Elucidating the Photocatalytic Behavior of TiO2-SnS2 Composites Based on Their Energy Band Structure
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Photocatalyst Synthesis and Immobilization
2.3. Photocatalyst Characterization
2.4. Photocatalytic Activity under Solar Irradiation
3. Results and Discussion
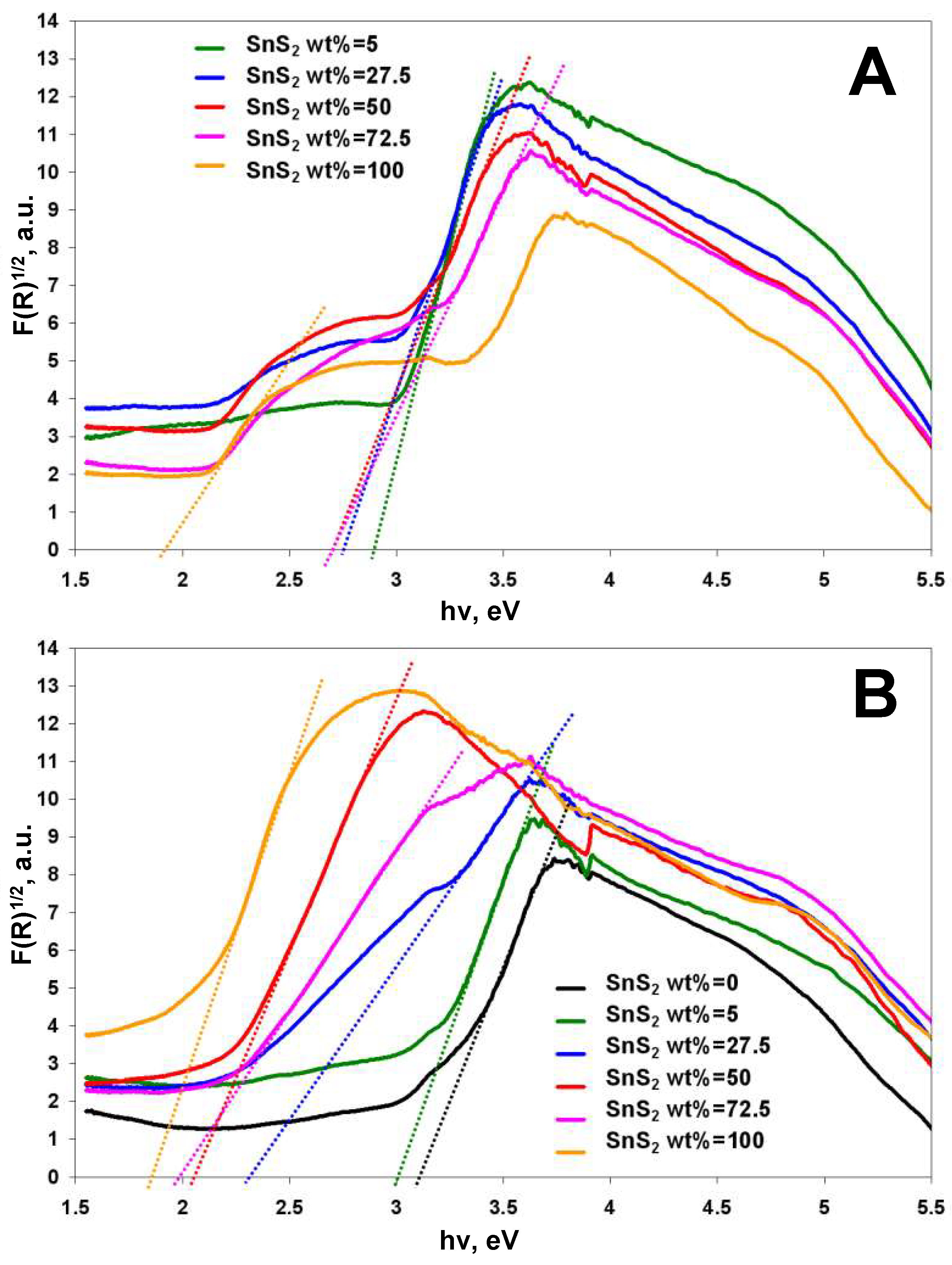
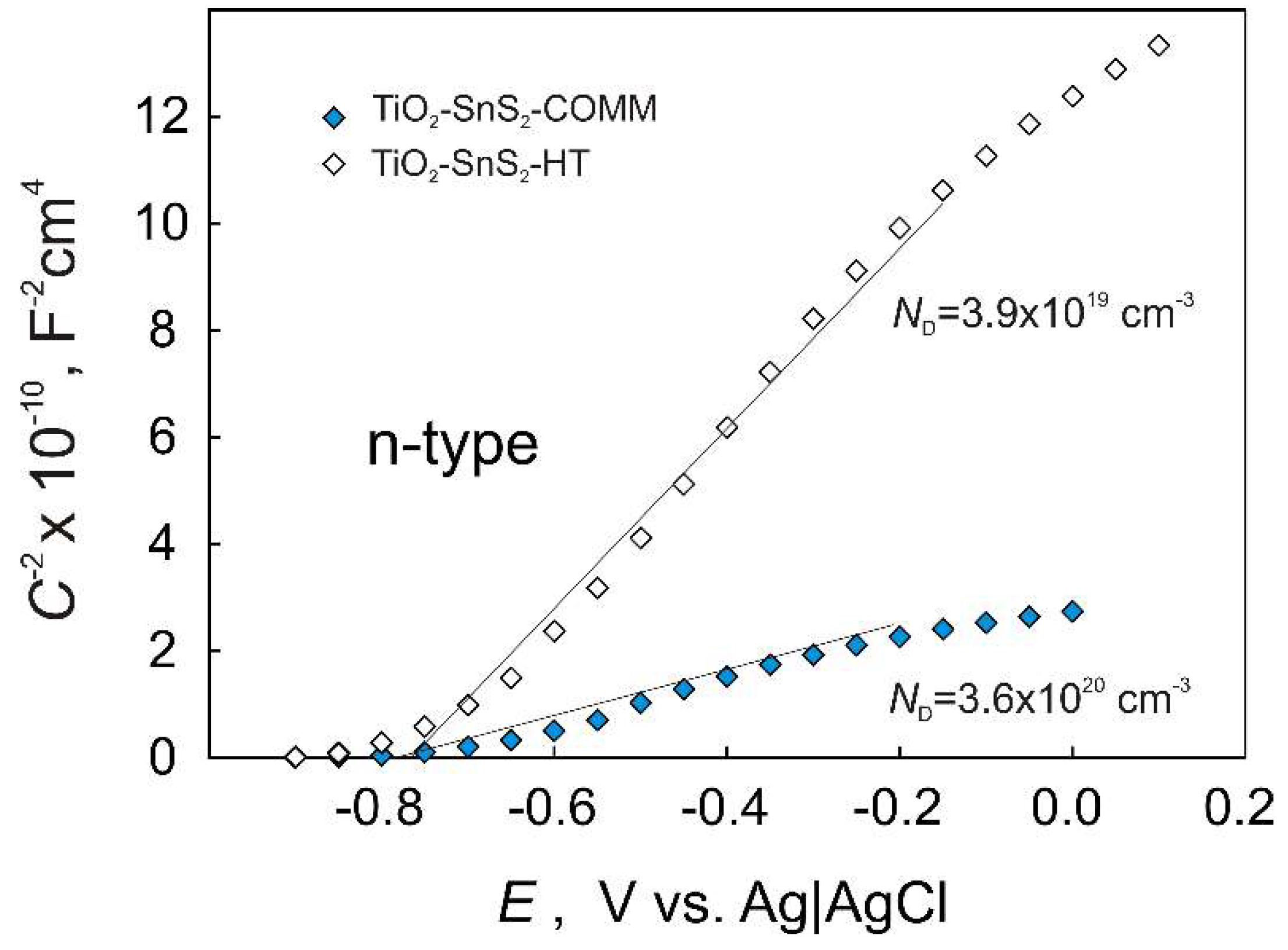
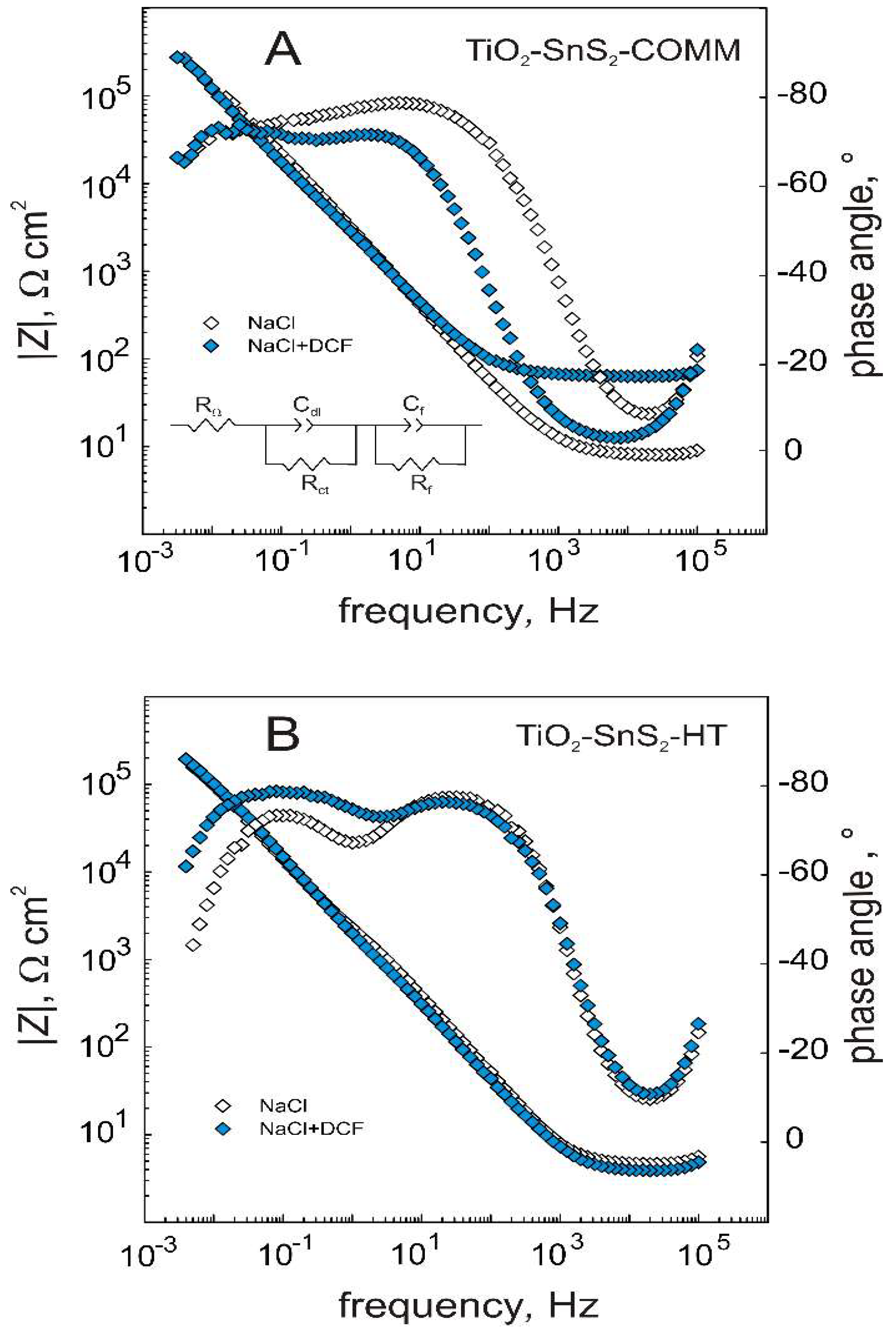
3.1. Semiconducting and Surface Properties of TiO2-SnS2 Composite Materials
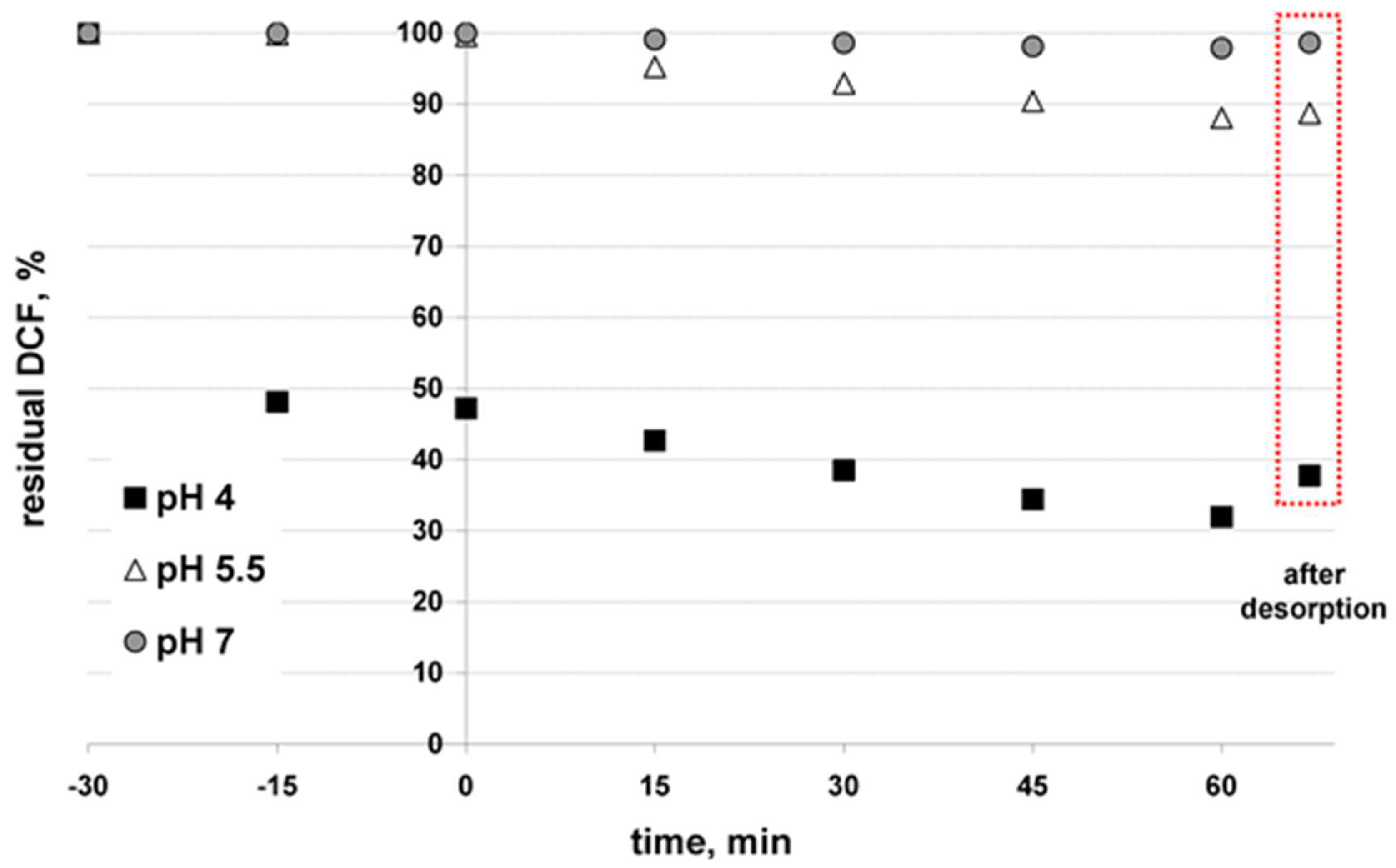
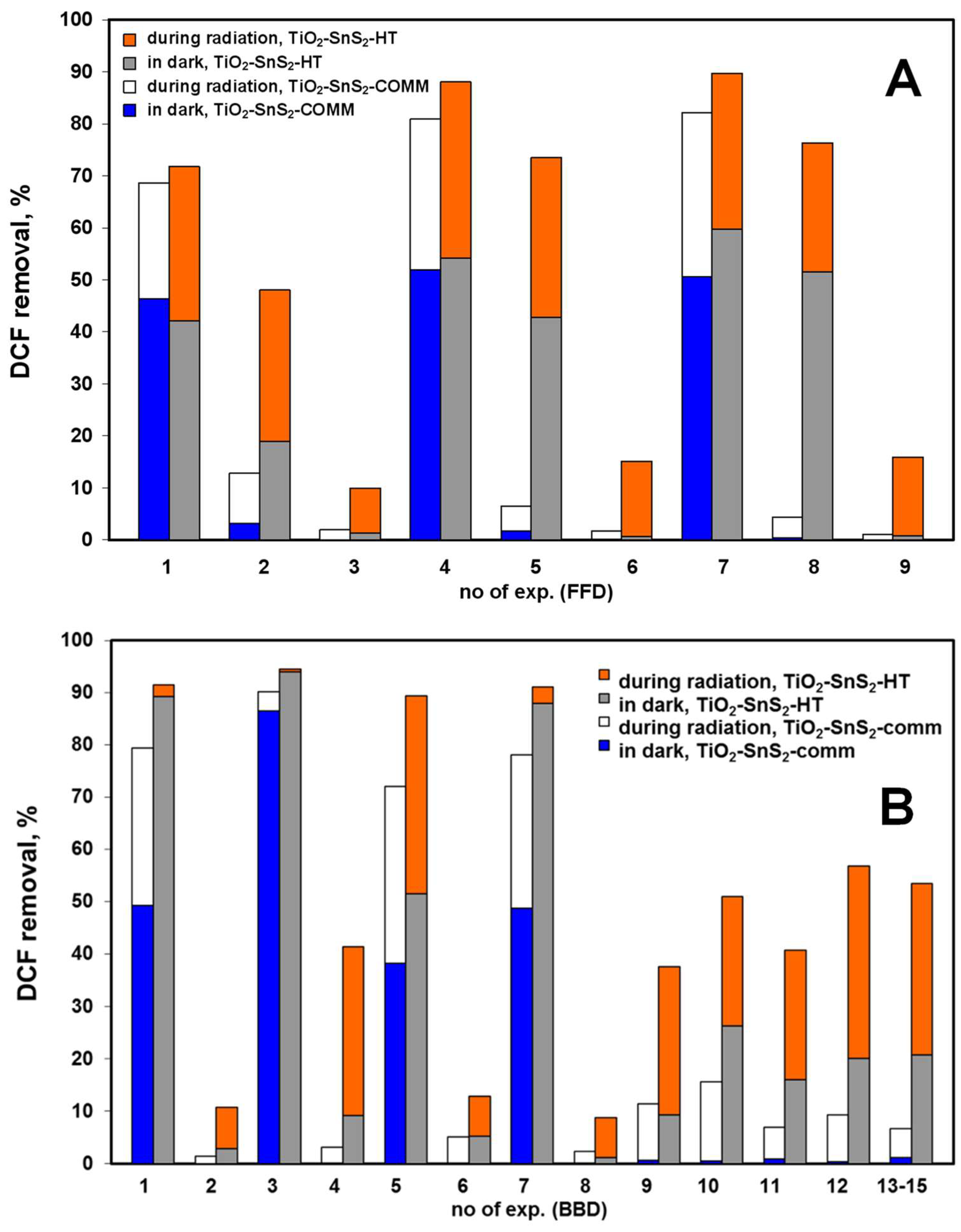
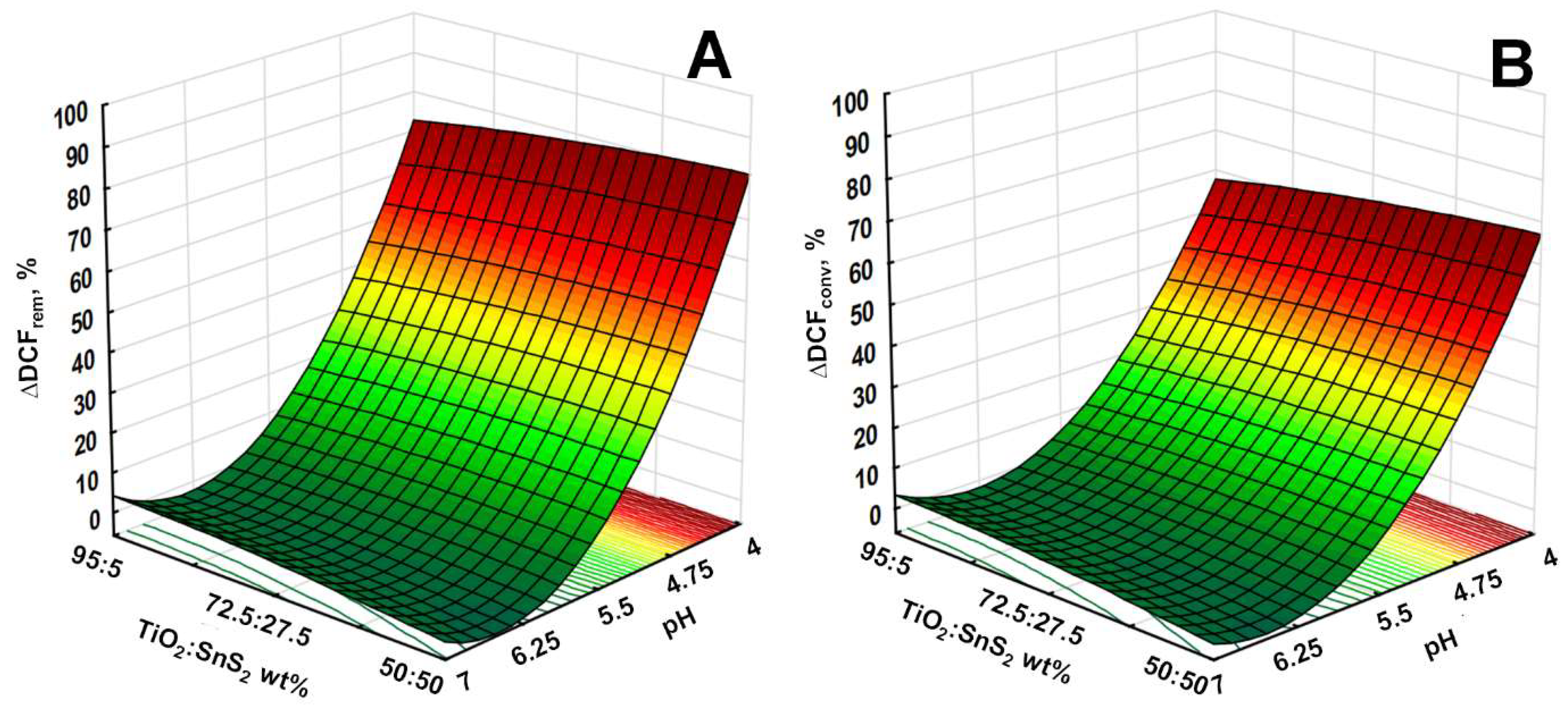
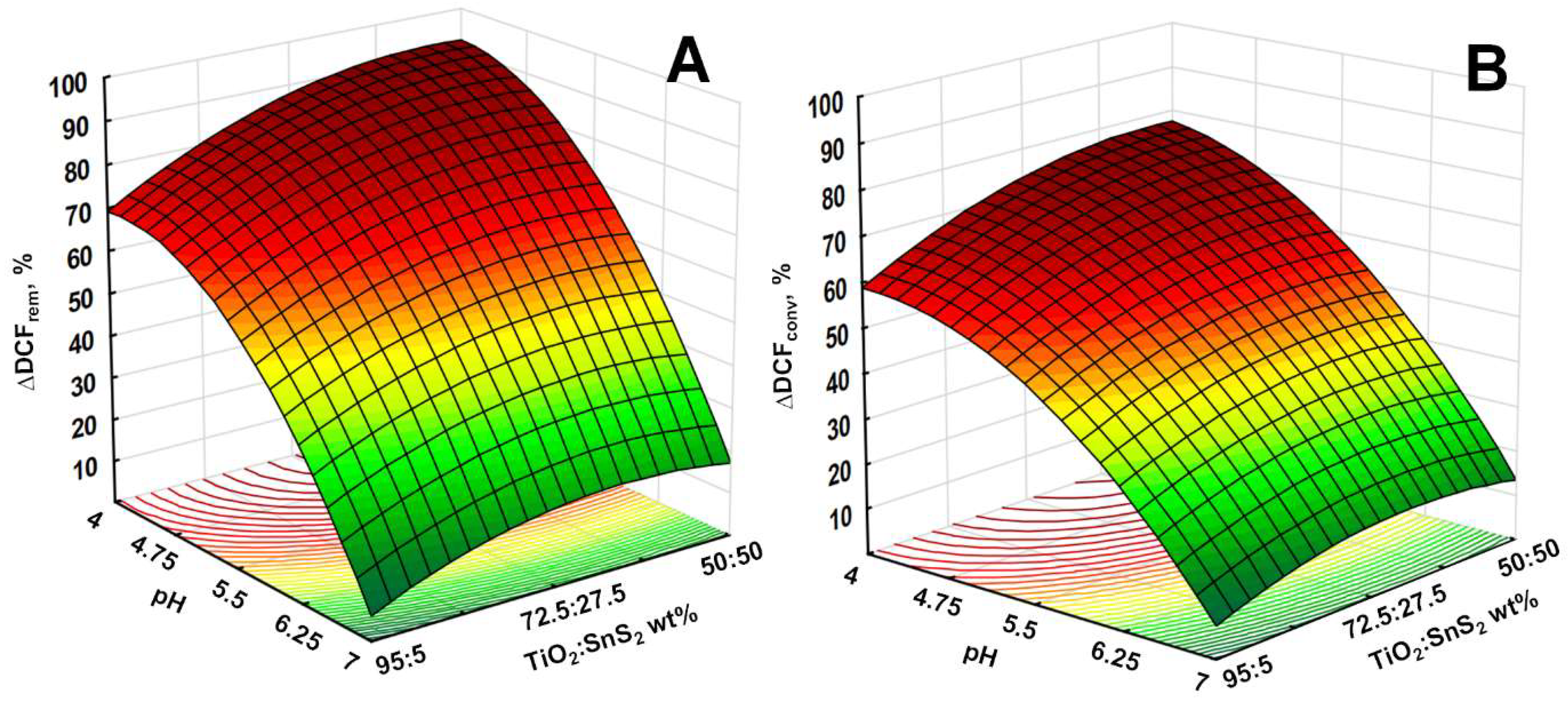
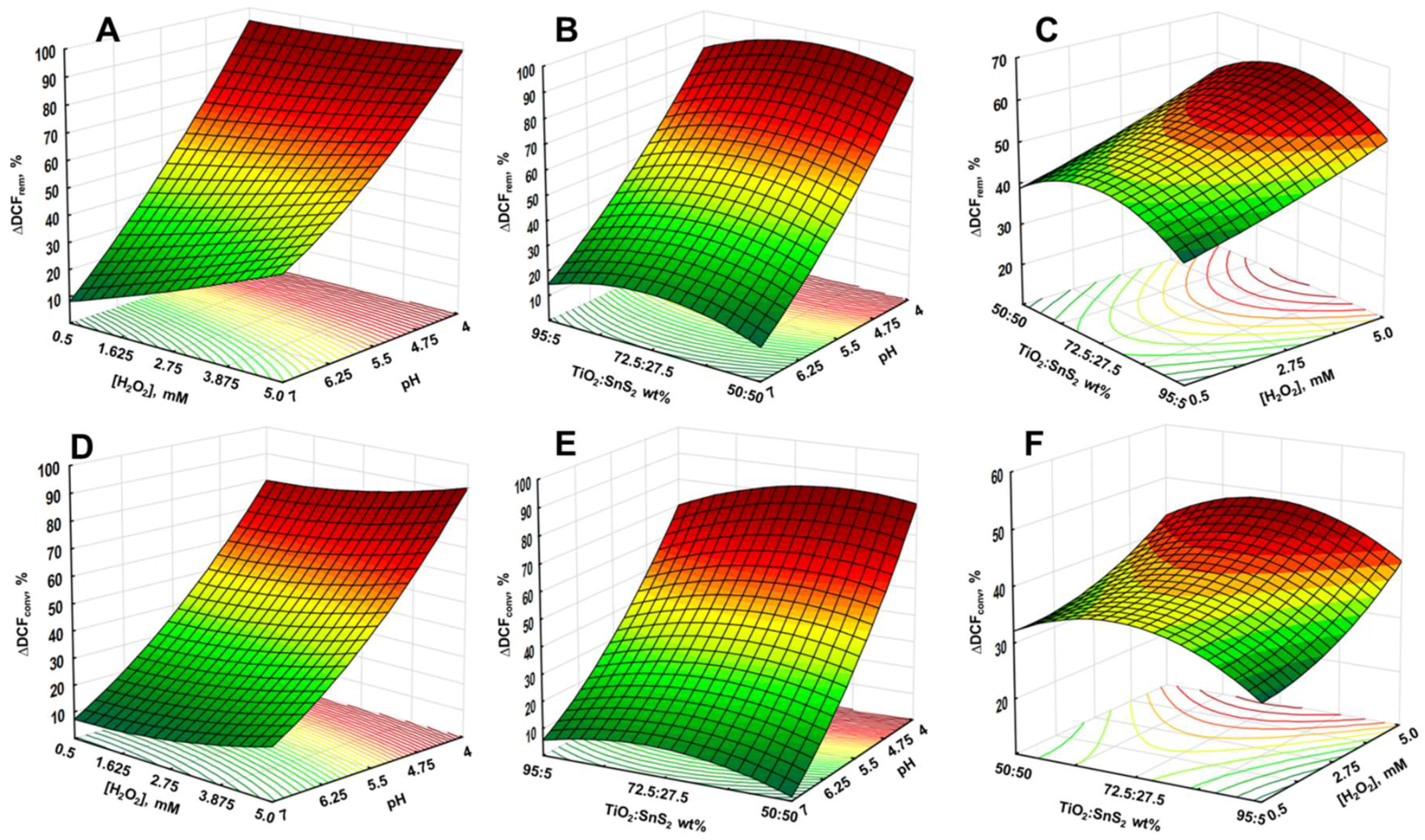
3.2. Solar-Driven Photocatalytic Treatment of DCF Using TiO2-SnS2 Catalysts
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wang, F.; Hu, S. Electrochemical sensors based on metal and semiconductor nanoparticles. Microchim. Acta 2009, 165, 1–22. [Google Scholar] [CrossRef]
- Oi, L.E.; Choo, M.Y.; Lee, H.V.; Ong, H.C.; Hamid, S.B.A.; Juan, J.C. Recent advances of titanium dioxide (TiO2) for green organic synthesis. RSC Adv. 2016, 6, 108741–108754. [Google Scholar] [CrossRef]
- Banerjee, S.; Dionysiou, D.D.; Pillai, S.C. Self-cleaning applications of TiO2 by photo-induced hydrophilicity and photocatalysis. Appl. Catal. B 2015, 176–177, 396–428. [Google Scholar] [CrossRef]
- Ni, M.; Leung, M.K.H.; Leung, D.Y.C.; Sumathy, K. A review and recent developments in photocatalytic water-splitting using TiO2 for hydrogen production. Renew. Sustain. Energy Rev. 2007, 11, 401–425. [Google Scholar] [CrossRef]
- Kovacic, M.; Salaeh, S.; Kusic, H.; Suligoj, A.; Fanetti, M.; Stangar, U.L.; Dionysiou, D.D.; Bozic, A.L. Solar-driven photocatalytic treatment of diclofenac using immobilized TiO2-based zeolite composites. Environ. Sci. Pollut. Res. 2016, 23, 17982–17994. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Zhou, H.; Cicek, N. Treatment of Organic Micropollutants in Water and Wastewater by UV-Based Processes: A Literature Review. Crit. Rev. Environ. Sci. Technol. 2014, 44, 1443–1476. [Google Scholar] [CrossRef]
- Méndez, E.; González-Fuentes, M.A.; Rebollar-Perez, G.; Méndez-Abores, A.; Torres, E. Emerging pollutant treatments in wastewater: Cases of antibiotics and hormones. J. Environ. Sci. Health Part A 2017, 52, 235–253. [Google Scholar] [CrossRef] [PubMed]
- Landmann, M.; Raus, E.; Schmidt, W.G. The electronic structure and optical response of rutile, anatase and brookite TiO2. J. Phys. Condens. Matter 2012, 24, 195503. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.C.; Li, J.; Xu, H.Y. One-step in situ solvothermal synthesis of SnS2/TiO2 nanocomposites with high performance in visible light-driven photocatalytic reduction of aqueous Cr(VI). Appl. Catal. B 2012, 123–124, 18–26. [Google Scholar] [CrossRef]
- Burton, L.A.; Colombara, D.; Abellon, R.D.; Grozema, F.C.; Peter, L.M.; Savenije, T.J.; Dennler, G.; Walsh, A. Synthesis, characterization, and electronic structure of single-crystal SnS, Sn2S3, and SnS2. Chem. Mater. 2013, 25, 4908–4916. [Google Scholar] [CrossRef]
- Vogel, R.; Hoyer, P.; Weller, H. Quantum-sized PbS, CdS, Ag2S, SbS3, and Bi2S3 particles as sensitizers for various nanoporous wide- bandgap semiconductors. J. Phys. Chem. 1994, 98, 3183–3188. [Google Scholar] [CrossRef]
- Velasco, M.V.R.; Sarruf, F.D.; Salgado-Santos, I.M.N.; Haroutiounian-Filho, C.A.; Kaneko, T.M.; Baby, A.R. Broad spectrum bioactive sunscreens. Int. J. Pharm. 2008, 363, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Meissner, D.; Benndorf, C.; Memming, R. Photocorrosion of cadmium sulfide: Analysis by photoelectron spectroscopy. Appl. Surf. Sci. 1987, 38, 423–436. [Google Scholar] [CrossRef]
- Ohko, Y.; Setani, M.; Sakata, T.; Mori, H.; Yoneyama, H. Preparation of monodisperse ZnS Nanoparticles by size selective photocorrosion. Chem. Lett. 1999, 7, 663–664. [Google Scholar] [CrossRef]
- Nguyen, C.C.; Vu, N.N.; Do, T.O. Recent advances in the development of sunlight-driven hollow structure photocatalysts and their application. J. Mater. Chem. A 2015, 3, 18345–18359. [Google Scholar] [CrossRef]
- Tachibana, Y.; Umekita, K.; Otsuka, Y.; Kuwabata, S. Charge Recombination Kinetics at an in Situ Chemical Bath-Deposited CdS/Nanocrystalline TiO2 Interface. J. Phys. Chem. C 2009, 113, 6852–6858. [Google Scholar] [CrossRef]
- Chi, Y.J.; Fu, H.G.; Qi, L.H.; Shi, K.Y.; Zhang, H.B.; Yu, H.T. Preparation and photoelectric performance of ITO/TiO2/CdS composite thin films. J. Photochem. Photobiol. A 2008, 195, 357–363. [Google Scholar] [CrossRef]
- Li, X.; Zhu, J.; Li, H.X. Comparative study on the mechanism in photocatalytic degradation of different-type organic dyes on SnS2 and CdS. Appl. Catal. B 2012, 123–124, 174–181. [Google Scholar] [CrossRef]
- Kusic, H.; Leszczynska, D. Altered toxicity of organic pollutants in water originated from simultaneous exposure to UV photolysis and CdSe/ZnS quantum dots. Chemosphere 2012, 89, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Burton, L.A.; Whittles, T.J.; Hesp, D.; Linhart, W.M.; Skelton, J.M.; Hou, B.; Webster, R.F.; O’Dowd, G.; Reece, C.; Cherns, D.; et al. Electronic and optical properties of single crystal SnS2: An earth-abundant disulfide photocatalyst. J. Mater. Chem. A 2016, 4, 1312–1318. [Google Scholar] [CrossRef]
- Hinojosa-Reyes, L.; Guzmán-Mar, J.L.; Villanueva-Rodríguez, M. Semiconductor Materials for Photocatalytic Oxidation of Organic Pollutants in Wastewater. In Photocatalytic Semiconductors: Synthesis, Characterization, and Environmental Applications; Hernández-Ramírez, A., Medina-Ramírez, I., Eds.; Springer International Publishing: Basel, Switzerland, 2015. [Google Scholar]
- Orazem, M.E.; Tribollet, B. Electrochemical Impedance Spectroscopy; John Wiley & Sons: New York, NY, USA, 2008. [Google Scholar]
- Katić, J.; Metikoš-Huković, M.; Šarić, I.; Petravić, M. Semiconducting properties of the oxide films formed on tin: Capacitive and XPS studies. J. Electrochem. Soc. 2016, 163, C221–C227. [Google Scholar] [CrossRef]
- Katić, J.; Metikoš-Huković, M.; Šarić, I.; Petravić, M. Electronic structure and redox behavior of tin sulfide films potentiostatically formed on tin. J. Electrochem. Soc. 2017, 164, C383–C389. [Google Scholar] [CrossRef]
- EU. Directive 2013/39/EU of the European Parliament and of the Council amending Directives 2000/60/EC and 2008/105/EC as regards priority substances in the field of water policy. Off. J. Eur. Commun. 2013, 226, 1–17. [Google Scholar]
- Postigo, C.; Barceló, D. Synthetic organic compounds and their transformation products in groundwater: Occurrence, fate and mitigation. Sci. Total Environ. 2015, 503–504, 32–47. [Google Scholar] [CrossRef] [PubMed]
- Stülten, D.; Zühlke, S.; Lamshöft, M.; Spiteller, M. Occurrence of diclofenac and selected metabolites in sewage effluents. Sci. Total Environ. 2008, 405, 310–316. [Google Scholar] [CrossRef] [PubMed]
- Kete, M.; Pavlica, E.; Fresno, F.; Bratina, G.; Stangar, U.L. Highly active photocatalytic coatings prepared by a low-temperature method. Environ. Sci. Pollut. Res. 2014, 21, 11238–11249. [Google Scholar] [CrossRef] [PubMed]
- Lopez, R.; Gomez, R. Band-gap energy estimation from diffuse reflectance measurements on sol–gel and commercial TiO2: A comparative study. J. Sol-Gel Sci. Technol. 2012, 61, 1–7. [Google Scholar] [CrossRef]
- Koci, K.; Obalova, L.; Matejova, L.; Placha, D.; Lacny, Z.; Jirkovsky, J.; Solcova, O. Effect of TiO2 particle size on the photocatalytic reduction of CO2. Appl. Catal. B 2009, 89, 494–502. [Google Scholar] [CrossRef]
- D’Amato, C.A.; Giovannetti, R.; Zannotti, M.; Rommozzi, E.; Ferraro, S.; Seghetti, C.; Minicucci, M.; Gunnella, R.; Di Cicco, A. Enhancement of visible-light photoactivity by polypropylene coated plasmonic Au/TiO2 for dye degradation in water solution. Appl. Surf. Sci. 2018, 441, 575–587. [Google Scholar] [CrossRef]
- Uppal, H.; Tawale, J.; Singh, N. Zinc peroxide functionalized synthetic graphite: An economical and efficient adsorbent for adsorption of arsenic (III) and (V). J. Environ. Chem. Eng. 2016, 4, 2964–2975. [Google Scholar] [CrossRef]
- Kovacic, M.; Kusic, H.; Fanetti, M.; Stangar, U.L.; Valant, M.; Dionysiou, D.D.; Bozic, A.L. TiO2-SnS2 nanocomposites; solar active photocatalytic materials for water treatment. Environ. Sci. Pollut. Res. 2017, 24, 19965–19979. [Google Scholar] [CrossRef] [PubMed]
- Boukamp, A. A nonlinear least squares fit procedure for analysis of immittance data of electrochemical systems. Solid State Ion. 1986, 20, 31–44. [Google Scholar] [CrossRef]
- Nogueira, F.R.; Oliveira, M.C.; Paterlini, W.C. Simple and fast spectrophotometric determination of H2O2 in photo-Fenton reactions using metavanadate. Talanta 2005, 66, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Kovacic, M.; Kopcic, N.; Kusic, H.; Bozic, A.L. Solar driven degradation of 17β-estradiol using composite photocatalytic materials and artificial irradiation source; influence of process and water matrix parameters. J. Photochem. Photobiol. A 2018, 361, 48–61. [Google Scholar] [CrossRef]
- Morrison, S.R. Electrochemistry at Semiconductor and Oxidized Metal Electrodes; Plenum Press: New York, NY, USA, 1980. [Google Scholar]
- Memming, R. Semiconductor Electrochemistry; Wiley-VCH: Weinheim, Germany, 2015. [Google Scholar]
- Bockris, J.O.M.; Khan, S.U.M. Surface Electrochemistry: A Molecular Level Approach; Plenum Press: New York, NY, USA, 1993. [Google Scholar]
- Chong, M.N.; Jin, B.; Chow, C.W.K.; Saint, C. Recent developments in photocatalytic water treatment technology: A review. Water Res. 2010, 44, 2997–3027. [Google Scholar] [CrossRef] [PubMed]
- Trovo, A.G.; Nogueira, R.F.P. Diclofenac abatement using modified solar photo-Fenton process with ammonium iron(III) citrate. J. Braz. Chem. Soc. 2011, 22, 1033–1039. [Google Scholar] [CrossRef]
- Pichat, P. Photocatalysis and Water Purification: From Fundamentals to Recent Applications; Wiley: Weinheim, Germany, 2013. [Google Scholar]
- Robertson, P.K.J.; Robertson, J.M.C.; Bahnemann, D.W. Removal of microorganisms and their chemical metabolites from water using semiconductor photocatalysis. J. Hazard. Mater. 2012, 211–212, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Bard, A.J.; Parsons, R.; Jordan, J. Standard Potentials in Aqueous Solution; IUPAC, Marcel Dekker Inc.: New York, NY, USA, 1985. [Google Scholar]
- Lide, D.R. (Ed.) CRC Handbook of Chemistry and Physics; CRC Press: Boca Raton, FL, USA, 1995. [Google Scholar]
- Perrone, J. (Ed.) Chemical Thermodynamics, Vol. 12 Chemical Thermodynamics of Tin; OECD Publications: Paris, France, 2012. [Google Scholar]
- Gerischer, H. The impact of semiconductors on the concepts of electrochemistry. Electrochim. Acta 1990, 35, 1677–1699. [Google Scholar] [CrossRef]


| Process Parameters | Model Variables/Coded Values | Level/Range | ||
|---|---|---|---|---|
| −1 | 0 | 1 | ||
| pH | X1 | 4 | 5.5 | 7 |
| H2O2, mM * | X2 | 0.5 | 2.75 | 5 |
| SnS2, wt % ** | X3 | 5 | 27.5 | 50 |
| TiO2-SnS2 Catalyst Band Gap; Eg, eV | ||||||
|---|---|---|---|---|---|---|
| Type | SnS2 wt % | |||||
| 0 | 5 | 27.5 | 50 | 72.5 | 100 | |
| TiO2-SnS2-COMM | 3.05 * | 2.89 | 2.75 | 2.67 | 2.66 | 1.91 |
| TiO2-SnS2-HT | 3.09 | 2.99 | 2.29 | 2.04 | 1.96 | 1.88 |
| Material | BET Surface Area, m2 g−1 | pHPZC |
|---|---|---|
| TiO2-SnS2-COMM | 96.00 ± 0.78 | 6.31 |
| TiO2-SnS2-HT | 160.58 ± 0.46 | 4.61 |
| TiO2-SnS2-COMM | |||||
| Electrolyte | RΩ, Ω cm2 | Cdl, μF cm−2 | Rct, kΩ cm2 | Cf, μF cm−2 | Rf, MΩ cm2 |
| NaCl | 7.6 | 45.0 | 17.63 | 49.8 | 1.21 |
| NaCl + DCF | 63.3 | 63.8 | 4.51 | 52.3 | 1.29 |
| TiO2-SnS2-HT | |||||
| NaCl | 4.5 | 61.5 | 0.95 | 47.8 | 0.24 |
| NaCl + DCF | 4.0 | 230.6 | 0.13 | 31.8 | 0.81 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kovacic, M.; Katic, J.; Kusic, H.; Loncaric Bozic, A.; Metikos Hukovic, M. Elucidating the Photocatalytic Behavior of TiO2-SnS2 Composites Based on Their Energy Band Structure. Materials 2018, 11, 1041. https://doi.org/10.3390/ma11061041
Kovacic M, Katic J, Kusic H, Loncaric Bozic A, Metikos Hukovic M. Elucidating the Photocatalytic Behavior of TiO2-SnS2 Composites Based on Their Energy Band Structure. Materials. 2018; 11(6):1041. https://doi.org/10.3390/ma11061041
Chicago/Turabian StyleKovacic, Marin, Jozefina Katic, Hrvoje Kusic, Ana Loncaric Bozic, and Mirjana Metikos Hukovic. 2018. "Elucidating the Photocatalytic Behavior of TiO2-SnS2 Composites Based on Their Energy Band Structure" Materials 11, no. 6: 1041. https://doi.org/10.3390/ma11061041
APA StyleKovacic, M., Katic, J., Kusic, H., Loncaric Bozic, A., & Metikos Hukovic, M. (2018). Elucidating the Photocatalytic Behavior of TiO2-SnS2 Composites Based on Their Energy Band Structure. Materials, 11(6), 1041. https://doi.org/10.3390/ma11061041







