Preparation and Characterization of Cyclotrimethylenetrinitramine (RDX) with Reduced Sensitivity
Abstract
:1. Introduction
2. Results and Discussion
2.1. Validity of Force Field
2.2. Simulated Growth Morphology of RDX in Vacuum
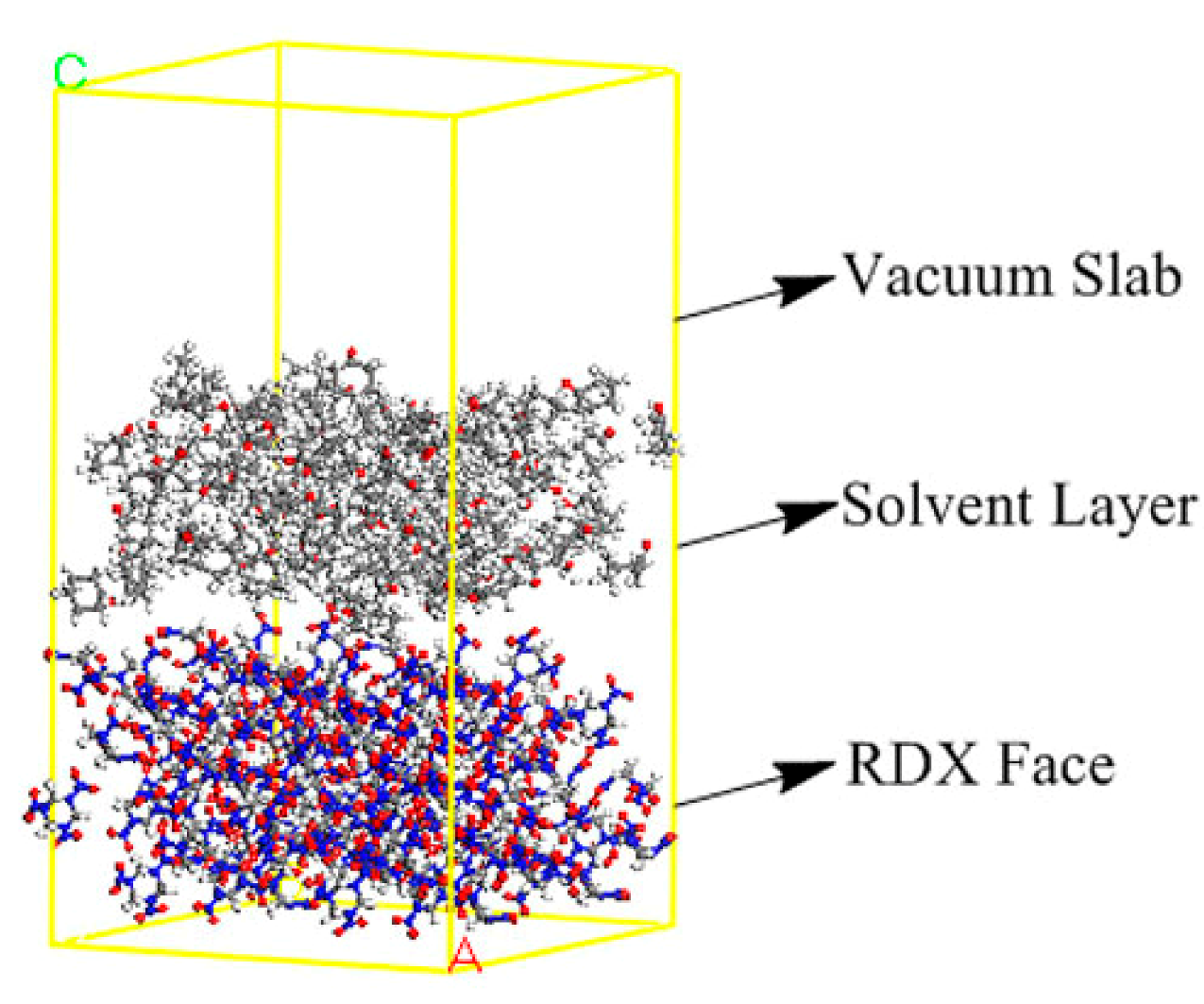
2.3. Calculated Interaction Energy (Eint) between RDX and Solvent
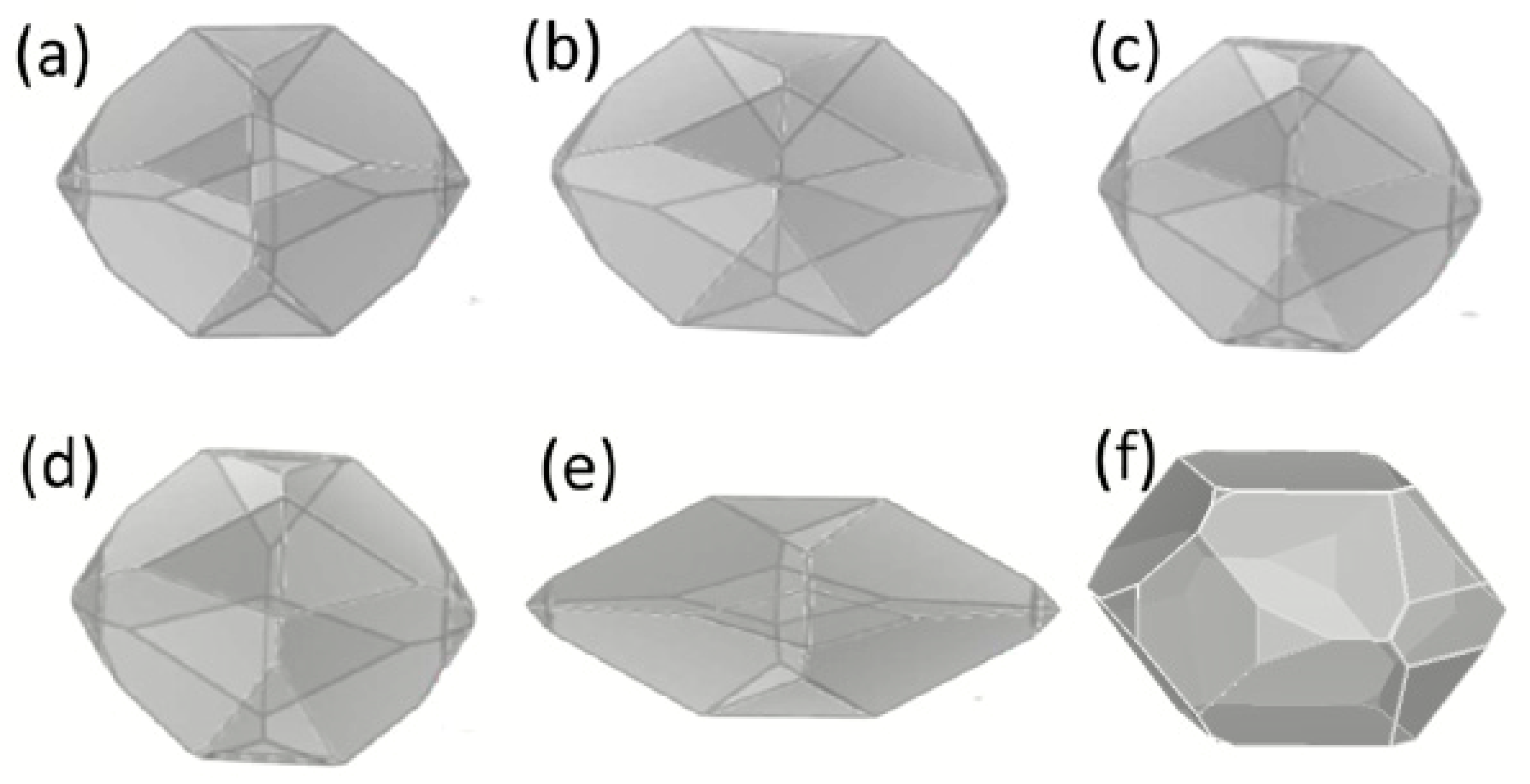
2.4. Re-Crystallization of RDX in Selected Solvents
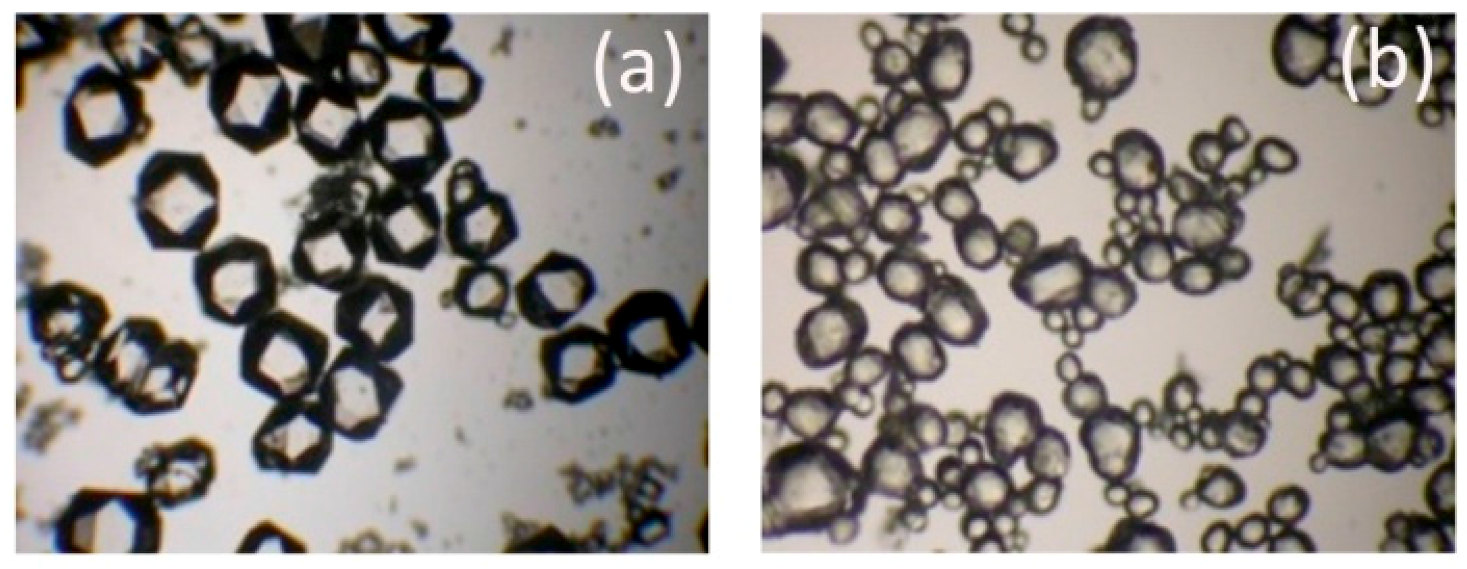
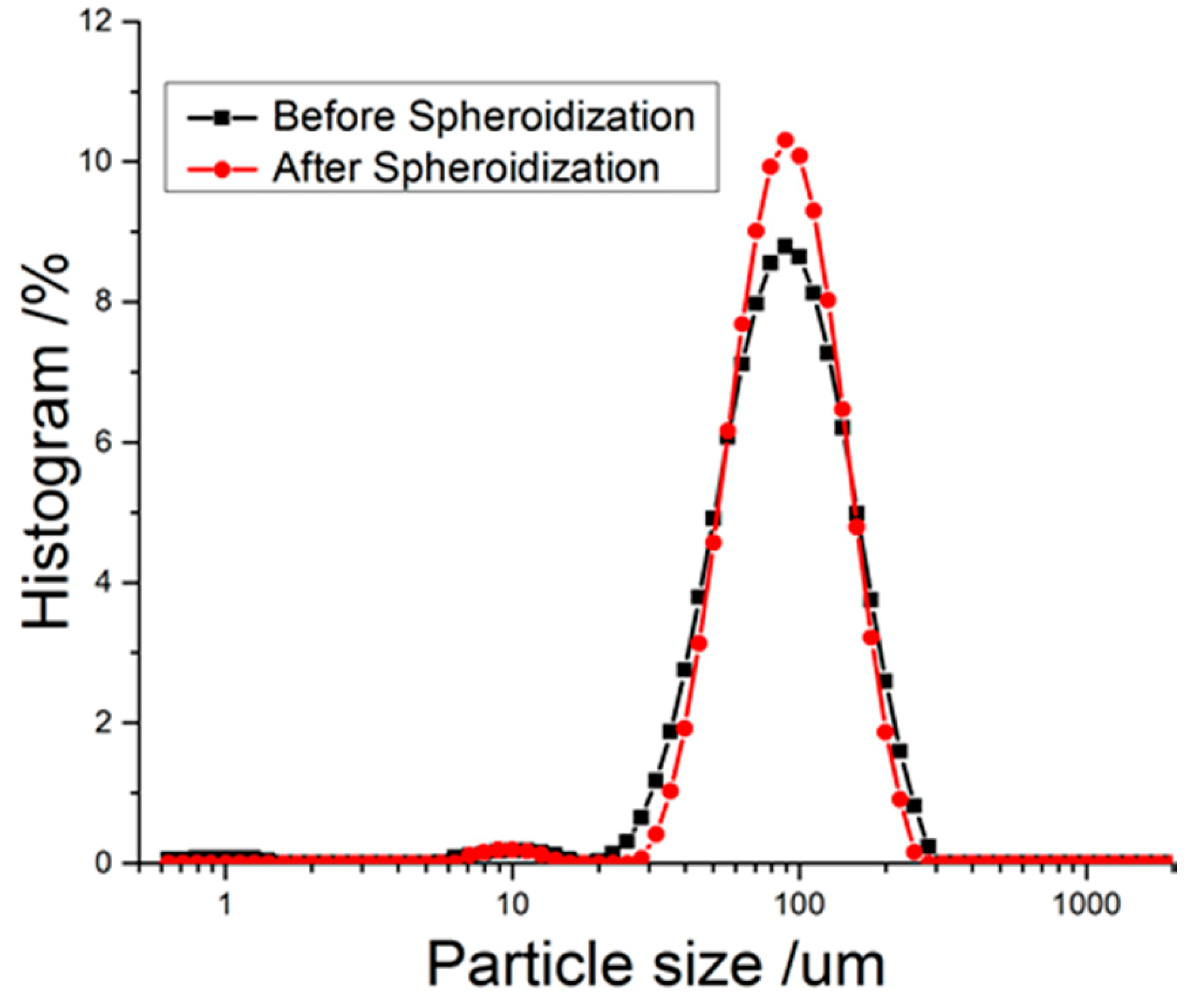
2.5. Spheroidization of RDX
2.6. Sensitivity of RDX
3. Materials and Methods
3.1. Computation
3.2. Experiment
3.2.1. Materials
3.2.2. Re-Crystallization of RDX
3.2.3. Spheroidization of RDX
3.2.4. Preparation of PBXN-109 and PBXW-115
3.2.5. Density Measurement
3.2.6. Purity Measurement
3.2.7. Size Distribution and Specific Surface Area Measurements
3.2.8. Sensitivity Test
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Johansen, Ø.H.; Kristiansen, J.D.; Gjersøe, R.; Berg, A.; Halvorsen, T.; Smith, K. RDX and HMX with Reduced Sensitivity Towards Shock Initiation—RS-RDX and RS-HMX. Propellants Explos. Pyrotech. 2008, 1, 20–24. [Google Scholar] [CrossRef]
- Spyckerelle, C.; Eck, G.; Berg, P.S.; Amnéus, A. Reduced Sensitivity RDX Obtained From Bachmann RDX. Propellants Explos. Pyrotech. 2008, 33, 14–19. [Google Scholar] [CrossRef]
- Doherty, R.M.; Watt, D.S. Relationship between RDX Properties and Sensitivity. Propellants Explos. Pyrotech. 2008, 33, 4–13. [Google Scholar] [CrossRef]
- Borne, L.; Patedoye, J.; Spyckerelle, C. Quantitative Characterization of Internal Defects in RDX Crystals. Propellants Explos. Pyrotech. 1999, 24, 255–259. [Google Scholar] [CrossRef]
- Oxley, J.; Smith, J.; Buco, R.; Huang, J. A Study of Reduced-Sensitivity RDX. J. Energ. Mater. 2007, 25, 141–160. [Google Scholar] [CrossRef]
- Van der Heijden, A.E.D.M.; Bouma, R.H.B.; van der Steen, A.C. Physicochemical Parameters of Nitramines Influencing Shock Sensitivity. Propellants Explos. Pyrotech. 2004, 29, 304–313. [Google Scholar] [CrossRef]
- Kim, J.; Kim, J.; Kim, H.; Koo, K. Characterization of Liquid Inclusion of RDX Crystals with a Cooling Crystallization. Cryst. Growth Des. 2009, 9, 2700–2706. [Google Scholar] [CrossRef]
- Chen, G.; Xia, M.; Lei, W.; Wang, F.; Gong, X. A study of the solvent effect on the morphology of RDX crystal by molecular modeling method. J. Mol. Model. 2013, 19, 5397–5406. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Li, L.; Jin, S.; Chen, S.; Jiao, Q. Effects of Additives on ε-HNIW Crystal Morphology and Impact Sensitivity. Propellants Explos. Pyrotech. 2012, 37, 77–82. [Google Scholar] [CrossRef]
- Duan, X.; Wei, C.; Liu, Y.; Pei, C. A molecular dynamics simulation of solvent effects on the crystal morphology of HMX. J. Hazard. Mater. 2010, 174, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Chen, S.; Li, Y.; Yang, J.; Wei, T.; Jin, S. An Investigation into the Effects of Additives on Crystal Characteristics and Impact Sensitivity of RDX. J. Energ. Mater. 2013, 32, 184–198. [Google Scholar] [CrossRef]
- Lavertu, R.R.S.; Godbout, A.C. Process for Spheroidization of RDX Crystals. U.S. Patent 4,065,529, 27 December 1977. [Google Scholar]
- Sun, H. COMPASS: An ab Initio Force-Field Optimized for Condensed-Phase Applicationss—Overview with Details on Alkane and Benzene Compounds. J. Phys. Chem. B 1998, 102, 7338–7364. [Google Scholar] [CrossRef]
- Pertsin, A.J. The Atom-Atom Potential Method; Springer: Berlin, Germany, 1987. [Google Scholar]
- Zhu, W.; Xiao, J.; Zhu, W.; Xiao, H. Molecular dynamics simulations of RDX and RDX-based plastic-bonded explosives. J. Hazard. Mater. 2009, 164, 1082–1088. [Google Scholar] [CrossRef] [PubMed]
- Borne, L.; Beaucamp, A. Effects of Explosive Crystal Internal Defects on Projectile Impact Initiation. In Proceedings of the 11th International Detonation Symposium, Snowmass Conference Center, Snowmass Village, CO, USA, 31 August–4 September 1998. [Google Scholar]
- Nguyen, A.M.; Nordborg, A.; Shchukarev, A.; Irgum, K. Thermally induced dissolution/precipitation—A simple approach for the preparation of macroporous monoliths from linear aliphatic polyamides. J. Sep. Sci. 2009, 32, 2619–2628. [Google Scholar] [CrossRef] [PubMed]
- Lochert, I.J.; Dexter, R.M.; Hamshere, B.L. Evaluation of Australian RDX in PBXN-109; DSTO-TN-0440; Weapons Systems Division: Canberra, Australia, 2002. [Google Scholar]
- Lochert, I.J.; Franson, M.D.; Hamshere, B.L. Reduced Sensitivity RDX Part I: Literature Review and DSTO Evaluation; Weapons Systems Division: Canberra, Australia, 2003. [Google Scholar]
- Choi, C.S.; Prince, E. The Crystal Structure of Cyelotrimethylene-trinitramine. Acta Cryst. 1972, 28, 2857–2862. [Google Scholar] [CrossRef]
- Hartman, P. The Attachment energy as a habit controlling factor III. application to corundum P. hartman. J. Cryst. Growth 1980, 49, 166–170. [Google Scholar] [CrossRef]
- Hartman, P. The attachment energy as ahabit controlling factor II. Application to anthracene, tin tetraiodide and orthorhombic sulphur P. Hartman. J. Cryst. Growth 1980, 49, 157–165. [Google Scholar] [CrossRef]
- Lu, J.P.; Kennedy, D.L. Modelling of PBXW-115 Using Kinetic Cheetah and the Dyna Codes; DSTO-TR-1496; Defence Science and Technology Organisation Salisbury (Australia) Systems Sciences Lab: Canberra, Australia, 2003. [Google Scholar]
- Hoffman, D.M. Voids and Density Distributions in 2,4,6,8,10,12-Hexanitro-2,4,6,8,10,12-Hexaazaisowurtzitane (CL-20) Prepared Under Various Conditions. Propellants Explos. Pyrotech. 2003, 28, 194–200. [Google Scholar] [CrossRef]
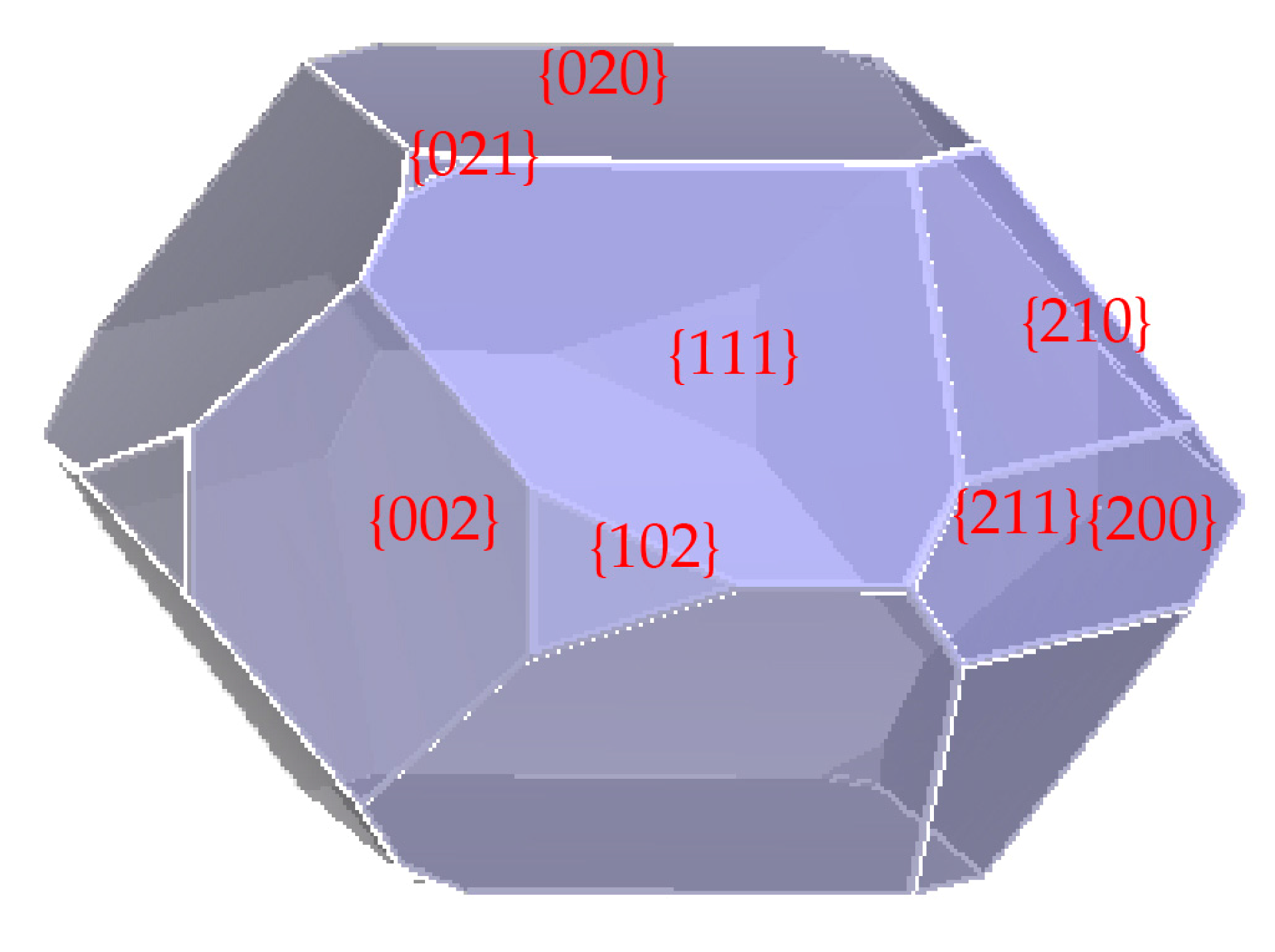

| {hkl} | N 1 | Eatt (kcal·mol−1) | S 2 (nm2) | Area Ratio 3 (%) |
|---|---|---|---|---|
| {111} | 8 | −14.3 | 1103.5 | 52.5 |
| {020} | 2 | −11.9 | 420.8 | 20.0 |
| {210} | 4 | −16.8 | 210.1 | 10.0 |
| {002} | 2 | −14.4 | 200.4 | 9.5 |
| {200} | 2 | −17.8 | 110.4 | 5.2 |
| {102} | 4 | −16.0 | 48.4 | 2.3 |
| {021} | 4 | −15.5 | 6.2 | 0.3 |
| {211} | 8 | −17.5 | 2.6 | 0.1 |
| {hkl} | Eint (kcal·mol−1) | ||||
|---|---|---|---|---|---|
| AC | CH | DMSO | GBL | NMP | |
| {111} | −7.4 | −8.0 | −7.0 | −8.4 | −9.2 |
| {020} | −4.0 | −3.6 | −3.7 | −4.2 | −4.6 |
| {210} | −8.0 | −7.9 | −6.9 | −8.7 | −7.9 |
| {002} | −3.1 | −3.7 | −3.2 | −4.0 | −4.0 |
| {200} | −4.5 | −4.5 | −4.1 | −5.1 | −4.6 |
| {102} | −8.7 | −8.8 | −8.9 | −10.3 | −9.7 |
| {021} | −9.2 | −9.2 | −8.0 | −9.3 | −9.8 |
| {211} | −9.3 | −9.0 | −8.6 | −10.7 | −10.1 |
| {hkl} | {111} | {020} | {210} | {002} | {200} | {102} | {021} | {211} | |
|---|---|---|---|---|---|---|---|---|---|
| (kcal·mol−1) | Vacuum | −14.3 | −11.9 | −16.8 | −14.4 | −17.8 | −16 | −15.5 | −17.5 |
| AC | −6.9 | −7.9 | −8.8 | −11.3 | −13.3 | −7.2 | −6.3 | −8.2 | |
| CH | −6.4 | −8.4 | −8.9 | −10.7 | −13.3 | −7.2 | −6.3 | −8.6 | |
| DMSO | −7.3 | −8.3 | −9.9 | −11.2 | −13.7 | −7.1 | −7.5 | −9 | |
| GBL | −6 | −7.7 | −8.1 | −10.4 | −12.6 | −5.6 | −6.2 | −6.8 | |
| NMP | −5.2 | −7.3 | −8.9 | −10.4 | −13.2 | −6.3 | −5.7 | −7.4 | |
| Solvent | AC | CH | DMSO | GBL | NMP | Vacuum |
|---|---|---|---|---|---|---|
| L/D | 1.62 | 1.67 | 1.62 | 1.61 | 1.93 | 1.66 |
| Parameters | Recrystallized RDX | Conventional RDX | ||||
|---|---|---|---|---|---|---|
| AC | CH | GBL | DMSO | NMP | ||
| Purity (%) | 99.90 | 99.92 | 99.71 | 99.76 | 99.85 | 99.27 |
| ρ (g·cm−3) | 1.813 | 1.811 | 1.806 | 1.803 | 1.809 | 1.796 |
| Experiment Number | Specific Surface Area (m2·g−1) | |
|---|---|---|
| Before | After | |
| 1 | 0.06739 | 0.04585 |
| 2 | 0.07658 | 0.04637 |
| 3 | 0.07269 | 0.04602 |
| Average | 0.07222 | 0.04608 |
| RDX | Impact Sensitivity (%) |
|---|---|
| Conventional RDX | 30 |
| High-quality RDX | 6 |
| Cast Cured PBX | RDX Source | Number of Cards |
|---|---|---|
| PBXN-109-1 | Conventional RDX | 99 |
| PBXN-109-2 | High-quality RDX | 69 |
| PBXW-115-1 | Conventional RDX | 91 |
| PBXW-115-2 | High-quality RDX | 59 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Li, X.; Chen, S.; Ma, X.; Yu, Z.; Jin, S.; Li, L.; Chen, Y. Preparation and Characterization of Cyclotrimethylenetrinitramine (RDX) with Reduced Sensitivity. Materials 2017, 10, 974. https://doi.org/10.3390/ma10080974
Wang Y, Li X, Chen S, Ma X, Yu Z, Jin S, Li L, Chen Y. Preparation and Characterization of Cyclotrimethylenetrinitramine (RDX) with Reduced Sensitivity. Materials. 2017; 10(8):974. https://doi.org/10.3390/ma10080974
Chicago/Turabian StyleWang, Yuqiao, Xin Li, Shusen Chen, Xiao Ma, Ziyang Yu, Shaohua Jin, Lijie Li, and Yu Chen. 2017. "Preparation and Characterization of Cyclotrimethylenetrinitramine (RDX) with Reduced Sensitivity" Materials 10, no. 8: 974. https://doi.org/10.3390/ma10080974
APA StyleWang, Y., Li, X., Chen, S., Ma, X., Yu, Z., Jin, S., Li, L., & Chen, Y. (2017). Preparation and Characterization of Cyclotrimethylenetrinitramine (RDX) with Reduced Sensitivity. Materials, 10(8), 974. https://doi.org/10.3390/ma10080974




