Cylindrical Polyurethane Scaffold Fabricated Using the Phase Inversion Method: Influence of Process Parameters on Scaffolds’ Morphology and Mechanical Properties
Abstract
1. Introduction
2. Results
2.1. Selection of Process Parameters
2.1.1. Composition of Nonsolvent Solution
2.1.2. Concentration of Polymer Solution
2.1.3. Time of the Process
2.1.4. Selection of Porogen
2.2. Influence of Porogen Addition on Physical and Mechanical Properties
3. Discussion
4. Materials and Methods
4.1. Materials
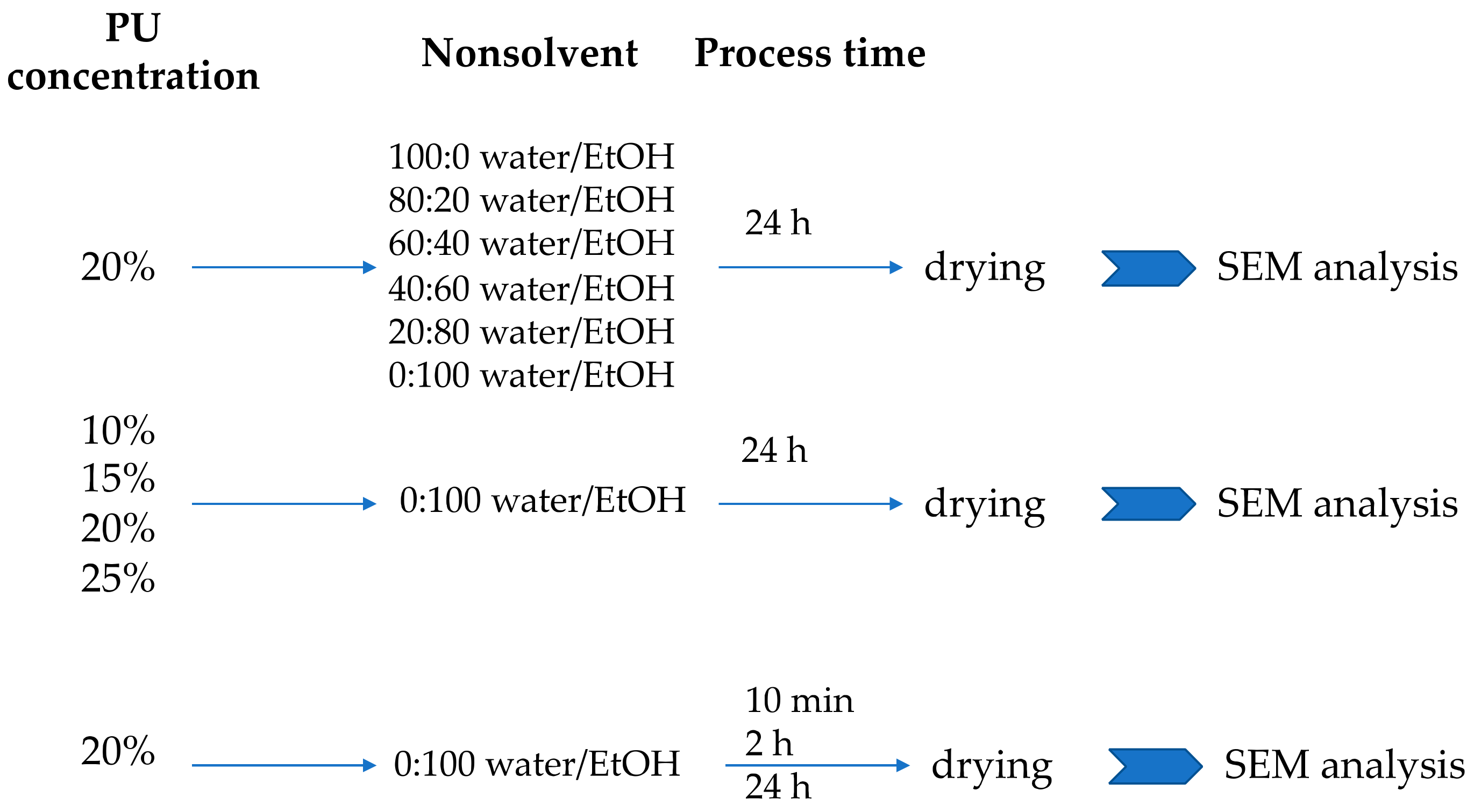
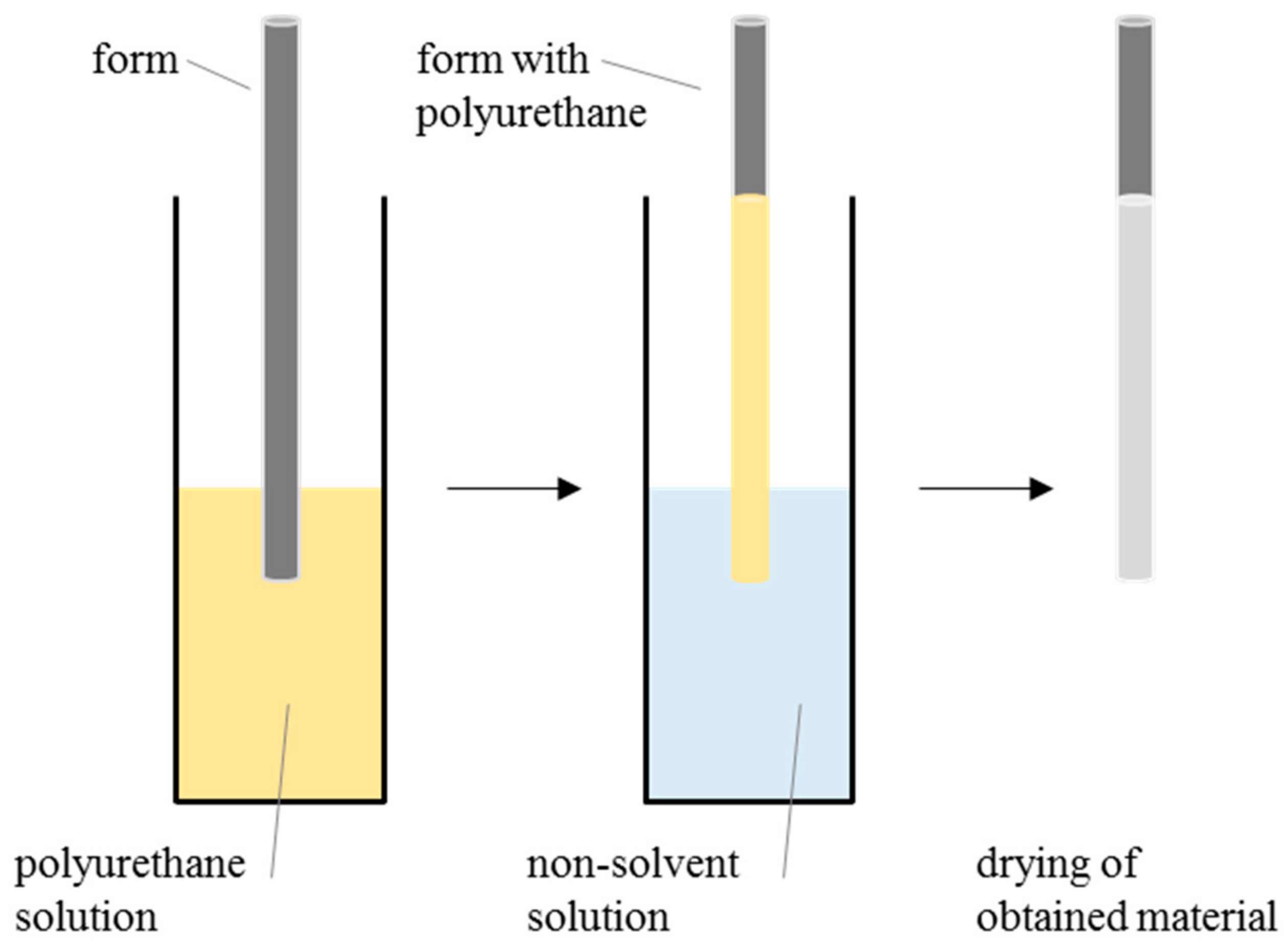
4.2. Preparation of Polyurethane Scaffolds—Selection of Process Parameters
4.3. Porogen Addition
4.4. Surface Characterization
4.5. Mechanical Testing
4.6. Porosity
4.7. Cytotoxicity Evaluation
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Adhikari, K.R.; Tucker, B.S.; Thomas, V. Tissue engineering of small-diameter vascular grafts. In Biointegration of Medical Implant Materials; Elsevier BV: Amsterdam, The Netherlands, 2020; Volume 1957, pp. 79–100. [Google Scholar] [CrossRef]
- Chlupac, J.; Filová, E.; Bačáková, L. Blood vessel replacement: 50 Years of development and tissue engineering paradigms in vascular surgery. Physiol. Res. 2009, 58, S119–S140. [Google Scholar] [CrossRef] [PubMed]
- Zdrahala, R.J. Small Caliber Vascular Grafts Part II: Polyurethanes Revisited. J. Biomater. Appl. 1996, 11, 37–61. Available online: http://hjb.sagepub.com.proxy.lib.umich.edu/content/9/2/183.full.pdf+html (accessed on 1 April 2021). [PubMed]
- Zilla, P.; Bezuidenhout, D.; Human, P. Prosthetic Vascular Grafts: Wrong Models, Wrong Questions and No Healing. Biomaterials 2007, 28, 5009–5027. Available online: http://www.ncbi.nlm.nih.gov/pubmed/17688939 (accessed on 1 April 2021). [CrossRef] [PubMed]
- Bergmeister, H.; Schreiber, C.; Grasl, C.; Walter, I.; Plasenzotti, R.; Stoiber, M.; Bernhard, D.; Schima, H. Healing characteristics of electrospun polyurethane grafts with various porosities. Acta Biomater. 2013, 9, 6032–6040. [Google Scholar] [CrossRef]
- Zubilewicz, T.; Becquemin, J.; Wronski, J.; Desgranges, P.; Kobeiter, H.; Feldo, M.; Przywara, S. Secondary interventions after endovascular repair of AAA. J. Mal. Vasc. 2004, 29, 30. [Google Scholar] [CrossRef]
- Boffito, M.; Sartori, S.; Ciardelli, G. Polymeric scaffolds for cardiac tissue engineering: Requirements and fabrication technologies. Polym. Int. 2013, 63, 2–11. [Google Scholar] [CrossRef]
- Naureen, B.; Haseeb, A.; Basirun, W.; Muhamad, F. Recent advances in tissue engineering scaffolds based on polyurethane and modified polyurethane. Mater. Sci. Eng. C 2021, 118, 111228. [Google Scholar] [CrossRef]
- Li, J.; Gao, Q.; Chen, Z.; Yang, X. Small diameter blood vessels with controllable micropore structure induced by centrifugal force for improved endothelialization. Eng. Life Sci. 2020, 20, 181–185. [Google Scholar] [CrossRef]
- Khorasani, M.; Shorgashti, S. Fabrication of microporous polyurethane by spray phase inversion method as small diameter vascular grafts material. J. Biomed. Mater. Res. Part A 2006, 77, 253–260. [Google Scholar] [CrossRef]
- Catto, V.; Farè, S.; Freddi, G.; Tanzi, M.C. Vascular Tissue Engineering: Recent Advances in Small Diameter Blood Vessel Regeneration. ISRN Vasc. Med. 2014, 2014, 1–27. [Google Scholar] [CrossRef]
- Baiguera, S.; Ribatti, D. Endothelialization approaches for viable engineered tissues. Angiogenesis 2013, 16, 1–14. [Google Scholar] [CrossRef]
- Adipurnama, I.; Yang, M.-C.; Ciach, T.; Butruk-Raszeja, B. Surface modification and endothelialization of polyurethane for vascular tissue engineering applications: A review. Biomater. Sci. 2016, 5, 22–37. [Google Scholar] [CrossRef]
- Rajendran, S. Implantable Materials: An Overview. In Medical and Healthcare Textiles; Woodhead Publishing Limited: Sawston, UK, 2010; pp. 329–333. [Google Scholar] [CrossRef]
- Leake, D.; Cenni, E.; Cavedagna, D.; Stea, S.; Ciapetti, G.; Pizzoferrato, A. Comparative study of the thromboresistance of Dacron® combined with various polyurethanes. Biomaterials 1989, 10, 441–444. [Google Scholar] [CrossRef]
- He, W.; Benson, R. Polymeric Biomaterials. In Applied Plastics Engineering Handbook: Processing, Materials, and Applications, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 145–164. [Google Scholar] [CrossRef]
- Cai, Q.; Liao, W.; Xue, F.; Wang, X.; Zhou, W.; Li, Y.; Zeng, W. Selection of different endothelialization modes and different seed cells for tissue-engineered vascular graft. Bioact. Mater. 2021, 6, 2557–2568. [Google Scholar] [CrossRef]
- Zhuang, Y.; Zhang, C.; Cheng, M.; Huang, J.; Liu, Q.; Yuan, G.; Lin, K.; Yu, H. Challenges and strategies for in situ endothelialization and long-term lumen patency of vascular grafts. Bioact. Mater. 2021, 6, 1791–1809. [Google Scholar] [CrossRef]
- Boffito, M.; Sartori, S.; Mattu, C.; Ciardelli, G. Polyurethanes for cardiac applications. In Advances in Polyurethane Biomaterials; Elsevier BV: Amsterdam, The Netherlands, 2016; pp. 387–416. [Google Scholar] [CrossRef]
- Ward, R.S.; Jones, R.L. Polyurethanes and Silicone Polyurethane Copolymers. In Comprehensive Biomaterials II; Elsevier: Amsterdam, The Netherlands, 2017; Volume 1, pp. 570–619. [Google Scholar]
- Fink, J.K. Poly(urethane)s. In Reactive Polymers Fundamentals and Applications, 2nd ed.; William Andrew: New York, NY, USA, 2013; Chapter 2; pp. 49–93. [Google Scholar]
- Burke, A.; Hasirci, N. Polyurethanes in Biomedical Applications. In Biomaterials: From Molecules to Engineered Tissues; Polyurethanes in Biomedical Applications; Plenum Publishers: New York, NY, USA, 2004; pp. 93–101. [Google Scholar]
- Wang, W.; Wang, C. Polyurethane for biomedical applications: A review of recent developments. In The Design and Manufacture of Medical Devices; Elsevier Masson SAS: Amsterdam, The Netherlands, 2012; pp. 115–151. [Google Scholar] [CrossRef]
- Chen, Q.; Liang, S.; Thouas, G.A. Elastomeric Biomaterials for Tissue Engineering. Prog. Polym. Sci. 2013, 38, 584–671. Available online: http://linkinghub.elsevier.com/retrieve/pii/S007967001200069X (accessed on 1 April 2021). [CrossRef]
- Mercado-Pagán, Á.E.; Kang, Y.; Findlay, M.W.; Yang, Y. Development and evaluation of elastomeric hollow fiber membranes as small diameter vascular graft substitutes. Mater. Sci. Eng. C 2015, 49, 541–548. [Google Scholar] [CrossRef]
- Vannozzi, L.; Ricotti, L.; Santaniello, T.; Terencio, T.; Oropesa-Nunez, R.; Canale, C.; Borghi, F.; Menciassi, A.; Lenardi, C.; Gerges, I. 3D porous polyurethanes featured by different mechanical properties: Characterization and interaction with skeletal muscle cells. J. Mech. Behav. Biomed. Mater. 2017, 75, 147–159. [Google Scholar] [CrossRef]
- Di Luccio, M.; Nobrega, R.; Borges, C.P. Microporous anisotropic phase inversion membranes from bisphenol-A polycarbonate: Study of a ternary system. Polymer (Guildf) 2000, 41, 4309–4315. [Google Scholar] [CrossRef]
- Qian, L.; Zhang, H. Porogen Incorporation and Phase Inversion. In Porous Polymers; Wiley: Hoboken, NJ, USA, 2011; pp. 79–117. [Google Scholar]
- Hołda, A.K.; Vankelecom, I.F. Understanding and guiding the phase inversion process for synthesis of solvent resistant nanofiltration membranes. J. Appl. Polym. Sci. 2015, 132, 1–17. [Google Scholar] [CrossRef]
- Wang, H.; Wang, L.; Liu, C.; Xu, Y.; Zhuang, Y.; Zhou, Y.; Gu, S.; Xu, W.; Yang, H. Effect of temperature on the morphology of poly (lactic acid) porous membrane prepared via phase inversion induced by water droplets. Int. J. Biol. Macromol. 2019, 133, 902–910. [Google Scholar] [CrossRef]
- Li, S.; Sengupta, D.; Chien, S. Vascular tissue engineering: From In Vitro to in situ. Wiley Interdiscip. Rev. Syst. Biol. Med. 2014, 6, 61–76. [Google Scholar] [CrossRef]
- Wu, J.; Hu, C.; Tang, Z.; Yu, Q.; Liu, X.; Chen, H. Tissue-engineered Vascular Grafts: Balance of the Four Major Requirements. Colloid Interface Sci. Commun. 2018, 23, 34–44. [Google Scholar] [CrossRef]
- Xie, F.; Zhang, T.; Bryant, P.; Kurusingal, V.; Colwell, J.M.; Laycock, B. Degradation and stabilization of polyurethane elastomers. Prog. Polym. Sci. 2019, 90, 211–268. [Google Scholar] [CrossRef]
- Tiwari, A.; Salacinski, H.; Seifalian, A.M.; Hamilton, G. New prostheses for use in bypass grafts with special emphasis on polyurethanes. Cardiovasc. Surg. 2002, 10, 191–197. [Google Scholar] [CrossRef]
- Khan, I.; Smith, N.; Jones, E.; Finch, D.S.; Cameron, R.E. Analysis and evaluation of a biomedical polycarbonate urethane tested in an In Vitro study and an ovine arthroplasty model. Part I: Materials selection and evaluation. Biomaterials 2005, 26, 621–631. [Google Scholar]
- Kuźmińska, A.; Butruk-Raszeja, B.A.; Stefanowska, A.; Ciach, T. Polyvinylpyrrolidone (PVP) hydrogel coating for cylindrical polyurethane scaffolds. Colloids Surf. B Biointerfaces 2020, 192, 111066. [Google Scholar] [CrossRef] [PubMed]
- Kidane, A.G.; Burriesci, G.; Edirisinghe, M.; Ghanbari, H.; Bonhoeffer, P.; Seifalian, A.M. A novel nanocomposite polymer for development of synthetic heart valve leaflets. Acta Biomater. 2009, 5, 2409–2417. [Google Scholar] [CrossRef] [PubMed]
- Nezarati, R.M.; Eifert, M.B.; Dempsey, D.K.; Cosgriff-Hernandez, E. Electrospun vascular grafts with improved compliance matching to native vessels. J. Biomed. Mater. Res. Part B Appl. Biomater. 2015, 103, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Feng, Y.; Fang, Z.; Yuan, W.; Khan, M. Co-electrospun blends of PU and PEG as potential biocompatible scaffolds for small-diameter vascular tissue engineering. Mater. Sci. Eng. C 2012, 32, 2306–2315. [Google Scholar] [CrossRef]
- Shi, C.; Yuan, W.; Khan, M.; Li, Q.; Feng, Y.; Yao, F.; Zhang, W. Hydrophilic PCU scaffolds prepared by grafting PEGMA and immobilizing gelatin to enhance cell adhesion and proliferation. Mater. Sci. Eng. C 2015, 50, 201–209. [Google Scholar] [CrossRef]
- ISO. Biological Evaluation of Medical Devices; BS EN ISO 10993-122004; European Committee for Standardization: Brussels, Belgium, 2004; pp. 1–17. [Google Scholar]
- François, S.; Chakfé, N.; Durand, B.; Laroche, G. A poly(l-lactic acid) nanofibre mesh scaffold for endothelial cells on vascular prostheses. Acta Biomater. 2009, 5, 2418–2428. [Google Scholar] [CrossRef]
- Łos, M.J.; Panigrahi, S.; Sielatycka, K.; Grillon, C. Successful biomaterial-based artificial organ-updates on artificial blood vessels. In Stem Cells and Biomaterials for Regenerative Medicine; Elsevier Inc.: Amsterdam, The Netherlands, 2018; pp. 203–222. [Google Scholar] [CrossRef]
- Kucinska-Lipka, J.; Gubanska, I.; Janik, H.; Sienkiewicz, M. Fabrication of polyurethane and polyurethane based composite fibres by the electrospinning technique for soft tissue engineering of cardiovascular system. Mater. Sci. Eng. C 2015, 46, 166–176. [Google Scholar] [CrossRef] [PubMed]
- Haghjooy Javanmard, S.; Anari, J.; Zargar Kharazi, A.; Vatankhah, E. In Vitro hemocompatibility and cytocompatibility of a three-layered vascular scaffold fabricated by sequential electrospinning of PCL, collagen, and PLLA nanofibers. J. Biomater. Appl. 2016, 31, 438–449. [Google Scholar] [CrossRef] [PubMed]
- Tan, Z.; Gao, X.; Liu, T.; Yang, Y.; Zhong, J.; Tong, C.; Tan, Y. Electrospun vein grafts with high cell infiltration for vascular tissue engineering. Mater. Sci. Eng. C 2017, 81, 407–415. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, X.S.; Guex, A.G.; Liu, S.S.; Müller, E.; Malini, R.I.; Zhao, H.J.; Rottmar, M.; Maniura-Weber, K.; Rossi, R.M.; et al. Corrigendum: A compliant and biomimetic three-layered vascular graft for small blood vessels (2017 Biofabrication 9 025010). Biofabrication 2017, 9, 029501. [Google Scholar] [CrossRef]
- Akentjew, T.L.; Terraza, C.; Suazo, C.; Maksimcuka, J.; Wilkens, C.A.; Vargas, F.; Zavala, G.; Ocaña, M.; Enrione, J.; García-Herrera, C.M.; et al. Rapid fabrication of reinforced and cell-laden vascular grafts structurally inspired by human coronary arteries. Nat. Commun. 2019, 10, 1–15. [Google Scholar]
- Tomecka, E.; Wojasinski, M.; Jastrzebska, E.; Chudy, M.; Ciach, T.; Brzozka, Z. Poly(l-lactic acid) and polyurethane nanofibers fabricated by solution blow spinning as potential substrates for cardiac cell culture. Mater. Sci. Eng. C 2017, 75, 305–316. [Google Scholar] [CrossRef] [PubMed]
- Nune, S.K.; Rama, K.S.; Dirisala, V.R.; Chavali, M.Y. Electrospinning of collagen nanofiber scaffolds for tissue repair and regeneration. In Nanostructures for Novel Therapy; Elsevier: Amsterdam, The Netherlands, 2017; pp. 281–311. [Google Scholar]
- Butruk-Raszeja, B.A.; Kuźmińska, A.; Wojasiński, M.; Piotrowska, Z. Physicochemical and Mechanical Properties of Blow Spun Nanofibrous Prostheses Modified with Acrylic Acid and REDV Peptide. Coatings 2020, 10, 1110. [Google Scholar] [CrossRef]
- Łopianiak, I.; Butruk-Raszeja, B.A. Evaluation of Sterilization/Disinfection Methods of Fibrous Polyurethane Scaffolds Designed for Tissue Engineering Applications. Int. J. Mol. Sci. 2020, 21, 8092. [Google Scholar] [CrossRef]
- Sarkar, S.; Burriesci, G.; Wojcik, A.; Aresti, N.; Hamilton, G.; Seifalian, A.M. Manufacture of small calibre quadruple lamina vascular bypass grafts using a novel automated extrusion-phase-inversion method and nanocomposite polymer. J. Biomech. 2009, 42, 722–730. [Google Scholar] [CrossRef]
- Janik, H.; Marzec, M. A review: Fabrication of porous polyurethane scaffolds. Mater. Sci. Eng. C 2015, 48, 586–591. [Google Scholar] [CrossRef]
- Martínez-Pérez, C.A.; Olivas-Armendariz, I.; Castro-Carmona, J.S.; García-Casillas, P.E. Scaffolds for Tissue Engineering Via Thermally Induced Phase Separation. Adv. Regen. Med. InTech Rijeka 2011, 21, 275–294. [Google Scholar]
- Strathmann, H.; Kock, K. The formation mechanism of phase inversion membranes. Desalination 1977, 21, 241–255. [Google Scholar] [CrossRef]
- Tan, A.; Gundogan, B.; Farhatnia, Y.; Nayyer, L.; Mahdibeiraghdar, S.; Rajadas, J.; De Coppi, P.; Davies, A.H.; Seifalian, A.M. Tissue engineering vascular grafts a fortiori: Looking back and going forward. Expert Opin. Biol. Ther. 2015, 15, 231–244. [Google Scholar]
- Wesolowski, S.; Fries, C.C.; Karlson, K.E.; De Bakey, M.; Sawyer, P.N. Porosity: Primary determinant of ultimate fate of synthetic vascular grafts. Surgery 1961, 50, 91–96. [Google Scholar] [CrossRef]
- Ju, Y.M.; Choi, J.S.; Atala, A.; Yoo, J.J.; Lee, S.J. Bilayered scaffold for engineering cellularized blood vessels. Biomaterials 2010, 31, 4313–4321. [Google Scholar] [CrossRef]
- Cafarelli, A.; Losi, P.; Salgarella, A.R.; Barsotti, M.C.; Di Cioccio, I.B.; Foffa, I.; Vannozzi, L.; Pingue, P.; Soldani, G.; Ricotti, L. Small-caliber vascular grafts based on a piezoelectric nanocomposite elastomer: Mechanical properties and biocompatibility. J. Mech. Behav. Biomed. Mater. 2019, 97, 138–148. [Google Scholar] [CrossRef]
- Gao, B.; Feng, Y.; Lu, J.; Zhang, L.; Zhao, M.; Shi, C.; Khan, M.; Guo, J. Grafting of phosphorylcholine functional groups on polycarbonate urethane surface for resisting platelet adhesion. Mater. Sci. Eng. C 2013, 33, 2871–2878. [Google Scholar] [CrossRef]
- Yuan, W.; Feng, Y.; Wang, H.; Yang, D.; An, B.; Zhang, W.; Khan, M.; Guo, J. Hemocompatible surface of electrospun nanofibrous scaffolds by ATRP modification. Mater. Sci. Eng. C 2013, 33, 3644–3651. [Google Scholar] [CrossRef]
- Khorasani, M.T.; Shorgashti, S. Fabrication of microporous thermoplastic polyurethane for use as small-diameter vascular graft material I. Phase-inversion method. J. Biomed. Mater. Res. Part B Appl. Biomater. 2005, 76, 41–48. [Google Scholar]
- Guan, J.; Fujimoto, K.L.; Sacks, M.S.; Wagner, W.R. Preparation and characterization of highly porous, biodegradable polyurethane scaffolds for soft tissue applications. Biomaterials 2005, 26, 3961–3971. [Google Scholar] [CrossRef] [PubMed]
- Nam, Y.S.; Park, T.G. Porous biodegradable polymeric scaffolds prepared by thermally induced phase separation. J. Biomed. Mater. Res. 1999, 47, 8–17. [Google Scholar] [CrossRef]
- Pennel, T.; Zilla, P.; Bezuidenhout, D. Biomaterials in Vascular Graft Surgery. In Reference Module in Materials Science and Materials Engineering; Elsevier: Amsterdam, The Netherlands, 2016; pp. 1–8. [Google Scholar] [CrossRef]
- Ahmed, M.; Ghanbari, H.; Cousins, B.G.; Hamilton, G.; Seifalian, A.M. Small calibre polyhedral oligomeric silsesquioxane nanocomposite cardiovascular grafts: Influence of porosity on the structure, haemocompatibility and mechanical properties. Acta Biomater. 2011, 7, 3857–3867. [Google Scholar] [CrossRef]
- Meng, X.; Wang, X.; Jiang, Y.; Zhang, B.; Li, K.; Li, Q. Suture retention strength of P(LLA-CL) tissue-engineered vascular grafts. RSC Adv. 2019, 9, 21258–21264. [Google Scholar] [CrossRef]
- Lin-Gibson, S.; Cooper, J.A.; Landis, F.A.; Cicerone, M.T. Systematic Investigation of Porogen Size and Content on Scaffold Morphometric Parameters and Properties. Biomacromolecules 2007, 8, 1511–1518. [Google Scholar] [CrossRef]
- Faturechi, R.; Hashemi, A.; Abolfathi, N.; Solouk, A. Mechanical guidelines on the properties of human healthy arteries in the design and fabrication of vascular grafts: Experimental tests and quasi-linear viscoelastic model. Acta Bioeng. Biomech. 2019, 21, 13–21. [Google Scholar]
- Karimi, A.; Rahmati, S.M.; Sera, T.; Kudo, S.; Navidbakhsh, M. A combination of experimental and numerical methods to investigate the role of strain rate on the mechanical properties and collagen fiber orientations of the healthy and atherosclerotic human coronary arteries. Bioengineered 2016, 8, 154–170. [Google Scholar] [CrossRef]
- Young, T.-H.; Chen, L.-W. Pore formation mechanism of membranes from phase inversion process. Desalination 1995, 103, 233–247. [Google Scholar] [CrossRef]
- Vermette, P.; Griesser, H.J.; Laroche, G.; Guidoin, R. Biomedical Applications of Polyurethanes; Tissue Engineering Intelligence Unit 6; Eurekah.com Inc.: London, UK, 2001; pp. 347–354. [Google Scholar]
- Vermette, P.; Griesser, H.J.; Laroche, G.G.; Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Shtilman, M.I. Polymeric Biomaterials. Part I. Polymer Implants; CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]
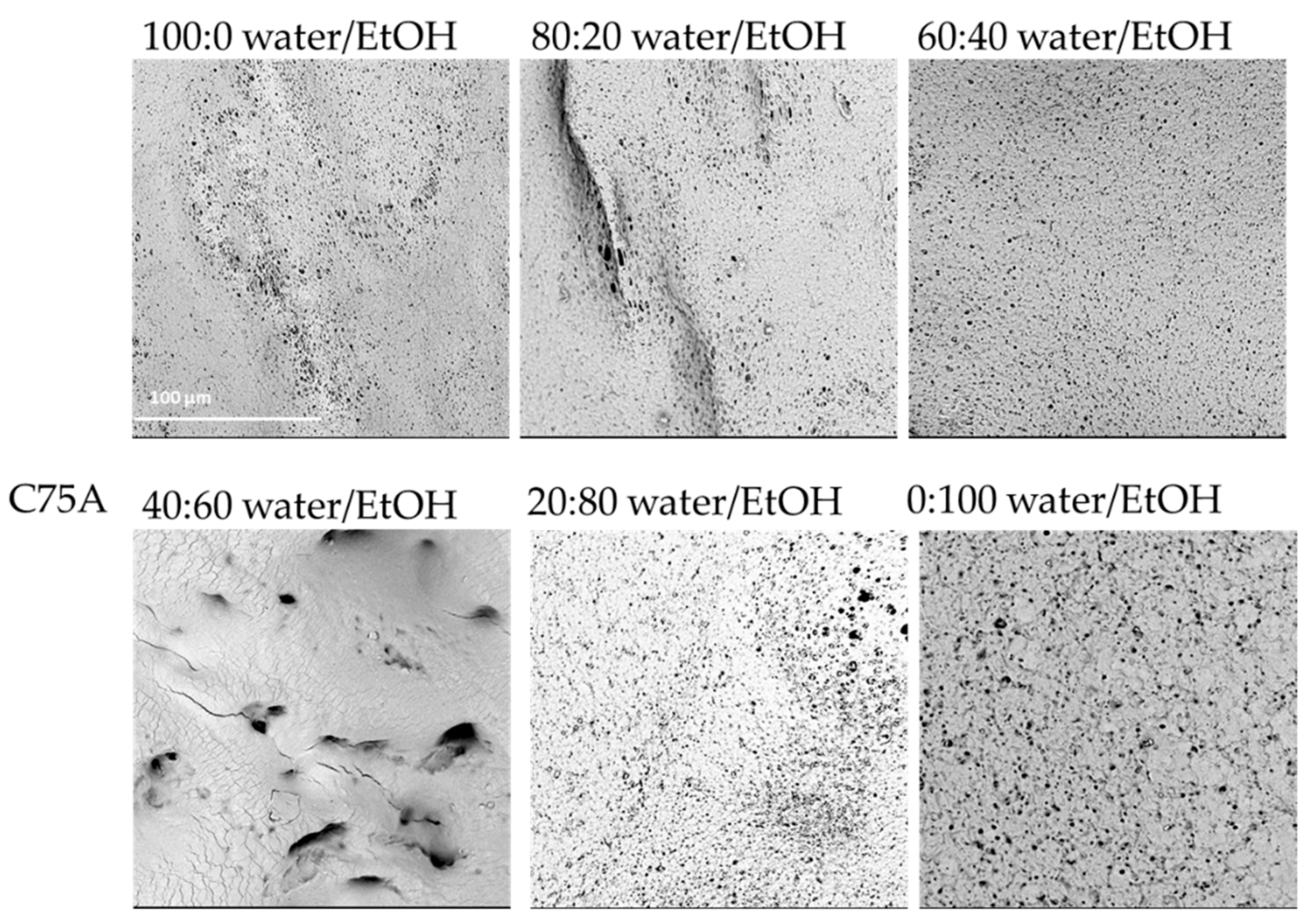
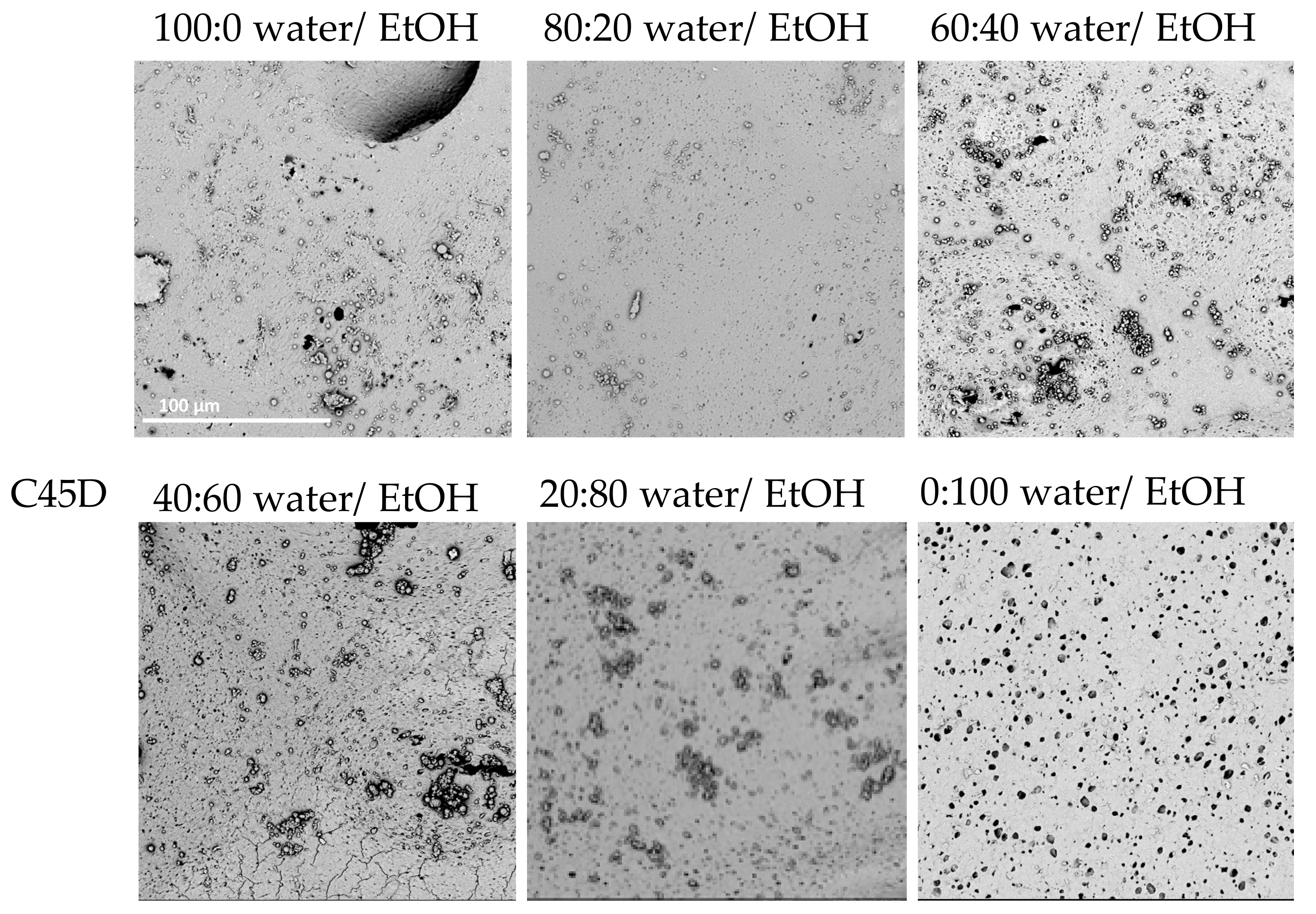
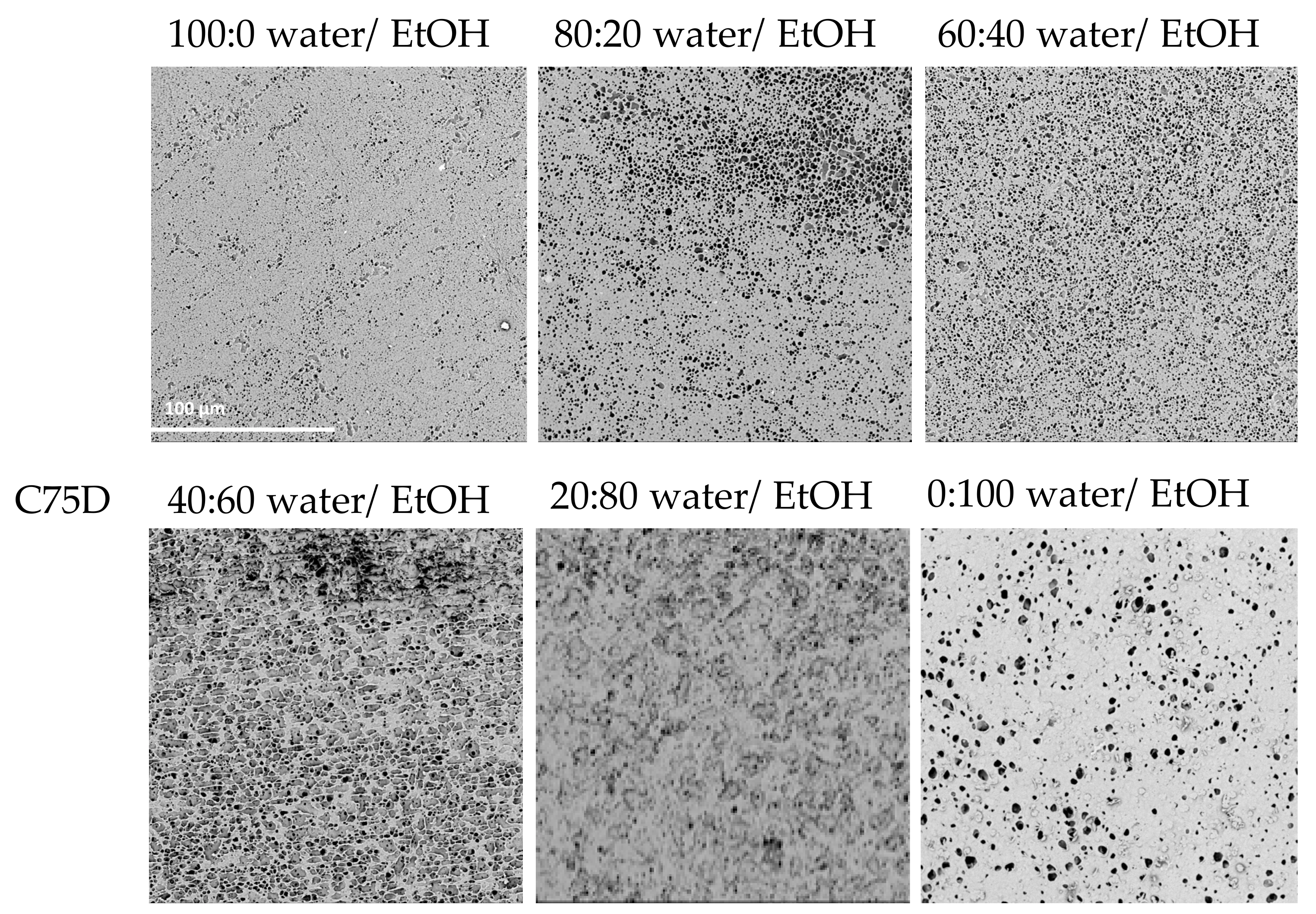


| 100:0 Water/EtOH | 80:20 Water/EtOH | 60:40 Water/EtOH | 40:60 Water/EtOH | 20:80 Water/EtOH | 0:100 Water/EtOH | ||
|---|---|---|---|---|---|---|---|
| C75A | (i) | + | + | + | ‒ | + | + |
| (ii) | ± | ± | ± | + | + | ||
| C45D | (i) | ‒ | ‒ | ± | ± | ± | + |
| (ii) | ± | ± | ± | + | + | + | |
| C75D | (i) | ± | + | + | ± | ± | + |
| (ii) | ‒ | – | ± | ± | ± | ± | |
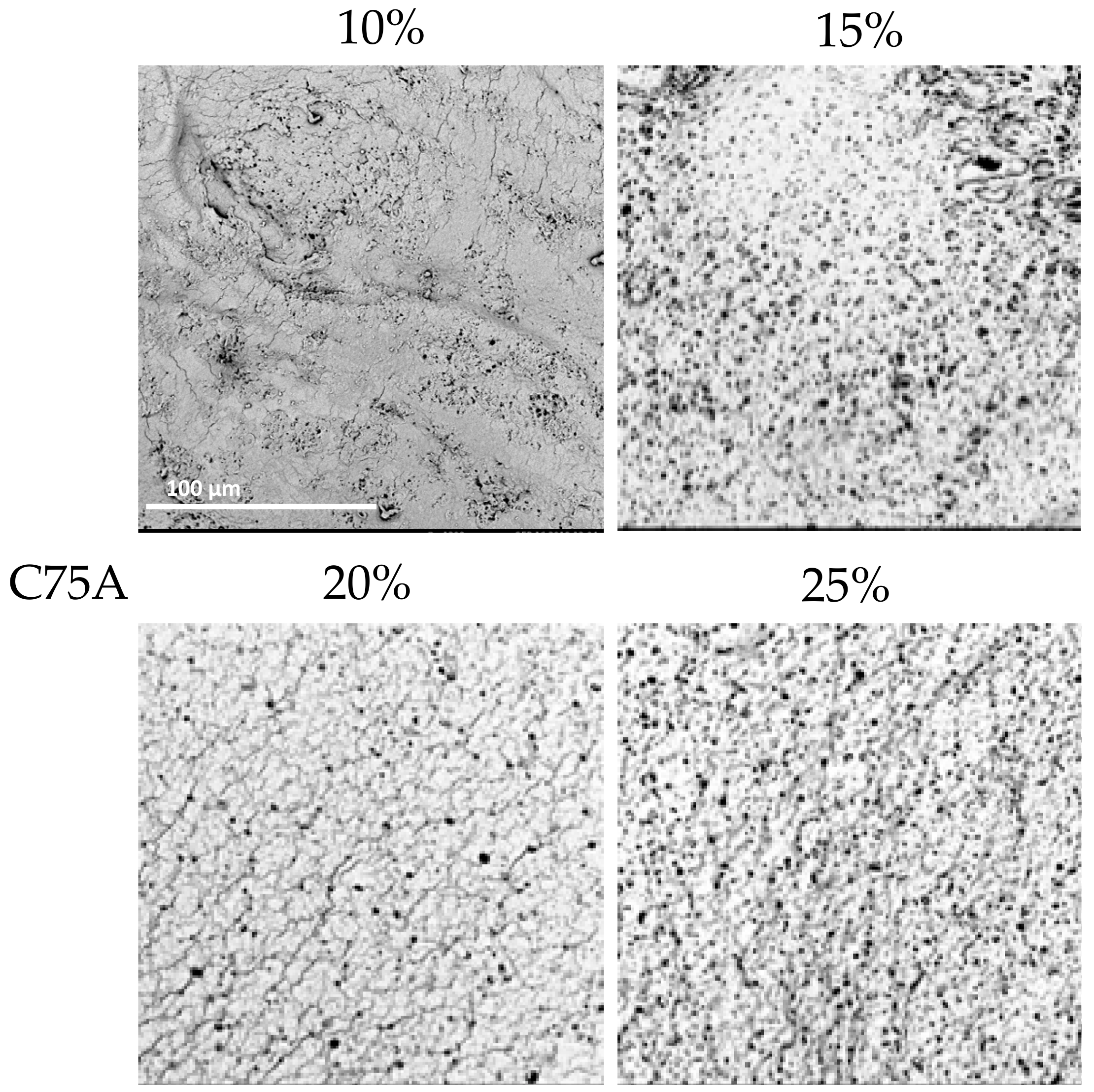
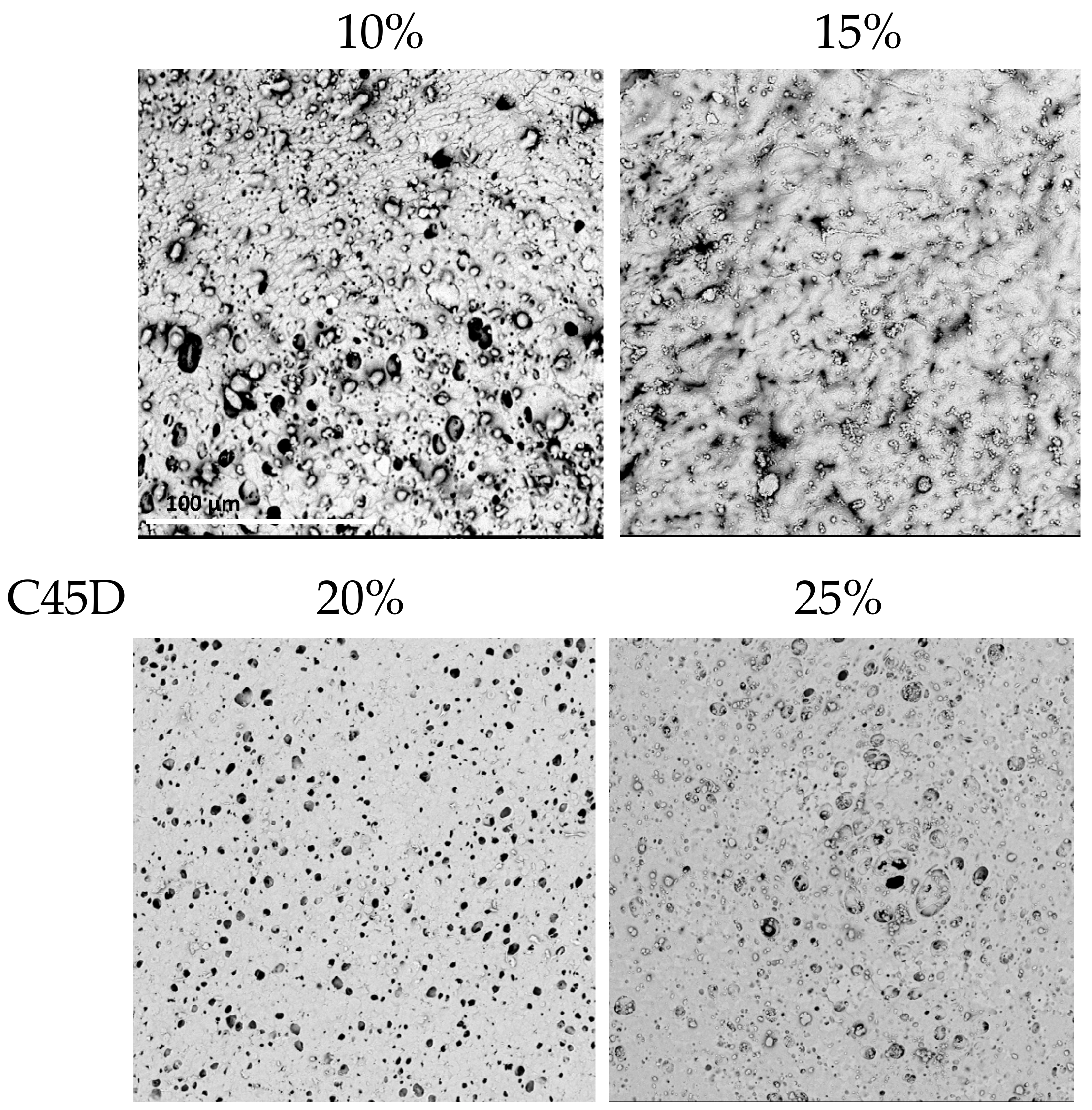
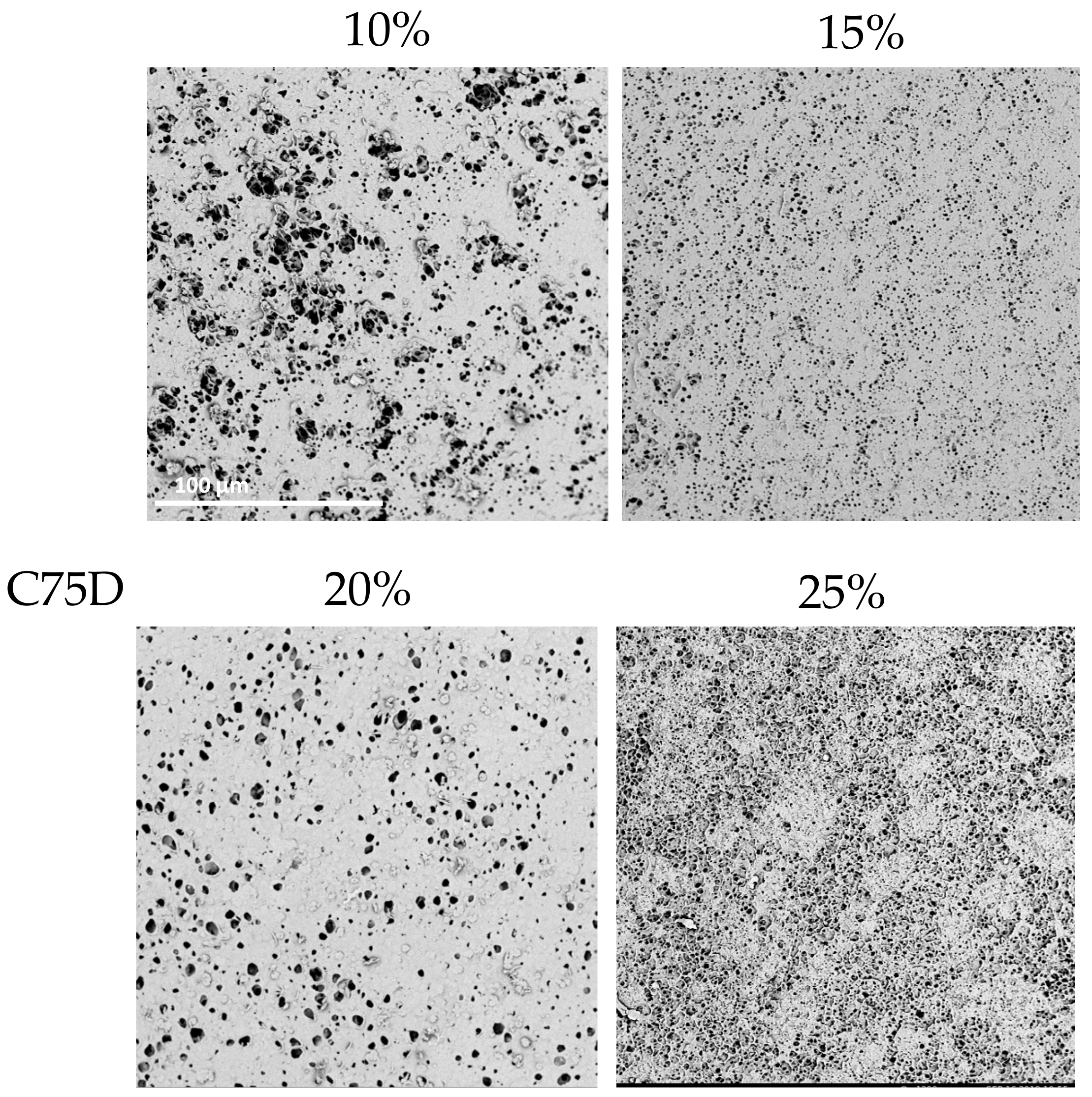
| 10% PU | 15% PU | 20% PU | 25% PU | ||
|---|---|---|---|---|---|
| C75A | (i) | ‒ | ± | + | + |
| (ii) | ‒ | ± | + | + | |
| C45D | (i) | ‒ | ‒ | + | + |
| (ii) | ‒ | + | + | ||
| C75D | (i) | ± | ± | + | ± |
| (ii) | ‒ | ± | ± | ± | |
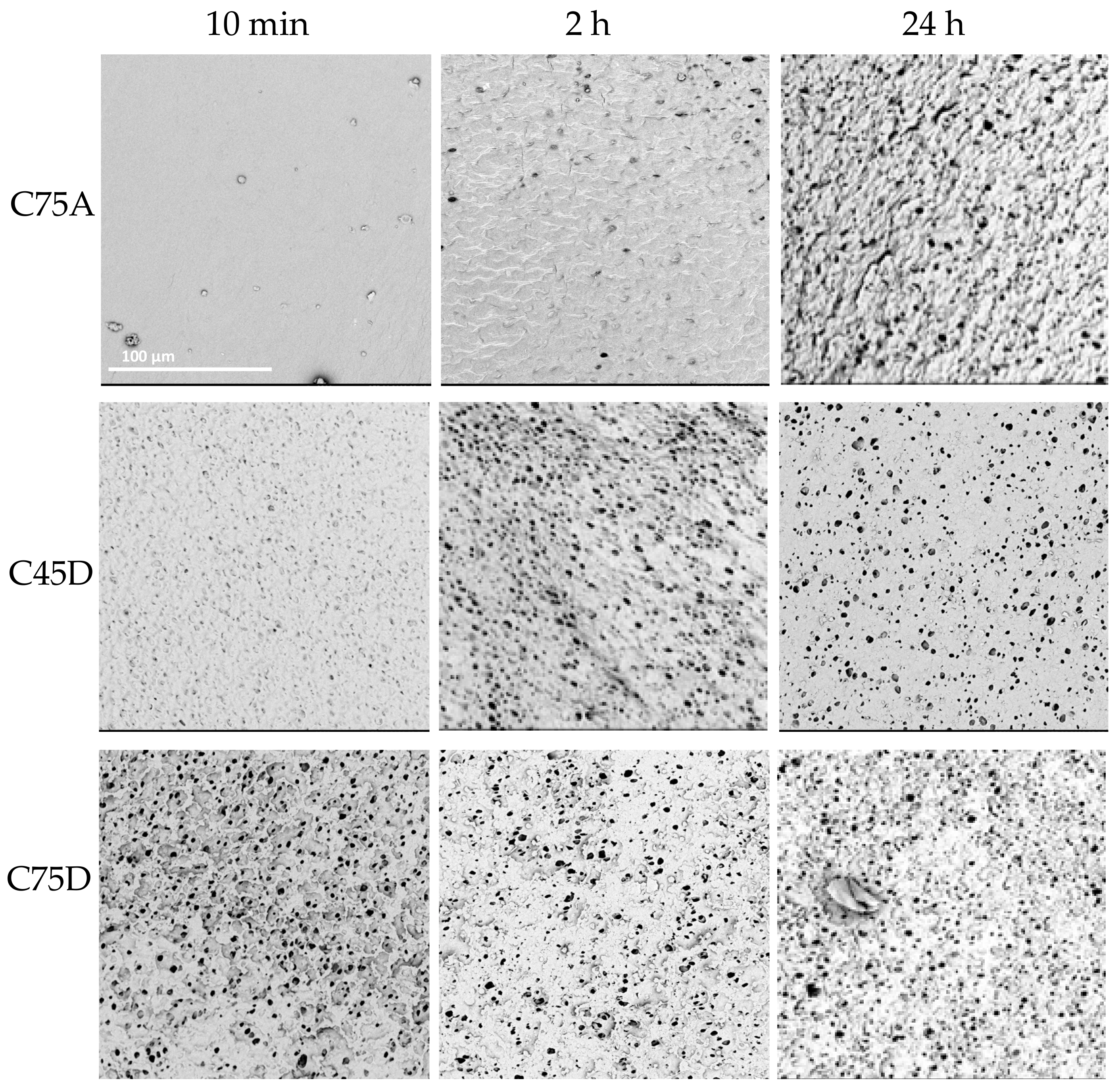
| 10 min | 2 h | 24 h | ||
|---|---|---|---|---|
| C75A | (i) | − | − | + |
| (ii) | ± | ± | + | |
| C45D | (i) | ± | + | + |
| (ii) | ± | ± | + | |
| C75D | (i) | + | + | + |
| (ii) | ± | ± | + | |
| Polymer Type | C75A | C45D | C75D |
|---|---|---|---|
| Polymer concentration (%) | 20 | ||
| Nonsolvent | 0:100 water/EtOH | ||
| Process time (h) | 24 h | ||
| Young’s modulus (MPa) | * | ||
| 2.4 ± 0.1 | 3.6 ± 1.5 | 38.5 ± 4.3 | |
| Tensile strength (MPa) | * | ||
| 9.8 ± 2.5 | 11.2 ± 1.2 | 4.1 ± 0.9 | |
| Elongation at break [mm/mm] | * | ||
| 7.8 ± 0.9 | 4.7 ± 0.4 | 0.8 ± 0.3 | |
| Cell viability (%) | 87 ± 4 | 92 ± 4 | 89 ± 4 |
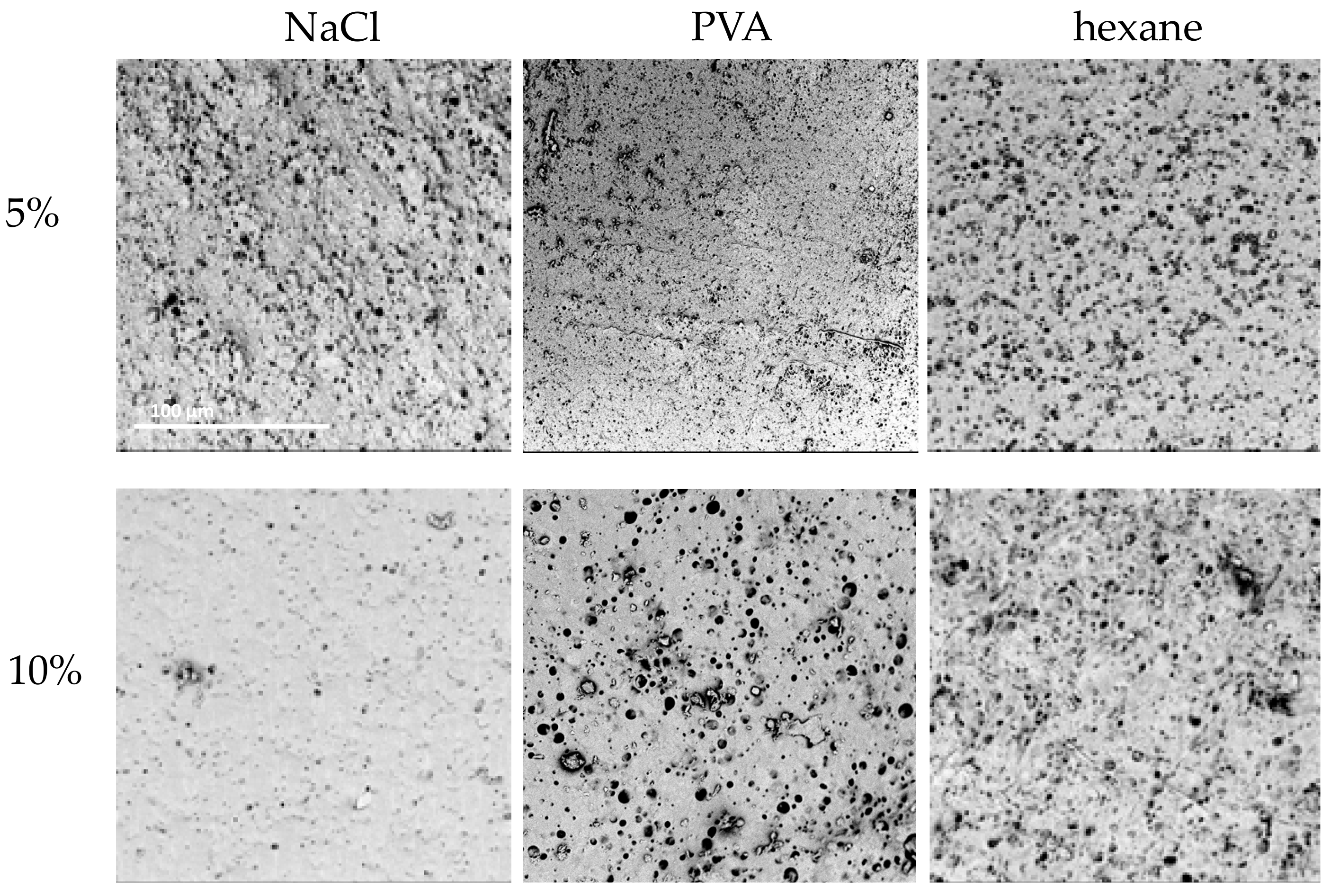
| 5% NaCl | 10% NaCl | 5% PVA | 10% PVA | 5% Hexane | 10% Hexane | |
|---|---|---|---|---|---|---|
| (i) | + | + | + | + | + | + |
| (ii) | ± | ± | ± | + | + | ± |
| PU C45D | PU C45D_10%PVA | ||
|---|---|---|---|
| Polymer type | C45D | ||
| Polymer concentration (%) | 20 | ||
| Nonsolvent | 0:100 water/EtOH | ||
| Process time (h) | 24 | ||
| Porogen | – | 10% PVA | |
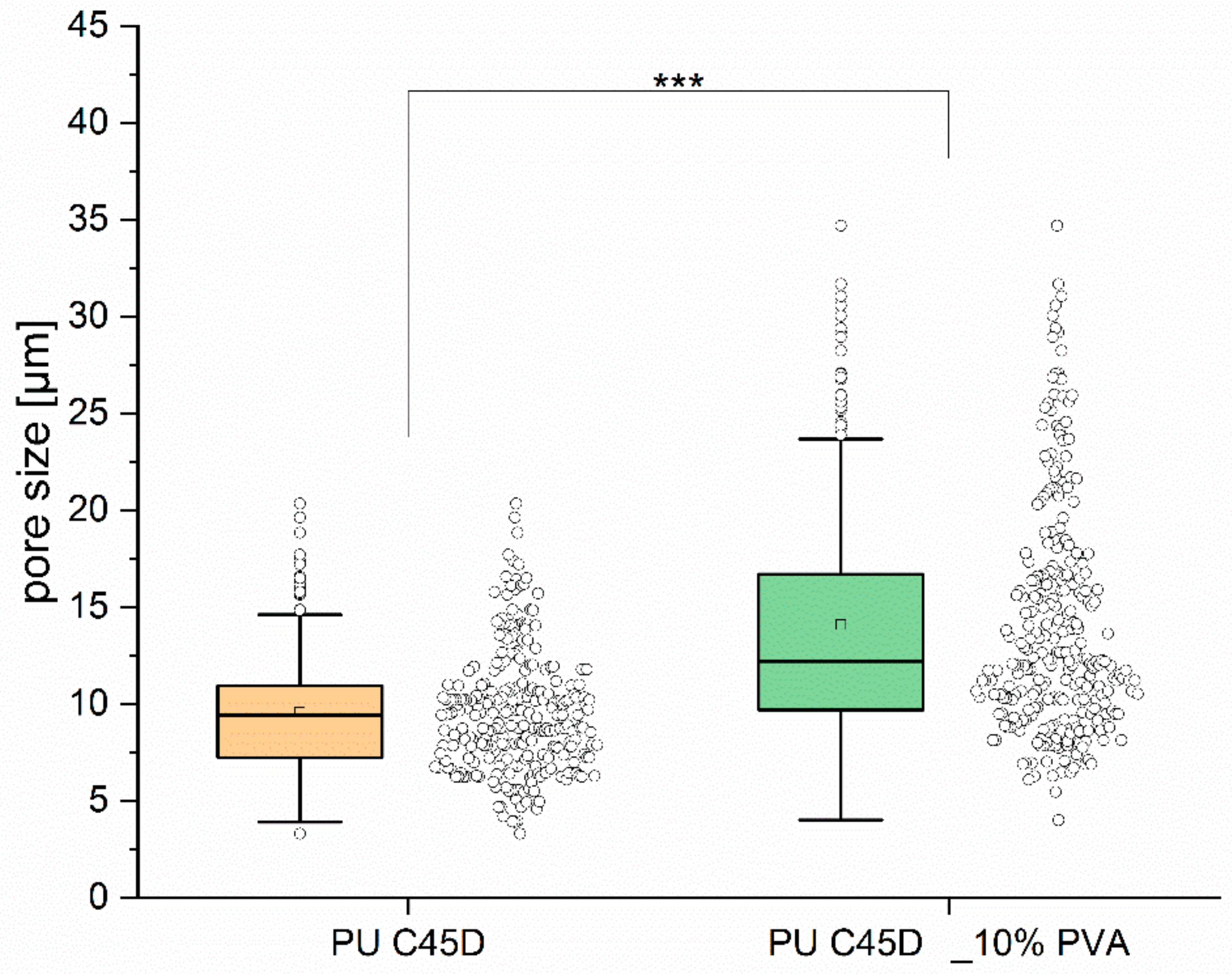
| Wall thickness (μm) | 202.0 ± 19.0 | * | 278.0 ± 78.0 |
| Internal diameter (mm) | 5.35 ± 0.1 | 5.41 ± 0.2 | |
| Average surface pore diameter (μm) | 9.6 ± 3.2 | * | 15.2 ± 6.4 |
| Min surface pore diameter (μm) | 3.3 | 5.5 | |
| Max surface pore diameter (μm) | 20.4 | 41.2 | |
| Porosity (%) | 59 ± 2 | * | 68 ± 3 |
| Young’s modulus (MPa) | 3.6 ± 1.5 | 9.7 ± 4.3 | |
| Tensile strength (MPa) | 11.2 ± 1.2 | * | 6.9 ± 2.3 |
| Elongation at break [mm/mm] | 4.7 ± 0.4 | * | 1.9 ± 0.2 |
| Cell viability (%) | 92 ± 4 | 86 ± 9 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kuźmińska, A.; Kwarta, D.; Ciach, T.; Butruk-Raszeja, B.A. Cylindrical Polyurethane Scaffold Fabricated Using the Phase Inversion Method: Influence of Process Parameters on Scaffolds’ Morphology and Mechanical Properties. Materials 2021, 14, 2977. https://doi.org/10.3390/ma14112977
Kuźmińska A, Kwarta D, Ciach T, Butruk-Raszeja BA. Cylindrical Polyurethane Scaffold Fabricated Using the Phase Inversion Method: Influence of Process Parameters on Scaffolds’ Morphology and Mechanical Properties. Materials. 2021; 14(11):2977. https://doi.org/10.3390/ma14112977
Chicago/Turabian StyleKuźmińska, Aleksandra, Dominika Kwarta, Tomasz Ciach, and Beata A. Butruk-Raszeja. 2021. "Cylindrical Polyurethane Scaffold Fabricated Using the Phase Inversion Method: Influence of Process Parameters on Scaffolds’ Morphology and Mechanical Properties" Materials 14, no. 11: 2977. https://doi.org/10.3390/ma14112977
APA StyleKuźmińska, A., Kwarta, D., Ciach, T., & Butruk-Raszeja, B. A. (2021). Cylindrical Polyurethane Scaffold Fabricated Using the Phase Inversion Method: Influence of Process Parameters on Scaffolds’ Morphology and Mechanical Properties. Materials, 14(11), 2977. https://doi.org/10.3390/ma14112977







