Comparison between Early Loaded Single Implants with Internal Conical Connection or Implants with Transmucosal Neck Design: A Non-Randomized Controlled Trial with 1-Year Clinical, Aesthetics, and Radiographic Evaluation
Abstract
:1. Introduction
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wennerberg, A.; Albrektsson, T. Current challenges in successful rehabilitation with oral implants. J. Oral Rehabil. 2011, 38, 286–294. [Google Scholar] [CrossRef] [PubMed]
- Dunican, E.M.; Fahy, J.V. The role of Type 2 inflammation in the pathogenesis of asthma exacerbations. Ann. Am. Thorac. 2015, 12 (Suppl. 2), S144–S149. [Google Scholar]
- Klopfleisch, R. Macrophage reaction against biomaterials in the mouse model—Phenotypes, functions and markers. Acta Biomater. 2019, 43, 3–13. [Google Scholar] [CrossRef]
- Trindade, R.; Albrektsson, T.; Tengvall, P.; Wennerberg, A. Foreign body reaction to biomaterials: On mechanisms for buildup and breakdown of osseointegration. Clin. Implant Dent. Relat. Res. 2016, 18, 192–203. [Google Scholar] [CrossRef] [PubMed]
- Tallarico, M.; Meloni, S.M. Retrospective analysis on survival rate, template-related complications, and prevalence of peri-implantitis of 694 anodized implants placed using computer-guided surgery: Results between 1 and 10 years of follow-up. Int. J. Oral Maxillofa. Implant. 2017, 32, 1162–1171. [Google Scholar] [CrossRef] [PubMed]
- Papaspyridakos, P.; Chen, C.J.; Singh, M.; Weber, H.P.; Gallucci, G.O. Success criteria in implant dentistry: A systematic review. J. Dent. Res. 2012, 91, 242–248. [Google Scholar] [CrossRef]
- Le Guehennec, L.; Goyenvalle, E.; Lopez-Heredia, M.A.; Weiss, P.; Amouriq, Y.; Layrolle, P. Histomorphometric analysis of the osseointegration of four different implant surfaces in the femoral epiphyses of rabbits. Clin. Oral Implant Res. 2008, 19, 1103–1110. [Google Scholar] [CrossRef]
- Le Guéhennec, L.; Soueidan, A.; Layrolle, P.; Amouriq, Y. Surface treatments of titanium dental implants for rapid osseointegration. Dent. Mater. 2007, 23, 844–854. [Google Scholar] [CrossRef]
- Schliephake, H.; Scharnweber, D.; Roesseler, S.; Dard, M.; Sewing, A.; Aref, A. Biomimetic calcium phosphate composite coating of dental implants. Int. J. Oral Maxillofac. Implant. 2006, 21, 738–746. [Google Scholar]
- Hermann, J.S.; Schoolfield, J.D.; Schenk, R.K.; Buser, D.; Cochran, D.L. Influence of the size of the microgap on crestal bone changes around titanium implants. A histometric evaluation of unloaded non-submerged implants in the canine mandible. J. Periodontol. 2001, 72, 1372–1383. [Google Scholar] [CrossRef] [Green Version]
- Albrektsson, T.; Zarb, G.; Worthington, P.; Eriksson, A.R. The long-term efficacy of currently used dental implants: A review and proposed criteria of success. Int. J. Oral Maxillofac. Implant. 1986, 1, 11–25. [Google Scholar]
- Meloni, S.M.; Lumbau, A.; Baldoni, E.; Pisano, M.; Spano, G.; Massarelli, O.; Tallarico, M. Platform switching versus regular platform single implants: 5-year post-loading results from a randomised controlled trial. Int. J. Oral Maxillofac. Implant. 2020, 13, 43–52. [Google Scholar]
- Pozzi, A.; Tallarico, M.; Moy, P.K. Immediate loading with a novel implant featured by variable-threaded geometry, internal conical connection and platform shifting: Three-year results from a prospective cohort study. Eur. J. Oral Implantol. 2015, 8, 51–63. [Google Scholar] [PubMed]
- Qian, J.; Wennerberg, A.; Albrektsson, T. Reasons for marginal bone loss around oral implants. Clin. Implant Dent. Relat. Res. 2012, 14, 792–807. [Google Scholar] [CrossRef] [PubMed]
- Pozzi, A.; Agliardi, E.; Tallarico, M.; Barlattani, A. Clinical and radiological outcomes of two implants with different prosthetic interfaces and neck configurations: Randomized, controlled, split-mouth clinical trial. Clin. Implant Dent. Relat. 2014, 16, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Norton, M.R. Marginal bone levels at single tooth implants with a conical fixture design. The influence of surface macro- and microstructure. Clin. Oral Implant. Res. 1998, 9, 91–99. [Google Scholar] [CrossRef]
- Galindo-Moreno, P.; León-Cano, A.; Ortega-Oller, I.; Monje, A.; O’Valle, F.; Catena, A. Marginal bone loss as success criterion in implant dentistry: Beyond 2 mm. Clin. Oral Implant. Res. 2015, 26, e28–e34. [Google Scholar] [CrossRef]
- Ceruso, F.M.; Ieria, I.; Martelli, M.; Lumbau, A.I.; Xhanari, E.; Gargari, M. New generation of fixture–Abutment connection combining soft tissue design and vertical screw-retained restoration: 1-Year clinical, aesthetics and radiographic preliminary evaluation. Dent. J. 2021, 9, 35. [Google Scholar] [CrossRef]
- Özcan, M.; Hämmerle, C. Titanium as a reconstruction and implant material in dentistry: Advantages and pitfalls. Materials 2012, 5, 1528–1545. [Google Scholar] [CrossRef] [Green Version]
- Amari, Y.; Piattelli, A.; Alccayhuaman KA, A.; Mesa, N.F.; Ferri, M.; Iezzi, G.; Botticelli, D. Bone healing at non-submerged implants installed with different insertion torques: A split-mouth histomorphometric randomized controlled trial. Int. J. Implant Dent. 2019, 5, 39. [Google Scholar] [CrossRef]
- Sul, Y.T.; Byon, E.; Wennerberg, A. Surface characteristics of electrochemically oxidized implants and acid-etched implants: Surface chemistry, morphology, pore configurations, oxide thickness, crystal structure, and roughness. Int. J. Oral Maxillofac. Implant. 2008, 23, 631–640. [Google Scholar]
- Hall, J.; Lausmaa, J. Properties of a new porous oxide surface on titanium implants. J. Ossetointegr. 2000, 1, 5–8. [Google Scholar]
- Schüpbach, P.; Glauser, R.; Rocci, A.; Martignoni, M.; Sennerby, L.; Lundgren, A.; Gottlow, J. The human bone-oxidized titanium implant interface: A light microscopic, scanning electron microscopic, back-scatter scanning electron microscopic, and energy-dispersive X-ray study of clinically retrieved dental implantsn. Clin. Implant Dent. Relat. Res. 2005, 7, S36–S43. [Google Scholar] [CrossRef] [PubMed]
- Esposito, M.; Coulthard, P.; Thomsen, P.; Worthington, H.V. The role of implant surface modifications, shape and material on the success of osseointegrated dental implants. A Cochrane systematic review. Eur. J. Prosthodont. Restor. Dent. 2005, 13, 15–31. [Google Scholar] [PubMed]
- Davies, J.E. Mechanisms of endosseous integration. Int. J. Prosthodont. 1998, 11, 391–401. [Google Scholar]
- Inoue, T.; Cox, J.E.; Pilliar, R.M.; Melcher, A.H. Effect of the surface geometry of smooth and porous-coated titanium alloy on the orientation of fibroblasts in vitro. J. Biomed. Mater. Res. 1987, 21, 107–126. [Google Scholar] [CrossRef]
- Roccuzzo, M.; Bonino, L.; Dalmasso, P.; Aglietta, M. Long-term results of a three arms prospective cohort study on implants in periodontally compromised patients: 10-year data around sandblasted and acid-etched (SLA) surface. Clin. Oral Implant. Res. 2014, 25, 1105–1112. [Google Scholar] [CrossRef]
- Malò, P.; Araújo Nobre, M.; Gonçalves, Y.; Lopes, A.; Ferro, A. Immediate function of anodically oxidized surface implants (TiUnite™) for fixed prosthetic rehabilitation: Retrospective study with 10 years of follow-up. BioMed. Res. Int. 2016, 2061237. [Google Scholar] [CrossRef] [PubMed]
- Tallarico, M.; Ceruso, F.M.; Muzzi, L.; Meloni, S.M.; Kim, Y.J.; Gargari, M.; Martinolli, M. Effect of simultaneous immediate implant placement and guided bone reconstruction with ultra-fine titanium mesh membranes on radiographic and clinical parameters after 18 months of loading. Materials 2019, 12, 1710. [Google Scholar] [CrossRef] [Green Version]
- French, D.; Larjava, H.; Tallarico, M. Retrospective study of 1087 anodized implants placed in private practice: Risk indicators associated with implant failure and relationship between bone levels and soft tissue health. Implant Dent. 2018, 27, 177–187. [Google Scholar] [CrossRef]
- Ceruso, F.M.; Ottria, L.; Martelli, M.; Gargari, M.; Barlattani, A. Transgingival Implants with a convergent collar (Prama). Surgical and screwed prosthetic approach. A case report. J. Biol. Regul. Homeost. Agents 2020, 34 (Suppl. 1), 71–77. [Google Scholar]
- Canullo, L.; Tallarico, M.; Pradies, G.; Marinotti, F.; Loi, I.; Cocchetto, R. Soft and hard tissue response to an implant with a convergent collar in the esthetic area: Preliminary report at 18 months. Int. J. Esthet. Dent. 2017, 12, 306–323. [Google Scholar]
- Ceruso, F.M.; Barnaba, P.; Mazzoleni, S.; Ottria, L.; Gargari, M.; Zuccon, A.; Bruno, G.; Di Fiore, A. Implant-abutment connections on single crowns: A systematic review. Oral Implantol. 2017, 10, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Tallarico, M.; Canullo, L.; Caneva, M.; Ozcan, M. Microbial colonization at the implant-abutment interface and its possible influence on periimplantitis: A systematic review and meta-analysis. J. Prosthodont. Res. 2017, 61, 233–241. [Google Scholar] [CrossRef] [Green Version]
- Tallarico, M.; Caneva, M.; Meloni, S.M.; Xhanari, E.; Covani, U.; Canullo, L. Definitive abutments placed at implant insertion and never removed: Is it an effective approach? a systematic review and meta-analysis of randomized controlled trials. J. Oral Maxillofac. Surg. 2018, 76, 316–324. [Google Scholar] [CrossRef] [PubMed]
- Cosola, S.; Marconcini, S.; Boccuzzi, M.; Menchini Fabris, G.B.; Covani, U.; Peñarrocha-Diago, M.; Peñarrocha-Oltra, D. Radiological outcomes of bone-level and tissue-level dental implants: Systematic review. Int. J. Environ. Res. Public Health 2020, 17, 6920. [Google Scholar] [CrossRef] [PubMed]
- Canullo, L.; Menini, M.; Bagnasco, F.; Di Tullio, N.; Pesce, P. Tissue-level versus bone-level single implants in the anterior area rehabilitated with feather-edge crowns on conical implant abutments: An up to 5-year retrospective study. J. Prosthet. Dent. 2021, in press. [Google Scholar] [CrossRef]



| General contraindications to implant surgery (e.g., uncontrolled diabetes) |
| Patients irradiated in the head and neck area |
| Immunosuppresed or immunocompromised patients |
| Patients treated or under treatment with intravenous amino-biphosphonates |
| Patients with untreated periodontitis |
| Immediate, postextractive implants |
| Patients with poor oral hygiene and motivation |
| Pregnancy or nursing |
| Substance abuser, psychiatric problems or unrealistic expectations |
| Patients unable to be followed-up |
| Patients with infection in the area intended for implant placement |
| NOBEL Biocare Parallel | PRAMA |
|---|---|
| Pure grade IV titanium | Pure grade IV titanium |
| TiUnite anodized surface | Pure ZIrTi Surface |
| 12° of conical connection | Transmucosal with a convergent part of 2 mm and a cylindrical part of 0.8 mm, without sharp edges |
| Roughness 1.35 μm | Roughness 1.4–1.7 μm |
| Primary Outcome | Implant failure | An implant was considered a failure if it presented any mobility, tested by tapping or rocking the implant head with a hand instrument and/ or any signs of radiolucency and/or fracture on an intraoral radiograph taken with a paralleling technique strictly perpendicular to the implant bone interface. The implant stability were assessed at initial loading and yearly without the prostheses removed. |
| Prosthetic failure | A prosthesis is considered a failure if it needs to be replaced by an alternative prosthesis. | |
| Complication | Any biological (pain, swelling, suppuration, etc.) and/or mechanical complication (fracture of the framework and/or the veneering material, screw loosening, etc.) was considered. | |
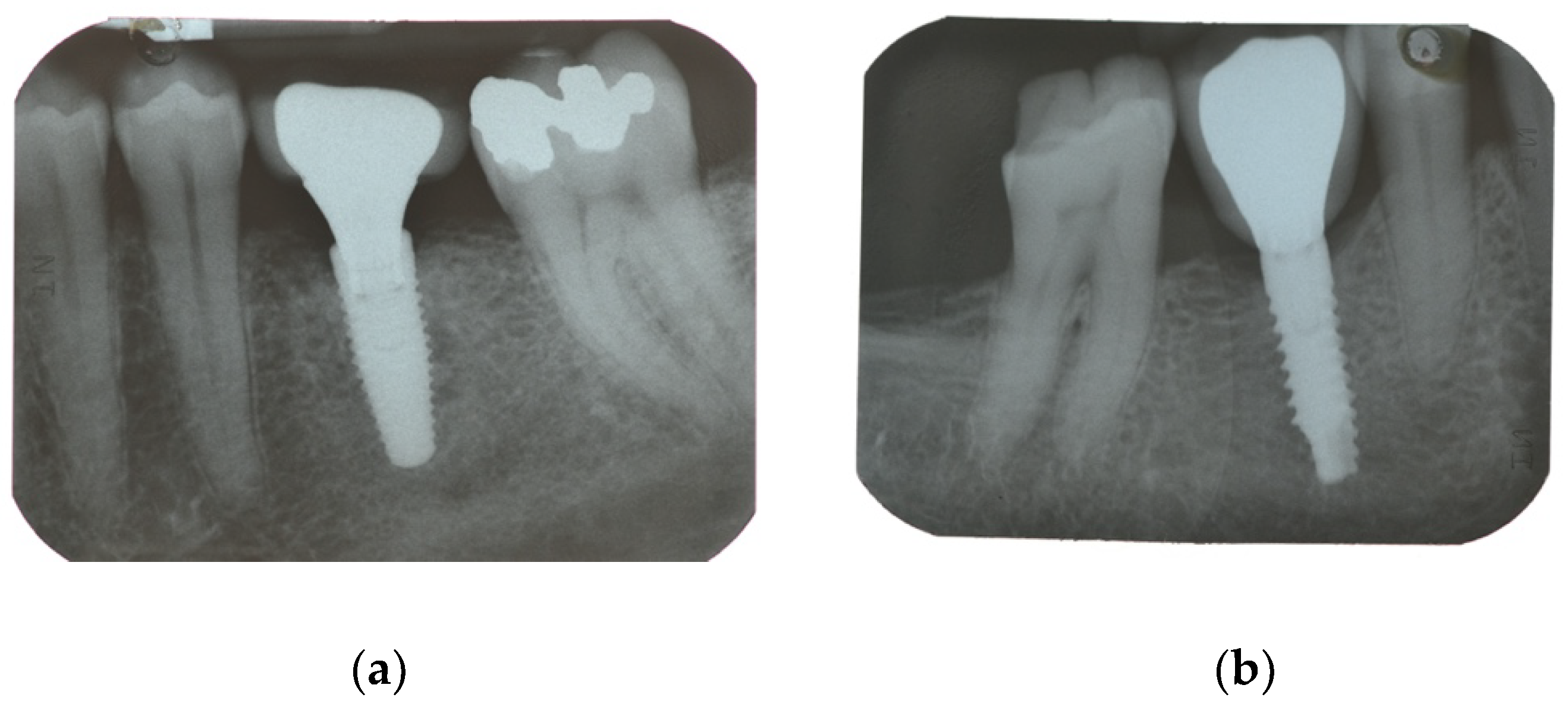
| Secondary Outcome | Marginal bone loss (MBL) | Marginal bone level changes were assessed using intraoral digital periapical radiographs at implant placement (baseline), and at after one year on function. Intraoral radiographs were taken with the parallel technique with customized holder. All the radiographs were evaluated under routine conditions. The software has been calibrated for every single image using the known distance of the implant diameter or length. The distance from the reference point at the implant neck to the first bone to implant contact were taken as the horizontal marginal bone level at both mesial and distal aspects. The average radiographic values of mesial and distal measurements were taken for each implant. Variation of the marginal bone levels at different time was taken as marginal bone loss. |
| BI and PI | Soft tissue parameters (BI and PI) around the implant/abutment interfaces were assessed yearly using a plastic periodontal probe (Plast-o-Probe, Dentsply Maillefer, Ballaigues, Switzerland). The BI were evaluated at four sites around each implant (mesial, distal, buccal and lingual) according to the Mombelli Index. The bleeding elicited 20 s after the careful insertion of a periodontal probe 1 mm into the mucosal sulcus, parallel to where the abutment wall will be assessed (0 = no bleeding; 1 = spot bleeding, 2 = linear bleeding, and 3 = spontaneous bleeding). The PI, defined as the presence of plaque (yes/no) on the abutment/restoration complex, was measured by running the periodontal probe parallel to the abutment surfaces, and scored at one site for implant. An independent blinded dental hygienist performed all periodontal measurements. | |
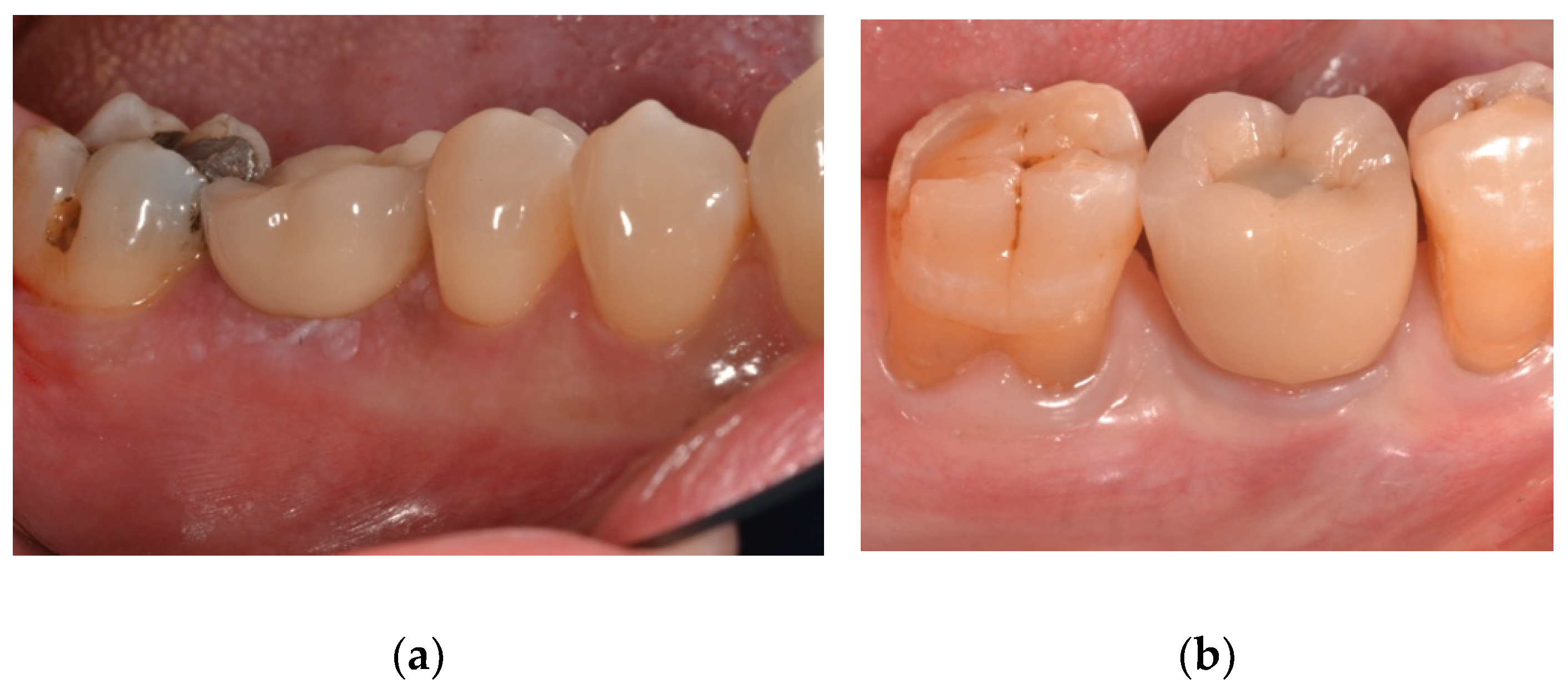
| PES | The aesthetic evaluation was performed according to the pink esthetic score (PES) on the vestibular and occlusal pictures taken including at least one adjacent tooth per side. The values will be assessed annually after definitive loading. Seven variabilities (mesial papilla, distal papilla, soft tissue level, soft tissue contour, alveolar process deficiency, soft tissue color and texture) were assessed at 0 to 2 score (0 being poorest and 2 being the best) by a blind outcome assessor. |
| NOBEL Parallel | PRAMA | p Value | |
|---|---|---|---|
| Mean age (SD) | 49.4(10.6) | 47.5(13.4) | 0.6759 |
| Sex (Male/Female) | 6/9 | 8/7 | 0.7152 |
| Smokers | 2 | 1 | 1.0 |
| Drop-Out | 1 | 2 | 1.0 |
| Maxilla/Mandible | 9/6 | 7/9 | 0.4795 |
| Molar implants | 9 | 7 | 0.4795 |
| Premolar implants | 5 | 7 | 0.7160 |
| Anterior implants | 1 | 1 | 1.0 |
| NOBEL Parallel | PRAMA | p Value | |
|---|---|---|---|
| Implant placement | 0.04 ± 0.06 (0.00 to 0.07); n = 15 | 0.01 ± 0.02 (0.00 to 0.02); n = 15 | 0.128 |
| One-year follow-up | 0.99 ± 0.71 (0.61 to 1.36); n = 14 | 0.65 ± 0.48 (0.40 to 0.91); n = 13 | 0.166 |
| Difference | 0.96 ± 0.72 (0.58 to 1.34) | 0.65 ± 0.48 (0.40 to 0.90) | 0.192 |
| NOBEL Parallel (n = 14) | PRAMA (n = 13) | p Value | |
|---|---|---|---|
| BI | 3 | 3 | 1.0 |
| PI | 4 | 3 | 1.0 |
| PES | 9.79 ± 2.61 (8.42 to 11.15) | 10.46 ± 2.30 (9.26 to 11.66) | 0.481 |
| p Value | 0.000 | 0.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ceruso, F.M.; Ieria, I.; Tallarico, M.; Meloni, S.M.; Lumbau, A.I.; Mastroianni, A.; Zotti, A.; Gargari, M. Comparison between Early Loaded Single Implants with Internal Conical Connection or Implants with Transmucosal Neck Design: A Non-Randomized Controlled Trial with 1-Year Clinical, Aesthetics, and Radiographic Evaluation. Materials 2022, 15, 511. https://doi.org/10.3390/ma15020511
Ceruso FM, Ieria I, Tallarico M, Meloni SM, Lumbau AI, Mastroianni A, Zotti A, Gargari M. Comparison between Early Loaded Single Implants with Internal Conical Connection or Implants with Transmucosal Neck Design: A Non-Randomized Controlled Trial with 1-Year Clinical, Aesthetics, and Radiographic Evaluation. Materials. 2022; 15(2):511. https://doi.org/10.3390/ma15020511
Chicago/Turabian StyleCeruso, Francesco Mattia, Irene Ieria, Marco Tallarico, Silvio Mario Meloni, Aurea Immacolata Lumbau, Alessandro Mastroianni, Alessio Zotti, and Marco Gargari. 2022. "Comparison between Early Loaded Single Implants with Internal Conical Connection or Implants with Transmucosal Neck Design: A Non-Randomized Controlled Trial with 1-Year Clinical, Aesthetics, and Radiographic Evaluation" Materials 15, no. 2: 511. https://doi.org/10.3390/ma15020511
APA StyleCeruso, F. M., Ieria, I., Tallarico, M., Meloni, S. M., Lumbau, A. I., Mastroianni, A., Zotti, A., & Gargari, M. (2022). Comparison between Early Loaded Single Implants with Internal Conical Connection or Implants with Transmucosal Neck Design: A Non-Randomized Controlled Trial with 1-Year Clinical, Aesthetics, and Radiographic Evaluation. Materials, 15(2), 511. https://doi.org/10.3390/ma15020511








