Solvent Effects on Gelation Behavior of the Organogelator Based on L-Phenylalanine Dihydrazide Derivatives
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
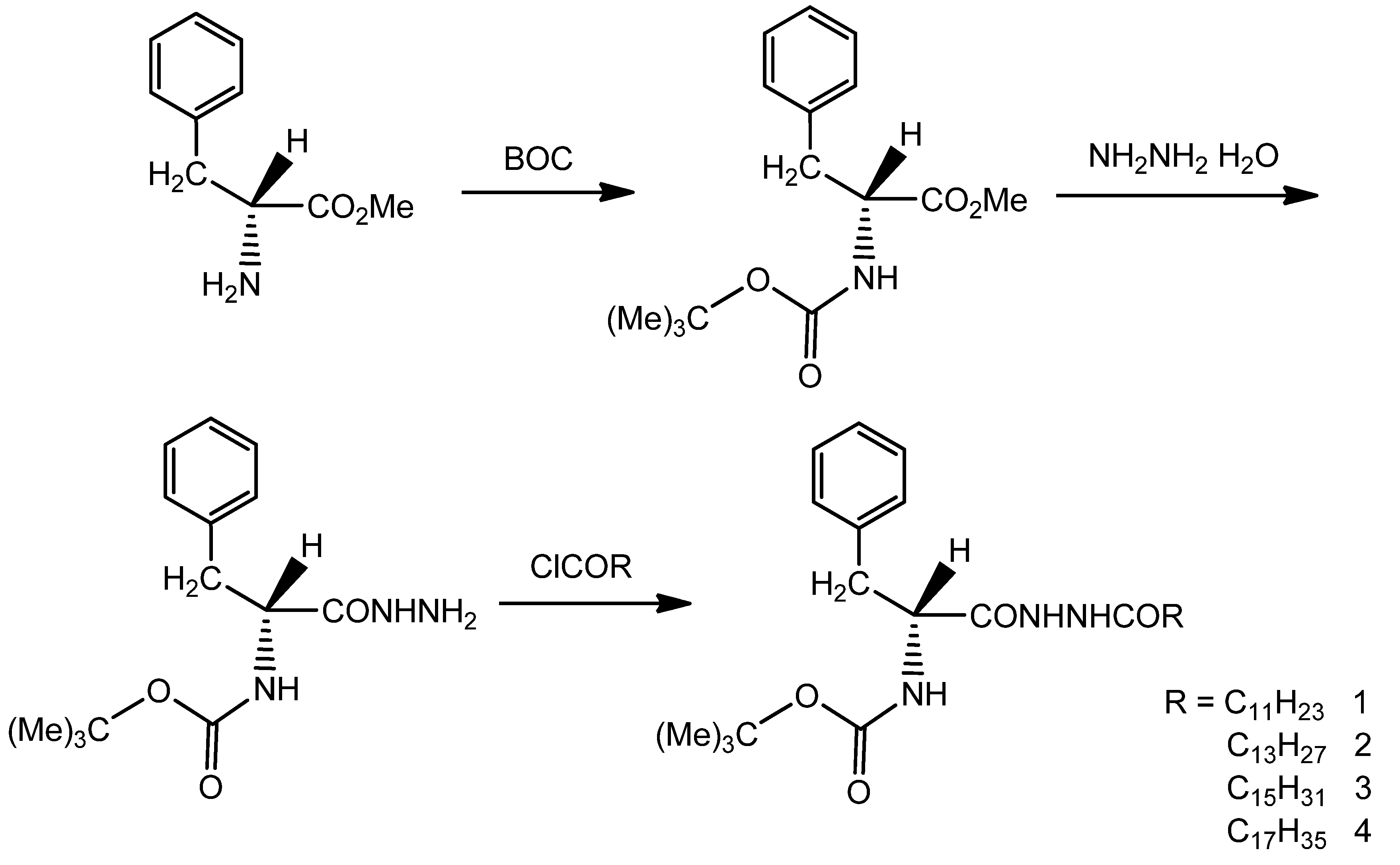
2.2. Synthesis
2.3. Measurements
3. Results and Discussion
3.1. Gelation Behaviors in Some Solvents
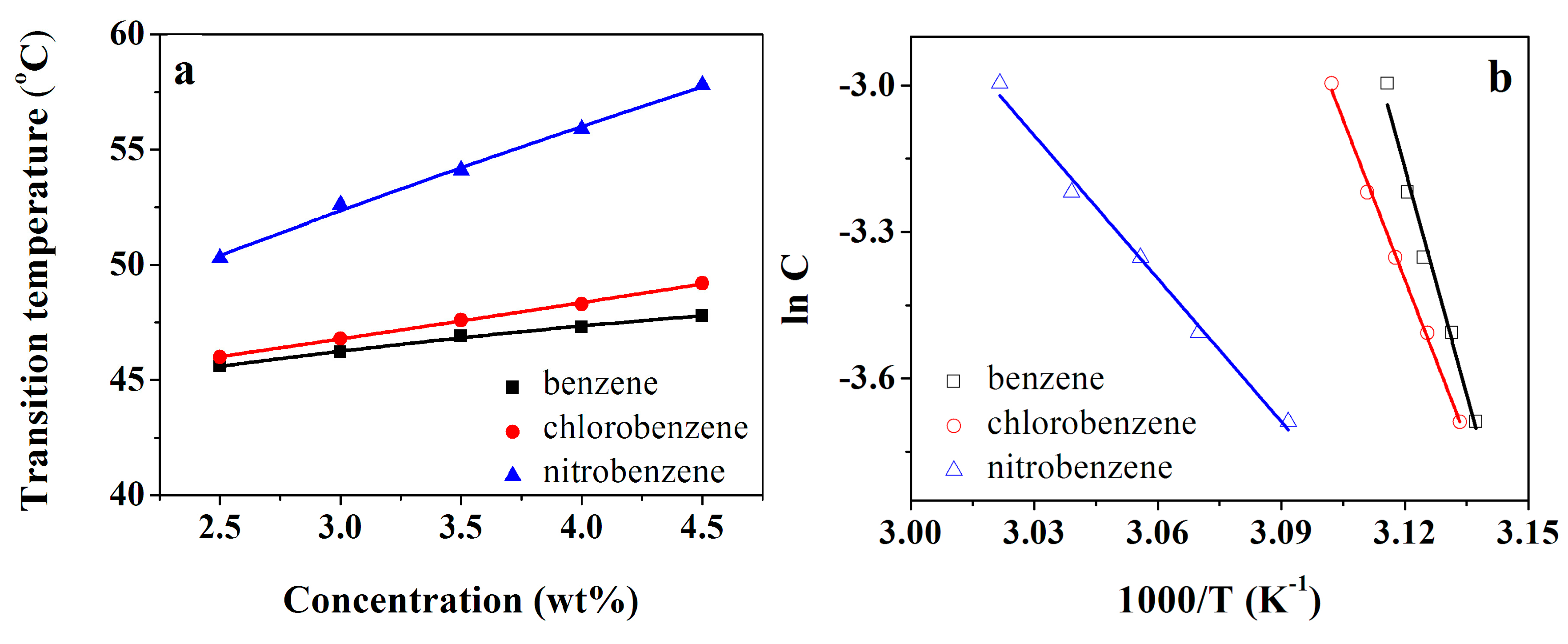
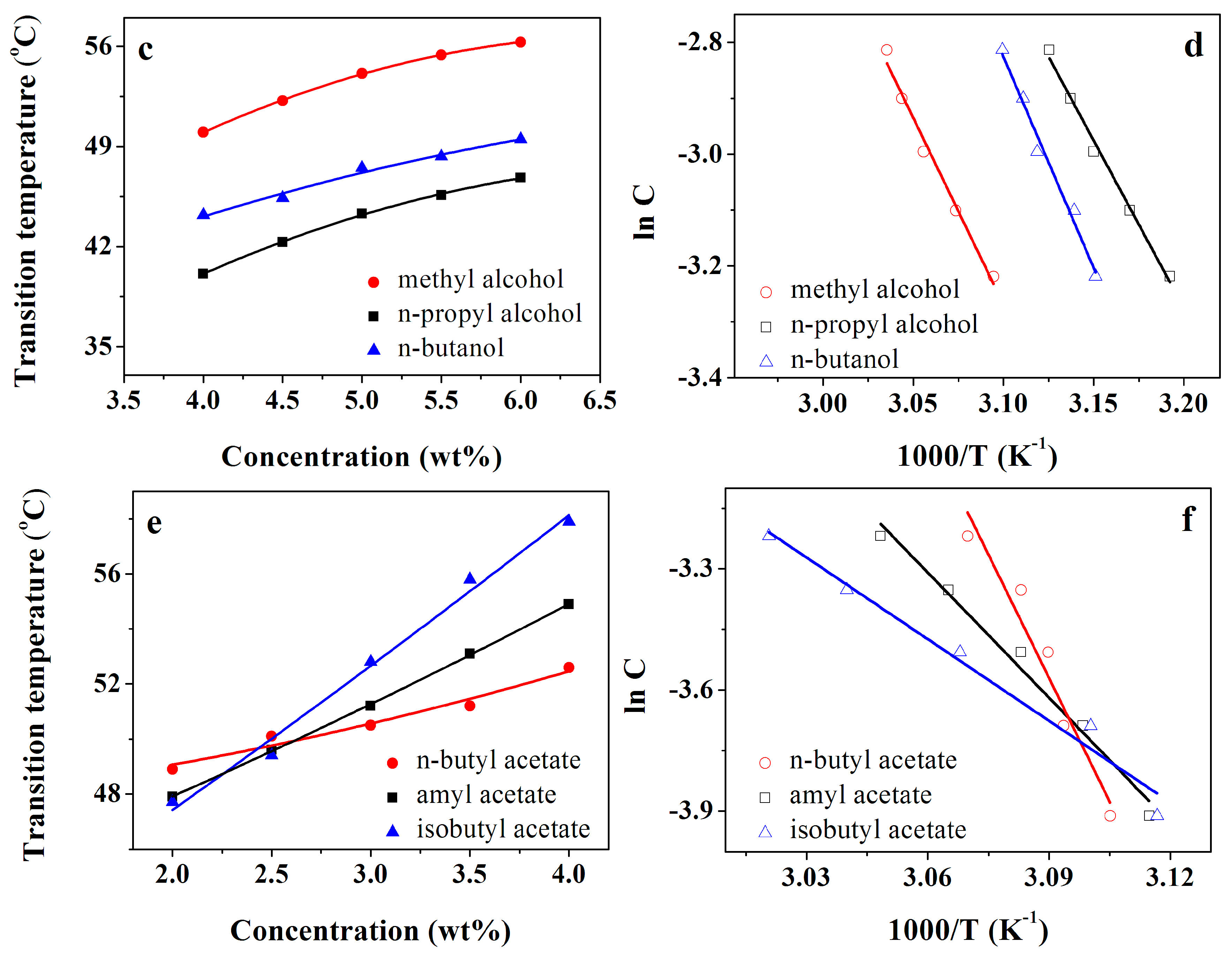
3.2. Gel Stability Research
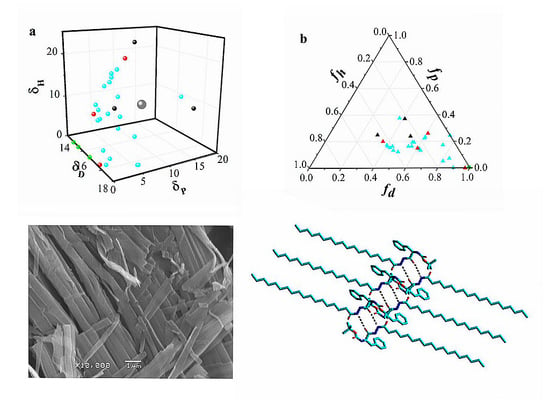
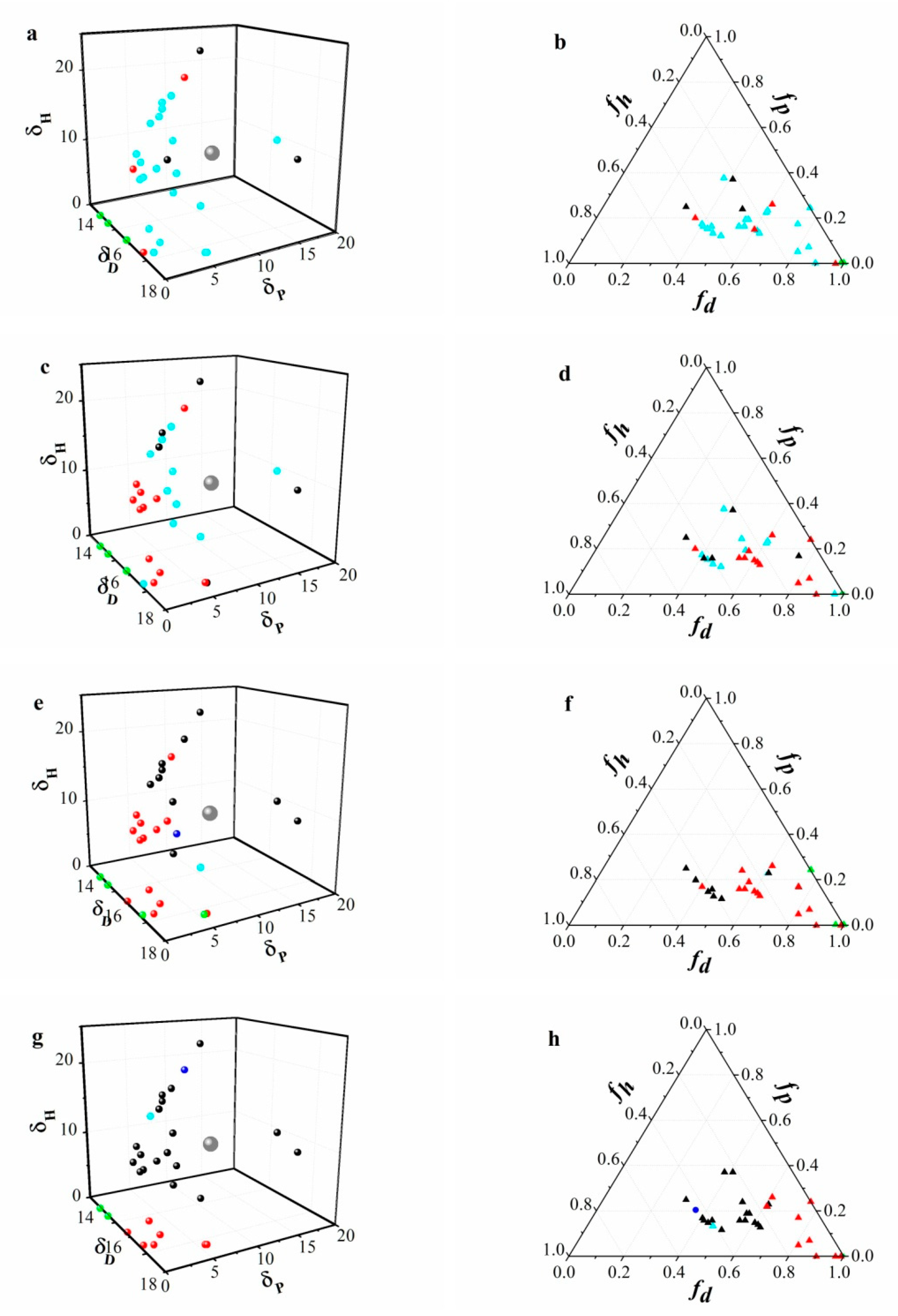
3.3. Hansen Solubility Parameter and Teas plot Presentation
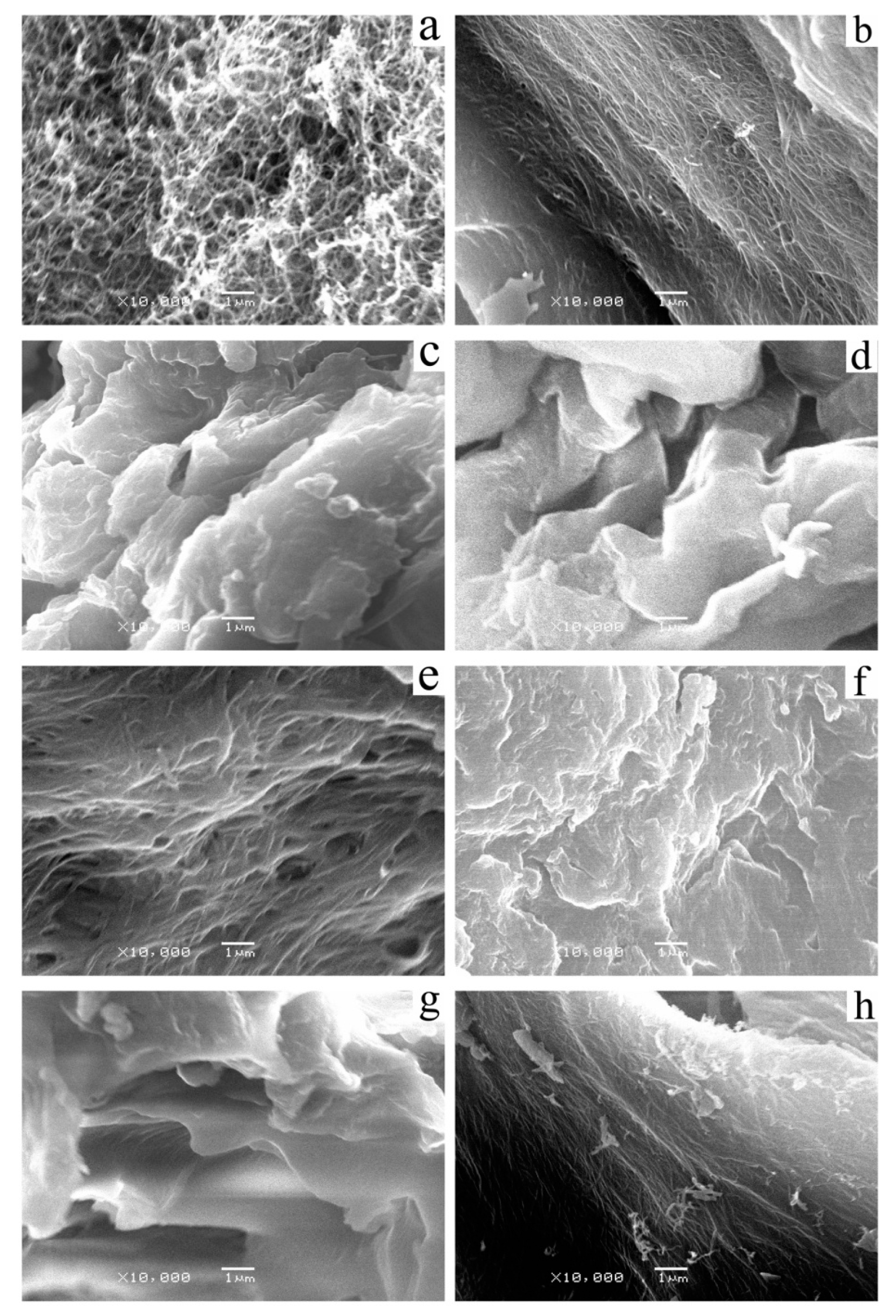
3.4. Morphology Studies of the Gels
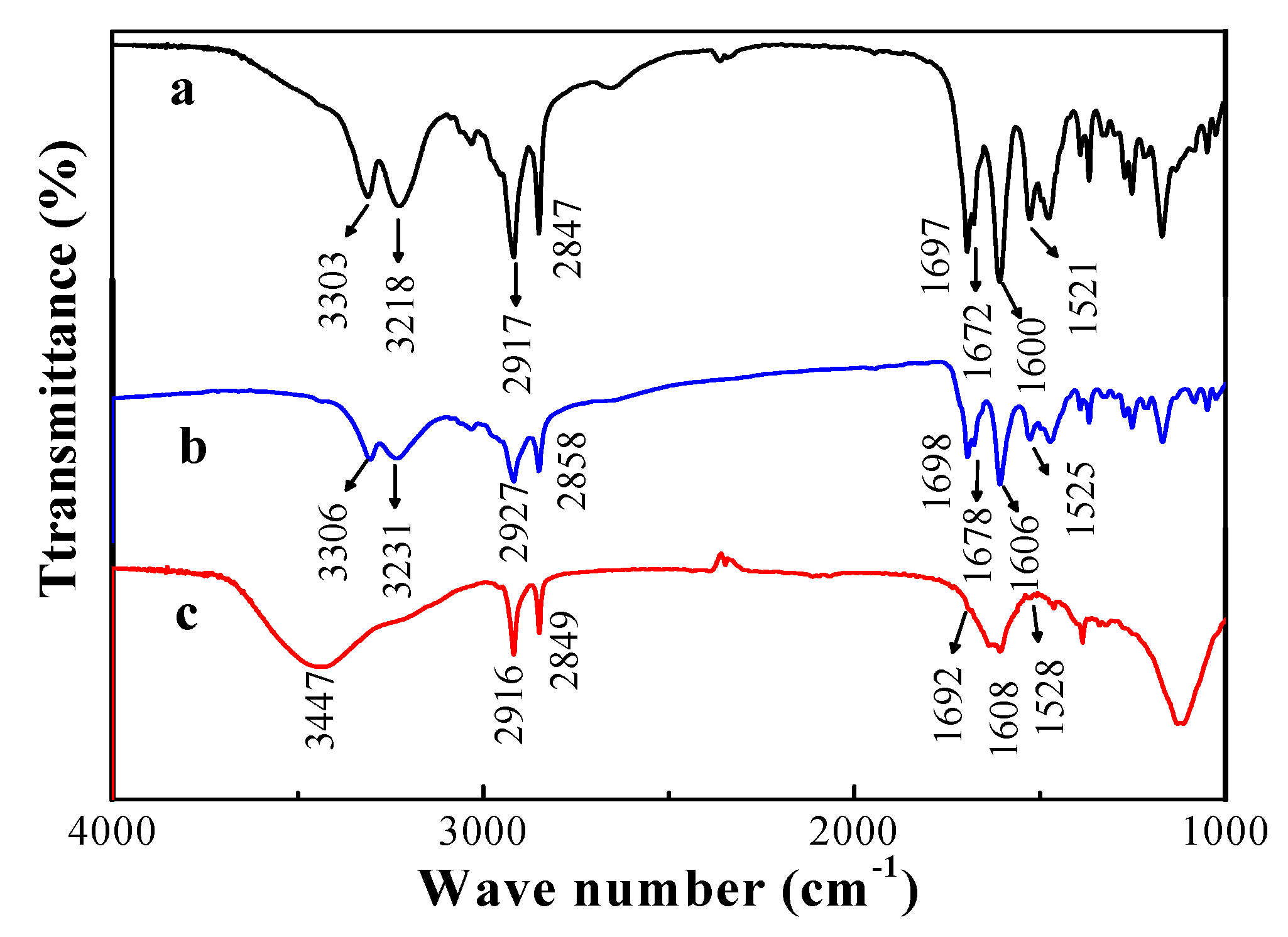
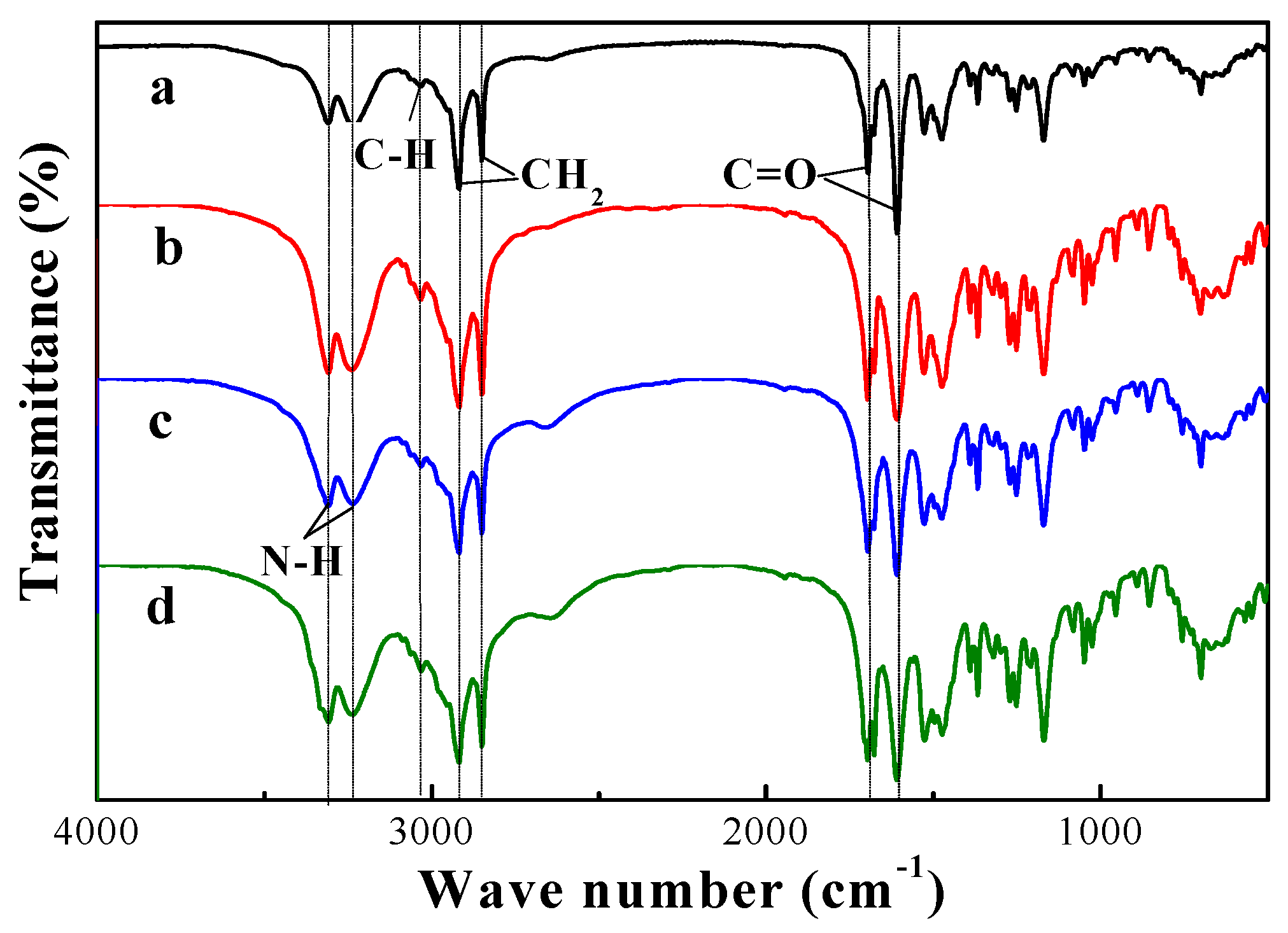
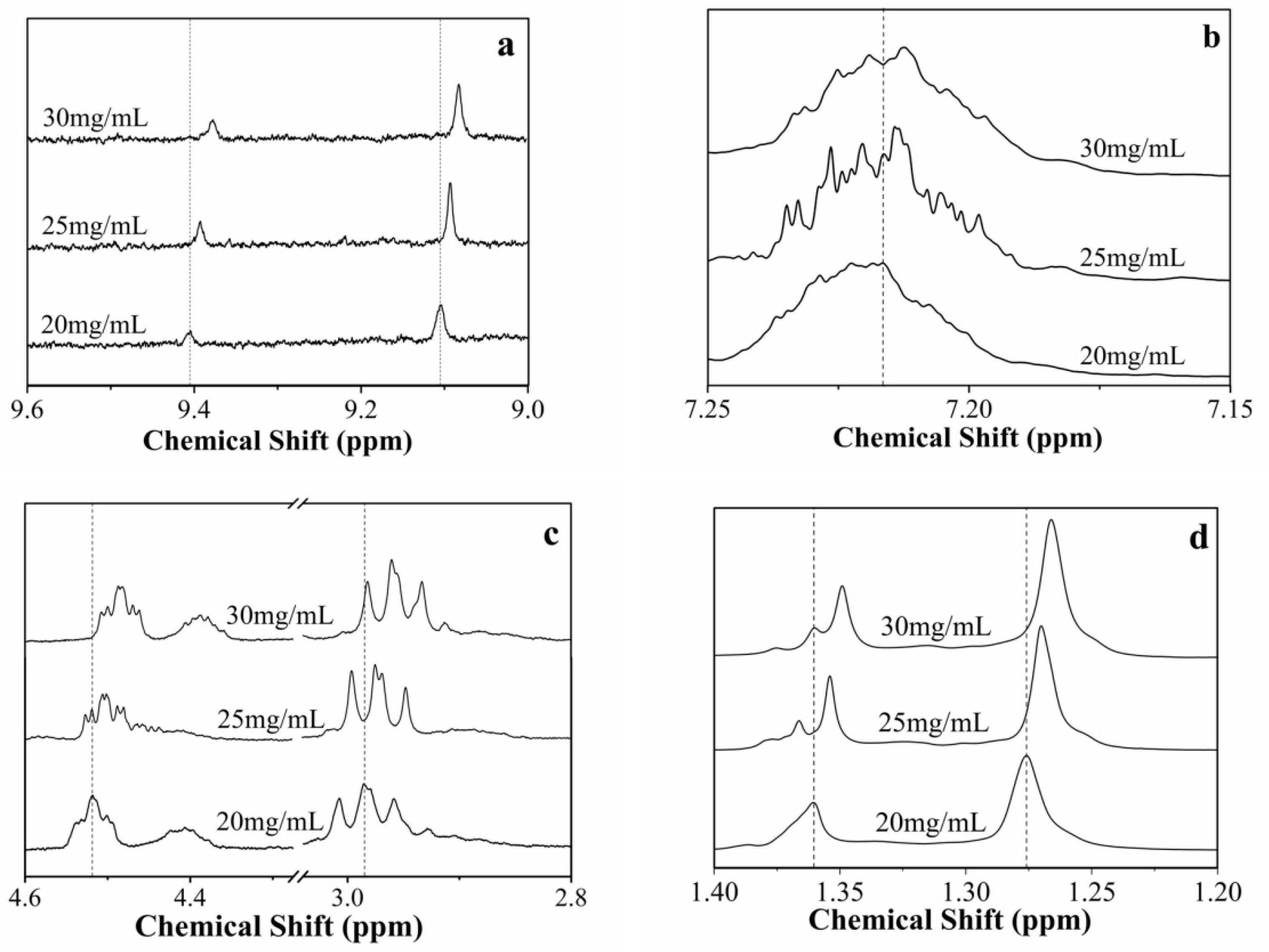
3.5. Spectral Studies of the Gels
3.6. X-ray Diffraction of the Gels
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Bielejewski, M.; Łapiński, A.; Luboradzki, R.; Tritt-Goc, J. Influence of solvent on the thermal stability and organization of self-assembling fibrillar networks in methyl-4,6-o-(p-nitrobenzylidene)-α-d-glucopyranoside gels. Tetrahedron 2011, 67, 7222–7230. [Google Scholar] [CrossRef]
- Löfman, M.; Koivukorpi, J.; Noponen, V.; Salo, H.; Sievänen, E. Bile acid alkylamide derivatives as low molecular weight organogelators: Systematic gelation studies and qualitative structural analysis of the systems. J. Colloid Interf. Sci. 2011, 360, 633–644. [Google Scholar] [CrossRef] [PubMed]
- Rogers, M.A.; Kim, J.H.J. Rheological assessment of the sol-gel transition for self-assembling low molecular weight gelators. Food Res. Int. 2011, 44, 1447–1451. [Google Scholar] [CrossRef]
- Kodama, M.; Kawasaki, Y.; Ohtaka, H. The main transition enthalpy of the gel-to-liquid crystal phases for a series of asymmetric chain length d-erythro (2S, 3R) sphingomyelins. Thermochim. Acta 2012, 532, 22–27. [Google Scholar] [CrossRef]
- Zhang, L.X.; Jiao, T.F.; Ma, K.; Xing, R.R.; Liu, Y.M.; Xiao, Y.; Zhou, J.X.; Zhang, Q.R.; Peng, Q.M. Self-assembly and drug release capacities of some amide compounds with aromatic substituent headgroups. Materials 2016, 9, 541. [Google Scholar] [CrossRef]
- Mondal, S.; Bairi, P.; Das, S.; Nandi, A.K. Phase selective organogel from an imine based gelator for use in oil spill recovery. J. Mater. Chem. A 2019, 7, 381–392. [Google Scholar] [CrossRef]
- Deval, V.; Kumar, A.; Gupta, V.; Sharma, A.; Gupta, A.; Tandon, P.; Kunimoto, K.K. Molecular structure (monomeric and dimeric) and hydrogen bonds in 5-benzyl 2-thiohydantoin studied by FT-IR and FT-Raman spectro-scopy and DFT calculations. Spectrochim. Acta A 2014, 132, 15–26. [Google Scholar] [CrossRef]
- Zheng, K.; Wang, H.Z.; Chow, H.F. Photochromic, organogelating and self-sorting behaviour of di-(dithienylethene) derivatives. Org. Biomol. Chem. 2019, 17, 3285–3291. [Google Scholar] [CrossRef]
- Ongaratto, R.; Conte, N.; D’Oca, C.R.M.; Brinkerhoff, R.C.; Ruas, C.P.; Gelesky, M.A.; D’Oca, M.G.M. In situ formation of AuNPs using fatty N-acylamino hydrazide organogelators as templates. New J. Chem. 2019, 43, 295–303. [Google Scholar] [CrossRef]
- Xing, R.R.; Liu, K.; Jiao, T.F.; Zhang, N.; Ma, K.; Zhang, R.Y.; Zou, Q.L.; Ma, G.H.; Yan, X.H. An injectable self-assembling collagen-gold hybrid hydrogel for combinatorial antitumor photothermal/photodynamic therapy. Adv. Mater. 2016, 28, 3669–3676. [Google Scholar] [CrossRef]
- Shan, Y.H.; Li, S.W.; Wang, R.; Zhu, S.Y.; Wu, F.; Fu, Y.; Zhu, L.N. Spontaneously hierarchical self-assembly of nanofibres into fluorescent spherical particles: A leap from organogels to macroscopic solidspheres. Soft Matter 2019, 15, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Vojta, D.; Vazdar, M. The study of hydrogen bonding and π⋯π interactions in phenol⋯ethynylbenzene complex by IR spectroscopy. Spectrochim. Acta A 2014, 132, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Ye, F.; Chen, S.; Tang, G.D.; Wang, X. Sonication induced morphological transformation between 3D gel network and globular structure in a two-component gelation system. Colloid Surface A 2014, 452, 165–172. [Google Scholar] [CrossRef]
- Bielejewski, M.; Puszkarska, A.; Tritt-Goc, J. Thermal properties, conductivity, and spin-lattice relaxation of gel electrolyte based on low molecular weight gelator and solution of high temperature ionic liquid. Electrochim. Acta 2015, 165, 122–129. [Google Scholar] [CrossRef]
- Boamah, P.O.; Huang, Y.; Hua, M.; Zhang, Q.; Liu, Y.; Onumah, J.; Wang, W.; Song, Y. Removal of cadmium from aqueous solution using low molecular weight chitosan derivative. Carbohyd. Polym. 2015, 122, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Flakus, H.T.; Hachula, B.; Holaj-Krzak, J.T. Long-distance inter-hydrogen bond coupling effects in the polarized IR spectra of succinic acid crystals. Spectrochim. Acta A 2015, 142, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Lu, Y.; Ma, H.; Ding, G.; Zhang, S.; Luo, Z.; Li, H.; Gao, F. Efficient enhancement of internal proton transfer of branched π-extended organic chromophore under one-photon and near-infrared two-photon irradiation. Chem. Phys. Lett. 2015, 619, 201–207. [Google Scholar] [CrossRef]
- Jiao, T.F.; Gao, F.Q.; Shen, X.H.; Zhang, Q.R.; Zhang, X.F.; Zhou, J.X.; Gao, F.M. Self-assembly and nanostructures in organogels based on a bolaform cholesteryl imide compound with conjugated aromatic spacer. Materials 2013, 6, 5893–5906. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.R.; Hu, J.S.; Liang, Y.H.; Cui, W.Q. The supramolecular organogel formed by self-assembly of ursolic acid appended with aromatic rings. Materials 2019, 12, 614. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Yuan, Y.; Tu, W.; Zhang, Y.; Zhang, M.; Qu, H. Preparation of efficient organogelators based on pyrazine-2,5-dicarboxylic acid showing room temperature mesophase. Tetrahedron 2015, 71, 3221–3230. [Google Scholar] [CrossRef]
- Ray, S.; Das, A. Studies on the π-π stacking features of imidazole units present in a series of 5-amino-1-alkylimidazole-4-carboxamides. J. Mol. Struct. 2015, 1089, 146–152. [Google Scholar] [CrossRef]
- Rekik, N.; Flakus, H.T.; Jarczyk-Jędryka, A.; Al-Agel, F.A.; Daouahi, M.; Jones, P.G.; Kusz, J.; Nowak, M. Elucidating the Davydov-coupling mechanism in hydrogen bond dimers: Experimental and theoretical investigation of the polarized IR spectra of 3-thiopheneacetic and 3-thiopheneacrylic acid crystals. J. Phys. Chem. Solids 2015, 77, 68–84. [Google Scholar] [CrossRef]
- Wu, Y.; Bai, B.; Zhang, C.; Zhang, Y.; Wang, H.; Wei, Z.; Li, M. Gelation properties of n,n-bis (3,4,5-tris(heptyloxy) benzoyl) fumarohydrazide in toluene and ethanol. Tetrahedron 2015, 71, 37–43. [Google Scholar] [CrossRef]
- Xu, L.; Hu, Y.; Liu, M.; Chen, J.; Huang, X.; Gao, W.; Wu, H. Gelation properties and glucose-sensitive behavior of phenylboronic acid based low-molecular-weight organogels. Tetrahedron 2015, 71, 2079–2088. [Google Scholar] [CrossRef]
- Tripathi, A.; Kumar, A.; Pandey, P.S. Visual chiral recognition of mandelic acid and α-amino acid derivatives by enantioselective gel formation and precipitation. Tetrahedron Lett. 2012, 53, 5745–5748. [Google Scholar] [CrossRef]
- Abdelkafi-Koubaa, Z.; Jebali, J.; Othman, H.; Morjen, M.; Aissa, I.; Zouari-Kesentini, R.; Bazaa, A.; Ellefi, A.A.; Majdoub, H.; Srairi-Abid, N.; et al. A thermoactive L-amino acid oxidase from cerastes snake venom: Purification, biochemical and molecular characterization. Toxicon 2014, 89, 32–44. [Google Scholar] [CrossRef] [PubMed]
- Das, T.; Häring, M.; Haldar, D.; David, D.D. Phenylalanine and derivatives as versatile low-molecular-weight gelators: Design, structure and tailored function. Biomater. Sci. 2018, 6, 38–59. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Tang, L.; Yu, J. Investigation on the assembled structure-property correlation of supramolecular hydrogel formed from low-molecular-weight gelator. J. Colloid Interface Sci. 2008, 319, 357–364. [Google Scholar] [CrossRef]
- Kowalczuk, J.; Jarosz, S.; Tritt-Goc, J. Characterization of low molecular-weight gelator methyl-4,6-o-(p-nitrobenzylidene)-α-d-glucopyranoside hydrogels and water diffusion in their networks. Tetrahedron 2009, 65, 9801–9806. [Google Scholar] [CrossRef]
- Bellomaria, A.; Nepravishta, R.; Mazzanti, U.; Marchetti, M.; Piccioli, P.; Paci, M. Determination of the presence of hyaluronic acid in preparations containing amino acids: The molecular weight characterization. Eur. J. pharm. Sci. 2014, 63, 199–203. [Google Scholar] [CrossRef] [PubMed]
- Puguan, J.M.C.; Yu, X.; Kim, H. Characterization of structure, physico-chemical properties and diffusion behavior of Ca-Alginate gel beads prepared by different gelation methods. J. Colloid Interface Sci. 2014, 432, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Kamlet, M.J.; Abboud, J.L.; Taft, R.W. The solvatochromic comparison method. 6. the π* scale of solvent polarities. J. Am. Chem. Soc. 1977, 99, 6027–6038. [Google Scholar] [CrossRef]
- Roy, A.; Maiti, M.; Roy, S. Effect of hydrocarbon chain and amide linkage on the interfacial and self-assembly properties of nicotinic acid amphiphiles. J. Chem. Eng. Data 2015, 60, 2209–2218. [Google Scholar] [CrossRef]
- Kamlet, M.J.; Abboud, J.L.M.; Abraham, M.H.; Taft, R.W. Linear solvation energy relationships. 23. a comprehensive collection of the solvatochromic parameters, π*, κ, and β, and some methods for simplifying the generalized solvatochromic equation. J. Org. Chem. 1983, 48, 2877–2887. [Google Scholar] [CrossRef]
- Hansen, C.M. Hansen Solubility Parameters: A User’s Handbook, 2nd ed.; CRC Press Inc.: Boca Raton, FL, USA, 2007. [Google Scholar]
- Stefanis, E.; Panayiotou, C. Prediction of Hansen solubility parameters with a new group-contribution method. Int. J. Thermophys. 2008, 29, 568–585. [Google Scholar] [CrossRef]
- Yu, Y.; Wang, S.; Jia, L.; Zhou, M.M.; Pan, Q.D.; Zhai, Y.C.; Wang, C.S. Organogels from different self-assembling novel L-proline dihydrazide derivatives: Gelation mechanism and morphology investigations. J. Sol-Gel. Sci. Technol. 2016, 78, 218–227. [Google Scholar] [CrossRef]
- Bielejewski, M.; Łapiński, A.; Kaszyńska, J.; Luboradzki, R.; Tritt-Goc, J. 1,2-o-(1-ethyl propylidene)-α-d-glucofuranose, a low molecular mass organogelator: Benzene gel formation and their thermal stabilities. Tetrahedron Lett. 2008, 49, 6685–6689. [Google Scholar] [CrossRef]
- Kolev, T. Influence of intermolecular hydrogen bonding on IR-spectroscopic properties of (R)-(−)-1-phenylglycinium hydrogen squarate monohydrate in solid-state. IR-LD, Raman spectroscopy and theoretical study. J. Mol. Struct. 2007, 846, 139–146. [Google Scholar] [CrossRef]
- Rybarczyk-Pirek, A.J.; Dubis, A.T.; Grabowski, S.J. Nawrot-Modranka, J. Intramolecular hydrogen bonds in crystals of thiophosphorylbenzopyrane derivatives X-ray and FT-IR studies. Chem. Phys. 2006, 320, 247–258. [Google Scholar] [CrossRef]
- Qu, S.; Li, M. Self assembly of linear shaped bi-dihydrazine derivative through intermolecular quadruple hydrogen bonding. Tetrahedron 2008, 64, 10890–10895. [Google Scholar] [CrossRef]


| Solvent | Compound 1 | Compound 2 | Compound 3 | Compound 4 | ||||
|---|---|---|---|---|---|---|---|---|
| CGC | State | CGC | State | CGC | State | CGC | State | |
| toluene | NF | - | 2.3 | TG | 1.0 | TG | 0.8 | TG |
| o-xylene | NF | - | 3.0 | TG | 0.5 | TG | 0.7 | TG |
| p-xylene | NF | - | 2.8 | OG | 0.5 | TG | 0.7 | TG |
| m-xylene | NF | - | 2.4 | TG | 1.1 | TG | 0.8 | TG |
| benzene | NF | - | 1.7 | TG | 0.9 | TG | 1.0 | TG |
| chlorobenzene | NF | - | 2.9 | OG | 0.7 | TG | 1.0 | TG |
| nitrobenzene | 2.5 | TG | 2.5 | TG | 1.2 | TG | 0.9 | TG |
| ethanol | 3.0 | TG | 2.9 | TG | 2.6 | OG | - | sol |
| methanol | 3.0 | OG | 2.6 | OG | 3.0 | OG | 1.6 | OG |
| n-propyl alcohol | NF | - | NF | - | 2.0 | TG | 2.8 | OG |
| n-butanol | NF | - | NF | - | 1.7 | OG | 1.5 | OG |
| iso-butanol | NF | - | 2.8 | OG | 2.6 | OG | 1.4 | OG |
| 1-pentanol | NF | - | NF | - | 1.7 | OG | NF | - |
| 1-hexanol | NF | - | NF | - | 1.8 | OG | 1.2 | OG |
| 2-propanol | NF | - | 2.7 | OG | 2.0 | OG | 1.5 | OG |
| methyl acetate | 3.0 | OG | NF | - | 1.2 | TG | 1.4 | OG |
| ethyl acetate | NF | - | 2.1 | TG | 1.7 | TG | 1.0 | OG |
| amyl acetate | NF | - | 2.2 | TG | 1.6 | TG | 1.3 | OG |
| n-butyl acetate | NF | - | 1.1 | TG | 1.5 | OG | 1.0 | OG |
| isobutyl acetate | 2.8 | TG | 1.3 | TG | 1.3 | TG | 1.0 | OG |
| n-propyl acetate | NF | - | 2.5 | TG | 1.8 | TG | 1.4 | OG |
| isopropyl acetate | NF | - | 1.4 | TG | 1.5 | TG | 2.0 | OG |
| cyclohexane | ins | - | ins | - | 0.5 | TG | 0.2 | TG |
| n-octane | ins | - | ins | - | - | ins | - | ins |
| n-hexane | ins | - | ins | - | - | ins | - | ins |
| tetrachloromethane | 3.0 | TG | NF | - | - | ins | 3.0 | TG |
| Solvent | π* | β | α | ΔHg (kJ·mol−1) |
|---|---|---|---|---|
| benzene | 0.59 | 0.10 | 0 | 198.4 |
| m-xylene | 0.47 | 0.10 | 0 | 171.8 |
| chlorobenzene | 0.71 | 0.07 | 0 | 140.3 |
| toluene | 0.54 | 0.11 | 0 | 132.3 |
| o-xylene | 0.43 | 0.12 | 0 | 121.9 |
| n-butanol | 0.47 | 0.88 | 0.79 | 70.25 |
| nitrobenzene | 1.01 | 0.39 | 0 | 69.8 |
| methanol | 0.60 | 0.62 | 0.93 | 37.33 |
| n-butyl acetate | 0.46 | NF | 0 | 93.70 |
| n-propyl alcohol | 0.52 | NF | 0.78 | 63.93 |
| isobutyl acetate | NF | NF | NF | 57.87 |
| amyl acetate | NF | NF | NF | 148.32 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, Y.; Chu, N.; Pan, Q.; Zhou, M.; Qiao, S.; Zhao, Y.; Wang, C.; Li, X. Solvent Effects on Gelation Behavior of the Organogelator Based on L-Phenylalanine Dihydrazide Derivatives. Materials 2019, 12, 1890. https://doi.org/10.3390/ma12121890
Yu Y, Chu N, Pan Q, Zhou M, Qiao S, Zhao Y, Wang C, Li X. Solvent Effects on Gelation Behavior of the Organogelator Based on L-Phenylalanine Dihydrazide Derivatives. Materials. 2019; 12(12):1890. https://doi.org/10.3390/ma12121890
Chicago/Turabian StyleYu, Yang, Ning Chu, Qiaode Pan, Miaomiao Zhou, Sheng Qiao, Yanan Zhao, Chuansheng Wang, and Xiangyun Li. 2019. "Solvent Effects on Gelation Behavior of the Organogelator Based on L-Phenylalanine Dihydrazide Derivatives" Materials 12, no. 12: 1890. https://doi.org/10.3390/ma12121890
APA StyleYu, Y., Chu, N., Pan, Q., Zhou, M., Qiao, S., Zhao, Y., Wang, C., & Li, X. (2019). Solvent Effects on Gelation Behavior of the Organogelator Based on L-Phenylalanine Dihydrazide Derivatives. Materials, 12(12), 1890. https://doi.org/10.3390/ma12121890