Structural and Luminescence Behavior of Nanocrystalline Orthophosphate KMeY(PO4)2: Eu3+ (Me = Ca, Sr) Synthesized by Hydrothermal Method
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. Structure and Morphology
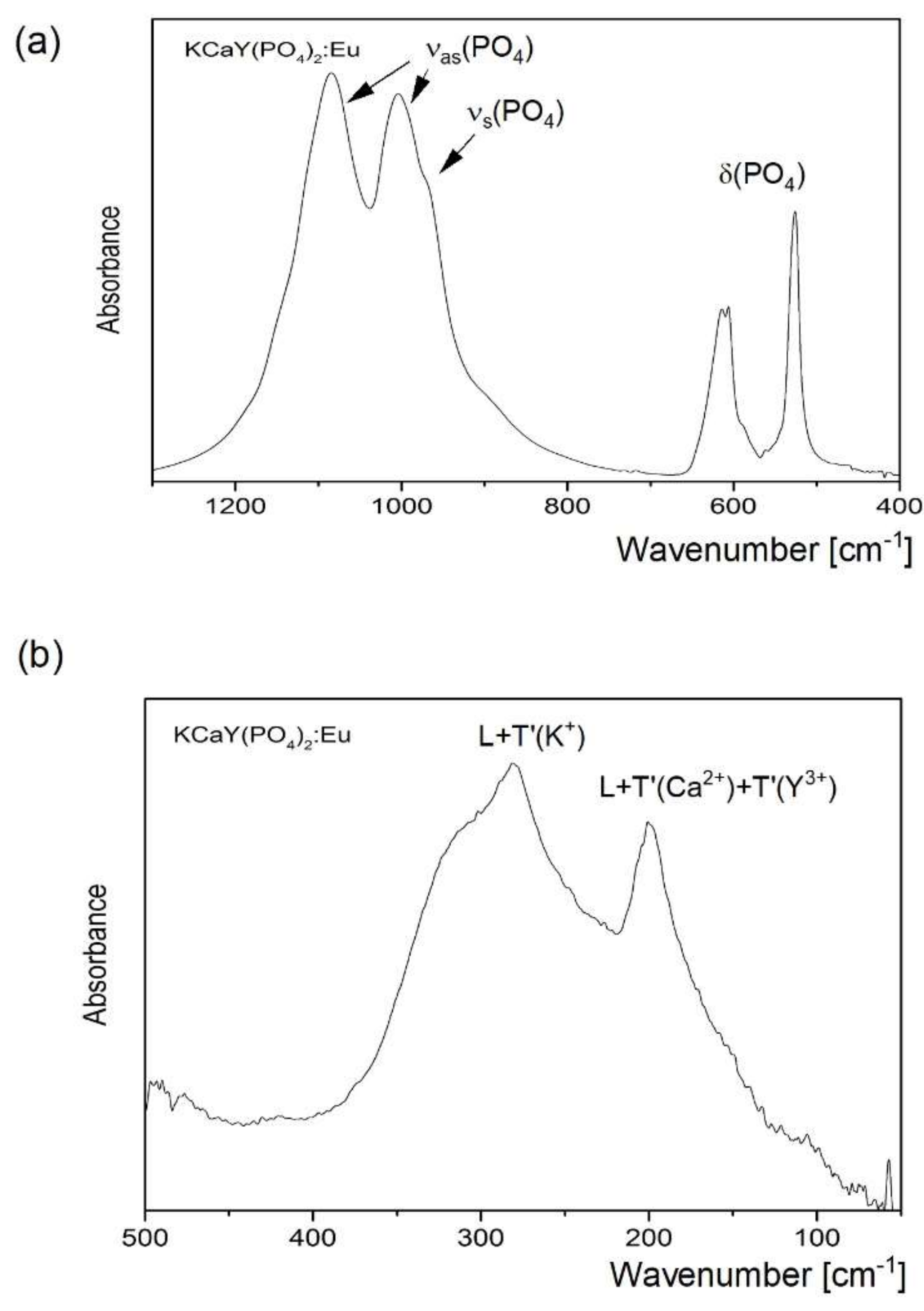
3.2. Raman and IR Studies
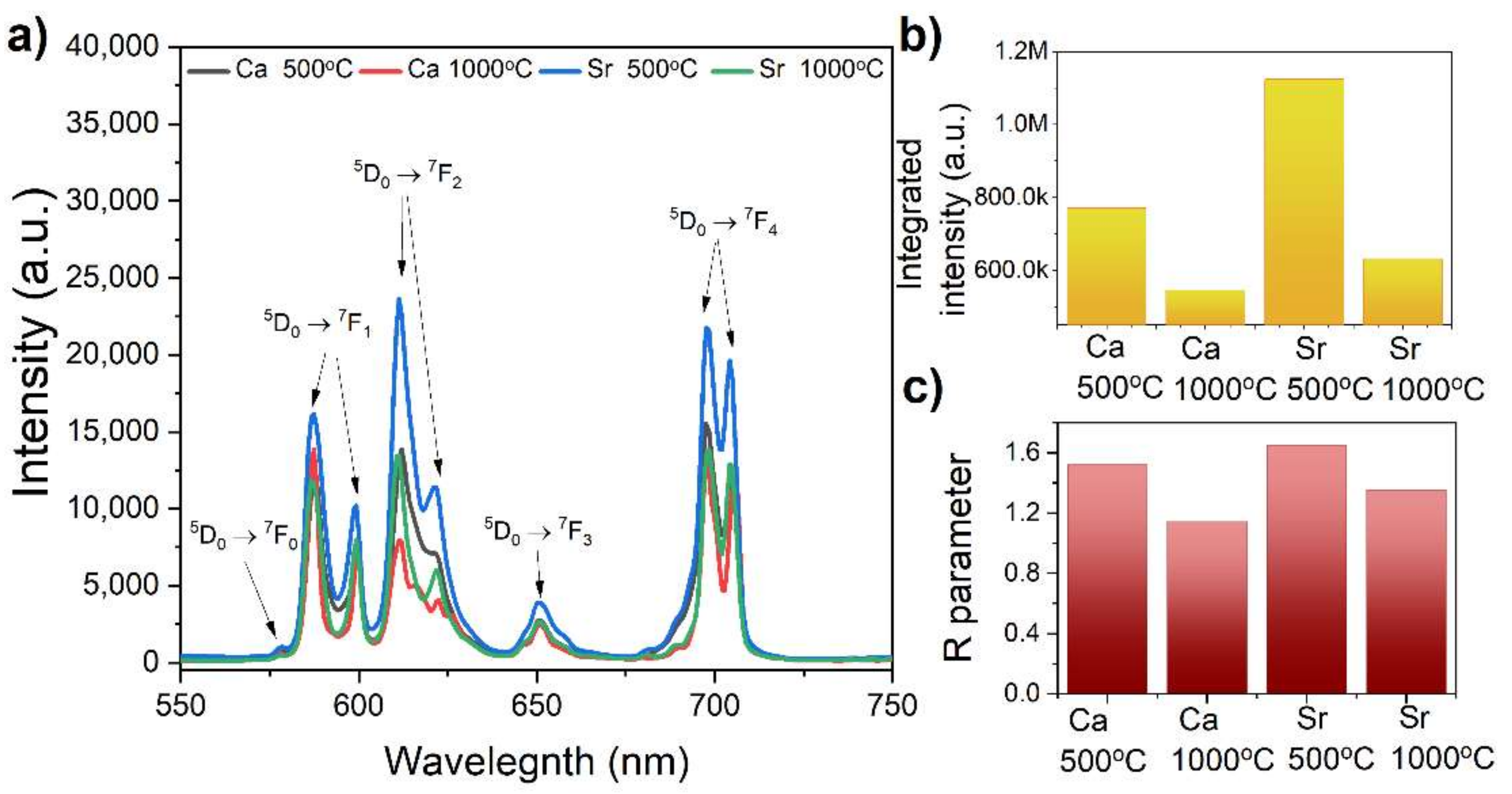
3.3. Optical Studies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Phogat, P.; Khatkar, S.P.; Taxak, V.B.; Malik, R.K. Eu3+ incorporated Bi4MgO4(PO4)2: Deviation of the novel nanophosphor by solution combustion and investigation in to crystallographic and photometric characteristic. Solid State Sci. 2022, 124, 106799. [Google Scholar] [CrossRef]
- Meena, M.L.; Lu, C.H.; Som, S.; Chaurasiya, R.; Lin, S.D. High efficient and thermally stable Eu3+ activated phosphate based phosphors for wLEDs: An experimental and DFT study. J. Alloys Comp. 2022, 895, 162670. [Google Scholar] [CrossRef]
- Zhao, J.; Dong, J.; Ye, X.; Wang, L. A promising novel red-emitting Eu3+-activated neodymium calcium phosphate phosphor with good thermal stability and excellent color purity for WLEDs. J. Mol. Struct. 2021, 1240, 130567. [Google Scholar] [CrossRef]
- Liu, W.R.; Huang, C.H.; Yeh, C.W.; Tsai, J.C.; Chiu, Y.C.; Yeh, Y.T.; Liu, R.S. A Study on the Luminescence and Energy Transfer of Single-Phase and Color-Tunable KCaY(PO4)2:Eu2+,Mn2+ Phosphor for Application in White-Light LEDs. Inorg. Chem. 2012, 51, 9636–9641. [Google Scholar] [CrossRef]
- Carrasco, I.; Bartosiewicz, K.; Piccinelli, F.; Nikl, M.; Bettinelli, M. Structural effects and 5d→4f emission transition shifts induced by Y co-doping in Pr-doped K3Lu1−xYx(PO4)2. J. Lumin. 2017, 189, 113–119. [Google Scholar] [CrossRef]
- Carrasco, I.; Piccinelli, F.; Bettinelli, M. Optical Spectroscopy of Ca9Tb1–xEux(PO4)7 (x=0, 0.1, 1): Weak Donor Energy Migration in the Whitlockite Structure. J. Phys. Chem. C 2017, 121, 16943–16950. [Google Scholar] [CrossRef]
- Yao, Q.; Hu, P.; Sun, P.; Liu, M.; Dong, R.; Chao, K.; Liu, Y.; Jiang, J.; Jiang, H. YAG:Ce3+ Transparent Ceramic Phosphors Brighten the Next-Generation Laser-Driven Lighting. Adv. Mater. 2020, 32, 1907888. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Gao, Y.; Tang, X.; Zhou, J.; Ye, S.; Wu, D.; Wu, Q. Strong f-f excitation in titanium silicate: Near-UV LED pumped red phosphors with outstanding temperature sensitivity. Ceram. Int. 2021, 47, 27157–27164. [Google Scholar] [CrossRef]
- Rodríguez-García, M.M.; Ciric, A.; Ristic, Z.; Williams, J.A.G.; Dramićanin, M.D.; Evans, I.R. Narrow-band red phosphors of high colour purity based on Eu3+ activated apatite-type Gd9.33(SiO4)6O2. J. Matter. Chem. C 2021, 9, 7474–7484. [Google Scholar] [CrossRef]
- Dereń, P.J.; Stefańska, D.; Ptak, M.; Wiśniewski, P. Method to Measure the Degree of Reduction of Eu3+ to Eu2+: How Anion and Cation Vacancies Influence the Degree of Reduction. J. Phys. Chem. C 2021, 125, 24505–24514. [Google Scholar] [CrossRef]
- Li, X.; Yang, C.; Liu, Q.; Wang, X.; Mi, X. Enhancement of luminescence properties of SrAl2Si2O8:Eu3+ red phosphor. Ceram. Int. 2020, 46, 17376–17382. [Google Scholar] [CrossRef]
- Li, S.; Wang, L.; Tang, D.; Cho, Y.; Liu, X.; Zhou, X.; Lu, L.; Zhang, L.; Takeda, T.; Hirosaki, N.; et al. Achieving High Quantum Efficiency Narrow-Band β-Sialon:Eu2+ Phosphors for High-Brightness LCD Backlights by Reducing the Eu3+ Luminescence Killer. Chem. Mater. 2018, 30, 494–505. [Google Scholar] [CrossRef]
- Wang, Y.; Seto, T.; Ishigaki, K.; Uwatoko, Y.; Xiao, G.; Zou, B.; Li, G.; Tang, Z.; Li, Z.; Wang, Y. Pressure-Driven Eu2+-Doped BaLi2Al2Si2N6: A New Color Tunable Narrow-Band Emission Phosphor for Spectroscopy and Pressure Sensor Applications. Adv. Funct. Mater. 2020, 30, 2001384. [Google Scholar] [CrossRef]
- Vlasse, M.; Bochu, P.; Parent, C.; Cheminade, J.P.; Daoudi, A.; le Flem, G.; Hagenmuller, P. Structure determination of calcium neodymium potassium double phosphate CaKNd(PO4)2. Acta Cryst. B 1982, 38, 2328–2331. [Google Scholar] [CrossRef]
- Keller, L.P.; McCarthy, G.J.; Garvey, R. Crystal chemistry of ABC(PO4)2 phases (A=K, Rb, Cs; B=Ca, Sr; C=Ln or Bi) with hexagonal LaPO4 structure. Mater. Res. Bull. 1987, 20, 459–462. [Google Scholar] [CrossRef]
- Parent, C.; Bochu, P.; le Flem, G.; Hagenmuller, P.; Bourcet, J.C.; Gaume-Mahn, F. Energy transfer mechanisms in the KCaLa1−x−yCexTby(PO4)2 phases. J. Phys. Chem. Solids 1984, 45, 39–45. [Google Scholar] [CrossRef]
- Tie, S.; Su, Q.; Yu, Y. Structure and site-symmetry investigation on the hexagonal KCaY(PO4)2. J. Alloys Compd. 1995, 227, 1–4. [Google Scholar] [CrossRef]
- Tie, S.; Su, Q.; Yu, Y. Investigation on the Luminescence of Ln3+ (Ln=Eu, Gd, and Dy) in Hexagonal KCaR(PO4)2 Phosphates (R=Gd, Y). Phys. Stat. Sol. A 1995, 147, 267–276. [Google Scholar] [CrossRef]
- Zhang, Z.J.; Yuan, J.L.; Duan, C.J.; Xiong, D.B.; Chen, H.H.; Zhao, J.T.; Zhang, G.B.; Shi, C.S. Vacuum ultraviolet spectroscopic properties of rare earth (RE=Ce, Tb, Eu, Tm, Sm -doped hexagonal KCaGd(PO4)2 phosphate. J. Appl. Phys. 2007, 102, 093514. [Google Scholar] [CrossRef]
- Chen, Y.C.; Huang, W.Y.; Chen, T.M. Enhancing the performance of photovoltaic cells by using down-converting KCaGd(PO4)2:Eu3+ phosphors. J. Rare Earths 2011, 29, 907–910. [Google Scholar] [CrossRef]
- Zhang, M.; Liang, Y.; Huang, W.; Xia, Z.; Yu, D.; Lan, Y.; Li, G.; Zhou, W. A novel single-composition tunable color emission KSrY(PO4)2:Ce3+, Tb3+, Mn2+ phosphor based on energy transfer. Mater. Res. Bull 2014, 57, 231–237. [Google Scholar] [CrossRef]
- Zhang, M.; Liang, Y.; Tong, M.; Wang, Q.; Yu, D.; Zhao, J.; Wu, J. Photoluminescence properties and energy transfer of a novel bluish-green tunable KSrY(PO4)2:Ce3+, Tb3+ phosphor. Ceram. Int. 2014, 40, 10407–10413. [Google Scholar] [CrossRef]
- Wang, X.F.; Yang, Q.; Wang, G.G.; Wang, X.Z.; Han, J.C. A new single-component KCaY(PO4)2:Dy3+, Eu3+ nanosized phosphor with high color-rendering index and excellent thermal resistance for warm-white NUV-LED. RSC Adv. 2016, 6, 96263–96274. [Google Scholar] [CrossRef]
- Wang, D.; Wang, Y. Spectroscope properties of KCaY(PO4)2:Eu3+ in vacuum ultraviolet region. Mater. Sci. Eng. B 2006, 133, 218–221. [Google Scholar] [CrossRef]
- Campayo, L.; Audubert, F.; Lartigue, J.-E.; Botuha, S.; Bernache-Assolant, D. Study of a phosphate-based material with rhabdophane structure for caesium immobilization: Synthesis, sintering and leaching behaviour. J. Nuclear Mater. 2008, 374, 101–108. [Google Scholar] [CrossRef]
- Campayo, L.; Audubert, F.; Bernache-Assolant, D. Synthesis study of alkaline-bearing rare earth phosphates with rhabdophane structure. Solid State Ionics 2005, 176, 2663–2669. [Google Scholar] [CrossRef]
- Matraszek, A.; Szczygieł, I.; Znamierowska, T. The synthesis and thermal stability of CaKCe(PO4)2 phosphate. Phase equalibria in the CaKPO4-CePO4 system. J. Alloys Comp. 2013, 555, 138–144. [Google Scholar] [CrossRef]
- Boultif, A.; Louer, D. Powder pattern indexing with the dichotomy method. J. Appl. Cryst. 2004, 37, 724–731. [Google Scholar] [CrossRef]
- Laugier, J.; Bochu, B. Checkcell, LMGP-Suite of Programs for the Interpretation of X-Ray Experiments; Ensp/Laboratoire des Materiaux et du Genie, Physique: Saint Martin d’Heres, France, 2014; Available online: http://ccp14.cryst.bbk.ac.uk/tutorial/lmgp/index.html (accessed on 15 January 2022).
- Macalik, L.; Tomaszewski, P.E.; Matraszek, A.; Szczygieł, I.; Solarz, P.; Godlewska, P.; Sobczyk, M.; Hanuza, J. Optical and structural characterization of pure and Pr3+ doped LaPO4 and CePO4 nanocrystals. J. Alloys Comp. 2011, 509, 7458–7465. [Google Scholar] [CrossRef]
- Matraszek, A.; Godlewska, P.; Macalik, L.; Hermanowicz, K.; Hanuza, J.; Szczygieł, I. Optical and thermal characterization of microcrystalline Na3RE(PO4)2:Yb orthoposphates synthesized by Pechini method (RE=Y, La, Gd). J. Alloys Comp. 2015, 619, 275–283. [Google Scholar] [CrossRef]
- Godlewska, P.; Macalik, A.M.L.; Hermanowicz, K.; Ptak, M.; Hanuza, J.; Szczygieł, I. Spectroscopic and structural properties of Na3RE(PO4):Yb orthoposphates synthesised by hydrothermal method (RE=Y, Gd). J. Alloys Comp. 2015, 628, 199–207. [Google Scholar] [CrossRef]
- Pelczarska, A.; Watras, A.; Godlewska, P.; Radomińska, E.; Macalik, L.; Szczygieł, I.; Hanuza, J.; Dereń, P.J. Structural, Raman, FT-IR and optical properties of Rb3Y2(PO4)3 and Rb3La(PO4)2 doped with Eu3+ ions. New J. Chem. 2015, 39, 8474–8483. [Google Scholar] [CrossRef]
- Bondzior, B.; Stefańska, D.; Vu, T.H.Q.; Miniajluk-Gaweł, N.; Dereń, P.J. Red luminescence with controlled rise time in La2MgTiO6:Eu3+. J. Alloys Comp. 2021, 852, 157074. [Google Scholar] [CrossRef]
- Rudnicka, D.; Dereń, P.J. Preliminary spectroscopic properties of K4SrSi3O9 doped with Eu3+. Opt. Mater. 2013, 35, 2531–2534. [Google Scholar] [CrossRef]
- Chaudan, E.; Kim, J.; Tusseau-Nenez, S.; Goldner, P.; Malta, O.L.; Peretti, J.; Gacoin, T. Polarized Luminescence of Anisotropic LaPO4:Eu Nanocrystal Polymorphs. J. Am. Chem. Soc. 2018, 140, 30. [Google Scholar] [CrossRef] [PubMed]
- Blasse, G. Luminescence from the Eu3+ ion in D4d Symmetry. Inorg. Chim. Acta 1988, 142, 153–154. [Google Scholar] [CrossRef]
- Ferreira, R.A.S.; Nobre, S.S.; Granadeiro, C.M.; Nogueira, H.I.S.; Carlos, L.D.; Malta, O.L. A theoretical interpretation of the abnormal 5D0→7F4 intensity based on the Eu3+ local coordination in the Na9[EuW10O36]∙14H2O polyoxometalate. J. Lumin. 2006, 121, 561–567. [Google Scholar] [CrossRef]
- Bettinelli, M.; Speghini, A.; Piccinelli, F.; Neto, A.N.C.; Malta, O.L. Luminescence spectroscopy of Eu3+ in Ca3Sc2Si3O12. J. Lumin. 2011, 131, 1026–1028. [Google Scholar] [CrossRef]
- Wiglusz, R.J.; Grzyb, T.; Watras, A.; Dereń, P.J.; Lis, S.; Strek, W. An impact of sintering temperature and doping level on structural and spectra properties of Eu-doped strontium aluminium oxide. J. Rare Earths 2011, 29, 1105–1110. [Google Scholar] [CrossRef]
- Limbu, S.; Singh, L.R. A comparative study of a tunable body-centered cubic (I) red-emitting nanophosphor for warm WLED applications. J. Solid State Chem. 2022, 308, 122929. [Google Scholar] [CrossRef]
- Strzep, A.; Watras, A.; Zawisza, K.; Boutinaud, P.; Wiglusz, R.J. Forgotten and Resurrected Chernovite-(Y): YAsO4 Doped with Eu3+ Ions as a Potential Nanosized Luminophore. Inorg. Chem. 2017, 56, 10914–10925. [Google Scholar] [CrossRef] [PubMed]
- Pązik, R.; Watras, A.; Macalika, L.; Dereń, P.J. One step urea assisted synthesis of polycrystalline Eu3+ doped KYP2O7–luminescence and emission thermal quenching properties. New J. Chem. 2014, 38, 1129–1137. [Google Scholar] [CrossRef]

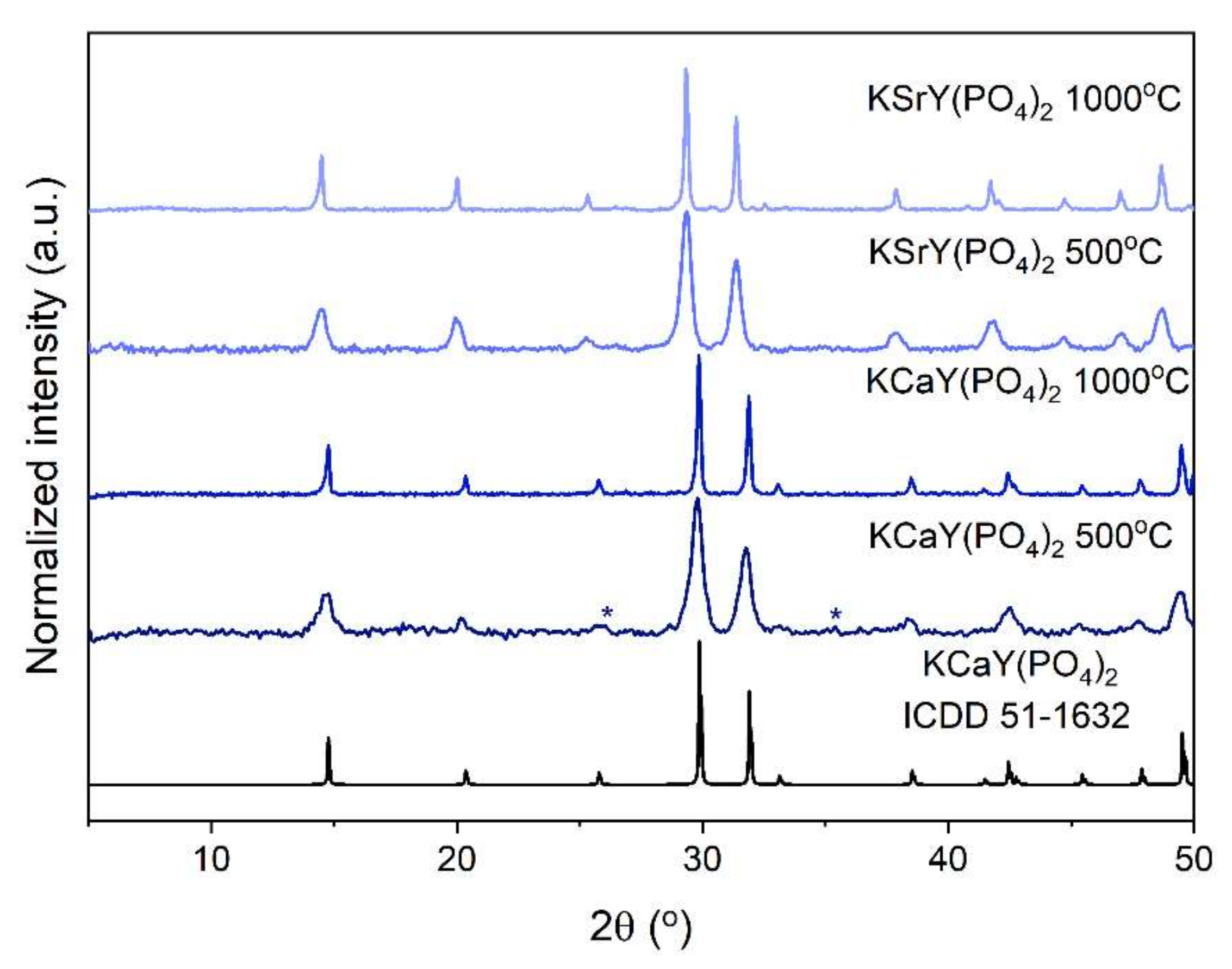
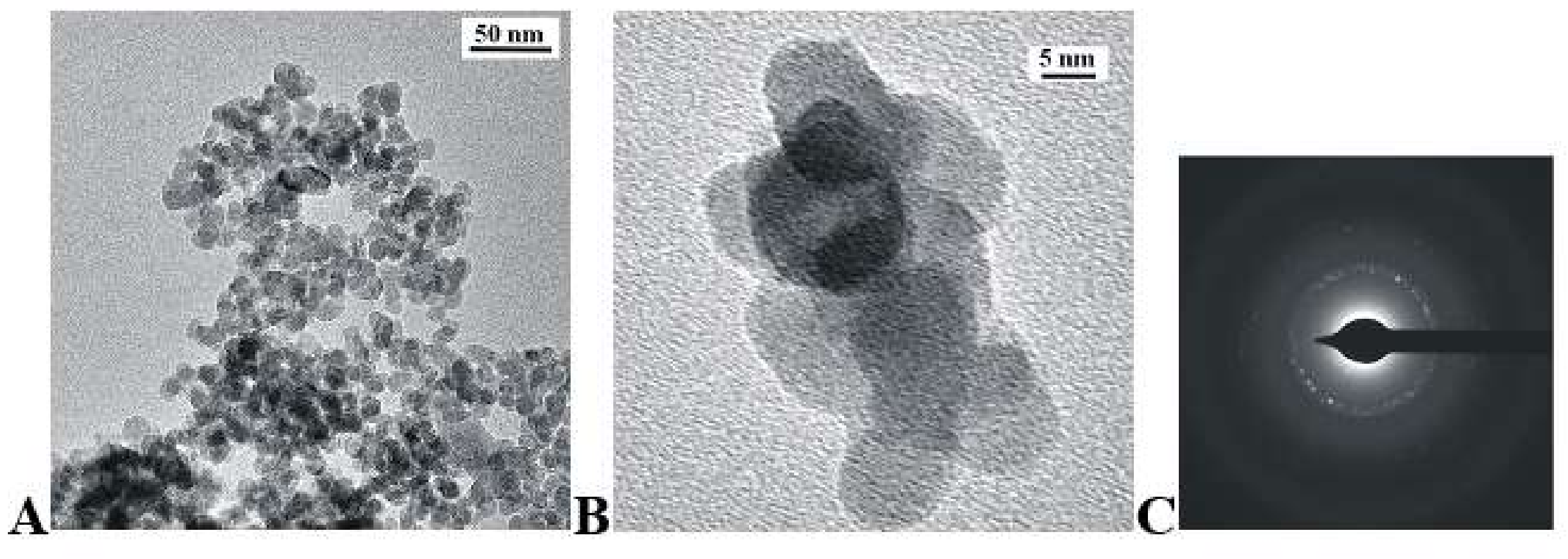
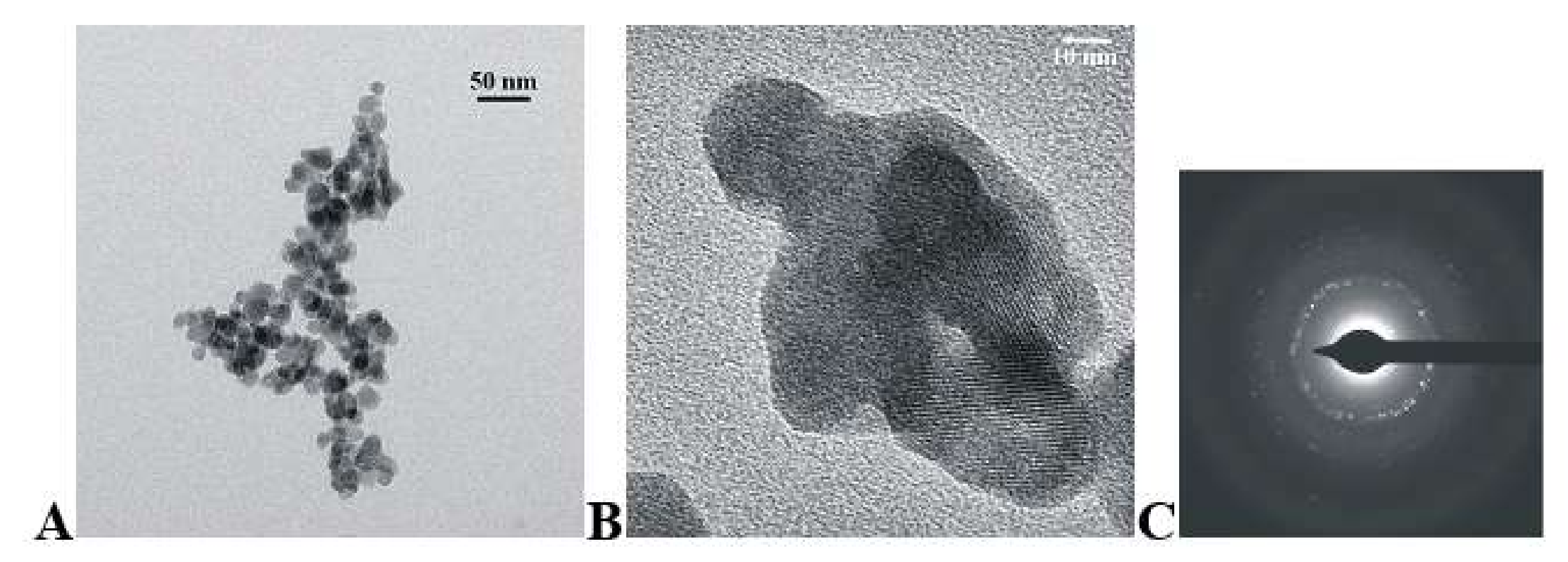



| Sample | Unit Cell Parameters (Å) | Cell Volume (Å3) | |
|---|---|---|---|
| a = b | c | ||
| KCaY(PO4)2:5% Eu3+ | 6.8873 ± 0.0045 | 6.3380 ± 0.0004 | 260.4 |
| KSrY(PO4)2:5% Eu3+ | 6.9911 ± 0.0015 | 6.4186 ± 0.0002 | 271.7 |
| Sample | τ1 (ms) | τ2 (ms) | R |
|---|---|---|---|
| KCaY(PO4)2:5% Eu3+/500 °C | 1.4 | 5.4 | 1.52 |
| KCaY(PO4)2:5% Eu3+/1000 °C | 4.7 | --- | 1.14 |
| KSrY(PO4)2:5% Eu3+/500 °C | 1.7 | 4.6 | 1.65 |
| KSrY(PO4)2:5% Eu3+/1000 °C | 3.8 | --- | 1.35 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pelczarska, A.J.; Stefańska, D.; Watras, A.; Macalik, L.; Szczygieł, I.; Hanuza, J. Structural and Luminescence Behavior of Nanocrystalline Orthophosphate KMeY(PO4)2: Eu3+ (Me = Ca, Sr) Synthesized by Hydrothermal Method. Materials 2022, 15, 1850. https://doi.org/10.3390/ma15051850
Pelczarska AJ, Stefańska D, Watras A, Macalik L, Szczygieł I, Hanuza J. Structural and Luminescence Behavior of Nanocrystalline Orthophosphate KMeY(PO4)2: Eu3+ (Me = Ca, Sr) Synthesized by Hydrothermal Method. Materials. 2022; 15(5):1850. https://doi.org/10.3390/ma15051850
Chicago/Turabian StylePelczarska, Aleksandra J., Dagmara Stefańska, Adam Watras, Lucyna Macalik, Irena Szczygieł, and Jerzy Hanuza. 2022. "Structural and Luminescence Behavior of Nanocrystalline Orthophosphate KMeY(PO4)2: Eu3+ (Me = Ca, Sr) Synthesized by Hydrothermal Method" Materials 15, no. 5: 1850. https://doi.org/10.3390/ma15051850
APA StylePelczarska, A. J., Stefańska, D., Watras, A., Macalik, L., Szczygieł, I., & Hanuza, J. (2022). Structural and Luminescence Behavior of Nanocrystalline Orthophosphate KMeY(PO4)2: Eu3+ (Me = Ca, Sr) Synthesized by Hydrothermal Method. Materials, 15(5), 1850. https://doi.org/10.3390/ma15051850








