Environmentally Sustainable and Ecosafe Polysaccharide-Based Materials for Water Nano-Treatment: An Eco-Design Study
Abstract
1. Introduction
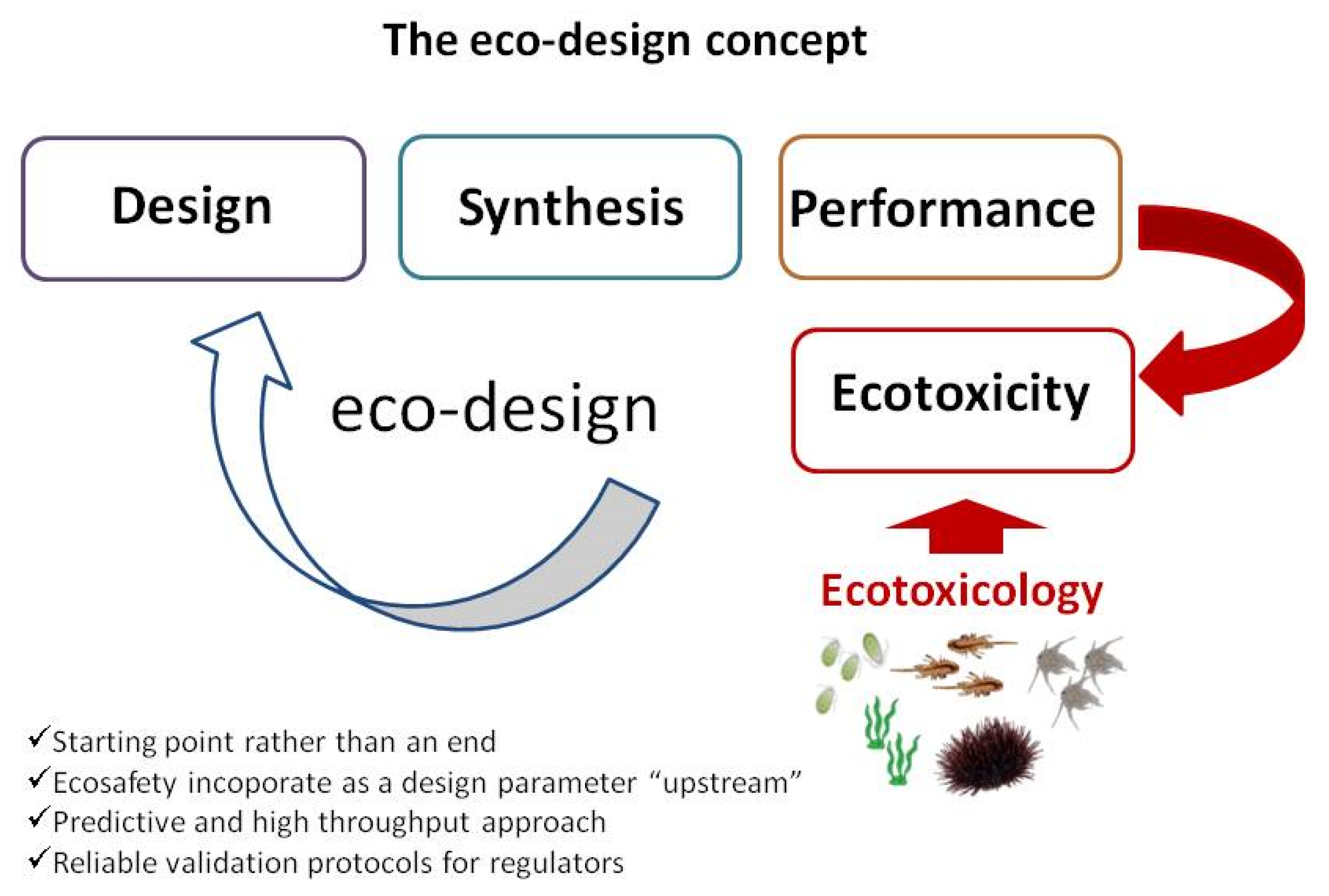
2. Eco-Design of ENMs for Environmental Remediation
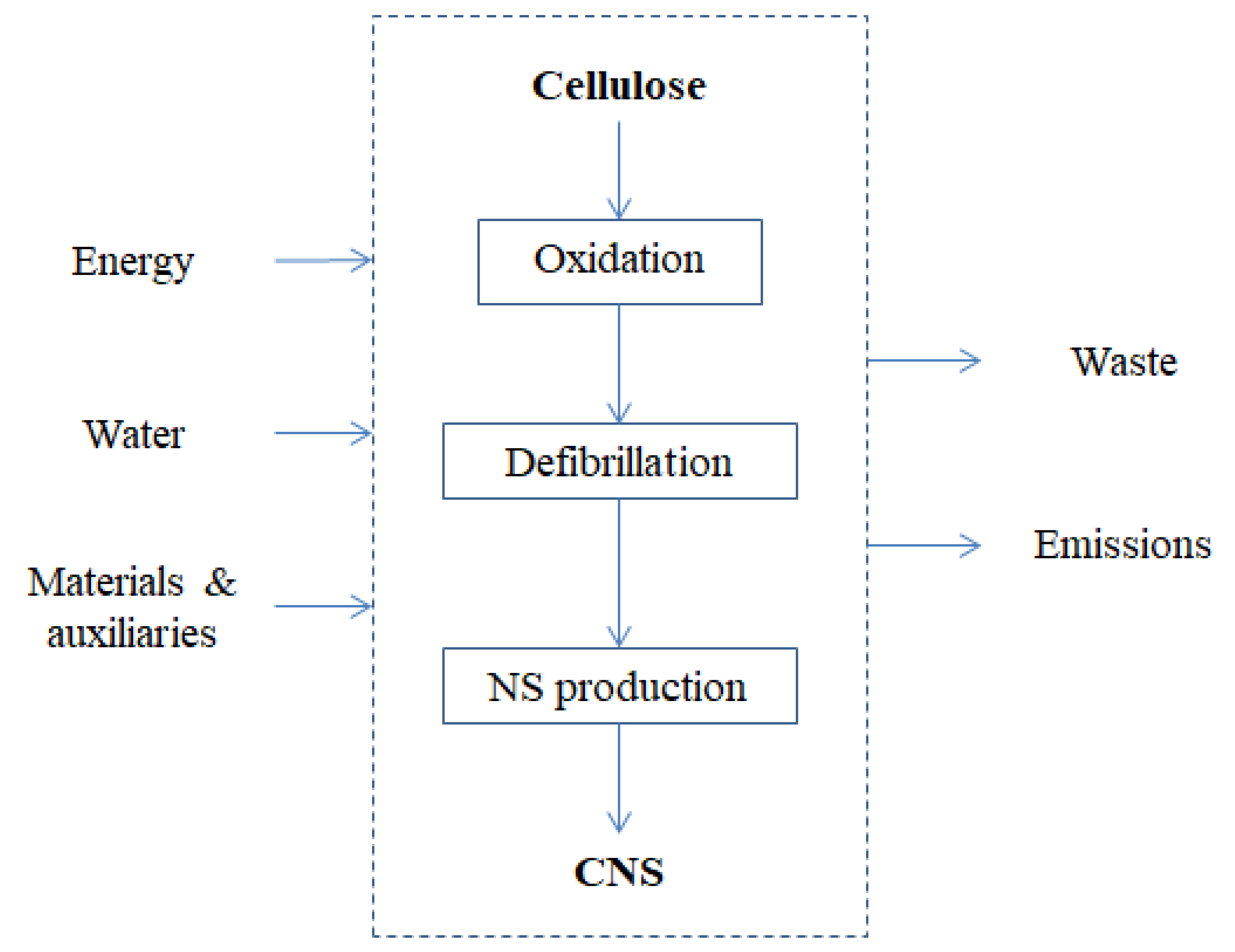
3. Life-Cycle Assessment of ENMs
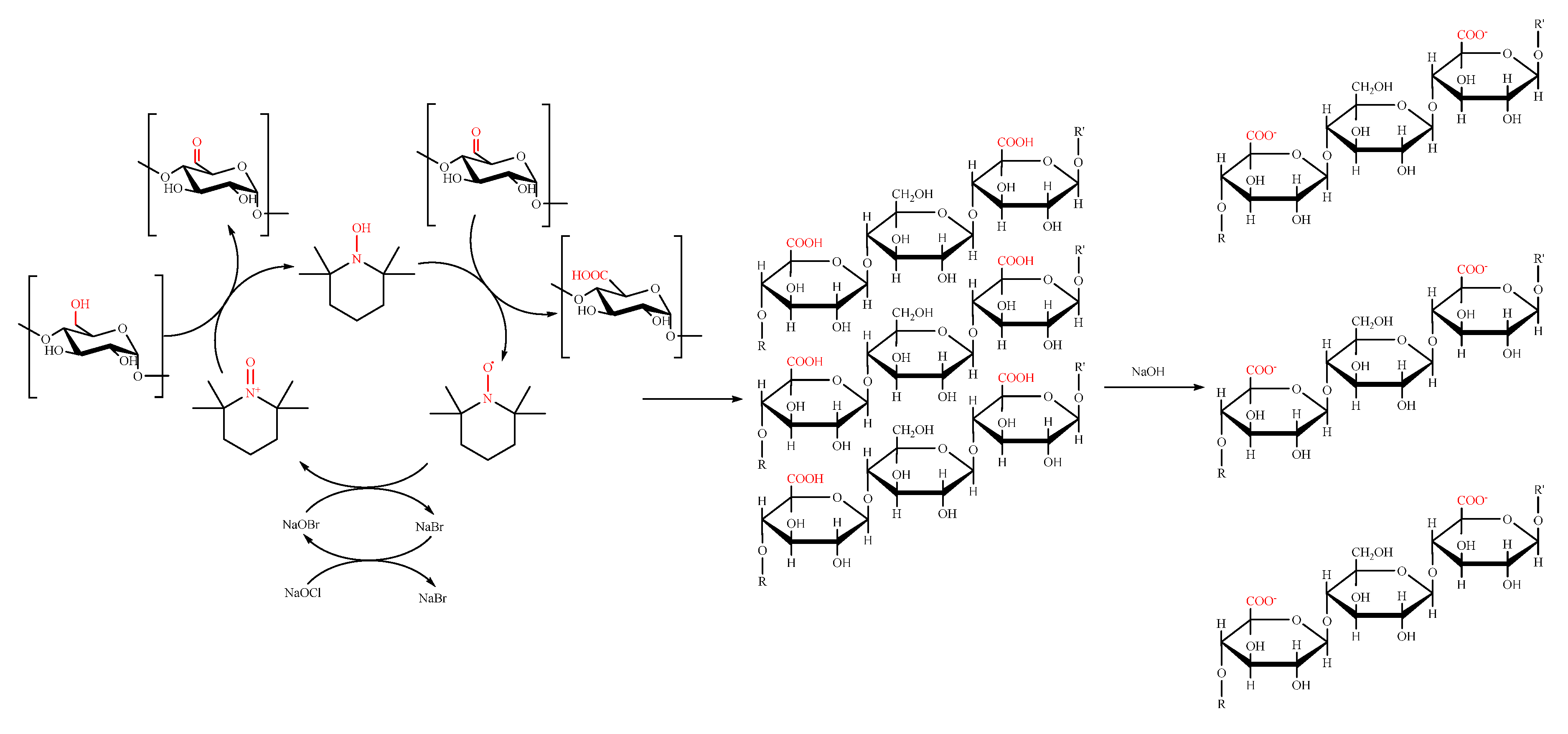
4. Cellulose- and Starch-Based Nanostructured Materials
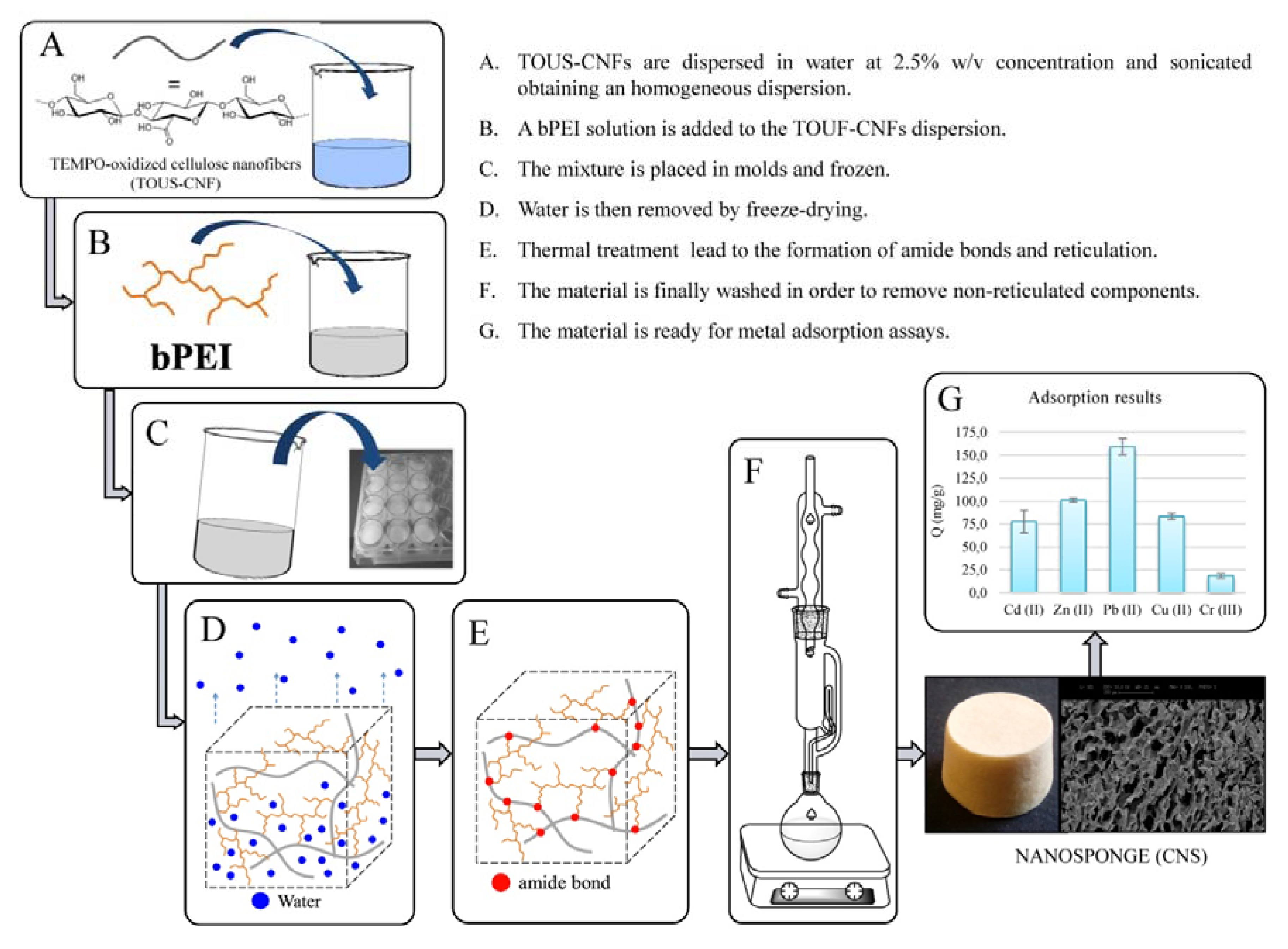
5. The NanoBonD Action
6. Conclusions and Outlook
Author Contributions
Funding
Conflicts of Interest
References
- Vaseashta, A.; Vaclavikova, M.; Vaseashta, S.; Gallios, G.; Roy, P.; Pummakarnchana, O. Nanostructures in environmental pollution detection, monitoring, and remediation. Sci. Technol. Adv. Mater. 2007, 8, 47–59. [Google Scholar] [CrossRef]
- Shevah, Y.; Waldman, M. In-situ and on-site treatment of groundwater (Technical Report). Pure Appl. Chem. 1995, 67, 1549–1561. [Google Scholar] [CrossRef]
- Otto, M.; Floyd, M.; Bajpai, S. Nanotechnology for site remediation. Remediat. J. 2008, 19, 99–108. [Google Scholar] [CrossRef]
- U.S. Environmental Protection Agency. Superfund: National Priorities List (NPL). Available online: https://www.epa.gov/superfund/superfund-national-priorities-list-npl (accessed on 23 May 2018).
- Holland, K.S. A Framework for Sustainable Remediation. Environ. Sci. Technol. 2011, 45, 7116–7117. [Google Scholar] [CrossRef] [PubMed]
- Karn, B.; Kuiken, T.; Otto, M. Nanotechnology and in Situ Remediation: A Review of the Benefits and Potential Risks. Environ. Health Perspect. 2009, 117, 1813–1831. [Google Scholar] [CrossRef] [PubMed]
- Grieger, K.D.; Hjorth, R.; Rice, J.; Kumar, N.; Bang, J. Nano-Remediation: Tiny Particles Cleaning up Big Environmental Problems; IUCN: Gland, Switzerland, 2015. [Google Scholar]
- Sánchez, A.; Recillas, S.; Font, X.; Casals, E.; González, E.; Puntes, V. Ecotoxicity of, and remediation with, engineered inorganic nanoparticles in the environment. TrAC Trends Anal. Chem. 2011, 30, 507–516. [Google Scholar] [CrossRef]
- Map of Contaminated Sites Utilizing Nano-Remediation (Oil, Manufacturing, Military, Private Properties, Residences). Available online: http://www.nanotechproject.org/inventories/remediation_map/ (accessed on 23 May 2018).
- Progress in Management of Contaminated Sites. Available online: https://www.eea.europa.eu/data-and-maps/indicators/progress-in-management-of-contaminated-sites/progress-in-management-of-contaminated-1 (accessed on 23 May 2018).
- Rickerby, D.; Morrison, M. Report from the Workshop on Nanotechnologies for Environmental Remediation; Joint Research Centre (JRC): Ispra, Italy, 2007. [Google Scholar]
- A little knowledge. Nat. Nanotechnol. 2007, 2, 731. [CrossRef]
- Godwin, H.; Nameth, C.; Avery, D.; Bergeson, L.L.; Bernard, D.; Beryt, E.; Boyes, W.; Brown, S.; Clippinger, A.J.; Cohen, Y.; et al. Nanomaterial Categorization for Assessing Risk Potential to Facilitate Regulatory Decision-Making. ACS Nano 2015, 9, 3409–3417. [Google Scholar] [CrossRef] [PubMed]
- Grieger, K.; Wickson, F.; Andersen, H.B.; Renn, O. Improving Risk Governance of Emerging Technologies through Public Engagement: The Neglected Case of Nano- Remediation? Int. J. Emerg. Technol. Soc. 2012, 10, 61–78. [Google Scholar]
- Patil, S.S.; Shedbalkar, U.U.; Truskewycz, A.; Chopade, B.A.; Ball, A.S. Nanoparticles for environmental clean-up: A review of potential risks and emerging solutions. Environ. Technol. Innov. 2016, 5, 10–21. [Google Scholar] [CrossRef]
- Matranga, V.; Corsi, I. Toxic effects of engineered nanoparticles in the marine environment: Model organisms and molecular approaches. Mar. Environ. Res. 2012, 76, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Scott-Fordsmand, J.; Peijnenburg, W.; Semenzin, E.; Nowack, B.; Hunt, N.; Hristozov, D.; Marcomini, A.; Irfan, M.; Jiménez, A.S.; Landsiedel, R.; et al. Environmental Risk Assessment Strategy for Nanomaterials. Int. J. Environ. Res. Public Health 2017, 14, 1251. [Google Scholar] [CrossRef] [PubMed]
- Kraas, M.; Schlich, K.; Knopf, B.; Wege, F.; Kägi, R.; Terytze, K.; Hund-Rinke, K. Long-term effects of sulfidized silver nanoparticles in sewage sludge on soil microflora. Environ. Toxicol. Chem. 2017, 36, 3305–3313. [Google Scholar] [CrossRef] [PubMed]
- Kaegi, R.; Voegelin, A.; Sinnet, B.; Zuleeg, S.; Siegrist, H.; Burkhardt, M. Transformation of AgCl nanoparticles in a sewer system—A field study. Sci. Total Environ. 2015, 535, 20–27. [Google Scholar] [CrossRef] [PubMed]
- Thalmann, B.; Voegelin, A.; Morgenroth, E.; Kaegi, R. Effect of humic acid on the kinetics of silver nanoparticle sulfidation. Environ. Sci. Nano 2016, 3, 203–212. [Google Scholar] [CrossRef]
- Gogos, A.; Thalmann, B.; Voegelin, A.; Kaegi, R. Sulfidation kinetics of copper oxide nanoparticles. Environ. Sci. Nano 2017, 4, 1733–1741. [Google Scholar] [CrossRef]
- Keller, A.A.; Wang, H.; Zhou, D.; Lenihan, H.S.; Cherr, G.; Cardinale, B.J.; Miller, R.; Ji, Z. Stability and Aggregation of Metal Oxide Nanoparticles in Natural Aqueous Matrices. Environ. Sci. Technol. 2010, 44, 1962–1967. [Google Scholar] [CrossRef] [PubMed]
- Brunelli, A.; Pojana, G.; Callegaro, S.; Marcomini, A. Agglomeration and sedimentation of titanium dioxide nanoparticles (n-TiO2) in synthetic and real waters. J. Nanopart. Res. 2013, 15. [Google Scholar] [CrossRef]
- Erhayem, M.; Sohn, M. Stability studies for titanium dioxide nanoparticles upon adsorption of Suwannee River humic and fulvic acids and natural organic matter. Sci. Total Environ. 2014, 468–469, 249–257. [Google Scholar] [CrossRef] [PubMed]
- Adeleye, A.S.; Conway, J.R.; Perez, T.; Rutten, P.; Keller, A.A. Influence of Extracellular Polymeric Substances on the Long-Term Fate, Dissolution, and Speciation of Copper-Based Nanoparticles. Environ. Sci. Technol. 2014, 48, 12561–12568. [Google Scholar] [CrossRef] [PubMed]
- Aiken, G.R.; Hsu-Kim, H.; Ryan, J.N. Influence of Dissolved Organic Matter on the Environmental Fate of Metals, Nanoparticles, and Colloids. Environ. Sci. Technol. 2011, 45, 3196–3201. [Google Scholar] [CrossRef] [PubMed]
- Schaumann, G.E.; Philippe, A.; Bundschuh, M.; Metreveli, G.; Klitzke, S.; Rakcheev, D.; Grün, A.; Kumahor, S.K.; Kühn, M.; Baumann, T.; et al. Understanding the fate and biological effects of Ag- and TiO2-nanoparticles in the environment: The quest for advanced analytics and interdisciplinary concepts. Sci. Total Environ. 2015, 535, 3–19. [Google Scholar] [CrossRef] [PubMed]
- Garner, K.L.; Keller, A.A. Emerging patterns for engineered nanomaterials in the environment: A review of fate and toxicity studies. J. Nanopart. Res. 2014, 16. [Google Scholar] [CrossRef]
- Yavuz, C.T.; Mayo, J.T.; Yu, W.W.; Prakash, A.; Falkner, J.C.; Yean, S.; Cong, L.; Shipley, H.J.; Kan, A.; Tomson, M.; et al. Low-Field Magnetic Separation of Monodisperse Fe3O4 Nanocrystals. Science 2006, 314, 964–967. [Google Scholar] [CrossRef] [PubMed]
- Nowack, B.; Bucheli, T.D. Occurrence, behavior and effects of nanoparticles in the environment. Environ. Pollut. 2007, 150, 5–22. [Google Scholar] [CrossRef] [PubMed]
- Nowack, B.; Ranville, J.F.; Diamond, S.; Gallego-Urrea, J.A.; Metcalfe, C.; Rose, J.; Horne, N.; Koelmans, A.A.; Klaine, S.J. Potential scenarios for nanomaterial release and subsequent alteration in the environment. Environ. Toxicol. Chem. 2012, 31, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Corsi, I.; Cherr, G.N.; Lenihan, H.S.; Labille, J.; Hassellov, M.; Canesi, L.; Dondero, F.; Frenzilli, G.; Hristozov, D.; Puntes, V.; et al. Common Strategies and Technologies for the Ecosafety Assessment and Design of Nanomaterials Entering the Marine Environment. ACS Nano 2014, 8, 9694–9709. [Google Scholar] [CrossRef] [PubMed]
- Lowry, G.V.; Gregory, K.B.; Apte, S.C.; Lead, J.R. Transformations of Nanomaterials in the Environment. Environ. Sci. Technol. 2012, 46, 6893–6899. [Google Scholar] [CrossRef] [PubMed]
- Petersen, E.J.; Flores-Cervantes, D.X.; Bucheli, T.D.; Elliott, L.C.C.; Fagan, J.A.; Gogos, A.; Hanna, S.; Kägi, R.; Mansfield, E.; Bustos, A.R.M.; et al. Quantification of Carbon Nanotubes in Environmental Matrices: Current Capabilities, Case Studies, and Future Prospects. Environ. Sci. Technol. 2016, 50, 4587–4605. [Google Scholar] [CrossRef] [PubMed]
- Trujillo-Reyes, J.; Peralta-Videa, J.R.; Gardea-Torresdey, J.L. Supported and unsupported nanomaterials for water and soil remediation: Are they a useful solution for worldwide pollution? J. Hazard. Mater. 2014, 280, 487–503. [Google Scholar] [CrossRef] [PubMed]
- Canesi, L.; Frenzilli, G.; Balbi, T.; Bernardeschi, M.; Ciacci, C.; Corsolini, S.; Della Torre, C.; Fabbri, R.; Faleri, C.; Focardi, S.; et al. Interactive effects of n-TiO2 and 2,3,7,8-TCDD on the marine bivalve Mytilus galloprovincialis. Aquat. Toxicol. 2014, 153, 53–65. [Google Scholar] [CrossRef] [PubMed]
- Della Torre, C.; Balbi, T.; Grassi, G.; Frenzilli, G.; Bernardeschi, M.; Smerilli, A.; Guidi, P.; Canesi, L.; Nigro, M.; Monaci, F.; et al. Titanium dioxide nanoparticles modulate the toxicological response to cadmium in the gills of Mytilus galloprovincialis. J. Hazard. Mater. 2015, 297, 92–100. [Google Scholar] [CrossRef] [PubMed]
- Della Torre, C.; Buonocore, F.; Frenzilli, G.; Corsolini, S.; Brunelli, A.; Guidi, P.; Kocan, A.; Mariottini, M.; Mottola, F.; Nigro, M.; et al. Influence of titanium dioxide nanoparticles on 2,3,7,8-tetrachlorodibenzo-p-dioxin bioconcentration and toxicity in the marine fish European sea bass (Dicentrarchus labrax). Environ. Pollut. 2015, 196, 185–193. [Google Scholar] [CrossRef] [PubMed]
- Nowack, B.; Baalousha, M.; Bornhöft, N.; Chaudhry, Q.; Cornelis, G.; Cotterill, J.; Gondikas, A.; Hassellöv, M.; Lead, J.; Mitrano, D.M.; et al. Progress towards the validation of modeled environmental concentrations of engineered nanomaterials by analytical measurements. Environ. Sci. Nano 2015, 2, 421–428. [Google Scholar] [CrossRef]
- Bour, A.; Mouchet, F.; Silvestre, J.; Gauthier, L.; Pinelli, E. Environmentally relevant approaches to assess nanoparticles ecotoxicity: A review. J. Hazard. Mater. 2015, 283, 764–777. [Google Scholar] [CrossRef] [PubMed]
- Garner, K.L.; Suh, S.; Keller, A.A. Assessing the Risk of Engineered Nanomaterials in the Environment: Development and Application of the nanoFate Model. Environ. Sci. Technol. 2017, 51, 5541–5551. [Google Scholar] [CrossRef] [PubMed]
- Gottschalk, F.; Sun, T.; Nowack, B. Environmental concentrations of engineered nanomaterials: Review of modeling and analytical studies. Environ. Pollut. 2013, 181, 287–300. [Google Scholar] [CrossRef] [PubMed]
- Holden, P.A.; Gardea-Torresdey, J.L.; Klaessig, F.; Turco, R.F.; Mortimer, M.; Hund-Rinke, K.; Cohen Hubal, E.A.; Avery, D.; Barceló, D.; Behra, R.; et al. Considerations of Environmentally Relevant Test Conditions for Improved Evaluation of Ecological Hazards of Engineered Nanomaterials. Environ. Sci. Technol. 2016, 50, 6124–6145. [Google Scholar] [CrossRef] [PubMed]
- Elliott, J. Toward achieving harmonization in a nano-cytotoxicity assay measurement through an interlaboratory comparison study. ALTEX 2017, 34, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Selck, H.; Handy, R.D.; Fernandes, T.F.; Klaine, S.J.; Petersen, E.J. Nanomaterials in the aquatic environment: A European Union–United States perspective on the status of ecotoxicity testing, research priorities, and challenges ahead. Environ. Toxicol. Chem. 2016, 35, 1055–1067. [Google Scholar] [CrossRef] [PubMed]
- Corsi, I.; Winther-Nielsen, M.; Sethi, R.; Punta, C.; Della Torre, C.; Libralato, G.; Lofrano, G.; Sabatini, L.; Aiello, M.; Fiordi, L.; et al. Ecofriendly nanotechnologies and nanomaterials for environmental applications: Key issue and consensus recommendations for sustainable and ecosafe nanoremediation. Ecotoxicol. Environ. Saf. 2018, 154, 237–244. [Google Scholar] [CrossRef] [PubMed]
- Petersen, E.J.; Diamond, S.A.; Kennedy, A.J.; Goss, G.G.; Ho, K.; Lead, J.; Hanna, S.K.; Hartmann, N.B.; Hund-Rinke, K.; Mader, B.; et al. Adapting OECD Aquatic Toxicity Tests for Use with Manufactured Nanomaterials: Key Issues and Consensus Recommendations. Environ. Sci. Technol. 2015, 49, 9532–9547. [Google Scholar] [CrossRef] [PubMed]
- Hjorth, R.; Coutris, C.; Nguyen, N.H.A.; Sevcu, A.; Gallego-Urrea, J.A.; Baun, A.; Joner, E.J. Ecotoxicity testing and environmental risk assessment of iron nanomaterials for sub-surface remediation–Recommendations from the FP7 project NanoRem. Chemosphere 2017, 182, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, N.H.A.; Von Moos, N.R.; Slaveykova, V.I.; Mackenzie, K.; Meckenstock, R.U.; Thűmmler, S.; Bosch, J.; Ševců, A. Biological effects of four iron-containing nanoremediation materials on the green alga Chlamydomonas sp. Ecotoxicol. Environ. Saf. 2018, 154, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Müller, N.C.; Nowack, B. Nano Zero Valent Iron—The Solution for Water and Soil Remediation? Observatory NANO Focus Report; EMPA: Dübendorf, Switzerland, 2010. [Google Scholar]
- Keller, A.A.; Garner, K.; Miller, R.J.; Lenihan, H.S. Toxicity of Nano-Zero Valent Iron to Freshwater and Marine Organisms. PLoS ONE 2012, 7, e43983. [Google Scholar] [CrossRef] [PubMed]
- Ševců, A.; El-Temsah, Y.S.; Joner, E.J.; Černík, M. Oxidative Stress Induced in Microorganisms by Zero-valent Iron Nanoparticles. Microbes Environ. 2011, 26, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Kim, J.Y.; Lee, W.I.; Nelson, K.L.; Yoon, J.; Sedlak, D.L. Bactericidal Effect of Zero-Valent Iron Nanoparticles on Escherichia coli. Environ. Sci. Technol. 2008, 42, 4927–4933. [Google Scholar] [CrossRef] [PubMed]
- Johnston, H.J.; Hutchison, G.R.; Christensen, F.M.; Peters, S.; Hankin, S.; Stone, V. Identification of the mechanisms that drive the toxicity of TiO2 particulates: The contribution of physicochemical characteristics. Part. Fibre Toxicol. 2009, 6, 33. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Li, G.; Yu, J.C. Inorganic materials for photocatalytic water disinfection. J. Mater. Chem. 2010, 20, 4529–4536. [Google Scholar] [CrossRef]
- Miller, R.J.; Bennett, S.; Keller, A.A.; Pease, S.; Lenihan, H.S. TiO2 Nanoparticles Are Phototoxic to Marine Phytoplankton. PLoS ONE 2012, 7, e30321. [Google Scholar] [CrossRef] [PubMed]
- Mathur, A.; Parashar, A.; Chandrasekaran, N.; Mukherjee, A. Nano-TiO2 enhances biofilm formation in a bacterial isolate from activated sludge of a waste water treatment plant. Int. Biodeterior. Biodegrad. 2017, 116, 17–25. [Google Scholar] [CrossRef]
- Callaghan, N.I.; MacCormack, T.J. Ecophysiological perspectives on engineered nanomaterial toxicity in fish and crustaceans. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2017, 193, 30–41. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Liu, Y.; Singhal, N.; Wang, L.; Gao, W. Comparative photocatalytic degradation of estrone in water by ZnO and TiO2 under artificial UVA and solar irradiation. Chem. Eng. J. 2012, 213, 150–162. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, C.; Hu, Z. Impact of metallic and metal oxide nanoparticles on wastewater treatment and anaerobic digestion. Envion. Sci. Process. Impacts 2013, 15, 39–48. [Google Scholar] [CrossRef]
- Chang, Y.-N.; Zhang, M.; Xia, L.; Zhang, J.; Xing, G. The Toxic Effects and Mechanisms of CuO and ZnO Nanoparticles. Materials 2012, 5, 2850–2871. [Google Scholar] [CrossRef]
- Auffan, M.; Rose, J.; Wiesner, M.R.; Bottero, J.-Y. Chemical stability of metallic nanoparticles: A parameter controlling their potential cellular toxicity in vitro. Environ. Pollut. 2009, 157, 1127–1133. [Google Scholar] [CrossRef] [PubMed]
- Franklin, N.M.; Rogers, N.J.; Apte, S.C.; Batley, G.E.; Gadd, G.E.; Casey, P.S. Comparative Toxicity of Nanoparticulate ZnO, Bulk ZnO, and ZnCl2 to a Freshwater Microalga (Pseudokirchneriella subcapitata): The Importance of Particle Solubility. Environ. Sci. Technol. 2007, 41, 8484–8490. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.J.; Lenihan, H.S.; Muller, E.B.; Tseng, N.; Hanna, S.K.; Keller, A.A. Impacts of Metal Oxide Nanoparticles on Marine Phytoplankton. Environ. Sci. Technol. 2010, 44, 7329–7334. [Google Scholar] [CrossRef] [PubMed]
- Aruoja, V.; Dubourguier, H.-C.; Kasemets, K.; Kahru, A. Toxicity of nanoparticles of CuO, ZnO and TiO2 to microalgae Pseudokirchneriella subcapitata. Sci. Total Environ. 2009, 407, 1461–1468. [Google Scholar] [CrossRef] [PubMed]
- Mortimer, M.; Kasemets, K.; Kahru, A. Toxicity of ZnO and CuO nanoparticles to ciliated protozoa Tetrahymena thermophila. Toxicology 2010, 269, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Volder, M.F.L.D.; Tawfick, S.H.; Baughman, R.H.; Hart, A.J. Carbon Nanotubes: Present and Future Commercial Applications. Science 2013, 339, 535–539. [Google Scholar] [CrossRef] [PubMed]
- Mitrano, D.M.; Motellier, S.; Clavaguera, S.; Nowack, B. Review of nanomaterial aging and transformations through the life cycle of nano-enhanced products. Environ. Int. 2015, 77, 132–147. [Google Scholar] [CrossRef] [PubMed]
- Hanna, S.K.; Miller, R.J.; Lenihan, H.S. Deposition of carbon nanotubes by a marine suspension feeder revealed by chemical and isotopic tracers. J. Hazard. Mater. 2014, 279, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Boncel, S.; Kyzioł-Komosińska, J.; Krzyżewska, I.; Czupioł, J. Interactions of carbon nanotubes with aqueous/aquatic media containing organic/inorganic contaminants and selected organisms of aquatic ecosystems—A review. Chemosphere 2015, 136, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Lo, I.M.C.; Hu, L.; Voon, C.P.; Lim, B.L.; Versaw, W.K. Environmental Risks of Nano Zerovalent Iron for Arsenate Remediation: Impacts on Cytosolic Levels of Inorganic Phosphate and MgATP2– in Arabidopsis thaliana. Environ. Sci. Technol. 2018, 52, 4385–4392. [Google Scholar] [CrossRef] [PubMed]
- Qualhato, G.; Rocha, T.L.; de Oliveira Lima, E.C.; e Silva, D.M.; Cardoso, J.R.; Koppe Grisolia, C.; de Sabóia-Morais, S.M.T. Genotoxic and mutagenic assessment of iron oxide (maghemite-γ-Fe2O3) nanoparticle in the guppy Poecilia reticulata. Chemosphere 2017, 183, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Hjorth, R.; van Hove, L.; Wickson, F. What can nanosafety learn from drug development? The feasibility of “safety by design”. Nanotoxicology 2017, 11, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Schiwy, A.; Maes, H.M.; Koske, D.; Flecken, M.; Schmidt, K.R.; Schell, H.; Tiehm, A.; Kamptner, A.; Thümmler, S.; Stanjek, H.; et al. The ecotoxic potential of a new zero-valent iron nanomaterial, designed for the elimination of halogenated pollutants, and its effect on reductive dechlorinating microbial communities. Environ. Pollut. 2016, 216, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, I.; Mukherjee, A.; Mukherjee, A. In planta genotoxicity of nZVI: Influence of colloidal stability on uptake, DNA damage, oxidative stress and cell death. Mutagenesis 2017, 32, 371–387. [Google Scholar] [CrossRef] [PubMed]
- Nogueira, V.; Lopes, I.; Rocha-Santos, T.A.P.; Rasteiro, M.G.; Abrantes, N.; Gonçalves, F.; Soares, A.M.V.M.; Duarte, A.C.; Pereira, R. Assessing the ecotoxicity of metal nano-oxides with potential for wastewater treatment. Environ. Sci. Pollut. Res. 2015, 22, 13212–13224. [Google Scholar] [CrossRef] [PubMed]
- Naveed Ul Haq, A.; Nadhman, A.; Ullah, I.; Mustafa, G.; Yasinzai, M.; Khan, I. Synthesis Approaches of Zinc Oxide Nanoparticles: The Dilemma of Ecotoxicity. Available online: https://www.hindawi.com/journals/jnm/2017/8510342/ (accessed on 23 April 2018).
- Mottier, A.; Mouchet, F.; Pinelli, É.; Gauthier, L.; Flahaut, E. Environmental impact of engineered carbon nanoparticles: From releases to effects on the aquatic biota. Curr. Opin. Biotechnol. 2017, 46, 1–6. [Google Scholar] [CrossRef] [PubMed]
- ISO 14040:2006—Environmental Management—Life Cycle Assessment—Principles and Framework. Available online: https://www.iso.org/standard/37456.html (accessed on 23 May 2018).
- ISO 14044:2006—Environmental Management—Life Cycle Assessment—Requirements and Guidelines. Available online: https://www.iso.org/standard/38498.html (accessed on 23 May 2018).
- Gavankar, S.; Suh, S.; Keller, A.F. Life cycle assessment at nanoscale: Review and recommendations. Int. J. Life Cycle Assess. 2012, 17, 295–303. [Google Scholar] [CrossRef]
- Grieger, K.D.; Laurent, A.; Miseljic, M.; Christensen, F.; Baun, A.; Olsen, S.I. Analysis of current research addressing complementary use of life-cycle assessment and risk assessment for engineered nanomaterials: have lessons been learned from previous experience with chemicals? J. Nanopart. Res. 2012, 14, 958. [Google Scholar] [CrossRef]
- Bauer, C.; Buchgeister, J.; Hischier, R.; Poganietz, W.R.; Schebek, L.; Warsen, J. Towards a framework for life cycle thinking in the assessment of nanotechnology. J. Clean. Prod. 2008, 16, 910–926. [Google Scholar] [CrossRef]
- European Commission. COM/2004/338—Towards a European Strategy for Nanotechnology; European Commission: Brussels, Belgium, 2004. [Google Scholar]
- European Commission. COM/2005/243—Nanosciences and Nanotechnologies: An Action Plan for Europe 2005–2009; European Commission: Brussels, Belgium, 2005. [Google Scholar]
- Hischier, R.; Walser, T. Life cycle assessment of engineered nanomaterials: State of the art and strategies to overcome existing gaps. Sci. Total Environ. 2012, 425, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Miseljic, M.; Olsen, S.I. Life-cycle assessment of engineered nanomaterials: A literature review of assessment status. J. Nanopart. Res. 2014, 16, 2427. [Google Scholar] [CrossRef]
- Upadhyayula, V.K.K.; Meyer, D.E.; Curran, M.A.; Gonzalez, M.A. Life cycle assessment as a tool to enhance the environmental performance of carbon nanotube products: A review. J. Clean. Prod. 2012, 26, 37–47. [Google Scholar] [CrossRef]
- Hischier, R. Framework for LCI modelling of releases of manufactured nanomaterials along their life cycle. Int. J. Life Cycle Assess. 2014, 19, 838–849. [Google Scholar] [CrossRef]
- Hischier, R. Life cycle assessment of manufactured nanomaterials: Inventory modelling rules and application example. Int. J. Life Cycle Assess. 2014, 19, 941–943. [Google Scholar] [CrossRef]
- Walser, T.; Demou, E.; Lang, D.J.; Hellweg, S. Prospective Environmental Life Cycle Assessment of Nanosilver T-Shirts. Environ. Sci. Technol. 2011, 45, 4570–4578. [Google Scholar] [CrossRef] [PubMed]
- Eckelman, M.J.; Mauter, M.S.; Isaacs, J.A.; Elimelech, M. New Perspectives on Nanomaterial Aquatic Ecotoxicity: Production Impacts Exceed Direct Exposure Impacts for Carbon Nanotoubes. Environ. Sci. Technol. 2012, 46, 2902–2910. [Google Scholar] [CrossRef] [PubMed]
- Salieri, B.; Righi, S.; Pasteris, A.; Olsen, S.I. Freshwater ecotoxicity characterisation factor for metal oxide nanoparticles: A case study on titanium dioxide nanoparticle. Sci. Total Environ. 2015, 505, 494–502. [Google Scholar] [CrossRef] [PubMed]
- Bekker, C.; Kuijpers, E.; Brouwer, D.H.; Vermeulen, R.; Fransman, W. Occupational Exposure to Nano-Objects and Their Agglomerates and Aggregates Across Various Life Cycle Stages; A Broad-Scale Exposure Study. Ann. Occup. Hyg. 2015, 59, 681–704. [Google Scholar] [CrossRef] [PubMed]
- Pirela, S.V.; Sotiriou, G.A.; Bello, D.; Shafer, M.; Bunker, K.L.; Castranova, V.; Thomas, T.; Demokritou, P. Consumer exposures to laser printer-emitted engineered nanoparticles: A case study of life-cycle implications from nano-enabled products. Nanotoxicology 2015, 9, 760–768. [Google Scholar] [CrossRef] [PubMed]
- Barberio, G.; Scalbi, S.; Buttol, P.; Masoni, P.; Righi, S. Combining life cycle assessment and qualitative risk assessment: The case study of alumina nanofluid production. Sci. Total Environ. 2014, 496, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Gavankar, S.; Suh, S.; Keller, A.A. The Role of Scale and Technology Maturity in Life Cycle Assessment of Emerging Technologies: A Case Study on Carbon Nanotubes. J. Ind. Ecol. 2015, 19, 51–60. [Google Scholar] [CrossRef]
- Tsuzuki, T. Life Cycle Thinking and Green Nanotechnology. Austin J. Nanomed. Nanotechnol. 2014, 2, 1010. [Google Scholar]
- Dhingra, R.; Naidu, S.; Upreti, G.; Sawhney, R. Sustainable Nanotechnology: Through Green Methods and Life-Cycle Thinking. Sustainability 2010, 2, 3323–3338. [Google Scholar] [CrossRef]
- Carpenter, A.W.; de Lannoy, C.-F.; Wiesner, M.R. Cellulose Nanomaterials in Water Treatment Technologies. Environ. Sci. Technol. 2015, 49, 5277–5287. [Google Scholar] [CrossRef] [PubMed]
- Gao, W.; Wang, J. The Environmental Impact of Micro/Nanomachines: A Review. ACS Nano 2014, 8, 3170–3180. [Google Scholar] [CrossRef] [PubMed]
- Bartke, S.; Hagemann, N.; Harries, N.; Hauck, J.; Bardos, P. Market potential of nanoremediation in Europe–Market drivers and interventions identified in a deliberative scenario approach. Sci. Total Environ. 2018, 619–620, 1040–1048. [Google Scholar] [CrossRef] [PubMed]
- Gil-Díaz, M.; Alonso, J.; Rodríguez-Valdés, E.; Gallego, J.R.; Lobo, M.C. Comparing different commercial zero valent iron nanoparticles to immobilize As and Hg in brownfield soil. Sci. Total Environ. 2017, 584–585, 1324–1332. [Google Scholar] [CrossRef] [PubMed]
- Gil-Díaz, M.; Diez-Pascual, S.; González, A.; Alonso, J.; Rodríguez-Valdés, E.; Gallego, J.R.; Lobo, M.C. A nanoremediation strategy for the recovery of an As-polluted soil. Chemosphere 2016, 149, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Xue, W.; Huang, D.; Zeng, G.; Wan, J.; Zhang, C.; Xu, R.; Cheng, M.; Deng, R. Nanoscale zero-valent iron coated with rhamnolipid as an effective stabilizer for immobilization of Cd and Pb in river sediments. J. Hazard. Mater. 2018, 341, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Azari, P.; Bostani, A.A. Reducing As availability in calcareous soils using nanoscale zero valent iron. Environ. Sci. Pollut. Res. 2017, 24, 20438–20445. [Google Scholar] [CrossRef] [PubMed]
- Bianco, C.; Patiño Higuita, J.E.; Tosco, T.; Tiraferri, A.; Sethi, R. Controlled Deposition of Particles in Porous Media for Effective Aquifer Nanoremediation. Sci. Rep. 2017, 7, 12992. [Google Scholar] [CrossRef] [PubMed]
- Khlestkin, V.K.; Peltek, S.E.; Kolchanov, N.A. Review of direct chemical and biochemical transformations of starch. Carbohydr. Polym. 2018, 181, 460–476. [Google Scholar] [CrossRef] [PubMed]
- Crini, G. Review: A History of Cyclodextrins. Chem. Rev. 2014, 114, 10940–10975. [Google Scholar] [CrossRef] [PubMed]
- Hu, Q.-D.; Tang, G.-P.; Chu, P.K. Cyclodextrin-Based Host–Guest Supramolecular Nanoparticles for Delivery: From Design to Applications. Acc. Chem. Res. 2014, 47, 2017–2025. [Google Scholar] [CrossRef] [PubMed]
- Webber, M.J.; Langer, R. Drug delivery by supramolecular design. Chem. Soc. Rev. 2017, 46, 6600–6620. [Google Scholar] [CrossRef] [PubMed]
- Prochowicz, D.; Kornowicz, A.; Lewiński, J. Interactions of Native Cyclodextrins with Metal Ions and Inorganic Nanoparticles: Fertile Landscape for Chemistry and Materials Science. Chem. Rev. 2017, 117, 13461–13501. [Google Scholar] [CrossRef] [PubMed]
- Caglieris, F.; Melone, L.; Canepa, F.; Lamura, G.; Castiglione, F.; Ferro, M.; Malpezzi, L.; Mele, A.; Punta, C.; Franchi, P.; et al. Effective magnetic moment in cyclodextrin–polynitroxides: Potential supramolecular vectors for magnetic resonance imaging. RSC Adv. 2015, 5, 76133–76140. [Google Scholar] [CrossRef]
- Caracciolo, F.; Carretta, P.; Filibian, M.; Melone, L. Dynamic Nuclear Polarization of β-Cyclodextrin Macromolecules. J. Phys. Chem. B 2017, 121, 2584–2593. [Google Scholar] [CrossRef] [PubMed]
- Melone, L.; Petroselli, M.; Pastori, N.; Punta, C. Functionalization of Cyclodextrins with N-Hydroxyphthalimide Moiety: A New Class of Supramolecular Pro-Oxidant Organocatalysts. Molecules 2015, 20, 15881–15892. [Google Scholar] [CrossRef] [PubMed]
- Landy, D.; Mallard, I.; Ponchel, A.; Monflier, E.; Fourmentin, S. Remediation technologies using cyclodextrins: An overview. Environ. Chem. Lett. 2012, 10, 225–237. [Google Scholar] [CrossRef]
- Crini, N.; Winterton, P.; Fourmentin, S.; Wilson, L.D.; Fenyvesi, É.; Crini, G. Water-insoluble β-cyclodextrin–epichlorohydrin polymers for removal of pollutants from aqueous solutions by sorption processes using batch studies: A review of inclusion mechanisms. Prog. Polym. Sci. 2017. [Google Scholar] [CrossRef]
- Zeiger, E.; Gollapudi, B.; Spencer, P. Genetic toxicity and carcinogenicity studies of glutaraldehyde? A review. Mutat. Res. Mutat. Res. 2005, 589, 136–151. [Google Scholar] [CrossRef] [PubMed]
- Hirakawa, B. Epichlorohydrin. In Encyclopedia of Toxicology; Elsevier: Amsterdam, The Netherlands, 2014; pp. 431–432. ISBN 978-0-12-386455-0. [Google Scholar]
- Trotta, F.; Cavalli, R.; Tumiatti, W.; Zerbinati, O.; Roggero, C.; Vallero, R. Ultrasound-Assisted Synthesis of Cyclodextrin-Based Nanosponges. WO Application WO2006002814A1, 12 January 2006. [Google Scholar]
- Trotta, F.; Tumiatti, W. Cross-Linked Polymers Based on Cyclodextrins for Removing Polluting Agents. U.S. EP1492822 (A1), 5 January 2005. [Google Scholar]
- Venuti, V.; Rossi, B.; Mele, A.; Melone, L.; Punta, C.; Majolino, D.; Masciovecchio, C.; Caldera, F.; Trotta, F. Tuning structural parameters for the optimization of drug delivery performance of cyclodextrin-based nanosponges. Expert Opin. Drug Deliv. 2017, 14, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Crupi, V.; Fontana, A.; Giarola, M.; Majolino, D.; Mariotto, G.; Mele, A.; Melone, L.; Punta, C.; Rossi, B.; Trotta, F.; et al. Connection between the vibrational dynamics and the cross-linking properties in cyclodextrins-based polymers. J. Raman Spectrosc. 2013, 44, 1457–1462. [Google Scholar] [CrossRef]
- Crupi, V.; Majolino, D.; Mele, A.; Melone, L.; Punta, C.; Rossi, B.; Toraldo, F.; Trotta, F.; Venuti, V. Direct evidence of gel–sol transition in cyclodextrin-based hydrogels as revealed by FTIR-ATR spectroscopy. Soft Matter 2014, 10, 2320–2326. [Google Scholar] [CrossRef] [PubMed]
- Venuti, V.; Rossi, B.; D’Amico, F.; Mele, A.; Castiglione, F.; Punta, C.; Melone, L.; Crupi, V.; Majolino, D.; Trotta, F.; et al. Combining Raman and infrared spectroscopy as a powerful tool for the structural elucidation of cyclodextrin-based polymeric hydrogels. Phys. Chem. Chem. Phys. 2015, 17, 10274–10282. [Google Scholar] [CrossRef] [PubMed]
- Rossi, B.; Venuti, V.; D’Amico, F.; Gessini, A.; Castiglione, F.; Mele, A.; Punta, C.; Melone, L.; Crupi, V.; Majolino, D.; et al. Water and polymer dynamics in a model polysaccharide hydrogel: The role of hydrophobic/hydrophilic balance. Phys. Chem. Chem. Phys. 2014, 17, 963–971. [Google Scholar] [CrossRef] [PubMed]
- Rossi, B.; Venuti, V.; D’Amico, F.; Gessini, A.; Mele, A.; Punta, C.; Melone, L.; Crupi, V.; Majolino, D.; Trotta, F.; et al. Toward an understanding of the thermosensitive behaviour of pH-responsive hydrogels based on cyclodextrins. Soft Matter 2015, 11, 5862–5871. [Google Scholar] [CrossRef] [PubMed]
- Rossi, B.; Venuti, V.; Mele, A.; Punta, C.; Melone, L.; D’Amico, F.; Gessini, A.; Crupi, V.; Majolino, D.; Trotta, F.; et al. Vibrational signatures of the water behaviour upon confinement in nanoporous hydrogels. Phys. Chem. Chem. Phys. 2016, 18, 12252–12259. [Google Scholar] [CrossRef] [PubMed]
- Bottari, C.; Comez, L.; Corezzi, S.; D’Amico, F.; Gessini, A.; Mele, A.; Punta, C.; Melone, L.; Pugliese, A.; Masciovecchio, C.; et al. Correlation between collective and molecular dynamics in pH-responsive cyclodextrin-based hydrogels. Phys. Chem. Chem. Phys. 2017, 19, 22555–22563. [Google Scholar] [CrossRef] [PubMed]
- Ferro, M.; Castiglione, F.; Punta, C.; Melone, L.; Panzeri, W.; Rossi, B.; Trotta, F.; Mele, A. Anomalous diffusion of Ibuprofen in cyclodextrin nanosponge hydrogels: An HRMAS NMR study. Beilstein J. Org. Chem. 2014, 10, 2715–2723. [Google Scholar] [CrossRef] [PubMed]
- Ferro, M.; Castiglione, F.; Punta, C.; Melone, L.; Panzeri, W.; Rossi, B.; Trotta, F.; Mele, A. Transport Properties of Ibuprofen Encapsulated in Cyclodextrin Nanosponge Hydrogels: A Proton HR-MAS NMR Spectroscopy Study. J. Vis. Exp. 2016. [Google Scholar] [CrossRef] [PubMed]
- Ferro, M.; Castiglione, F.; Pastori, N.; Punta, C.; Melone, L.; Panzeri, W.; Rossi, B.; Trotta, F.; Mele, A. Dynamics and interactions of ibuprofen in cyclodextrin nanosponges by solid-state NMR spectroscopy. Beilstein J. Org. Chem. 2017, 13, 182–194. [Google Scholar] [CrossRef] [PubMed]
- Rossi, B.; Paciaroni, A.; Venuti, V.; Fadda, G.C.; Melone, L.; Punta, C.; Crupi, V.; Majolino, D.; Mele, A. SANS investigation of water adsorption in tunable cyclodextrin-based polymeric hydrogels. Phys. Chem. Chem. Phys. 2017, 19, 6022–6029. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Repo, E.; Yin, D.; Meng, Y.; Jafari, S.; Sillanpää, M. EDTA-Cross-Linked β-Cyclodextrin: An Environmentally Friendly Bifunctional Adsorbent for Simultaneous Adsorption of Metals and Cationic Dyes. Environ. Sci. Technol. 2015, 49, 10570–10580. [Google Scholar] [CrossRef] [PubMed]
- Kargarzadeh, H.; Ahmad, I.; Thomas, S.; Dufresne, A. Handbook of Nanocellulose and Cellulose Nanocomposites; John Wiley & Sons: Hoboken, NJ, USA, 2017; ISBN 978-3-527-68998-9. [Google Scholar]
- Lee, K.-Y. Nanocellulose and Sustainability: Production, Properties, Applications, and Case Studies; CRC Press: Boca Raton, FL, USA, 2018; ISBN 978-1-351-26290-3. [Google Scholar]
- Chin, K.-M.; Ting, S.S.; Ong, H.L.; Omar, M. Surface functionalized nanocellulose as a veritable inclusionary material in contemporary bioinspired applications: A review. J. Appl. Polym. Sci. 2018, 135, 46065. [Google Scholar] [CrossRef]
- Mahfoudhi, N.; Boufi, S. Nanocellulose as a novel nanostructured adsorbent for environmental remediation: A review. Cellulose 2017, 24, 1171–1197. [Google Scholar] [CrossRef]
- Mohammed, N.; Grishkewich, N.; Tam, K.C. Cellulose nanomaterials: Promising sustainable nanomaterials for application in water/wastewater treatment processes. Environ. Sci. Nano 2018, 5, 623–658. [Google Scholar] [CrossRef]
- Chang, J.; Zhang, L.; Wang, P. Intelligent environmental nanomaterials. Environ. Sci. Nano 2018, 5, 811–836. [Google Scholar] [CrossRef]
- Voisin, H.; Bergström, L.; Liu, P.; Mathew, A.P. Nanocellulose-Based Materials for Water Purification. Nanomaterials 2017, 7, 57. [Google Scholar] [CrossRef] [PubMed]
- Bossa, N.; Carpenter, A.W.; Kumar, N.; de Lannoy, C.-F.; Wiesner, M. Cellulose nanocrystal zero-valent iron nanocomposites for groundwater remediation. Environ. Sci. Nano 2017, 4, 1294–1303. [Google Scholar] [CrossRef] [PubMed]
- Pierre, G.; Punta, C.; Delattre, C.; Melone, L.; Dubessay, P.; Fiorati, A.; Pastori, N.; Galante, Y.M.; Michaud, P. TEMPO-mediated oxidation of polysaccharides: An ongoing story. Carbohydr. Polym. 2017, 165, 71–85. [Google Scholar] [CrossRef] [PubMed]
- Isogai, A.; Saito, T.; Fukuzumi, H. TEMPO-oxidized cellulose nanofibers. Nanoscale 2011, 3, 71–85. [Google Scholar] [CrossRef] [PubMed]
- Melone, L.; Rossi, B.; Pastori, N.; Panzeri, W.; Mele, A.; Punta, C. TEMPO-Oxidized Cellulose Cross-Linked with Branched Polyethyleneimine: Nanostructured Adsorbent Sponges for Water Remediation. ChemPlusChem 2015, 80, 1408–1415. [Google Scholar] [CrossRef]
- Chen, W.; Li, Q.; Wang, Y.; Yi, X.; Zeng, J.; Yu, H.; Liu, Y.; Li, J. Comparative Study of Aerogels Obtained from Differently Prepared Nanocellulose Fibers. ChemSusChem 2014, 7, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Melone, L.; Altomare, L.; Alfieri, I.; Lorenzi, A.; De Nardo, L.; Punta, C. Ceramic aerogels from TEMPO-oxidized cellulose nanofibre templates: Synthesis, characterization, and photocatalytic properties. J. Photochem. Photobiol. Chem. 2013, 261, 53–60. [Google Scholar] [CrossRef]
- Panzella, L.; Melone, L.; Pezzella, A.; Rossi, B.; Pastori, N.; Perfetti, M.; D’Errico, G.; Punta, C.; d’Ischia, M. Surface-Functionalization of Nanostructured Cellulose Aerogels by Solid State Eumelanin Coating. Biomacromolecules 2016, 17, 564–571. [Google Scholar] [CrossRef] [PubMed]
- Ge, H.; Huang, H.; Xu, M.; Chen, Q. Cellulose/poly(ethylene imine) composites as efficient and reusable adsorbents for heavy metal ions. Cellulose 2016, 23, 2527–2537. [Google Scholar] [CrossRef]
- Zhang, N.; Zang, G.-L.; Shi, C.; Yu, H.-Q.; Sheng, G.-P. A novel adsorbent TEMPO-mediated oxidized cellulose nanofibrils modified with PEI: Preparation, characterization, and application for Cu(II) removal. J. Hazard. Mater. 2016, 316, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.; Repo, E.; Song, Y.; Yin, D.; Hammouda, S.B.; Chen, L.; Kalliola, S.; Tang, J.; Tam, K.C.; Sillanpää, M. Polyethylenimine-cross-linked cellulose nanocrystals for highly efficient recovery of rare earth elements from water and a mechanism study. Green Chem. 2017, 19, 4816–4828. [Google Scholar] [CrossRef]
- Melone, L.; Bonafede, S.; Tushi, D.; Punta, C.; Cametti, M. Dip in colorimetric fluoride sensing by a chemically engineered polymeric cellulose/ bPEI conjugate in the solid state. RSC Adv. 2015, 5, 83197–83205. [Google Scholar] [CrossRef]
- Fiorati, A.; Turco, G.; Travan, A.; Caneva, E.; Pastori, N.; Cametti, M.; Punta, C.; Melone, L. Mechanical and Drug Release Properties of Sponges from Cross-linked Cellulose Nanofibers. ChemPlusChem 2017, 82, 848–858. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef] [PubMed]
- Bonilla-Petriciolet, A.; Mendoza-Castillo, D.I.; Reynel-Avila, H.E. Adsorption Processes for Water Treatment and Purification; Springer: Berlin/Heidelberg, Germany, 2017; ISBN 978-3-319-58135-4. [Google Scholar]
- Rene, E.R.; Sahinkaya, E.; Lewis, A.; Lens, P.N.L. Sustainable Heavy Metal Remediation; Springer: Berlin/Heidelberg, Germany, 2017; Volumes 1 & 2, ISBN 978-3-319-58621-2. [Google Scholar]
| Nanoparticles Type | Remediation Mechanism | Remediated Contaminants | Potential Toxicity | Test Organisms | Reference |
|---|---|---|---|---|---|
| nZVI | Adsorption; oxidation; reduction | metals; chlorinated pollutants | Algal growth inhibition; ROS generation; oxidative stress; disruption of membrane integrity; genotoxicity; morphological alterations of roots; oxygen consumption | bacteria; freshwater microalga; freshwater crustaceans; earthworm; plant | [48,49,74,75] |
| Iron-based ENMs | Adsorption; oxidation; reduction | metals; microbiological contaminants | Algal growth inhibition; ROS generation; oxidative stress; disruption of membrane integrity; genotoxicity; mutagenicity; reproduction impairment | bacteria; freshwater microalga; freshwater crustaceans; earthworm; plant; fish | [48,49,72] |
| TiO2 | Photodegradation | organic contaminants | ROS generation; oxidative stress; membrane damage; cell viability reduction; reproduction impairment; tissues alterations and gill histopathology; neurotoxicity | bacteria; crustacean; plant; fish | [57,58,60,76] |
| ZnO | Photocatalysis; photodegradation; adsorption | organic contaminants; heavy metals | Algal growth inhibition; ROS generation; gill damage; embryotoxicity; metal stress via dissolution and ion release; membrane damages | bacteria; freshwater microalga; crustacean; fish | [58,60,61,77] |
| CNT-based ENMs | Catalytic facilitation; adsorption | organic contaminants; heavy metals | toxicity enhancement of contaminants; carrying of pollutants; ROS generation; growth rate inhibition; membrane damage | bacteria; microalga; crustaceans; mollusks | [58,69,70,78] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Corsi, I.; Fiorati, A.; Grassi, G.; Bartolozzi, I.; Daddi, T.; Melone, L.; Punta, C. Environmentally Sustainable and Ecosafe Polysaccharide-Based Materials for Water Nano-Treatment: An Eco-Design Study. Materials 2018, 11, 1228. https://doi.org/10.3390/ma11071228
Corsi I, Fiorati A, Grassi G, Bartolozzi I, Daddi T, Melone L, Punta C. Environmentally Sustainable and Ecosafe Polysaccharide-Based Materials for Water Nano-Treatment: An Eco-Design Study. Materials. 2018; 11(7):1228. https://doi.org/10.3390/ma11071228
Chicago/Turabian StyleCorsi, Ilaria, Andrea Fiorati, Giacomo Grassi, Irene Bartolozzi, Tiberio Daddi, Lucio Melone, and Carlo Punta. 2018. "Environmentally Sustainable and Ecosafe Polysaccharide-Based Materials for Water Nano-Treatment: An Eco-Design Study" Materials 11, no. 7: 1228. https://doi.org/10.3390/ma11071228
APA StyleCorsi, I., Fiorati, A., Grassi, G., Bartolozzi, I., Daddi, T., Melone, L., & Punta, C. (2018). Environmentally Sustainable and Ecosafe Polysaccharide-Based Materials for Water Nano-Treatment: An Eco-Design Study. Materials, 11(7), 1228. https://doi.org/10.3390/ma11071228








