Adsorption of Pb (II) Ions onto Hydroxyapatite Nanopowders in Aqueous Solutions
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of Hydroxyapatite Nanoparticles (HAp)
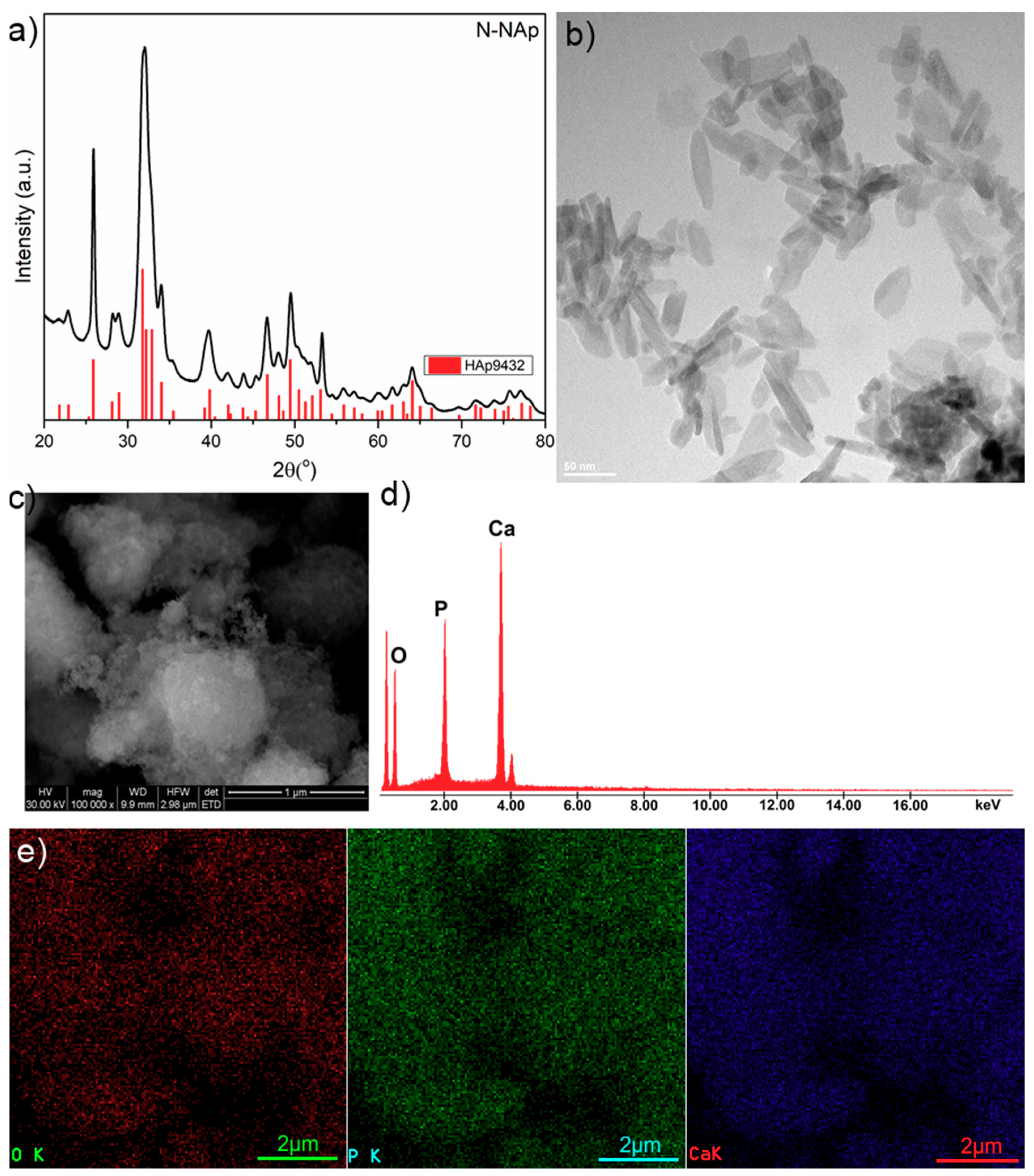
2.2. Structural and Morphological Characterizations
2.3. Batch Adsorption Experiments
3. Results and Discussions
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liao, D.; Zheng, W.; Li, X.; Yang, Q.; Yue, X.; Guo, L.; Zeng, G. Removal of lead(II) from aqueous solutions using carbonate hydroxyapatite extracted from eggshell waste. J. Hazard. Mater. 2010, 177, 126–130. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.H.; Min, B.G.; Jeong, Y.G.; Lyoo, W.S.; Lee, S.C. Removal of lead ions in aqueous solution by hydroxyapatite/polyurethane composite foams. J. Hazard. Mater. 2008, 152, 1285–1292. [Google Scholar] [CrossRef] [PubMed]
- Raita, Ş.; Cornilă, N.; Danacu, V.; Rosu, P.; Barbuceanu, F. Histological researches concerning the pancreas in Struthio camelus Anatomia. Blackwell Verlag GmbH Ana. Histol. Embryol. 2014, 43, 74–75. [Google Scholar]
- Iconaru, S.L.; Prodan, A.M.; Turculet, C.S.; Beuran, M.; Ghita, R.V.; Costescu, A.; Groza, A.; Chifiriuc, M.C.; Chapon, P.; Gaiaschi, S.; et al. Enamel Based Composite Layers Deposited on Titanium Substrate with Antifungal Activity. J. Spectroscop. 2016, 4361051. [Google Scholar] [CrossRef]
- Gillham, R.W.; O’Hannesin, R.S. Enhanced degradation of halogentaed aliphatics by zero-valent iron. Ground Water 1994, 32, 958–967. [Google Scholar] [CrossRef]
- Biswas, P.; Wu, C.Y. Nanoparticles and the Environment, Critical Review Paper. J. Air Waste Manag. Assoc. 2005, 55, 708–746. [Google Scholar] [CrossRef] [PubMed]
- Elliott, D.W.; Zhang, W.X. Field assessment of nanoscale bimetallic particles for groundwater treatment. Environ. Sci. Technol. 2000, 35, 4922–4926. [Google Scholar] [CrossRef]
- Suciu, I.; Vârlan, C.; Dimitriu, B.; Sorițău, O.; Amza, O.; Moldoveanu, G.F.; Burlibașa, M.; Gheorghe, I.; Lazar, V.; Gheorghiu, I. Biocompatibility Study of Several Esthetic Dental Restorative Materials. Rom. Biotechnol. Lett. 2016, 21, 11438–11442. [Google Scholar]
- Predoi, D.; Predoi, M.V.; Iconaru, S.L.; Ech Cherif El Kettani, M.; Leduc, D.; Prodan, A.M. Ultrasonic Measurements on β Cyclodextrin/Hydroxyapatite Composites for Potential Water Depollution. Materials 2017, 10, 681. [Google Scholar] [CrossRef] [PubMed]
- Raita, S.; Cornilă, N.; Dănacu, V.; Belu, C.; Georgescu, B.; Roșu, P.; Bărbuceanu, F. Morphological studies on the liver in Struthio camelus. Anatomia. Blackwell Verlag GmbH Ana. Histol. Embryol. 2014, 43, 74–75. [Google Scholar]
- Prodan, A.M.; Beuran, M.; Turculet, C.S.; Popa, M.; Andronescu, E.; Bleotu, C.; Raita, S.M.; Soare, M.; Lupescu, O. In vitro evaluation of glycerol coated iron oxide nanoparticles in solution. Rom. Biotechnol. Lett. 2018, 23, 13901–13908. [Google Scholar]
- Turculet, C.S.; Prodan, A.M.; Negoi, I.; Teleanu, G.; Popa, M.; Andronescu, E.; Beuran, M.; Stanciu, G.A.; Hristu, R.; Badea, M.L.; et al. Preliminary evaluation of the antifungal activity of samarium doped hydroxyapatite thin films. Rom. Biotechnol. Lett. 2018, 23, 13927–13932. [Google Scholar]
- Zamani, S.; Salahi, E.; Mobasherpour, I. Removal of Nickel from aqueous solution by nano hydroxyapatite originated from Persian Gulf corals. Can. Chem. Trans. 2013, 1, 173–190. [Google Scholar]
- Zhu, R.; Yu, R.; Yao, J.; Mao, D.; Xing, C.; Wang, D. Removal of Cd2+ from aqueous solutions by hydroxyapatite. Catal. Today 2008, 139, 94–99. [Google Scholar] [CrossRef]
- Milonjić, S.K. Comments on “factors influencing the removal of divalent cations by hydroxyapatite”. J. Hazard. Mater. 2009, 162, 1588–1589. [Google Scholar] [CrossRef] [PubMed]
- Iconaru, S.L.; Motelica-Heino, M.; Guegan, R.; Predoi, M.V.; Prodan, A.M.; Predoi, D. Removal of Zinc Ions Using Hydroxyapatite and Study of Ultrasound Behavior of Aqueous Media. Materials 2018, 11, 1350. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, T.; Hatsushika, T.; Hayakawa, Y. Synthetic hydroxyapatites employed as inorganic cation-exchangers. J. Chem. Soc. Faraday Trans. 1981, 77, 1059–1062. [Google Scholar] [CrossRef]
- Gupta, S.S.; Bhattacharyya, K.G. Kinetics of adsorption of metal ions on inorganic materials: A review. Adv. Colloid Inter. Sci. 2011, 162, 39–58. [Google Scholar] [CrossRef] [PubMed]
- Di Natale, F.; Erto, A.; Lancia, A.; Musmarra, D. A descriptive model for metallic ions adsorption from aqueous solutions onto activated carbons. J. Hazard. Mater. 2009, 169, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Erto, A.; Di Natale, F.; Musmarra, D.; Lancia, A. Modeling of single and competitive adsorption of cadmium and zinc onto activated carbon. Adsorption 2015, 21, 611–621. [Google Scholar] [CrossRef]
- Suzuki, Y.; Takeuchi, Y. Uptake of a few divalent heavy metal ionic species by a fixed bed of hydroxyapatite particles. J. Chem. Eng. Jpn. 1994, 27, 571–576. [Google Scholar] [CrossRef]
- Suzuki, T.; Ishigaki, K.; Miyake, M. Hydroxyapatites as inorganic cation exchangers. Part 3: Exchange characteristics of lead ions (Pb2+). J. Chem. Soc. Faraday Trans. 1984, 80, 3157–3165. [Google Scholar] [CrossRef]
- Martı́nez, C.E.; McBride, M.B. Aging of coprecipitated Cu in alumina: Changes in structural location, chemical form, and solubility. Geochim. Cosmochim. Acta 2000, 64, 1729–1736. [Google Scholar] [CrossRef]
- Borum, L.; Wilson, O.C., Jr. Surface modification of hydroxyapatite. Part II. Silica. Biomaterials 2003, 24, 3681–3688. [Google Scholar] [CrossRef]
- Ciobanu, C.S.; Constantin, L.V.; Predoi, D. Structural and physical properties of antibacterial Ag-doped nano-hydroxyapatite synthesized at 100 C. Nanoscale Res. Lett. 2011, 6, 613. [Google Scholar] [CrossRef] [PubMed]
- Ciobanu, C.S.; Iconaru, S.L.; Le Coustumer, P.; Constantin, L.V.; Predoi, D. Antibacterial activity of silver-doped hydroxyapatite nanoparticles against gram-positive and gram-negative bacteria. Nanoscale Res. Lett. 2012, 7, 324. [Google Scholar] [CrossRef] [PubMed]
- Ciobanu, C.S.; Iconaru, S.L.; Popa, C.L.; Motelica-Heino, M.; Predoi, D. Evaluation of samarium doped hydroxyapatite, ceramics for medical application: Antimicrobial activity. J. Nanomater. 2015, 2015, 849216. [Google Scholar] [CrossRef]
- Ciobanu, C.S.; Andronescu, E.; Predoi, D. Bet and Xrd Studies on the Hydroxyapatite and Europium Doped Hydroxyapatite. Dig. J. Nanomater. Biostruct. 2011, 6, 1239–1244. [Google Scholar]
- Ciobanu, C.S.; Iconaru, S.L.; Massuyeau, F.; Constantin, L.V.; Costescu, A.; Predoi, D. Synthesis, structure, and luminescent properties of europium-doped hydroxyapatite nanocrystalline powders. J. Nanomater. 2012, 942801. [Google Scholar] [CrossRef]
- Ciobanu, C.S.; Iconaru, S.L.; Le Coustumer, P.; Predoi, D. Vibrational Investigations of Silver-Doped Hydroxyapatite with Antibacterial Properties. J. Spectrosc. 2013, 471061. [Google Scholar] [CrossRef]
- Ciobanu, C.S.; Andronescu, E.; Stoicu, A.; Florea, O.; Le Coustumer, P.; Galaup, S.; Djouadi, A.; Mevellec, J.Y.; Musa, I.; Massuyeau, F.; et al. Influence of Annealing Treatment of Nano-Hydroxyapatite Bioceramics on the Vibrational Properties. Dig. J. Nanomater. Biostruct. 2011, 6, 609–624. [Google Scholar]
- Ciobanu, C.S.; Iconaru, S.L.; Popa, C.L.; Costescu, A.; Motelica-Heino, M.; Predoi, D. Porous Methyltrimethoxysilane Coated Nanoscale-Hydroxyapatite for Removing Lead Ions from Aqueous Solutions. J. Nanomater. 2014, 361061. [Google Scholar] [CrossRef]
- Ślósarczyk, A.; Paszkiewicz, Z.; Paluszkiewicz, C. FTIR and XRD evaluation of carbonated hydroxyapatite powders synthesized by wet methods. J. Mol. Struct. 2005, 744–747, 657–661. [Google Scholar] [CrossRef]
- Costescu, A.; Ciobanu, C.S.; Iconaru, S.L.; Ghita, R.V.; Chifiriuc, C.M.; Marutescu, L.G.; Predoi, D. Fabrication, Characterization, and Antimicrobial Activity, Evaluation of Low Silver Concentrations in Silver-Doped Hydroxyapatite Nanoparticles. J. Nanomater. 2013, 194854. [Google Scholar] [CrossRef]
- Cusco, I.R.; Guitian, F.; de Aza, S.; Arttis, L. Differentiation between Hydroxyapatite and β-Tricalcium Phosphate by Means of p-Raman Spectroscopy. J. Eur. Ceram. Soc. 1998, 18, 1301–1305. [Google Scholar] [CrossRef]
- Brunauer, S.; Emet, P.H.; Teller, E. Adsorption of Gases in Multimolecular Layers. J. Am. Chem. Soc. 1938, 60, 309–319. [Google Scholar] [CrossRef]
- Barrett, E.P.; Joyner, L.G.; Halenda, P.P. The determination of pore volume and area distributions in porous substances. I. Computations from nitrogen isotherms. J. Am. Chem. Soc. 1951, 73, 373–380. [Google Scholar] [CrossRef]
- Cozmuta, L.M.; Cozmuta, A.M.; Peter, A.; Nicula, C.; Nsimba, E.B.; Tutu, H. The influence of pH on the adsorption of lead by Na-clinoptilolite: Kinetic and equilibrium studies. Water SA 2012, 38, 269–278. [Google Scholar] [CrossRef]
- Zhang, M.; Jin, C.C.; Xu, L.H.; Ding, T. Effect of temperature, salinity, and pH on the adsorption of lead by sediment of a tidal river in east China. In Proceedings of the 2012 International Conference on Biomedical Engineering and Biotechnology, Macao, China, 28–30 May 2012. [Google Scholar]
- Malek, A.; Farooq, S. Comparison of isotherm models for hydrocarbon adsorption on activated carbon. AIChE J. 1996, 42, 3191–3201. [Google Scholar] [CrossRef]
- Langmuir, I. The constitution and fundamental properties of solids and liquids. J. Am. Chem. Soc. 1916, 38, 2221–2295. [Google Scholar] [CrossRef]
- Bulut, E.; Ozacar, M.; Sengil, I.A. Adsorption of malachite green onto bentonite: Equilibrium and kinetic studies and process design. Micropor. Mesopor. Mater. 2008, 115, 234–246. [Google Scholar] [CrossRef]
- Ncibi, M.C. Applicability of some statistical tools to predict optimum adsorption isotherm after linear and non-linear regression analysis. J. Hazard. Mater. 2008, 153, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Vijayaraghavan, K.; Padmesh, T.V.N.; Palanivelu, K.; Velan, M. Biosorption of nickel(II) ions onto Sargassum wightii: Application of two-parameterand three parameter isotherm models. J. Hazard. Mater. 2006, B133, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Kundu, S.; Gupta, A.K. Arsenic adsorption onto iron oxide-coated cement (IOCC): Regression analysis of equilibrium data with several isotherm models and their optimization. Chem. Eng. J. 2006, 122, 93–106. [Google Scholar] [CrossRef]
- Allen, S.J.; Mckay, G.; Porter, J.F. Adsorption isotherm models for basic dyea dsorption by peat in single and binary component systems. J. Colloid Interface Sci. 2004, 280, 322–333. [Google Scholar] [CrossRef] [PubMed]
- Langmuir, I. The adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 1918, 40, 1361–1403. [Google Scholar] [CrossRef]
- Ho, Y.S.; Huang, C.T.; Huang, H.W. Equilibrium sorption isotherm for metal ions on tree fern. Process. Biochem. 2002, 37, 1421–1430. [Google Scholar] [CrossRef]
- Ho, Y.S. Selection of optimum sorption isotherm. Carbon. 2004, 42, 2115–2116. [Google Scholar] [CrossRef]
- He, J.; Hong, S.; Zhang, L.; Gan, F.; Ho, Y.S. Equilibrium and Thermodynamic parameters of adsorption of merhylene blue onto rectorite. Fresen. Environ. Bull. 2010, 19, 2651–2656. [Google Scholar]
- Minh, D.P.; Tran, N.D.; Nzihou, A.; Sharrock, P. Hydroxyapatite gel for the improved removal of Pb2+ ions from aqueous. Solution Chem. Eng. J. 2013, 232, 128–138. [Google Scholar] [CrossRef]
- Freundlich, H.M.F. Over the adsorption in solution. J. Phys. Chem. 1906, 57, 385–471. [Google Scholar]
- Adamson, A.W.; Gast, A.P. Physical Chemistry of Surfaces, 6th ed.; Wiley-Interscience: New York, NY, USA, 1997. [Google Scholar]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Zeldowitsch, J. Adsorption site energy distribution. Acta Phys. Chim. URSS 1934, 1, 961–973. [Google Scholar]
- Voudrias, E.; Ytianos, F.; Bozani, E. Sorption Description isotherms of Dyes from aqueous solutions and Waste Waters with Different Sorbent materials, Global Nest. Int. J. 2002, 4, 75–83. [Google Scholar]
- Mohan, S.; Karthikeyan, J. Removal of lignin and tannin color from aqueous solution by adsorption on to activated carbon solution by adsorption on to activated charcoal. Environ. Pollut. 1997, 97, 183–187. [Google Scholar] [CrossRef]
- Goldberg, S. Equations and Models Describing Adsorption Processes in Soils. In Chemical Processes in Soils; Soil Science Society of America: Madison, WI, USA, 2005; p. 489. [Google Scholar]
- Sangeetha, K.; Vidhya, G.; Vasugi, G.; Girija, E.K. Lead and cadmium removal from single and binary metal ion solution by novel hydroxyapatite/alginate/gelatin nanocomposites. J. Environ. Chem. Eng. 2018, 6, 1118–1126. [Google Scholar] [CrossRef]
- Xu, Y.; Schwartz, F.W. Lead immobilization by hydroxyapatite in aqueous solutions. J. Contam. Hydrol. 1994, 15, 187–206. [Google Scholar] [CrossRef]
- Mousavi, H.Z.; Seyedi, S.R. Kinetic and equilibrium studies on the removal of Pb (II) from aqueous solution using nettle ash. J. Chil. Chem. Soc. 2010, 55, 307–311. [Google Scholar] [CrossRef]
- Zheng, W.; Li, X.; Yang, Q.; Zeng, G.; Shen, X.; Zhang, Y.; Liu, J. Adsorption of Cd(II) and Cu(II) from aqueous solution by carbonate hydroxylapatite derived from eggshell waste. J. Hazard. Mater. 2007, 147, 534–539. [Google Scholar] [CrossRef] [PubMed]
- Zhan, X.M.; Zhao, X. Mechanism of lead adsorption from aqueous solutions using an adsorbent synthesized from natural condensed tannin. Water Res. 2003, 37, 3905–3912. [Google Scholar] [CrossRef]
- Mousa, S.M.; Ammar, N.S.; Ibrahim, H.A. Removal of lead ions using hydroxyapatite nano-material prepared from phosphogypsum wast. J. Saudi. Chem. Soc. 2016, 20, 357–365. [Google Scholar] [CrossRef]
- Hafsteinsdóttir, E.G.; Camenzuli, D.; Rocavert, A.L.; Walworth, J.; Gore, D.B. Chemical immobilization of metals and metalloids by phosphates. Appl. Geochem. 2015, 59, 47–62. [Google Scholar] [CrossRef]
- Peng, Q.; Guo, J.; Zhan, Q.; Xiang, J.; Liu, B.; Zhou, A.; Liu, R.; Tian, Y. Unique Lead Adsorption Behavior of Activated Hydroxyl Group in Two-Dimensional Titanium Carbide. J. Am. Chem. Soc. 2014, 136, 4113–4116. [Google Scholar] [CrossRef] [PubMed]
- Günay, A.; Arslankaya, A.; Tosun, I. Lead removal from aqueous solution by natural and pretreated clinoptilolite: Adsorption equilibrium and kinetics. J. Hazard. Mater. 2007, 146, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Vila, M.; Sandra Sánchez-Salcedo, S.; Vallet-Regí, M. Hydroxyapatite foams for the immobilization of heavy metals: From waters to the human body. Inorg. Chim. Acta 2012, 393, 24–35. [Google Scholar] [CrossRef]
- Ma, Q.Y.; Traina, S.J.; Logan, T.J.; Ryan, J. In situ lead immobilization by apatite. Environ. Sci. Technol. 1993, 27, 1803–1810. [Google Scholar] [CrossRef]
- Gandhi, M.R.; Meenakshi, G.N.K.S. Removal of copper(II) using chitin/chitosan nano-hydroxyapatite composite. Int. J. Biol. Macromol. 2011, 48, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Liang, W.; Zhan, L.; Piao, L.; Rüssel, C. Lead and copper removal fromaqueous solutions by porous glass derived calcium hydroxyapatite. Mater. Sci. Eng. B 2011, 176, 1010–1014. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, M.; Chen, W.; Zhu, S.; Liu, N.; Zhu, L. Immobilization of lead and cadmium from aqueous solution and contaminated sediment using nano-hydroxyapatite. Environ. Pollut. 2010, 158, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Corami, A.; Mignardi, S.; Ferrini, V. Cadmium removal from single- and multi-metal (Cd + Pb + Zn + Cu) solutions by sorption on hydroxyapatite. J. Colloid Interface Sci. 2008, 317, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Fang, Z.; Zheng, L.; Cheng, W.; Tsang, P.E.; Fang, J.; Zhao, D. Remediation of lead contaminated soil by biochar-supported nano-hydroxyapatite. Ecotoxicol. Environ. Saf. 2016, 132, 224–230. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Fang, Z.; Tsang, P.E.; Fang, J.; Zhao, D. In situ remediation and phytotoxicity assessment of lead-contaminated soil by biochar-supported nHAP. J. Environ. Manag. 2016, 182, 247–251. [Google Scholar] [CrossRef] [PubMed]
- Đorđević, M.P.; Maletaškić, J.; Stanković, N.; Babić, B.; Yoshidab, K.; Yano, T.; Matović, B. In-situ immobilization of Sr radioactive isotope using nanocrystalline hydroxyapatite. Ceram. Int. 2018, 44, 1771–1777. [Google Scholar] [CrossRef]







| N-HAp Information from BJH Analysis | N-HAp | |
|---|---|---|
| Cumulative pore surface area (Adsorption) obtained by the BJH method (m2/g) | 91.3 | |
| Cumulative pore surface area (Desorption) obtained by the BJH method (m2/g) | 100.1 | |
| Pore volume | Total pore volume (Absorption) (cm³/g) | 0.025 |
| Total pore volume (Desorption) (cm³/g) | 0.4 | |
| Pore size | Average pore size (Absorption) (nm) | 13.6 |
| Average pore size (Desorption) (nm) | 22.6 | |
| The average pore diameter (Absorption) obtained by the BJH method (nm) | 18.23 | |
| The average pore diameter (Desorption) obtained by the BJH method (nm) | 16.66 | |
| Particle size (nm) | 83.37 | |
| Sample | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| N-HAp | R2 | qm (mg/g) | KL (L/mg) | R2 | n | kf |
| 0.997 | 99.304 ± 0.2 | 2.347 ± 0.2 | 0.943 | 1.83 ± 0.3 | 69.785 ± 0.3 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iconaru, S.L.; Motelica-Heino, M.; Guegan, R.; Beuran, M.; Costescu, A.; Predoi, D. Adsorption of Pb (II) Ions onto Hydroxyapatite Nanopowders in Aqueous Solutions. Materials 2018, 11, 2204. https://doi.org/10.3390/ma11112204
Iconaru SL, Motelica-Heino M, Guegan R, Beuran M, Costescu A, Predoi D. Adsorption of Pb (II) Ions onto Hydroxyapatite Nanopowders in Aqueous Solutions. Materials. 2018; 11(11):2204. https://doi.org/10.3390/ma11112204
Chicago/Turabian StyleIconaru, Simona Liliana, Mikael Motelica-Heino, Regis Guegan, Mircea Beuran, Adrian Costescu, and Daniela Predoi. 2018. "Adsorption of Pb (II) Ions onto Hydroxyapatite Nanopowders in Aqueous Solutions" Materials 11, no. 11: 2204. https://doi.org/10.3390/ma11112204
APA StyleIconaru, S. L., Motelica-Heino, M., Guegan, R., Beuran, M., Costescu, A., & Predoi, D. (2018). Adsorption of Pb (II) Ions onto Hydroxyapatite Nanopowders in Aqueous Solutions. Materials, 11(11), 2204. https://doi.org/10.3390/ma11112204








