Recent Progress in the Synthesis of MoS2 Thin Films for Sensing, Photovoltaic and Plasmonic Applications: A Review
Abstract
1. Introduction
2. Fabrication Techniques of 2D-MoS2
2.1. Mechanical and Chemical Exfoliations
2.2. Chemical Vapor Deposition
2.3. Atomic Layer Deposition
2.4. Pulsed Laser Deposition
2.5. Other Processing Routes
3. Characterizations of MoS2 Thin Films
4. Band Structures and Electronic Properties
5. MoS2 Applications
5.1. MoS2 for Solar Energy Harvesting
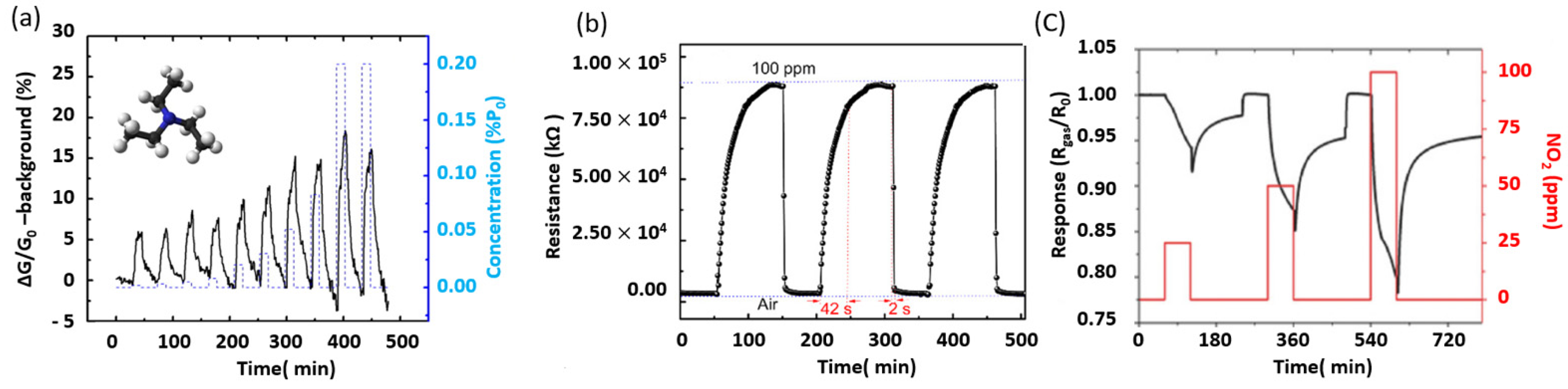
5.2. MoS2 for Gas Sensing Applications
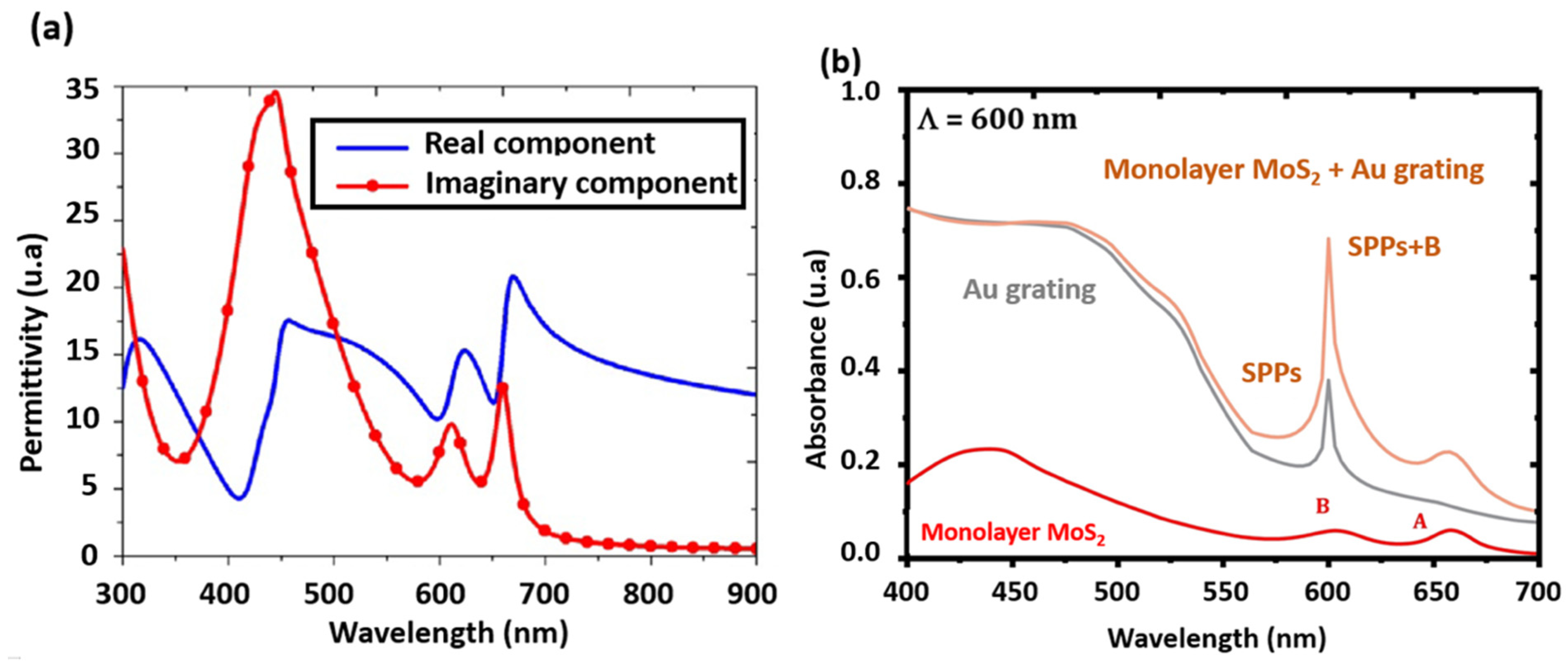
5.3. MoS2 for Plasmonic Applications
6. Summary and Outlook
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 1T | Tetragonal |
| 2D | Two-dimension |
| 2H | Hexagonal |
| 3R | Rhombohedral |
| Δω | Raman shift between the peak positions of E12g and A1g |
| ALD | Atomic layer deposition |
| A1g | MoS2 out-of-plane Raman vibration mode |
| BL | Buffer layer |
| CNTs | Carbon nanotubes |
| CVD | Chemical vapor deposition |
| DFT | Density-functional theory |
| E12g | MoS2 in-plane Raman vibration mode |
| ETL | Electron transport layer |
| EQE | External quantum efficiency |
| FEE | Field electron emission |
| FET | Field-effect transistor |
| Gr | Graphene |
| HIT | Heterojunction with intrinsic thin layer |
| HLDG | Hybrid Lorentz-Drude-Gaussian model |
| HNPs | Hexagonal-shaped nanoplates |
| HRTEM | High-Resolution Transmission Electron Microscopy |
| J-V | current density versus voltage |
| Jsc | Short circuit current density |
| KPFM | Kelvin probe force microscopy |
| LSPR | Localized surface plasmon |
| NPs | Nanoparticles |
| NSs | Nanosheets |
| PCE | Power conversion efficiency |
| PDMS | Polydimethylsiloxane polymer |
| PL | Photoluminescence |
| PLD | Pulsed laser deposition |
| PPB | Particles per billions |
| PPM | Particles per millions |
| PSCs | Perovskite solar cells |
| Ra | Resistance of the sensing element in the presence of atmospheric air |
| Rg | Resistance of the sensing element in the presence of the target gas |
| RH | Relative humidity |
| SCs | Solar cells |
| SEM | Scanning electron microscopy |
| Smax | The maximum value of the sensing response |
| SP | Surface potential |
| TCE | Transparent conducting electrode |
| TEM | Transmission electron microscopy |
| TD | Thermal decomposition |
| TMDs | Transition metal dichalcogenides |
| TRPL | Time-resolved photoluminescence |
| TVD | Thermal vapor deposition |
| TVS | Thermal vapor sulfurization |
| UV | Ultraviolet |
| Voc | Open circuit voltage |
| XPS | X-ray photoelectron spectroscopy |
References
- Mas-Ballesté, R.; Gómez-Navarro, C.; Gómez-Herrero, J.; Zamora, F. 2D materials: To graphene and beyond. Nanoscale 2011, 3, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Allen, M.J.; Tung, V.C.; Kaner, R.B. Honeycomb carbon: A review of graphene. Chem. Rev. 2010, 110, 132–145. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Murali, S.; Cai, W.; Li, X.; Suk, J.W.; Potts, J.R.; Ruoff, R.S. Graphene and graphene oxide: Synthesis, properties, and applications. Adv. Mater. 2010, 22, 3906–3924. [Google Scholar] [CrossRef] [PubMed]
- Mattevi, C.; Kim, H.; Chhowalla, M. A review of chemical vapour deposition of graphene on copper. J. Mater. Chem. 2011, 21, 3324–3334. [Google Scholar] [CrossRef]
- Deokar, G.; Avila, J.; Razado-Colambo, I.; Codron, J.L.; Boyaval, C.; Galopin, E.; Asensio, M.C.; Vignaud, D. Towards high quality CVD graphene growth and transfer. Carbon N. Y. 2015, 89, 82–92. [Google Scholar] [CrossRef]
- Deokar, G.; Casanova-Cháfer, J.; Rajput, N.S.; Aubry, C.; Llobet, E.; Jouiad, M.; Costa, P.M.F.J. Wafer-scale few-layer graphene growth on Cu/Ni films for gas sensing applications. Sens. Actuators B Chem. 2020, 305, 127458. [Google Scholar] [CrossRef]
- Minkin, A.S.; Lebedeva, I.V.; Popov, A.M.; Knizhnik, A.A. The Application of Empirical Potentials for Calculation of Elastic Properties of Graphene. Tech. Phys. Lett. 2019, 45, 111–114. [Google Scholar] [CrossRef]
- Gupta, A.; Sakthivel, T.; Seal, S. Recent development in 2D materials beyond graphene. Prog. Mater. Sci. 2015, 73, 44–126. [Google Scholar] [CrossRef]
- Castro Neto, A.H.; Guinea, F.; Peres, N.M.R.; Novoselov, K.S.; Geim, A.K. The electronic properties of graphene. Rev. Mod. Phys. 2009, 81, 109–162. [Google Scholar] [CrossRef]
- Neto Castro, A.H.; Novoselov, K. Two-dimensional crystals: Beyond graphene. Mater. Express 2011, 1, 10–17. [Google Scholar] [CrossRef]
- Tan, C.; Zhang, H. Two-dimensional transition metal dichalcogenide nanosheet-based composites. Chem. Soc. Rev. 2015, 44, 2713–2731. [Google Scholar] [CrossRef]
- Lu, Q.; Yu, Y.; Ma, Q.; Chen, B.; Zhang, H. 2D Transition-Metal-Dichalcogenide-Nanosheet-Based Composites for Photocatalytic and Electrocatalytic Hydrogen Evolution Reactions. Adv. Mater. 2016, 28, 1917–1933. [Google Scholar] [CrossRef] [PubMed]
- Ansari, L.; Monaghan, S.; McEvoy, N.; Coileáin, C.; Cullen, C.P.; Lin, J.; Siris, R.; Stimpel-Lindner, T.; Burke, K.F.; Mirabelli, G.; et al. Quantum confinement-induced semimetal-to-semiconductor evolution in large-area ultra-thin PtSe2 films grown at 400 °C. npj 2D Mater. Appl. 2019, 3, 1–8. [Google Scholar] [CrossRef]
- Stanford, M.G.; Rack, P.D.; Jariwala, D. Emerging nanofabrication and quantum confinement techniques for 2D materials beyond graphene. npj 2D Mater. Appl. 2018, 2, 1–15. [Google Scholar] [CrossRef]
- Butoi, C.I.; Langdon, B.T.; Kelley, D.F. Electron-Transfer Dynamics in DTDCI / MoS 2 and DTDCI / WS 2 Nanoclusters. J. Phys. Chem. B 1998, 102, 9635–9639. [Google Scholar] [CrossRef]
- Wu, X.L.; Fan, J.Y.; Qiu, T.; Yang, X.; Siu, G.G.; Chu, P.K. Experimental evidence for the quantum confinement effect in 3C-SiC nanocrystallites. Phys. Rev. Lett. 2005, 94, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Gan, Z.X.; Liu, L.Z.; Wu, H.Y.; Hao, Y.L.; Shan, Y.; Wu, X.L.; Chu, P.K. Quantum confinement effects across two-dimensional planes in MoS2 quantum dots. Appl. Phys. Lett. 2015, 106, 233113. [Google Scholar] [CrossRef]
- Ellis, J.K.; Lucero, M.J.; Scuseria, G.E. The indirect to direct band gap transition in multilayered MoS 2 as predicted by screened hybrid density functional theory. Appl. Phys. Lett. 2011, 99, 261908. [Google Scholar] [CrossRef]
- Wang, S.W.; Medina, H.; Hong, K.B.; Wu, C.C.; Qu, Y.; Manikandan, A.; Su, T.Y.; Lee, P.T.; Huang, Z.Q.; Wang, Z.; et al. Thermally Strained Band Gap Engineering of Transition-Metal Dichalcogenide Bilayers with Enhanced Light-Matter Interaction toward Excellent Photodetectors. ACS Nano 2017, 11, 8768–8776. [Google Scholar] [CrossRef]
- Yu, Z.; Ong, Z.Y.; Li, S.; Xu, J.B.; Zhang, G.; Zhang, Y.W.; Shi, Y.; Wang, X. Analyzing the Carrier Mobility in Transition-Metal Dichalcogenide MoS2 Field-Effect Transistors. Adv. Funct. Mater. 2017, 27, 1604093. [Google Scholar] [CrossRef]
- Wang, S.; Zhao, W.; Giustiniano, F.; Eda, G. Effect of oxygen and ozone on p-type doping of ultra-thin WSe2 and MoSe2 field effect transistors. Phys. Chem. Chem. Phys. 2016, 18, 4304–4309. [Google Scholar] [CrossRef]
- Wang, X.; Wang, P.; Wang, J.; Hu, W.; Zhou, X.; Guo, N.; Huang, H.; Sun, S.; Shen, H.; Lin, T.; et al. Ultrasensitive and Broadband MoS2 Photodetector Driven by Ferroelectrics. Adv. Mater. 2015, 27, 6575–6581. [Google Scholar] [CrossRef]
- Yin, Z.; Zhang, X.; Cai, Y.; Chen, J.; Wong, J.I.; Tay, Y.Y.; Chai, J.; Wu, J.; Zeng, Z.; Zheng, B.; et al. Preparation of MoS2-MoO3 hybrid nanomaterials for light-emitting diodes. Angew. Chemie - Int. Ed. 2014, 53, 12560–12565. [Google Scholar]
- Liu, Z.; Liu, K.; Zhang, F.; Jain, S.M.; He, T.; Jiang, Y.; Liu, P.; Yang, J.; Liu, H.; Yuan, M. CH3NH3PbI3:MoS2 heterostructure for stable and efficient inverted perovskite solar cell. Sol. Energy 2020, 195, 436–445. [Google Scholar] [CrossRef]
- Wang, H.; Li, C.; Fang, P.; Zhang, Z.; Zhang, J.Z. Synthesis, properties, and optoelectronic applications of two-dimensional MoS2 and MoS2-based heterostructures. Chem. Soc. Rev. 2018, 47, 6101–6127. [Google Scholar] [CrossRef]
- Gupta, D.; Chauhan, V.; Kumar, R. A comprehensive review on synthesis and applications of molybdenum disulfide (MoS2) material: Past and recent developments. Inorg. Chem. Commun. 2020, 121, 108200. [Google Scholar] [CrossRef]
- Krishnan, U.; Kaur, M.; Singh, K.; Kumar, M.; Kumar, A. A synoptic review of MoS 2: Synthesis to applications. Superlattices Microstruct. 2019, 128, 274–297. [Google Scholar] [CrossRef]
- Nalwa, H.S. A review of molybdenum disulfide (MoS2) based photodetectors: From ultra-broadband, self-powered to flexible devices. RSC Adv. 2020, 10, 30529–30602. [Google Scholar] [CrossRef]
- Sun, J.; Li, X.; Guo, W.; Zhao, M.; Fan, X.; Dong, Y.; Xu, C.; Deng, J.; Fu, Y. Synthesis methods of two-dimensional MoS2: A brief review. Crystals 2017, 7, 198. [Google Scholar] [CrossRef]
- Kumar, R.; Sahoo, S.; Joanni, E.; Singh, R.K.; Yadav, R.M.; Verma, R.K.; Singh, D.P.; Tan, W.K.; Pérez del Pino, A.; Moshkalev, S.A.; et al. A review on synthesis of graphene, h-BN and MoS2 for energy storage applications: Recent progress and perspectives. Nano Res. 2019, 12, 2655–2694. [Google Scholar] [CrossRef]
- Xu, H.; Yi, J.; She, X.; Liu, Q.; Song, L.; Chen, S.; Yang, Y.; Song, Y.; Vajtai, R.; Lou, J.; et al. 2D heterostructure comprised of metallic 1T-MoS2/Monolayer O-g-C3N4 towards efficient photocatalytic hydrogen evolution. Appl. Catal. B Environ. 2018, 220, 379–385. [Google Scholar] [CrossRef]
- Backes, C.; Berner, N.C.; Chen, X.; Lafargue, P.; LaPlace, P.; Freeley, M.; Duesberg, G.S.; Coleman, J.N.; McDonald, A.R. Functionalization of liquid-exfoliated two-dimensional 2H-MoS2. Angew. Chemie - Int. Ed. 2015, 54, 2638–2642. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.; Willatzen, M.; Wang, Z.L. Prediction of strong piezoelectricity in 3R-MoS2 multilayer structures. Nano Energy 2019, 56, 512–515. [Google Scholar] [CrossRef]
- Liu, K.K.; Zhang, W.; Lee, Y.H.; Lin, Y.C.; Chang, M.T.; Su, C.Y.; Chang, C.S.; Li, H.; Shi, Y.; Zhang, H.; et al. Growth of large-area and highly crystalline MoS 2 thin layers on insulating substrates. Nano Lett. 2012, 12, 1538–1544. [Google Scholar] [CrossRef]
- Yu, H.; Liao, M.; Zhao, W.; Liu, G.; Zhou, X.J.; Wei, Z.; Xu, X.; Liu, K.; Hu, Z.; Deng, K.; et al. Wafer-Scale Growth and Transfer of Highly-Oriented Monolayer MoS2 Continuous Films. ACS Nano 2017, 11, 12001–12007. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Zhang, S.; Pan, S.; Tang, B.; Liang, Y.; Zhao, X.; Zhang, Z.; Shi, J.; Huan, Y.; Shi, Y.; et al. Epitaxial Growth of Centimeter-Scale Single-Crystal MoS2 Monolayer on Au(111). ACS Nano 2020, 14, 5036–5045. [Google Scholar] [CrossRef]
- Chen, X.P.; Xing, G.J.; Xu, L.F.; Lian, H.Q.; Wang, Y. Vertically aligned MoS2 films prepared by RF-magnetron sputtering method as electrocatalysts for hydrogen evolution reactions. Compos. Interfaces 2020, 1–10. [Google Scholar] [CrossRef]
- Hu, Z.; Wang, L.; Zhang, K.; Wang, J.; Cheng, F.; Tao, Z.; Chen, J. MoS2 Nanoflowers with Expanded Interlayers as High-Performance Anodes for Sodium-Ion Batteries. Angew. Chemie - Int. Ed. 2014, 53, 12794–12798. [Google Scholar] [CrossRef]
- Chen, J.; Kuriyama, N.; Yuan, H.; Takeshita, H.T.; Sakai, T. Electrochemical hydrogen storage in MoS2 nanotubes. J. Am. Chem. Soc. 2001, 123, 11813–11814. [Google Scholar] [CrossRef]
- Li, W.J.; Shi, E.W.; Ko, J.M.; Chen, Z.Z.; Ogino, H.; Fukuda, T. Hydrothermal synthesis of MoS2 nanowires. J. Cryst. Growth 2003, 250, 418–422. [Google Scholar] [CrossRef]
- Hwang, H.; Kim, H.; Cho, J. MoS2 nanoplates consisting of disordered graphene-like layers for high rate lithium battery anode materials. Nano Lett. 2011, 11, 4826–4830. [Google Scholar] [CrossRef]
- Deokar, G.; Vancsó, P.; Arenal, R.; Ravaux, F.; Casanova-Cháfer, J.; Llobet, E.; Makarova, A.; Vyalikh, D.; Struzzi, C.; Lambin, P.; et al. MoS2–Carbon Nanotube Hybrid Material Growth and Gas Sensing. Adv. Mater. Interfaces 2017, 4, 1–10. [Google Scholar] [CrossRef]
- Deokar, G.; Vignaud, D.; Arenal, R.; Louette, P.; Colomer, J. Synthesis and characterization of MoS2 nanosheets. Nanotechnology 2016, 27, 075604. [Google Scholar] [CrossRef]
- Deokar, G.; Rajput, N.S.; Vancsó, P.; Ravaux, F.; Jouiad, M.; Vignaud, D.; Cecchet, F.; Colomer, J.F. Large area growth of vertically aligned luminescent MoS2 nanosheets. Nanoscale 2017, 9, 277–287. [Google Scholar] [CrossRef]
- Gan, X.; Gao, Y.; Fai Mak, K.; Yao, X.; Shiue, R.J.; Van Der Zande, A.; Trusheim, M.E.; Hatami, F.; Heinz, T.F.; Hone, J.; et al. Controlling the spontaneous emission rate of monolayer MoS2 in a photonic crystal nanocavity. Appl. Phys. Lett. 2013, 103, 181119. [Google Scholar] [CrossRef] [PubMed]
- Eda, G.; Fujita, T.; Yamaguchi, H.; Voiry, D.; Chen, M.; Chhowalla, M. Coherent atomic and electronic heterostructures of single-layer MoS 2. ACS Nano 2012, 6, 7311–7317. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Chen, S.; Pang, H.; Xue, H.; Yu, Y. MoS2-Based Nanocomposites for Electrochemical Energy Storage. Adv. Sci. 2017, 4, 1600289. [Google Scholar] [CrossRef] [PubMed]
- Shokri, A.; Salami, N. Gas sensor based on MoS2 monolayer. Sensors Actuators, B Chem. 2016, 236, 378–385. [Google Scholar] [CrossRef]
- Deokar, G.; Rajput, N.S.; Li, J.; Deepak, F.L.; Ou-Yang, W.; Reckinger, N.; Bittencourt, C.; Colomer, J.F.; Jouiad, M. Toward the use of CVD-grown MoS2 nanosheets as field-emission source. Beilstein J. Nanotechnol. 2018, 9, 1686–1694. [Google Scholar] [CrossRef]
- Ma, J.; Bai, H.; Zhao, W.; Yuan, Y.; Zhang, K. High efficiency graphene/MoS2/Si Schottky barrier solar cells using layer-controlled MoS2 films. Sol. Energy 2018, 160, 76–84. [Google Scholar] [CrossRef]
- Arulraj, A.; Ramesh, M.; Subramanian, B.; Senguttuvan, G. In-situ temperature and thickness control grown 2D-MoS2 via pulsed laser ablation for photovoltaic devices. Sol. Energy 2018, 174, 286–295. [Google Scholar] [CrossRef]
- Guo, F.; Li, M.; Ren, H.; Huang, X.; Hou, W.; Wang, C.; Shi, W.; Lu, C. Fabrication of p-n CuBi2O4/MoS2 heterojunction with nanosheets-on-microrods structure for enhanced photocatalytic activity towards tetracycline degradation. Appl. Surf. Sci. 2019, 491, 88–94. [Google Scholar] [CrossRef]
- Splendiani, A.; Sun, L.; Zhang, Y.; Li, T.; Kim, J.; Chim, C.Y.; Galli, G.; Wang, F. Emerging photoluminescence in monolayer MoS2. Nano Lett. 2010, 10, 1271–1275. [Google Scholar] [CrossRef] [PubMed]
- Eda, G.; Yamaguchi, H.; Voiry, D.; Fujita, T.; Chen, M.; Chhowalla, M. Photoluminescence from chemically exfoliated MoS 2. Nano Lett. 2011, 11, 5111–5116. [Google Scholar] [CrossRef] [PubMed]
- Rafiee, J.; Mi, X.; Gullapalli, H.; Thomas, A.V.; Yavari, F.; Shi, Y.; Ajayan, P.M.; Koratkar, N.A. Wetting transparency of graphene. Nat. Mater. 2012, 11, 217–222. [Google Scholar] [CrossRef]
- Kozbial, A.; Zhou, F.; Li, Z.; Liu, H.; Li, L. Are Graphitic Surfaces Hydrophobic? Acc. Chem. Res. 2016, 49, 2765–2773. [Google Scholar] [CrossRef]
- Marbou, K.; Ghaferi, A.A.; Jouiad, M. In-situ Characterization of Wettability Alteration in HOPG. SOP Trans. Nanotechnol 2015, 2374, 1–10. [Google Scholar]
- Huang, Y.; Pan, Y.H.; Yang, R.; Bao, L.H.; Meng, L.; Luo, H.L.; Cai, Y.Q.; Liu, G.D.; Zhao, W.J.; Zhou, Z.; et al. Universal mechanical exfoliation of large-area 2D crystals. Nat. Commun. 2020, 11, 1–9. [Google Scholar] [CrossRef]
- Magda, G.Z.; Petõ, J.; Dobrik, G.; Hwang, C.; Biró, L.P.; Tapasztó, L. Exfoliation of large-area transition metal chalcogenide single layers. Sci. Rep. 2015, 5, 3–7. [Google Scholar] [CrossRef]
- Kim, S.; Park, W.; Kim, D.; Kang, J.; Lee, J.; Jang, H.Y.; Song, S.H.; Cho, B.; Lee, D. Novel exfoliation of high-quality 2h-mos2 nanoflakes for solution-processed photodetector. Nanomaterials 2020, 10, 1045. [Google Scholar] [CrossRef]
- Pirzado, A.A.; Le Normand, F.; Romero, T.; Paszkiewicz, S.; Papaefthimiou, V.; Ihiawakrim, D.; Janowska, I. Few-layer graphene from mechanical exfoliation of graphite-based materials: Structure-dependent characteristics. ChemEngineering 2019, 3, 1–10. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Jiang, D.; Schedin, F.; Booth, T.J.; Khotkevich, V.V.; Morozov, S.V.; Geim, A.K.; Benka, S.G. Two-dimensional atomic crystals. Phys. Today 2005, 58, 9. [Google Scholar] [CrossRef] [PubMed]
- Janica, I.; Iglesias, D.; Ippolito, S.; Ciesielski, A.; Samorì, P. Effect of temperature and exfoliation time on the properties of chemically exfoliated MoS2nanosheets. Chem. Commun. 2020, 56, 15573–15576. [Google Scholar] [CrossRef] [PubMed]
- Guan, Z.; Wang, C.; Li, W.; Luo, S.; Yao, Y.; Yu, S.; Sun, R.; Wong, C.P. A facile and clean process for exfoliating MoS2 nanosheets assisted by a surface active agent in aqueous solution. Nanotechnology 2018, 29, 425702. [Google Scholar] [CrossRef]
- Lin, H.; Wang, J.; Luo, Q.; Peng, H.; Luo, C.; Qi, R.; Huang, R.; Travas-Sejdic, J.; Duan, C.G. Rapid and highly efficient chemical exfoliation of layered MoS2and WS2. J. Alloys Compd. 2017, 699, 222–229. [Google Scholar] [CrossRef]
- Yang, Y.Q.; Tye, C.T.; Smith, K.J. Influence of MoS2 catalyst morphology on the hydrodeoxygenation of phenols. Catal. Commun. 2008, 9, 1364–1368. [Google Scholar] [CrossRef]
- Liu, H.F.; Wong, S.L.; Chi, D.Z. CVD Growth of MoS2-based Two-dimensional Materials. Chem. Vap. Depos. 2015, 21, 241–259. [Google Scholar] [CrossRef]
- Wang, Q.H.; Kalantar-Zadeh, K.; Kis, A.; Coleman, J.N.; Strano, M.S. Electronics and optoelectronics of two-dimensional transition metal dichalcogenides. Nat. Nanotechnol. 2012, 7, 699–712. [Google Scholar] [CrossRef]
- Zeng, T.; You, Y.; Wang, X.; Hu, T.; Tai, G. Chemical vapor deposition and device application of two-dimensional molybdenum disulfide-based atomic crystals. Prog. Chem. 2016, 28, 459–470. [Google Scholar]
- Balendhran, S.; Ou, J.Z.; Bhaskaran, M.; Sriram, S.; Ippolito, S.; Vasic, Z.; Kats, E.; Bhargava, S.; Zhuiykov, S.; Kalantar-Zadeh, K. Atomically thin layers of MoS 2via a two step thermal evaporation-exfoliation method. Nanoscale 2012, 4, 461–466. [Google Scholar] [CrossRef]
- Nam Trung, T.; Kamand, F.Z.; Al tahtamouni, T.M. Elucidating the mechanism for the chemical vapor deposition growth of vertical MoO2/MoS2 flakes toward photoelectrochemical applications. Appl. Surf. Sci. 2020, 505, 144551. [Google Scholar] [CrossRef]
- Ahn, C.; Lee, J.; Kim, H.U.; Bark, H.; Jeon, M.; Ryu, G.H.; Lee, Z.; Yeom, G.Y.; Kim, K.; Jung, J.; et al. Low-Temperature Synthesis of Large-Scale Molybdenum Disulfide Thin Films Directly on a Plastic Substrate Using Plasma-Enhanced Chemical Vapor Deposition. Adv. Mater. 2015, 27, 5223–5229. [Google Scholar] [CrossRef]
- Sojková, M.; Siffalovic, P.; Babchenko, O.; Vanko, G.; Dobročka, E.; Hagara, J.; Mrkyvkova, N.; Majková, E.; Ižák, T.; Kromka, A.; et al. Carbide-free one-zone sulfurization method grows thin MoS 2 layers on polycrystalline CVD diamond. Sci. Rep. 2019, 9, 2–12. [Google Scholar] [CrossRef]
- Withanage, S.S.; Kalita, H.; Chung, H.S.; Roy, T.; Jung, Y.; Khondaker, S.I. Uniform Vapor-Pressure-Based Chemical Vapor Deposition Growth of MoS2 Using MoO3 Thin Film as a Precursor for Coevaporation. ACS Omega 2018, 3, 18943–18949. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Rong, Y.; Fan, Y.; Pacios, M.; Bhaskaran, H.; He, K.; Warner, J.H. Shape evolution of monolayer MoS2 crystals grown by chemical vapor deposition. Chem. Mater. 2014, 26, 6371–6379. [Google Scholar] [CrossRef]
- Jeon, J.; Jang, S.K.; Jeon, S.M.; Yoo, G.; Jang, Y.H.; Park, J.H.; Lee, S. Layer-controlled CVD growth of large-area two-dimensional MoS2 films. Nanoscale 2015, 7, 1688–1695. [Google Scholar] [CrossRef] [PubMed]
- Hyun, C.M.; Choi, J.H.; Lee, S.W.; Park, J.H.; Lee, K.T.; Ahn, J.H. Synthesis mechanism of MoS2 layered crystals by chemical vapor deposition using MoO3 and sulfur powders. J. Alloys Compd. 2018, 765, 380–384. [Google Scholar] [CrossRef]
- Lin, Z.; Zhao, Y.; Zhou, C.; Zhong, R.; Wang, X.; Tsang, Y.H.; Chai, Y. Controllable Growth of Large-Size Crystalline MoS2 and Resist-Free Transfer Assisted with a Cu Thin Film. Sci. Rep. 2015, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Rotunno, E.; Bosi, M.; Seravalli, L.; Salviati, G.; Fabbri, F. Influence of organic promoter gradient on the MoS2growth dynamics. Nanoscale Adv. 2020, 2, 2352–2362. [Google Scholar] [CrossRef]
- Le, D.; Rawal, T.B.; Rahman, T.S. Single-Layer MoS2 with Sulfur Vacancies: Structure and Catalytic Application. J. Phys. Chem. C 2014, 118, 5346–5351. [Google Scholar] [CrossRef]
- Jurca, T.; Moody, M.J.; Henning, A.; Emery, J.D.; Wang, B.; Tan, J.M.; Lohr, T.L.; Lauhon, L.J.; Marks, T.J. Low-Temperature Atomic Layer Deposition of MoS2 Films. Angew. Chemie - Int. Ed. 2017, 56, 4991–4995. [Google Scholar] [CrossRef]
- Tan, L.K.; Liu, B.; Teng, J.H.; Guo, S.; Low, H.Y.; Loh, K.P. Atomic layer deposition of a MoS2 film. Nanoscale 2014, 6, 10584–10588. [Google Scholar] [CrossRef] [PubMed]
- Mattinen, M.; Hatanpää, T.; Sarnet, T.; Mizohata, K.; Meinander, K.; King, P.J.; Khriachtchev, L.; Räisänen, J.; Ritala, M.; Leskelä, M. Atomic Layer Deposition of Crystalline MoS2 Thin Films: New Molybdenum Precursor for Low-Temperature Film Growth. Adv. Mater. Interfaces 2017, 4, 1700123. [Google Scholar] [CrossRef]
- Jin, Z.; Shin, S.; Kwon, D.H.; Han, S.J.; Min, Y.S. Novel chemical route for atomic layer deposition of MoS2 thin film on SiO2/Si substrate. Nanoscale 2014, 6, 14453–14458. [Google Scholar] [CrossRef]
- Browning, R.; Padigi, P.; Solanki, R.; Tweet, D.J.; Schuele, P.; Evans, D. Atomic layer deposition of MoS2 thin films. Mater. Res. Express 2015, 2, 12–17. [Google Scholar] [CrossRef]
- Liu, H.; Chen, L.; Zhu, H.; Sun, Q.Q.; Ding, S.J.; Zhou, P.; Zhang, D.W. Atomic layer deposited 2D MoS2 atomic crystals: From material to circuit. Nano Res. 2020, 13, 1644–1650. [Google Scholar] [CrossRef]
- Chen, C.; Raza, M.H.; Amsalem, P.; Schultz, T.; Koch, N.; Pinna, N. Morphology-Controlled MoS2 by Low-Temperature Atomic Layer Deposition. Nanoscale 2020, 12, 20404–20412. [Google Scholar]
- Yang, J.; Liu, L. Nanotribological properties of 2-D MoS2 on different substrates made by atomic layer deposition (ALD). Appl. Surf. Sci. 2020, 502, 144402. [Google Scholar] [CrossRef]
- Huang, Y.; Liu, L.; Sha, J.; Chen, Y. Size-dependent piezoelectricity of molybdenum disulfide (MoS2) films obtained by atomic layer deposition (ALD). Appl. Phys. Lett. 2017, 111, 063902. [Google Scholar] [CrossRef]
- Jang, Y.; Yeo, S.; Lee, H.B.R.; Kim, H.; Kim, S.H. Wafer-scale, conformal and direct growth of MoS 2 thin films by atomic layer deposition. Appl. Surf. Sci. 2016, 365, 160–165. [Google Scholar] [CrossRef]
- Pandiyan, R.; Oulad Elhmaidi, Z.; Sekkat, Z.; Abd-lefdil, M.; El Khakani, M.A. Reconstructing the energy band electronic structure of pulsed laser deposited CZTS thin films intended for solar cell absorber applications. Appl. Surf. Sci. 2017, 396, 1562–1570. [Google Scholar] [CrossRef]
- Brassard, D.; El Khakani, M.A. Pulsed-laser deposition of high- k titanium silicate thin films. J. Appl. Phys. 2005, 98, 054912. [Google Scholar] [CrossRef]
- Daghrir, R.; Drogui, P.; Dimboukou-Mpira, A.; El Khakani, M.A. Photoelectrocatalytic degradation of carbamazepine using Ti/TiO2 nanostructured electrodes deposited by means of a pulsed laser deposition process. Chemosphere 2013, 93, 2756–2766. [Google Scholar] [CrossRef]
- Ka, I.; Le Borgne, V.; Ma, D.; El Khakani, M.A. Pulsed laser ablation based direct synthesis of single-wall carbon nanotube/PbS quantum dot nanohybrids exhibiting strong, spectrally wide and fast photoresponse. Adv. Mater. 2012, 24, 6289–6294. [Google Scholar] [CrossRef]
- Late, D.J.; Shaikh, P.A.; Khare, R.; Kashid, R.V.; Chaudhary, M.; More, M.A.; Ogale, S.B. Pulsed laser-deposited MoS2 thin films on W and Si: Field emission and photoresponse studies. ACS Appl. Mater. Interfaces 2014, 6, 15881–15888. [Google Scholar] [CrossRef] [PubMed]
- Rai, R.H.; Pérez-Pacheco, A.; Quispe-Siccha, R.; Glavin, N.R.; Muratore, C. Pulsed laser annealing of amorphous two-dimensional transition metal dichalcogenides. J. Vac. Sci. Technol. A 2020, 38, 052201. [Google Scholar] [CrossRef]
- Wang, R.; Sun, P.; Wang, H.; Wang, X. Pulsed laser deposition of amorphous molybdenum disulfide films for efficient hydrogen evolution reaction. Electrochim. Acta 2017, 258, 876–882. [Google Scholar] [CrossRef]
- Loh, T.A.J.; Chua, D.H.C. Growth mechanism of pulsed laser fabricated few-layer MoS2 on metal substrates. ACS Appl. Mater. Interfaces 2014, 6, 15966–15971. [Google Scholar] [CrossRef] [PubMed]
- McDevitt, N.T.; Bultman, J.E.; Zabinski, J.S. Study of amorphous MoS2 films grown by pulsed laser deposition. Appl. Spectrosc. 1998, 52, 1160–1164. [Google Scholar] [CrossRef]
- Mosleh, M.; Laube, S.J.P.; Suh, N.P. Friction of undulated surfaces coated with mos2 by pulsed laser deposition. Tribol. Trans. 1999, 42, 495–502. [Google Scholar] [CrossRef]
- Serrao, C.R.; Diamond, A.M.; Hsu, S.L.; You, L.; Gadgil, S.; Clarkson, J.; Carraro, C.; Maboudian, R.; Hu, C.; Salahuddin, S. Highly crystalline MoS2 thin films grown by pulsed laser deposition. Appl. Phys. Lett. 2015, 106, 052101. [Google Scholar] [CrossRef]
- Barvat, A.; Prakash, N.; Singh, D.K.; Dogra, A.; Khanna, S.P.; Singh, S.; Pal, P. Mixed Phase Compositions of MoS2 Ultra Thin Film Grown by Pulsed Laser Deposition. Mater. Today Proc. 2018, 5, 2241–2245. [Google Scholar] [CrossRef]
- Siegel, G.; Venkata Subbaiah, Y.P.; Prestgard, M.C.; Tiwari, A. Growth of centimeter-scale atomically thin MoSfilms by pulsed laser deposition. APL Mater. 2015, 3, 056103. [Google Scholar] [CrossRef]
- Kumar, S.; Sharma, A.; Ho, Y.T.; Pandey, A.; Tomar, M.; Kapoor, A.K.; Chang, E.Y.; Gupta, V. High performance UV photodetector based on MoS2 layers grown by pulsed laser deposition technique. J. Alloys Compd. 2020, 835, 155222. [Google Scholar] [CrossRef]
- Lopez-Sanchez, O.; Lembke, D.; Kayci, M.; Radenovic, A.; Kis, A. Ultrasensitive photodetectors based on monolayer MoS 2. Nat. Nanotechnol. 2013, 8, 497–501. [Google Scholar] [CrossRef] [PubMed]
- Alkis, S.; Öztaş, T.; Aygün, L.E.; Bozkurt, F.; Okyay, A.K.; Ortaç, B. Thin film MoS_2 nanocrystal based ultraviolet photodetector. Opt. Express 2012, 20, 21815. [Google Scholar] [CrossRef] [PubMed]
- Huo, N.; Konstantatos, G. Ultrasensitive all-2D MoS2 phototransistors enabled by an out-of-plane MoS2 PN homojunction. Nat. Commun. 2017, 8, 1–6. [Google Scholar] [CrossRef]
- Tsai, D.S.; Liu, K.K.; Lien, D.H.; Tsai, M.L.; Kang, C.F.; Lin, C.A.; Li, L.J.; He, J.H. Few-layer MoS2 with high broadband photogain and fast optical switching for use in harsh environments. ACS Nano 2013, 7, 3905–3911. [Google Scholar] [CrossRef] [PubMed]
- Goel, N.; Kumar, R.; Roul, B.; Kumar, M.; Krupanidhi, S.B. Wafer-scale synthesis of a uniform film of few-layer MoS2 on GaN for 2D heterojunction ultraviolet photodetector. J. Phys. D. Appl. Phys. 2018, 51, 374003. [Google Scholar] [CrossRef]
- Donley, M.S.; Murray, P.T.; Barber, S.A.; Haas, T.W. Deposition and properties of MoS2 thin films grown by pulsed laser evaporation. Surf. Coatings Technol. 1988, 36, 329–340. [Google Scholar] [CrossRef]
- Walck, S.D.; Donley, M.S.; Zabinski, J.S.; Dyhouse, V.J. Characterization of Pulsed Laser Deposited PbO/MoS2 by Transmission Electron Microscopy. J. Mater. Res. 1994, 9, 236–245. [Google Scholar] [CrossRef]
- Barvat, A.; Prakash, N.; Satpati, B.; Singha, S.S.; Kumar, G.; Singh, D.K.; Dogra, A.; Khanna, S.P.; Singha, A.; Pal, P. Emerging photoluminescence from bilayer large-area 2D MoS2 films grown by pulsed laser deposition on different substrates. J. Appl. Phys. 2017, 122, 015304. [Google Scholar] [CrossRef]
- Wang, R.; Shao, Q.; Yuan, Q.; Sun, P.; Nie, R.; Wang, X. Direct growth of high-content 1T phase MoS2 film by pulsed laser deposition for hydrogen evolution reaction. Appl. Surf. Sci. 2020, 504, 144320. [Google Scholar] [CrossRef]
- Wang, S.; Yu, H.; Zhang, H.; Wang, A.; Zhao, M.; Chen, Y.; Mei, L.; Wang, J. Broadband few-layer MoS2 saturable absorbers. Adv. Mater. 2014, 26, 3538–3544. [Google Scholar] [CrossRef]
- Fominski, V.Y.; Markeev, A.M.; Nevolin, V.N.; Prokopenko, V.B.; Vrublevski, A.R. Pulsed laser deposition of MoSx films in a buffer gas atmosphere. Thin Solid Films 1994, 248, 240–246. [Google Scholar] [CrossRef]
- Jiao, L.; Jie, W.; Yang, Z.; Wang, Y.; Chen, Z.; Zhang, X.; Tang, W.; Wu, Z.; Hao, J. Layer-dependent photoresponse of 2D MoS 2 films prepared by pulsed laser deposition. J. Mater. Chem. C 2019, 7, 2522–2529. [Google Scholar] [CrossRef]
- Serna, M.I.; Yoo, S.H.; Moreno, S.; Xi, Y.; Oviedo, J.P.; Choi, H.; Alshareef, H.N.; Kim, M.J.; Minary-Jolandan, M.; Quevedo-Lopez, M.A. Large-Area Deposition of MoS2 by Pulsed Laser Deposition with in Situ Thickness Control. ACS Nano 2016, 10, 6054–6061. [Google Scholar] [CrossRef] [PubMed]
- Jiao, L.; Wang, Y.; Zhi, Y.; Cui, W.; Chen, Z.; Zhang, X.; Jie, W.; Wu, Z. Fabrication and Characterization of Two-Dimensional Layered MoS2 Thin Films by Pulsed Laser Deposition. Adv. Condens. Matter Phys. 2018, 2018, 23–28. [Google Scholar] [CrossRef]
- Pradhan, G.; Sharma, A.K. Anomalous Raman and photoluminescence blue shift in mono- and a few layered pulsed laser deposited MoS2 thin films. Mater. Res. Bull. 2018, 102, 406–411. [Google Scholar] [CrossRef]
- Walck, S.D.; Zabinski, J.S.; Donley, M.S.; Bultman, J.E. Evolution of surface topography in pulsed-laser-deposited thin films of MoS2. Surf. Coatings Technol. 1993, 62, 412–416. [Google Scholar] [CrossRef]
- Ho, Y.T.; Ma, C.H.; Luong, T.T.; Wei, L.L.; Yen, T.C.; Hsu, W.T.; Chang, W.H.; Chu, Y.C.; Tu, Y.Y.; Pande, K.P.; et al. Layered MoS2 grown on c -sapphire by pulsed laser deposition. Phys. Status Solidi - Rapid Res. Lett. 2015, 9, 187–191. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, S.; Yu, H.; Zhang, H.; Chen, Y.; Mei, L.; Di Lieto, A.; Tonelli, M.; Wang, J. Atomic-layer molybdenum sulfide optical modulator for visible coherent light. Sci. Rep. 2015, 5, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, S.; Wang, D.; Yu, H.; Zhang, H.; Chen, Y.; Mei, L.; Di Lieto, A.; Tonelli, M.; Wang, J. Atomic-layer molybdenum sulfide passively modulated green laser pulses. IEEE Photonics Technol. Lett. 2016, 28, 197–200. [Google Scholar] [CrossRef]
- Miao, P.; Ma, Y.; Sun, M.; Li, J.; Xu, P. Tuning the SERS activity and plasmon-driven reduction of p-nitrothiophenol on a Ag@MoS2 film. Faraday Discuss. 2019, 214, 297–307. [Google Scholar] [CrossRef] [PubMed]
- Xie, M.Z.; Zhou, J.Y.; Ji, H.; Ye, Y.; Wang, X.; Jiang, K.; Shang, L.Y.; Hu, Z.G.; Chu, J.H. Annealing effects on sulfur vacancies and electronic transport of MoS2 films grown by pulsed-laser deposition. Appl. Phys. Lett. 2019, 115, 121901. [Google Scholar]
- Su, B.; He, H.; Ye, Z. Large-area ZnO/MoS2 heterostructure grown by pulsed laser deposition. Mater. Lett. 2019, 253, 187–190. [Google Scholar] [CrossRef]
- Pang, X.; Zhang, Q.; Shao, Y.; Liu, M.; Zhang, D.; Zhao, Y. A flexible pressure sensor based on magnetron sputtered mos2. Sensors (Switzerland) 2021, 21, 1130. [Google Scholar] [CrossRef]
- Tao, J.; Chai, J.; Lu, X.; Wong, L.M.; Wong, T.I.; Pan, J.; Xiong, Q.; Chi, D.; Wang, S. Growth of wafer-scale MoS2 monolayer by magnetron sputtering. Nanoscale 2015, 7, 2497–2503. [Google Scholar] [CrossRef]
- Kaindl, R.; Bayer, B.C.; Resel, R.; Müller, T.; Skakalova, V.; Habler, G.; Abart, R.; Cherevan, A.S.; Eder, D.; Blatter, M.; et al. Growth, structure and stability of sputter-deposited MoS2 thin films. Beilstein J. Nanotechnol. 2017, 8, 1115–1126. [Google Scholar] [CrossRef] [PubMed]
- Rowley-Neale, S.J.; Ratova, M.; Fugita, L.T.N.; Smith, G.C.; Gaffar, A.; Kulczyk-Malecka, J.; Kelly, P.J.; Banks, C.E. Magnetron Sputter-Coated Nanoparticle MoS2 Supported on Nanocarbon: A Highly Efficient Electrocatalyst toward the Hydrogen Evolution Reaction. ACS Omega 2018, 3, 7235–7242. [Google Scholar] [CrossRef]
- Tian, L.; Wu, R.; Liu, H.Y. Synthesis of Au-nanoparticle-loaded 1T@2H-MoS 2 nanosheets with high photocatalytic performance. J. Mater. Sci. 2019, 54, 9656–9665. [Google Scholar] [CrossRef]
- Nan, H.; Wang, Z.; Wang, W.; Liang, Z.; Lu, Y.; Chen, Q.; He, D.; Tan, P.; Miao, F.; Wang, X.; et al. Strong photoluminescence enhancement of MoS2 through defect engineering and oxygen bonding. ACS Nano 2014, 8, 5738–5745. [Google Scholar] [CrossRef]
- Feng, Y.; Zhang, K.; Li, H.; Wang, F.; Zhou, B.; Fang, M.; Wang, W.; Wei, J.; Wong, H.S.P. In situ visualization and detection of surface potential variation of mono and multilayer MoS2 under different humidities using Kelvin probe force microscopy. Nanotechnology 2017, 28, 295705. [Google Scholar] [CrossRef]
- Mouri, S.; Miyauchi, Y.; Matsuda, K. Tunable Photoluminescence of Monolayer MoS2 via Chemical Doping. Nano Lett. 2013, 13, 5944–5948. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Yan, H.; Brus, L.E.; Heinz, T.F.; Hone, J.; Ryu, S. Anomalous lattice vibrations of single- and few-layer MoS2. ACS Nano 2010, 4, 2695–2700. [Google Scholar] [CrossRef]
- Mak, K.F.; Lee, C.; Hone, J.; Shan, J.; Heinz, T.F. Atomically thin MoS2: A new direct-gap semiconductor. Phys. Rev. Lett. 2010, 105, 2–5. [Google Scholar] [CrossRef] [PubMed]
- Cheiwchanchamnangij, T.; Lambrecht, W.R.L. Quasiparticle band structure calculation of monolayer, bilayer, and bulk MoS 2. Phys. Rev. B - Condens. Matter Mater. Phys. 2012, 85, 205302. [Google Scholar] [CrossRef]
- Li, H.; Zhang, Q.; Yap, C.C.R.; Tay, B.K.; Edwin, T.H.T.; Olivier, A.; Baillargeat, D. From bulk to monolayer MoS 2: Evolution of Raman scattering. Adv. Funct. Mater. 2012, 22, 1385–1390. [Google Scholar] [CrossRef]
- Ahmad, S.; Mukherjee, S. A Comparative Study of Electronic Properties of Bulk MoS2 and Its Monolayer Using DFT Technique: Application of Mechanical Strain on MoS2 Monolayer. Graphene 2014, 03, 52–59. [Google Scholar] [CrossRef]
- Erfanifam, S.; Jamilpanah, L.; Sangpour, P.; Haddadi, F.; Hamdi, M.; Erfanifam, M.; Chanda, G.; Herrmannsdörfer, T.; Sazgari, V.; Sadeghi, A.; et al. Electrical and optical properties of MoS2,MoOx=2,3(MoSO)/RGO heterostructure. arXiv 2018, 3, 1–6. [Google Scholar]
- Zhang, Z.; Qian, Q.; Li, B.; Chen, K.J. Interface Engineering of Monolayer MoS2/GaN Hybrid Heterostructure: Modified Band Alignment for Photocatalytic Water Splitting Application by Nitridation Treatment. ACS Appl. Mater. Interfaces 2018, 10, 17419–17426. [Google Scholar] [CrossRef]
- Dolui, K.; Rungger, I.; Das Pemmaraju, C.; Sanvito, S. Possible doping strategies for MoS2 monolayers: An ab initio study. Phys. Rev. B - Condens. Matter Mater. Phys. 2013, 88, 075420. [Google Scholar] [CrossRef]
- Zahid, F.; Liu, L.; Zhu, Y.; Wang, J.; Guo, H. A generic tight-binding model for monolayer, bilayer and bulk MoS 2. AIP Adv. 2013, 3, 052111. [Google Scholar] [CrossRef]
- Liang, M.; Ali, A.; Belaidi, A.; Hossain, M.I.; Ronan, O.; Downing, C.; Tabet, N.; Sanvito, S.; EI-Mellouhi, F.; Nicolosi, V. Improving stability of organometallic-halide perovskite solar cells using exfoliation two-dimensional molybdenum chalcogenides. npj 2D Mater. Appl. 2020, 4, 1–8. [Google Scholar] [CrossRef]
- Singh, E.; Kim, K.S.; Yeom, G.Y.; Nalwa, H.S. Atomically thin-layered molybdenum disulfide (MoS2) for bulk-heterojunction solar cells. ACS Appl. Mater. Interfaces 2017, 9, 3223–3245. [Google Scholar] [CrossRef]
- J rvinen, T.; Lorite, G.S.; Per ntie, J.; Toth, G.; Saarakkala, S.; Virtanen, V.K.; Kordas, K. WS2 and MoS2 thin film gas sensors with high response to NH3 in air at low temperature. Nanotechnology 2019, 30, 405501. [Google Scholar] [CrossRef]
- Perkins, F.K.; Friedman, A.L.; Cobas, E.; Campbell, P.M.; Jernigan, G.G.; Jonker, B.T. Chemical Vapor Sensing with Monolayer MoS2. Nano Lett. 2013, 13, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, B.; Simsek, E. Utilization of monolayer MoS2 in Bragg stacks and metamaterial structures as broadband absorbers. Opt. Commun. 2016, 369, 89–93. [Google Scholar] [CrossRef]
- He, X.; Liu, F.; Lin, F.; Xiao, G.; Shi, W. Tunable MoS2 modified hybrid surface plasmon waveguides. Nanotechnology 2019, 30, 125201. [Google Scholar] [CrossRef] [PubMed]
- Mawlong, L.P.L.; Paul, K.K.; Giri, P.K. Exciton-plasmon coupling and giant photoluminescence enhancement in monolayer MoS2 through hierarchically designed TiO2/Au/MoS2 ternary core − Shell heterostructure. Nanotechnology 2021, 32, 215201. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Pan, R.; Tian, S.; Gu, C.; Li, J. Plasmonic hybrids of mos2 and 10-nm nanogap arrays for photoluminescence enhancement. Micromachines 2020, 11, 1109. [Google Scholar] [CrossRef]
- Rahmati, B.; Hajzadeh, I.; Taheri, M.; Karimzadeh, R.; Mohajerzadeh, S.; Mohseni, S.M. Plasmonic improvement photoresponse of vertical-MoS2 nanostructure photodetector by Au nanoparticles. Appl. Surf. Sci. 2019, 490, 165–171. [Google Scholar] [CrossRef]
- Tsai, M.L.; Su, S.H.; Chang, J.K.; Tsai, D.S.; Chen, C.H.; Wu, C.I.; Li, L.J.; Chen, L.J.; He, J.H. Monolayer MoS2 heterojunction solar cells. ACS Nano 2014, 8, 8317–8322. [Google Scholar] [CrossRef]
- Burgelman, M.; Verschraegen, J.; Minnaert, B.; Marlein, J. Numerical simulation of thin film solar cells: Practical exercises with SCAPS. Numos Work. 2007, 357–366. [Google Scholar]
- Burgelman, M.; Verschraegen, J.; Degrave, S.; Nollet, P. Modeling thin-film PV devices. Prog. Photovoltaics Res. Appl. 2004, 12, 143–153. [Google Scholar] [CrossRef]
- Verschraegen, J.; Burgelman, M. Numerical modeling of intra-band tunneling for heterojunction solar cells in scaps. Thin Solid Films 2007, 515, 6276–6279. [Google Scholar] [CrossRef]
- Chen, X.; Wu, Z.; Xu, S.; Wang, L.; Huang, R.; Han, Y.; Ye, W.; Xiong, W.; Han, T.; Long, G.; et al. Probing the electron states and metal-insulator transition mechanisms in molybdenum disulphide vertical heterostructures. Nat. Commun. 2015, 6, 1–8. [Google Scholar] [CrossRef]
- Rashid, H.; Rahman, K.S.; Hossain, M.I.; Tabet, N.; Alharbi, F.H.; Amin, N. Prospects of molybdenum disulfide (MoS2) as an alternative absorber layer material in thin film solar cells from numerical modeling. Chalcogenide Lett. 2014, 11, 397–403. [Google Scholar]
- Deng, Q.; Li, Y.; Shen, Y.; Chen, L.; Wang, G.; Wang, S. Numerical simulation on n-MoS2/p-Si heterojunction solar cells. Mod. Phys. Lett. B 2017, 31, 1750079. [Google Scholar] [CrossRef]
- Tousif, N.; Mohammad, S.; Ferdous, A.A.; Hoque, A. Investigation of Different Materials as Buffer Layer in CZTS Solar Cells Using SCAPS. J. Clean Energy Technol. 2018, 6, 293–296. [Google Scholar] [CrossRef]
- Heidariramsheh, M.; Haghighi, M.; Dabbagh, M.M.; Mahdavi, S.M. Pure sulfide Cu2ZnSnS4 layers through a one-step low-temperature PLD technique: Insight into simulation on modified back contact to overcome the barrier of MoS2. Mater. Sci. Eng. B Solid-State Mater. Adv. Technol. 2020, 262, 114701. [Google Scholar] [CrossRef]
- Bouarissa, A.; Gueddim, A.; Bouarissa, N.; Maghraoui-Meherezi, H. Modeling of ZnO/MoS2/CZTS photovoltaic solar cell through window, buffer and absorber layers optimization. Mater. Sci. Eng. B Solid-State Mater. Adv. Technol. 2021, 263, 114816. [Google Scholar] [CrossRef]
- Zaidi, B.; Ullah, M.S.; Hadjoudja, B.; Gagui, S.; Houaidji, N.; Chouial, B.; Shekhar, C. Role of TCO films in improving the efficiency of CdS/MoS 2 heterojunction solar cells. J. Nano- Electron. Phys. 2019, 11, 4–7. [Google Scholar] [CrossRef]
- Kohnehpoushi, S.; Nazari, P.; Nejand, B.A.; Eskandari, M. MoS2: A two-dimensional hole-transporting material for high-efficiency, low-cost perovskite solar cells. Nanotechnology 2018, 29, 205201. [Google Scholar] [CrossRef] [PubMed]
- Joshi, N.; Hayasaka, T.; Liu, Y.; Liu, H.; Oliveira, O.N.; Lin, L. A review on chemiresistive room temperature gas sensors based on metal oxide nanostructures, graphene and 2D transition metal dichalcogenides. Microchim. Acta 2018, 185, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Donarelli, M.; Ottaviano, L. 2d materials for gas sensing applications: A review on graphene oxide, mos2, ws2 and phosphorene. Sensors (Switzerland) 2018, 18, 3638. [Google Scholar] [CrossRef] [PubMed]
- Ramanathan, A.A. Defect Functionalization of MoS2 nanostructures as toxic gas sensors: A review. IOP Conf. Ser. Mater. Sci. Eng. 2018, 305, 012001. [Google Scholar] [CrossRef]
- Li, W.; Zhang, Y.; Long, X.; Cao, J.; Xin, X.; Guan, X.; Peng, J.; Zheng, X. Gas Sensors Based on Mechanically Exfoliated MoS2 Nanosheets for Room-Temperature NO2 Detection. Sensors (Basel). 2019, 19, 2123. [Google Scholar] [CrossRef]
- Cho, B.; Hahm, M.G.; Choi, M.; Yoon, J.; Kim, A.R.; Lee, Y.J.; Park, S.G.; Kwon, J.D.; Kim, C.S.; Song, M.; et al. Charge-transfer-based gas sensing using atomic-layer MoS2. Sci. Rep. 2015, 5, 8052. [Google Scholar] [CrossRef]
- Zhang, Y.; Guo, S.; Xin, X.; Song, Y.; Yang, L.; Wang, B.; Tan, L.; Li, X. Plasmonic MoO2 as co-catalyst of MoS2 for enhanced photocatalytic hydrogen evolution. Appl. Surf. Sci. 2020, 504, 144291. [Google Scholar] [CrossRef]
- Li, Y.; Wang, H.; Xie, L.; Liang, Y.; Hong, G.; Dai, H. MoS2 Nanoparticles Grown on Graphene: An Advanced Catalyst for the Hydrogen Evolution Reaction. J. Am. Chem. Soc. 2011, 133, 7296–7299. [Google Scholar] [CrossRef]
- Chaojian, H.; Bo, L.; Qingwei, L.; Lijun, Y.; Yang, W.; Zhan, Y.; Lixin, D. Plasmon-Enhanced Photovoltaic Characteristics of Black Phosphorus-MoS 2 Heterojunction. IEEE Open J. Nanotechnol. 2021, 2, 41–51. [Google Scholar] [CrossRef]
- Mukherjee, B.; Tseng, F.; Gunlycke, D.; Amara, K.K.; Eda, G.; Simsek, E. Complex electrical permittivity of the monolayer molybdenum disulfide (MoS_2) in near UV and visible. Opt. Mater. Express 2015, 5, 447. [Google Scholar] [CrossRef]
- Jiang, Y.; Chen, W.; Wang, J. Broadband MoS 2 -based absorber investigated by a generalized interference theory. Opt. Express 2018, 26, 24403. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Lu, L.; Cheng, Q.; Luo, Z. Surface plasmon-enhanced optical absorption in monolayer MoS2 with one-dimensional Au grating. J. Quant. Spectrosc. Radiat. Transf. 2018, 211, 138–143. [Google Scholar] [CrossRef]
- Chen, W.; Wang, L.; Jiang, Y.; Wang, J. A Perfect Absorber Based on Monolayer MoS2 and Nano-Silver in the Visible Regime. 2018 Int. Conf. Microw. Millim. Wave Technol. (ICMMT). IEEE 2018, 1–3. [Google Scholar]
- Camellini, A.; Mazzanti, A.; Mennucci, C.; Martella, C.; Lamperti, A.; Molle, A.; Buatier de Mongeot, F.; Della Valle, G.; Zavelani-Rossi, M. Evidence of Plasmon Enhanced Charge Transfer in Large-Area Hybrid Au–MoS2 Metasurface. Adv. Opt. Mater. 2020, 8, 2000653. [Google Scholar] [CrossRef]
- Muhammad, N.; Chen, Y.; Qiu, C.W.; Wang, G.P. Optical Bound States in Continuum in MoS2-Based Metasurface for Directional Light Emission. Nano Lett. 2021, 21, 967–972. [Google Scholar] [CrossRef]
- Deng, M.; Li, Z.; Rong, X.; Luo, Y.; Li, B.; Zheng, L.; Wang, X.; Lin, F.; Meixner, A.J.; Braun, K.; et al. Light-Controlled Near-Field Energy Transfer in Plasmonic Metasurface Coupled MoS2 Monolayer. Small 2020, 16, 2003539. [Google Scholar] [CrossRef]
- Liu, J.T.; Tong, H.; Wu, Z.H.; Huang, J.B.; Zhou, Y.S. Greatly enhanced light emission of MoS2 using photonic crystal heterojunction. Sci. Rep. 2017, 7, 1–8. [Google Scholar] [CrossRef]
- Xie, Y.; Liang, F.; Chi, S.; Wang, D.; Zhong, K.; Yu, H.; Zhang, H.; Chen, Y.; Wang, J. Defect Engineering of MoS2 for Room-Temperature Terahertz Photodetection. ACS Appl. Mater. Interfaces 2020, 12, 7351–7357. [Google Scholar] [CrossRef] [PubMed]
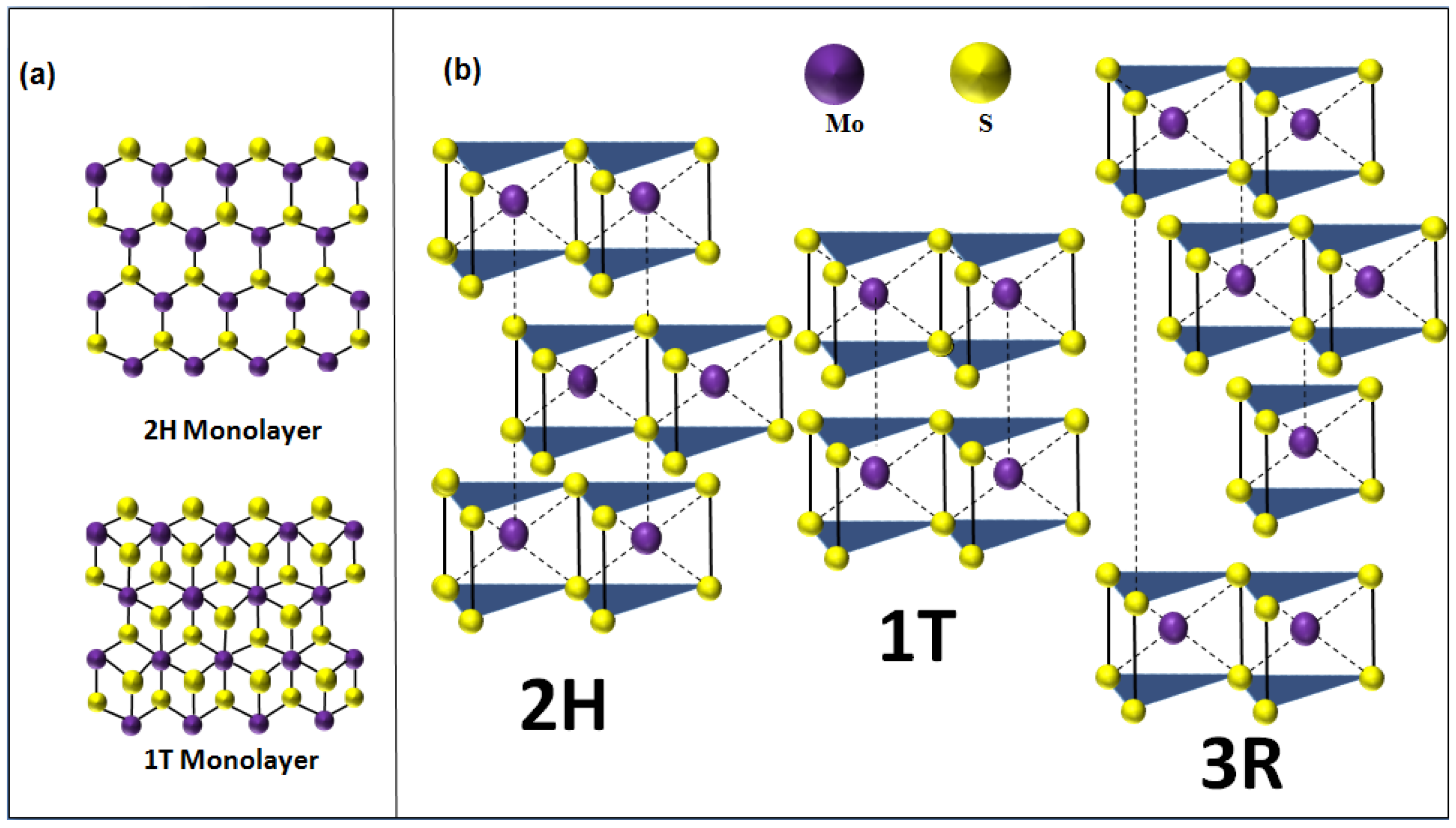
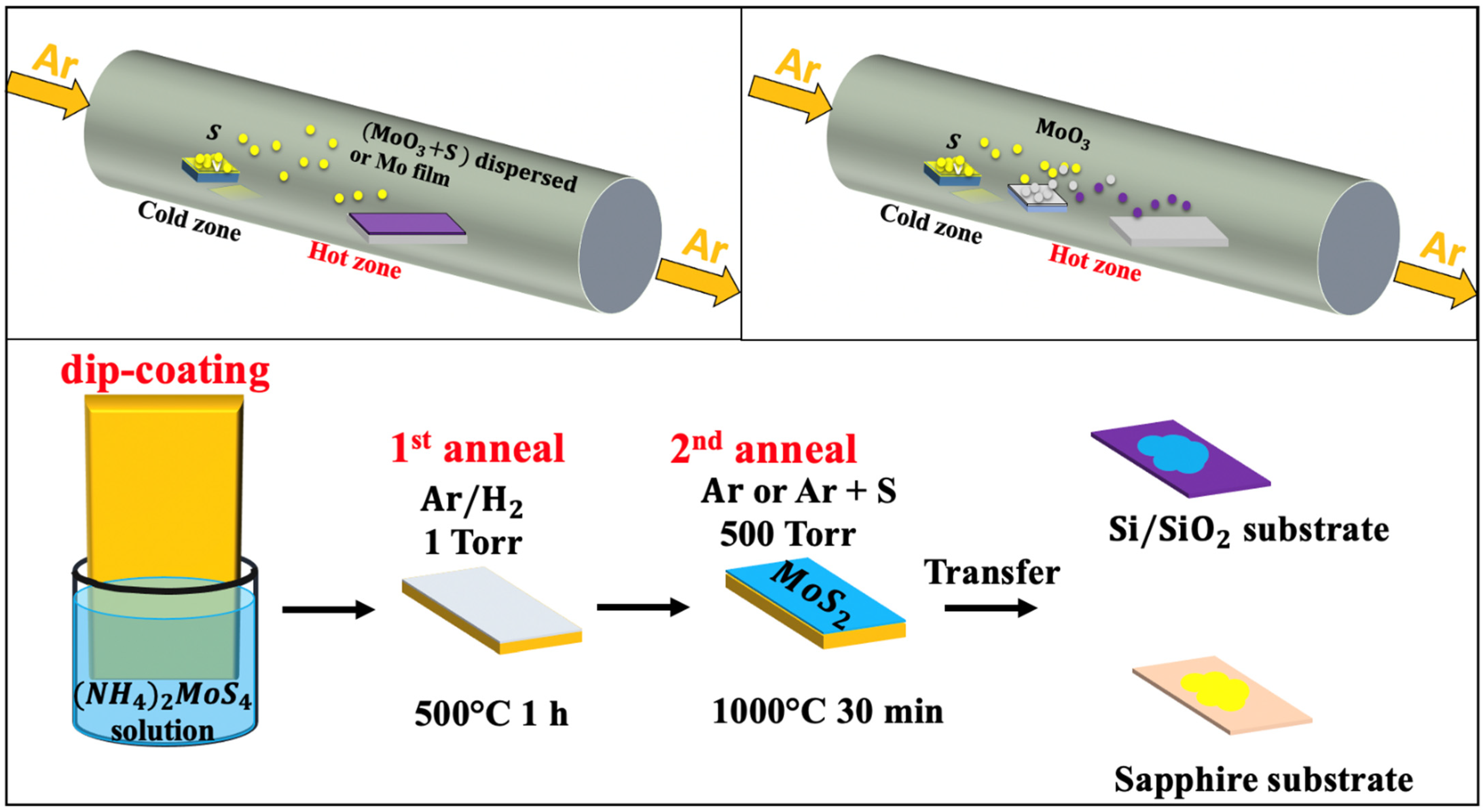
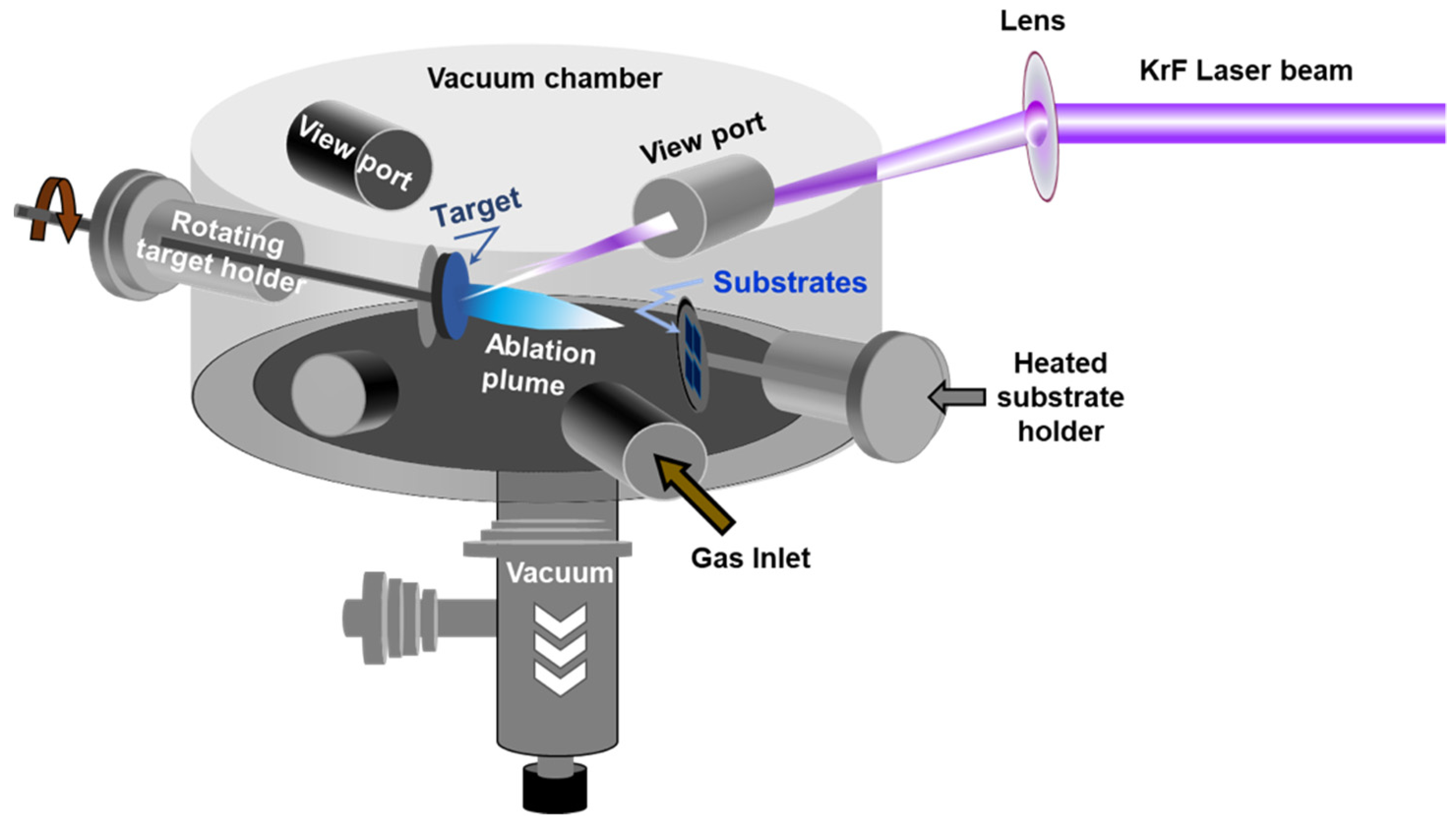
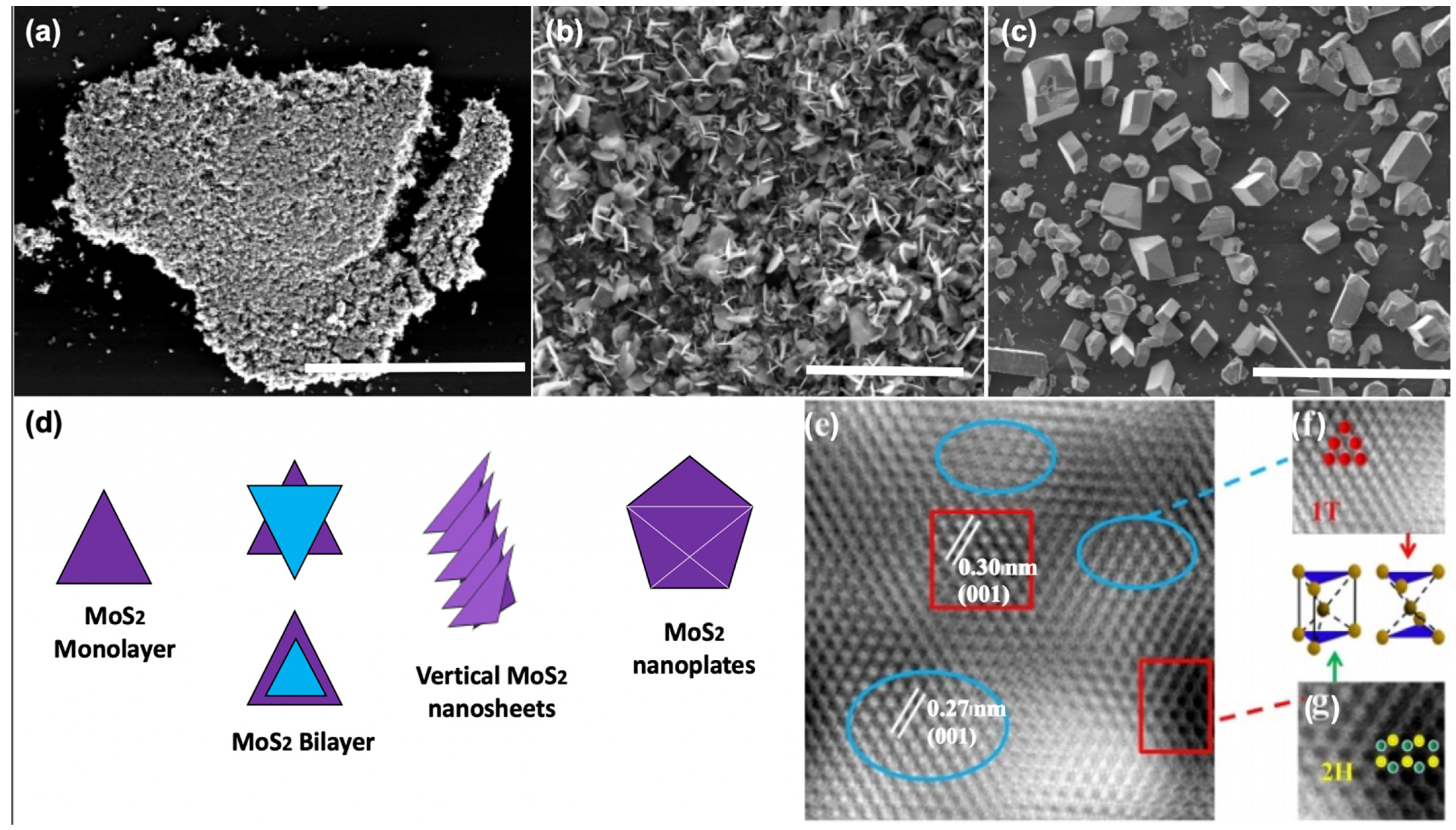
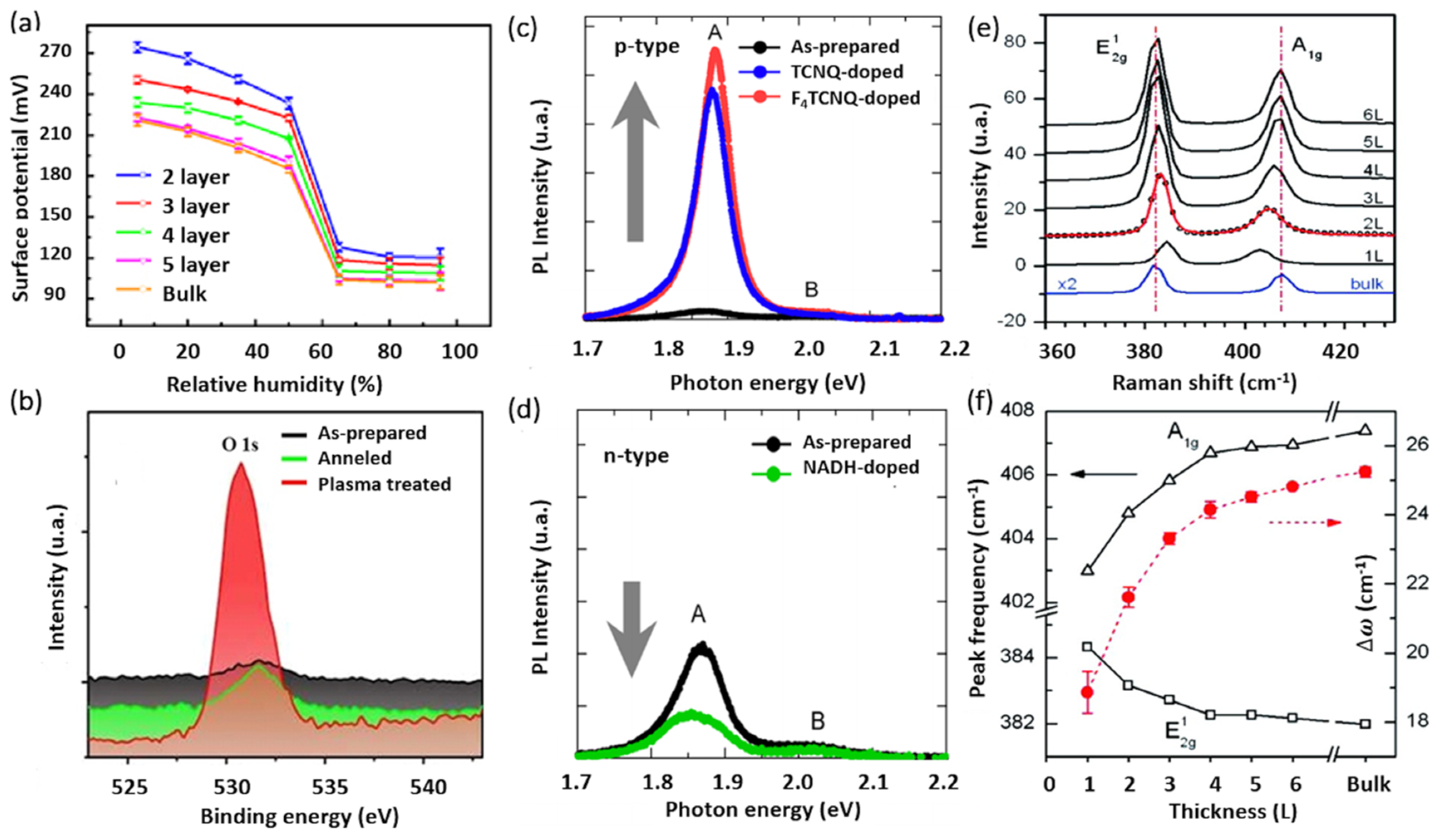
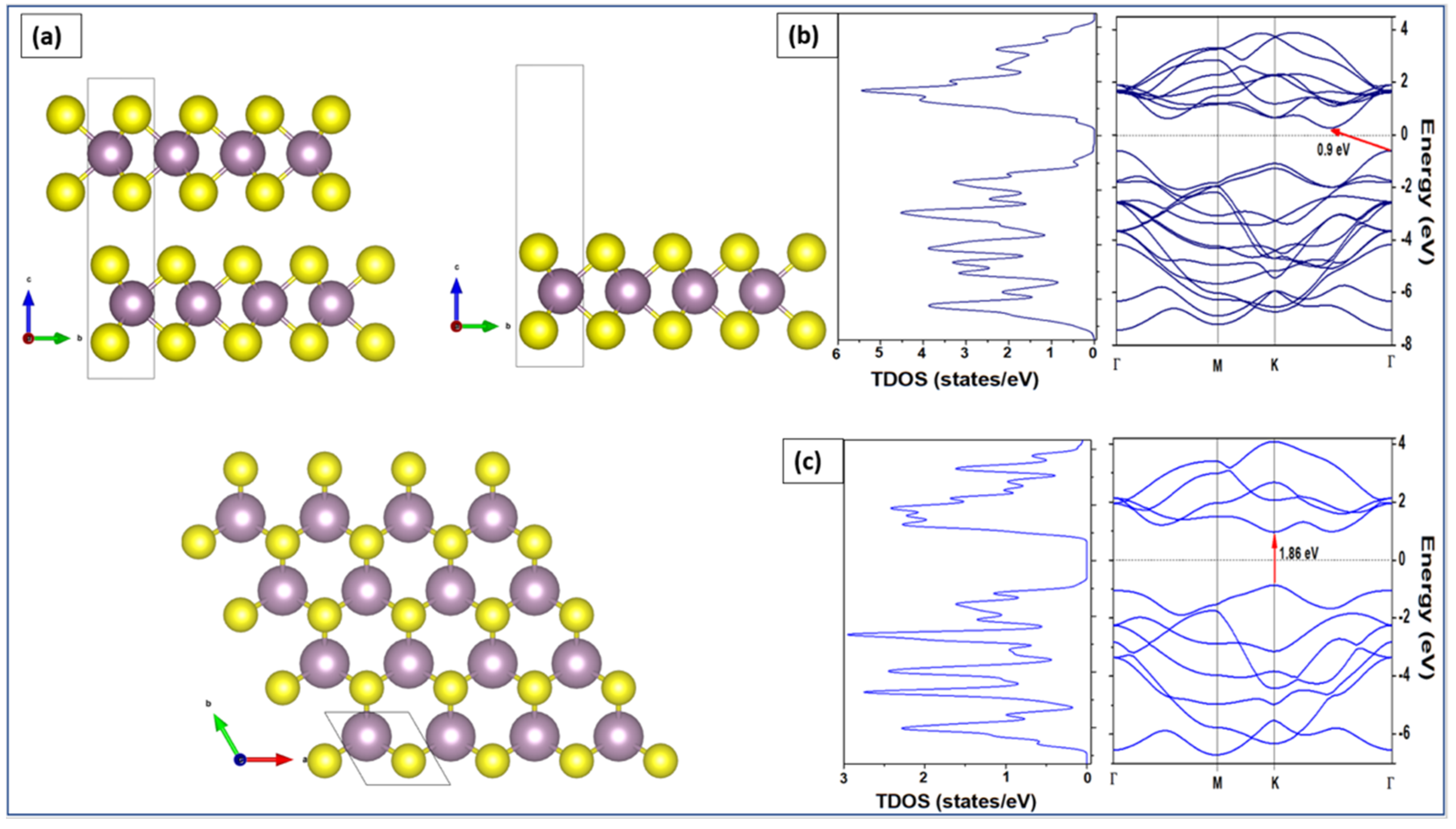
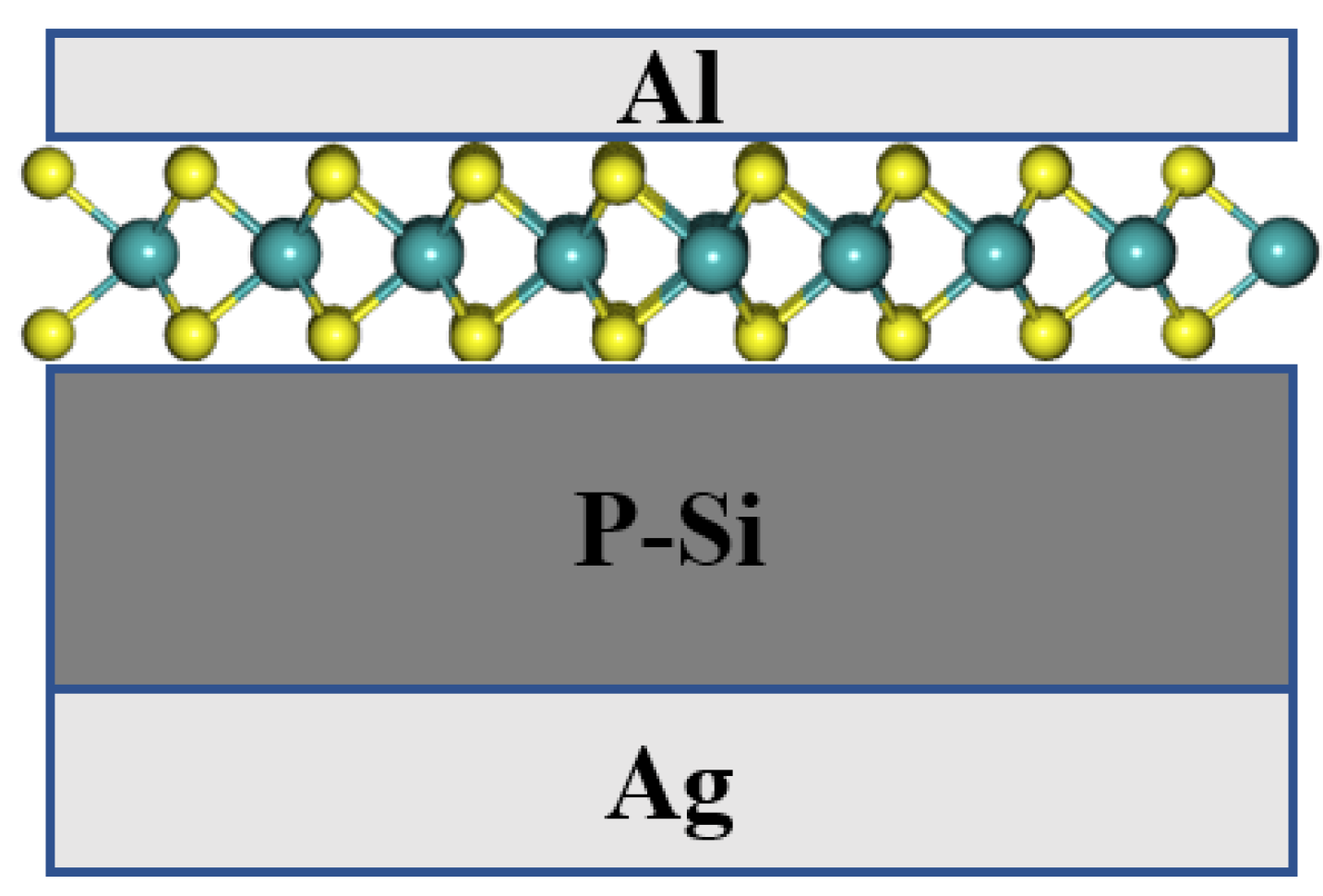
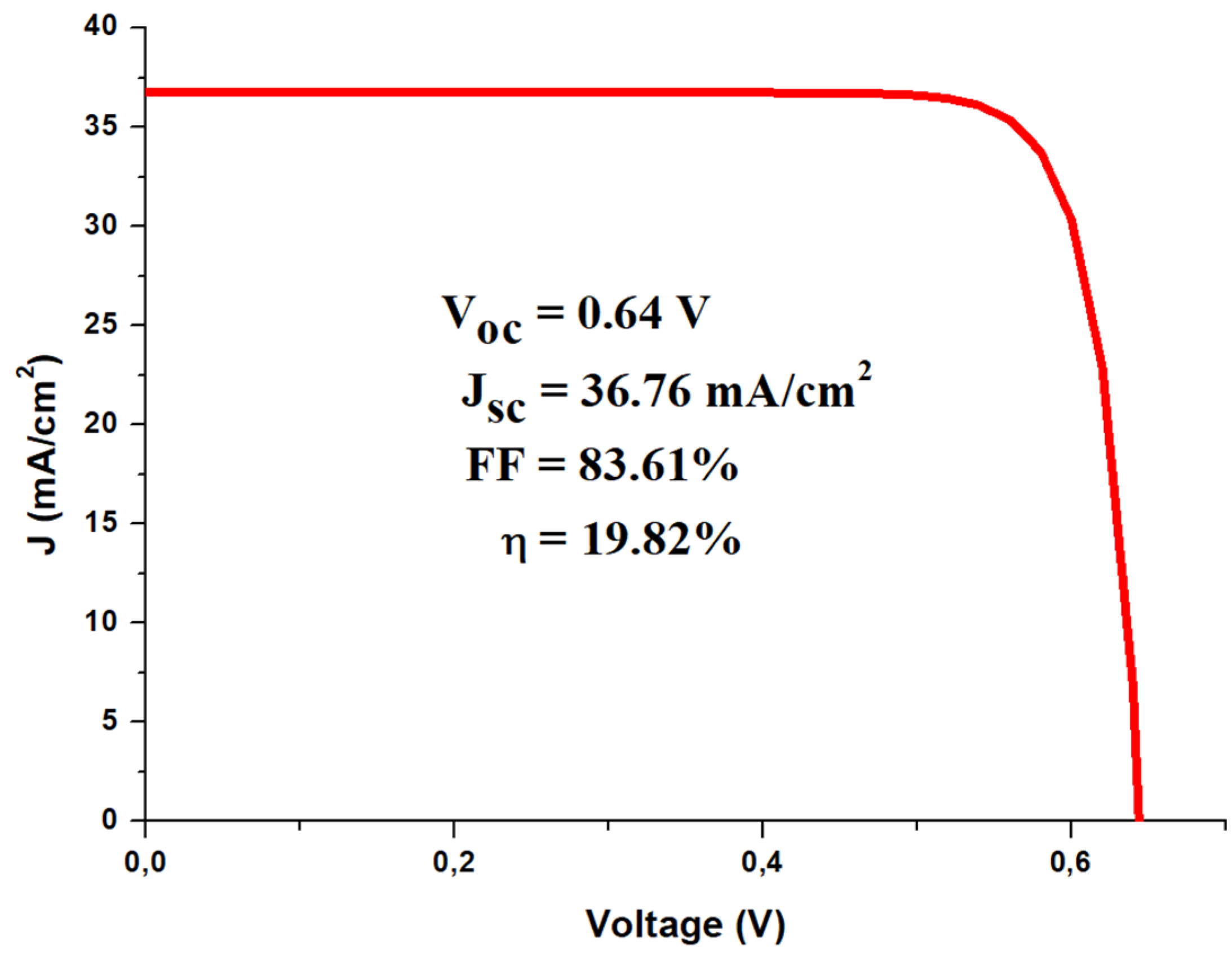
| Polymorphic Structure | Lattice Parameter | Point Group | Electronic Behavior | Ref |
|---|---|---|---|---|
| 1T | a = 5.60 Å, c = 5.99 Å | D6d | Metal | [31] |
| 2H | a = 3.15 Å, c = 12.30 Å | D6h | Semiconductor | [32] |
| 3R | a = 3.17 Å, c = 18.38 Å. | C3v | Semiconductor | [33] |
| Substrate | Precursors | Growth Conditions | Morphology | Ref |
|---|---|---|---|---|
| Si | MoO3 and S powders dispersed on substrate | MoO3 and S powders dispersed on substrate at 850 °C; S powder at 400 °C; Ar-0.725 L/min; time reaction = 30 min | MoS2 nanosheets | [43] |
| Si [001] | S powder and Mo film deposited on substrate | Mo deposited on Silicon at 850 °C, S at 400 °C; Ar-0.725 L/min; time reaction = 30 min | MoS2 nanosheets | [44] |
| Si/SiO2 | S powder and Mo film deposited on substrate | Mo deposited on Silicon at 850 °C, S at 400 °C; Ar-0.725 L/min; time reaction = 30 min | MoS2 nanosheets | [49] |
| Diamond substrate | S powder and Mo deposited on substrate | Mo deposited on Silicon with S powder at 800 °C; N2; ambient pressure; time reaction = 30 min | Horizontally and vertically MoS2 | [73] |
| Si/SiO2 | S powder and MoO3 deposited on substrate | MoO3 film deposited on Silicon at 750–850 °C, 600 mg of S powder at 100 °C; Ar-0.01 L/min; time reaction = 10 min | Mono-to few-layers of MoS2 | [74] |
| Substrate/Setup | MoO3 (mg) | S (mg) | D * (cm) | Gas, Flow (sccm) | T (°C), Time (min) | Morphology | Ref |
|---|---|---|---|---|---|---|---|
| Si face-down | 15 | 80 | 18 | Ar 10 to 500 | 700, 30 | Flake size between 5.1–47.9 µm | [75] |
| SiO2/Si face-up | 10 | 200 | 30 | Ar, 100 | 850, 20 | Monolayer, bilayer and trilayer MoS2 | [76] |
| SiO2/Si face-down | 10 | 100 | – | N2, 20 | 650, 20 | MoS2 monolayer | [77] |
| SiO2/Si face-down | 10-30 | – | 25 | Ar, 150 | 800, 10 | MoS2 triangular flakes | [78] |
| SiO2/Si face-up | 50 | 175 | – | N2, 300 | 750, 15 | MoS2 monolayer with lateral size of 50 µm | [79] |
| Substrate | Precursors | P (Torr) | T (°C) | Cycles | Thickness | Ref |
|---|---|---|---|---|---|---|
| SiO2/Si | Mo hexacarbonyl and dimethyldisulfide | 1.4–3.3 | 100 | 100 | ≈11 nm | [84] |
| SiO2/n-Si | MoCl5 and H2S | 0.75 | 350–450 | 100 | ≈9 nm | [85] |
| Al2O3 | Mo(NMe2)4 and H2S | – | 60 | 100 | ≈12 nm | [81] |
| Al2O3 2-inch wafer | MoCl5 and H2S | 0.001 | 300 | 50 | ≈9 nm | [82] |
| SiO2/Si | Mo(thd)3 (thd = 2,2,6,6 tetramethylheptane 3,5-dionato) and H2S | 3.75 | 300 | 100 | ≈25 nm | [83] |
| Al2O3 c-plane | MoCl5 and hexamethyldisilathiane | 3.75 | 350 | 250 | ≈22 nm | [86] |
| Carbon nanotubes, Si-wafers and glass | bis(tbutylimino)bis(dimethylamino) Mo (VI) and H2S | 300 | 100–250 | 100 | ≈11 nm | [87] |
| Si, SiO2, Al2O3 | MoCl5 and H2S | 3.75 | 430–480 | 1 | 1 layer | [88] |
| Si | MoCl5 and H2S | – | 390–480 | 100 | ≈21.5 nm | [89] |
| SiO2 | Mo hexacarbonyl and H2S | – | 175 | 100 | ≈5 nm | [90] |
| Substrate | Target | P(Pa) | T(°C) | Laser Energy | Thickness | Properties | Ref |
|---|---|---|---|---|---|---|---|
| Stainless steel | MoS2 | 2.66 × 10−6 | RT/200/300/450 | 5 mJ | ≈400 nm | Granular structure stoichiometric, crystalline MoS2 | [110] |
| Stainless steel | MoS2 | 10−6 | RT/300 | 100 mJ | ≈70 nm | Stoichiometric single crystal MoS2 | [111] |
| c-Al2O3 (0001) and Si/SiO2 | 2H-MoS2 | 9.33 × 10−4 | 600 | 500 mJ/cm2 | ≈1.4 nm | Stoichiometric 2H phase Flake size ≈ 10 µm | [112] |
| GaN/c-Al2O3 (0001) | 2H-MoS2 | 8 × 10−4 | 700 | 50 mJ | Few layers | Mixed phase Roughness ≈0.11 nm | [102] |
| Titanium foil | p-MoS2 | 1.33 × 10−2 | RT | – | 0.65 nm | 1T phase MoS2 | [113] |
| SiO2 on Si [100] | MoS2 | 1.33 × 10−2 | 800 | 200 mJ/cm2 | ≈20–60 nm | 2H phase MoS2 | [104] |
| Gold-coated carbon cloth | Amorphous MoS2 | 1.33 × 10−2 | RT | 220 mJ/cm2 | ≈200 nm | 2H phase MoS2 | [97] |
| Quartz | MoS2 | 9 × 10−5 | 300 | 8500 mJ/cm2 | 30 layers | Mixed phase | [114] |
| Al2O3 (0001) | MoS2+S Powder | 1.33 × 10−2 | 700 | 50 mJ | 1–15 Layers of MoS2 | p-MoS2 2H phase MoS2 Roughness of 0.27 nm | [101] |
| Si | MoS2 | 4 × 10−4 | RT | 5/10/100/400 mJ/cm2 | ≈100–200 nm | Various compositions of MoSx (x ≤ 2.2) | [115] |
| SiO2 | MoS2 | 3 × 10−5 | 700 | 200 mJ | 1–5 layers | 2H phase MoS2 | [116] |
| W (100)-tip | MoS2+poly(vinl) | 5 × | 700 | 2000 mJ/cm2 | ≈20–60 nm | nearly stoichiometric 2H phase MoS2 | [95] |
| n-Si and p-Si | MoS2+poly(vinl) | 5 × | 700 | 500 mJ/cm2 | ≈20–60 nm | nearly stoichiometric 2H phase MoS2 | [95] |
| Al, Ag, Ni, Cu | MoS2 | 2.6 × 10−5 | 500 | 50 mJ | ≈5 nm | Epitaxial growth of 2H phase MoS2 | [98] |
| Sapphire Quartz SiO2 HfO2 | MoS2 +S powder | 1.33 × 10−2 | 700 | 30 mJ | 1 monolayer—2.8 nm | large-area growth of stoichiometric layered 2H phase MoS2 | [117] |
| SiO2/Si | MoS2 | 10−5 | 700 | 200 mJ | few-layer | 2H phase MoS2 | [118] |
| SiO2/Si | MoS2 powder | 5 × 10−4 | 600 | 2200 mJ/cm2 | 13 nm | Epitaxial growth of 2H phase MoS2 | [119] |
| Si | MoS2 | 10−4 | RT | 100 mJ | 129–1900 nm | Stoichiometric films | [120] |
| c-plane sapphire | MoS2 | 10−3 | 800 | 2000–3000 mJ/cm2 | 1–5 layers | Epitaxial growth of 2H phase MoS2 | [121] |
| Quartz glass | Polycrystalline MoS2 powder | 5 × 10−4 | 300 | 8500 mJ/cm2 | 9–10 monolayers | nearly stoichiometric 2H phase MoS2 | [122] |
| Quartz | MoS2 | 8.9 × 10−5 | 600 mJ | ≈5.8 nm | 2H phase MoS2 | [123] | |
| SiO2/Si | MoS2@Ag | 1.33 × 10−7 | 500 | 1000–2000 mJ/cm2 | ≈1.3–12.8 nm | 2H phase MoS2 | [124] |
| fluorophlogopite mica | MoS2 | 10−5 | 700 | 4000 mJ/cm2 | ≈3.3 nm | 2H phase MoS2 | [125] |
| Al2O3 (0001) | MoS2 | 10−3 | 650 | 100 mJ | ≈400 nm | 2H phase MoS2 | [126] |
| Techniques | Advantages | Limitations |
|---|---|---|
| Mechanical exfoliation |
|
|
| Chemical exfoliation |
|
|
| Chemical vapor deposition |
|
|
| Atomic layer deposition |
|
|
| Pulsed laser deposition |
|
|
| Sputtering |
|
|
| Parameters | p-Si [SCAPS] | n-MoS2 |
|---|---|---|
| Thickness (nm) | 200 | 0.32 |
| Bandgap (eV) | 1.12 | 1.9 [153] |
| Electron affinity (eV) | 4.5 | 4.2 [153] |
| Dielectric permittivity (relative) | 11.9 | 10.5 [157] |
| CB effective density of states (1/cm3) | 2.8 × 1019 | 2.2 × 1018 [158] |
| VB effective density of states (1/cm3) | 1.04 × 1019 | 1.8 × 1019 [158] |
| Electron thermal velocity (cm/s) | 1 × 107 | 1 × 107 [159] |
| Hole thermal velocity (cm/s) | 1 × 107 | 1 × 107 [159] |
| Electron mobility (cm2/Vs) | 1500 | 150 [20] |
| Hole mobility (cm2/Vs) | 4500 | 86 [159] |
| Shallow uniform donor density (1/cm3) | 0 | 1 × 1017 [159] |
| Shallow uniform acceptor density NA (1/cm3) | 1 × 1016 | 0 |
| PV Parameters | Reported Values and References | |||
|---|---|---|---|---|
| Bandgap | 1.29 eV [158,160,161] | 1.2–1.8 eV [159] | 1.23 eV [162] | 1.8 eV [163] |
| Electron affinity | 4.2 eV [158,160,161,162,163] | 4–4.7 eV [159] | 4.22 eV [163] | – |
| Relative dielectric permittivity | 3 [164] | 4 [160,161,162] | 7 [159] | 13.6 [158] |
| Effective density of states in conduction band | 1016 cm−3 [163] | 7.5 × 1017 cm−3 [160,162] | 2.2 × 1018 cm−3 [158,161] | 1019, 2.5 × 1020 cm−3 [159,164] |
| Effective density of states in valance band | 1017 cm−3 [163] | 1.8 × 1018 cm [160,162] | ~1019 cm−3 [158,161,164] | 2.5 × 1020 cm−3 [159] |
| Electron thermal velocity | 105 cm/s [162] | 2.12 × 107 cm/s | – | – |
| Hole thermal velocity | 107 cm/s [162] | 1.18 × 107 cm/s [161] | – | – |
| Electron mobility | 44 cm2/Vs [159] | 50 cm2/Vs [161] | 100 cm2/Vs [158,160,162] | – |
| Hole mobility | 30 cm2/Vs [161] | 86 cm2/Vs [159] | 150 cm2/Vs [158,160,162] | |
| Shallow uniform donor density | 1016 [161] | 1017 [164] | 1018 [162] | – |
| Shallow uniform acceptor density | 10 cm−3 [161] | 1017 cm−3 (MoS2 type P) [158] | 1021 cm−3 (MoS2 type P) [160] | – |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mouloua, D.; Kotbi, A.; Deokar, G.; Kaja, K.; El Marssi, M.; EL Khakani, M.A.; Jouiad, M. Recent Progress in the Synthesis of MoS2 Thin Films for Sensing, Photovoltaic and Plasmonic Applications: A Review. Materials 2021, 14, 3283. https://doi.org/10.3390/ma14123283
Mouloua D, Kotbi A, Deokar G, Kaja K, El Marssi M, EL Khakani MA, Jouiad M. Recent Progress in the Synthesis of MoS2 Thin Films for Sensing, Photovoltaic and Plasmonic Applications: A Review. Materials. 2021; 14(12):3283. https://doi.org/10.3390/ma14123283
Chicago/Turabian StyleMouloua, Driss, Ahmed Kotbi, Geetanjali Deokar, Khaled Kaja, Mimoun El Marssi, My Ali EL Khakani, and Mustapha Jouiad. 2021. "Recent Progress in the Synthesis of MoS2 Thin Films for Sensing, Photovoltaic and Plasmonic Applications: A Review" Materials 14, no. 12: 3283. https://doi.org/10.3390/ma14123283
APA StyleMouloua, D., Kotbi, A., Deokar, G., Kaja, K., El Marssi, M., EL Khakani, M. A., & Jouiad, M. (2021). Recent Progress in the Synthesis of MoS2 Thin Films for Sensing, Photovoltaic and Plasmonic Applications: A Review. Materials, 14(12), 3283. https://doi.org/10.3390/ma14123283









