Rollover Prevention Model for Stratified Liquefied Natural Gas in Storage Tanks
Abstract
1. Introduction
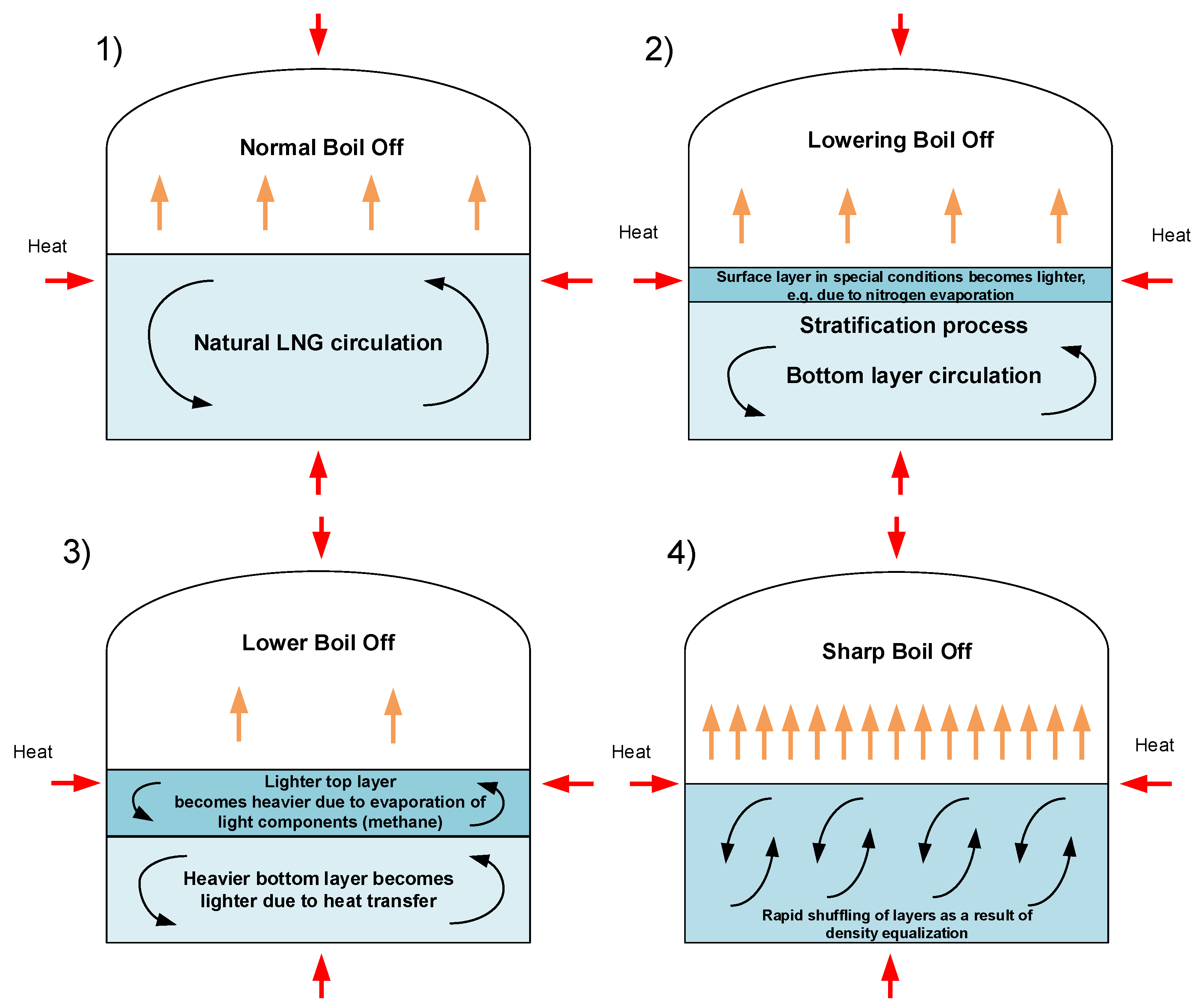
2. LNG Properties and Stratification Causes
3. LNG Stratification and Rollover
4. Analytical Prevention Model—Stable Stratification Model (SSM)
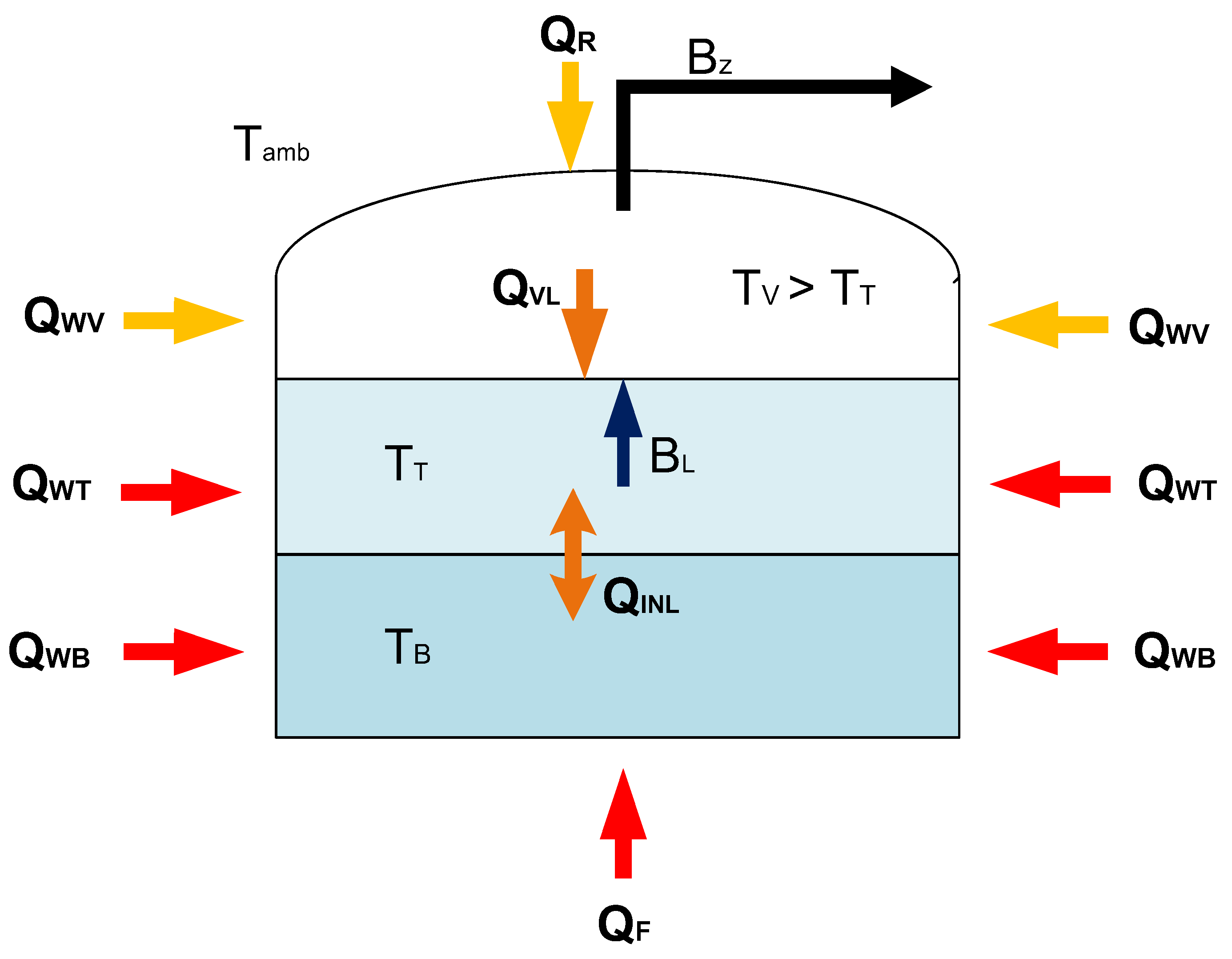
4.1. Physical Description
4.2. Model Algorithm
5. Assumptions
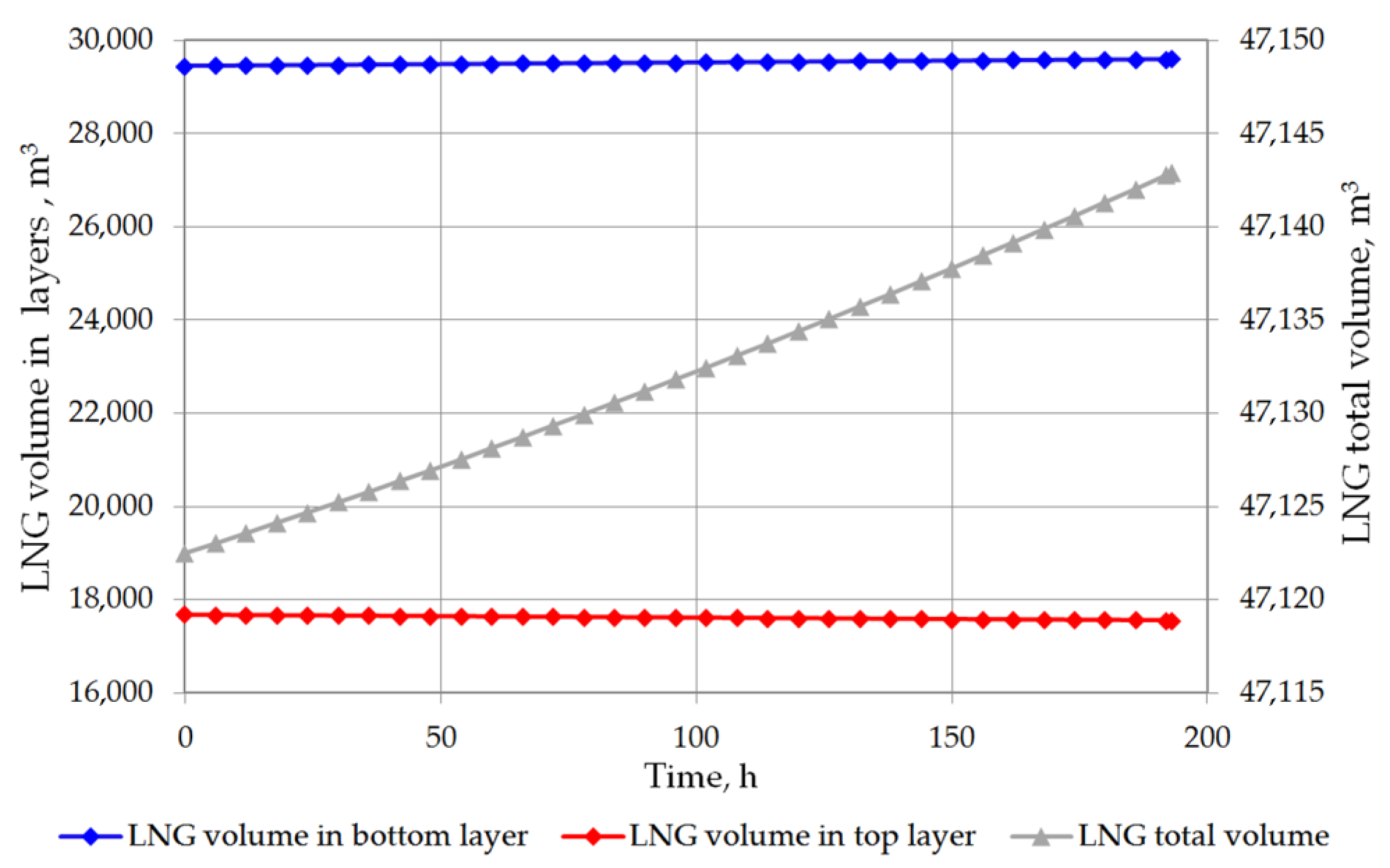
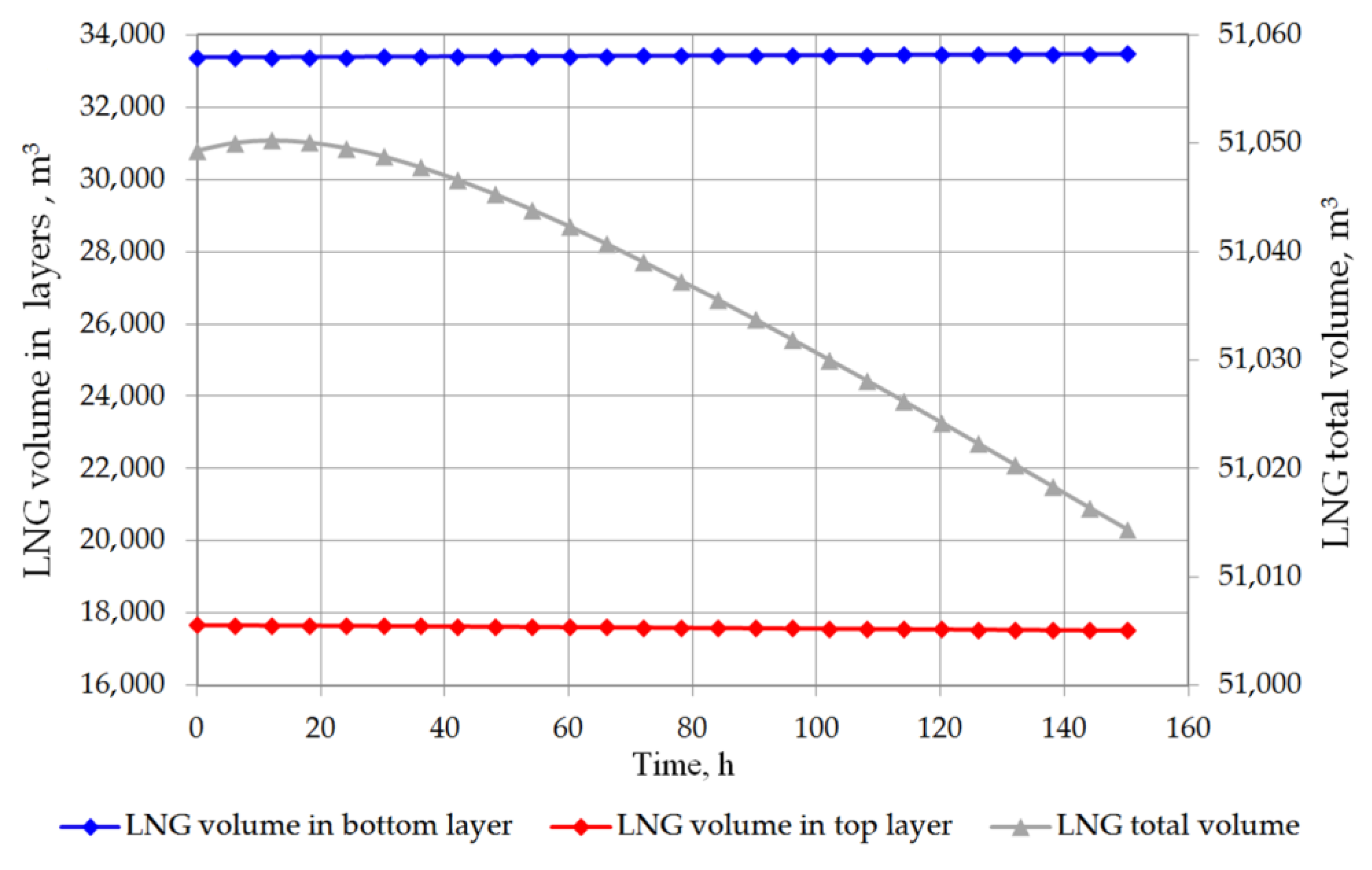
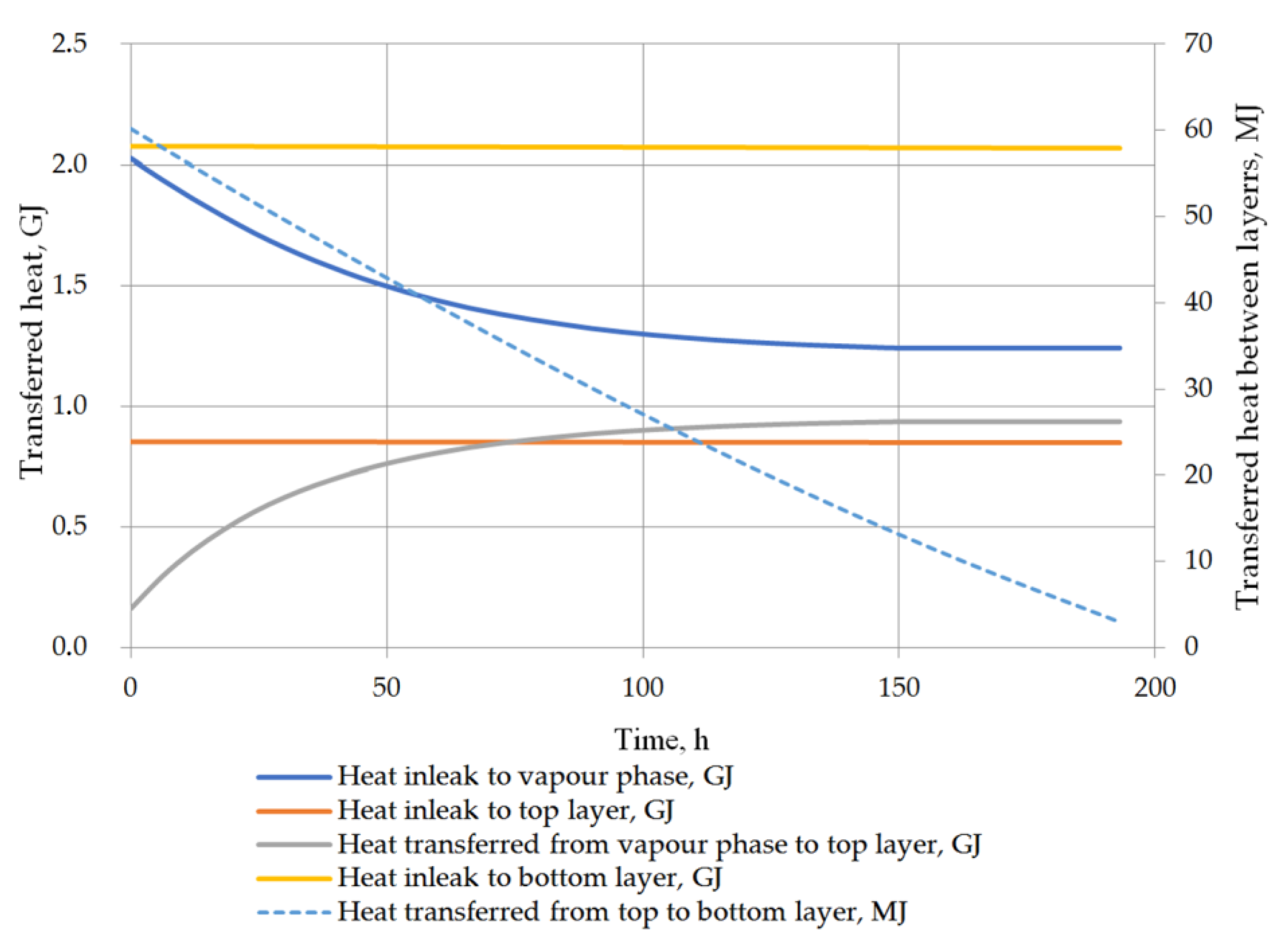
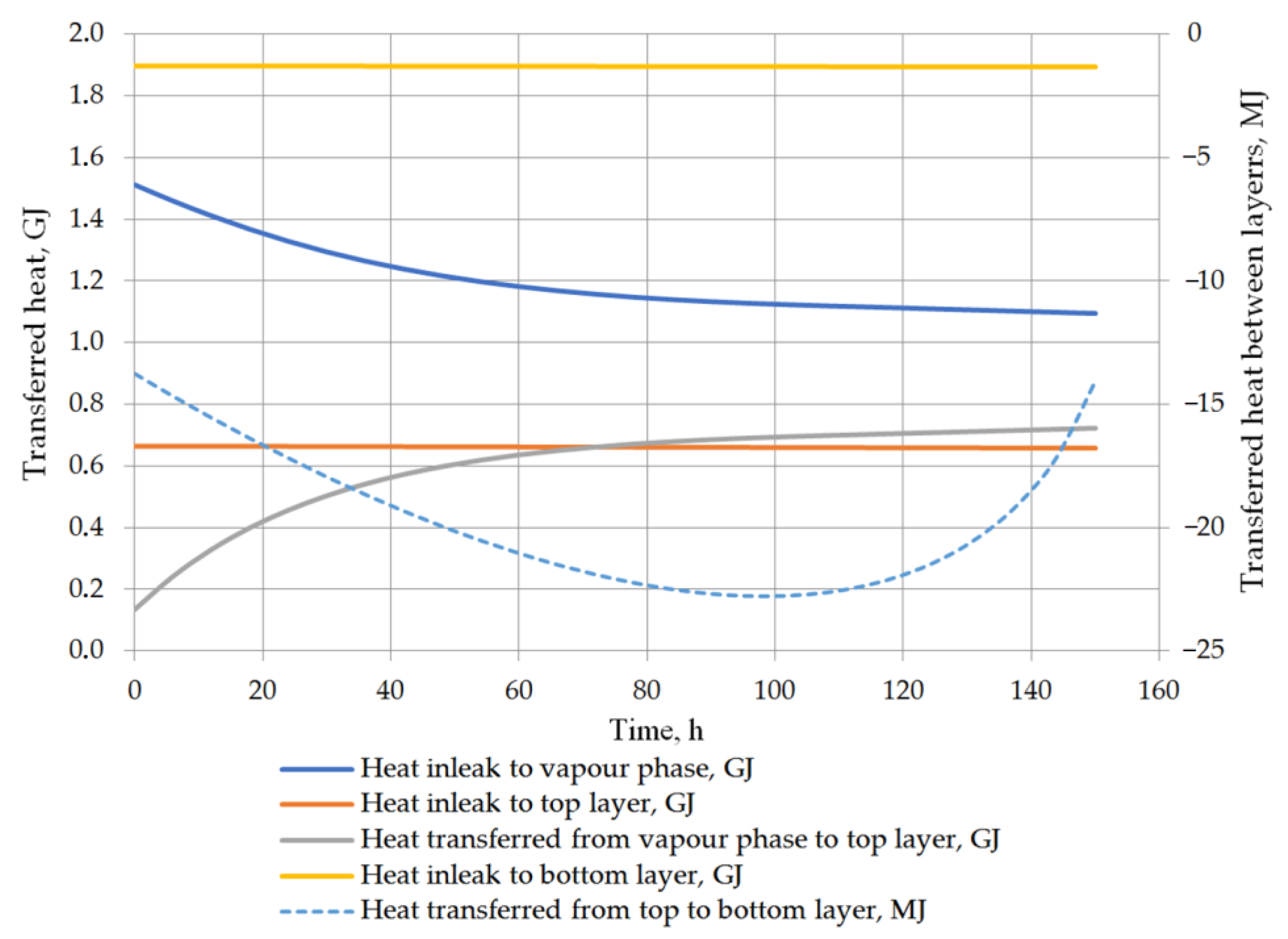
6. Results and Discussion
- (1)
- densities of top and bottom layers;
- (2)
- temperatures of the bottom layer and the top layer of LNG;
- (3)
- vapor phase temperature;
- (4)
- boil off rate (BOR);
- (5)
- thicknesses of bottom and top layers;
- (6)
- heat transferred into storage tank and between phases and layers in tank.
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Nomenclature
| A | area of heat transfer, m2 |
| BL | liquid vaporized (boil off gas), moles |
| B | vapor vented from tank, moles |
| Cp | isobaric heat capacity, J/mole |
| f | fugacity, Pa |
| H | enthalpy, J |
| h | molar enthalpy, J/mole |
| k | thermal conductivity, W/(m·K) |
| N | total moles, moles |
| Q | heat transferred, J |
| T | temperature, K |
| U | heat transfer coefficient, W/(m2·K) |
| z | height, m |
| Greek symbols | |
| α | thermal diffusivity, m2/s |
| ν | kinematic viscosity, m2/s |
| ρ | density, kg/m3 |
| φ | fugacity coefficient |
| Subscripts | |
| amb | ambient |
| av | average |
| B | liquid bottom layer |
| F | y tank bottom slab (foundation) |
| i | initial, beginning of time step |
| i + 1 | end of time step, initial of next step |
| in | inleak |
| INL | interlayer |
| R | by tank roof |
| T | liquid top layer |
| V | vapor phase (vapor layer) |
| VL | vapor to liquid |
| VZ | vapor phase at z height |
| W | by tank side wall |
| Acronyms | |
| LNG | Liquefied natural gas |
| BOR | Boil off rate |
| SSM | Stable stratification model |
References
- International Gas Union. World LNG Report—2015 Edition; C/O Statoil ASA; International Gas Union, Office of the Secretary General: Oslo, Norway, 2015. [Google Scholar]
- GIIGNL. LNG Custody Transfer Handbook, 4th ed.; (Version 4); GIIGNL: Paris, France, 2015; p. 104. [Google Scholar]
- Sarsten, J.A. LNG stratification and rollover. Pipeline Gas J. 1972, 199, 37–42. [Google Scholar]
- Baker, N.; Creed, M. Stratification and rollover in liquefied natural gas storage tanks. Trans. IChemE 74 (Part B) 1996, 74 Pt B, 25–30. [Google Scholar]
- Hubert, A.; Dembele, S.; Denissenko, P.; Wen, J. Predicting Liquefied Natural Gas (LNG) rollovers using Computational Fluid Dynamics. J. Loss Prev. Process Ind. 2019, 62, 103922. [Google Scholar] [CrossRef]
- Kulitsa, M.; Wood, D.A. LNG rollover challenges and their mitigation on Floating Storage and Regasification Units: New perspectives in assessing rollover consequences. J. Loss Prev. Process Ind. 2018, 54, 352–372. [Google Scholar] [CrossRef]
- Nakano, R.; Sugawara, Y.; Yamagata, S.; Nakamura, J. An experimental study on the mixing of stratified layers using liquid freon. In Proceedings of the Gastech 82 LNG-LPG Conference, Paris, France, 5–8 October 1982; pp. 383–391. [Google Scholar]
- Morioka, M.; Enya, S. Natural convection of density-stratified layers in a vessel. Heat Transf. Jpn. Res. 1984, 12, 48–69. [Google Scholar]
- Gorieu, O.; Uznanski, D.; DuPont, P. How to operate LNG terminals with flexibility/ safety despite the diversification of unloaded LNG qualities. In Proceedings of the 14th International Conference and Exhibition of Liquefied Natural Gas, Doha, Qatar, 21–24 March 2004; pp. 1–10. [Google Scholar]
- Chatterjee, N.; Geist, J.M. The effects of stratification on boil-off rate in LNG tanks. Pipeline Gas J. 1972, 99, 40–45. [Google Scholar]
- Germeles, A.E. A model for LNG tank Rollover. Adv. Cryog. Eng. 1975, 21, 326–336. [Google Scholar]
- Heestand, J.; Shipman, C.W.; Meader, J.W. A predictive model for rollover in stratified LNG tanks. AIChE J. 1983, 29, 199–207. [Google Scholar] [CrossRef]
- Bates, S.; Morrison, D. Modelling the behaviour of stratified liquid natural gas in storage tanks: A study of the rollover phenomenon. Int. J. Heat Mass Transf. 1997, 40, 1875–1884. [Google Scholar] [CrossRef]
- Deshpande, K.B.; Zimmerman, W.B.; Tennant, M.T.; Webster, M.B.; Lukaszewski, M.W. Optimization methods for the real-time inverse problem posed by modelling of liquefied natural gas storage. Chem. Eng. J. 2011, 170, 44–52. [Google Scholar] [CrossRef][Green Version]
- Kamiya, A.; Tahsita, M.; Sugawara, Y. An Experimental Study of LNG Rollover Phenomenon; American Society of Mechanical Engineers: New York, NY, USA, 1985; pp. 1–11. [Google Scholar]
- Agbabi, T. Rollover and Interfacial Studies in LNG Mixtures. Institute of Cryogenics. Ph.D. Thesis, University of Southampton, Southampton, UK, 1987; p. 187. [Google Scholar]
- Morrison, D.S.; Richardson, A. An experimental study on the stability of stratified layers and rollover in LNG storage tanks. In Proceedings of the Low Temperature Engineering and Cryogenics Conference, Southampton, UK, 13–15 July 1990. [Google Scholar]
- Gursu, S.; Sherif, S.A.; Veziroglu, T.N.; Sheffield, J.W. Analysis and Optimization of Thermal Stratification and Self-Pressurization Effects in Liquid Hydrogen Storage Systems—Part 1: Model Development. J. Energy Resour. Technol. 1993, 115, 221–227. [Google Scholar] [CrossRef]
- Shi, J.; Beduz, C.; Scurlock, R. Numerical modelling and flow visualization of mixing of stratified layers and rollover in LNG. Cryogenics 1993, 33, 1116–1124. [Google Scholar] [CrossRef]
- Munakata, T.; Lior, N. A study of double diffusive rollover in cylindrical enclosures. Proc. ASME Heat Tran. Div. 1995, 317, 279–291. [Google Scholar]
- Tanasawa, I. Experimental techniques in natural convection. Exp. Therm. Fluid Sci. 1995, 10, 503–518. [Google Scholar] [CrossRef]
- Sheats, D.; Capers, M. Density stratification in LNG storage. Cold Facts 1999, 15, 3. [Google Scholar]
- SIGTTO. Guidance for the Prevention of Rollover in LNG Ships, 1st ed.; SIGTTO: London, UK, 2012. [Google Scholar]
- Scurlock, R.G. Stratification, Rollover and Handling of LNG, LPG and Others Cryogenic Mixtures; Springer International Publishing: Berlin/Heidelberg, Germany, 2016; 134p, ISBN 978-3-319-20695-0. [Google Scholar]
- Arjomandnia, P. Simulation of Rollover in Stratified LNG Storage Tanks. Master’s Thesis, University of Western Australia, Perth, Australia, 2010. [Google Scholar]
- Arjomandnia, P.; Tade, M.O.; Pareek, V.; May, E.F. Analysis of available data from liquefied natural gas rollover incidents to determine critical stability ratios. AIChE J. 2013, 60, 362–374. [Google Scholar] [CrossRef]
- Zimmerman, W.B.; Rees, J.M. Rollover instability due to double diffusion in a stably stratified cylindrical tank. Phys. Fluids 2007, 19, 123604. [Google Scholar] [CrossRef]
- Kim, H.; Shin, M.-W.; Yoon, E.S. Optimization of operating procedure of LNG storage facilities using rigorous BOR model. In Proceedings of the 17th World Congress the International Federation of Automatic Control, Seoul, Republic of Korea, 6–11 July 2008; pp. 10923–10926. [Google Scholar]
- Ludwig, C.; Dreyer, M.; Hopfinger, E. Pressure variations in a cryogenic liquid storage tank subjected to periodic excitations. Int. J. Heat Mass Transf. 2013, 66, 223–234. [Google Scholar] [CrossRef]
- Ren, J.-J.; Shi, J.-Y.; Liu, P.; Bi, M.-S.; Jia, K. Simulation on thermal stratification and de-stratification in liquefied gas tanks. Int. J. Hydrog. Energy 2013, 38, 4017–4023. [Google Scholar] [CrossRef]
- Shi, J.; Ren, J.; Liu, P.; Bi, M. Experimental research on the effects of fluid and heater on thermal stratification of liquefied gas. Exp. Therm. Fluid Sci. 2013, 50, 29–36. [Google Scholar] [CrossRef]
- Migliore, C.; Salehi, A.; Vesovic, V. A non-equilibrium approach to modelling the weathering of stored Liquefied Natural Gas (LNG). Energy 2017, 124, 684–692. [Google Scholar] [CrossRef]
- Migliore, C.; Salehi, A.; Vesovic, V. Weathering of stored Liquefied Natural Gas (LNG). In Proceedings of the 10th International Conference on Thermal Engineering: Theory and Applications, Muscat, Oman, 26–28 February 2017. [Google Scholar]
- Migliore, C.; Tubilleja, C.; Vesovic, V. Weathering prediction model for stored liquefied natural gas (LNG). J. Nat. Gas Sci. Eng. 2015, 26, 570–580. [Google Scholar] [CrossRef]
- Pellegrini, L.A.; Moioli, S.; Brignoli, F.; Bellini, C. LNG Technology: The Weathering in Above-Ground Storage Tanks. Ind. Eng. Chem. Res. 2014, 53, 3931–3937. [Google Scholar] [CrossRef]
- Zakaria, Z.; Baslasl, M.S.O.; Samsuri, A.; Ismail, I.; Supee, A.; Haladin, N.B. Rollover Phenomenon in Liquefied Natural Gas Storage Tank. J. Fail. Anal. Prev. 2019, 19, 1439–1447. [Google Scholar] [CrossRef]
- Lu, Y.; Liang, C. Rollover of Liquid Natural Gas in a Storage Tank: A Numerical Simulation. In Proceedings of the 2nd International Conference on Green Energy, Environment and Sustainable Development (GEESD2021), Shanghai, China, 26–27 June 2021; Dobrota, D., Cheng, C., Eds.; IOS Press: Amsterdam, The Netherlands, 2021. [Google Scholar]
- Wang, Z.; Han, F.; Liu, Y.; Li, W. Evolution Process of Liquefied Natural Gas from Stratification to Rollover in Tanks of Coastal Engineering with the Influence of Baffle Structure. J. Mar. Sci. Eng. 2021, 9, 95. [Google Scholar] [CrossRef]
- Zhao, B.; Han, S.; Xu, L.; Shi, C.; Gao, D.; Zhang, Y.D. Rollover Mechanism Methodology of LNG Tank with Gas-Liquid Stratification Based on Curvelet Finite Element Method and Large Eddy Simulation Technology. J. Appl. Fluid Mech. 2018, 11, 779–786. [Google Scholar] [CrossRef]
- Saleem, A.; Farooq, S.; Karimi, I.A.; Banerjee, R. CFD Analysis of Stratification and Rollover Phenomena in an Industrial-Scale LNG Storage Tank. Ind. Eng. Chem. Res. 2020, 59, 14126–14144. [Google Scholar] [CrossRef]
- Hubert, A.; Dembele, S.; Denissenko, P.; Wen, J. Numerical Predictions of the Rollover Phenomenon Using Computational Fluid Dynamics; Symposum Series No. 162; IChemE: Rugby, UK, 2017. [Google Scholar]
- Yumeng, L.; Jinshu, L.; Junjie, S. Effect of Rolling on the Stratification and Rollover Phenomenon of LNG Cargo Tank, ISOPE-I-21-3202. In Proceedings of the 31st International Ocean and Polar Engineering Conference, Rhodes, Greece, 26 June–1 July 2021. [Google Scholar]
- Huang, B.; Li, J.; Fu, C.; Guo, T.; Feng, S. A Comprehensive Review of Stratification and Rollover Behavior of Liquefied Natural Gas in Storage Tanks. Processes 2022, 10, 1360. [Google Scholar] [CrossRef]
- International Group of LNG Exporters (GIIGNL). Annual Report 2018; International Group of LNG Exporters: Paris, France, 2018. [Google Scholar]
- Miana, M.; del Hoyo, R.; Rodrigálvarez, V.; Valdés, J.R.; Llorens, R. Calculation models of liquefied natural gas (LNG) ageing during ship transportation. Appl. Energy 2010, 87, 1687–1700. [Google Scholar] [CrossRef]
- Miana, M.; del Hoyo, R.; Rodrigálvarez, V. Comparison of evaporation rate and heat flow models for prediction of Liquefied Natural Gas (LNG) ageing during ship transportation. Fuel 2016, 177, 87–106. [Google Scholar] [CrossRef]
- Kulitsa, M.; Wood, D.A. Floating storage and regasification units face specific LNG rollover challenges: Consideration of saturated vapor pressure provides insight and mitigation options. Nat. Gas Ind. B 2018, 5, 391–414. [Google Scholar]
- Łaciak, M. Liquefied Natural Gas storage of variable composition. Arch. Min. Sci. 2015, 60, 225–238. [Google Scholar]
- Łaciak, M. Thermodynamic processes involving liquefied natural gas at the receiving terminals. Arch. Min. Sci. 2013, 58, 349–359. [Google Scholar]
- Bashiri, A. Modelling and simulation of Rollover in LNG Storage Tanks. In Proceedings of the 23rd World Gas Conference, Amsterdam, The Netherlands, 5–9 June 2006. [Google Scholar]
- Bird, R.; Warren, S.; Lightfoot, E. Transport Phenomena, 2nd ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2007. [Google Scholar]
- Huerta, F.; Vesovic, V. A realistic vapour phase heat transfer model for the weathering of LNG stored in large tanks. Energy 2019, 174, 280–291. [Google Scholar] [CrossRef]




| Composition | Methane | Ethane | Propane | C4+ | Nitrogen |
|---|---|---|---|---|---|
| Source | %mole | %mole | %mole | %mole | %mole |
| Australia NWS | 87.33 | 8.33 | 3.33 | 0.97 | 0.04 |
| Australia Darwin | 87.64 | 9.97 | 1.96 | 0.33 | 0.10 |
| Algieria–Skikda | 91.40 | 7.35 | 0.57 | 0.05 | 0.63 |
| Brunei | 90.12 | 5.34 | 3.02 | 1.48 | 0.04 |
| Egypt–Idku | 95.31 | 3.58 | 0.74 | 0.35 | 0.02 |
| Egypt–Damietta | 97.25 | 2.49 | 0.12 | 0.12 | 0.02 |
| Indonesia–Badak | 90.14 | 5.46 | 2.98 | 1.41 | 0.01 |
| Libya | 81.39 | 12.44 | 3.51 | 0.64 | 2.02 |
| Malaysia–Bintulu | 91.69 | 4.64 | 2.60 | 0.93 | 0.14 |
| Nigeria | 91.70 | 5.52 | 2.17 | 0.58 | 0.03 |
| Norway | 92.03 | 5.75 | 1.31 | 0.45 | 0.46 |
| Oman | 90.68 | 5.75 | 2.12 | 1.25 | 0.20 |
| Peru | 89.06 | 10.26 | 0.10 | 0.01 | 0.57 |
| Qatar | 90.91 | 6.43 | 1.66 | 0.73 | 0.27 |
| Russia–Sakhalin | 92.54 | 4.47 | 1.97 | 0.95 | 0.07 |
| Trynidad | 96.78 | 2.78 | 0.37 | 0.06 | 0.01 |
| USA–Alaska | 99.70 | 0.09 | 0.03 | 0.01 | 0.17 |
| Yemen | 93.17 | 5.92 | 0.77 | 0.12 | 0.02 |
| Parameter | Value | Unit |
|---|---|---|
| Inner tank diameter | 50 | m |
| Inner tank wall thickness | 40 | mm |
| Primary thermal insulation thickness | 800 | mm |
| Secondary thermal insulation thickness | 400 | mm |
| Outer concrete wall thickness | 500 | mm |
| Foundation plate thickness | 1200 | mm |
| Foundation thermal insulation thickness | 1000 | mm |
| Thickness of suspended roof | 40 | mm |
| Thickness of roof insulation | 500 | mm |
| Thermal conductivity of inner tank wall | 90 | W/(m·K) |
| Thermal conductivity of primary insulation | 0.05 | W/(m·K) |
| Thermal conductivity of secondary insulation | 0.1 | W/(m·K) |
| Thermal conductivity of concrete wall and foundation plate | 1.8 | W/(m·K) |
| Thermal conductivity of foundation plate insulation | 0.1 | W/(m·K) |
| Tank height | 43 | m |
| First Variant (1) | Second Variant (2) | |
|---|---|---|
| Ambient air temperature | 293 K | 293 K |
| Ground temperature | 285 K | 285 K |
| Initial temperature in bottom layer | 116.12 K | 115 K |
| Tank operational pressure | 1.5 bar(a) | 1.3 bar(a) |
| Initial density of top layer | 451.19 kg/m3 | 434.01 kg/m3 |
| Initial density of bottom layer | 452.68 kg/m3 | 435.52 kg/m3 |
| Initial level of LNG in tank | 24 m | 26 m |
| Initial thickness of top layer | 9 m | 9 m |
| Composition of top layer | ||
| methane | 90.0% | 95.5% |
| ethane | 6.7% | 3.2% |
| propane | 2.3% | 0.9% |
| n-butane | 0.5% | 0.0% |
| iso-butane | 0.5% | 0.3% |
| nitrogen | 0.0% | 0.1% |
| Composition of bottom layer | ||
| methane | 90.1% | 94.8% |
| ethane | 6.4% | 3.3% |
| propane | 2.1% | 1.0% |
| n-butane | 0.4% | 0.2% |
| iso-butane | 0.5% | 0.1% |
| nitrogen | 0.5% | 0.6% |
| Time Step | t = 0 h | t =30 h | t = 60 h | t = 90 h | t = 120 h | t = 150 h | t = 180 h |
|---|---|---|---|---|---|---|---|
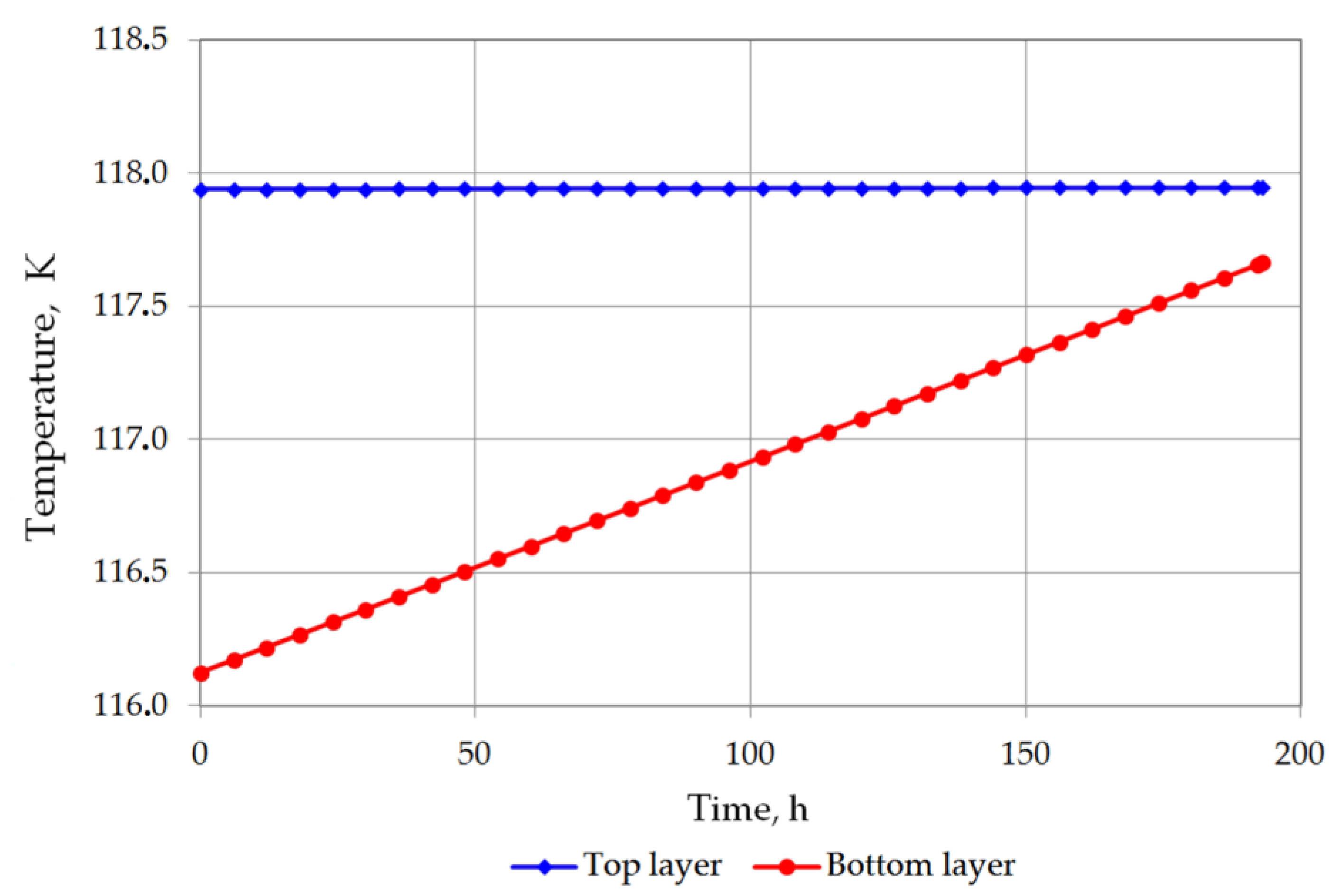
| Temperature of bottom layer, K | 116.124 | 116.3609 | 116.5512 | 116.79 | 117.0298 | 117.2703 | 117.5601 |
| Temperature of top layer, K | 117.9392 | 117.9403 | 117.9413 | 117.9425 | 117.9438 | 117.945 | 117.9464 |
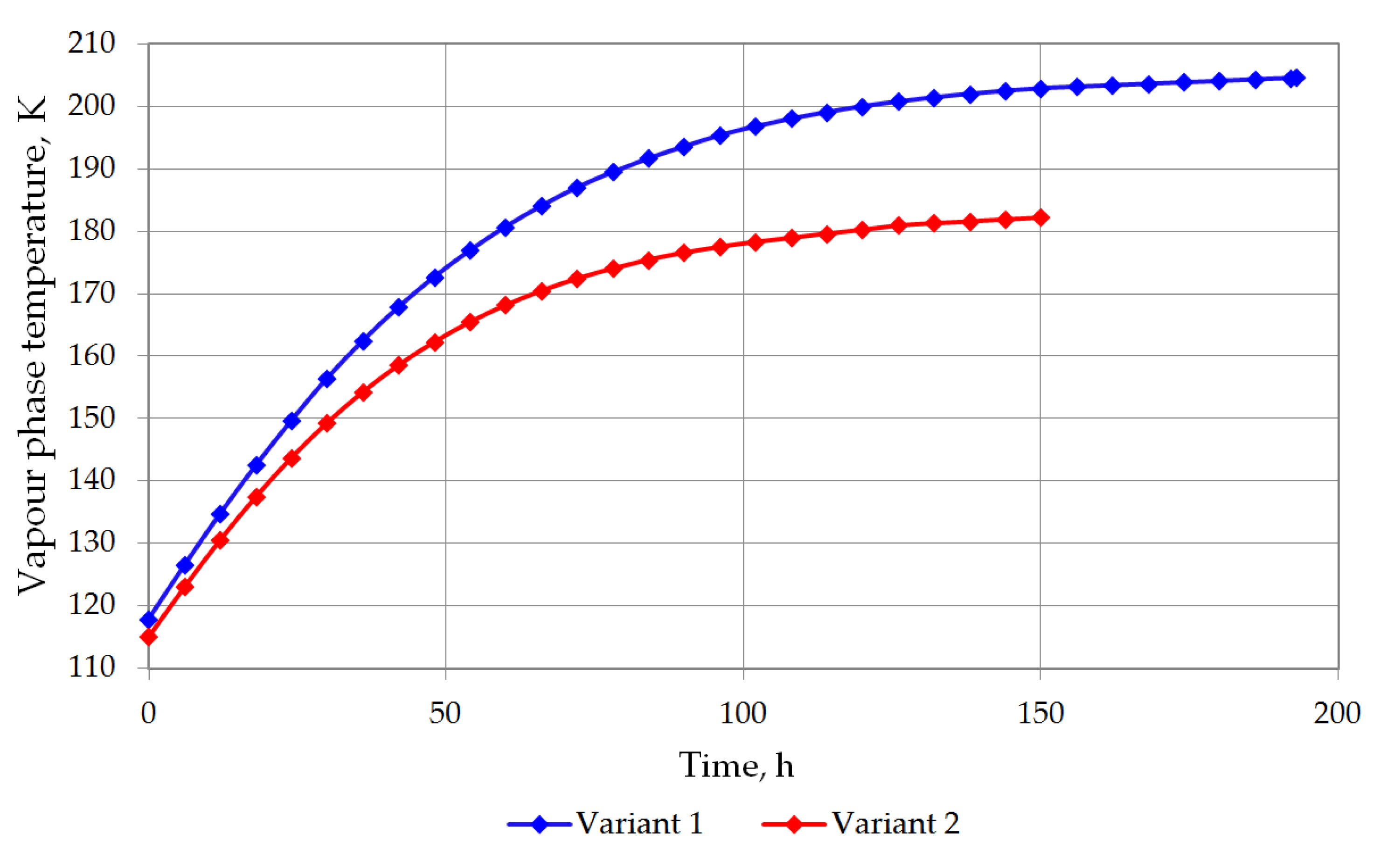
| Temperature of gas phase, K | 126.5542 | 162.533 | 180.7731 | 193.6171 | 200.0772 | 202.9652 | 203.3263 |
| Density of top layer, kg/m3 | 451.2055 | 451.2373 | 451.2655 | 451.3007 | 451.336 | 451.3713 | 451.4137 |
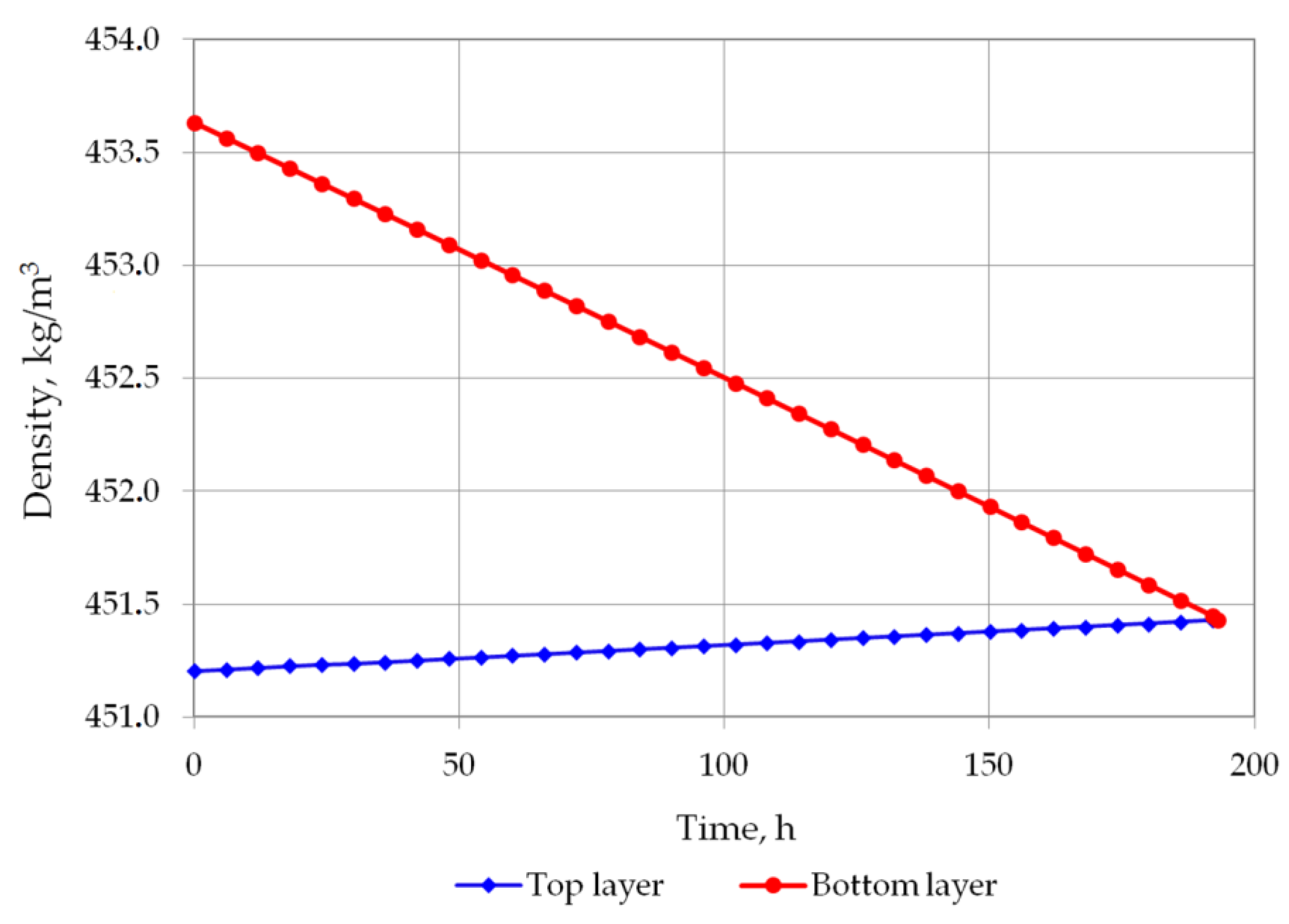
| Density of bottom layer, kg/m3 | 453.6301 | 453.2938 | 453.0234 | 452.6838 | 452.3425 | 451.9996 | 451.5862 |
| Density difference, kg/m3 | 2.424581 | 2.056484 | 1.75796 | 1.383114 | 1.006509 | 0.628253 | 0.172478 |
| Composition of top layer: | |||||||
| Methane | 0.900 | 0.899904 | 0.899822 | 0.899719 | 0.899617 | 0.899514 | 0.899390 |
| Ethane | 0.067 | 0.067069 | 0.067124 | 0.067192 | 0.067261 | 0.06733 | 0.067413 |
| Propane | 0.023 | 0.023019 | 0.023038 | 0.023061 | 0.023085 | 0.023109 | 0.023137 |
| n-Butane | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| iso-Butane | 0.010 | 0.010008 | 0.010016 | 0.010027 | 0.010037 | 0.010047 | 0.01006 |
| Nitrogen | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Time Step | t = 0 h | t =30 h | t = 60 h | t = 90 h | t = 120 h | t = 150 h |
|---|---|---|---|---|---|---|
| Temperature of bottom layer, K | 115.5 | 115.697 | 115.8942 | 116.0915 | 116.2888 | 116.4857 |
| Temperature of top layer, K | 115.0313 | 115.0415 | 115.054 | 115.067 | 115.08 | 115.0926 |
| Temperature of gas phase, K | 123.0159 | 154.2574 | 170.4496 | 177.5019 | 181.4353 | 181.9967 |
| Density of top layer, kg/m3 | 434.0142 | 434.01 | 434.0081 | 434.0074 | 434.0078 | 434.0093 |
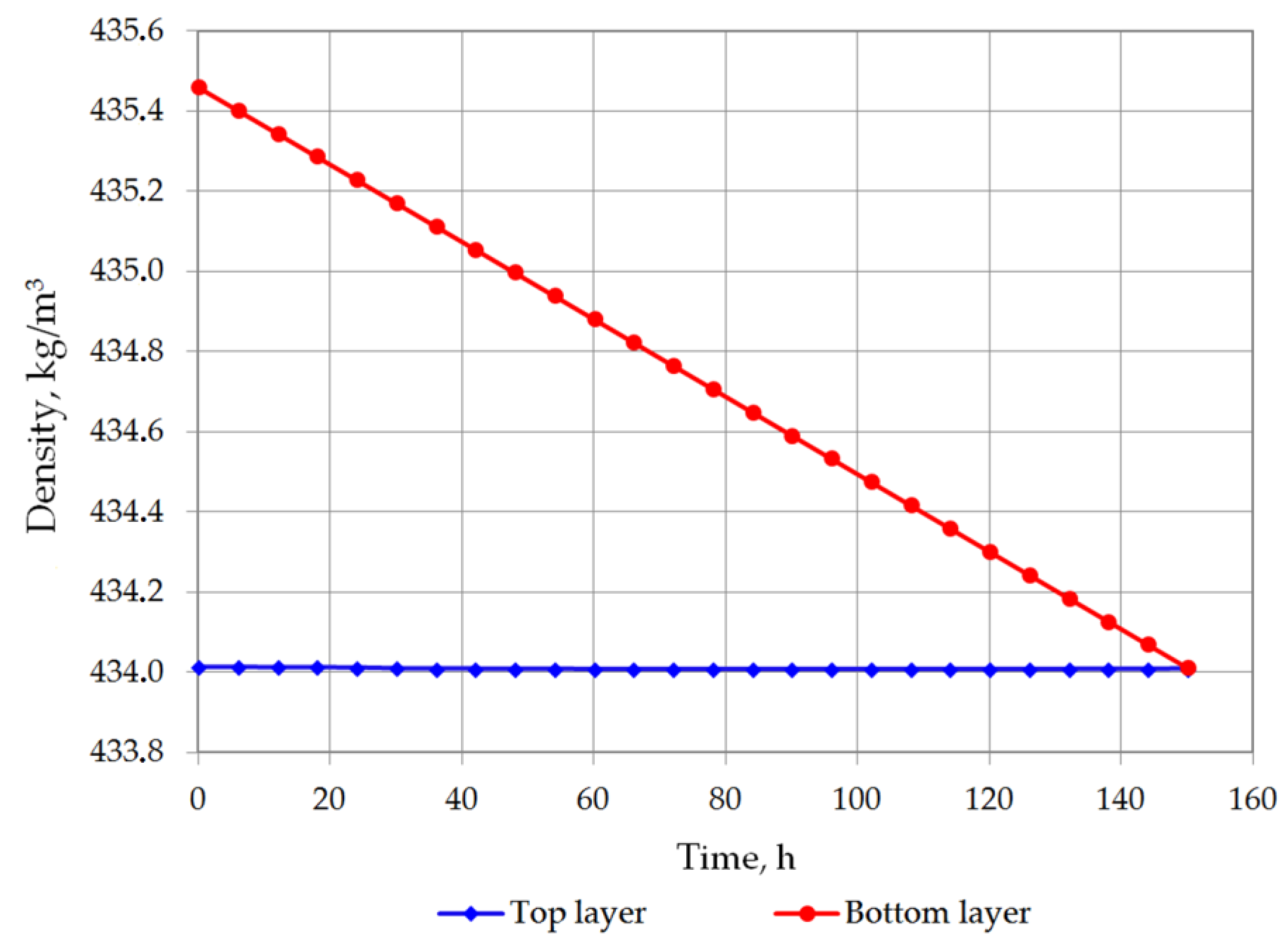
| Density of bottom layer, kg/m3 | 435.4609 | 435.1719 | 434.8822 | 434.592 | 434.3017 | 434.0117 |
| Density difference, kg/m3 | 1.446645 | 1.161924 | 0.874034 | 0.584666 | 0.293898 | 0.002427 |
| Composition of top layer: | ||||||
| Methane | 0.955 | 0.954976 | 0.954948 | 0.954909 | 0.954858 | 0.954813 |
| Ethane | 0.032 | 0.032041 | 0.032083 | 0.032138 | 0.032208 | 0.032267 |
| Propane | 0.009 | 0.009009 | 0.009021 | 0.009036 | 0.009056 | 0.009073 |
| n-Butane | 0 | 0 | 0 | 0 | 0 | 0 |
| iso-Butane | 0.003 | 0.003003 | 0.003007 | 0.003012 | 0.003019 | 0.003024 |
| Nitrogen | 0.001 | 0.00097 | 0.000941 | 0.000904 | 0.000859 | 0.000823 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Włodek, T.; Łaciak, M. Rollover Prevention Model for Stratified Liquefied Natural Gas in Storage Tanks. Energies 2023, 16, 7666. https://doi.org/10.3390/en16227666
Włodek T, Łaciak M. Rollover Prevention Model for Stratified Liquefied Natural Gas in Storage Tanks. Energies. 2023; 16(22):7666. https://doi.org/10.3390/en16227666
Chicago/Turabian StyleWłodek, Tomasz, and Mariusz Łaciak. 2023. "Rollover Prevention Model for Stratified Liquefied Natural Gas in Storage Tanks" Energies 16, no. 22: 7666. https://doi.org/10.3390/en16227666
APA StyleWłodek, T., & Łaciak, M. (2023). Rollover Prevention Model for Stratified Liquefied Natural Gas in Storage Tanks. Energies, 16(22), 7666. https://doi.org/10.3390/en16227666






