Development of a New Modified CaCl2·6H2O Composite Phase Change Material
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Equipment
3. Result and Discussion
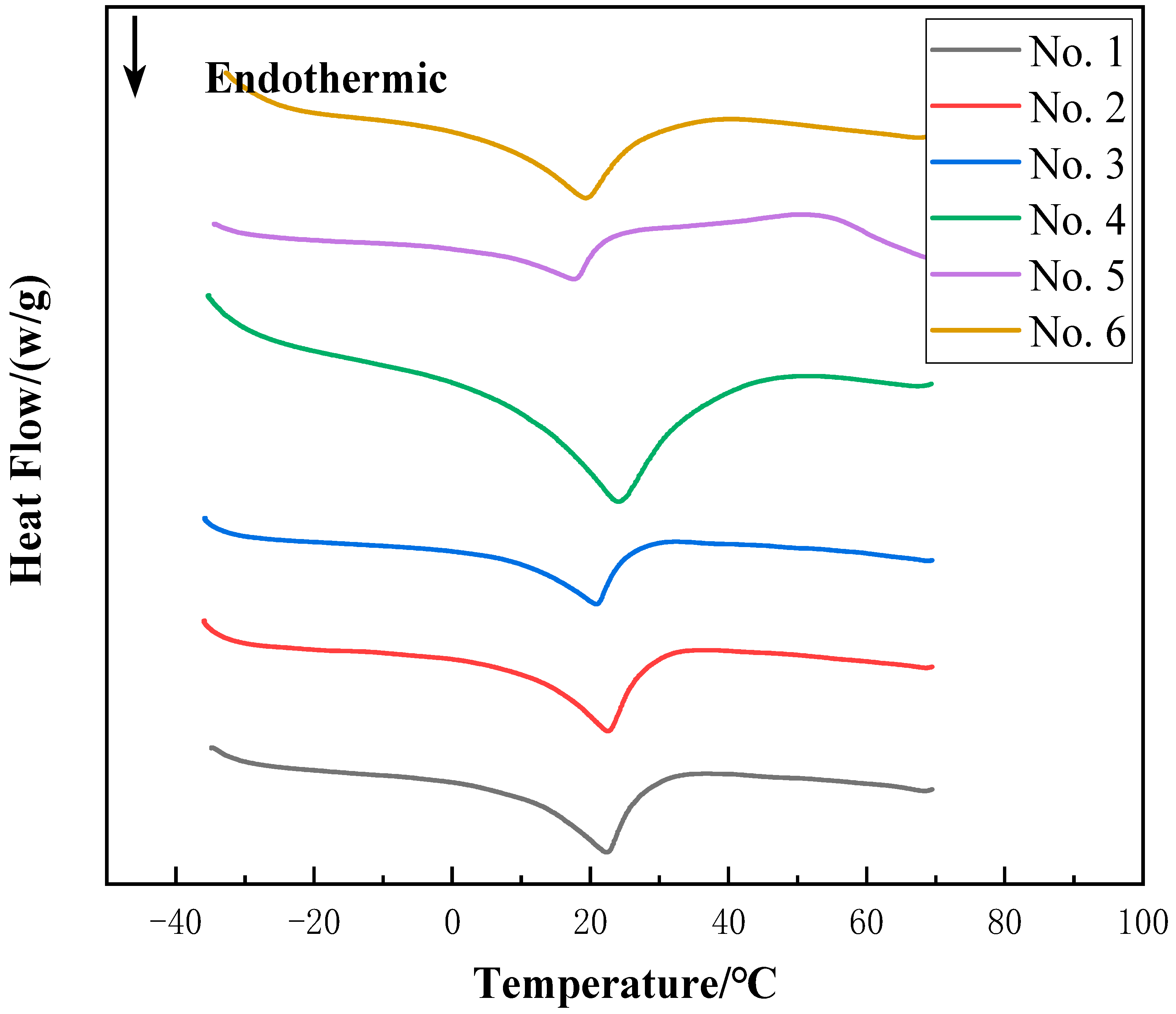
3.1. Development of CPCM
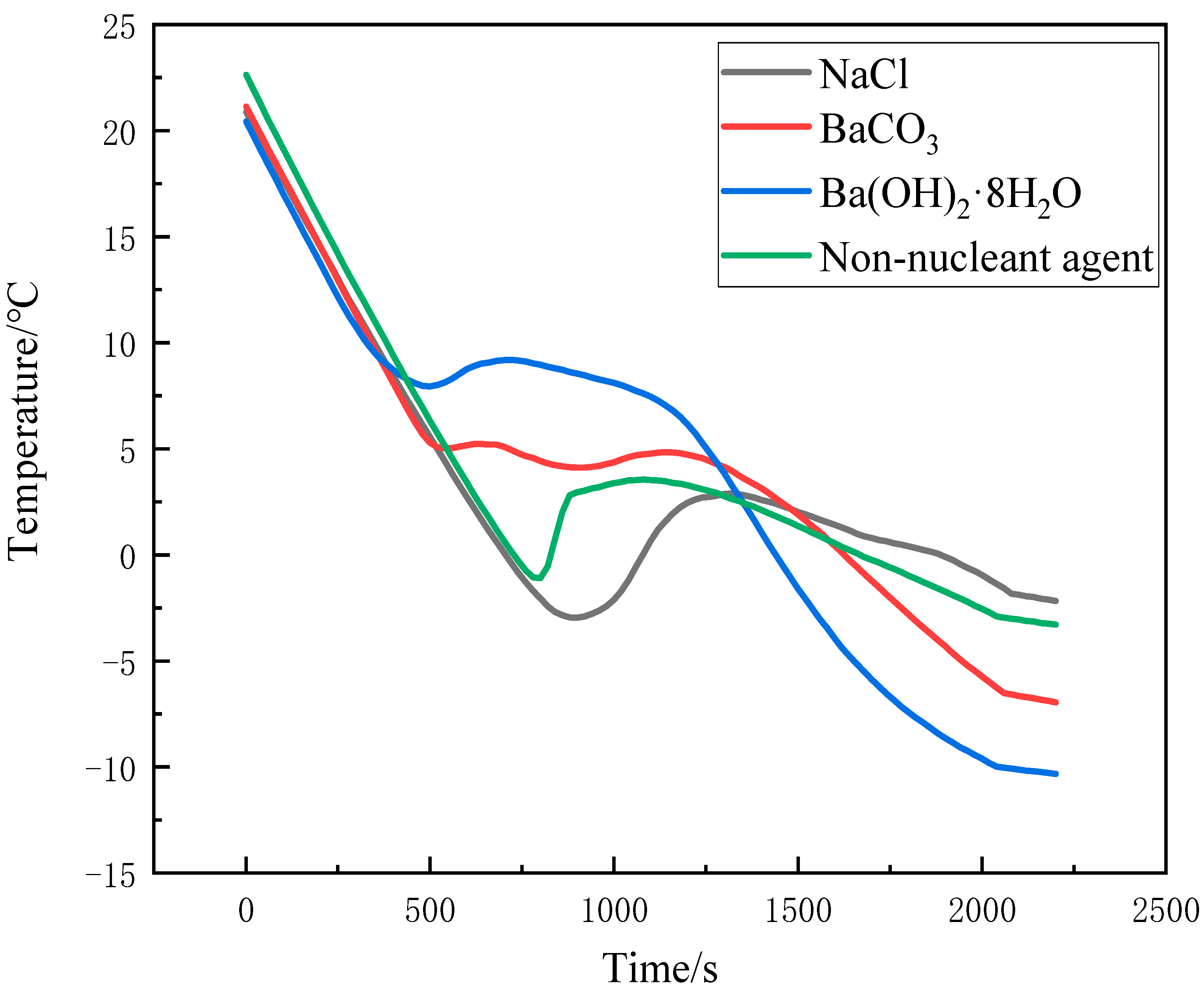
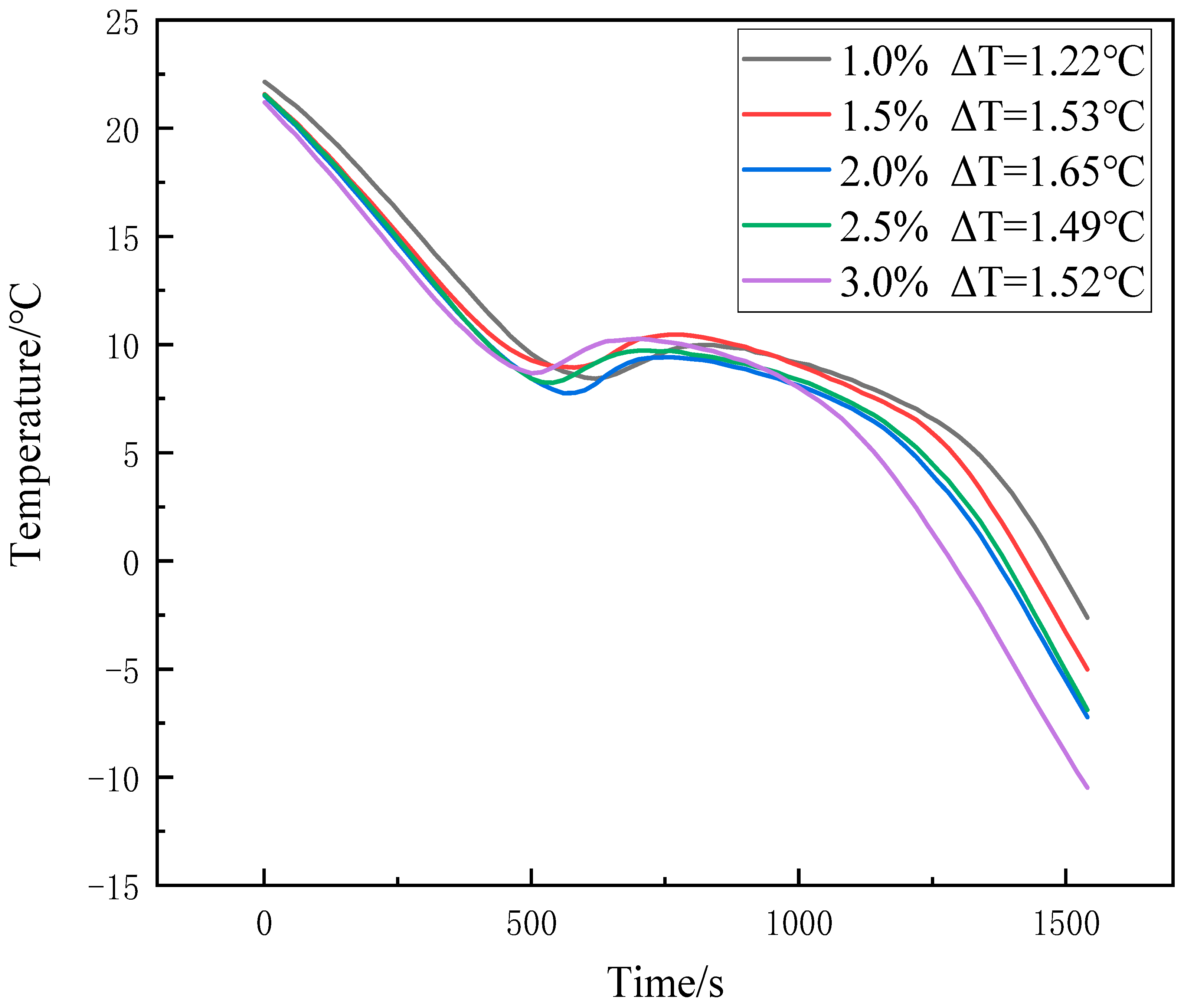
3.2. Improvement of Subcooling
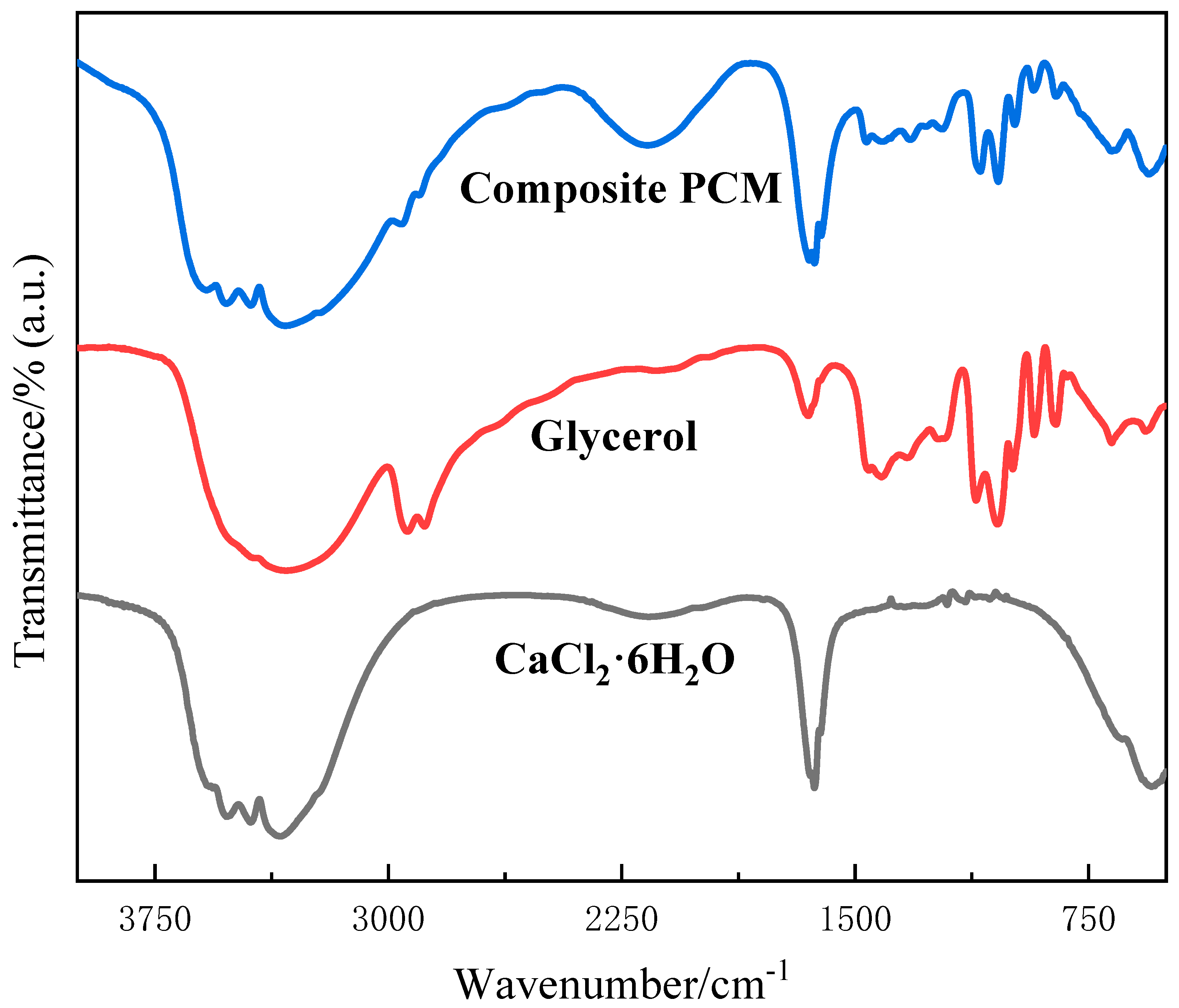
3.3. FTIR Analysis
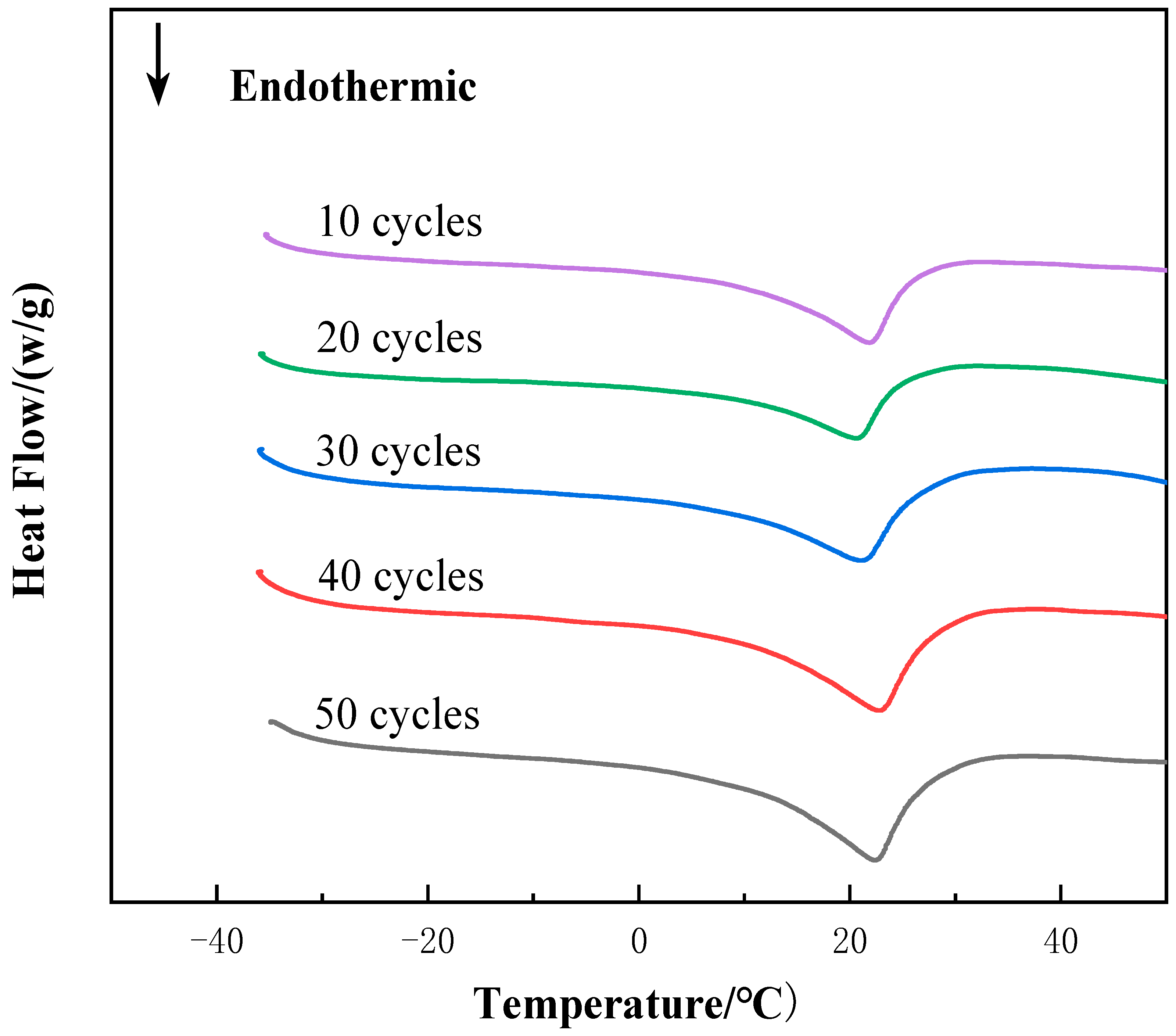
3.4. Thermal Reliability
4. Conclusions
- After DSC test, it was found that when the mass ratio of calcium chloride hexahydrate to glycerol was 85:15, the phase change temperature of the composite phase change material was 11.8 °C and the enthalpy of phase change was 112.86 J/g, which was suitable for the field of air-conditioning.
- FTIR confirmed that the material was successfully developed and proved that the two reagents only acted physically, without chemical reaction.
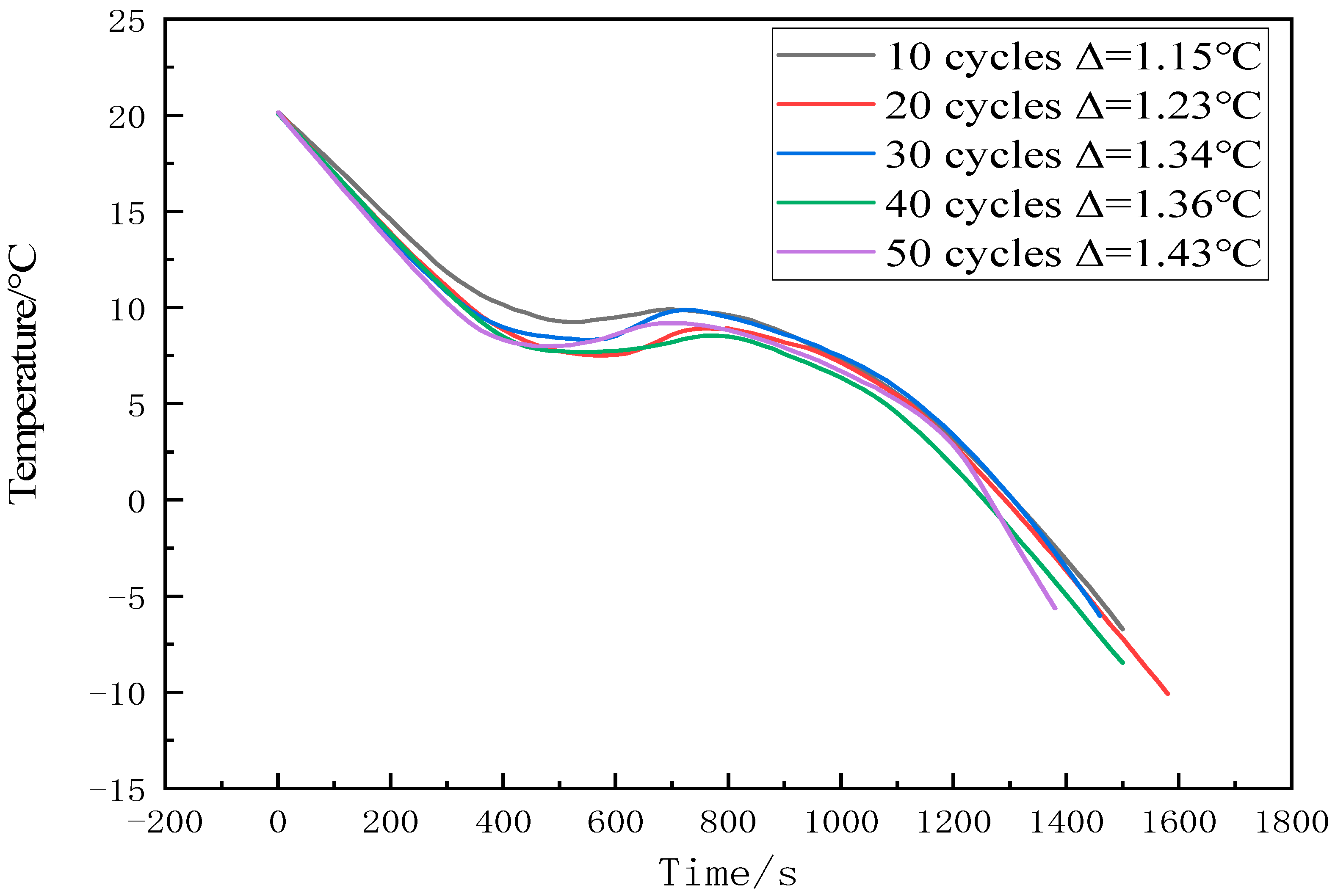
- The material still showed good thermal stability after 50 thermal cycles. The subcooling of the new CPCM only increased by 0.28 °C after 50 cycles and the enthalpy of CPCM has almost no change.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, J.; Li, Z.; Yang, S.; Tao, H.; Yao, H.; Wu, C. Application of Dueling-DQN in air conditioning energy saving control. Comput. Syst. Appl. 2021, 30, 271–279. [Google Scholar]
- Navidbakhsh, M.; Shirazi, A.; Sanaye, S. Four E analysis and multi-objective optimization of an ice storage system incorporating PCM as the partial cold storage for air-conditioning applications. Appl. Therm. Eng. 2013, 58, 30–41. [Google Scholar] [CrossRef]
- Nagano, K.; Ogawa, K.; Mochida, T.; Hayashi, K.; Ogoshi, H. Thermal characteristics of magnesium nitrate hexahydrate and magnesium chloride hexahydrate mixture as a phase change material for effective utilization of urban waste heat. Appl. Therm. Eng. 2004, 24, 221–232. [Google Scholar] [CrossRef]
- Cunha, J. Thermal energy storage for low and medium temperature applications using phase change materials-A review. Appl. Energy 2016, 177, 227–238. [Google Scholar] [CrossRef] [Green Version]
- Sar, A. Eutectic mixtures of some fatty acids for latent heat storage: Thermal properties and thermal reliability with respect to thermal cycling. Energy Convers. Manag. 2006, 47, 1207–1221. [Google Scholar] [CrossRef]
- Luo, X.; Zhang, X.; Hou, X.; Hua, W.; Han, X.; Li, J. Experimental Analysis on Heat Release of Pulsating Heat Pipe Phase Change Heat Accumulator With Anhydrous Ethanol. J. Eng. Thermophys. 2018, 39, 1524–1531. [Google Scholar]
- Han, B.; Choi, J.; Dantzig, J.; Bischof, J. A quantitative analysis on latent heat of an aqueous binary mixture. Cryobiology 2006, 52, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Oro, E.; Miro, L.; Barreneche, C.; Martorell, I.; Farid, M.; Cabeza, L. Corrosion of metal and polymer containers for use in PCM cold storage. Appl. Energy 2013, 109, 449–453. [Google Scholar] [CrossRef]
- Shi, T.; Xu, H.; Qi, C.; Lei, B.; Wu, Y.; Zhao, C. Multi-physics modeling of thermochemical heat storage with enhance heat transfer. Appl. Therm. Eng. 2021, 198, 117508. [Google Scholar] [CrossRef]
- Xu, H.; Wang, Y.; Han, X. Analytical considerations of thermal storage and interface evolution of a PCM with/without porous media. Int. J. Numer. Methods Heat Fluid Flow 2019, 30, 373–400. [Google Scholar] [CrossRef]
- Dong, Y.; Wang, F.; Yang, L.; Shi, X.; Zhang, G.; Shuai, Y. Thermal Performance Analysis of PCM Capsules Packed-Bed System with Biomimetic Leaf Hierarchical Porous Structure. J. Therm. Sci. 2021, 30, 1559–1571. [Google Scholar] [CrossRef]
- Dong, Y.; Wang, F.; Zhang, Y.; Shi, X.; Zhang, A.; Shuai, Y. Experimental and numerical study on flow characteristic and thermal performance of macro-capsules phase change material with biomimetic oval structure. Energy 2022, 238, 121830. [Google Scholar] [CrossRef]
- Wang, F.; Dong, Y.; Li, Y.; Xu, J.; Zhang, G. Numerical study on the thermal performance of packed-bed latent heat thermal energy storage system with biomimetic alveoli structure capsule. Sci. China Technol. Sci. 2021, 64, 1544–1554. [Google Scholar] [CrossRef]
- Ying, Y.; Shen, H. Investigation on Cryogenics Cool Thermal Energy Storage Phase Change Composition Material. J. Low Temp. Phys. 2009, 31, 144–147. [Google Scholar]
- Li, J.; Xie, R.; Liu, G. Development of New Low-temperature Phase Change Material and Equipment Used in Refrigerated Transportation. J. Refrig. 2018, 39, 32–37. [Google Scholar]
- Kenisarin, M.; Mahkamov, K. Salt hydrates as latent heat storage materials: Thermophysical properties and costs. Solar Energy Mater. Sol. Cells 2016, 145, 255–286. [Google Scholar] [CrossRef]
- Zhang, X.; Zhou, Y.; Li, X.; Shen, Y.; Hai, C.; Dong, O.; Ren, X.; Zeng, J.; Sun, Y.; Wang, S.; et al. Research progress of inorganic hydrated salt phase change energy storage materials based on calcium chloride hexahydrate. Energy Storage Sci. Technol. 2018, 7, 40–47. [Google Scholar]
- Zhang, X.; Zhou, S.; Liu, S.; Li, Y.; Xu, X. Cold Storage Characteristics of n-Octanoic-Lauric Acid Nanocomposite Phase Change Material. J. Tianjin Univ. Sci. Technol. 2019, 52, 71–77. [Google Scholar]
- Carlsson, B.; Stymne, H.; Wettermark, G. An incongruent heat-of-fusion system-CaCl2·6H2O- Made congruent through modification of the chemical composition of the system. Sol. Energy 1979, 23, 343–350. [Google Scholar] [CrossRef]
- Paris, J.; Jolly, R. Observations sur le comportement a la fusion-solidification du chlorure de calcium hexahydrate. Thermochim. Acta 1989, 152, 271–278. [Google Scholar] [CrossRef]
- Zhang, X.; Zeng, T.; Chen, Y. Development of improved calcium chloride hexahydrate phase change cold storage material. In Proceedings of the 13th Yangtze River Delta Energy Forum, Hangzhou, China, 1 October 2016; pp. 251–256. [Google Scholar]
- Ji, J.; Zeng, T.; Zhang, X.; Gao, Y.; Chen, Y. Preparation and properties of calcium chloride hexahydrate phase change material after modified. Cryog. Supercond. 2017, 45, 71–76. [Google Scholar]
- Liu, X.; Wu, J.; Xian, T.; Feng, Y. Preparation and properties of CaCl2 /expanded graphite composite phase change materials. J. Zhejiang Univ. 2019, 53, 1291–1297. [Google Scholar]
- Bayes-Garcia, L.; Ventola, L.; Cordobilla, R.; Benages, R.; Cuevas-Diarte, M.A. Phase Change Materials (PCM) microcapsules with different shell compositions: Preparation, characterization and thermal stability. Sol. Energy Mater. Sol. Cells 2010, 94, 1235–1240. [Google Scholar] [CrossRef]
- Yu, J.; Liu, T. Preparation and Characterization of Microencapsulated Phase Change Coating. Appl. Mech. Mater. 2012, 204–208, 4173–4176. [Google Scholar] [CrossRef]
- Fu, W.; Zou, T.; Liang, X. Thermal properties and thermal conductivity enhancement of composite phase change material using sodium acetate trihydrate-urea/expanded graphite for radiant floor heating system. Appl. Therm. Eng. 2018, 138, 618–626. [Google Scholar] [CrossRef]
- Zou, T.; Fu, W.; Liang, X. Preparation and performance of modified calcium chloride hexahydrate composite phase change material for air-conditioning cold storage. Int. J. Refrig. 2018, 95, 175–181. [Google Scholar] [CrossRef]
- Zou, T.; Liang, X.; Wang, S. Effect of expanded graphite size on performances of modified CaCl2·6H2O phase change material for cold energy storage. Microporous Mesoporous Mater. 2020, 305, 110403. [Google Scholar] [CrossRef]
- Wang, X.; Dennis, M.; Hou, L. Clathrate hydrate technology for cold storage in air conditioning systems. Renew. Sustain. Energy Rev. 2014, 36, 34–51. [Google Scholar] [CrossRef]
- Gang, L.; Hwang, Y.; Radermacher, R. Review of cold storage materials for air conditioning application—Science Direct. Int. J. Refrig. 2012, 35, 2053–2077. [Google Scholar]
- Luo, X.; Lu, X.; Cao, S.; Zou, C. Analysis on the Improvement of Thermal Performance of Phase Change Material Ba (OH)2 · 8H2O. Energies 2021, 14, 7761. [Google Scholar] [CrossRef]
- Liu, D.; Xu, Y. Effect of nucleating agent on thermal storage properties of CaCl2·6H2O phase change materials. J. Sol. Energy 2007, 028, 732–738. [Google Scholar]


| Number | CaCl · 6H2O/wt.% | C3H8O3/wt.% |
|---|---|---|
| No.1 | 85 | 15 |
| No.2 | 84 | 16 |
| No.3 | 83 | 17 |
| No.4 | 82 | 18 |
| No.5 | 81 | 19 |
| No.6 | 80 | 20 |
| Additives Content | C3H8O3 Content (wt.%) | |||||
|---|---|---|---|---|---|---|
| 15% (No.1) | 16% (No.2) | 17% (No.3) | 18% (No.4) | 19% (No.5) | 20% (No.6) | |
| Tm (°C) | 11.8 | 12.2 | 14.6 | 12.5 | 9.2 | 88.4 |
| ΔH (J/g) | 112.86 | 116.78 | 103.56 | 109.07 | 102.38 | 84.18 |
| No. of Cycles | 10 | 20 | 30 | 40 | 50 |
|---|---|---|---|---|---|
| Tm(°C) | 11.7 | 10.3 | 11.9 | 11.3 | 11.8 |
| ΔH (J/g) | 111.33 | 119.72 | 119.39 | 115.43 | 111.48 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, S.; Luo, X.; Han, X.; Lu, X.; Zou, C. Development of a New Modified CaCl2·6H2O Composite Phase Change Material. Energies 2022, 15, 824. https://doi.org/10.3390/en15030824
Cao S, Luo X, Han X, Lu X, Zou C. Development of a New Modified CaCl2·6H2O Composite Phase Change Material. Energies. 2022; 15(3):824. https://doi.org/10.3390/en15030824
Chicago/Turabian StyleCao, Shibo, Xiaoxue Luo, Xiaochun Han, Xiaohui Lu, and Changzhen Zou. 2022. "Development of a New Modified CaCl2·6H2O Composite Phase Change Material" Energies 15, no. 3: 824. https://doi.org/10.3390/en15030824
APA StyleCao, S., Luo, X., Han, X., Lu, X., & Zou, C. (2022). Development of a New Modified CaCl2·6H2O Composite Phase Change Material. Energies, 15(3), 824. https://doi.org/10.3390/en15030824






