 1H), 3.52–3.56 (m, 1H), 3.64–3.68 (m, 1H), 4.40–4.44 (m, 1H), 5.14 (t, J = 5.6 Hz, 1H), 7.39–7.42 (m, 1Har), 7.53–7.61 (m, 1Har), 7.81 (dd, J1 = 1.4 Hz, J2 = 7.0 Hz, 1Har), 8.01–8.04 (m, 1Har) 13C NMR (700 MHz, DMSO) δ = 11.09 (CH3), 24.98 (CH2), 56.94 (CH), 63.77 (CH2), 125.96 (2xCHar), 127.72 (CHar), 128.58 (Car), 131.63 (CHar), 140.55 (Car), 167.32 (C=O) 77Se NMR (700 MHz, DMSO) δ = 839.29 ppm; IR = 3224, 2959, 2871, 1590, 1562, 1444, 1338, 1310, 1252, 1082, 1020, 791, 739, 676, 601, 549, 483, 422 cm−1. Elemental Anal. Calcd for C11H13NO2Se (271.01): C, 48.90; H, 4.85; N, 5.18; Found C, 48.92; H, 4.85; N, 5.11.
N-[(R)-(−)-1-hydroxy-2-butanyl]-1,2-benzisoselenazol-3(2H)-one 21a
Yield: 57%; mp 109–110 °C; = −41 (c = 0.83, CHCl3);
1H NMR (700 MHz, DMSO) δ = 0.81 (t, J = 7.7 Hz, 3H), 1.53–1.60 (m, 1H), 1.71–1.77 (m, 1H), 3.51–3.57 (m, 1H), 3.64–3.68 (m, 1H), 4.38–4.44 (m, 1H), 5.12 (t, J = 4.9 Hz, 1H), 7.39-7.42 (m, 1Har), 7.57–7.61 (m, 1Har), 7.80–7.83 (m, 1Har), 8.02 (d, J = 7.7 Hz, 1Har) 13C NMR (400 MHz, DMSO) δ = 11.10 (CH3), 24.96 (CH2), 56.87 (CH), 63.73 (CH2), 125.96 (CHar), 125.98 (CHar), 127.70 (CHar), 128.57 (Car), 131.63 (CHar), 140.56 (Car), 167.30 (C=O) 77Se NMR (400 MHz, DMSO) δ = 837.92 ppm; IR = 3233, 2959, 2871, 1591, 1562, 1444, 1338, 1310, 1252, 1081, 1020, 791, 741, 676, 600, 549, 484, 421 cm−1. Elemental Anal. Calcd for C11H13NO2Se (271.01): C, 48.90; H, 4.85; N, 5.18; Found C, 48.93; H, 4.84; N, 5.18.
N-[(R)-(−)-1,2,3,4-tetrahydro-1-napthyl]-1,2-benzisoselenazol-3(2H)-one 22a
Yield: 96%; mp 217–220 °C;
= −50 (c = 0.58, CHCl
3) (lit. mp 212–214 °C;
= −0.53 (c = 0.075, CHCl
3) [
17])
1H NMR (700 MHz, DMSO) δ = 1.81–1.87 (m, 1H), 1.95–2.05 (m, 2H), 2.09–2.15 (m, 1H), 2.76–2.81 (m, 1H), 2.87–2.92 (m, 1H), 5.60 (t, J = 7 Hz, 1H), 7.02–7.06 (m, 1Har), 7.13–7.25 (m, 3Har), 7.43–7.47 (m, 1Har), 7.59–7.62 (m, 1Har), 7.84–7.89 (m, 1Har), 7.96 (d, J = 7.7 Hz, 1H) 13C NMR (400 MHz, DMSO) δ = 20.57 (CH2), 29.16 (CH2), 30.77 (CH2), 51.93 (CH), 126.05 (CHar), 126.32 (CHar), 126.48 (CHar), 127.86 (CHar), 128.13 (CHar), 128.75 (CHar), 129.40 (CHar), 129.50 (CHar), 131.95 (Car), 136.21 (Car), 138.12 (Car), 139.77 (Car), 166.88 (C=O) 77Se NMR (400 MHz, DMSO) δ = 830.37 ppm; IR = 2919, 2857, 1588, 1561, 1491, 1441, 1308, 1268, 1245, 1154, 1082, 769, 733, 677, 572, 534, 511, 481, 418 cm−1. Elemental Anal. Calcd for C17H15NOSe (329.03): C, 62.20; H, 4.61; N, 4.27; Found C, 62.25; H, 4.62; N, 4.29.
N-[(S)-(+)-1,2,3,4-tetrahydro-1-napthyl]-1,2-benzisoselenazol-3(2H)-one 23a
Yield: 94%; mp 218–220 °C; = +54 (c = 0.42, CHCl3);
1H NMR (700 MHz, DMSO) δ = 1.77–1.84 (m, 1H), 1.92–2.02 (m, 2H), 2.06–2.12 (m, 1H), 2.72–2.79 (m, 1H), 2.83–2.89 (m, 1H), 5.57 (t, J = 7 Hz, 1H), 7.00–7.03 (m, 1Har), 7.10–7.22 (m, 3Har), 7.40–7.44 (m, 1Har), 7.55–7.59 (m, 1Har), 7.84–7.86 (m, 1Har), 7.93 (d, J = 7.7 Hz, 1H) 13C NMR (400 MHz, DMSO) δ = 20.58 (CH2), 29.16 (CH2), 30.77 (CH2), 51.92 (CH), 126.06 (CHar), 126.31 (CHar), 126.47 (CHar), 127.85 (CHar), 128.11 (CHar), 128.76 (CHar), 129.40 (CHar), 129.50 (CHar), 131.93 (Car), 136.22 (Car), 138.12 (Car), 139.77 (Car), 166.86 (C=O) 77Se NMR (400 MHz, DMSO) δ = 830.01 ppm; IR = 2921, 2853, 1589, 1561, 1492, 1441, 1308, 1269, 1245, 1154, 1081, 769, 734, 676, 572, 534, 512, 481, 419 cm−1. Elemental Anal. Calcd for C17H15NOSe (329.03): C, 62.20; H, 4.61; N, 4.27; Found C, 62.29; H, 4.62; N, 4.21.
N-[(R)-(+)-α-methylbenzyl]-1,2-benzisoselenazol-3(2H)-one 24a
Yield: 49%; mp 108–110 °C;
= +128 (c = 1.04, CHCl
3) (lit. mp 116–117.5 °C;
= +120 (c = 1.00, C
2H
5OH) [
13])
1H NMR (700 MHz, DMSO) δ = 1.68 (d, J = 7 Hz, 3H), 5.67 (q, J = 7 Hz, 1H), 7.30–7.33 (m, 1Har) 7.36–7.42 (m, 4Har), 7.43–7.45 (m, 1Har), 7.58–7.61 (m, 1Har), 7.83 (dd, J1 = 0.7 Hz, J2 = 7.7 Hz, 1Har), 7.99 (d, J = 7.7 Hz, 1H) 13C NMR (700 MHz, DMSO) δ = 20.86 (CH3), 52.37 (CH), 126.18 (CHar), 126.32 (CHar), 127.40 (2xCHar), 127.83 (CHar), 128.11 (CHar), 128.99 (2xCHar), 129.03 (Car), 131.88 (CHar), 139.58 (Car), 142.61 (Car), 166.34 (C=O) 77Se NMR (700 MHz, DMSO) δ = 819.68 ppm; IR = 2921, 1590, 1560, 1492, 1441, 1308, 1249, 1154, 1115, 1059, 759, 738, 695, 610, 562, 538, 504, 480, 420 cm−1. Elemental Anal. Calcd for C15H13NOSe (303.02): C, 59.61; H, 4.34; N, 4.63; Found C, 59.68; H, 4.35; N, 4.60.
N-[(S)-(−)-α-methylbenzyl]-1,2-benzisoselenazol-3(2H)-one 25a
Yield: 95%; mp 109–111 °C;
= −123 (c = 1.02, CHCl
3) (lit. mp 116–117.5 °C;
= −120 (c = 1.00, C
2H
5OH) [
13])
1H NMR (700 MHz, DMSO) δ = 1.68 (d, J = 7 Hz, 3H), 5.67 (q, J = 7Hz, 1H), 7.30–7.33 (m, 1Har) 7.37–7.42 (m, 4Har), 7.42–7.45 (m, 1Har), 7.58–7.61 (m, 1Har), 7.83 (dd, J1 = 1.4 Hz, J2 = 7.7 Hz, 1Har), 8.00 (d, J = 7.7 Hz, 1H) 13C NMR (700 MHz, DMSO) δ = 20.86 (CH3), 52.41 (CH), 126.18 (CHar), 126.32 (CHar), 127.40 (2xCHar), 127.83 (CHar), 128.11 (CHar), 128.99 (2xCHar), 129.02 (Car), 131.89 (CHar), 139.59 (Car), 142.60 (Car), 166.36 (C=O) 77Se NMR (700 MHz, DMSO) δ = 820.47 ppm; IR = 2924, 1590, 1561, 1492, 1441, 1307, 1245, 1158 1059, 759, 738, 695, 610, 560, 538, 504, 448, 416 cm−1. Elemental Anal. Calcd for C15H13NOSe (303.02): C, 59.61; H, 4.34; N, 4.63; Found C, 59.63; H, 4.34; N, 4.58.
N-[(S)-(−)-1-(1-napthyl)ethyl]-1,2-benzisoselenazol-3(2H)-one 26a
Yield: 76%; mp 136–138 °C; = −241 (c = 0.72, CHCl3);
1H NMR (700 MHz, DMSO) δ = 1.78 (d, J = 7.0 Hz, 3H), 6.37 (q, J = 6.3 Hz, 1H), 7.40–7.44 (m, 1Har), 7.50–7.56 (m, 3Har), 7.60–7.63 (m, 1Har), 7.82 (d, J = 7 Hz, 1Har), 7.84–7.89 (m, 2Har), 7.94–8.02 (m, 3Har) 13C NMR (700 MHz, DMSO) δ = 19,72 (CH3), 48,32 (CH), 123.37 (CHar), 123.97 (CHar), 125.67 (CHar), 126.13 (CHar), 126.30 (CHar), 126.44 (CHar), 127.29 (CHar), 127.90 (CHar), 129.00 (Car), 129.21 (CHar), 129.48 (CHar), 131.63 (Car), 131.82 (CHar), 133.99 (Car), 137.91 (Car), 139.73 (Car), 165.81 (C=O) 77Se NMR (700 MHz, DMSO) δ = 816.43 ppm; IR = 2924, 2853, 1590, 1562, 1442, 1341, 1309, 1245, 1079, 1020, 798, 736, 674, 605, 523, 505, 453 cm−1. Elemental Anal. Calcd for C19H15NOSe (353.03): C, 64.78; H, 4.29; N, 3.98; Found C, 64.82; H, 4.29; N, 3.90.
N-[(R)-(+)-1-(1-napthyl)ethyl]-1,2-benzisoselenazol-3(2H)-one 27a
Yield: 65%; mp 135–137 °C;
= +245 (c = 0.62, CHCl
3) (lit. mp 130–132 °C;
= +2.62 (c = 0.28, CHCl
3) [
17])
1H NMR (700 MHz, DMSO) δ = 1.78 (d, J = 7.7 Hz, 3H), 6.37 (q, J = 6.3 Hz, 1H), 7.40–7.44 (m, 1Har), 7.50–7.56 (m, 3Har), 7.60–7.63 (m, 1Har), 7.82 (d, J = 7 Hz, 1Har), 7.84–7.89 (m, 2Har), 7.94–8.02 (m, 3Har) 13C NMR (700 MHz, DMSO) δ = 19.74 (CH3), 48.29 (CH), 123.38 (CHar), 123.96 (CHar), 125.68 (CHar), 126.14 (CHar), 126.30 (CHar), 126.44 (CHar), 127.28 (CHar), 127.88 (CHar), 129.02 (Car), 129.21 (CHar), 129.46 (CHar), 131.63 (Car), 131.80 (CHar), 133.99 (Car), 137.94 (Car), 139.73 (Car), 165.77 (C=O) 77Se NMR (700 MHz, DMSO) δ = 815.51 ppm; IR = 2923, 2853, 1590, 1561, 1456, 1440, 1343, 1309, 1268, 1079, 1020, 797, 739, 675, 606, 522, 504, 453 cm−1. Elemental Anal. Calcd for C19H15NOSe (353.03): C, 64.78; H, 4.29; N, 3.98; Found C, 64.77; H, 4.30; N, 3.89.
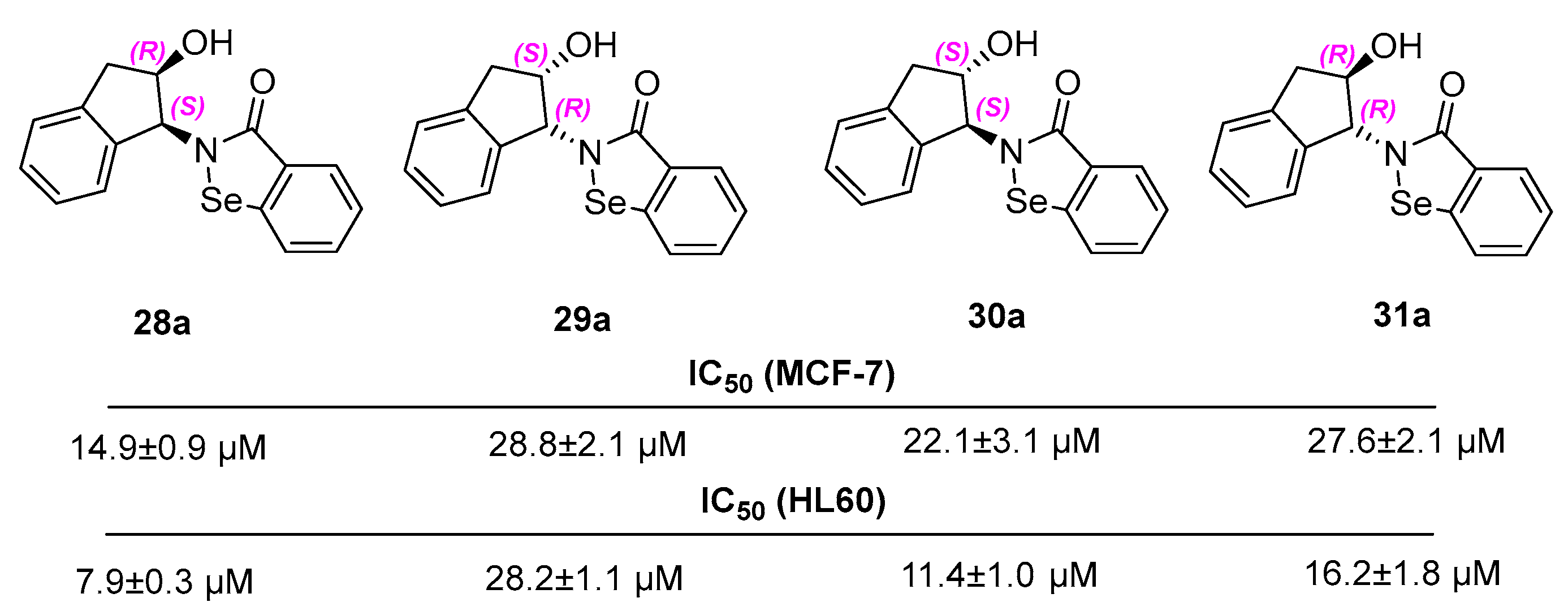
N-[(1S,2R)-(−)-cis-2-hydroxy-1-indanyl]-1,2-benzisoselenazol-3(2H)-one 28a
Yield: 71%; mp 187–189 °C; = −97 (c = 0.61, CHCl3);
1H NMR (700 MHz, DMSO) δ = 2.86–2.89 (m, 1H), 3.14–3.18 (m, 1H), 4.57–4.61 (m, 1H), 5.56 (d, J = 4.9 Hz, 1H), 5.95 (d, J = 4.9 Hz, 1H), 6.90 (d, J = 7.7 Hz, 1Har), 7.14 (t, J = 7.0 Hz, 1Har), 7.23 (t, J = 7.7 Hz, 1Har), 7.28–7.31 (m, 1Har), 7.38–7.41 (m, 1Har), 7.55–7.59 (m, 1Har), 7.87–7.90 (m, 1Har), 7.95 (d, J = 7.7 Hz, 1H) 13C NMR (400 MHz, DMSO) δ = 39.52 (CH2), 61.36 (CH), 73.06 (CH), 124.73 (CHar), 125.68 (CHar), 125.71 (CHar), 125.78 (CHar), 126.91 (CHar), 127.76 (CHar), 127.86 (CHar), 128.37 (CHar), 131.83 (CHar), 141.06 (Car), 141.46 (Car), 142.28 (Car), 168.25 (C=O) 77Se NMR (400 MHz, DMSO) δ = 877.05 ppm; IR = 3259, 2922, 2853, 1621, 1533, 1458, 1446, 1349, 1311, 1259, 1047, 1026, 733, 680, 517, 475, 415 cm−1. Elemental Anal. Calcd for C16H13NO2Se (331.01): C, 58.19; H, 3.97; N, 4.24; Found C, 58.11; H, 3.96; N, 4.29.
N-[(1R,2S)-(+)-cis-2-hydroxy-1-indanyl]-1,2-benzisoselenazol-3(2H)-one 29a
Yield: 51%; mp 187–189 °C; = +100 (c = 0.59, CHCl3);
1H NMR (700 MHz, DMSO) δ = 2.89–2.92 (m, 1H), 3.17–3.21 (m, 1H), 4.60–4.64 (m, 1H), 5.59 (d, J = 4.2 Hz, 1H), 5.98 (d, J = 4.9 Hz, 1H), 6.93 (d, J = 7.7 Hz, 1Har), 7.16 (t, J = 4.9 Hz, 1Har), 7.26 (t, J = 7.7 Hz, 1Har), 7.31–7.34 (m, 1Har), 7.41–7.44 (m, 1Har), 7.58–7.62 (m, 1Har), 7.90–7.93 (m, 1Har), 7.98 (d, J = 7.7 Hz, 1H) 13C NMR (400 MHz, DMSO) δ = 39.56 (CH2), 61.37 (CH), 74.07 (CH), 124.74 (CHar), 125.68 (CHar), 125.71 (CHar), 125.78 (CHar), 126.90 (CHar), 127.76 (CHar), 127.86 (CHar), 128.37 (CHar), 131.82 (CHar), 141.06 (Car), 141.47 (Car), 142.26 (Car), 168.24 (C=O) 77Se NMR (400 MHz, DMSO) δ = 877.00 ppm; IR = 3234, 2950, 2853, 1602, 1566, 1457, 1445, 1337, 1312, 1271, 1061, 1019, 756, 738, 677, 517, 476, 417 cm−1. Elemental Anal. Calcd for C16H13NO2Se (331.01): C, 58.19; H, 3.97; N, 4.24; Found C, 58.13; H, 3.98; N, 4.25.
N-[(1S,2S)-(+)-trans-2-hydroxy-1-indanyl]-1,2-benzisoselenazol-3(2H)-one 30a
Yield: 87%; mp 204–206 °C; = +212 (c = 0.55, CHCl3);
1H NMR (700 MHz, DMSO) δ = 2.80–2.84 (m, 1H), 3.24–3.28 (m, 1H), 4.43 (kw, J = 5.6 Hz, 1H), 5.57 (d, J = 4.9 Hz, 1H), 5.70 (d, J = 5.6 Hz, 1H), 7.10 (d, J = 7.7 Hz, 1Har), 7.20–7.24 (m, 1Har), 7.31 (d, J = 4.1 Hz, 2Har), 7.44–7.48 (m, 1Har), 7.60–7.63 (m, 1Har), 7.87–7.90 (m, 1Har), 7.98 (d, J = 7.7 Hz, 1H) 13C NMR (300 MHz, DMSO) δ = 39.01 (CH2), 65.63 (CH), 78.88 (CH), 125.18 (CHar), 125.46 (CHar), 126.14 (CHar), 126.31 (CHar), 127.32 (CHar), 127.80 (CHar), 128.88 (CHar), 129.01 (Car), 131.95 (CHar), 139.56 (Car), 140.85 (Car), 140.95 (Car), 167.38 (C=O) 77Se NMR (400 MHz, DMSO) δ = 824.06 ppm; IR = 3091, 2923, 2850, 1590, 1562, 1460, 1445, 1329, 1312, 1268, 1072, 1021, 793, 743, 733, 676, 516, 458 cm−1. Elemental Anal. Calcd for C16H13NO2Se (331.01): C, 58.09; H, 3.98; N, 4.26; Found C, 58.15; H, 3.97; N, 4.22.
N-[(1R,2R)-(−)-trans-2-hydroxy-1-indanyl]-1,2-benzisoselenazol-3(2H)-one 31a
Yield: 66%; mp 205–207 °C; = −214 (c = 0.57, CHCl3);
1H NMR (700 MHz, DMSO) δ = 2.80–2.85 (m, 1H), 3.24–3.28 (m, 1H), 4.43 (kw, J = 5.6 Hz, 1H), 5.56 (d, J = 4.9 Hz, 1H), 5.69 (d, J = 5.6 Hz, 1H), 7.10 (d, J = 9.1 Hz, 1Har), 7.20–7.24 (m, 1Har), 7.31 (d, J = 7.7 Hz, 2Har), 7.44–7.47 (m, 1Har), 7.60–7.63 (m, 1Har), 7.88–7.90 (m, 1Har), 7.98 (d, J = 7.7 Hz, 1H) 13C NMR (400 MHz, DMSO) δ = 39.33 (CH2), 65.71 (CH), 78.93 (CH), 125.24 (CHar), 125.51 (CHar), 126.20 (CHar), 126.35 (CHar), 127.36 (CHar), 127.85 (CHar), 128.95 (CHar), 129.04 (Car), 131.98 (CHar), 139.62 (Car), 140.94 (Car), 141.03 (Car), 167.42 (C=O) 77Se NMR (400 MHz, DMSO) δ = 823.38 ppm; IR = 3090, 2923, 2852, 1590, 1562, 1460, 1445, 1329, 1312, 1269, 1071, 1021, 793, 744, 733, 676, 516, 457 cm−1. Elemental Anal. Calcd for C16H13NO2Se (331.01): C, 58.19; H, 3.97; N, 4.24; Found C, 58.15; H, 3.97; N, 4.22.
2,2′-diselenobis[N-(S)-(+)-sec-butylbezamide] 18b
Yield: 66%; mp 233–235 °C; = +43 (c = 0.24, CHCl3);
1H NMR (700 MHz, DMSO) δ = 0.92 (t, J = 7.7 Hz, 3H), 1.18 (d, J = 7.0 Hz, 3H), 1.50–1.60 (m, 2H), 3.92–3.97 (m, 1H), 7.31–7.34 (m, 1Har), 7.36–7.39 (m, 1Har), 7.69 (d, J = 7.7 Hz, 1Har), 7.79 (d, J = 7.7 Hz, 1Har), 8.44 (d, J = 7.7 Hz, 1Har) 13C NMR (700 MHz, DMSO) δ = 11.15 (CH3), 20.59 (CH3), 29.26 (CH2), 47.33 (CH), 126.55 (CHar), 128.30 (CHar), 130.41 (CHar), 131.79 (CHar), 132.31 (Car), 134.35 (Car), 167.36 (C=O) 77Se NMR (700 MHz, DMSO) δ = 443.25′ ppm; IR: 3312, 2968,, 2927, 2870, 1738, 1612, 1583, 1535, 1448, 1435, 1366, 1353, 1302, 1285, 1229, 1216, 1163, 1146, 1027, 871, 743, 677, 645, 541, 472, 446 cm−1. Elemental Anal. Calcd for C22H28N2O2Se2 (512.08): C, 51.77; H, 5.53; N, 5.49; Found C, 51.72; H, 5.53; N, 5.52.
2,2′-diselenobis[N-(R)-(−)-sec-butylbezamide] 19b
Yield: 74%; mp 234–235 °C; = −41 (c = 0.30, CHCl3);
1H NMR (700 MHz, DMSO) δ = 0.91 (t, J = 7.7 Hz, 3H), 1.18 (d, J = 7.0 Hz, 3H), 1.50–1.60 (m, 2H), 3.92–3.97 (m, 1H), 7.31–7.34 (m, 1Har), 7.36–7.39 (m, 1Har), 7.69 (d, J = 8.4 Hz, 1Har), 7.79 (d, J = 7.7 Hz, 1Har), 8.44 (d, J = 7.7 Hz, 1Har) 13C NMR (700 MHz, DMSO) δ = 11.15 (CH3), 20.60 (CH3), 29.27 (CH2), 47.32 (CH), 126.54 (CHar), 128.30 (CHar), 130.41 (CHar), 131.78 (CHar), 132.32 (Car), 134.36 (Car), 167.35 (C=O) 77Se NMR (700 MHz, DMSO) δ = 443.17 ppm; IR: 3311, 2968, 2927, 2870, 1738, 1613, 1583, 1535, 1448, 1435, 1366, 1353, 1302, 1284, 1229, 1216, 1164, 1145, 1027, 872, 743, 678, 645, 528, 472, 446 cm−1. Elemental Anal. Calcd for C22H28N2O2Se2 (512.08): C, 51.77; H, 5.53; N, 5.49; Found C, 51.70; H, 5.53; N, 5.45.
2,2′-diselenobis[N-(S)-(+)-1-hydroxy-2-butanylbezamide] 20b
Yield: 52%; mp 200–202 °C; = +65 (c = 0.31, CHCl3);
1H NMR (700 MHz, DMSO) δ = 0.92 (t, J = 7.7 Hz, 3H), 1.44–1.52 (m, 1H), 1.66–1.73 (m, 1H), 3.41–3.46 (m, 1H), 3.49–3.53 (m, 1H), 3.86–3.93 (m, 1H), 4.73 (t, J = 6.3 Hz, 1H), 7.31–7.35 (m, 1Har), 7.36–7.40 (m, 1Har), 7.70 (dd, J1 = 1.4 Hz, J2 = 8.4 Hz, 1Har), 7.84 (dd, J1 = 1.4 Hz, J2 = 8.4 Hz, 1Har), 8.31 (d, J = 8.4 Hz, 1Har) 13C NMR (700 MHz, DMSO) δ = 11.05 (CH3), 24.12 (CH2), 53.94 (CH), 63.51 (CH2), 126.50 (CHar), 128.43 (CHar), 130.40 (CHar), 131.82 (CHar), 132.38 (Car), 134.27 (Car), 167.91 (C=O) 77Se NMR (700 MHz, DMSO) δ = 444.00 ppm; IR: 3278, 3045, 2969, 2870, 1738, 1622, 1583, 1530, 1453, 1366, 1306, 1283, 1216, 1179, 1165, 1076, 1060, 1048, 1025, 873, 727, 692, 644, 557, 447 cm−1. Elemental Anal. Calcd for C22H28N2O4Se2 (544.04): C, 48.72; H, 5.20; N, 5.16; Found C, 48.65; H, 5.21; N, 5.11.
2,2′-diselenobis[N-(R)-(−)-1-hydroxy-2-butanylbezamide] 21b
Yield: 46%; mp 199–201 °C; = −67 (c = 0.41, CHCl3);
1H NMR (700 MHz, DMSO) δ = 0.89 (t, J = 7.0 Hz, 3H), 1.41–1.49 (m, 1H), 1.63–1.70 (m, 1H), 3.38–3.43 (m, 1H), 3.46–3.50 (m, 1H), 3.83–3.89 (m, 1H), 4.69 (t, J = 5.6 Hz, 1H), 7.27–7.31 (m, 1Har), 7.33–7.37 (m, 1Har), 7.67 (dd, J1 = 0.7 Hz, J2 = 8.4 Hz, 1Har), 7.81 (dd, J1 = 1.4 Hz, J2 = 7.7 Hz, 1Har), 8.27 (d, J = 8.4 Hz, 1Har) 13C NMR (700 MHz, DMSO) δ = 11.05 (CH3), 24.12 (CH2), 53.95 (CH), 63.51 (CH2), 126.50 (CHar), 128.43 (CHar), 130.42 (CHar), 131.82 (CHar), 132.36 (Car), 134.27 (Car), 167.92 (C=O) 77Se NMR (700 MHz, DMSO) δ= 443.96 ppm; IR: 3276, 3047, 2969, 2870, 1738, 1622, 1584, 1530, 1453, 1365, 1306, 1283, 1216, 1179, 1165, 1076, 1060, 1047, 1025, 873, 727, 691, 644, 557, 448 cm−1. Elemental Anal. Calcd for C22H28N2O4Se2 (544.04): C, 48.72; H, 5.20; N, 5.16; Found C, 48.69; H, 5.20; N, 5.09.
2,2′-diselenobis[N-(R)-(−)-1,2,3,4-tetrahydro-1-napthylbezamide] 22b
Yield: 58%; mp 266–268 °C; = −167 (c = 0.21, CHCl3);
1H NMR (700 MHz, DMSO) δ = 1.69–1.81 (m, 1H), 1.81–1.88 (m, 1H), 1.94–2.05 (m, 2H), 2.72–2.82 (m, 2H), 5.25 (q, J = 7Hz, 1H), 7.09–7.19 (m, 3Har), 7.23–7.31 (m, 2Har), 7.34–7.39 (m, 1Har), 7.74 (dd, J1 = 1.4 Hz, J2 = 7.7 Hz, 1Har), 7.81 (dd, J1 = 1.4 Hz, J2 = 7.7 Hz, 1Har), 9.05 (d, J = 9.1 Hz, 1H) 13C NMR (700 MHz, DMSO) δ = 20.84 (CH2), 29.37 (CH2), 30.30 (CH2), 48.02 (CH), 126.35 (CHar), 126.60 (CHar), 127.24 (CHar), 128.35 (CHar), 128.56 (CHar), 129.27 (CHar), 130.52 (CHar), 131.99 (Car), 132.62 (CHar), 134.02 (Car), 137.73 (Car), 137.76 (Car), 167.61 (C=O) 77Se NMR (700 MHz, DMSO) δ= 445.03 ppm; IR: 3279, 2940, 2923, 1627, 1583, 1530, 1486, 1430, 1335, 1256, 1080, 1024, 878, 865, 758, 682, 593, 447 cm−1. Elemental Anal. Calcd for C34H32N2O2Se2 (660.08): C, 62.01; H, 4.90; N, 4.25; Found C, 61.95; H, 4.91; N, 4.17.
2,2′-diselenobis[N-(S)-(+)-1,2,3,4-tetrahydro-1-napthylbezamide] 23b
Yield: 86%; mp 266–268 °C; = +166 (c = 0.25, CHCl3);
1H NMR (700 MHz, DMSO) δ = 1.72–1.84 (m, 1H), 1.85–1.91 (m, 1H), 1.97–2.08 (m, 2H), 2.74–2.85 (m, 2H), 5.28 (q, J = 8.4 Hz, 1H), 7.12–7.21 (m, 3Har), 7.26–7.34 (m, 2Har), 7.38–7.42 (m, 1Har), 7.77 (dd, J1 = 1.4 Hz, J2 = 7.7 Hz, 1Har), 7.85 (dd, J1 = 1.4 Hz, J2 = 7.7 Hz, 1Har), 9.08 (d, J = 9.1 Hz, 1H) 13C NMR (700 MHz, DMSO) δ = 20.84 (CH2), 29.37 (CH2), 30.30 (CH2), 48.02 (CH), 126.36 (CHar), 126.60 (CHar), 127.24 (CHar), 128.35 (CHar), 128.56 (CHar), 129.28 (CHar), 130.52 (CHar), 131.99 (Car), 132.62 (CHar), 134.01 (Car), 137.72 (Car), 137.76 (Car), 167.62 (C=O) 77Se NMR (700 MHz, DMSO) δ = 445.22 ppm; IR: 3275, 2939, 2922, 1627, 1583, 1530, 1486, 1430, 1334, 1255, 1079, 1024, 878, 864, 758, 681, 593, 427 cm−1. Elemental Anal. Calcd for C34H32N2O2Se2 (660.08): C, 62.01; H, 4.90; N, 4.25; Found C, 62.00; H, 4.89; N, 4.21.
2,2′-diselenobis[N-(R)-(+)-α-methylbenzylbezamide] 24b
Yield: 92%; mp 200–202 °C;
= +101 (c = 0.47, CHCl
3) (lit. mp 219–220°C;
= +172 (c = 1.00, dioxane) [
13])
1H NMR (700 MHz, DMSO) δ = 1.52 (d, J = 7 Hz, 3H), 5.17–5.22 (m, 1H), 7.23–7.27 (m, 1Har), 7.33–7.39 (m, 4Har), 7.43–7.45 (m, 2Har), 7.67 (dd, J1 = 1.4 Hz, J2 = 7.7 Hz, 1Har), 7.92 (dd, J1 = 1.4 Hz, J2 = 7.7 Hz, 1Har), 9.09 (d, J = 7.7 Hz, 1H) 13C NMR (700 MHz, DMSO) δ = 22.61 (CH3), 49.30 (CH), 126.57 (2xCHar), 126.58 (CHar), 127.19 (CHar), 128.60 (CHar), 128.75 (2xCHar), 130.46 (CHar), 132.03 (CHar), 132.53 (Car), 133.78 (Car), 144.95 (Car), 167.15 (C=O) 77Se NMR (700 MHz, DMSO) δ = 444.99 ppm; IR: 3312, 3061, 3033, 2976, 2930, 1625, 1583, 1524, 1451, 1431, 1376, 1325, 1207, 1133, 1091, 873, 709, 696, 674, 644, 472, 443 cm-1. Elemental Anal. Calcd for C30H28N2O2Se2 (608.05): C, 59.41; H, 4.65; N, 4.62; Found C, 59.31; H, 4.64; N, 4.55.
2,2′-diselenobis[N-(S)-(−)-α-methylbenzylbezamide] 25b
Yield: 81%; mp 200–202 °C;
= −104 (c = 0.63, CHCl
3) (lit. mp 219–220 °C;
= −172 (c = 1.00, dioxane) [
13])
1H NMR (700 MHz, DMSO) δ = 1.52 (d, J = 7 Hz, 3H), 5.17–5.23 (m, 1H), 7.23–7.27 (m, 1Har) 7.32–7.40 (m, 4Har), 7.43–7.46 (m, 2Har), 7.66–7.70 (m, 1Har), 7.92 (dd, J1 = 0.7 Hz, J2 = 7.7 Hz, 1Har), 9.09 (d, J = 8.4 Hz, 1H) 13C NMR (700 MHz, DMSO) δ = 22.62 (CH3), 49.30 (CH), 126.57 (2xCHar), 126.58 (CHar), 127.19 (CHar), 128.61 (CHar), 128.75 (2xCHar), 130.45 (CHar), 132.03 (CHar), 132.52 (Car), 133.76 (Car), 144.96 (Car), 167.15 (C=O) 77Se NMR (700 MHz, DMSO) δ= 445.01 ppm; IR: 3310, 3059, 3032, 2975, 1623, 1583, 1522, 1448, 1431, 1376, 1324, 1207, 1132, 1090, 872, 696, 671, 644, 471, 443 cm−1. Elemental Anal. Calcd for C30H28N2O2Se2 (608.05): C, 59.41; H, 4.65; N, 4.62; Found C, 59.32; H, 4.65; N, 4.59.
2,2′-diselenobis[N-(S)-(−)-1-(1-napthyl)ethylbezamide] 26b
Yield: 70%; mp 262–64 °C; = −96 (c = 0.35, CHCl3);
1H NMR (700 MHz, DMSO) δ = 1.66 (d, J = 7.7 Hz, 3H), 5.95–6.01 (m, 1H), 7.31–7.36 (m, 2Har), 7.49–7.56 (m, 2Har), 7.58–7.62 (m, 1Har), 7.65–7.69 (m, 2Har), 7.86 (d, J = 8.4 Hz, 1Har), 7.90–7.94 (m, 1Har), 7.95–7.99 (m, 1Har), 8.26 (d, J = 8.4 Hz, 1Har), 9.26 (d, J = 7.7 Hz, 1H) 13C NMR (700 MHz, DMSO) δ = 21.87 (CH3), 45.64 (CH), 123.11 (CHar), 123.63 (CHar), 125.95 (CHar), 126.09 (CHar), 126.60 (CHar), 126.73 (CHar), 127.85 (CHar), 128.67 (CHar), 129.17 (CHar), 130.40 (CHar), 130.90 (CHar), 132.03 (Car), 132.45 (Car), 133.76 (Car), 133.90 (Car), 140.44 (Car), 167.10 (C=O) 77Se NMR (700 MHz, DMSO) δ = 442.72 ppm; IR: 3290, 2922, 1625, 1583, 1530, 1452, 1429, 1338, 1308, 1284, 1258, 1180, 1132, 1025, 871, 792, 773, 735, 693, 641, 448 cm−1.Elemental Anal. Calcd for C38H32N2O2Se2 (708.08): C, 64.59; H, 4.56; N, 3.96; Found C, 64.46; H, 4.55; N, 3.89.
2,2′-diselenobis[N-(R)-(+)-1-(1-napthyl)ethylbezamide] 27b
Yield: 73%; mp 262–264 °C; = +98 (c = 0.32, CHCl3);
1H NMR (700 MHz, DMSO) δ = 1.66 (d, J = 7.7 Hz, 3H), 5.55–6.01 (m, 1H), 7.31–7.36 (m, 2Har), 7.49–7.56 (m, 2Har), 7.58–7.61 (m, 1Har), 7.66–7.69 (m, 2Har), 7.86 (d, J = 8.4 Hz, 1Har), 7.90–7.93 (m, 1Har), 7.96–7.98 (m, 1Har), 8.26 (d, J = 8.4 Hz, 1Har), 9.26 (d, J = 7.7 Hz, 1H) 13C NMR (700 MHz, DMSO) δ = 21.87 (CH3), 45.64 (CH), 123.12 (CHar), 123.64 (CHar), 125.96 (CHar), 126.09 (CHar), 126.59 (CHar), 126.71 (CHar), 127.85 (CHar), 128.67 (CHar), 129.17 (CHar), 130.41 (CHar), 130.91 (CHar), 132.03 (Car), 132.64 (Car), 133.79 (Car), 133.91 (Car), 140.45 (Car), 167.10 (C=O) 77Se NMR (700 MHz, DMSO) δ = 443.08 ppm; IR: 3292, 3063, 2976, 2927, 1624, 1583, 1531, 1450, 1432, 1337, 1306, 1283, 1258, 1180, 1131, 1083, 1026, 997, 878, 793, 773, 736, 725, 692, 641, 447 cm−1. Elemental Anal. Calcd for C38H32N2O2Se2 (708.08): C, 64.59; H, 4.56; N, 3.96; Found C, 64.52; H, 4.57; N, 3.90.
2,2′-diselenobis[N-(1S,2R)-(−)-cis-2-hydroxy-1-indanylbezamide] 28b
Yield: 70%; mp 241–243 °C; = −149 (c = 0.39, CHCl3);
1H NMR (700 MHz, DMSO) δ = 2.90–2.95 (m, 1H), 3.14 (dd, J1 = 4.9 Hz, J2 = 9.1 Hz, 1H), 4.56–4.61 (m, 1H), 5.17–5.21 (m, 1H), 5.49 (dd, J1 = 5.6 Hz, J2 = 8.4 Hz, 1H), 7.21–7.27 (m, 2Har), 7.28–7.35 (m, 3Har), 7.40–7.44 (m, 1Har), 7.78 (d, J = 8.4 Hz, 1Har), 8.01 (d, J = 7.7 Hz, 1Har), 8.59 (d, J = 8.4 Hz, 1Har) 13C NMR (700 MHz, DMSO) δ= 39.92 (CH2), 58.10 (CH), 72.54 (CH), 124.94 (CHar), 125.35 (CHar), 126.63 (CHar), 126.86 (CHar), 128.00 (CHar), 129.00 (CHar), 130.42 (CHar), 132.16 (CHar), 132.66 (Car), 133.62 (Car), 141.44 (Car), 142.02 (Car), 168.21 (C=O) 77Se NMR (700 MHz, DMSO) δ = 445.85 ppm; IR: 3288, 3046, 2976, 2926, 1622, 1582, 1529, 1428, 1337, 1305, 1281, 1216, 1080, 1047, 997, 873, 792, 772, 735, 691, 558, 447 cm−1. Elemental Anal. Calcd for C32H28N2O4Se2 (664.04): C, 58.01; H, 4.26; N, 4.23; Found C, 58.05; H, 4.27; N, 4.17.
2,2′-diselenobis[N-(1R,2S)-(+)-cis-2-hydroxy-1-indanylbezamide] 29b
Yield: 73%; mp 241–243 °C; = +152 (c = 0.41, CHCl3);
1H NMR (700 MHz, DMSO) δ = 2.91–2,95 (m, 1H), 3.14 (dd, J1 = 4.9 Hz, J2 = 16.1 Hz, 1H), 4.46 (qd, J1 = 2.1 Hz, J2 = 4.9 Hz, 1H), 5.18 (d, J = 4.9 Hz, 1H), 5.49 (dd, J1 = 5.6 Hz, J2 = 8.4 Hz, 1H), 7.21–7.27 (m, 2Har), 7.28–7.35 (m, 3Har), 7.40–7.44 (m, 1Har), 7.78 (d, J = 8.4 Hz, 1Har), 8.01 (d, J = 7.7 Hz, 1Har), 8.59 (d, J = 8.4 Hz, 1Har) 13C NMR (700 MHz, DMSO) δ = 39.70 (CH2), 58.10 (CH), 72.53 (CH), 124.94 (CHar), 125.34 (CHar), 126.63 (CHar), 126.86 (CHar), 128.00 (CHar), 129.00 (CHar), 130.42 (CHar), 132.15 (CHar), 132.65 (Car), 133.62 (Car), 141.43 (Car), 142.02 (Car), 168.21 (C=O) 77Se NMR (700 MHz, DMSO) δ = 445.85 ppm; IR: 3289, 3047, 2974, 2927, 1622, 1583, 1530, 1431, 1337, 1306, 1282, 1210, 1082, 1046, 997, 877, 793, 772, 735, 692, 558, 447 cm−1. Elemental Anal. Calcd for C32H28N2O4Se2 (664.04): C, 58.01; H, 4.26; N, 4.23; Found C, 58.09; H, 4.27; N, 4.17.
2,2′-diselenobis[N-(1S,2S)-(−)-trans-2-hydroxy-1-indanylbezamide] 30b
Yield: 93%; mp 234–236 °C; = +153 (c = 0.33, CHCl3);
1H NMR (700 MHz, DMSO) δ = 2.76 (dd, J1 = 7.7 Hz, J2 = 15.4 Hz, 1H), 3.18 (dd, J1 = 7.7 Hz, J2 = 15.4 Hz, 1H), 4.46 (kwintet, J = 7 Hz, 1H), 5.29 (t, J = 7.7 Hz, 1H), 5.41 (d, J = 5.6 Hz, 1H), 7.17–7.23 (m, 4Har), 7.30–7.34 (m, 1Har), 7.37–7.41 (m, 1Har), 7.76 (dd, J1 = 0.7 Hz, J2 = 7.7 Hz, 1Har), 7.88 (dd, J1 = 0.7 Hz, J2 = 7.7 Hz, 1Har), 9.03 (d, J = 8.4 Hz, 1H) 13C NMR (700 MHz, DMSO) δ= 39.34 (CH2), 62.39 (CH), 77.89 (CH), 124.41 (CHar), 125.16 (CHar), 126.59 (CHar), 127.19 (CHar), 128.17 (CHar), 128.64 (CHar), 130.54 (CHar), 132.09 (CHar), 132.67 (Car), 133.88 (Car), 140.46 (Car), 142.18 (Car), 168.32 (C=O) 77Se NMR (700 MHz, DMSO) δ = 445.85 ppm; IR: 3254, 2919, 1630, 1584, 1530, 1456, 1456, 1428, 1345, 1272, 1214, 1122, 1065, 919, 871, 685, 646, 579, 517, 447 cm−1. Elemental Anal. Calcd for C32H28N2O4Se2 (664.04): C, 58.01; H, 4.26; N, 4.23; Found C, 58.05; H, 4.27; N, 4.19.
2,2′-diselenobis[N-(1R,2R)-(−)-trans-2-hydroxy-1-indanylbezamide] 31b
Yield: 86%; mp 233–235 °C; = −150 (c = 0.33, CHCl3);
1H NMR (700 MHz, DMSO) δ = 2.76 (dd, J1 = 7.7 Hz, J2 = 15.4 Hz, 1H), 3.18 (dd, J1 = 7.7 Hz, J2 = 15.4 Hz, 1H), 4.46 (kwintet, J = 7 Hz, 1H), 5.30 (t, J = 7.7 Hz, 1H), 5.40 (d, J = 5.6 Hz, 1H), 7.15–7.24 (m, 4Har), 7.30–7.34 (m, 1Har), 7.36–7.42 (m, 1Har), 7.76 (dd, J1 = 0.7 Hz, J2 = 7.7 Hz, 1Har), 7.88 (dd, J1 = 0.7 Hz, J2 = 7.7 Hz, 1Har), 9.01 (d, J = 8.4 Hz, 1H) 13C NMR (700 MHz, DMSO) δ = 39.31 (CH2), 62.36 (CH), 77.88 (CH), 124.41 (CHar), 125.16 (CHar), 126.59 (CHar), 127.20 (CHar), 128.17 (CHar), 128.64 (CHar), 130.52 (CHar), 132.09 (CHar), 132.67 (Car), 133.84 (Car), 140.44 (Car), 142.17 (Car), 168.32 (C=O) 77Se NMR (700 MHz, DMSO) δ = 445.76 ppm; IR: 3257, 2920, 1630, 1584, 1531, 1458, 1429, 1345, 1272, 1213, 1123, 1066, 920, 871, 658, 649, 580, 518, 448 cm−1. Elemental Anal. Calcd for C32H28N2O4Se2 (664.04): C, 58.01; H, 4.26; N, 4.23; Found C, 57.92; H, 4.27; N, 4.18.