Enhancing the Oxidation Resistance of Al2O3-SiC-C Castables via Introducing Micronized Andalusite
Abstract
:1. Introduction
2. Experimental Procedures
2.1. Materials
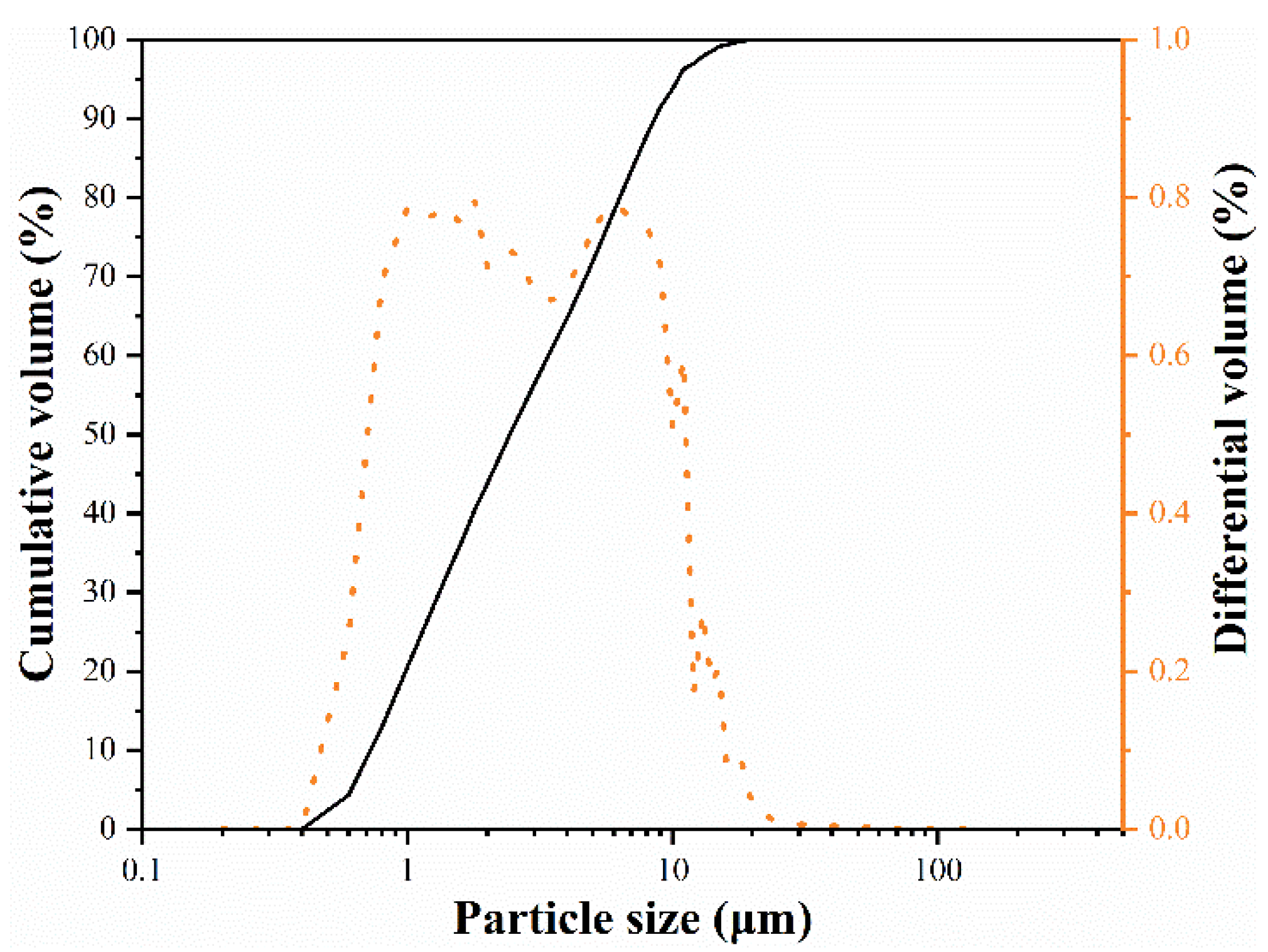
2.2. Sample Preparation and Characterization
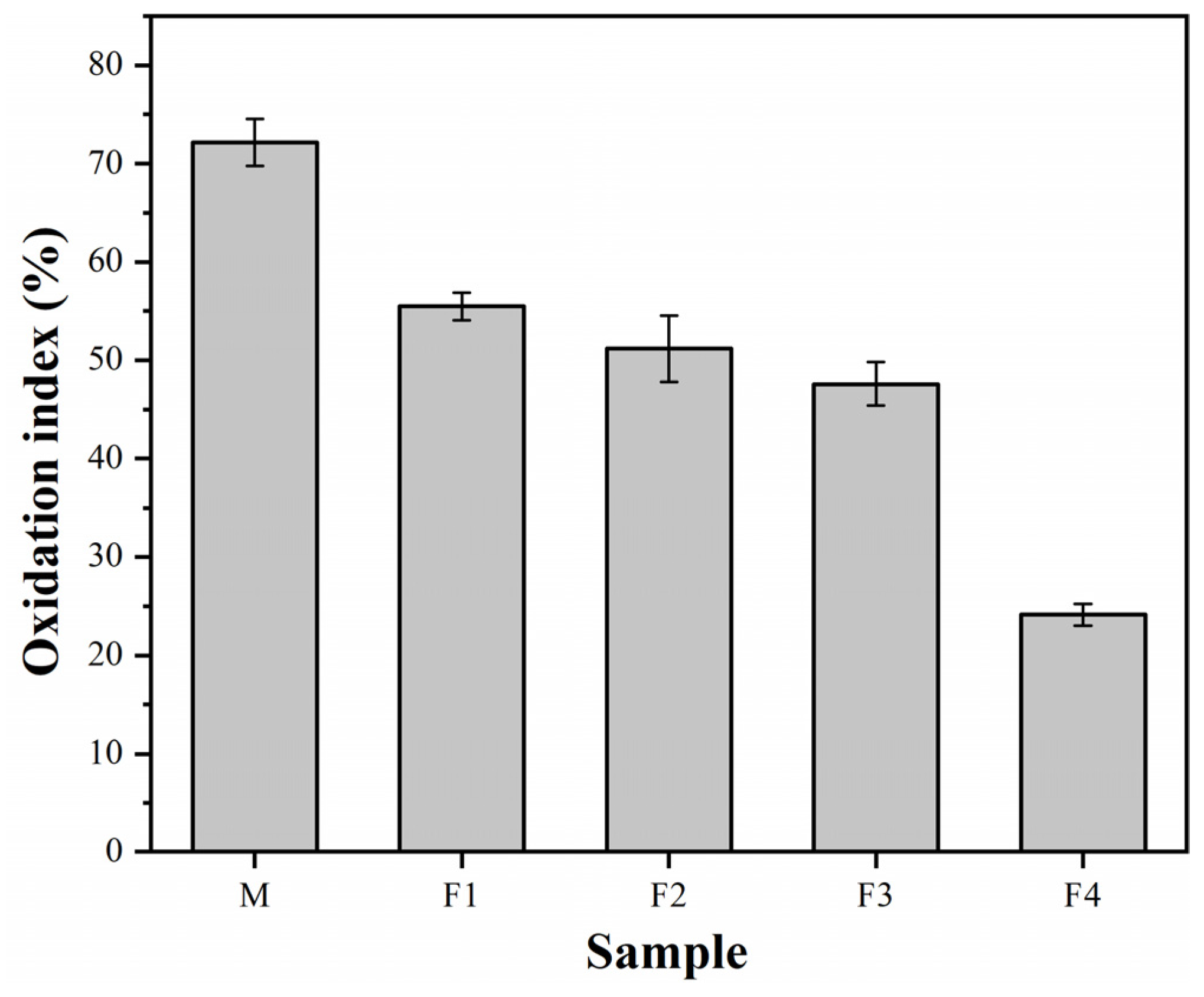
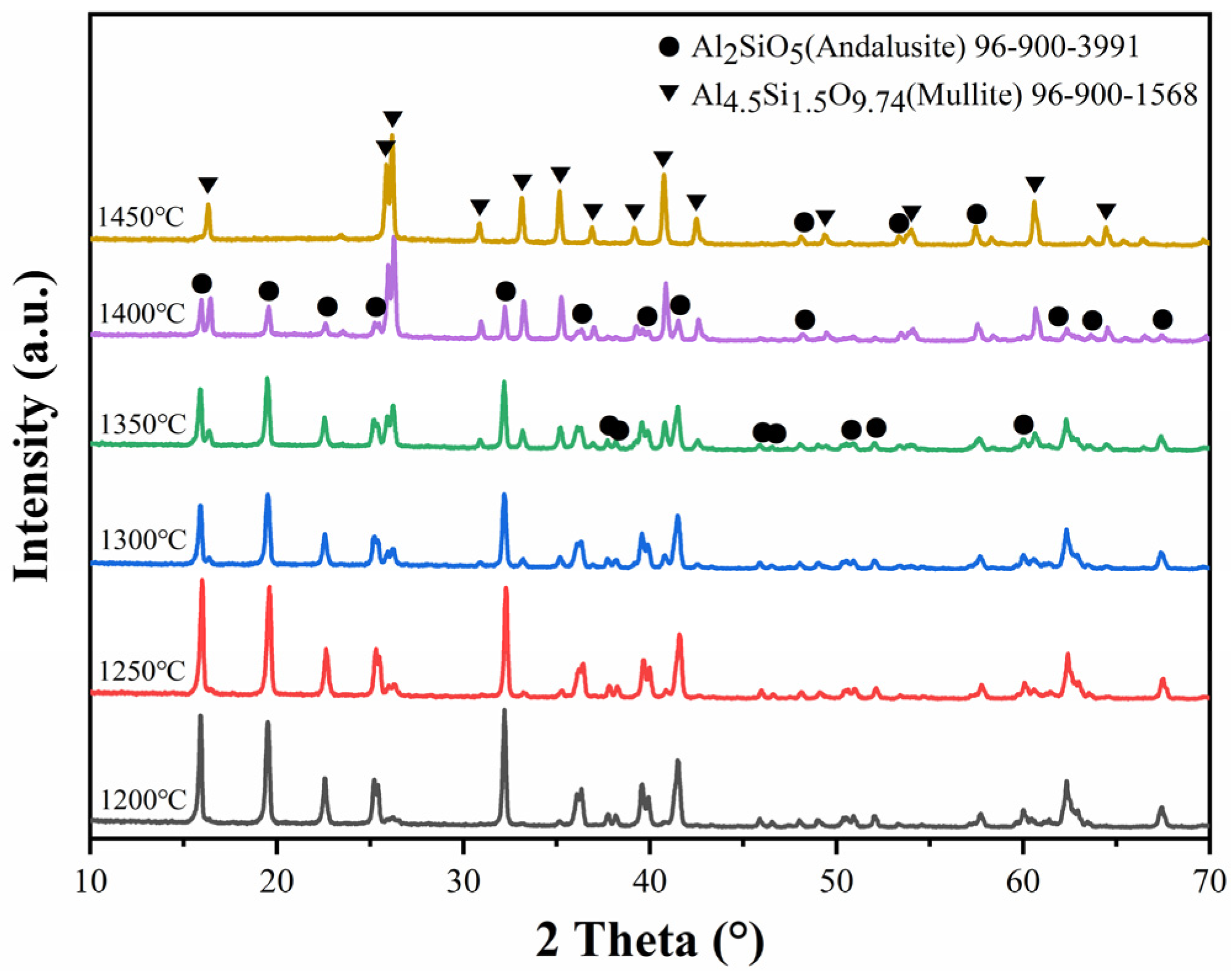
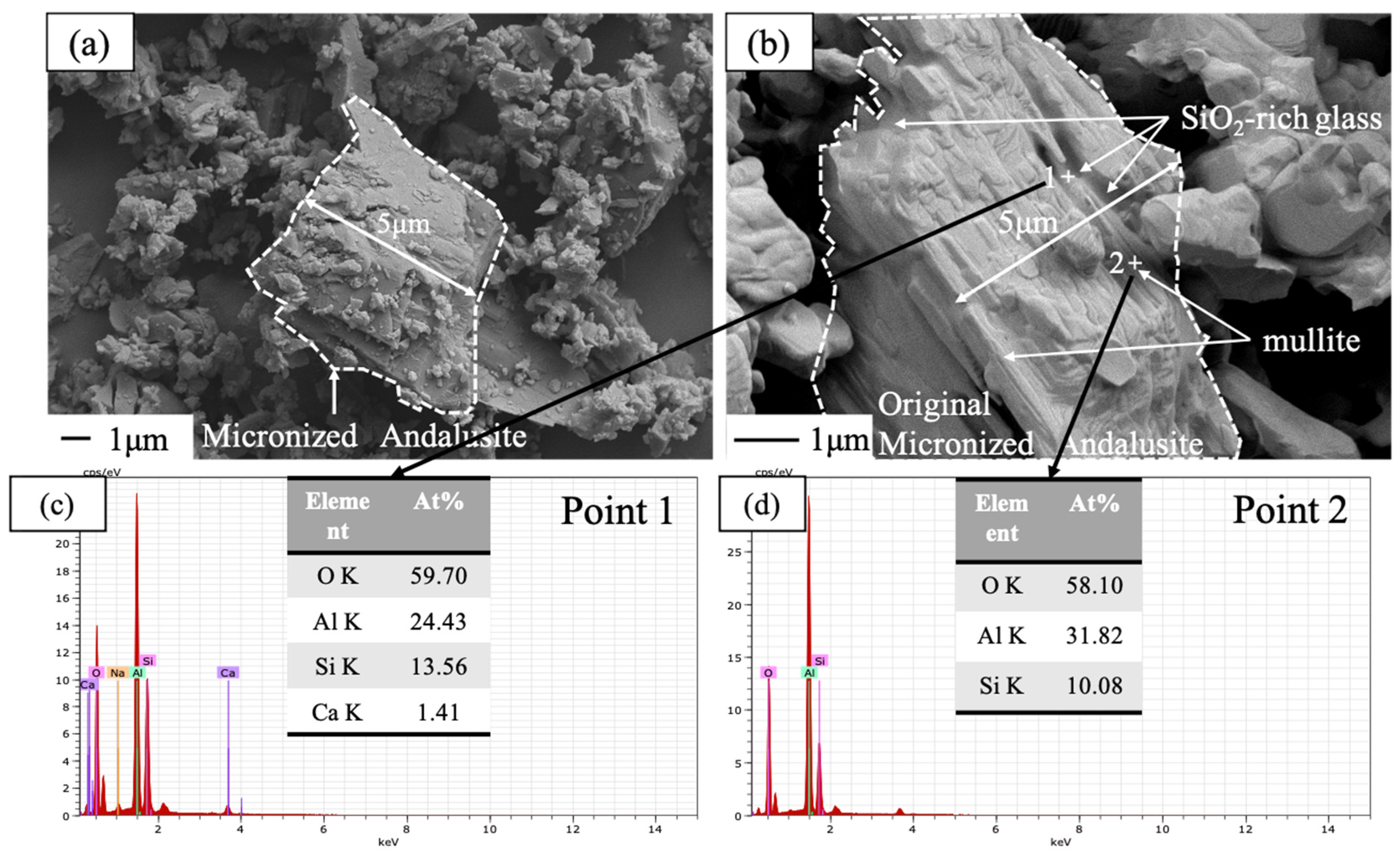
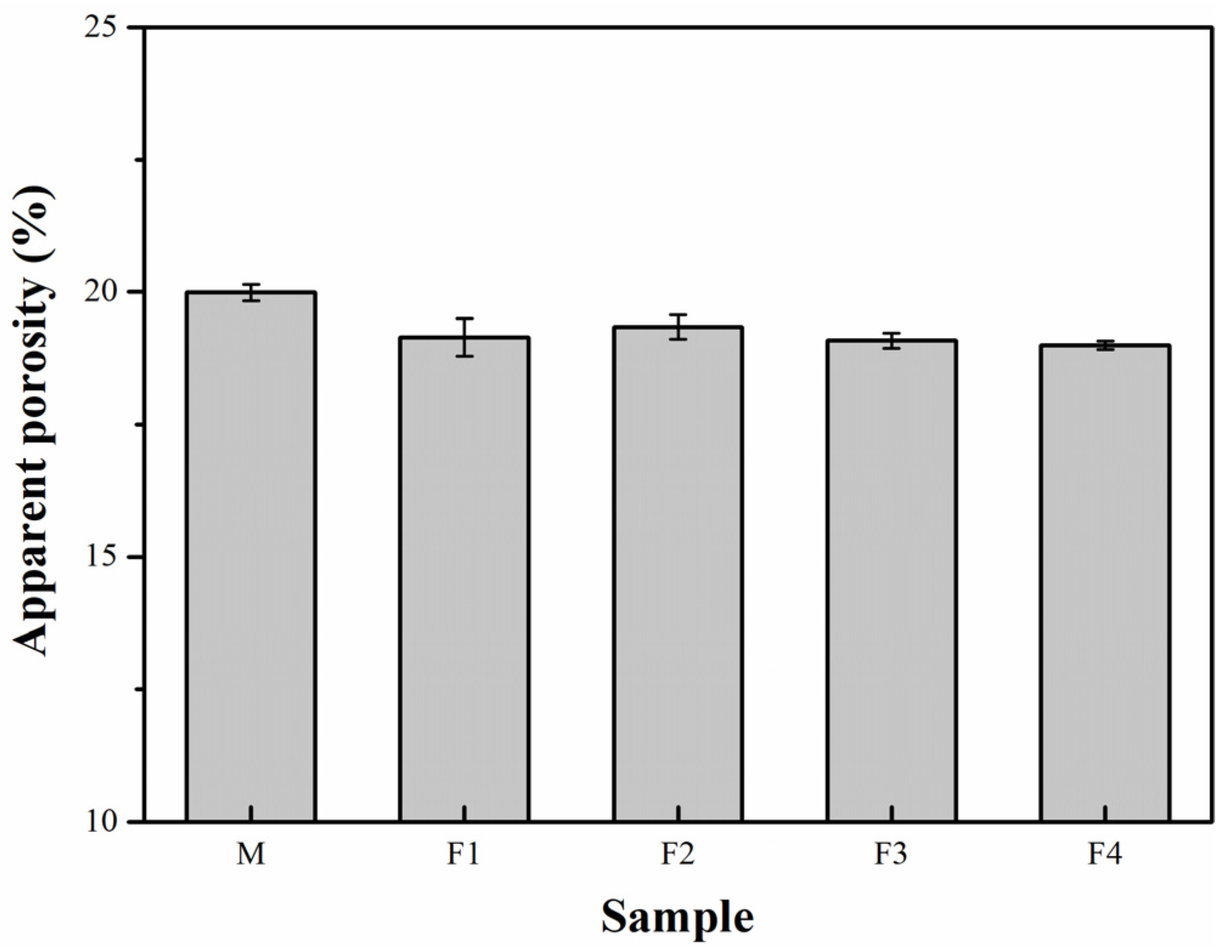
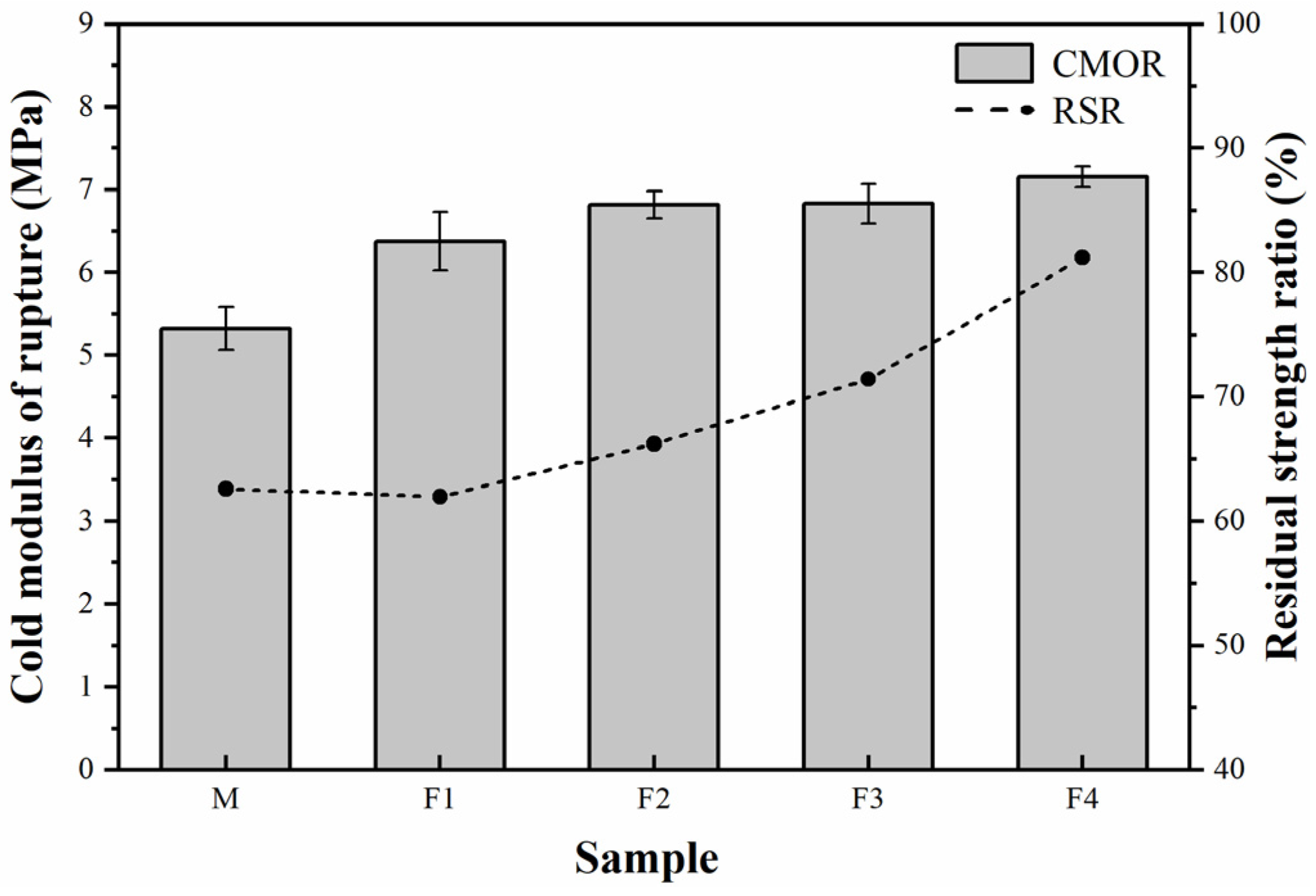
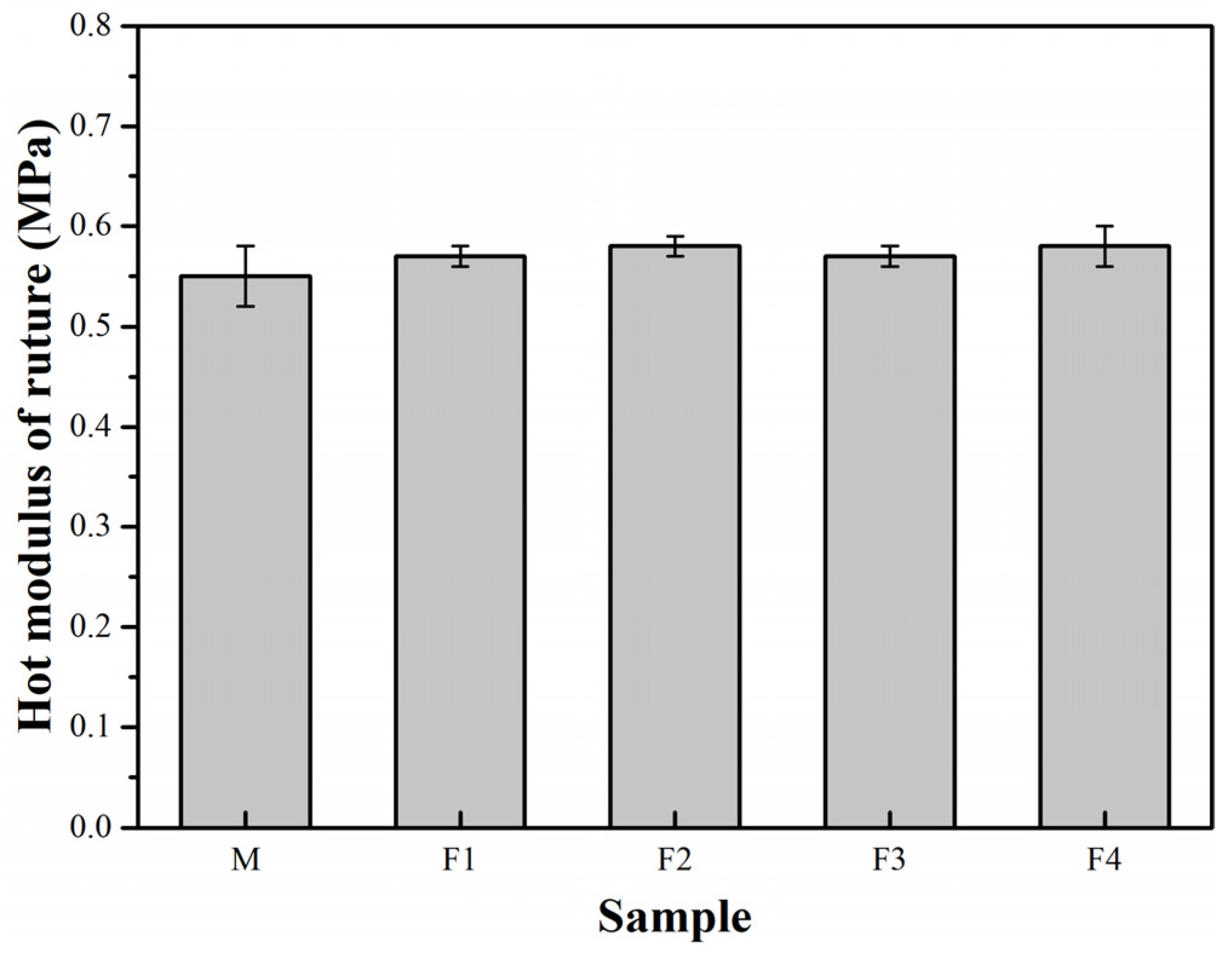
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Prompt, N.; Ouedraogo, E. High temperature mechanical characterization of an alumina refractory concrete for Blast Furnace main trough: Part I. General context. J. Eur. Ceram. Soc. 2008, 28, 2859–2865. [Google Scholar] [CrossRef]
- Zhang, S.; Lee, W.E. Carbon containing castables: Current status and future prospects. Br. Ceram. Trans. 2002, 101, 1–8. [Google Scholar] [CrossRef]
- Gallet-Doncieux, A.; Bahloul, O.; Gault, C.; Huger, M.; Chotard, T. Investigations of SiC aggregates oxidation: Influence on SiC castables refractories life time at high temperature. J. Eur. Ceram. Soc. 2012, 32, 737–743. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhao, S.; Nie, J. Effect of ball pitch on properties of Al2O3-SiC-C castables for iron trough. Refractories 2010, 44, 126–128. [Google Scholar]
- Wu, J.; Bu, N.; Li, H.; Zhen, Q. Effect of B4C on the properties and microstructure of Al2O3-SiC-C based trough castable refractories. Ceram. Int. 2017, 43, 1402–1409. [Google Scholar] [CrossRef]
- Liu, G.; Jin, X.; Qiu, W.; Ruan, G.; Li, Y. The effect of microsilica on the oxidation resistance of Al2O3-SiC-SiO2-C castables with Si and B4C additives. Ceram. Int. 2016, 42, 251–262. [Google Scholar] [CrossRef]
- Bi, Y.; Zhang, H.; Wang, H.; Duan, S.; Jia, Q.; Ge, S.; Zhang, S. Catalytic Fabrication of SiC/SiO2 coated graphite and its behaviour in Al2O3–C castable systems. Ceram. Int. 2019, 45, 16180–16187. [Google Scholar] [CrossRef]
- Ding, D.; Tian, X.; Ye, G. Effect of andalusite aggregates on oxidation resistance of Al2O3-SiC-C. castables. Ceram. Int. 2019, 45, 19237–19241. [Google Scholar] [CrossRef]
- Chen, A.; Wang, X.; Zhou, W.; Mu, Y.; Parr, C.; Ye, G. Oxidation resistance of Al2O3-SiC-C castables with different grades of andalusite. J. Alloys Compd. 2020, 851, 156836. [Google Scholar] [CrossRef]
- Tian, X.; Liu, M.; Luan, J.; Zhu, L.; Zhao, J.; Ye, G. Effect of andalusite aggregate pre-fired at different temperatures on volume stability and oxidation resistance of Al2O3-SiC-C. castables. Ceram. Int. 2020, 46, 22745–22751. [Google Scholar] [CrossRef]
- Tian, X.; Ding, D.; Zhang, P.; Liao, G.; Ahouanto, F.; Ye, G. Effect of andalusite with different particle sizes on properties of Al2O3-SiC-C castables. Refractories 2018, 52, 422–425. [Google Scholar]
- Prigent, P.; Bouchetou, M.L.; Poirier, J. Andalusite: An amazing refractory raw material with excellent corrosion resistance to sodium vapours. Ceram. Int. 2011, 37, 2287–2296. [Google Scholar] [CrossRef]
- Rebouillat, L.; Rigaud, M. Andalusite-based high-alumina castables. J. Am. Ceram. Soc. 2002, 85, 373–378. [Google Scholar] [CrossRef]
- Dubreuil, P.; Filari, E.; Sobolev, V.M. Use of andalusite refractories in ferrous metallurgy. Refract. Ind. Ceram. 1999, 40, 252–259. [Google Scholar] [CrossRef]
- Hülsmans, A.; Schmücker, M.; Mader, W.; Schneider, H. The transformation of andalusite to mullite and silica: Part I. Transformation mechanism in [1] A direction. Am. Mineral. 2000, 85, 980–986. [Google Scholar] [CrossRef]
- Hülsmans, A.; Schmücker, M.; Mader, W.; Schneider, H. The transformation of andalusite to mullite and silica: Part Ⅱ. Transformation mechanism in [100] and [10] A direction. Am. Mineral. 2000, 85, 987–992. [Google Scholar] [CrossRef]
- Ding, D.; Ye, G.; Li, N.; Liao, G.; Tian, X.; Chen, L. Andalusite transformation and properties of andalusite-bearing refractories fired in different atmospheres. Ceram. Int. 2019, 45, 3186–3191. [Google Scholar] [CrossRef]
- Ouedraogo, E.; Prompt, N. High-temperature mechanical characterization of an alumina refractory concrete for Blast Furnace main trough: Part II. Material behaviour. J. Eur. Ceram. Soc. 2008, 28, 2867–2875. [Google Scholar] [CrossRef]
- Liao, G.; He, K.; Lil, L.; Jiang, M. Study on application of alumina in high-purity andalusite based refractory. J. Miner. Mater. Charact. Eng. 2004, 3, 81–89. [Google Scholar] [CrossRef]
- Kakroudi, M.G.; Huger, M.; Gault, C.; Chotard, T. Anisotropic behaviour of andalusite particles used as aggregates on refractory castables. J. Eur. Ceram. Soc. 2009, 29, 571–579. [Google Scholar] [CrossRef]
- Li, C.; Bian, C.; Han, Y.; Wang, C.A.; An, L. Mullite whisker reinforced porous anorthite cermics with low thermal conductivity and high strength. J. Eur. Ceram. Soc. 2016, 36, 761–765. [Google Scholar] [CrossRef]
- Ribeiro, G.C.; Resende, W.S.; Rodrigues, J.A.; Ribeiro, S. Thermal shock resistance of a refractory castable containing andalusite aggregate. Ceram. Int. 2016, 42, 19167–19171. [Google Scholar] [CrossRef]
- Bouchetou, M.L.; Ildefonse, J.P.; Poirier, J.; Daniellou, P. Mullite grown from fired andalusite grains: Role of impurities and of the high temperature liquid phase on the kinetics of mullitization and consequences on thermal shocks resistance. Ceram. Int. 2005, 31, 999–1005. [Google Scholar] [CrossRef]


| Raw Material | Particle Size (mm) | wt (%) | ||||
|---|---|---|---|---|---|---|
| M | F1 | F2 | F3 | F4 | ||
| Brown fused alumina | 8–5 | 19 | 19 | 19 | 19 | 19 |
| 5–3 | 19 | 19 | 19 | 19 | 19 | |
| 3–1 | 19 | 19 | 19 | 19 | 19 | |
| 1–0 | 7.5 | 7.5 | 7.5 | 7.5 | 7.5 | |
| ≤0.074 | 5 | 4 | 3 | 2 | 0 | |
| Micronized andalusite | ≤0.005 | 0 | 1 | 2 | 3 | 5 |
| Pitch | 1–0.2 | 3 | 3 | 3 | 3 | 3 |
| SiC | 1–0 | 8 | 8 | 8 | 8 | 8 |
| ≤0.045 | 8 | 8 | 8 | 8 | 8 | |
| Silica fume | 2 | 2 | 2 | 2 | 2 | |
| Secar 71 | 3 | 3 | 3 | 3 | 3 | |
| α-Al2O3 micro powder | 5 | 5 | 5 | 5 | 5 | |
| Si powder | 1.5 | 1.5 | 1.5 | 1.5 | 1.5 | |
| Al powder | +0.1 | +0.1 | +0.1 | +0.1 | +0.1 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Wang, S.; Mu, Y.; Zhao, R.; Wang, Q.; Parr, C.; Ye, G. Enhancing the Oxidation Resistance of Al2O3-SiC-C Castables via Introducing Micronized Andalusite. Materials 2021, 14, 4775. https://doi.org/10.3390/ma14174775
Wang X, Wang S, Mu Y, Zhao R, Wang Q, Parr C, Ye G. Enhancing the Oxidation Resistance of Al2O3-SiC-C Castables via Introducing Micronized Andalusite. Materials. 2021; 14(17):4775. https://doi.org/10.3390/ma14174775
Chicago/Turabian StyleWang, Xiaoyu, Saixin Wang, Yuandong Mu, Ruijie Zhao, Qingfeng Wang, Chris Parr, and Guotian Ye. 2021. "Enhancing the Oxidation Resistance of Al2O3-SiC-C Castables via Introducing Micronized Andalusite" Materials 14, no. 17: 4775. https://doi.org/10.3390/ma14174775
APA StyleWang, X., Wang, S., Mu, Y., Zhao, R., Wang, Q., Parr, C., & Ye, G. (2021). Enhancing the Oxidation Resistance of Al2O3-SiC-C Castables via Introducing Micronized Andalusite. Materials, 14(17), 4775. https://doi.org/10.3390/ma14174775






