1. Introduction
Nickel compounds are one of the most dangerous environmental pollutants. The release of nickel-containing effluents into the environment can cause severe soil and water pollution [
1]. Nickel is used in a wide range of applications in numerous industrial processes, the most important of which are mineral processing, electroplating, and production of paints, chemicals, and batteries [
1,
2].
Traditional techniques such as ion-exchange, membrane filtration, chemical precipitation, and adsorption are used for nickel removal from wastewater. The application of these techniques is limited due to the resulting generation of substantial volumes of sludge, which leads to secondary environmental pollution, as well as the high cost (including sludge disposal), high energy requirements, and impracticality at low metal concentrations in wastewater [
2,
3]. Biosorption using dry or alive microorganisms can be considered as an alternative to conventional techniques [
2].
Among the microorganisms used as biosorbents, bacteria have an important role due to their ubiquity, large surfaces, resistance to metal ions at high concentrations, and the capability to reduce metal ions to less toxic forms. Often, biofilms alone show low adsorption capacity and their removal from treated effluents is a difficult task. To face this challenge, the immobilization of bacteria onto cheap, porous materials is a suitable option [
4,
5]. Zeolites are excellent ion exchangers which are characterized by a structural negative charge, which makes them perfect candidates for the removal of cations. Zeolites are readily available materials possessing excellent thermal and radiation stability and are characterized by a three-dimensional tetrahedral network and uniform porous structure [
6]. At the same time, it should be mentioned that zeolites possess a low affinity for anions. The application of hybrid biosorbents (zeolite and bacteria) can improve the removal capacity for heavy metals, including anions [
7].
An
Escherichia coli biofilm placed on zeolite was applied for Cu(II) and Zn(II) ion removal from aqueous solutions [
4]. Quintelas et al. [
8] studied the efficiency of chromium(VI), cadmium, nickel, and iron removal by
Escherichia coli biofilm supported on zeolite NaY. The biosorption behavior of a biofilm of
Arthrobacter viscosus placed on 13 X zeolite towards Ni(II) was investigated by Lameiras et al. [
9].
Arthrobacter viscosus supported on NaY zeolite was applied for dye and Cr(VI) removal [
7]. The bacterial biofilm of
Arthrobacter viscosus placed on activated carbon and natural zeolite was used to adsorb Cr (VI) from solutions [
10]. The possibility of using a biofilm of
Shewanella xiamenensis placed on zeolite for the treatment of complex chromium-containing effluents was reported by Zinicovscaia et al. [
5].
In the present study, the biosorption capacity of a hybrid adsorbent, which consisted of Shewanella xiamenensis biofilm and zeolite, for the treatment of synthetic and real nickel-containing effluents under different experimental conditions was investigated. Adsorption equilibrium data were described using Langmuir, Freundlich, and Temkin models. The kinetics data were analyzed using pseudo-first, pseudo-second-order, Elovich, and Webber Morris models. The thermodynamic parameters of the biosorption were investigated. The effect of pH, sorbent dosage, and effluent dilution on Ni(II) removal from real effluent was assessed.
4. Discussion
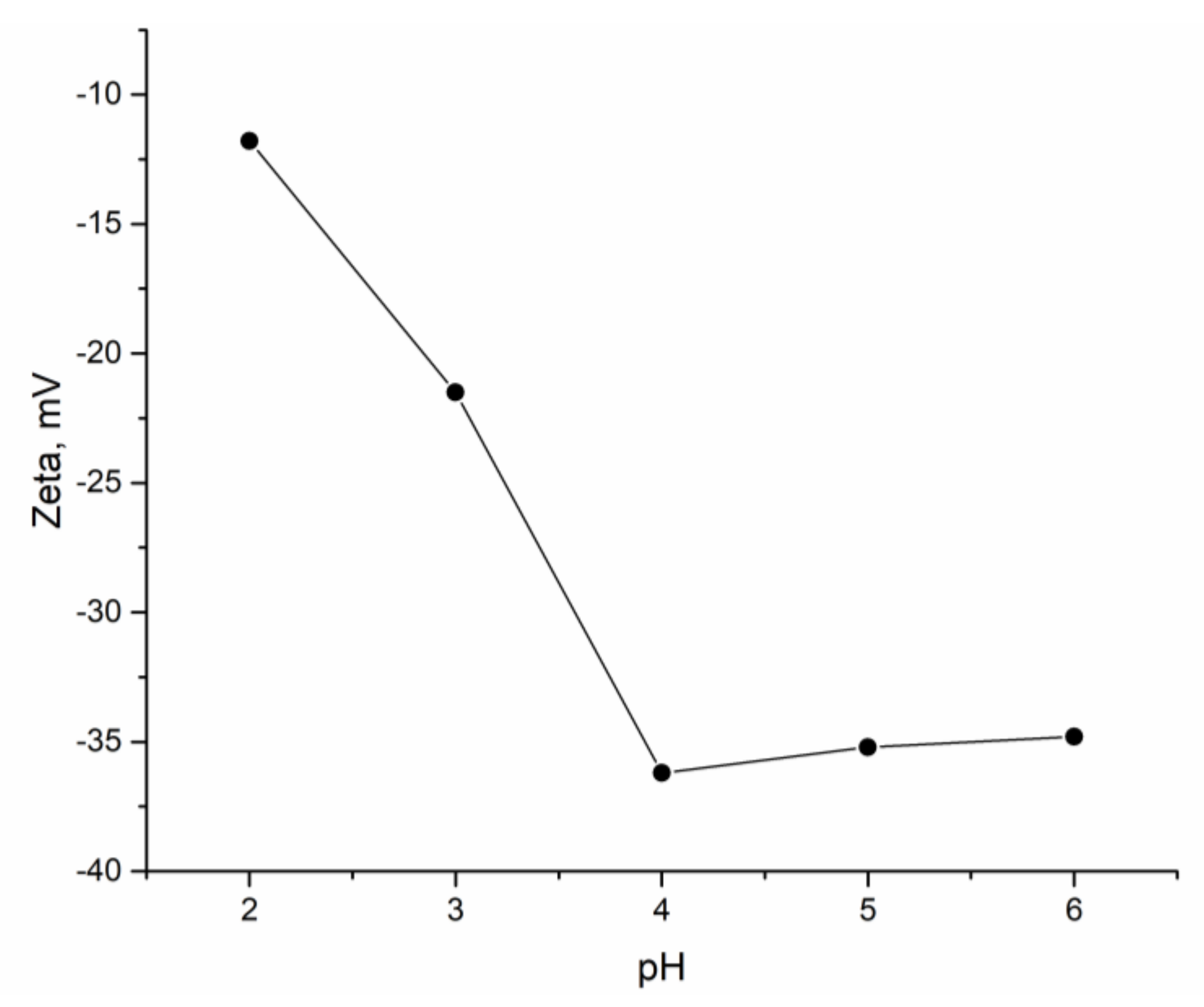
A study on the effect of pH on the sorption capacity of sorbent showed that the obtained hybrid adsorbent (
Shewanella xiamenensis biofilm placed on zeolite) was effective for metal cation removal. The highest removal of Ni(II) was achieved in Ni(II) and Ni(II)-Cr(VI)-Fe(III) systems, in which there was no competition of other metal cations with Ni(II) for binding sites. The negative zeta potential values obtained at the studied pH range created unfavorable conditions for the sorption of anions. According to the thermodynamic calculations (
Figure S1), at the pH range 2–8.2, nickel was present in the solution in dissolved form, while at pH > 8.2, low-soluble Ni(OH)
2 was formed. In the case of the studied hybrid adsorbent, it is suggested that Ni(II) can interact with functional groups of
Shewanella xiamenensis, as well as with zeolite. In our previous research, it was shown that at Ni(II) concentration in a solution of 10–14 mg/L, removal by cyanobacteria did not exceed 66% [
28]. Bacteria showed higher Ni(II) removal and up to 80% of Ni(II) was removed by the species
Pseudomonas cepacian 120S and
Bacillus subtilis 117S from a solution at pH 7.0 [
29].
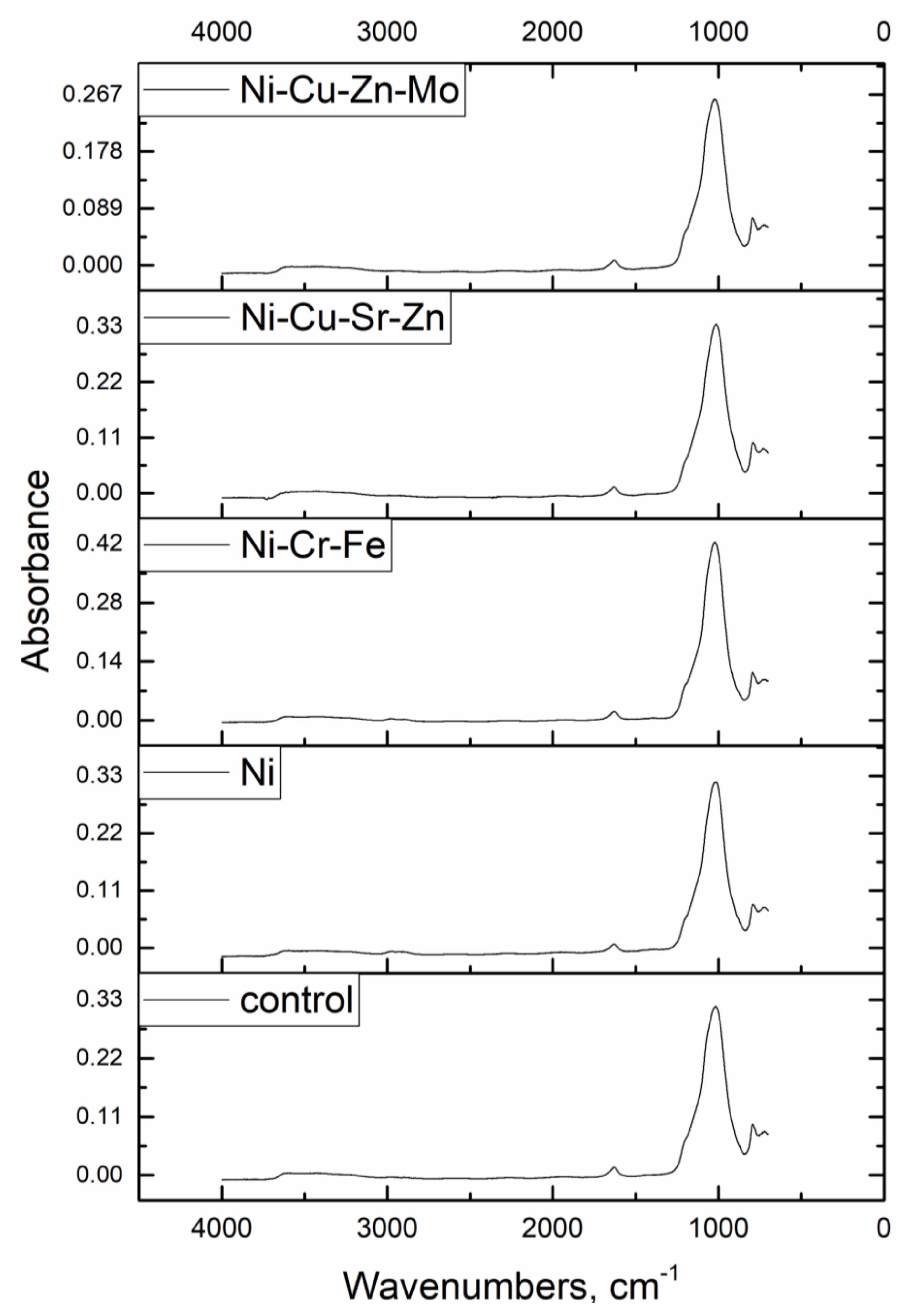
Hydroxyl, carboxyl, carbonyl, and amino groups play an important role in Ni(II) binding by microorganisms [
2,
15]. Zeolites are minerals in which the process of chemical immobilization of heavy metals is based on the exchange of the alkali and alkaline earth metal cation with heavy metals [
5]. FTIR analysis showed that biofilm functional groups were not involved in metal ion trapping. Thus, ion exchange can be proposed as the main mechanism of metal ion uptake from the analyzed solutions. Data obtained for Na and K using neutron activation analysis confirmed this fact since their content in the metal-loaded adsorbent decreased by 2–18% in respect to the control adsorbent. The theoretical exchange capacity of zeolite for Ni(II) was 4–11 times higher than the actual exchange capacity [
30].
In a study by Handley-Sidhu et al [
31], it was shown that the biofilm was responsible for less than 30% of Sr
2+ sorption by biogenic hydroxyapatite. Ni(II) removal by
Arthrobacter viscosus supported on zeolite 13 X constituted 94.1%. Authors showed that Ni(II) removal by zeolite was only faster than that by bacteria, suggesting that the main role in the removal of nickel belongs to zeolite, while bacteria slightly improve it [
9]. Experiments performed on zeolite and on
Shewanella xiamenensis separately (data not shown) demonstrated that zeolite was able to remove 70% of Ni(II) ions from the Ni(II) system; however, equilibrium was attained in 24 h.
Shewanella xiamenensis removed 60% of Ni(II) in 30 min and then equilibrium was attained. Thus, it can be concluded that the main role in metal removal from the analyzed systems belonged to zeolite and bacterial biofilm accelerated the metal sorption process.
In the Ni(II)-Cr(III)-Fe(III) system, at the studied pH range, nickel was present in the solution in dissolved form and the chromium dominant form was hydrochromate ions, HCrO
4− (
Figure S2). Iron(III) started to form a solid mineral phase at pH > 4.2. Ni(II) was almost completely removed in this system, while Cr(VI) and Fe(II) removal did not occur. According to literature data, it is known that Cr(VI) and Fe(II) are efficiently removed from solution at low pH values [
27,
32]. Low removal of Cr(VI) and Fe(II) by the studied biosorbent can be explained by possible biofilm disruption at acidic pH and the negative charge of the hybrid adsorbent, as zeta potential data showed.
In the other two systems, Ni(II) removal decreased almost by 30% in comparison with the one previously described. This can be associated with the presence of other metal cations in the analyzed systems and their competition for binding sites. In the Ni(II)-Cu(II)-Sr(II)-Zn(II) system, metal ions are present in the solution in dissolved form at the studied pH range (
Figure S3). The preference of biosorbent for the analyzed elements changed in the following order: Zn > Sr > Cu > Ni. The adsorption capacity of zeolite for lead was significantly higher than for nickel [
30]. Potassium content in the metal-loaded sorbent decreased by 15% in comparison with the control, while Na content was not affected by the sorption process.
In the Ni(II)-Zn(II)-Mo(VI)-Cu(II) system, Zn(II) and Ni(II) were present in soluble forms. Molybdenum in the analyzed complex system is present in the solution in dissolved form at pH 2.0–3.0, and at the pH range of 3.0–6.0, it is present in the form of copper molybdate. Copper is present both in dissolved form and in precipitated form as molybdate, CuMoO
4 (
Figure S4). The presence of Mo(VI) in the analyzed system did not affect the removal of other metal cations, while its removal from the solution did not occur. Besides ion exchange, metal precipitation can be expected as another mechanism of metal removal.
As was previously mentioned, the removal of anions by zeolite is hampered by the negative charge of the material. The maximum removal of chromium by a biofilm of
Escherichia coli supported on NaY zeolite was achieved at pH in the range 4.6–5.1, of iron at pH 2.7–3.5, and of nickel at pH 5.7–6.2 [
8]. The maximum removal of copper (54.98 %) and zinc (57.32%) by
Escherichia coli biofilm placed on zeolite was attained at pH values of 4.8–5.7 and 4.5–5.5, respectively [
4]. In experiments described by refs. [
5,
7], it was possible to achieve 50% and 35% removal of Cr(VI) by bacterial biofilm supported by zeolite. In the presented studies, living bacteria were used, which resulted in Cr(VI) reduction to Cr(III) and further sorption. In the present study, dried biosorbent was used and only sorption processes took place. Molybdenum was adsorbed at pH 3.0 on zeolite as a FeOMoO
2(OH)·2H
2O inner-sphere complex [
33].
The optimal time for maximum removal of metal ions from the analyzed systems varied from 60 to 150 min. Removal of Cr(VI) by natural zeolite was lower and lasted longer than removal by grape and olive wastes used as adsorbents [
34]. Optimum nickel uptake by natural zeolite was achieved at a contact time of 56–68 min and a pH of 4.8–6 [
1]. The equilibrium time for adsorption of copper and zinc on
Escherichia coli biofilm placed on zeolite was achieved after 5 and 4 days, respectively [
4].
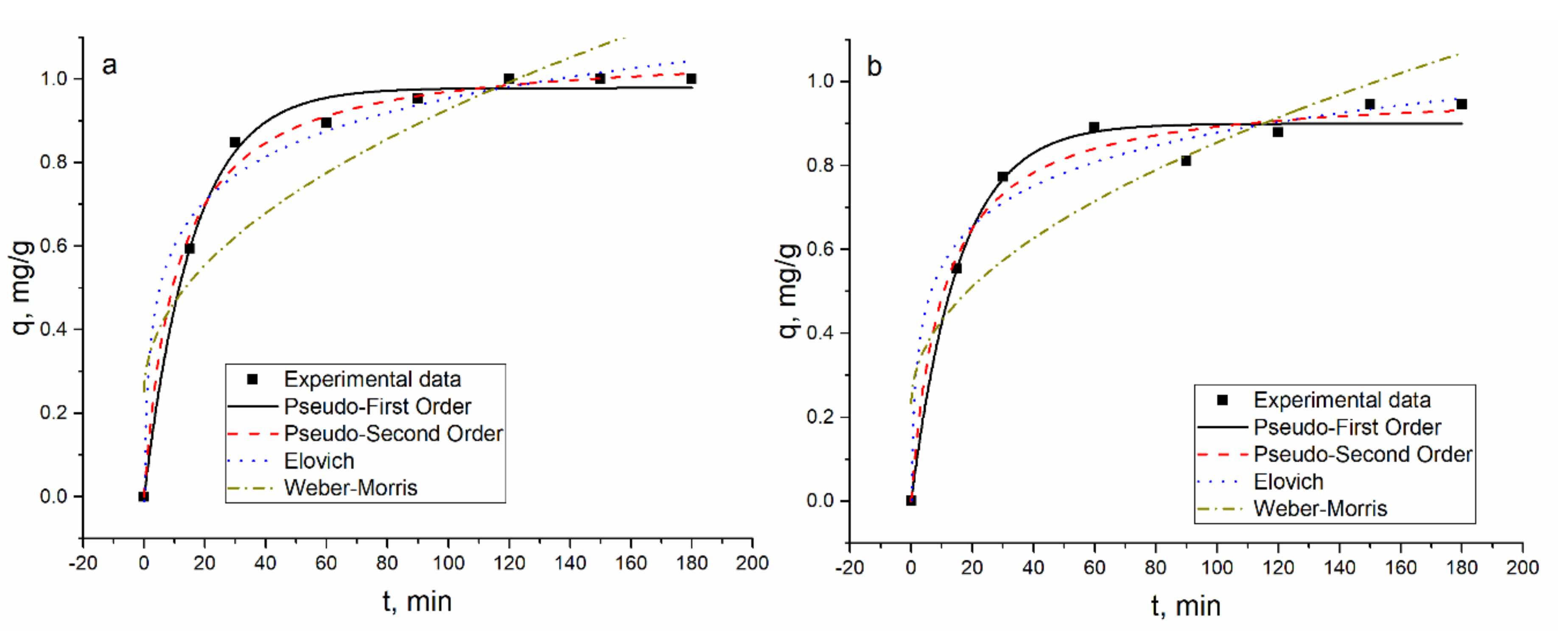
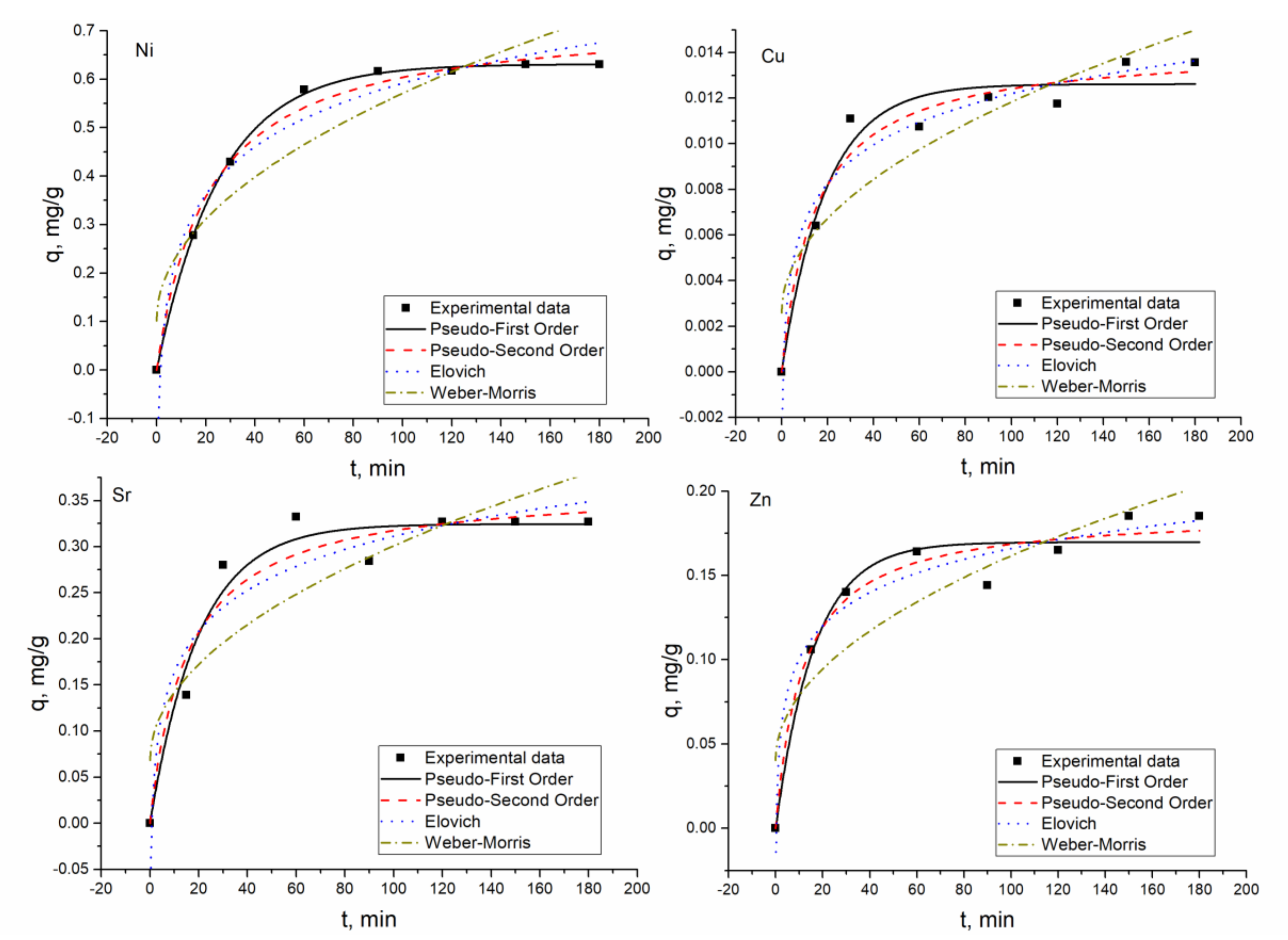
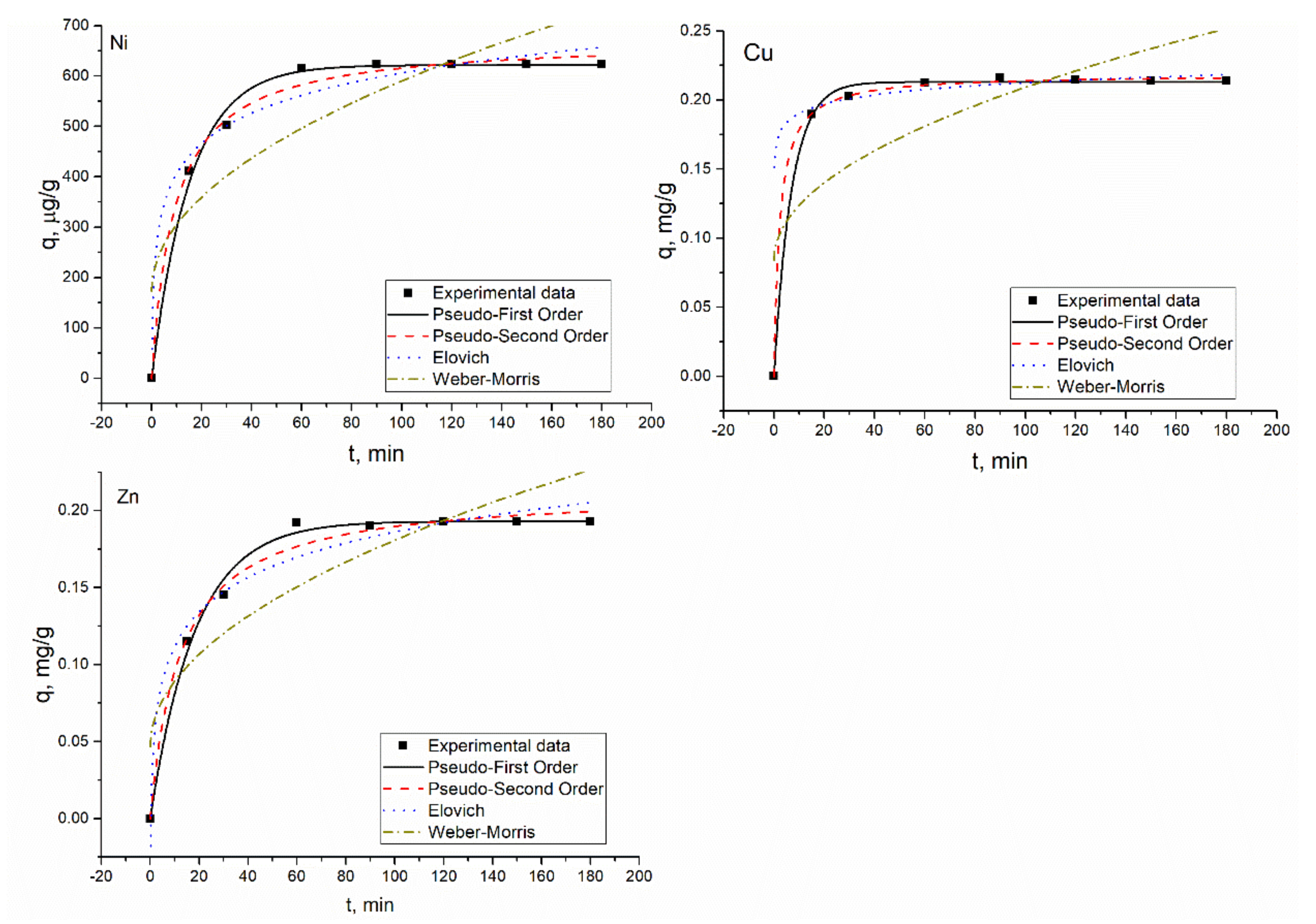
According to the coefficients of determination of the applied models, the PFO, PSO, and EM were found to be applicable to describe experimentally obtained data. The applicability of PSO and EM showed that chemical absorption and ion exchange are the main mechanisms of metal sorption by the hybrid adsorbent [
27]. The R
2 values for most of the elements in the analyzed systems were similar and ESS were calculated additionally. The EES values for both models, PFO and PSO, were very similar, indicating that the used models fit the data well. Since the R
2 and ESS values for both models were very close, adsorption rate values were used as criteria to determine the best fitting model. For all elements except Ni(II) in Ni(II)-Zn(II)-Mo(VI)-Cu(II), the adsorption rate values calculated for the PSO model were higher than for the PFO model, pointing to a higher rate of adsorption. The PSO model fits better Ni(II) biosorption on
Arthrobacter viscosus biofilm supported on 13 X zeolite [
9].
The increase in Ni(II) concentration in the analyzed solutions up to 100 mg/L led to increased biosorbent adsorption capacity, up to 2.9 mg/g in Ni(II), Ni(II)-Cr(VI)-Fe(III), and Ni(II)-Sr(II)-Cu(II)-Zn(II) systems, while in the Ni(II)-Zn(II)-Mo(VI)-Cu(II) system, it constituted only 1.7 mg/g. At the same time, the Ni(II) removal efficiency decreased as a result of the increase in its concentration in the solution. At low metal concentrations, there are enough binding sites on the sorbent surface, and at higher metal concentrations, the decrease in the absorption is explained by the saturation of the adsorption sites [
1]. In Ni(II)-Cr(VI)-Fe(III) and Ni(II)-Sr(II)-Cu(II)-Zn(II) systems, Ni(II) adsorption was not affected by the presence of other metal ions in the solution. Since equilibrium experiments were performed at pH 6.0, optimal for Ni(II) removal, the removal of Cr(VI) and Fe(III) did not occur and the same was observed for Mo(VI) in the Ni(II)-Zn(II)-Mo(VI)-Cu(II) system. In the Ni(II)-Sr(II)-Cu(II)-Zn(II) system, Ni(II) sorption was similar to that of the Ni(II) system, indicating that metal ions present in the complex system did not affect its removal. The decrease in Na and K content in the metal-loaded adsorbent did not exceed 10%. The lowest adsorption of Ni(II) was in the Ni(II)-Zn(II)-Mo(VI)-Cu(II) system. It is important to mention that in this system, a significant reduction of Na (by 20%) and K (by 30%) took place, which may indicate their involvement in ion exchange. Cu(II) removal was almost unaffected by the increase in nickel concentration in the solution, while Zn(II) removal drastically decreased. At a nickel concentration in the solution of 10 mg/L, 99% of Zn(II) was removed from the solution but a 10-fold increase in nickel concentration led to a decrease in its removal up to 39%.
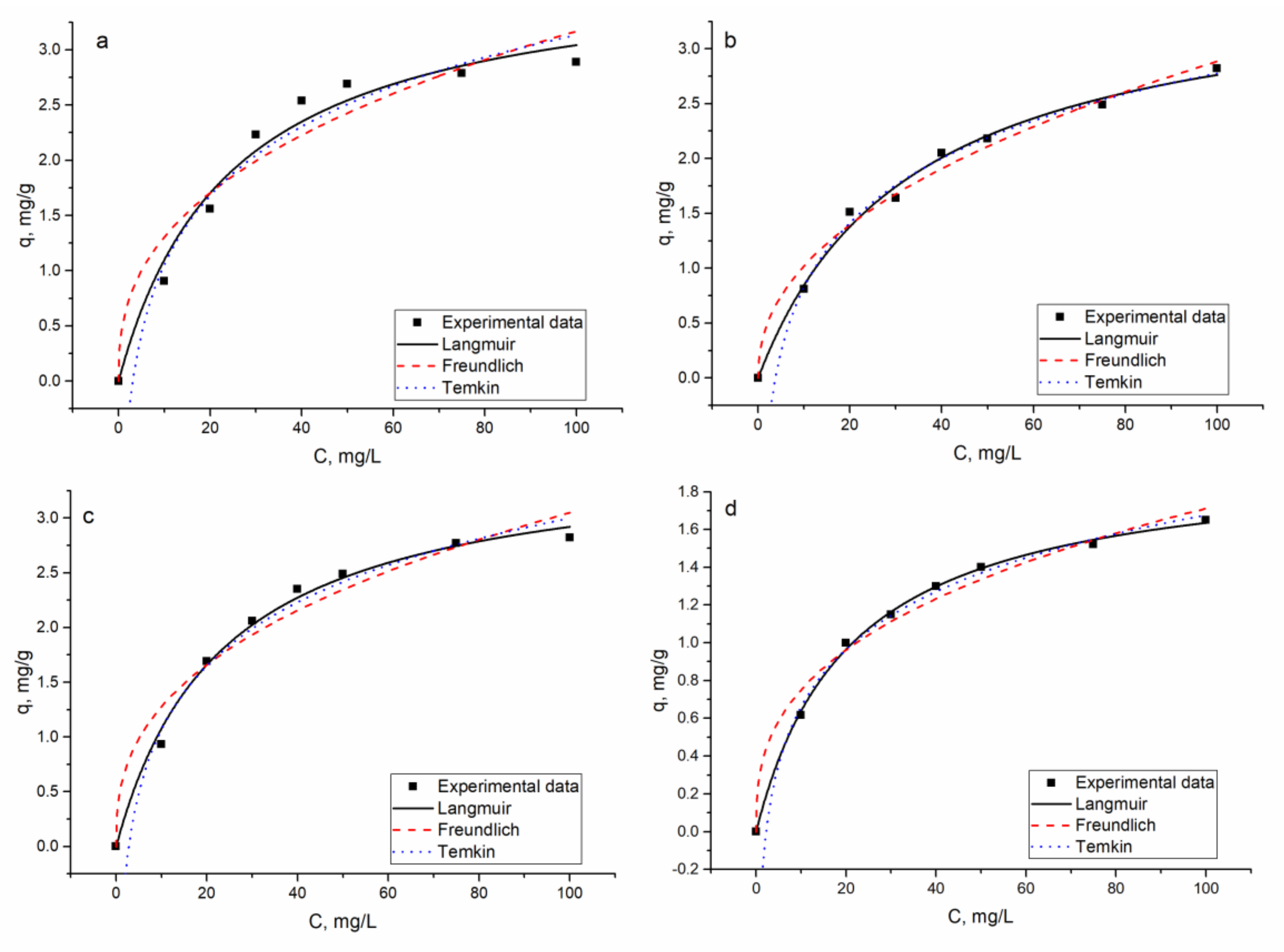
The Langmuir model, which assumes a monolayer sorption, fit well the data obtained for Ni(II). The maximum sorption capacity in three of four systems was on the level of 3.6–3.9 mg/g, while in the Ni(II)-Zn(II)-Mo(VI)-Cu(II) system, it was significantly lower (1.98 mg/g). The R
2 values for the Freundlich model were lower. For the Temkin isotherm, R
2 values were on the level of the Langmuir model and the constant (B) values related to the heat of adsorption were in the range 0.4–0.9 kJ/mol. Adsorption is considered a physical process if bonding energy is in the range of 5–40 kJ/mol and the chemical one in the energy range 40–800 kJ/mol. The low values of energy obtained in the Temkin model in the present study suggested weak ionic interaction between the sorbate and the sorbent [
35].
The sorption capacity of the analyzed hybrid sorbent toward Ni(II) was compared with data available for other sorbents (
Table 7). The maximum sorption capacity obtained in the present study was lower than the data presented in the main part of the research, except for El-Sadaawy et al.’s [
36] study.
The rise in temperature in the experimental solutions resulted in growth of Ni(II), Sr(II), and Zn(II) removal, while Cu(II) removal was not dependent on the temperature. The temperature increase to 50 °C in all analyzed systems had a positive effect on Ni(II) removal efficiency. According to the Gibbs energy values, the biosorption in the analyzed systems was spontaneous physical sorption [
40]. Positive
∆Hº values were obtained for all metal ions and indicated by the endothermic character of the biosorption. Positive
ΔS° values acquired for cations in the analyzed systems suggested high affinity and the presence of a low energy barrier of metal adsorption processes using
Shewanella xiamenensis biofilm placed on zeolite [
41].
The dosage of biosorbent and pH are the key parameters for efficient metal ion removal from industrial effluents. In the studied effluent, Ni(II) concentration was 125 mg/L, concentrations of other metal ions were considerably lower, and they were excluded from further discussion. The maximum removal of Ni(II) was achieved at pH range 4.0–6.0 and amounted to 17%, which was considerably lower than values obtained in batch experiments. Ni(II) removal by natural zeolite from wastewater constituted 60%; however, the initial Ni(II) concentration was almost 35 times lower than in the present study [
42]. One of the ways to increase metal ion removal is to increase the dosage of the adsorbent. The increase in sorbent dosage from 0.5 to 2.0 g resulted in a rise in Ni(II) removal only by 10%. The percent of Ni(II) adsorbed by coir pith from an effluent containing 145 mg/L of Ni(II) was 88% at 5% (w/v) adsorbent dosage and a further increase in sorbent dosage did not increase nickel removal [
43]. The low rate of Ni(II) removal with the increase in sorbent dosage can be explained by sorbent particle agglomeration, which reduces the specific surface area and increases the diffusion path length [
44]. In batch experiments, it was shown that with the increase in Ni(II) concentration in solution, its removal significantly decreases from 90% at Ni(II) concentration 10 mg/L to 28% at Ni(II) concentration in solution 100 mg/L. Thus, in the next experiment, the effluent was diluted two and twelve times and 0.5 or 1.0 g of sorbent was added to it. Ni(II) removal from the effluent diluted two times did not exceed 27%; however, a 12-fold effluent dilution resulted in the removal of 72% of Ni(II). Thus, it can be concluded that applied biosorbents are better suited for output wastewater which has been pretreated by traditional techniques or for wastewater with low Ni(II) concentrations. The efficiency of zinc and copper removal from the input raw wastewater into the treatment plant and output wastewater by
E. coli biofilm placed on the zeolite was higher for wastewater treated previously by traditional techniques [
4].
Besides the sorption capacity, one of the most important parameters of the biosorbent which determines its wide application is the cost. The price of zeolite in Russia varies from 78 to 916 USD per ton, depending on the supplier. The price of zeolite used in the present study was 380 USD per ton. Bacteria
Shewanella xiamenensis do not require special growth conditions and the cost of mineral salt used for growth constituted 0.2 USD per liter of cultivation medium. The final price of the hybrid adsorbent would constitute 530 USD per ton of sorbent. The price of the studied hybrid adsorbent would be comparable with the price of ALO sorbent (200–600 USD per ton) [
6], higher than the price of activated charcoal (approximately 100 USD per ton) [
45] but lower than the price of single-walled carbon nanotubes, multiwalled carbon nanotubes, and granular activated carbon (90,000, 12,000, and 1000 USD per ton, respectively) [
37]. It should be mentioned that the zeolite used in the present study was purchased from a commercial company in a small amount; its procurement from the producer in large quantities would significantly decrease the price.