Liquid-Film Assisted Mechanism of Reactive Flash Sintering in Oxide Systems
Abstract
1. Introduction
2. Process Analysis
2.1. Formation of Intermediate Phase by RFS
2.2. Flash Sintering of Doped Oxide
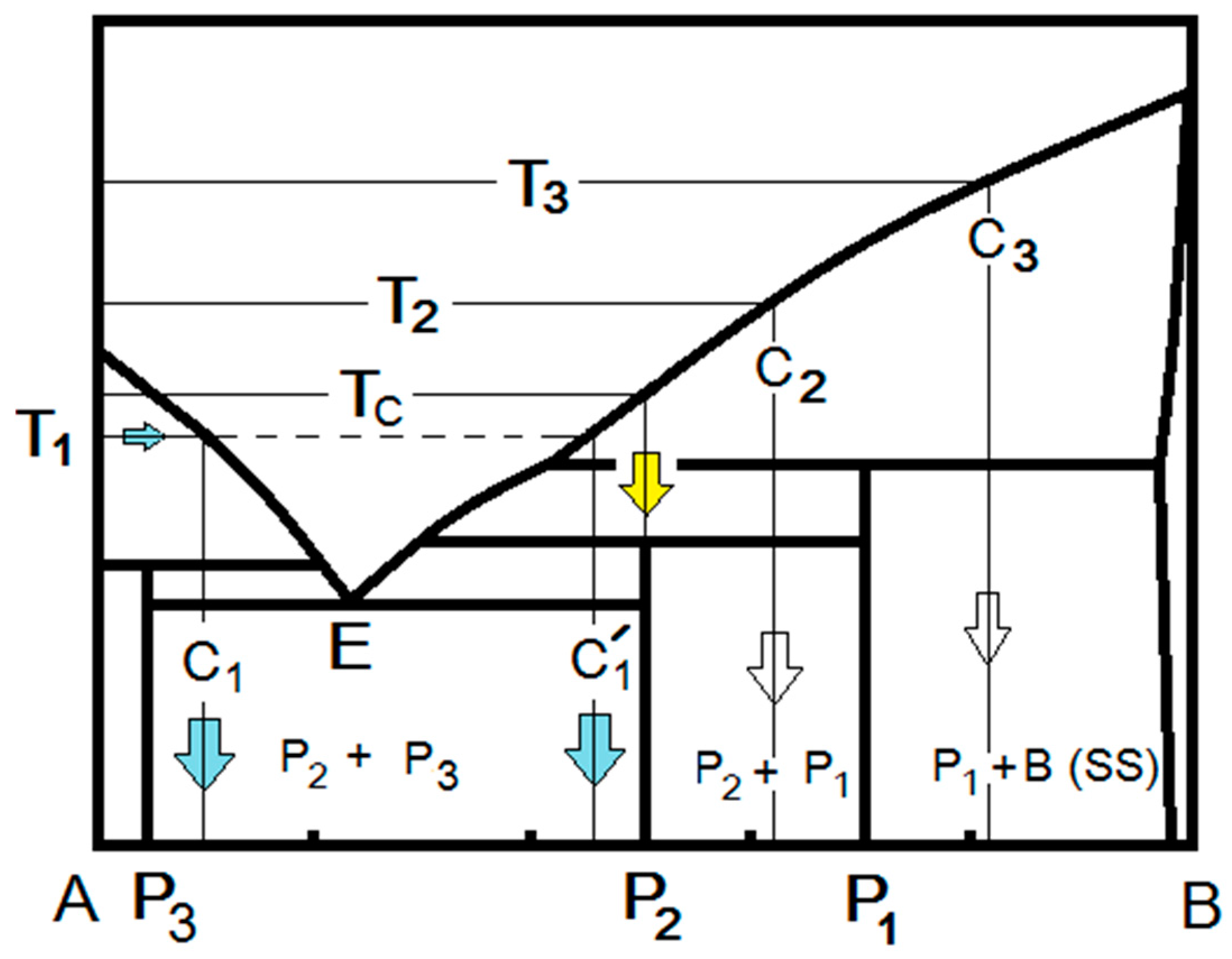
2.3. RFS in Multicomponent Systems
3. Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BBR | Black body radiation |
| FS | Flash sintering |
| RFS | Reaction flash sintering |
| SEM | Scanning electron microscopy |
| WDS | (X-ray) Wavelength dispersive spectroscopy |
References
- Cologna, M.; Francis, J.S.C.; Raj, R. Field assisted and flash sintering of alumina and its relationship to conductivity and MgO-doping. J. Am. Ceram. Soc. 2011, 31, 2827–2837. [Google Scholar] [CrossRef]
- Schmerbauch, C.; Gonzalez-Julian, J.; Röder, R.; Ronning, C.; Guillon, O. Flash sintering of nanocrystalline Zinc oxide and its influence on microstructure and defect formation. J. Am. Ceram. Soc. 2014, 97, 1728–1735. [Google Scholar] [CrossRef]
- Rheinheimer, W.; Phuah, X.L.; Wang, H.; Lemke, F.; Hoffmann, M.J.; Wang, H. The role of point defects and defect gradients in flash sintering of perovskite oxides. Acta Mater. 2019, 106, 398–408. [Google Scholar] [CrossRef]
- Narayan, J. Grain growth model for electric-assisted processing and flash sintering of materials. Scr. Mater. 2013, 68, 785–788. [Google Scholar] [CrossRef]
- Chaim, R. Liquid film capillary mechanism for densification of ceramic powders during flash sintering. Materials 2016, 9, 280. [Google Scholar] [CrossRef] [PubMed]
- Chaim, R. Particle surface softening as universal behaviour during flash sintering of oxide nano-powders. Materials 2017, 10, 179. [Google Scholar] [CrossRef]
- Chaim, R.; Chevallier, G.; Weibel, A.; Estournès, C. Flash sintering of dielectric nanoparticles as a percolation phenomenon through a softened film. J. Appl. Phys. 2017, 121, 145103. [Google Scholar] [CrossRef]
- Yu, M.; Grasso, S.; Mckinnon, R.; Saunders, T.; Reece, M.J. Review of flash sintering: Materials, mechanisms and modelling. Adv. Appl. Ceram. 2017, 116, 24–60. [Google Scholar] [CrossRef]
- Biesuz, M.; Sglavo, V.M. Flash sintering of ceramics. J. Eur. Ceram. Soc. 2019, 39, 115–143. [Google Scholar] [CrossRef]
- Morisaki, N.; Yoshida, H.; Kobayashi, T.; Tokunaga, T.; Yamamoto, T. Intergranular amorphous films formed by DC electric field in pure zirconia. J. Am. Ceram. Soc. 2018, 101, 3282–3287. [Google Scholar] [CrossRef]
- Corapcioglu, G.; Gulgun, M.A.; Kisslinger, K.; Sturm, S.; Jha, S.K.K.; Raj, R. Microstructure and microchemistry of flash sintered K0.5Na0.5NbO3. J. Ceram. Soc. Jpn. 2016, 124, 321–328. [Google Scholar] [CrossRef]
- Uehashi, A.; Sasaki, K.; Tokunaga, T.; Yoshida, H.; Yamamoto, T. Formation of secondary phase at grain boundary of flash-sintered BaTiO3. Microscopy 2014, 63, i19–i20. [Google Scholar] [CrossRef] [PubMed]
- Du, B.; Gucci, F.; Porwal, H.; Grasso, S.; Mahajan, A.; Reece, M.J. Flash spark plasma sintering of magnesium silicide stannide with improved thermoelectric properties. J. Mater. Chem. C 2017, 5, 1514–1521. [Google Scholar] [CrossRef]
- Biesuz, M.; Sglavo, V.M. Current-induced abnormal and oriented grain growth in corundum upon flash sintering. Scr. Mater. 2018, 150, 82–86. [Google Scholar] [CrossRef]
- Chaim, R.; Estournès, C. On thermal runaway and local endothermic/exothermic reactions during flash sintering of ceramic nanoparticles. J. Mater. Sci. 2018, 53, 6378–6389. [Google Scholar] [CrossRef]
- Chaim, R. On the kinetics of liquid-assisted densification during flash sintering of ceramic nanoparticles. Scr. Mater. 2019, 158, 88–90. [Google Scholar] [CrossRef]
- Schie, M.; Menzel, S.; Robertson, J.; Waser, R.; De Souza, R.A. Field-enhanced route to generating anti-Frenkel pairs in HfO2. Phys. Rev. Mater. 2018, 2, 035002. [Google Scholar] [CrossRef]
- Pérez-Maqueda, L.A.; Gil-González, E.; Perejón, A.; Lebrun, J.M.; Sánchez-Jiménez, P.E.; Raj, R. Flash sintering of highly insulating nanostructured phase-pure BiFeO3. J. Am. Ceram. Soc. 2017, 100, 3365–3369. [Google Scholar] [CrossRef]
- Jha, S.K.; Lebrun, J.M.; Raj, R. Phase transformation in the alumina-titania system during flash sintering experiments. J. Eur. Ceram. Soc. 2016, 36, 733–739. [Google Scholar] [CrossRef]
- Gil-González, E.; Perejón, A.; Sánchez-Jiménez, P.E.; Sayagués, M.J.; Raj, R.; Pérez Maqueda, L.A. Phase-pure BiFeO3 produced by reaction flash sintering of Bi2O3 and Fe2O3. J. Mater. Chem. A 2018, 6, 5356–5366. [Google Scholar] [CrossRef]
- Kok, D.; Yadav, D.; Sortino, E.; McCormack, S.J.; Tseng, K.P.; Kriven, W.M.; Raj, R.; Mecartney, M.L. α-alumina and spinel react into single-phase high-alumina spinel in <3 s during flash sintering. J. Am. Ceram. Soc. 2018. [Google Scholar] [CrossRef]
- Gaskell, D.R. Introduction to the Thermodynamics of Materials, 5th ed.; Taylor & Francis Group LLC: New York, NY, USA, 2008. [Google Scholar]
- German, R.M.; Farooq, S.; Kipphut, C.M. Kinetics of liquid sintering. Mater. Sci. Eng. A 1988, 105–106, 215–224. [Google Scholar] [CrossRef]
- Lu, J.; Qiao, L.J.; Fu, P.Z.; Wu, Y.C. Phase equilibrium of Bi2O3-Fe2O3 pseudo-binary system and growth of BiFeO3 single crystal. J. Cryst. Growth 2011, 318, 936–941. [Google Scholar] [CrossRef]
- Ping, L.R.; Azad, A.M.; Dung, T.W. Magnesium aluminate (MgAl2O4) spinel produced via self-heat-sustained (SHS) technique. Mater. Res. Bull. 2001, 36, 1417–1430. [Google Scholar] [CrossRef]
- Alper, A.M.; McNally, R.N.; Ribbe, P.H.; Doman, R.C. The system MgO-MgAl2O4. J. Am. Ceram. Soc. 1962, 45, 263–268. [Google Scholar] [CrossRef]
- Chiang, Y.M.; Birnie, D.P., III; Kingery, W.D. Physical Ceramics, Principles for Ceramic Science and Engineering; Wiley: New York, NY, USA, 1997. [Google Scholar]
- Jung, I.H.; Eriksson, G.; Wu, P.; Pelton, A. Thermodynamic modeling of the Al2O3-Ti2O3-TiO2 system and its application to the Fe-Al-Ti-O inclusion diagram. ISIJ Int. 2009, 49, 1290–1297. [Google Scholar] [CrossRef]
- Biesuz, M.; Sglavo, V.M. Flash sintering of alumina: Effect of different operating conditions on densification. J. Eur. Ceram. Soc. 2016, 36, 2535–2542. [Google Scholar] [CrossRef]
- Roy, S.K.; Coble, R.L. Solubility of Magnesia, Titania, and Magnesium Titanate in aluminum oxide. J. Am. Ceram. Soc. 1968, 51, 1–6. [Google Scholar] [CrossRef]
- Ando, K.; Momoda, M. Solubility of MgO in single-crystal Al2O3. J. Ceram. Soc. Jpn. 1987, 95, 381–386. [Google Scholar] [CrossRef]
- Greskovich, C.; Brewer, J.A. Solubility of magnesia in polycrystalline alumina at high temperatures. J. Am. Ceram. Soc. 2001, 84, 420–425. [Google Scholar] [CrossRef]
- Miller, L.; Avishai, A.; Kaplan, W.D. Solubility limit of MgO in Al2O3 at 1600 °C. J. Am. Ceram. Soc. 2006, 89, 350–353. [Google Scholar] [CrossRef]
- Biesuz, M.; Luchi, P.; Quaranta, A.; Sglavo, V.M. Theoretical and phenomenological analogies between flash sintering and dielectric breakdown in α-alumina. J. Appl. Phys. 2016, 120, 145107. [Google Scholar] [CrossRef]
- Mishra, R.S.; Mukherjee, A.K.; Yamazaki, K.; Shoda, K. Effect of TiO2 doping on rapid densification of alumina by plasma activated sintering. J. Mater. Res. 1996, 11, 1144–1148. [Google Scholar] [CrossRef]
- Biesuz, M.; Sglavo, V.M. Liquid phase flash sintering in magnesia silicate glass-containing alumina. J. Eur. Ceram. Soc. 2017, 37, 705–713. [Google Scholar] [CrossRef]
- Yoon, B.; Yadav, D.; Ghose, S.; Sarin, P.; Raj, R. On the synchronicity of flash sintering and phase transformation. J. Am. Ceram. Soc. 2019, 102, 3110–3116. [Google Scholar] [CrossRef]
- Kumar, M.; Yadav, D.; Lebrun, J.M.; Raj, R. Flash sintering with current-rate: A different approach. J. Am. Ceram. Soc. 2019, 102, 823–835. [Google Scholar] [CrossRef]
- Chaim, R.; Estournès, C. Effects of the fundamental oxide properties on the electric field—Flash temperature during flash sintering. Scr. Mater. 2019, 163, 130–132. [Google Scholar] [CrossRef]
- Pavlyuchkov, D.; Savinykh, G.; Fabrichnaya, O. Experimental investigation and thermodynamic modeling of the ZrO2-MgO-Al2O3 system. J. Eur. Ceram. Soc. 2014, 34, 1397–1408. [Google Scholar] [CrossRef]

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaim, R.; Amouyal, Y. Liquid-Film Assisted Mechanism of Reactive Flash Sintering in Oxide Systems. Materials 2019, 12, 1494. https://doi.org/10.3390/ma12091494
Chaim R, Amouyal Y. Liquid-Film Assisted Mechanism of Reactive Flash Sintering in Oxide Systems. Materials. 2019; 12(9):1494. https://doi.org/10.3390/ma12091494
Chicago/Turabian StyleChaim, Rachman, and Yaron Amouyal. 2019. "Liquid-Film Assisted Mechanism of Reactive Flash Sintering in Oxide Systems" Materials 12, no. 9: 1494. https://doi.org/10.3390/ma12091494
APA StyleChaim, R., & Amouyal, Y. (2019). Liquid-Film Assisted Mechanism of Reactive Flash Sintering in Oxide Systems. Materials, 12(9), 1494. https://doi.org/10.3390/ma12091494




