Multifunctional Carbon Aerogels Derived by Sol–Gel Process of Natural Polysaccharides of Different Botanical Origin
Abstract
1. Introduction
2. Materials and Methods
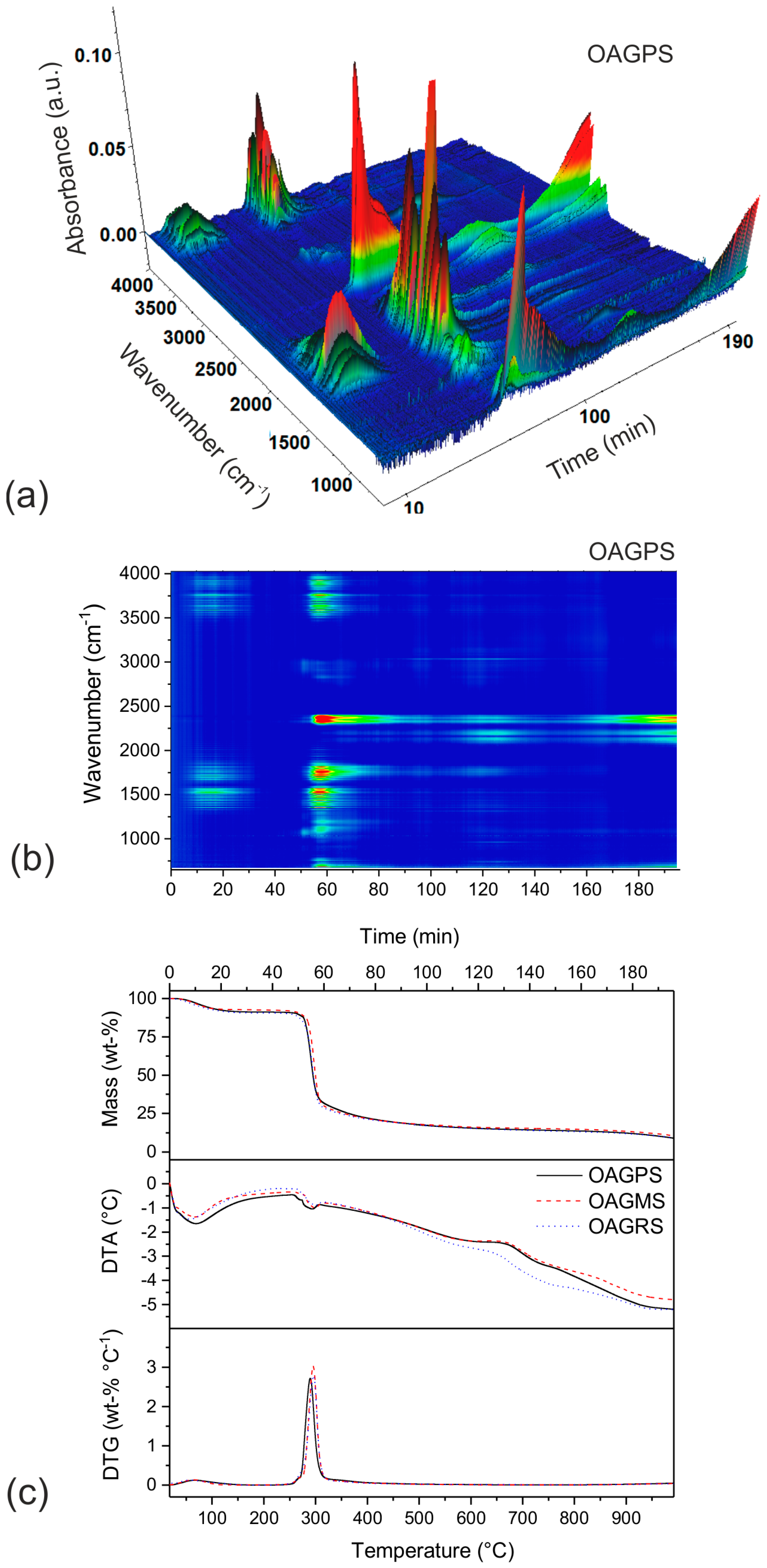
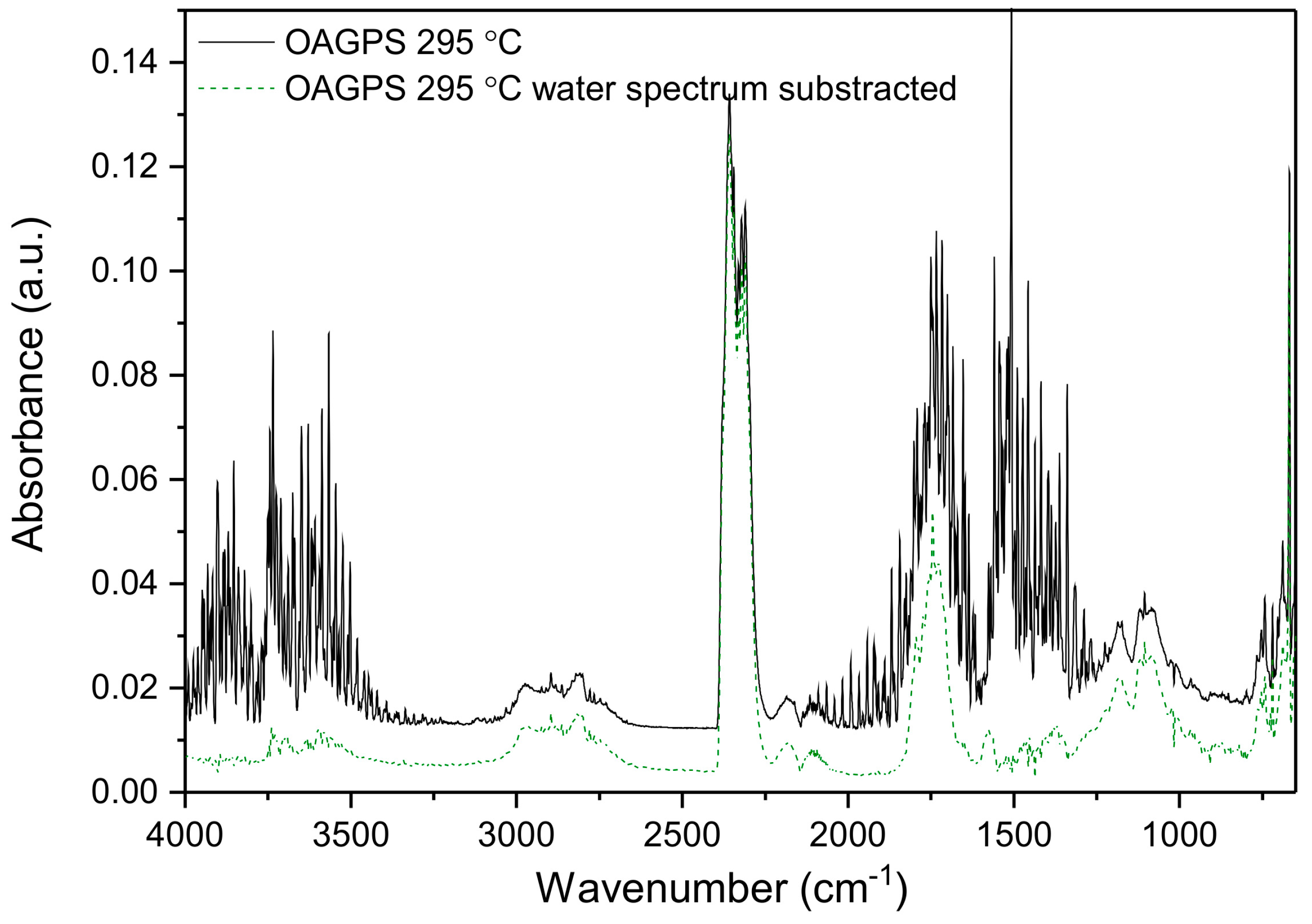
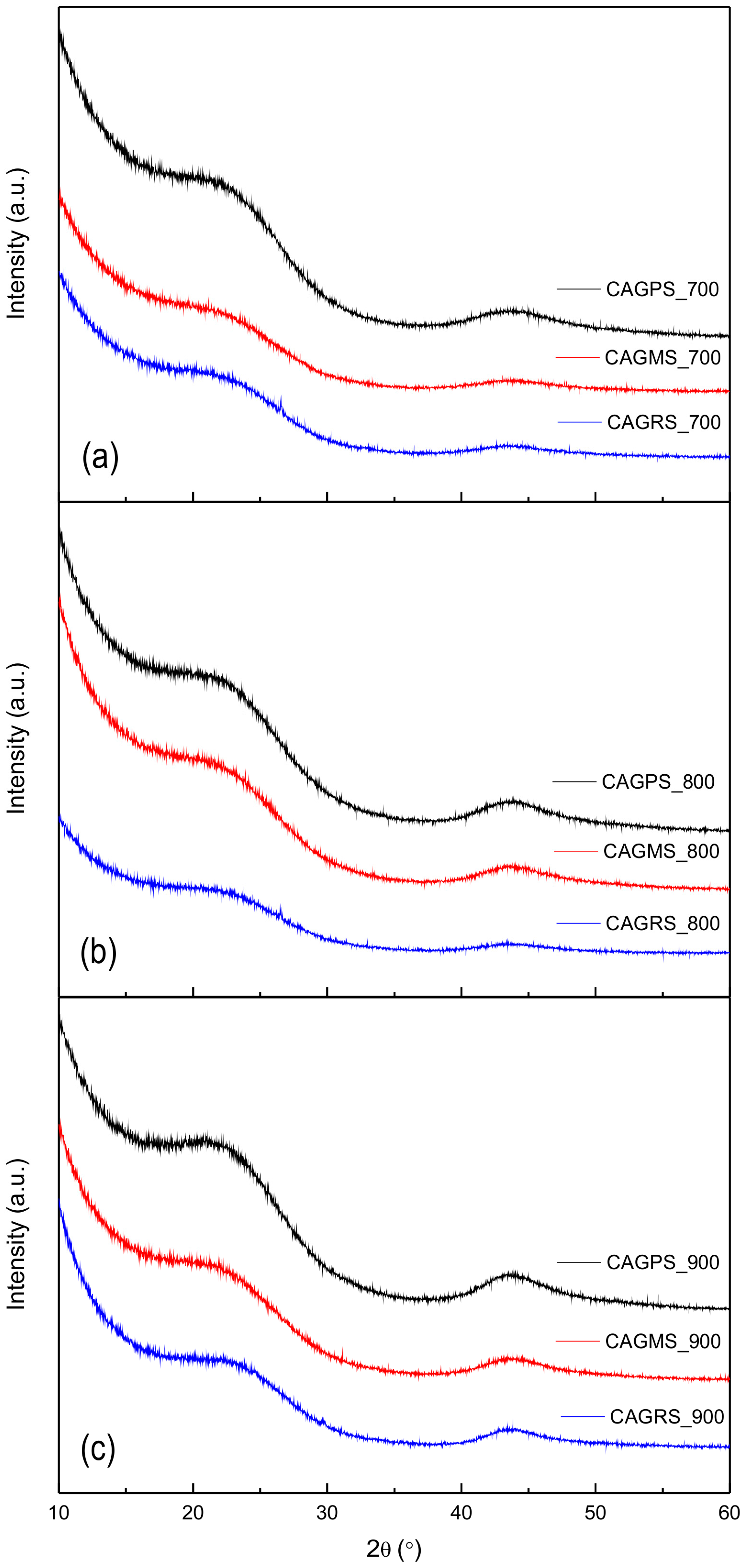
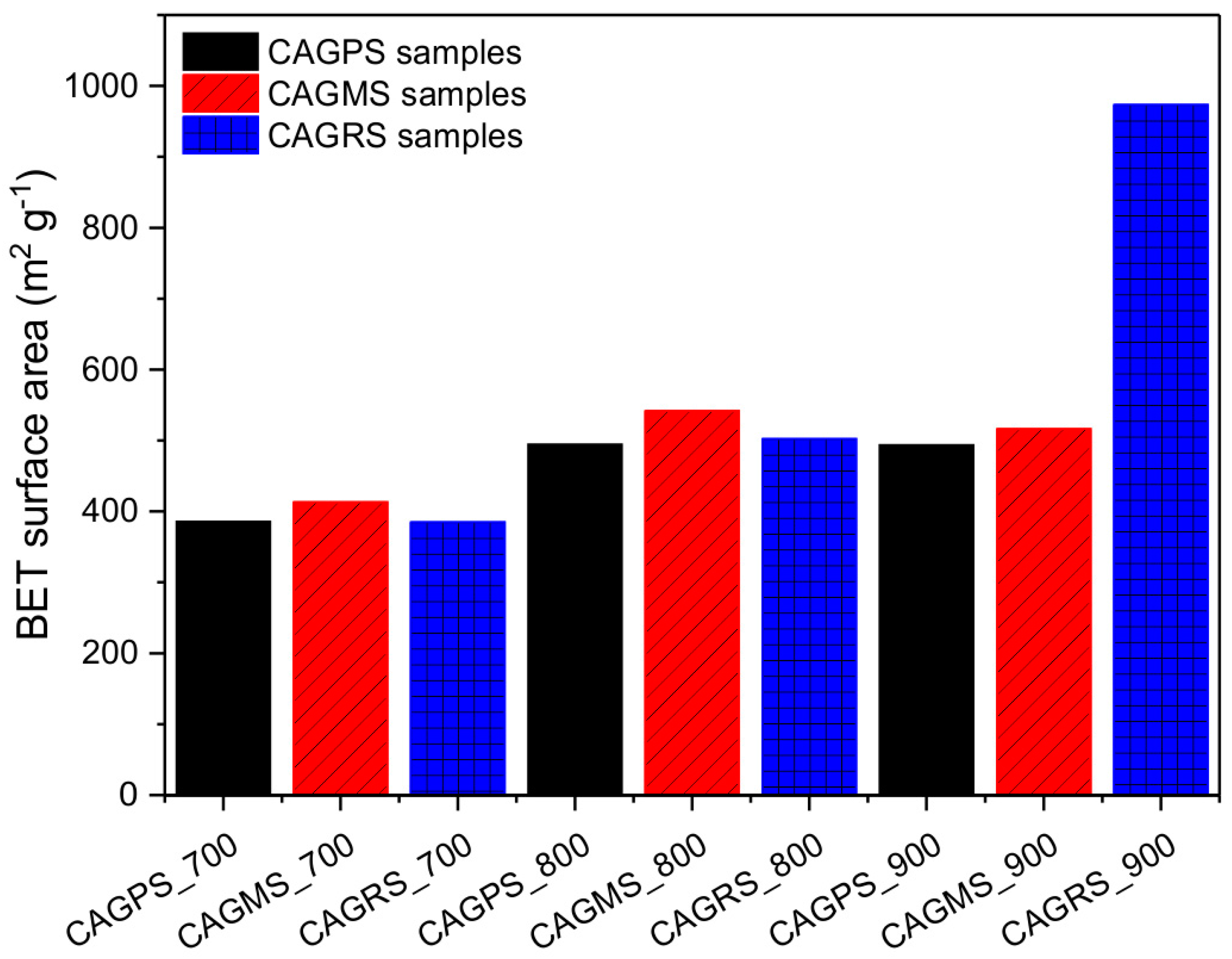
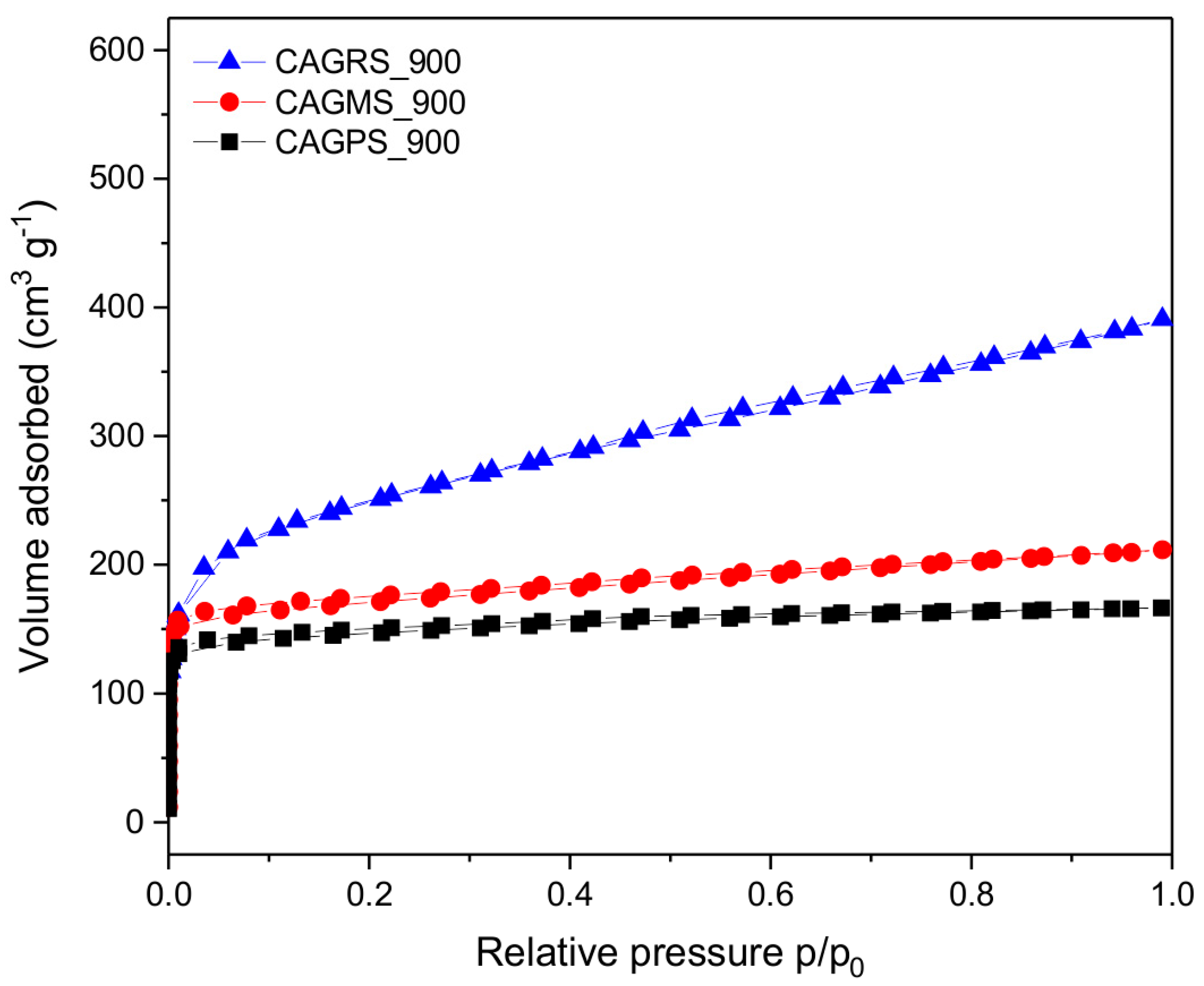
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kistler, S.S. Coherent expanded aerogels and jellies. Nature 1931, 127, 741. [Google Scholar] [CrossRef]
- Kistler, S.S. Coherent expanded aerogels. J. Phys. Chem. 1932, 36, 52–64. [Google Scholar] [CrossRef]
- Pekala, R.W. Low Density, Resorcinol-Formaldehyde Aerogels. U.S. Patent 4873218 A, 26 May 1988. [Google Scholar]
- Shariff, A.M.; Beshir, D.M.; Bustam, M.A.; Maitra, S. Some studies on the synthesis and characterization of carbon aerogel. Trans. Indian Ceram. Soc. 2010, 69, 83–88. [Google Scholar] [CrossRef]
- Lu, X.; Nilsson, O.; Fricke, J.; Pekala, R.W. Thermal and electrical conductivity of monolithic carbon aerogels. J. Appl. Phys. 1993, 73, 581–584. [Google Scholar] [CrossRef]
- Wiener, M.; Reichenauer, G.; Braxmeier, S.; Hemberger, F.; Ebert, H.P. Carbon aerogel-based high-temperature thermal insulation. Int. J. Thermophys. 2009, 30, 1372–1385. [Google Scholar] [CrossRef]
- Feng, J.; Zhang, C.; Feng, J. Carbon fiber reinforced carbon aerogel composites for thermal insulation prepared by soft reinforcement. Mater. Lett. 2012, 67, 266–268. [Google Scholar] [CrossRef]
- Meena, A.K.; Mishra, G.K.; Rai, P.K.; Rajagopal, C.; Nagar, P.N. Removal of heavy metal ions from aqueous solutions using carbon aerogel as an adsorbent. J. Hazard. Mater. 2005, 122, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Kadirvelu, K.; Goel, J.; Rajagopal, C. Sorption of lead, mercury and cadmium ions in multi-component system using carbon aerogel as adsorbent. J. Hazard. Mater. 2008, 153, 502–507. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.Q.; Samad, Y.A.; Polychronopoulou, K.; Alhassan, S.M.; Liao, K. Carbon aerogel from winter melon for highly efficient and recyclable oils and organic solvents absorption. ACS Sustain. Chem. Eng. 2014, 2, 1492–1497. [Google Scholar] [CrossRef]
- Li, J.; Wang, X.; Huang, Q.; Gamboa, S.; Sebastian, P.J. Studies on preparation and performances of carbon aerogel electrodes for the application of supercapacitor. J. Power Sources 2006, 158, 784–788. [Google Scholar] [CrossRef]
- Fang, B.; Binder, L. A modified activated carbon aerogel for high-energy storage in electric double layer capacitors. J. Power Sources 2006, 163, 616–622. [Google Scholar] [CrossRef]
- Mirzaeian, M.; Hall, P.J. Preparation of controlled porosity carbon aerogels for energy storage in rechargeable lithium oxygen batteries. Electrochem. Acta 2009, 54, 7444–7451. [Google Scholar] [CrossRef]
- Biener, J.; Stadermann, M.; Suss, M.; Worsley, M.A.; Biener, M.M.; Rose, K.A.; Baumann, T.F. Advanced carbon aerogels for energy applications. Energy Environ. Sci. 2011, 4, 656–667. [Google Scholar] [CrossRef]
- Moreno-Castilla, C.; Maldonado-Hódar, F.J. Carbon aerogels for catalysis applications: An overview. Carbon 2005, 43, 455–465. [Google Scholar] [CrossRef]
- Smirnova, A.; Dong, X.; Hara, H.; Vasiliev, A.; Sammes, N. Novel carbon aerogel-supported catalysts for PEM fuel cell application. Int. J. Hydrogen Energy 2005, 30, 149–158. [Google Scholar] [CrossRef]
- Du, H.; Li, B.; Kang, F.; Fu, R.; Zeng, Y. Carbon aerogel supported Pt–Ru catalysts for using as the anode of direct methanol fuel cells. Carbon 2007, 45, 429–435. [Google Scholar] [CrossRef]
- García-González, C.A.; Alnaief, M.; Smirnova, I. Polysaccharide-based aerogels—Promising biodegradable carriers for drug delivery systems. Carbohydr. Polym. 2011, 86, 1425–1438. [Google Scholar] [CrossRef]
- Rinki, K.; Dutta, P.K.; Hunt, A.J.; Macquarrie, D.J.; Clark, J.H. Chitosan aerogels exhibiting high surface area for biomedical application: Preparation, characterization, and antibacterial study. Int. J. Polym. Mater. 2011, 60, 988–999. [Google Scholar] [CrossRef]
- Kruss, S.; Hilmer, A.J.; Zhang, J.; Reuel, N.F.; Mu, B.; Strano, M.S. Carbon nanotubes as optical biomedical sensors. Adv. Drug Deliv. Rev. 2013, 65, 1933–1950. [Google Scholar] [CrossRef] [PubMed]
- Pekala, R.W. Organic aerogels from the polycondensation of resorcinol with formaldehyde. J. Mater. Sci. 1989, 24, 3221–3227. [Google Scholar] [CrossRef]
- García-González, C.A.; Camino-Rey, M.C.; Alnaief, M.; Zetzl, C.; Smirnova, I. Supercritical drying of aerogels using CO2: Effect of extraction time on the end material textural properties. J. Supercrit. Fluids 2012, 66, 297–306. [Google Scholar] [CrossRef]
- Tamon, H.; Ishizaka, H.; Yamamoto, T.; Suzuki, T. Influence of freeze-drying conditions on the mesoporosity of organic gels as carbon precursors. Carbon 2000, 38, 1099–1105. [Google Scholar] [CrossRef]
- Chang, X.; Chen, D.; Jiao, X. Starch-derived carbon aerogels with high performance for sorption of cationic dyes. Polymer 2010, 51, 3801–3807. [Google Scholar] [CrossRef]
- Bakierska, M.; Molenda, M.; Majda, D.; Dziembaj, R. Functional starch based carbon aerogels for energy applications. Procedia Eng. 2014, 98, 14–19. [Google Scholar] [CrossRef]
- Zhu, Y.; Hu, H.; Li, W.C.; Zhang, X. Cresol–formaldehyde based carbon aerogel as electrode material for electrochemical capacitor. J. Power Sources 2006, 162, 738–742. [Google Scholar] [CrossRef]
- Pekala, R.W. Melamine-Formaldehyde Aerogels. U.S. Patent 5086085 A, 19 June 1991. [Google Scholar]
- Mulik, S.; Sotiriou-Leventis, C.; Leventis, N. Macroporous electrically conducting carbon networks by pyrolysis of isocyanate-cross-linked resorcinol-formaldehyde aerogels. Chem. Mater. 2008, 20, 6985–6997. [Google Scholar] [CrossRef]
- Yamashita, J.; Ojima, T.; Shioya, M.; Hatori, H.; Yamada, Y. Organic and carbon aerogels derived from poly(vinyl chloride). Carbon 2003, 41, 285–294. [Google Scholar] [CrossRef]
- Wu, D.; Fu, R.; Sun, Z.; Yu, Z. Low-density organic and carbon aerogels from the sol–gel polymerization of phenol with formaldehyde. J. Non-Cryst. Solids 2005, 351, 915–921. [Google Scholar] [CrossRef]
- Wu, D.; Fu, R.; Zhang, S.; Dresselhaus, M.S.; Dresselhaus, G. The preparation of carbon aerogels based upon the gelation of resorcinol–furfural in isopropanol with organic base catalyst. J. Non-Cryst. Solids 2004, 336, 26–31. [Google Scholar] [CrossRef]
- BeMiller, J.; Whistler, R. Starch: Chemistry and Technology, 3rd ed.; Elsevier: New York, NY, USA, 2009; ISBN 978-0-12-746275-2. [Google Scholar]
- Zhai, Y.; Peng, W.; Zeng, G.; Fu, Z.; Lan, Y.; Chen, H.; Wang, C.; Fan, X. Pyrolysis characteristics and kinetics of sewage sludge for different sizes and heating rates. J. Therm. Anal. Calorim. 2012, 107, 1015–1022. [Google Scholar] [CrossRef]
- Yu, L.; Falco, C.; Weber, J.; White, R.J.; Howe, J.Y.; Titirici, M.M. Carbohydrate-derived hydrothermal carbons: A thorough characterization study. Langmuir 2012, 28, 12373–12383. [Google Scholar] [CrossRef] [PubMed]
- Worzakowska, M.; Torres-Garcia, E.; Grochowicz, M. Degradation kinetics of starch-g-poly(phenyl methacrylate) copolymers. Thermochim. Acta 2015, 619, 8–15. [Google Scholar] [CrossRef]
- Tang, X.; Savut Jan, S.; Qian, Y.; Xia, H.; Ni, J.; Savilov, S.V.; Aldoshin, S.M. Graphene wrapped ordered LiNi0.5Mn1.5O4 nanorods as promising cathode material for lithium-ion batteries. Sci. Rep. 2015, 7, 11958. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Su, D.; Park, J.; Ahn, H.; Wang, G. Graphene-supported SnO2 nanoparticles prepared by a solvothermal approach for an enhanced electrochemical performance in lithium-ion batteries. Nanoscale Res. Lett. 2012, 7, 215. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Huang, F.; Zhao, W.; Liu, Z.; Wan, D. Synthesis of graphene-supported Li4Ti5O12 nanosheets for high rate battery application. J. Mater. Chem. 2012, 22, 11257–11260. [Google Scholar] [CrossRef]
- Wallace, P.R. The Band Theory of Graphite. Phys. Rev. 1947, 71, 622–634. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Wei, L.; Sevilla, M.; Fuertes, A.B.; Mokaya, R.; Yushin, G. Hydrothermal carbonization of abundant renewable natural organic chemicals for high-performance supercapacitor electrodes. Adv. Energy Mater. 2011, 1, 356–361. [Google Scholar] [CrossRef]
- Zhao, S.; Wang, C.Y.; Chen, M.M.; Wang, J.; Shi, Z.Q. Potato starch-based activated carbon spheres as electrode material for electrochemical capacitor. J. Phys. Chem. Solids 2009, 70, 1256–1260. [Google Scholar] [CrossRef]

| Sample | Starch Type | Carbon Yield (wt %) | ||
|---|---|---|---|---|
| 700 °C | 800 °C | 900 °C | ||
| OAGPS | potato starch | 16.1 | 15.2 | 13.9 |
| OAGMS | maize starch | 17.1 | 16.2 | 14.8 |
| OAGRS | rice starch | 15.8 | 14.6 | 13.3 |
| Sample | C (wt %) | H (wt %) | N (wt %) | O (wt %) |
|---|---|---|---|---|
| CAGPS_700 | 91.8 | 1.7 | 0.4 | 6.1 |
| CAGPS_800 | 92.1 | 1.4 | 0.4 | 6.1 |
| CAGPS_900 | 92.5 | 1.0 | 0.6 | 5.9 |
| CAGMS_700 | 91.8 | 1.7 | 0.3 | 6.2 |
| CAGMS_800 | 90.9 | 1.3 | 0.6 | 7.2 |
| CAGMS_900 | 92.3 | 1.1 | 0.3 | 7.3 |
| CAGRS_700 | 86.8 | 1.9 | 2.0 | 9.3 |
| CAGRS_800 | 88.6 | 1.5 | 1.3 | 8.6 |
| CAGRS_900 | 93.4 | 1.1 | 0.4 | 5.1 |
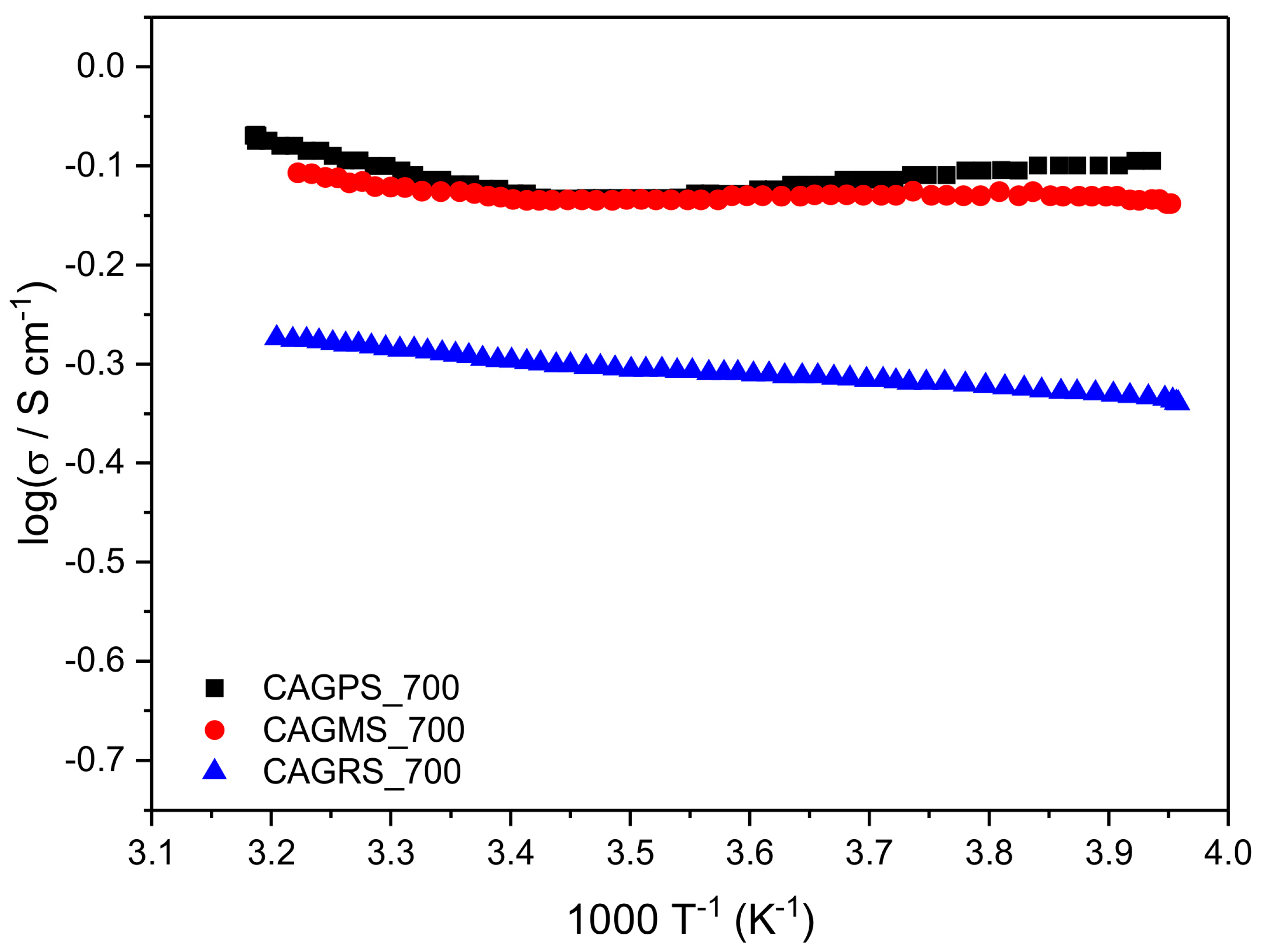
| Sample | Ea (eV) | σ at ~25 °C (S∙cm−1) |
|---|---|---|
| CAGPS_700 | 0.007 | 0.833 |
| CAGMS_700 | 0.004 | 0.751 |
| CAGRS_700 | 0.015 | 0.513 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bakierska, M.; Chojnacka, A.; Świętosławski, M.; Natkański, P.; Gajewska, M.; Rutkowska, M.; Molenda, M. Multifunctional Carbon Aerogels Derived by Sol–Gel Process of Natural Polysaccharides of Different Botanical Origin. Materials 2017, 10, 1336. https://doi.org/10.3390/ma10111336
Bakierska M, Chojnacka A, Świętosławski M, Natkański P, Gajewska M, Rutkowska M, Molenda M. Multifunctional Carbon Aerogels Derived by Sol–Gel Process of Natural Polysaccharides of Different Botanical Origin. Materials. 2017; 10(11):1336. https://doi.org/10.3390/ma10111336
Chicago/Turabian StyleBakierska, Monika, Agnieszka Chojnacka, Michał Świętosławski, Piotr Natkański, Marta Gajewska, Małgorzata Rutkowska, and Marcin Molenda. 2017. "Multifunctional Carbon Aerogels Derived by Sol–Gel Process of Natural Polysaccharides of Different Botanical Origin" Materials 10, no. 11: 1336. https://doi.org/10.3390/ma10111336
APA StyleBakierska, M., Chojnacka, A., Świętosławski, M., Natkański, P., Gajewska, M., Rutkowska, M., & Molenda, M. (2017). Multifunctional Carbon Aerogels Derived by Sol–Gel Process of Natural Polysaccharides of Different Botanical Origin. Materials, 10(11), 1336. https://doi.org/10.3390/ma10111336







