Current Advances in the Regeneration of Degenerated Articular Cartilage: A Literature Review on Tissue Engineering and Its Recent Clinical Translation
Abstract
:1. Introduction
2. Materials and Methods
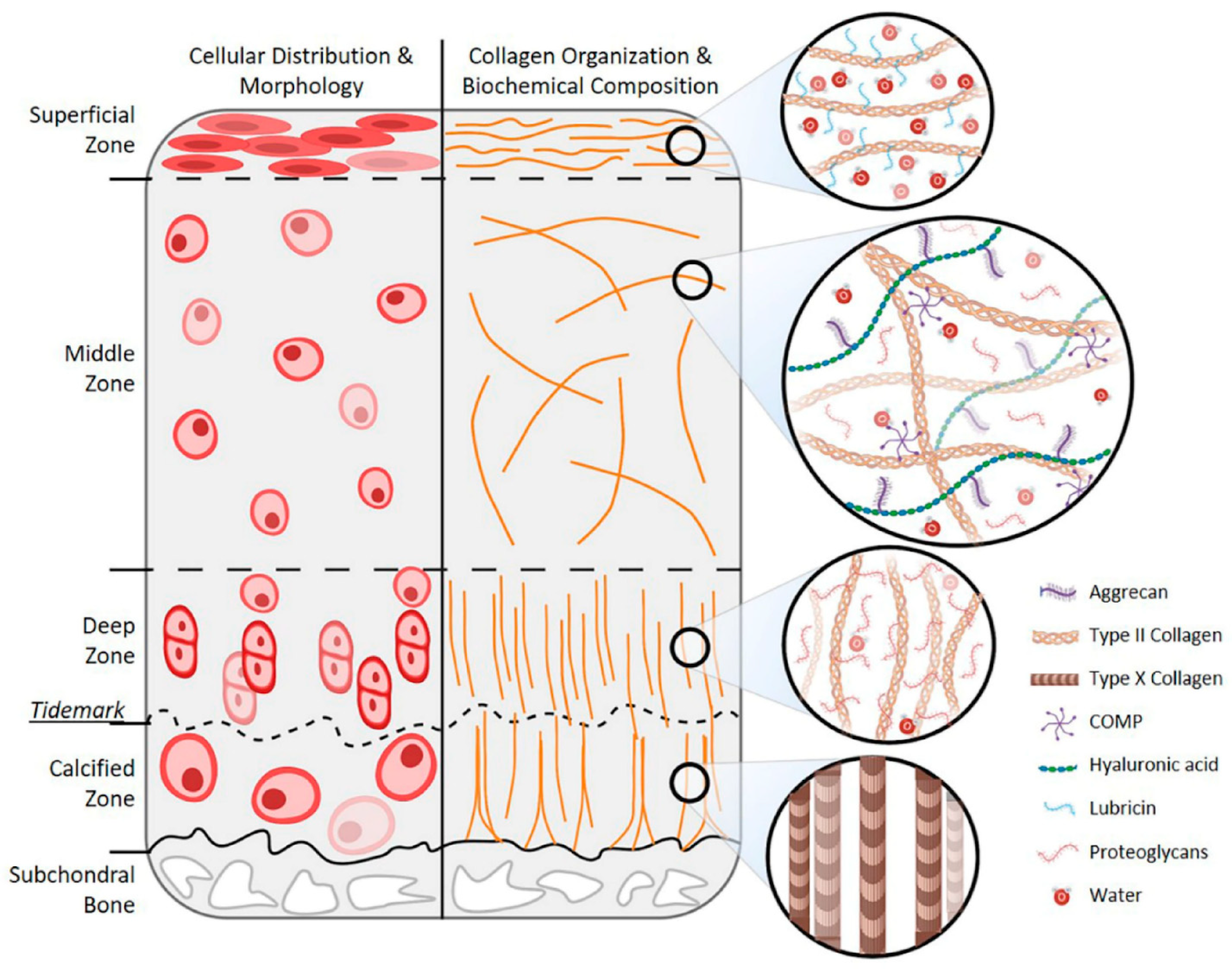
3. Age-Related Changes in Articular Cartilage
- ICRS Grade 0: Normal;
- ICRS Grade 1: Nearly Normal (superficial lesions);
- ICRS Grade 2: Abnormal (lesions extending down to <50% of cartilage depth);
- ICRS Grade 3: Severely Abnormal (cartilage defects extending down >50% of cartilage depth);
- ICRS Grade 4: Severely Abnormal (cartilage defects extending through the subchondral bone).
3.1. Age-Related Changes in Chondrocytes
3.2. Age-Related Changes in Cartilage ECM
4. Tissue Engineering Strategies for Articular Cartilage Regeneration
4.1. Cell-Based Tissue Engineering Strategies
4.1.1. Scaffold-Based Strategies
4.1.2. Scaffold-Free Strategies
4.1.3. Injectables
4.1.4. Clinical Studies
4.2. Cell-Free Tissue Engineering Strategies
4.2.1. Scaffold-Based Strategies
4.2.2. Injectables
4.2.3. Clinical Studies
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ACCs | articular cartilage chondrocytes |
| ACI | autologous chondrocyte implantation |
| ACLT | anterior cruciate ligament transection |
| AD-MSCs | adipose-derived mesenchymal stem cells |
| AGEs | advanced glycation end-products |
| AGG | aggrecan |
| antimiR-221 | microRNA inhibitor targeting miR-221 |
| ApoPep-1 | apoptosis-targeting peptide-1 |
| BGP | β-glycerophosphate |
| BM-MSCs | bone marrow-derived mesenchymal stromal cells |
| BMP | bone morphogenetic protein |
| C-ACT | collagen-augmented chondrogenesis technique |
| CCP | costal chondrocyte-derived pellet-type |
| Col-1 | collagen type I |
| Col-2 | collagen type II |
| CPCs | chondrogenic progenitor cells |
| CS-MA | methacrylated chondroitin sulfate |
| ECM | extracellular matrix |
| EPL | ε-poly-L-lysine |
| GAG | glycosaminoglycan |
| GelMA | gelatin–methacrylamide |
| GO | graphene oxide |
| HA | hyaluronic acid |
| haMPCs | human adipose-derived mesenchymal progenitor cells |
| hAMSCs | human amniotic mesenchymal stem cells |
| HAV | histidine-alanine-valine |
| hMSCs | human mesenchymal stem cells |
| HTO | high tibial osteotomy |
| hWJ-MSC-Exos | hWJ-MSC-derived exosomes |
| hWJ-MSC | human Wharton’s Jelly mesenchymal stem cells |
| ICRS | International Cartilage Repair Society |
| IGF-1 | insulin-like growth factor-1 |
| IPFP | infrapatellar fat pad |
| JS-Paint | joint surface paint |
| KGN | kartogenin |
| KL Grade | Kellgren and Lawrence Grade |
| LAA | L-ascorbic acid |
| MACI | matrix-induced autologous chondrocyte implantation |
| MAP | microporous annealed particle |
| MeHA | methacrylated hyaluronic acid |
| MMPs | matrix metalloproteinases |
| MRI | magnetic resonance imaging |
| MSC-Exos | MSC-derived exosomes |
| MSCs | mesenchymal stem cells |
| NB | N-(2-aminoethyl)-4-(4-(hydroxymethyl)-2-methoxy-5-nitrosophenoxy) butanamide |
| NOD/SCID | non-obese diabetic/severe combined immunodeficient |
| OA | Osteoarthritis |
| OLT | osteochondral lesion of the talus |
| OPF | oligo(poly(ethylene glycol) fumarate) |
| PCL | poly(ε-caprolactone) |
| PCM | pericellular matrix |
| PD sheets | allogeneic polydactyly-derived chondrocyte sheets |
| PDLLA | poly-D, L-lactic acid/polyethylene glycol |
| PEG | polyethylene glycol |
| PL | platelet lysate |
| PLA | polylactic acid |
| PLEL | poly(d,L-lactide)-poly(ethylene glycol)-poly(d,L-lactide) |
| PLGA | poly(lactic-co-glycolic acid) |
| PMPC | poly(2-methacryloyloxyethyl phosphorylcholine) |
| PRP | platelet-rich plasma |
| PTOA | post-traumatic osteoarthritis |
| RAGE | receptor for advanced glycation endproducts |
| RCTs | randomized clinical trials |
| RGD | arginine-glycine-aspartate |
| rhSDF-1α | recombinant human stromal cell–derived factor 1α |
| rhTG-4 | recombinant human transglutaminase 4 |
| ROS | reactive oxygen species |
| SASP | senescence-associated secretory phenotype |
| SDF-1 | stromal cell-derived factor-1 |
| SDS | sodium dodecyl sulfate |
| SF-MSCs | synovial fluid-derived mesenchymal stem cells |
| SF | silk fibroin |
| Si-HPMC | siloxane derived hydroxypropylmethylcellulose |
| SMSCs | synovium-derived mesenchymal stem cells |
| SS | silk spidroin |
| SVF | stromal vascular fraction |
| TAN | tanshinone IIA |
| TCO | trans-cyclooctene |
| TGF-β | transforming growth factor-β |
| TMJ-OA | temporomandibular joint osteoarthritis |
| UC-derived WJ | umbilical cord-derived Wharton’s Jelly |
References
- Di Micco, R.; Krizhanovsky, V.; Baker, D.; d’Adda di Fagagna, F. Cellular Senescence in Ageing: From Mechanisms to Therapeutic Opportunities. Nat. Rev. Mol. Cell Biol. 2021, 22, 75–95. [Google Scholar] [CrossRef] [PubMed]
- Baar, M.P.; Perdiguero, E.; Muñoz-Cánoves, P.; de Keizer, P.L. Musculoskeletal Senescence: A Moving Target Ready to Be Eliminated. Curr. Opin. Pharmacol. 2018, 40, 147–155. [Google Scholar] [CrossRef] [PubMed]
- Lotz, M.; Loeser, R.F. Effects of Aging on Articular Cartilage Homeostasis. Bone 2012, 51, 241–248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, L.-R.; Marston, G.; Martin, A. Anatomy, Cartilage; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Hjelle, K.; Solheim, E.; Strand, T.; Muri, R.; Brittberg, M. Articular Cartilage Defects in 1000 Knee Arthroscopies. Arthroscopy 2002, 18, 730–734. [Google Scholar] [CrossRef] [PubMed]
- Flanigan, D.C.; Harris, J.D.; Trinh, T.Q.; Siston, R.A.; Brophy, R.H. Prevalence of Chondral Defects in Athletes’ Knees: A Systematic Review: A Systematic Review. Med. Sci. Sports Exerc. 2010, 42, 1795–1801. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A.S.; Loeser, R.F. Why Is Osteoarthritis an Age-Related Disease? Best Pract. Res. Clin. Rheumatol. 2010, 24, 15–26. [Google Scholar] [CrossRef] [Green Version]
- Cieza, A.; Causey, K.; Kamenov, K.; Hanson, S.W.; Chatterji, S.; Vos, T. Global Estimates of the Need for Rehabilitation Based on the Global Burden of Disease Study 2019: A Systematic Analysis for the Global Burden of Disease Study 2019. Lancet 2021, 396, 2006–2017. [Google Scholar] [CrossRef]
- World Population Ageing. Highlights. 2020. Available online: https://www.un.org/development/desa/pd/sites/www.un.org.development.desa.pd/files/files/documents/2020/Sep/un_pop_2020_pf_ageing_10_key_messages.pdf (accessed on 14 July 2021).
- Medvedeva, E.V.; Grebenik, E.A.; Gornostaeva, S.N.; Telpuhov, V.I.; Lychagin, A.V.; Timashev, P.S.; Chagin, A.S. Repair of Damaged Articular Cartilage: Current Approaches and Future Directions. Int. J. Mol. Sci. 2018, 19, 2366. [Google Scholar] [CrossRef] [Green Version]
- Pogliacomi, F.; Schiavi, P.; Paraskevopoulos, A.; Leigheb, M.; Pedrazzini, A.; Ceccarelli, F.; Vaienti, E. When Is Indicated Viscosupplementation in Hip Osteoarthritis? Acta Biomed. 2018, 90, 67–74. [Google Scholar]
- Leigheb, M.; Bosetti, M.; de Consoli, A.; Borrone, A.; Cannas, M.; Grassi, F. Chondral Tissue Engineering of the Reumatoid Knee with Collagen Matrix Autologous Chondrocytes Implant. Acta Biomed. 2017, 88, 107–113. [Google Scholar]
- Moreira-Teixeira, L.S.; Georgi, N.; Leijten, J.; Wu, L.; Karperien, M. Cartilage Tissue Engineering. Endocr. Dev. 2011, 21, 102–115. [Google Scholar] [CrossRef] [PubMed]
- Sophia Fox, A.J.; Bedi, A.; Rodeo, S.A. The Basic Science of Articular Cartilage: Structure, Composition, and Function. Sports Health 2009, 1, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Thorp, H.; Kim, K.; Kondo, M.; Maak, T.; Grainger, D.W.; Okano, T. Trends in Articular Cartilage Tissue Engineering: 3D Mesenchymal Stem Cell Sheets as Candidates for Engineered Hyaline-like Cartilage. Cells 2021, 10, 643. [Google Scholar] [CrossRef]
- International Cartilage Repair Society (ICRS) Cartilage Injury Evaluation Package. Available online: https://cartilage.org/content/uploads/2014/10/ICRS_evaluation.pdf (accessed on 18 October 2021).
- Loeser, R.F. Aging and Osteoarthritis: The Role of Chondrocyte Senescence and Aging Changes in the Cartilage Matrix. Osteoarthr. Cartil. 2009, 17, 971–979. [Google Scholar] [CrossRef] [Green Version]
- Loeser, R.F.; Yammani, R.R.; Carlson, C.S.; Chen, H.; Cole, A.; Im, H.-J.; Bursch, L.S.; Yan, S.D. Articular Chondrocytes Express the Receptor for Advanced Glycation End Products: Potential Role in Osteoarthritis. Arthritis Rheum. 2005, 52, 2376–2385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cecil, D.L.; Johnson, K.; Rediske, J.; Lotz, M.; Schmidt, A.M.; Terkeltaub, R. Inflammation-Induced Chondrocyte Hypertrophy Is Driven by Receptor for Advanced Glycation End Products. J. Immunol. 2005, 175, 8296–8302. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yammani, R.R.; Carlson, C.S.; Bresnick, A.R.; Loeser, R.F. Increase in Production of Matrix Metalloproteinase 13 by Human Articular Chondrocytes Due to Stimulation with S100A4: Role of the Receptor for Advanced Glycation End Products. Arthritis Rheum. 2006, 54, 2901–2911. [Google Scholar] [CrossRef] [PubMed]
- Mitsuyama, H.; Healey, R.M.; Terkeltaub, R.A.; Coutts, R.D.; Amiel, D. Calcification of Human Articular Knee Cartilage is Primarily an Effect of Aging Rather than Osteoarthritis. Osteoarthr. Cartil. 2007, 15, 559–565. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, A.C.; Temple, M.M.; Ng, D.M.; Verzijl, N.; DeGroot, J.; TeKoppele, J.M.; Sah, R.L. Induction of Advanced Glycation End Products and Alterations of the Tensile Properties of Articular Cartilage. Arthritis Rheum. 2002, 46, 3212–3217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwon, H.; Brown, W.E.; Lee, C.A.; Wang, D.; Paschos, N.; Hu, J.C.; Athanasiou, K.A. Surgical and Tissue Engineering Strategies for Articular Cartilage and Meniscus Repair. Nat. Rev. Rheumatol. 2019, 15, 550–570. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Hu, D.A.; Wu, D.; He, F.; Wang, H.; Huang, L.; Shi, D.; Liu, Q.; Ni, N.; Pakvasa, M.; et al. Applications of Biocompatible Scaffold Materials in Stem Cell-Based Cartilage Tissue Engineering. Front. Bioeng. Biotechnol. 2021, 9, 603444. [Google Scholar] [CrossRef]
- Johnson, K.; Zhu, S.; Tremblay, M.S.; Payette, J.N.; Wang, J.; Bouchez, L.C.; Meeusen, S.; Althage, A.; Cho, C.Y.; Wu, X.; et al. A Stem Cell-Based Approach to Cartilage Repair. Science 2012, 336, 717–721. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Teng, B.; Zhang, S.; Pan, J.; Zeng, Z.; Chen, Y.; Hei, Y.; Fu, X.; Li, Q.; Ma, M.; Sui, Y.; et al. A Chondrogenesis Induction System Based on a Functionalized Hyaluronic Acid Hydrogel Sequentially Promoting HMSC Proliferation, Condensation, Differentiation, and Matrix Deposition. Acta Biomater. 2021, 122, 145–159. [Google Scholar] [CrossRef]
- Shen, H.; Lin, H.; Sun, A.X.; Song, S.; Wang, B.; Yang, Y.; Dai, J.; Tuan, R.S. Acceleration of Chondrogenic Differentiation of Human Mesenchymal Stem Cells by Sustained Growth Factor Release in 3D Graphene Oxide Incorporated Hydrogels. Acta Biomater. 2020, 105, 44–55. [Google Scholar] [CrossRef]
- Nürnberger, S.; Schneider, C.; Keibl, C.; Schädl, B.; Heimel, P.; Monforte, X.; Teuschl, A.H.; Nalbach, M.; Thurner, P.J.; Grillari, J.; et al. Repopulation of Decellularised Articular Cartilage by Laser-Based Matrix Engraving. EBioMedicine 2021, 64, 103196. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-K.; Bae, H.C.; Ro, D.H.; Lee, S.; Lee, M.C.; Han, H.-S. Enhancement of Cartilage Regeneration of Synovial Stem Cells/Hydrogel by Using Transglutaminase-4. Tissue Eng. Part A 2021, 27, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Dang, Q.T.; Huynh, T.D.; Inchingolo, F.; Dipalma, G.; Inchingolo, A.D.; Cantore, S.; Paduanelli, G.; Nguyen, K.C.D.; Ballini, A.; Isacco, C.G.; et al. Human Chondrocytes from Human Adipose Tissue-Derived Mesenchymal Stem Cells Seeded on a Dermal-Derived Collagen Matrix Sheet: Our Preliminary Results for a Ready to Go Biotechnological Cartilage Graft in Clinical Practice. Stem Cells Int. 2021, 2021, 6664697. [Google Scholar] [CrossRef]
- Özdemir, E.; Emet, A.; Hashemihesar, R.; Yürüker, A.C.S.; Kılıç, E.; Uçkan Çetinkaya, D.U.; Turhan, E. Articular Cartilage Regeneration Utilizing Decellularized Human Placental Scaffold, Mesenchymal Stem Cells and Platelet Rich Plasma. Tissue Eng. Regen. Med. 2020, 17, 901–908. [Google Scholar] [CrossRef] [PubMed]
- Barlian, A.; Judawisastra, H.; Ridwan, A.; Wahyuni, A.R.; Lingga, M.E. Chondrogenic Differentiation of Wharton’s Jelly Mesenchymal Stem Cells on Silk Spidroin-Fibroin Mix Scaffold Supplemented with L-Ascorbic Acid and Platelet Rich Plasma. Sci. Rep. 2020, 10, 19449. [Google Scholar] [CrossRef] [PubMed]
- Fan, G.-W.; Gao, X.-M.; Wang, H.; Zhu, Y.; Zhang, J.; Hu, L.-M.; Su, Y.-F.; Kang, L.-Y.; Zhang, B.-L. The Anti-Inflammatory Activities of Tanshinone IIA, an Active Component of TCM, Are Mediated by Estrogen Receptor Activation and Inhibition of INOS. J. Steroid Biochem. Mol. Biol. 2009, 113, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Zhu, H.; Chen, Z.; Ma, Z.; Tan, H.; Xiao, C.; Tang, X.; Zhang, B.; Wang, Y.; Gao, Y. Tanshinone IIA Protects Endothelial Cells from H₂O₂-Induced Injuries via PXR Activation. Biomol. Ther. 2017, 25, 599–608. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fu, J.; Huang, H.; Liu, J.; Pi, R.; Chen, J.; Liu, P. Tanshinone IIA Protects Cardiac Myocytes against Oxidative Stress-Triggered Damage and Apoptosis. Eur. J. Pharmacol. 2007, 568, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Xu, Y.; Li, H.; Dai, Y.; Zhou, G.; Zhou, Z.; Xia, H.; Liu, H. Tanshinone IIA Delivery Silk Fibroin Scaffolds Significantly Enhance Articular Cartilage Defect Repairing via Promoting Cartilage Regeneration. ACS Appl. Mater. Interfaces 2020, 12, 21470–21480. [Google Scholar] [CrossRef] [PubMed]
- Rubí-Sans, G.; Recha-Sancho, L.; Pérez-Amodio, S.; Mateos-Timoneda, M.Á.; Semino, C.E.; Engel, E. Development of a Three-Dimensional Bioengineered Platform for Articular Cartilage Regeneration. Biomolecules 2019, 10, 52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dufour, A.; Lafont, J.E.; Buffier, M.; Verset, M.; Cohendet, A.; Contamin, H.; Confais, J.; Sankar, S.; Rioult, M.; Perrier-Groult, E.; et al. Repair of Full-Thickness Articular Cartilage Defects Using IEIK13 Self-Assembling Peptide Hydrogel in a Non-Human Primate Model. Sci. Rep. 2021, 11, 4560. [Google Scholar] [CrossRef] [PubMed]
- Scalzone, A.; Ferreira, A.M.; Tonda-Turo, C.; Ciardelli, G.; Dalgarno, K.; Gentile, P. The Interplay between Chondrocyte Spheroids and Mesenchymal Stem Cells Boosts Cartilage Regeneration within a 3D Natural-Based Hydrogel. Sci. Rep. 2019, 9, 14630. [Google Scholar] [CrossRef] [Green Version]
- Duan, W.; Zhao, Y.; Ren, X.; Zhao, R.; Li, Q.; Sun, Z.; Song, W.; Yang, Y.; Li, P.; Wei, X. Combination of Chondrocytes and Chondrons Improves Extracellular Matrix Production to Promote the Repairs of Defective Knee Cartilage in Rabbits. J. Orthop. Translat. 2021, 28, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Owida, H.A.; de Las Heras Ruiz, T.; Dhillon, A.; Yang, Y.; Kuiper, N.J. Co-Culture of Chondrons and Mesenchymal Stromal Cells Reduces the Loss of Collagen VI and Improves Extracellular Matrix Production. Histochem. Cell Biol. 2017, 148, 625–638. [Google Scholar] [CrossRef]
- Castilho, M.; Mouser, V.; Chen, M.; Malda, J.; Ito, K. Bi-Layered Micro-Fibre Reinforced Hydrogels for Articular Cartilage Regeneration. Acta Biomater. 2019, 95, 297–306. [Google Scholar] [CrossRef] [PubMed]
- Zhu, D.; Tong, X.; Trinh, P.; Yang, F. Mimicking Cartilage Tissue Zonal Organization by Engineering Tissue-Scale Gradient Hydrogels as 3D Cell Niche. Tissue Eng. Part A 2018, 24, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Steele, J.A.M.; McCullen, S.D.; Callanan, A.; Autefage, H.; Accardi, M.A.; Dini, D.; Stevens, M.M. Combinatorial Scaffold Morphologies for Zonal Articular Cartilage Engineering. Acta Biomater. 2014, 10, 2065–2075. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cochis, A.; Bonetti, L.; Sorrentino, R.; Negrini, N.C.; Grassi, F.; Leigheb, M.; Rimondini, L.; Farè, S. 3D Printing of Thermo-Responsive Methylcellulose Hydrogels for Cell-Sheet Engineering. Materials 2018, 11, 579. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Altomare, L.; Cochis, A.; Carletta, A.; Rimondini, L.; Farè, S. Thermo-Responsive Methylcellulose Hydrogels as Temporary Substrate for Cell Sheet Biofabrication. J. Mater. Sci. Mater. Med. 2016, 27, 95. [Google Scholar] [CrossRef] [PubMed]
- Wongin, S.; Wangdee, C.; Nantavisai, S.; Banlunara, W.; Nakbunnum, R.; Waikakul, S.; Chotiyarnwong, P.; Roytrakul, S.; Viravaidya-Pasuwat, K. Evaluation of Osteochondral-Like Tissues Using Human Freeze-Dried Cancellous Bone and Chondrocyte Sheets to Treat Osteochondral Defects in Rabbits. Biomater. Sci. 2021, 9, 4701–4716. [Google Scholar] [CrossRef]
- Takizawa, D.; Sato, M.; Okada, E.; Takahashi, T.; Maehara, M.; Tominaga, A.; Sogo, Y.; Toyoda, E.; Watanabe, M. Regenerative Effects of Human Chondrocyte Sheets in a Xenogeneic Transplantation Model Using Immune-Deficient Rats. J. Tissue Eng. Regen. Med. 2020, 14, 1296–1306. [Google Scholar] [CrossRef] [PubMed]
- Thorp, H.; Kim, K.; Kondo, M.; Grainger, D.W.; Okano, T. Fabrication of Hyaline-Like Cartilage Constructs Using Mesenchymal Stem Cell Sheets. Sci. Rep. 2020, 10, 20869. [Google Scholar] [CrossRef] [PubMed]
- You, Q.; Liu, Z.; Zhang, J.; Shen, M.; Li, Y.; Jin, Y.; Liu, Y. Human Amniotic Mesenchymal Stem Cell Sheets Encapsulating Cartilage Particles Facilitate Repair of Rabbit Osteochondral Defects. Am. J. Sports Med. 2020, 48, 599–611. [Google Scholar] [CrossRef] [PubMed]
- Wasai, S.; Toyoda, E.; Takahashi, T.; Maehara, M.; Okada, E.; Uchiyama, R.; Akamatsu, T.; Watanabe, M.; Sato, M. Development of Injectable Polydactyly-Derived Chondrocyte Sheets. Int. J. Mol. Sci. 2021, 22, 3198. [Google Scholar] [CrossRef] [PubMed]
- Takagi, T.; Kabata, T.; Hayashi, K.; Fang, X.; Kajino, Y.; Inoue, D.; Ohmori, T.; Ueno, T.; Yoshitani, J.; Ueoka, K.; et al. Periodic Injections of Adipose-Derived Stem Cell Sheets Attenuate Osteoarthritis Progression in an Experimental Rabbit Model. BMC Musculoskelet. Disord. 2020, 21, 691. [Google Scholar] [CrossRef]
- Köhnke, R.; Ahlers, M.O.; Birkelbach, M.A.; Ewald, F.; Krueger, M.; Fiedler, I.; Busse, B.; Heiland, M.; Vollkommer, T.; Gosau, M.; et al. Temporomandibular Joint Osteoarthritis: Regenerative Treatment by a Stem Cell Containing Advanced Therapy Medicinal Product (ATMP)-an in Vivo Animal Trial. Int. J. Mol. Sci. 2021, 22, 443. [Google Scholar] [CrossRef] [PubMed]
- Qu, M.; Liao, X.; Jiang, N.; Sun, W.; Xiao, W.; Zhou, X.; Khademhosseini, A.; Li, B.; Zhu, S. Injectable Open-Porous PLGA Microspheres as Cell Carriers for Cartilage Regeneration. J. Biomed. Mater. Res. Part A 2021, 109, 2091–2100. [Google Scholar] [CrossRef] [PubMed]
- Prasadam, I.; Akuien, A.; Friis, T.E.; Fang, W.; Mao, X.; Crawford, R.W.; Xiao, Y. Mixed Cell Therapy of Bone Marrow-Derived Mesenchymal Stem Cells and Articular Cartilage Chondrocytes Ameliorates Osteoarthritis Development. Lab. Investig. 2018, 98, 106–116. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Co, C.M.; Izuagbe, S.; Zhou, J.; Zhou, N.; Sun, X.; Borrelli, J.; Tang, L. Click Chemistry-Based Pre-Targeting Cell Delivery for Cartilage Regeneration. Regen. Biomater. 2021, 8, rbab018. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Liang, Y.; Li, X.; Ouyang, K.; Wang, M.; Cao, T.; Li, W.; Liu, J.; Xiong, J.; Li, B.; et al. Exosome-Mediated Delivery of Kartogenin for Chondrogenesis of Synovial Fluid-Derived Mesenchymal Stem Cells and Cartilage Regeneration. Biomaterials 2021, 269, 120539. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Li, H.; Xiang, D.; Shao, J.; Fu, Q.; Han, Y.; Zhu, J.; Chen, Y.; Qian, Q. The Clinical Efficacy of Arthroscopic Therapy with Knee Infrapatellar Fat Pad Cell Concentrates in Treating Knee Cartilage Lesion: A Prospective, Randomized, and Controlled Study. J. Orthop. Surg. Res. 2021, 16, 87. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Z.; Tang, J.; Yue, B.; Wang, J.; Zhang, J.; Xuan, L.; Dai, C.; Li, S.; Li, M.; Xu, C.; et al. Human Adipose-Derived Mesenchymal Progenitor Cells plus Microfracture and Hyaluronic Acid for Cartilage Repair: A Phase IIa Trial. Regen. Med. 2020, 15, 1193–1214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, Y.S.; Chung, P.K.; Suh, D.S.; Heo, D.B.; Tak, D.H.; Koh, Y.G. Implantation of Mesenchymal Stem Cells in Combination with Allogenic Cartilage Improves Cartilage Regeneration and Clinical Outcomes in Patients with Concomitant High Tibial Osteotomy. Knee Surg. Sports Traumatol. Arthrosc. 2020, 28, 544–554. [Google Scholar] [CrossRef]
- Garza, J.R.; Campbell, R.E.; Tjoumakaris, F.P.; Freedman, K.B.; Miller, L.S.; Santa Maria, D.; Tucker, B.S. Clinical Efficacy of Intra-Articular Mesenchymal Stromal Cells for the Treatment of Knee Osteoarthritis: A Double-Blinded Prospective Randomized Controlled Clinical Trial. Am. J. Sports Med. 2020, 48, 588–598. [Google Scholar] [CrossRef]
- Lu, L.; Dai, C.; Du, H.; Li, S.; Ye, P.; Zhang, L.; Wang, X.; Song, Y.; Togashi, R.; Vangsness, C.T.; et al. Intra-Articular Injections of Allogeneic Human Adipose-Derived Mesenchymal Progenitor Cells in Patients with Symptomatic Bilateral Knee Osteoarthritis: A Phase I Pilot Study. Regen. Med. 2020, 15, 1625–1636. [Google Scholar] [CrossRef] [PubMed]
- Yoon, K.-H.; Park, J.-Y.; Lee, J.-Y.; Lee, E.; Lee, J.; Kim, S.-G. Costal Chondrocyte-Derived Pellet-Type Autologous Chondrocyte Implantation for Treatment of Articular Cartilage Defect. Am. J. Sports Med. 2020, 48, 1236–1245. [Google Scholar] [CrossRef]
- Cochis, A.; Grad, S.; Stoddart, M.J.; Farè, S.; Altomare, L.; Azzimonti, B.; Alini, M.; Rimondini, L. Bioreactor Mechanically Guided 3D Mesenchymal Stem Cell Chondrogenesis Using a Biocompatible Novel Thermo-Reversible Methylcellulose-Based Hydrogel. Sci. Rep. 2017, 7, 45018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szychlinska, M.A.; Calabrese, G.; Ravalli, S.; Dolcimascolo, A.; Castrogiovanni, P.; Fabbi, C.; Puglisi, C.; Lauretta, G.; di Rosa, M.; Castorina, A.; et al. Evaluation of a Cell-Free Collagen Type I-Based Scaffold for Articular Cartilage Regeneration in an Orthotopic Rat Model. Materials 2020, 13, 2369. [Google Scholar] [CrossRef] [PubMed]
- Gavenis, K.; Schneider, U.; Maus, U.; Mumme, T.; Muller-Rath, R.; Schmidt-Rohlfing, B.; Andereya, S. Cell-Free Repair of Small Cartilage Defects in the Goettinger Minipig: Which Defect Size Is Possible? Knee Surg. Sports Traumatol. Arthrosc. 2012, 20, 2307–2314. [Google Scholar] [CrossRef] [PubMed]
- Schneider, U.; Schmidt-Rohlfing, B.; Gavenis, K.; Maus, U.; Mueller-Rath, R.; Andereya, S. A Comparative Study of 3 Different Cartilage Repair Techniques. Knee Surg. Sports Traumatol. Arthrosc. 2011, 19, 2145–2152. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhang, X.; Hong, Y.; Fu, Q.; He, Q.; Mechakra, A.; Zhu, Q.; Zhou, F.; Liang, R.; Li, C.; et al. Tissue-Adhesive Paint of Silk Microparticles for Articular Surface Cartilage Regeneration. ACS Appl. Mater. Interfaces 2020, 12, 22467–22478. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Han, L.; Sun, T.; Ma, J.; Sun, S.; Ma, L.; Wu, B. Extracellular Matrix Derived from Allogenic Decellularized Bone Marrow Mesenchymal Stem Cell Sheets for the Reconstruction of Osteochondral Defects in Rabbits. Acta Biomater. 2020, 118, 54–68. [Google Scholar] [CrossRef]
- Dai, Y.; Shen, T.; Ma, L.; Wang, D.; Gao, C. Regeneration of Osteochondral Defects in Vivo by a Cell-Free Cylindrical Poly (Lactide-Co-Glycolide) Scaffold with a Radially Oriented Microstructure. J. Tissue Eng. Regen. Med. 2018, 12, e1647–e1661. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Lock, J.; Sallee, A.; Liu, H. Magnetic Nanocomposite Hydrogel for Potential Cartilage Tissue Engineering: Synthesis, Characterization, and Cytocompatibility with Bone Marrow Derived Mesenchymal Stem Cells. ACS Appl. Mater. Interfaces 2015, 7, 20987–20998. [Google Scholar] [CrossRef] [PubMed]
- Lebourg, M.; Martínez-Díaz, S.; García-Giralt, N.; Torres-Claramunt, R.; Gómez-Tejedor, J.A.; Ribelles, J.L.G.; Vila-Canet, G.; Monllau, J.C. Cell-Free Cartilage Engineering Approach Using Hyaluronic Acid-Polycaprolactone Scaffolds: A Study in Vivo. J. Biomater. Appl. 2014, 28, 1304–1315. [Google Scholar] [CrossRef] [PubMed]
- Milner, P.E.; Parkes, M.; Puetzer, J.L.; Chapman, R.; Stevens, M.M.; Cann, P.; Jeffers, J.R.T. A Low Friction, Biphasic and Boundary Lubricating Hydrogel for Cartilage Replacement. Acta Biomater. 2018, 65, 102–111. [Google Scholar] [CrossRef]
- Lolli, A.; Sivasubramaniyan, K.; Vainieri, M.L.; Oieni, J.; Kops, N.; Yayon, A.; van Osch, G.J.V.M. Hydrogel-Based Delivery of AntimiR-221 Enhances Cartilage Regeneration by Endogenous Cells. J. Control. Release 2019, 309, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Brouillette, M.J.; Seol, D.; Zheng, H.; Buckwalter, J.A.; Martin, J.A. Use of Recombinant Human Stromal Cell-Derived Factor 1α-Loaded Fibrin/Hyaluronic Acid Hydrogel Networks to Achieve Functional Repair of Full-Thickness Bovine Articular Cartilage via Homing of Chondrogenic Progenitor Cells: Sdf-1α for Articular Cartilage Repair upon Chondrogenic Induction. Arthritis rheumatol. 2015, 67, 1274–1285. [Google Scholar] [PubMed]
- Crecente-Campo, J.; Borrajo, E.; Vidal, A.; Garcia-Fuentes, M. New Scaffolds Encapsulating TGF-Β3/BMP-7 Combinations Driving Strong Chondrogenic Differentiation. Eur. J. Pharm. Biopharm. 2017, 114, 69–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, I.L.; Pfeifer, C.G.; Fisher, M.B.; Saxena, V.; Meloni, G.R.; Kwon, M.Y.; Kim, M.; Steinberg, D.R.; Mauck, R.L.; Burdick, J.A. Fibrous Scaffolds with Varied Fiber Chemistry and Growth Factor Delivery Promote Repair in a Porcine Cartilage Defect Model. Tissue Eng. Part A 2015, 21, 2680–2690. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, C.H.; Cook, J.L.; Mendelson, A.; Moioli, E.K.; Yao, H.; Mao, J.J. Regeneration of the Articular Surface of the Rabbit Synovial Joint by Cell Homing: A Proof of Concept Study. Lancet 2010, 376, 440–448. [Google Scholar] [CrossRef] [Green Version]
- Lu, S.; Lam, J.; Trachtenberg, J.E.; Lee, E.J.; Seyednejad, H.; van den Beucken, J.J.J.P.; Tabata, Y.; Wong, M.E.; Jansen, J.A.; Mikos, A.G.; et al. Dual Growth Factor Delivery from Bilayered, Biodegradable Hydrogel Composites for Spatially-Guided Osteochondral Tissue Repair. Biomaterials 2014, 35, 8829–8839. [Google Scholar] [CrossRef] [Green Version]
- Re’em, T.; Witte, F.; Willbold, E.; Ruvinov, E.; Cohen, S. Simultaneous Regeneration of Articular Cartilage and Subchondral Bone Induced by Spatially Presented TGF-Beta and BMP-4 in a Bilayer Affinity Binding System. Acta Biomater. 2012, 8, 3283–3293. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.; Tian, G.; Yang, Z.; Gao, X.; Wang, F.; Li, J.; Tian, Z.; Huang, B.; Wei, F.; Sang, X.; et al. Enhancement of Acellular Cartilage Matrix Scaffold by Wharton’s Jelly Mesenchymal Stem Cell-Derived Exosomes to Promote Osteochondral Regeneration. Bioact. Mater. 2021, 6, 2711–2728. [Google Scholar] [CrossRef] [PubMed]
- Yuan, T.; Li, Z.; Zhang, Y.; Shen, K.; Zhang, X.; Xie, R.; Liu, F.; Fan, W. Injectable Ultrasonication-Induced Silk Fibroin Hydrogel for Cartilage Repair and Regeneration. Tissue Eng. Part A 2021, 27, 1213–1224. [Google Scholar] [CrossRef]
- Schaeffer, C.; Pfaff, B.N.; Cornell, N.J.; Salopek, L.S.; Shan, S.; Viyar, J.; Omesiete, W.; Griffin, D.R.; Cottler, P.S.; DeGeorge, B.R., Jr. Injectable Microannealed Porous Scaffold for Articular Cartilage Regeneration. Ann. Plast. Surg. 2020, 84, S446–S450. [Google Scholar] [CrossRef]
- Buchtová, N.; Réthoré, G.; Boyer, C.; Guicheux, J.; Rambaud, F.; Vallé, K.; Belleville, P.; Sanchez, C.; Chauvet, O.; Weiss, P.; et al. Nanocomposite Hydrogels for Cartilage Tissue Engineering: Mesoporous Silica Nanofibers Interlinked with Siloxane Derived Polysaccharide. J. Mater. Sci. Mater. Med. 2013, 24, 1875–1884. [Google Scholar] [CrossRef] [PubMed]
- Tang, Q.; Lim, T.; Shen, L.-Y.; Zheng, G.; Wei, X.-J.; Zhang, C.-Q.; Zhu, Z.-Z. Well-Dispersed Platelet Lysate Entrapped Nanoparticles Incorporate with Injectable PDLLA-PEG-PDLLA Triblock for Preferable Cartilage Engineering Application. Biomaterials 2021, 268, 120605. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Shen, L.; Zhu, Z.; Luo, X.; Zhai, Y.; Hua, X.; Zhao, S.; Cen, L.; Zhang, Z. A Cell-Free Therapy for Articular Cartilage Repair Based on Synergistic Delivery of SDF-1 & KGN with HA Injectable Scaffold. Chem. Eng. J. 2020, 393, 124649. [Google Scholar]
- Cole, B.J.; Haunschild, E.D.; Carter, T.; Meyer, J.; Fortier, L.A.; Gilat, R.; BC (BioCartilage) Study Group. Clinically Significant Outcomes Following the Treatment of Focal Cartilage Defects of the Knee with Microfracture Augmentation Using Cartilage Allograft Extracellular Matrix: A Multicenter Prospective Study. Arthroscopy 2021, 37, 1512–1521. [Google Scholar] [CrossRef] [PubMed]
- Wolf, M.T.; Zhang, H.; Sharma, B.; Marcus, N.A.; Pietzner, U.; Fickert, S.; Lueth, A.; Albers, G.H.R.; Elisseeff, J.H. Two-Year Follow-up and Remodeling Kinetics of ChonDux Hydrogel for Full-Thickness Cartilage Defect Repair in the Knee. Cartilage 2020, 11, 447–457. [Google Scholar] [CrossRef]
- Lee, Y.K.; Young, K.W.; Kim, J.S.; Lee, H.S.; Cho, W.-J.; Kim, H.N. Arthroscopic Microfracture with Atelocollagen Augmentation for Osteochondral Lesion of the Talus: A Multicenter Randomized Controlled Trial. BMC Musculoskelet. Disord. 2020, 21, 716. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Chun, C.H.; Wang, J.H.; Kim, J.G.; Kang, S.-B.; Yoo, J.D.; Chon, J.-G.; Kim, M.K.; Moon, C.W.; Chang, C.B.; et al. Microfractures versus a Porcine-Derived Collagen-Augmented Chondrogenesis Technique for Treating Knee Cartilage Defects: A Multicenter Randomized Controlled Trial. Arthroscopy 2020, 36, 1612–1624. [Google Scholar] [CrossRef] [PubMed]
- Roessler, P.P.; Pfister, B.; Gesslein, M.; Figiel, J.; Heyse, T.J.; Colcuc, C.; Lorbach, O.; Efe, T.; Schüttler, K.F. Short-Term Follow up after Implantation of a Cell-Free Collagen Type I Matrix for the Treatment of Large Cartilage Defects of the Knee. Int. Orthop. 2015, 39, 2473–2479. [Google Scholar] [CrossRef] [PubMed]
- Schüttler, K.-F.; Götschenberg, A.; Klasan, A.; Stein, T.; Pehl, A.; Roessler, P.P.; Figiel, J.; Heyse, T.J.; Efe, T. Cell-Free Cartilage Repair in Large Defects of the Knee: Increased Failure Rate 5 Years after Implantation of a Collagen Type I Scaffold. Arch. Orthop. Trauma Surg. 2019, 139, 99–106. [Google Scholar] [CrossRef]
- Efe, T.; Theisen, C.; Fuchs-Winkelmann, S.; Stein, T.; Getgood, A.; Rominger, M.B.; Paletta, J.R.J.; Schofer, M.D. Cell-Free Collagen Type I Matrix for Repair of Cartilage Defects-Clinical and Magnetic Resonance Imaging Results. Knee Surg. Sports Traumatol. Arthrosc. 2012, 20, 1915–1922. [Google Scholar] [CrossRef]
- Schüttler, K.F.; Schenker, H.; Theisen, C.; Schofer, M.D.; Getgood, A.; Roessler, P.P.; Struewer, J.; Rominger, M.B.; Efe, T. Use of Cell-Free Collagen Type I Matrix Implants for the Treatment of Small Cartilage Defects in the Knee: Clinical and Magnetic Resonance Imaging Evaluation. Knee Surg. Sports Traumatol. Arthrosc. 2014, 22, 1270–1276. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Maffulli, N.; Rodriguez, H.C.; Lee, C.E.; Levy, H.J.; El-Amin, S.F., III. Umbilical Cord-Derived Wharton’s Jelly for Treatment of Knee Osteoarthritis: Study Protocol for a Non-Randomized, Open-Label, Multi-Center Trial. J. Orthop. Surg. Res. 2021, 16, 143. [Google Scholar] [CrossRef] [PubMed]
- Slattery, C.; Kweon, C.Y. Classifications in Brief: Outerbridge Classification of Chondral Lesions. Clin. Orthop. Relat. Res. 2018, 476, 2101–2104. [Google Scholar] [CrossRef] [PubMed]
- Leigheb, M.; Guzzardi, G.; Barini, M.; Abruzzese, M.; Riva, S.; Paschè, A.; Pogliacomi, F.; Rimondini, L.; Stecco, A.; Grassi, F.A.; et al. Role of Low Field MRI in Detecting Knee Lesions. Acta Biomed. 2018, 90, 116–122. [Google Scholar] [PubMed]
- Jiang, S.; Guo, W.; Tian, G.; Luo, X.; Peng, L.; Liu, S.; Sui, X.; Guo, Q.; Li, X. Clinical Application Status of Articular Cartilage Regeneration Techniques: Tissue-Engineered Cartilage Brings New Hope. Stem Cells Int. 2020, 2020, 5690252. [Google Scholar] [CrossRef] [PubMed]
- Vinatier, C.; Guicheux, J. Cartilage Tissue Engineering: From Biomaterials and Stem Cells to Osteoarthritis Treatments. Ann. Phys. Rehabil. Med. 2016, 59, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Kwan, H.; Chisari, E.; Khan, W.S. Cell-Free Scaffolds as a Monotherapy for Focal Chondral Knee Defects. Materials 2020, 13, 306. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Cell-Based Tissue Engineering Strategies for Cartilage Defects | Ref. | ||
|---|---|---|---|
| 01 | Scaffold-Based Strategies | MeHA/RGD/HAV + BM-MSCs + KGN in PLGA microspheres | [25] |
| PDLLA/GO nanosheets + BM-MSCs + TGF-β3 | [26] | ||
| Human dermal-derived collagen + AD-MSCs + collagen substrates | [29] | ||
| SS/SF scaffold + hWJ-MSCs + LAA or PRP | [31] | ||
| SF scaffold + chondrocytes + TAN | [32] | ||
| PCL/RAD16-I + BM-MSCs | [33] | ||
| Fibrin/IEIK13 ± chondrocytes | [34] | ||
| Chitosan/BGP + BM-MSCs/chondrocytes | [35] | ||
| GelMA with bilayered architecture + chondrocytes + TGF-ß1 or mechanical stimulation | [38] | ||
| Gradient PEG-norbornene/PEG-dithiol/CS-MA + chondrocytes or MSCs | [39] | ||
| Bilayered PCL scaffold/porogens + chondrocytes | [40] | ||
| 02 | Scaffold-Free Strategies | Pre-differentiated BM-MSCs cell sheets | [45] |
| 03 | Injectables | Allogeneic PD cell sheet fragments | [47] |
| Autologous AD-MSCs cell sheets | [48] | ||
| 04 | Clinical Studies | IPFP cell concentrates | [54] |
| ACI + CCP | [59] |
| Cell-Based Tissue Engineering Strategies for Cartilage Defects Associated with Osteochondral Defects and Osteoarthritis (OA) | Ref. | ||
|---|---|---|---|
| 01 | Scaffold-Based Strategies | Human articular cartilage ECM + AD-MSCs/chondrocytes | [27] |
| HA/collagen/fibrinogen + SMSCs + rhTG-4 | [28] | ||
| Human placenta + BM-MSCs ± PRP | [30] | ||
| Alginate spheres + chondrocytes/chondrons | [36] | ||
| 02 | Scaffold-Free Strategies | Human freeze-dried cancellous bone + human chondrocyte sheets | [43] |
| Human chondrocytes ± human synoviocytes | [44] | ||
| hAMSCs cell sheets + cartilage particles | [46] | ||
| 03 | Injectables | AD-MSCs ± HA | [49] |
| Open-porous PLGA microspheres + BM-MSCs | [50] | ||
| MSCs + chondrocytes | [51] | ||
| PEG/ApoPep-1/TCO + chondrocytes | [52] | ||
| E7-Exo + SF-MSCs + KGN | [53] | ||
| 04 | Clinical Studies | Microfracture ± HA ± haMPCs | [55] |
| AD-MSCs ± allogeneic cartilage from fresh cadavers | [56] | ||
| Intra-articular injection of autologous SVF | [57] | ||
| Intra-articular injections of allogeneic haMPCs | [58] |
| Cell-Free Tissue Engineering Strategies for Cartilage Defects | Ref. | ||
|---|---|---|---|
| 01 | Scaffold-Based Strategies | Col-1-based scaffolds | [61,62,63] |
| Col-2/HA/PEG/magnetic nanoparticles | [67] | ||
| HA/PCL scaffolds | [68] | ||
| PMPC/DN biphasic gel | [69] | ||
| Fibrin/HA + rhSDF-1α | [71] | ||
| PLGA + TGF-β3 + BMP-7 | [72] | ||
| Modified HA fibers/PCL fibers + TGF-β3 + microfracture | [73] | ||
| 02 | Injectables | Si-HPMC/mesoporous silica nanofibers | [80] |
| HA hydrogel + PLGA microspheres co-encapsulating KGN and SDF-1 | [82] | ||
| 03 | Clinical Studies | Commercial cartilage allograft ECM + microfracture | [83] |
| Photoreactive chondroitin-sulfate/PEG HA hydrogel + microfracture | [84] |
| Cell-Based Tissue Engineering Strategies for Cartilage Defects Associated with Osteochondral Defects and Osteoarthritis (OA) | Ref. | ||
|---|---|---|---|
| 01 | Scaffold-Based Strategies | SF microparticles coated with NB | [64] |
| ECM scaffold derived from allogeneic BM-MSCs | [65] | ||
| PLGA scaffold with radially oriented microtubular pores | [66] | ||
| Fibrin/HA + antimiR-221 ± lipofectamine | [70] | ||
| HA/PCL + TGF-β3 or TGF-β3-free collagen solution | [74] | ||
| OPF-based scaffold /gelatin microparticles + IGF-1 ± BMP-2 | [75] | ||
| Bilayer alginate scaffold + TGF-β1 + BMP-4 | [76] | ||
| Porcine-derived acellular cartilage ECM scaffold + hWJMSC-Exos | [77] | ||
| 02 | Injectables | SF injectable | [78] |
| Photo-annealed MAP gel | [79] | ||
| PDLLA-PEG-PDLLA + heparin/EPL nanoparticles + PL | [81] | ||
| 03 | Clinical Studies | Atelocollagen/thrombin/fibrinogen + microfracture | [85] |
| Porcine-derived C-ACT | [86] | ||
| Col-1-based scaffolds | [87,88,89,90] | ||
| UC-derived WJ | [91] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Daou, F.; Cochis, A.; Leigheb, M.; Rimondini, L. Current Advances in the Regeneration of Degenerated Articular Cartilage: A Literature Review on Tissue Engineering and Its Recent Clinical Translation. Materials 2022, 15, 31. https://doi.org/10.3390/ma15010031
Daou F, Cochis A, Leigheb M, Rimondini L. Current Advances in the Regeneration of Degenerated Articular Cartilage: A Literature Review on Tissue Engineering and Its Recent Clinical Translation. Materials. 2022; 15(1):31. https://doi.org/10.3390/ma15010031
Chicago/Turabian StyleDaou, Farah, Andrea Cochis, Massimiliano Leigheb, and Lia Rimondini. 2022. "Current Advances in the Regeneration of Degenerated Articular Cartilage: A Literature Review on Tissue Engineering and Its Recent Clinical Translation" Materials 15, no. 1: 31. https://doi.org/10.3390/ma15010031
APA StyleDaou, F., Cochis, A., Leigheb, M., & Rimondini, L. (2022). Current Advances in the Regeneration of Degenerated Articular Cartilage: A Literature Review on Tissue Engineering and Its Recent Clinical Translation. Materials, 15(1), 31. https://doi.org/10.3390/ma15010031









