Lignin Modifications, Applications, and Possible Market Prices
Abstract
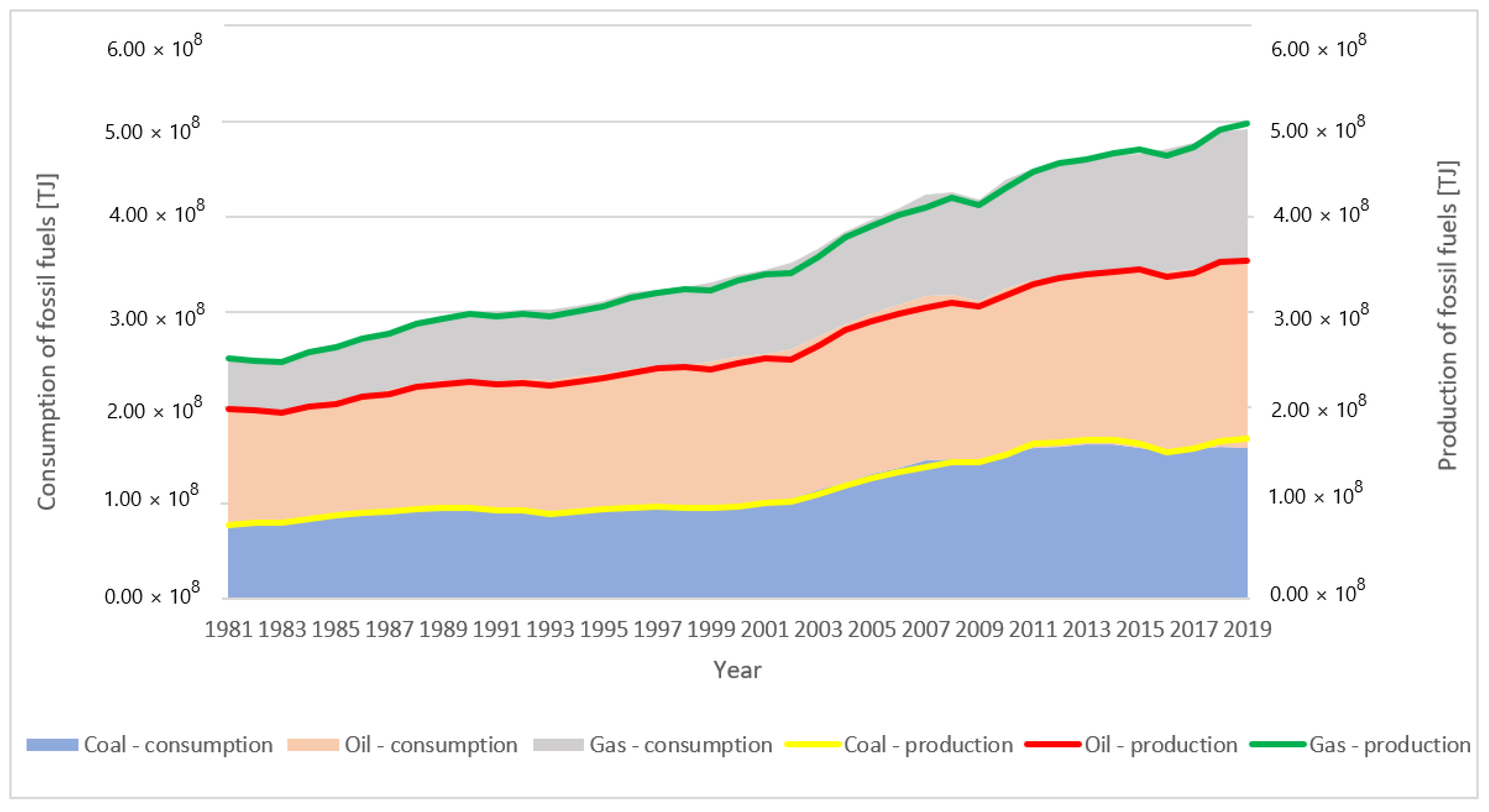
1. Introduction
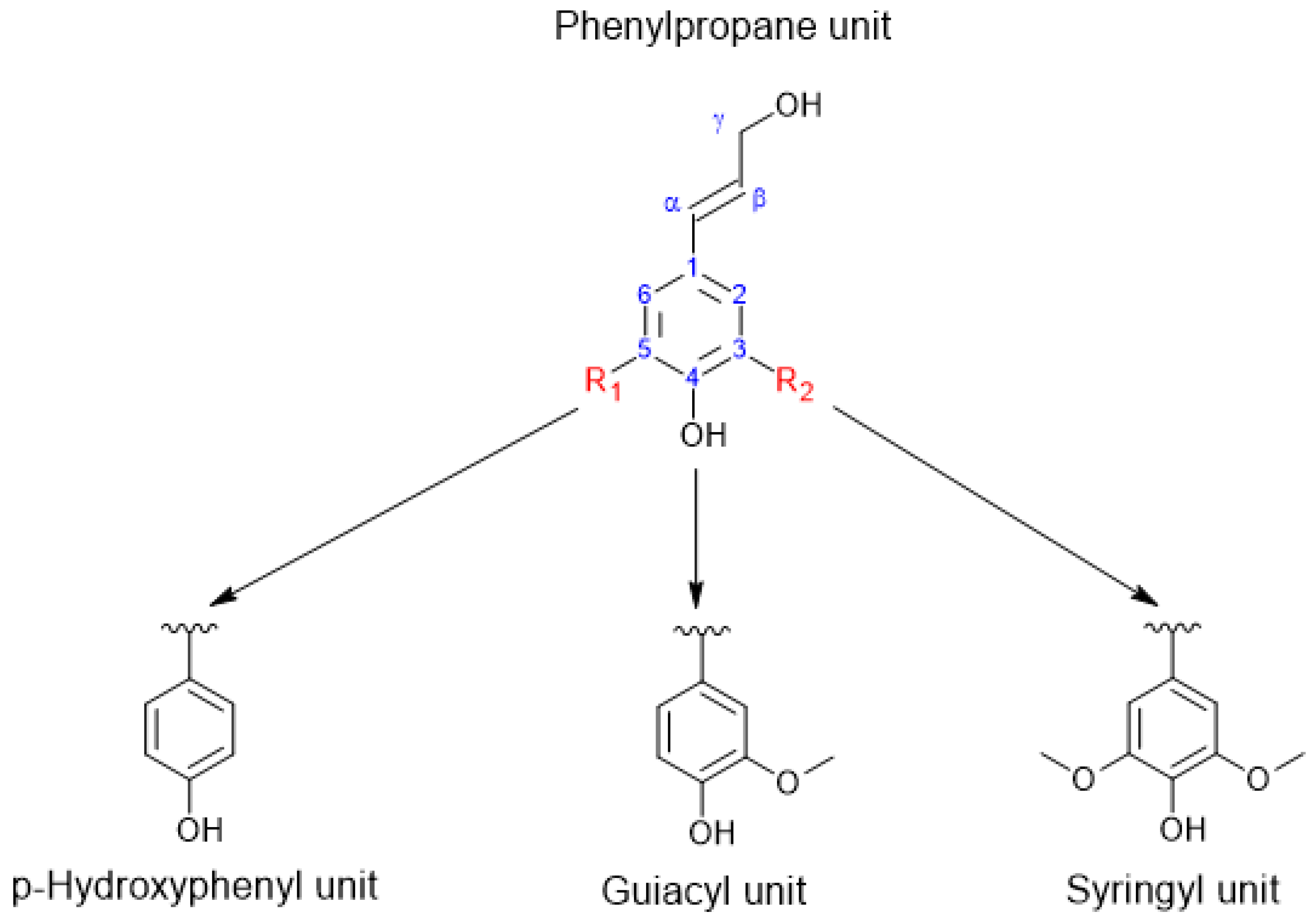
2. Lignin Structure
- Polysaccharides are removed from the plant and lignin stays as residual compounds (methods according to Klasson, Komarov, Willstätter, Purves, etc.).
- Lignin is dissolved and removed from the plant, and polysaccharides remain unmodified (Kraft, sulfite, neutral sulfite semi-chemical, soda pulping, etc.).
- The wood content is mechanically removed, and lignin stays as residual material (milled wood lignin, isolated with enzymes).
- For analytical determination of the lignin content.
- For the preparation of the lignin sample for further investigation.
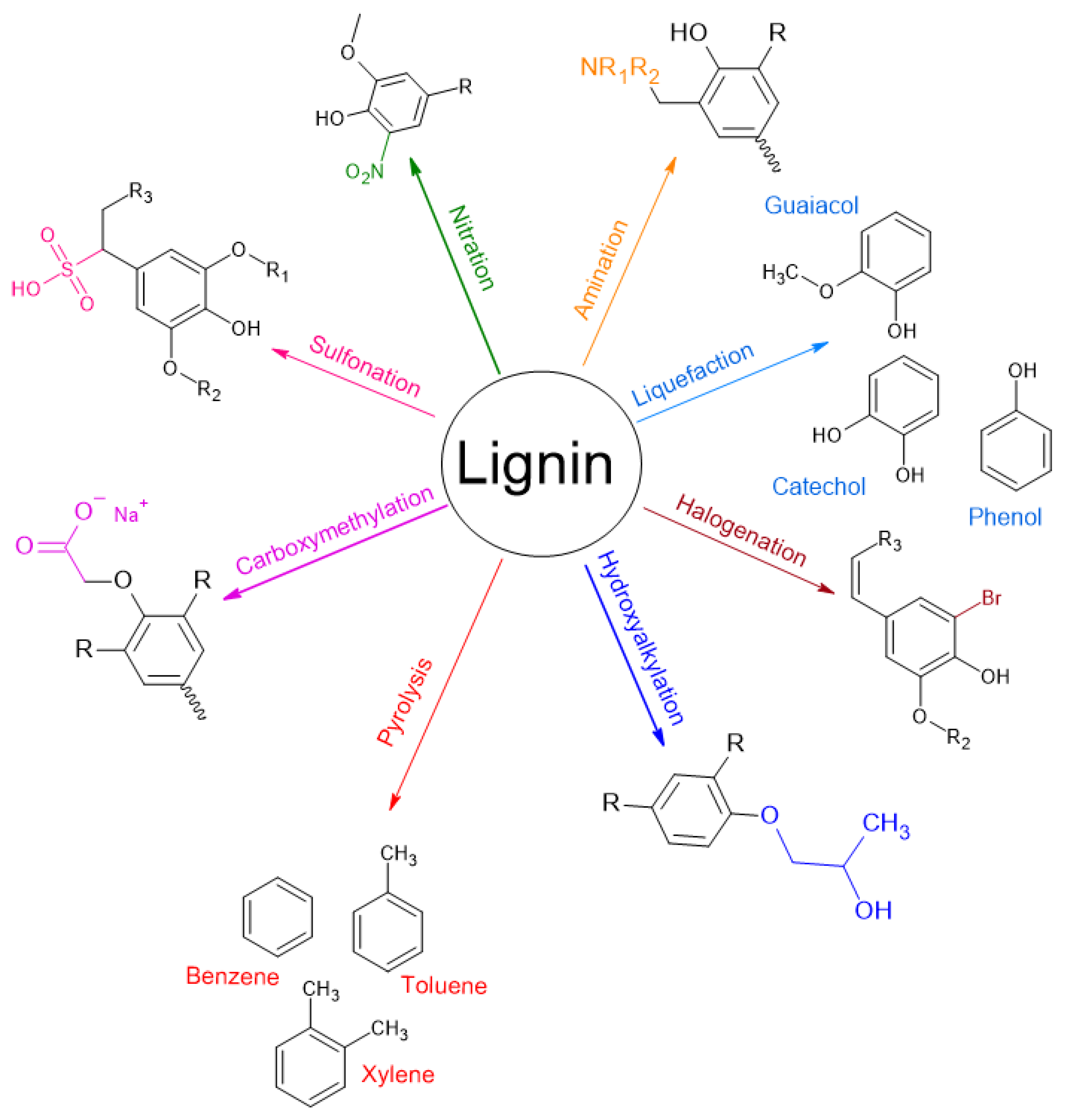
3. Modifications of Lignin
- The material is sustainable and renewable;
- There is an immense generation of lignin as a by-product in the pulp/paper industry and the cellulosic ethanol industry;
- There are various functional groups present in the lignin structure that enable a variety range of chemical reactions;
- Lignin has intrinsic biodegradability, and it is believed that lignin-incorporated polymers are more biodegradable than petroleum-based polymers.
4. Lignin Fractionation
4.1. Depolymerisation/Fragmentation of Lignin
4.2. Modification of Functional Groups
5. Lignin Products
5.1. High Purity Lignin
5.2. Lignin Biocomposites Reinforced with Plant Fibres
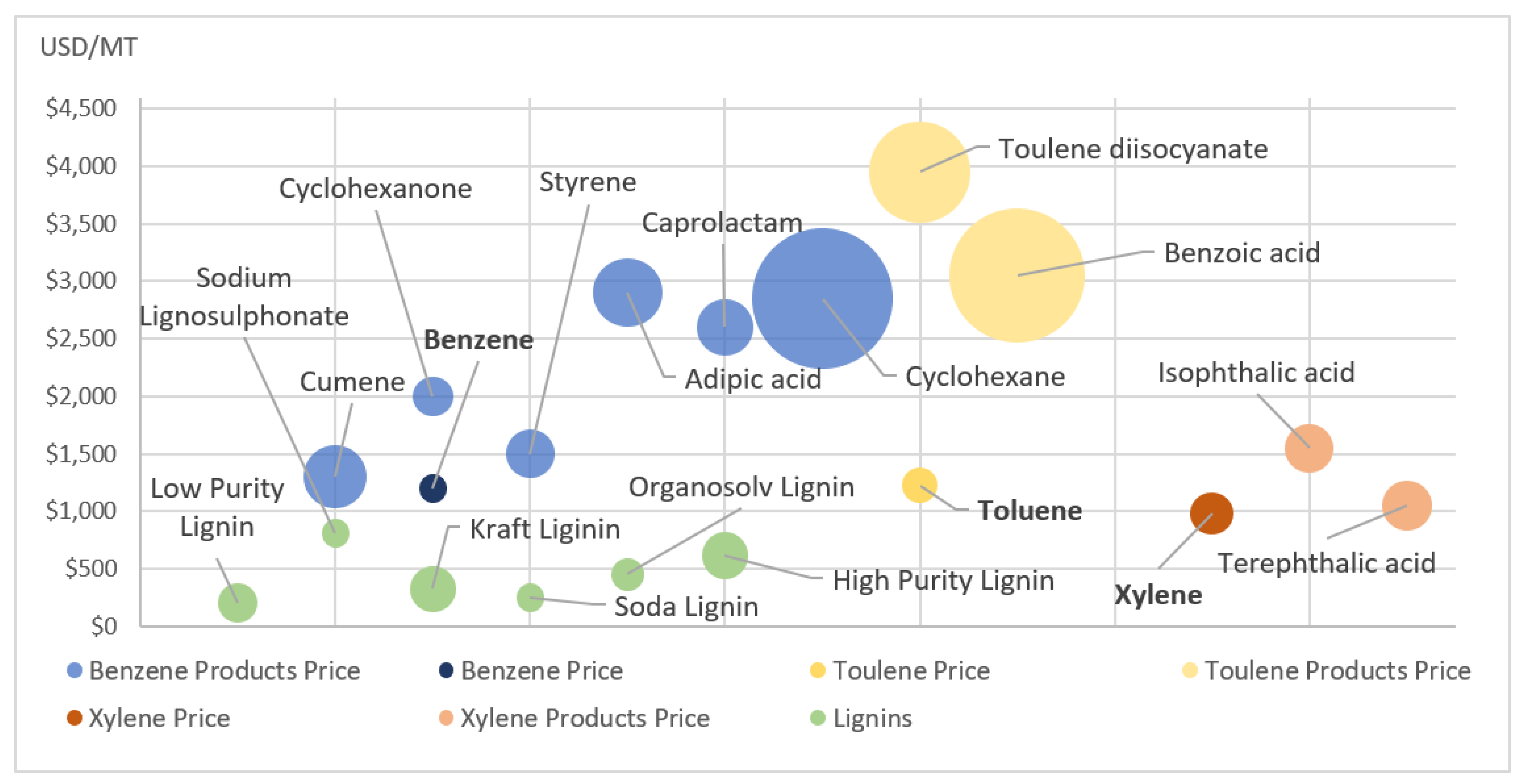
5.3. Benzene, Toluene, Xylene
5.4. Phenol Resins
5.5. Lignin Bio-Oil
5.6. Lignin-Based Carbon Nanofibers
5.7. Lignin-Based Polyurethane and Polyurethanes Foams
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- BP. Statistical Review of World Energy Globally Consistent Data on World Energy Markets and Authoritative Publications in the Field of Energy; Statistical Review; BP: London, UK, 2021; Volume 70. [Google Scholar]
- Bar-On, Y.M.; Phillips, R.; Milo, R. The biomass distribution on Earth. Proc. Natl. Acad. Sci. USA 2018, 115, 6506–6511. [Google Scholar] [CrossRef]
- Ritchie, H.; Roser, M. Fossil Fuels. Available online: https://ourworldindata.org/fossil-fuels (accessed on 12 November 2020).
- Rosillo-Calle, F.; de Groot, P.; Hemstock, S.L. Non Woody Biomass and Secondary Fuels. In The Biomass Assessment Handbook: Energy for a Sustainable Environment; Rosillo-Calle, F., de Groot, P., Hemstock, S.L., Woods, J., Eds.; Routledge: London, UK, 2015; pp. 1–336. ISBN 9781138019652. [Google Scholar]
- Zakzeski, J.; Bruijnincx, P.C.A.; Jongerius, A.L.; Weckhuysen, B.M. The Catalytic Valorization of Lignin for the Production of Renewable Chemicals. Chem. Rev. 2010, 110, 3552–3599. [Google Scholar] [CrossRef] [PubMed]
- Keyoumu, A.; Sjödahl, R.; Henriksson, G.; Ek, M.; Gellerstedt, G.; Lindström, M.E. Continuous nano- and ultra-filtration of kraft pulping black liquor with ceramic filters: A method for lowering the load on the recovery boiler while generating valuable side-products. Ind. Crop. Prod. 2004, 20, 143–150. [Google Scholar] [CrossRef]
- Vanholme, R.; Demedts, B.; Morreel, K.; Ralph, J.; Boerjan, W. Lignin Biosynthesis and Structure. Plant Physiol. 2010, 153, 895–905. [Google Scholar] [CrossRef] [PubMed]
- Fengel, D.; Wegener, G. Wood: Chemistry, Ultrastructure, Reactions; Walter de Gruyter: Berlin, Germany, 2011; ISBN 3935638396. [Google Scholar]
- Nakashima, J.; Chen, F.; Jackson, L.; Shadle, G.; Dixon, R.A. Multi-site genetic modification of monolignol biosynthesis in alfalfa ( Medicago sativa ): Effects on lignin composition in specific cell types. New Phytol. 2008, 179, 738–750. [Google Scholar] [CrossRef]
- Uzal, E.N.; Ros, L.V.G.; Pomar, F.; Bernal, M.A.; Paradela, A.; Albar, J.P.; Barceló, A.R. The presence of sinapyl lignin inGinkgo bilobacell cultures changes our views of the evolution of lignin biosynthesis. Physiol. Plant. 2009, 135, 196–213. [Google Scholar] [CrossRef]
- Boerjan, W.; Ralph, J.; Baucher, M. Lignin Biosynthesis. Annu. Rev. Plant Biol. 2003, 54, 519–546. [Google Scholar] [CrossRef] [PubMed]
- Patil, N.D.; Tanguy, N.R.; Yan, N. Lignin Interunit Linkages and Model Compounds; Elsevier: Amsterdam, The Netherlands, 2016; ISBN 9780323355667. [Google Scholar]
- Blažej, A.; Košík, M. Covalent and Supermolecular Structure of Basic Components of Plant Raw Materials: Structure of the Cell Wall. In PHYTOMASS a Raw Material for Chemistry and Biotechnology; Kemp, T.J., Kennedy, J.F., Eds.; VEDA Publishing house of Slovak Academy od Science: Bratislava, Slovakia, 1993; pp. 32–47. ISBN 0-13-666801-1. [Google Scholar]
- Sjöström, E. Wood pulping. In Wood Chemistry; Elsevier: Amsterdam, The Netherlands, 1993; pp. 114–164. [Google Scholar]
- Abhilash, M.; Thomas, D. Biopolymers for Biocomposites and Chemical Sensor Applications; Elsevier: Amsterdam, The Netherlands, 2017; ISBN 9780081009741. [Google Scholar]
- Sjöström, E. Wood Chemistry Fundamentals and Applications: Lignin. In Wood Chemistry: Fundamentals and Applications; Elsevier: Amsterdam, The Netherlands, 1993; pp. 1–36. [Google Scholar] [CrossRef]
- Rana, R.; Nanda, S.; Meda, V.; Dalai, A.K. A Review of Lignin Chemistry and Its Biorefining Conversion Technologies. J. Biochem. Eng. Bioprocess Technol. 2018, 1, 1–14. [Google Scholar]
- Chakar, F.S.; Ragauskas, A.J. Review of current and future softwood kraft lignin process chemistry. Ind. Crop. Prod. 2004, 20, 131–141. [Google Scholar] [CrossRef]
- Adler, E. Lignin chemistry? past, present and future. Wood Sci. Technol. 1977, 11, 169–218. [Google Scholar] [CrossRef]
- Liu, X.; Bouxin, F.P.; Fan, J.; Budarin, V.L.; Hu, C.; Clark, J.H. Recent Advances in the Catalytic Depolymerization of Lignin towards Phenolic Chemicals: A Review. ChemSusChem 2020, 13, 4296–4317. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Fu, S.; Gan, L. (Eds.) Structure and Characteristics of Lignin. In Lignin Chemistry and Applications; Elsevier: Amsterdam, The Netherlands, 2019; pp. 25–50. [Google Scholar] [CrossRef]
- Cao, L.; Yu, I.K.; Liu, Y.; Ruan, X.; Tsang, D.C.; Hunt, A.J.; Ok, Y.S.; Song, H.; Zhang, S. Lignin valorization for the production of renewable chemicals: State-of-the-art review and future prospects. Bioresour. Technol. 2018, 269, 465–475. [Google Scholar] [CrossRef]
- Grossman, A.; Wilfred, V. Lignin-Based Polymers and Nanomaterials. Curr. Opin. Biotechnol. 2019, 56, 112–120. [Google Scholar] [CrossRef]
- Kalami, S.; Arefmanesh, M.; Master, E.; Nejad, M. Replacing 100% of phenol in phenolic adhesive formulations with lignin. J. Appl. Polym. Sci. 2017, 134, 45124. [Google Scholar] [CrossRef]
- Solt, P.; Jääskeläinen, A.S.; Lingenfelter, P.; Konnerth, J.; van Herwijnen, H.W.G. Impact of Molecular Weight of Kraft Lignin on Adhesive Performance of Lignin-Based Phenol-Formaldehyde Resins. For. Prod. J. 2019, 68, 365–371. [Google Scholar]
- Wang, H.; Pu, Y.; Ragauskas, A.; Yang, B. From lignin to valuable products–strategies, challenges, and prospects. Bioresour. Technol. 2018, 271, 449–461. [Google Scholar] [CrossRef]
- Puziy, A.M.; Poddubnaya, O.I.; Sevastyanova, O. Carbon Materials from Technical Lignins: Recent Advances. Top Curr. Chem. 2018, 376, 95–128. [Google Scholar] [CrossRef]
- Luo, H.; Abu-Omar, M.M. Chemicals from Lignin Synthesis of Novel Nanocrystal Materials for Solar and Themoelectric Device Applications. View Project Catalytic Depolymerization of Lignin View Project; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar] [CrossRef]
- Aro, T.; Fatehi, P. Production and Application of Lignosulfonates and Sulfonated Lignin. ChemSusChem 2017, 10, 1861–1877. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, P.; Lintinen, K.; Hirvonen, J.T.; Kostiainen, M.A.; Santos, H.A. Properties and chemical modifications of lignin: Towards lignin-based nanomaterials for biomedical applications. Prog. Mater. Sci. 2018, 93, 233–269. [Google Scholar] [CrossRef]
- Thakur, V.K.; Thakur, M.K. Recent advances in green hydrogels from lignin: A review. Int. J. Biol. Macromol. 2015, 72, 834–847. [Google Scholar] [CrossRef] [PubMed]
- Feng, Q.; Chen, F.; Wu, H. Thermosensitive Lignin Hydrogel. BioResources 2011, 6, 4942–4952. [Google Scholar]
- Aso, T.; Koda, K.; Kubo, S.; Yamada, T.; Nakajima, I.; Uraki, Y. Preparation of Novel Lignin-Based Cement Dispersants from Isolated Lignins. J. Wood Chem. Technol. 2013, 33, 286–298. [Google Scholar] [CrossRef]
- Ouyang, X.; Ke, L.; Qiu, X.; Guo, Y.; Pang, Y. Sulfonation of Alkali Lignin and Its Potential Use in Dispersant for Cement. J. Dispers. Sci. Technol. 2009, 30, 1–6. [Google Scholar] [CrossRef]
- Ge, Y.; Li, Z. Application of Lignin and Its Derivatives in Adsorption of Heavy Metal Ions in Water: A Review. ACS Sustain. Chem. Eng. 2018, 6, 7181–7192. [Google Scholar] [CrossRef]
- Cayla, A.; Rault, F.; Giraud, S.; Salaün, F.; Fierro, V.; Celzard, A. PLA with Intumescent System Containing Lignin and Ammonium Polyphosphate for Flame Retardant Textile. Polymers 2016, 8, 331. [Google Scholar] [CrossRef] [PubMed]
- Tavares, L.B.; Boas, C.R.S.V.; Schleder, G.; Nacas, A.; Rosa, D.D.S.; Santos, L.T. Bio-based polyurethane prepared from Kraft lignin and modified castor oil. Express Polym. Lett. 2016, 10, 927–940. [Google Scholar] [CrossRef]
- Hatakeyama, H. Polyurethanes containing lignin. In Chemical Modification, Properties, and Usage of Lignin; Hu, T.Q., Ed.; Springer Science & Business Media: New York, NY, USA, 2002; p. 291. ISBN 978-1-4613-5173-3. [Google Scholar]
- Nakano, J.; Izuta, Y.; Orita, T.; Hatakeyama, H.; Kobashigawa, K.; Teruya, K.; Hirose, S. Thermal and Mechanical Properties of Polyurethanes Derived from Fractionated Kraft Lignin. Sen’i Gakkaishi 1997, 53, 416–422. [Google Scholar] [CrossRef]
- Huang, J.; Fu, S.; Gan, L. Introduction. In Lignin Chemistry and Applications; Huang, J., Fu, S., Gan, L., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 1–24. [Google Scholar]
- Meister, J.J. Modification of lignin. J. Macromol. Sci. Part C Polym. Rev. 2002, 42, 235–289. [Google Scholar] [CrossRef]
- Tribot, A.; Amer, G.; Alio, M.A.; de Baynast, H.; Delattre, C.; Pons, A.; Mathias, J.-D.; Callois, J.-M.; Vial, C.; Michaud, P.; et al. Wood-lignin: Supply, extraction processes and use as bio-based material. Eur. Polym. J. 2019, 112, 228–240. [Google Scholar] [CrossRef]
- Sun, Z.; Fridrich, B.; de Santi, A.; Elangovan, S.; Barta, K. Bright Side of Lignin Depolymerization: Toward New Platform Chemicals. Chem. Rev. 2018, 118, 614–678. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Ferdosian, F. Green Chemistry and Sustainable Technology Conversion of Lignin into Bio-Based Chemicals and Materials. In Conversion of Lignin into Bio-Based Chemicals and Materials; Springer: New York, NY, USA, 2017. [Google Scholar]
- Cateto, C.; Barreiro, M.; Rodrigues, A. Monitoring of lignin-based polyurethane synthesis by FTIR-ATR. Ind. Crop. Prod. 2008, 27, 168–174. [Google Scholar] [CrossRef]
- Delmas, G.-H.; Benjelloun-Mlayah, B.; Le Bigot, Y.; Delmas, M. Biolignin™ based epoxy resins. J. Appl. Polym. Sci. 2013, 127, 1863–1872. [Google Scholar] [CrossRef]
- Cateto, C.A.; Barreiro, M.F.; Rodrigues, A.E.; Belgacem, M.N. Kinetic study of the formation of lignin-based polyurethanes in bulk. React. Funct. Polym. 2011, 71, 863–869. [Google Scholar] [CrossRef]
- Belgacem, M.N.; Blayo, A.; Gandini, A. Organosolv lignin as a filler in inks, varnishes and paints. Ind. Crop. Prod. 2003, 18, 145–153. [Google Scholar] [CrossRef]
- Hajirahimkhan, S. Chemical Modification of Lignin into Advanced Materials. Ph.D. Thesis, The University of Western Ontario, London, ON, Canada, 2018. [Google Scholar]
- Gellerstedt, G.; Majtnerova, A.; Zhang, L. Towards a new concept of lignin condensation in kraft pulping. Initial results. Comptes Rendus. Biol. 2004, 327, 817–826. [Google Scholar] [CrossRef] [PubMed]
- Pérez, J.; Muñoz-Dorado, J.; De La Rubia, T.; Martínez, J. Biodegradation and biological treatments of cellulose, hemicellulose and lignin: An overview. Int. Microbiol. 2002, 5, 53–63. [Google Scholar] [CrossRef]
- Tian, M.; Wen, J.; MacDonald, D.; Asmussen, R.M.; Chen, A. A novel approach for lignin modification and degradation. Electrochem. Commun. 2010, 12, 527–530. [Google Scholar] [CrossRef]
- Kaneko, M.; Nemoto, J.; Ueno, H.; Gokan, N.; Ohnuki, K.; Horikawa, M.; Saito, R.; Shibata, T. Photoelectrochemical reaction of biomass and bio-related compounds with nanoporous TiO2 film photoanode and O2-reducing cathode. Electrochem. Commun. 2006, 8, 336–340. [Google Scholar] [CrossRef]
- Antunes, C.S.A.; Bietti, M.; Salamone, M.; Scione, N. Early stages in the TiO2-photocatalyzed degradation of simple phenolic and non-phenolic lignin model compounds. J. Photochem. Photobiol. A Chem. 2004, 163, 453–462. [Google Scholar] [CrossRef]
- Wool, R.P.; Sun, X. Bio-Based Polymers and Composites; Elsevier Academic Press: Amsterdam, The Netherlands, 2005; ISBN 9780080454344. [Google Scholar]
- Acha, B.A.; Marcovich, N.E.; Reboredo, M.M. Lignin in jute fabric-polypropylene composites. J. Appl. Polym. Sci. 2009, 113, 1480–1487. [Google Scholar] [CrossRef]
- Raiskila, S. The Effect of Lignin Content and Lignin Modification on Norway Spruce Wood Properties and Decay Resistance. Ph.D Thesis, University of Helsinki, Helsinki, Finland, 2008. [Google Scholar]
- Simionescu, C.; Rusan, V.; Macoveanu, M.M.; Cazacu, G.; Lipsa, R.; Vasile, C.; Stoleriu, A.; Ioanid, A. Lignin/epoxy composites. Compos. Sci. Technol. 1993, 48, 317–323. [Google Scholar] [CrossRef]
- el Mansouri, N.E.; Yuan, Q.; Huang, F. Characterization of Alkaline Lignins for Use in Phenol-Formaldehyde and Epoxy Resins. Bioresources 2011, 6, 2647–2662. [Google Scholar]
- Wang, M.; Leitch, M.; Xu, C. Synthesis of phenol–formaldehyde resol resins using organosolv pine lignins. Eur. Polym. J. 2009, 45, 3380–3388. [Google Scholar] [CrossRef]
- Miyagawa, H.; Misra, M.; Drzal, L.T.; Mohanty, A.K. Biobased Epoxy/Layered Silicate Nanocomposites: Thermophysical Properties and Fracture Behavior Evaluation. J. Polym. Environ. 2005, 13, 87–96. [Google Scholar] [CrossRef]
- Evtugin, D.V.; Gandini, A. Polyesters based on oxygen-organosolv lignin. Acta Polym. 1996, 47, 344–350. [Google Scholar] [CrossRef]
- Bonini, C.; D’Auria, M.; Emanuele, L.; Ferri, R.; Pucciariello, R.; Sabia, A.R. Polyurethanes and polyesters from lignin. J. Appl. Polym. Sci. 2005, 98, 1451–1456. [Google Scholar] [CrossRef]
- Pouteau, C.; Dole, P.; Cathala, B.; Averous, L.; Boquillon, N. Antioxidant properties of lignin in polypropylene. Polym. Degrad. Stab. 2003, 81, 9–18. [Google Scholar] [CrossRef]
- Domenek, S.; Louaifi, A.; Guinault, A.; Baumberger, S. Potential of Lignins as Antioxidant Additive in Active Biodegradable Packaging Materials. J. Polym. Environ. 2013, 21, 692–701. [Google Scholar] [CrossRef]
- Vinardell, M.; Ugartondo, V.; Mitjans, M. Potential applications of antioxidant lignins from different sources. Ind. Crop. Prod. 2008, 27, 220–223. [Google Scholar] [CrossRef]
- Falkehag, S. Lignin in Materials. J. Appl. Polym. Sci. 1975, 28, 247–257. [Google Scholar]
- De Chirico, A.; Armanini, M.; Chini, P.; Cioccolo, G.; Provasoli, F.; Audisio, G. Flame retardants for polypropylene based on lignin. Polym. Degrad. Stab. 2003, 79, 139–145. [Google Scholar] [CrossRef]
- Yu, L.; Dean, K.; Li, L. Polymer blends and composites from renewable resources. Prog. Polym. Sci. 2006, 31, 576–602. [Google Scholar] [CrossRef]
- Bismarck, A.; Baltazar-Y-Jimenez, A.; Sarikakis, K. Green Composites as Panacea? Socio-Economic Aspects of Green Materials. Environ. Dev. Sustain. 2006, 8, 445–463. [Google Scholar] [CrossRef]
- Pouteau, C.; Baumberger, S.; Cathala, B.; Dole, P. Lignin–polymer blends: Evaluation of compatibility by image analysis. Comptes Rendus. Biol. 2004, 327, 935–943. [Google Scholar] [CrossRef] [PubMed]
- Laurichesse, S.; Avérous, L. Chemical modification of lignins: Towards biobased polymers. Prog. Polym. Sci. 2014, 39, 1266–1290. [Google Scholar] [CrossRef]
- Li, Q.; Xie, S.; Serem, W.K.; Naik, M.T.; Liu, L.; Yuan, J.S. Quality carbon fibers from fractionated lignin. Green Chem. 2017, 19, 1628–1634. [Google Scholar] [CrossRef]
- Xie, S.; Li, Q.; Karki, P.; Zhou, F.; Yuan, J.S. Lignin as Renewable and Superior Asphalt Binder Modifier. ACS Sustain. Chem. Eng. 2017, 5, 2817–2823. [Google Scholar] [CrossRef]
- Brodin, I.; Sjöholm, E.; Gellerstedt, G. Kraft lignin as feedstock for chemical products: The effects of membrane filtration. Holzforschung 2009, 63, 290–297. [Google Scholar] [CrossRef]
- Sevastyanova, O.; Helander, M.; Chowdhury, S.; Lange, H.; Wedin, H.; Zhang, L.; Ek, M.; Kadla, J.F.; Crestini, C.; Lindström, M.E. Tailoring the molecular and thermo-mechanical properties of kraft lignin by ultrafiltration. J. Appl. Polym. Sci. 2014, 131, 40799. [Google Scholar] [CrossRef]
- Li, Q.; Naik, M.T.; Lin, H.-S.; Hu, C.; Serem, W.K.; Liu, L.; Karki, P.; Zhou, F.; Yuan, J.S. Tuning hydroxyl groups for quality carbon fiber of lignin. Carbon 2018, 139, 500–511. [Google Scholar] [CrossRef]
- Jönsson, A.-S.; Nordin, A.-K.; Wallberg, O. Concentration and purification of lignin in hardwood kraft pulping liquor by ultrafiltration and nanofiltration. Chem. Eng. Res. Des. 2008, 86, 1271–1280. [Google Scholar] [CrossRef]
- Holmqvist, A.; Wallberg, O.; Jönsson, A.-S. Ultrafiltration of Kraft Black Liquor from Two Swedish Pulp Mills. Chem. Eng. Res. Des. 2005, 83, 994–999. [Google Scholar] [CrossRef]
- Klett, A.S. Purification, Fractionation, and Characterization of Lignin from Kraft Black Liquor for Use as a Renewable Biomaterial. Ph.D Thesis, Clemson University, Clemson, SC, USA, 2017. [Google Scholar]
- Compere, A.L.; Griffith, W.L.; Leitten, C.F.; Shaffer, J.T. Low Cost Carbon Fiber from Renewable Resources. In Proceedings of the International SAMPE Technical Conference, Oak Ridge, TN, USA, 10 August 2001. [Google Scholar]
- Pineda, A.; Lee, A.F. Heterogeneously catalyzed lignin depolymerization. Appl. Petrochem. Res. 2016, 6, 243–256. [Google Scholar] [CrossRef] [PubMed]
- Joffres, B.; Laurenti, D.; Charon, N.; Daudin, A.; Quignard, A.; Geantet, C. Thermochemical Conversion of Lignin for Fuels and Chemicals: A Review. Oil Gas Sci. Technol.—Rev. d’IFP Energ. Nouv. 2013, 68, 753–763. [Google Scholar] [CrossRef]
- Wang, B.; Sun, Y.-C.; Sun, R.-C. Fractionational and structural characterization of lignin and its modification as biosorbents for efficient removal of chromium from wastewater: A review. J. Leather Sci. Eng. 2019, 1, 1–25. [Google Scholar] [CrossRef]
- Lai, C.; Tu, M.; Xia, C.; Shi, Z.; Sun, S.; Yong, Q.; Yu, S. Lignin Alkylation Enhances Enzymatic Hydrolysis of Lignocellulosic Biomass. Energy Fuels 2017, 31, 12317–12326. [Google Scholar] [CrossRef]
- Rodriguez Couto, S.; Toca-Herrera, J.L. Industrial and biotechnological applications of laccases: A review. Biotechnol. Adv. 2006, 24, 500–513. [Google Scholar] [CrossRef]
- Xu, Q.; Qin, M.; Shi, S.; Jin, L.; Fu, Y. Structural changes in lignin during the deinking of old newsprint with laccase–violuric acid system. Enzym. Microb. Technol. 2006, 39, 969–975. [Google Scholar] [CrossRef]
- Li, Y.; Zhao, R.; Pang, Y.; Qiu, X.; Yang, D. Microwave-assisted synthesis of high carboxyl content of lignin for enhancing adsorption of lead. Colloids Surf. A Physicochem. Eng. Asp. 2018, 553, 187–194. [Google Scholar] [CrossRef]
- Shweta, K.; Jha, H. Synthesis and characterization of crystalline carboxymethylated lignin–TEOS nanocomposites for metal adsorption and antibacterial activity. Bioresour. Bioprocess. 2016, 3, 1–16. [Google Scholar] [CrossRef]
- Lignin (Alkaline), TCI America|Fisher Scientific. Available online: https://www.fishersci.com/shop/products/lignin-alkaline-tci-america-2/L008225G (accessed on 26 August 2022).
- Lignin, Alkali—Lignin, Kraft. Available online: https://www.sigmaaldrich.com/SK/en/substance/ligninalkali123458068051?gclid=EAIaIQobChMI3MXqg8zk-QIVRwGLCh289w9wEAAYASAAEgJoc_D_BwE (accessed on 26 August 2022).
- Fraunhofer ISI; Directorate-General for Research and Innovation(European Comission); University of Bologna. Detailed Case Studies on the Top 20 Innovative Bio-Based Products. In Top 20 Innovative Bio-Based Products; EU Publications: Luxembourg, 2018; pp. 53–242. ISBN 9789276034209. [Google Scholar]
- Väisänen, T.; Das, O.; Tomppo, L. A review on new bio-based constituents for natural fiber-polymer composites. J. Clean. Prod. 2017, 149, 582–596. [Google Scholar] [CrossRef]
- Trebostad, M. Renewable Chemicals from Lignin-A Conceptual Evaluation of Thermochemical Processes for Production of Valuable Chemicals from Lignin. Master’s Thesis, Norwegian University of Life Science, As, Norway, 2016. [Google Scholar]
- Xylene, Xylene Suppliers and Manufacturers at Alibaba.Com. Available online: https://www.alibaba.com/showroom/xylene.html?fsb=y&IndexArea=product_en&CatId=&SearchText=xylene&isGalleryList=G (accessed on 26 November 2020).
- Bajwa, D.S.; Pourhashem, G.; Ullah, A.H.; Bajwa, S.G. A concise review of current lignin production, applications, products and their environmental impact. Ind. Crop. Prod. 2019, 139, 111526. [Google Scholar] [CrossRef]
- Agrawal, A.; Kaushik, N.; Biswas, S. Derivatives and Applications of Lignin—An Insight. SciTech J. 2014, 1, 30–36. [Google Scholar]
- Gosselink, R.; de Jong, E.; Guran, B.; Abächerli, A. Co-ordination network for lignin—Standardisation, production and applications adapted to market requirements (EUROLIGNIN). Ind. Crop. Prod. 2004, 20, 121–129. [Google Scholar] [CrossRef]
- Stewart, D. Lignin as a base material for materials applications: Chemistry, application and economics. Ind. Crop. Prod. 2008, 27, 202–207. [Google Scholar] [CrossRef]
- Park, Y.; Doherty, W.; Halley, P. Developing lignin-based resin coatings and composites. Ind. Crop. Prod. 2008, 27, 163–167. [Google Scholar] [CrossRef]
- Siddiqui, H. Production of Lignin-Based Phenolic Resins Using De-Polymerized Kraft Lignin and Process Optimization. Master’s Thesis, The University of Western Ontario, London, ON, Canada, 2013. [Google Scholar]
- Phenolic Resin, Phenolic Resin Suppliers and Manufacturers at Alibaba.Com. Available online: https://www.alibaba.com/showroom/phenolic+resin.html?fsb=y&IndexArea=product_en&CatId=&SearchText=phenolic+resin&isGalleryList=G (accessed on 26 November 2020).
- Triantafyllidis, K.S.; Lappas, A.A.; Stöcker, M. (Eds.) The Role of Catalysis for the Sustainable Production of Bio-Fuels and Bio-Chemicals, 1st ed.; Elsevier: Amsterdam, The Netherlands; Boston, MA, USA, 2013; ISBN 978-0-444-56330-9. [Google Scholar]
- Collard, F.-X.; Blin, J. A review on pyrolysis of biomass constituents: Mechanisms and composition of the products obtained from the conversion of cellulose, hemicelluloses and lignin. Renew. Sustain. Energy Rev. 2014, 38, 594–608. [Google Scholar] [CrossRef]
- Gill, A.S.; Visotsky, D.; Mears, L.; Summers, J.D. Cost Estimation Model for PAN Based Carbon Fiber Manufacturing Process. In Proceedings of the International Manufacturing Science and Engineering Conference, Novy Smokovec, Slovakia, 6–10 June 2016. [Google Scholar] [CrossRef]
- High-Strength Carbon Fiber Price per Kg for Professional Uses—Alibaba.Com. Available online: https://www.alibaba.com/showroom/carbon-fiber-price-per-kg.html (accessed on 26 November 2020).
- Kubo, S.; Kadla, J.F. Lignin-based Carbon Fibers: Effect of Synthetic Polymer Blending on Fiber Properties. J. Polym. Environ. 2005, 13, 97–105. [Google Scholar] [CrossRef]
- GreenLight—Cost Effective Lignin based Carbon Fibres for Innovative Light weight Applications. Available online: https://greenlight-project.eu/ (accessed on 26 November 2020).
- Xu, J.; Jiang, J.; Hse, C.-Y.; Shupe, T.F. Preparation of polyurethane foams using fractionated products in liquefied wood. J. Appl. Polym. Sci. 2013, 131, 40096. [Google Scholar] [CrossRef]
- Mahmood, N.; Yuan, Z.; Schmidt, J.; Xu, C. Depolymerization of lignins and their applications for the preparation of polyols and rigid polyurethane foams: A review. Renew. Sustain. Energy Rev. 2016, 60, 317–329. [Google Scholar] [CrossRef]
- Lim, H.; Kim, S.H.; Kim, B.K. Effects of silicon surfactant in rigid polyurethane foams. Express Polym. Lett. 2008, 2, 194–200. [Google Scholar] [CrossRef]
- Cinelli, P.; Anguillesi, I.; Lazzeri, A. Green synthesis of flexible polyurethane foams from liquefied lignin. Eur. Polym. J. 2013, 49, 1174–1184. [Google Scholar] [CrossRef]
- Mahmood, N.; Yuan, Z.; Schmidt, J.; Xu, C. Production of polyols via direct hydrolysis of kraft lignin: Effect of process parameters. Bioresour. Technol. 2013, 139, 13–20. [Google Scholar] [CrossRef]
- Polyurethane Foam, Polyurethane Foam Suppliers and Manufacturers at Alibaba.Com. Available online: https://www.alibaba.com/showroom/polyurethane+foam.html?fsb=y&IndexArea=product_en&CatId=&SearchText=polyurethane+foam&isGalleryList=G (accessed on 26 November 2020).
| Linkage Type | Dimer Structure | Approximate Percentage in Grasses | Approximate Percentage in Hardwood | Approximate Percentage in Softwood | References |
|---|---|---|---|---|---|
| β-O-4 | Phenylpropane β-aryl ether | 69–94 | 50–65 | 45–50 | [18,19,20,21] |
| α-O-4 | Phenylpropane α-aryl ether | 11–14 | 1–8 | 5–8 | [18,19,20,21] |
| β-5 | Phenylcoumaran | 5–11 | 3–11 | 9–12 | [18,19] |
| 5-5 | Biphenyl and dibenzodioxocin | n. i. | 1–5 | 5–25 | [18,19,20,21] |
| 4-O-5 | Diaryl ether | n. i. | 6–7 | 3.5–8 | [18,19,20,21] |
| β-1 | 1,2-Diaryl propane | 0–2 | 1–7 | 1–10 | [18,19,20,21] |
| β-β | β-β-linked structures | 1–15 | 3–12 | 2–6 | [18,19,20,21] |
| Functional groups of lignin | |||||
| Carbonyl [mol/ lignin unit] | - | 0.15 | 0.20 | [21] | |
| Aliphatic hydroxyl [mol/ lignin unit] | 0.56–1.13 | 1.10–1.15 | 1.15–1.20 | [21] | |
| Phenolic hydroxyl [mol/ lignin unit] | 0.36–0.44 | 0.10–0.20 | 0.20–0.30 | [21] | |
| Methoxy [mol/ lignin unit] | - | 1.40–1.60 | 0.90–0.95 | [21] | |
| Material Category | Products | Potential Applications | References |
|---|---|---|---|
| Aromatic macromolecules and fine chemicals | Lignin monomers and dimers, aromatic phenols, alkyl phenols, aromatic aldehydes, aromatic alcohols, acids, aryl ketones, antioxidants, dispersants, PU, phenolic resins, vanillin | Industrial chemicals, biobased adhesives, multifunctional materials, building blocks for biobased products | [22,23,24,25,26] |
| Carbon materials, Biofuels | Biochar, bio-oil, syngas, activated carbon, carbon fibers, carbon black | Light-weight polymer composites, adsorbents, electrochemical devices, automotive | [27,28] |
| Polymer and nanomaterials | 3D Printing resin (anionic surfactant), Scaffolds, lignin nanotubes, hydrogels | Biomedical applications, tissue engineering, drug delivery | [29,30,31] |
| Energy Storage | Li-ion, Na-ion batteries (electrodes), super capacitors, solar cells | Energy devices, batteries, fuels cells | [32] |
| Building Materials | Bitumen, cement additive, dispersant, reinforcement | Construction pavements, cement panels | [22,27] |
| Specialized Applications | Soil conditioner, in fertilizers and pesticides as controlled release agent, sequestering agent, material absorbing soil contaminants, fire retardant | Agriculture, textiles, soil reclamation, water purification, fire suppression | [22,27,33,34,35,36] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nadányi, R.; Ház, A.; Lisý, A.; Jablonský, M.; Šurina, I.; Majová, V.; Baco, A. Lignin Modifications, Applications, and Possible Market Prices. Energies 2022, 15, 6520. https://doi.org/10.3390/en15186520
Nadányi R, Ház A, Lisý A, Jablonský M, Šurina I, Majová V, Baco A. Lignin Modifications, Applications, and Possible Market Prices. Energies. 2022; 15(18):6520. https://doi.org/10.3390/en15186520
Chicago/Turabian StyleNadányi, Richard, Aleš Ház, Anton Lisý, Michal Jablonský, Igor Šurina, Veronika Majová, and Andrej Baco. 2022. "Lignin Modifications, Applications, and Possible Market Prices" Energies 15, no. 18: 6520. https://doi.org/10.3390/en15186520
APA StyleNadányi, R., Ház, A., Lisý, A., Jablonský, M., Šurina, I., Majová, V., & Baco, A. (2022). Lignin Modifications, Applications, and Possible Market Prices. Energies, 15(18), 6520. https://doi.org/10.3390/en15186520







