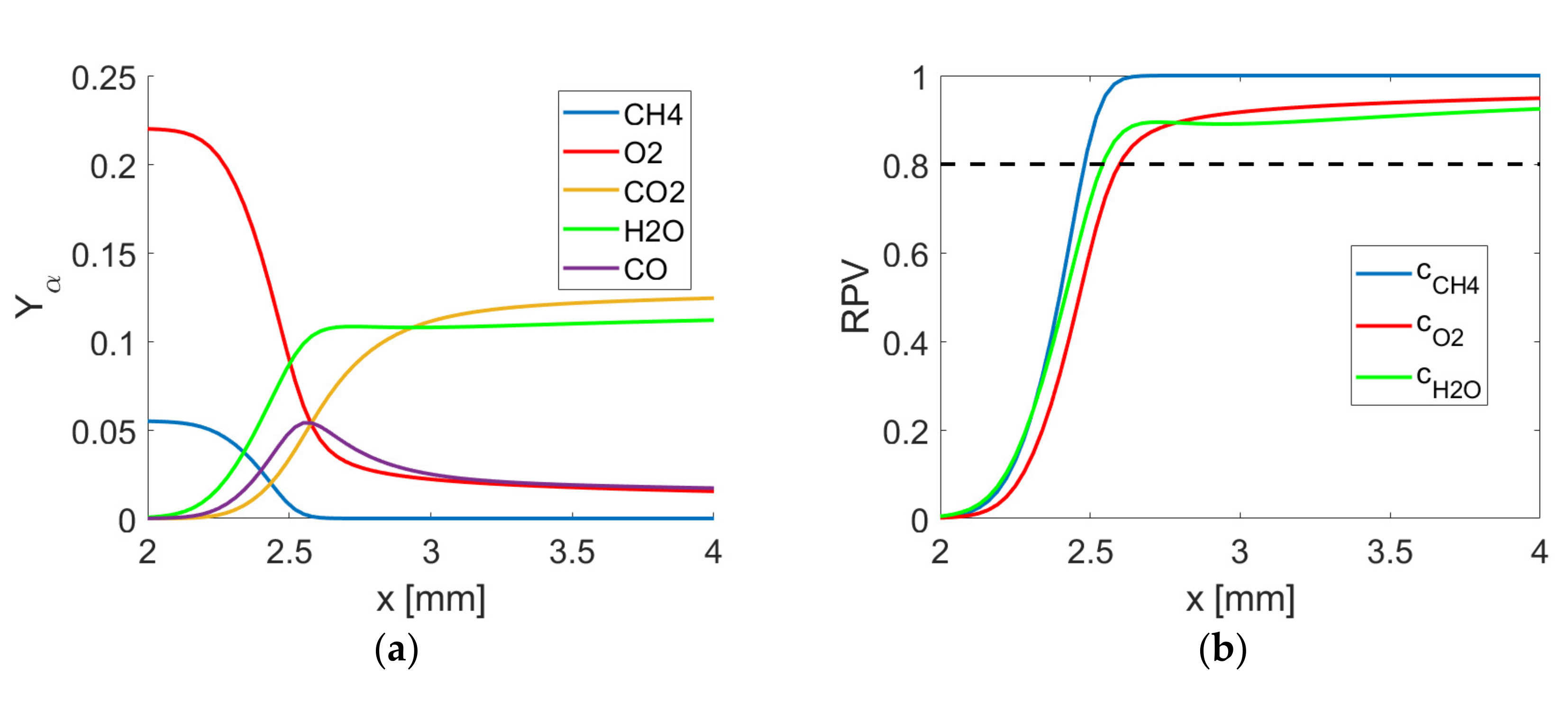
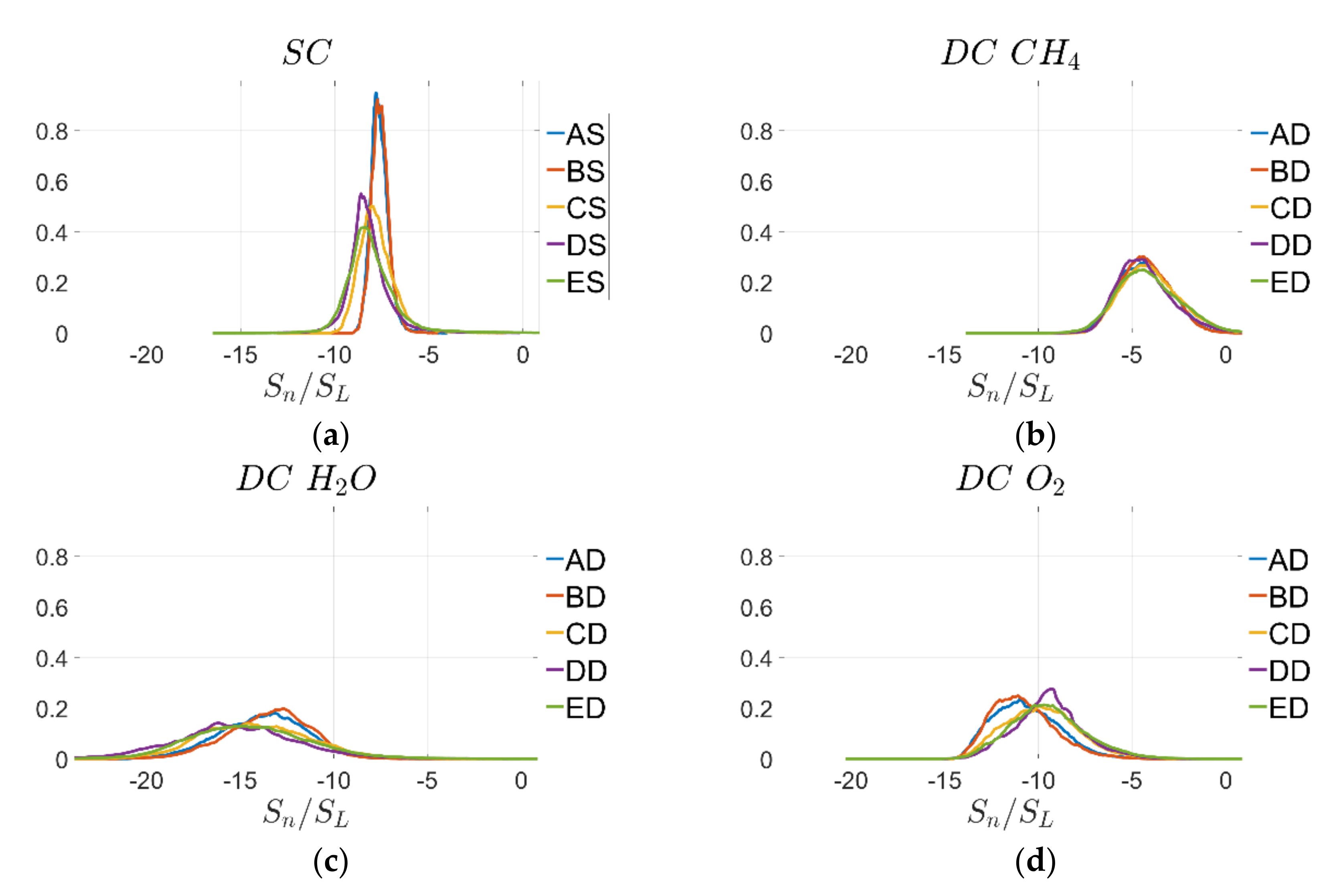
3.1. Mean Variation and Statistics of Displacement Speed and Its Components
Mean variations of normalised displacement speed
across the flame for all cases are shown in
Figure 5 for SC and DC using three different definitions for reaction progress variable based on
and
mass fractions. The variations between the different cases can be explained by stretch effects [
23] and are consistent for all cases shown in
Figure 5a–d. However, there are some non-negligible quantitative differences between the different definitions of
. The highest displacement speed values are obtained for the SC case and DC based on
mass fraction while the lowest values are achieved for
mass fraction.
An advantage of the mass fraction-based reaction progress variable definition is that the displacement speed can be evaluated throughout the flame without any problems, while the mass fraction-based definition becomes singular due to the non-monotonicity of -based reaction progress variable, as discussed earlier. Displacement speed defined by mass fraction-based reaction progress variable becomes difficult to evaluate towards the burned gas side because assumes very small values before reaches a value of unity. It has to be admitted that there are quantitative differences between displacement speed statistics based on SC and DC simulations, however, they are of the same magnitude as the differences based on DC simulations using different definitions of reaction progress variable.
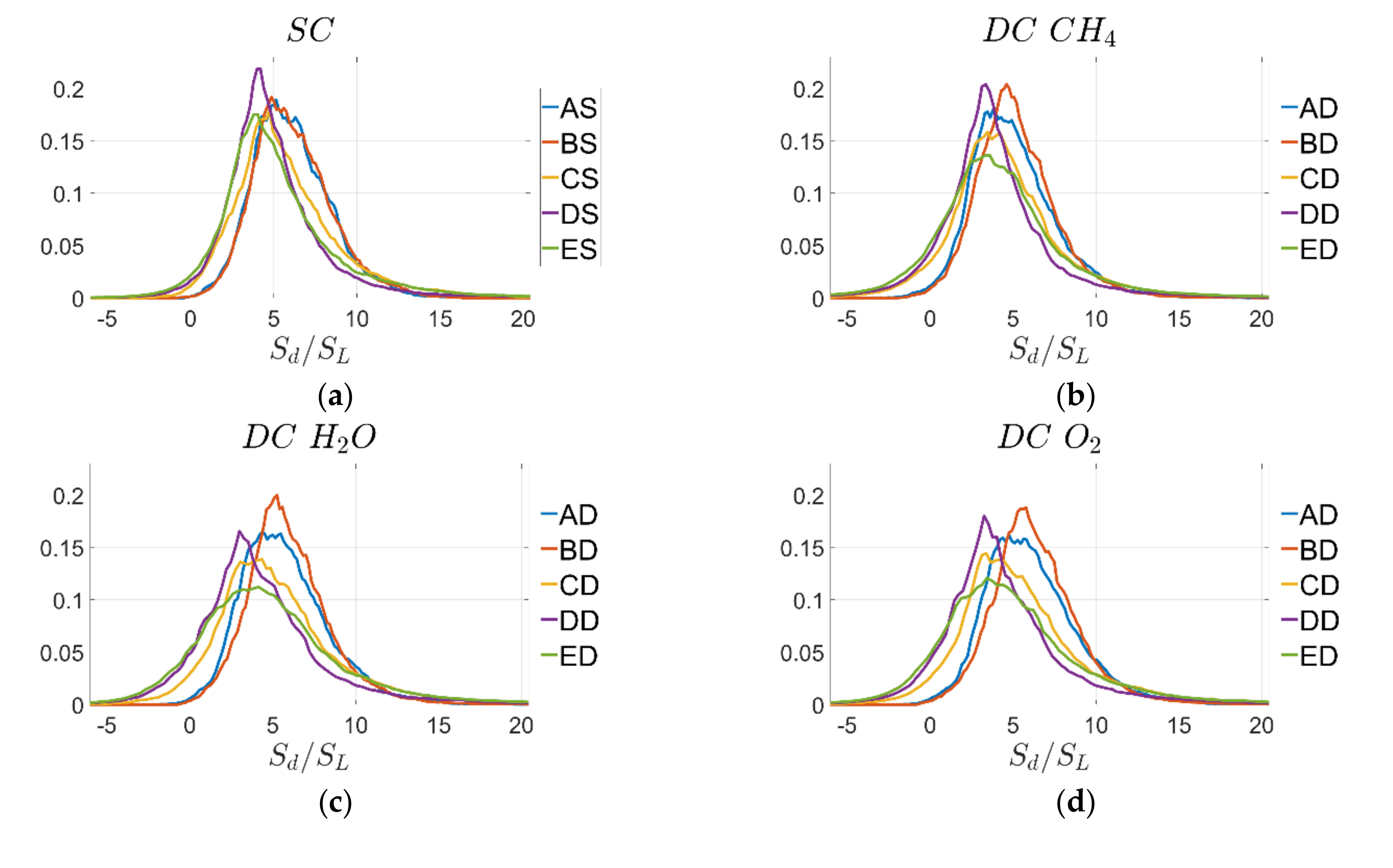
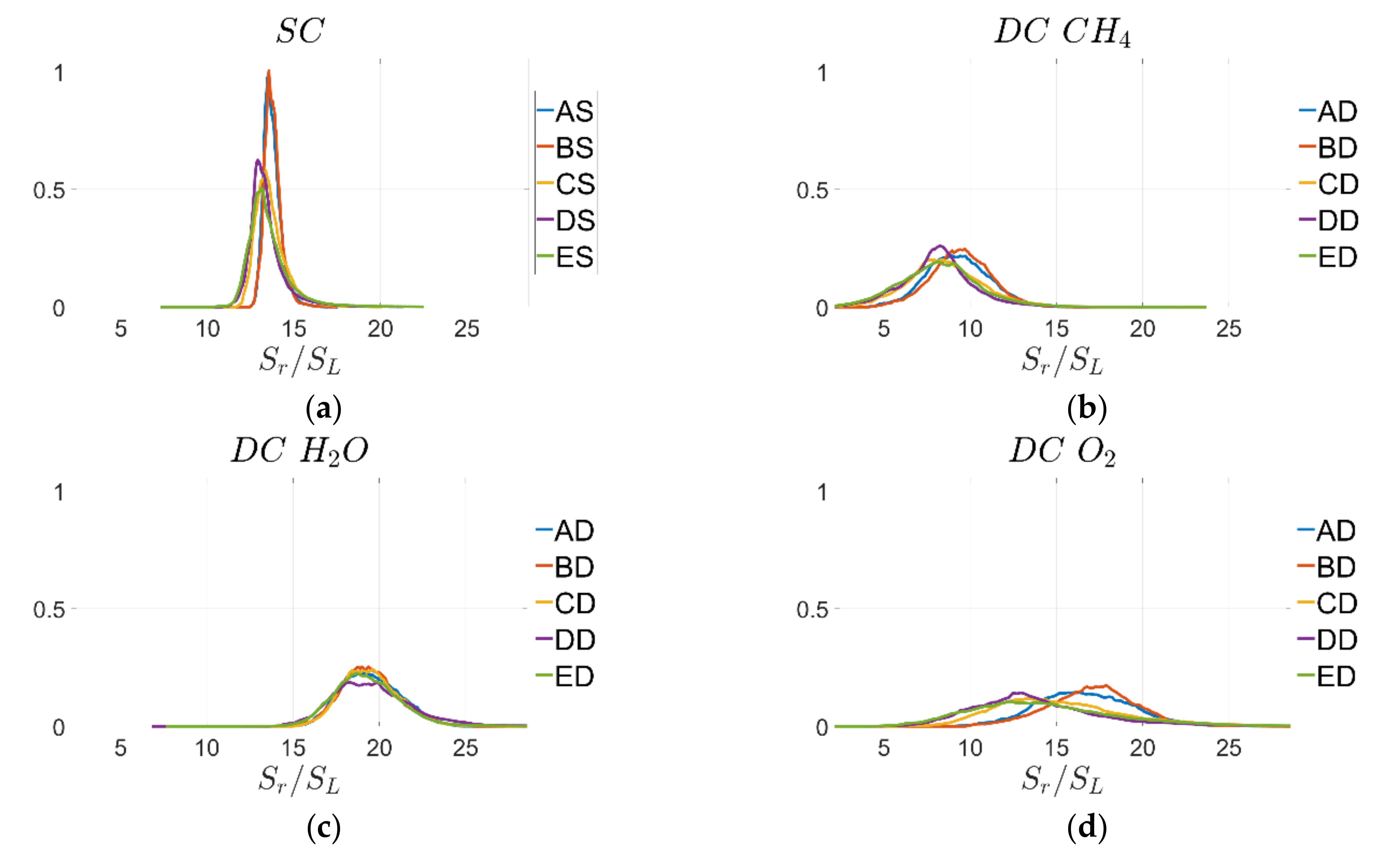
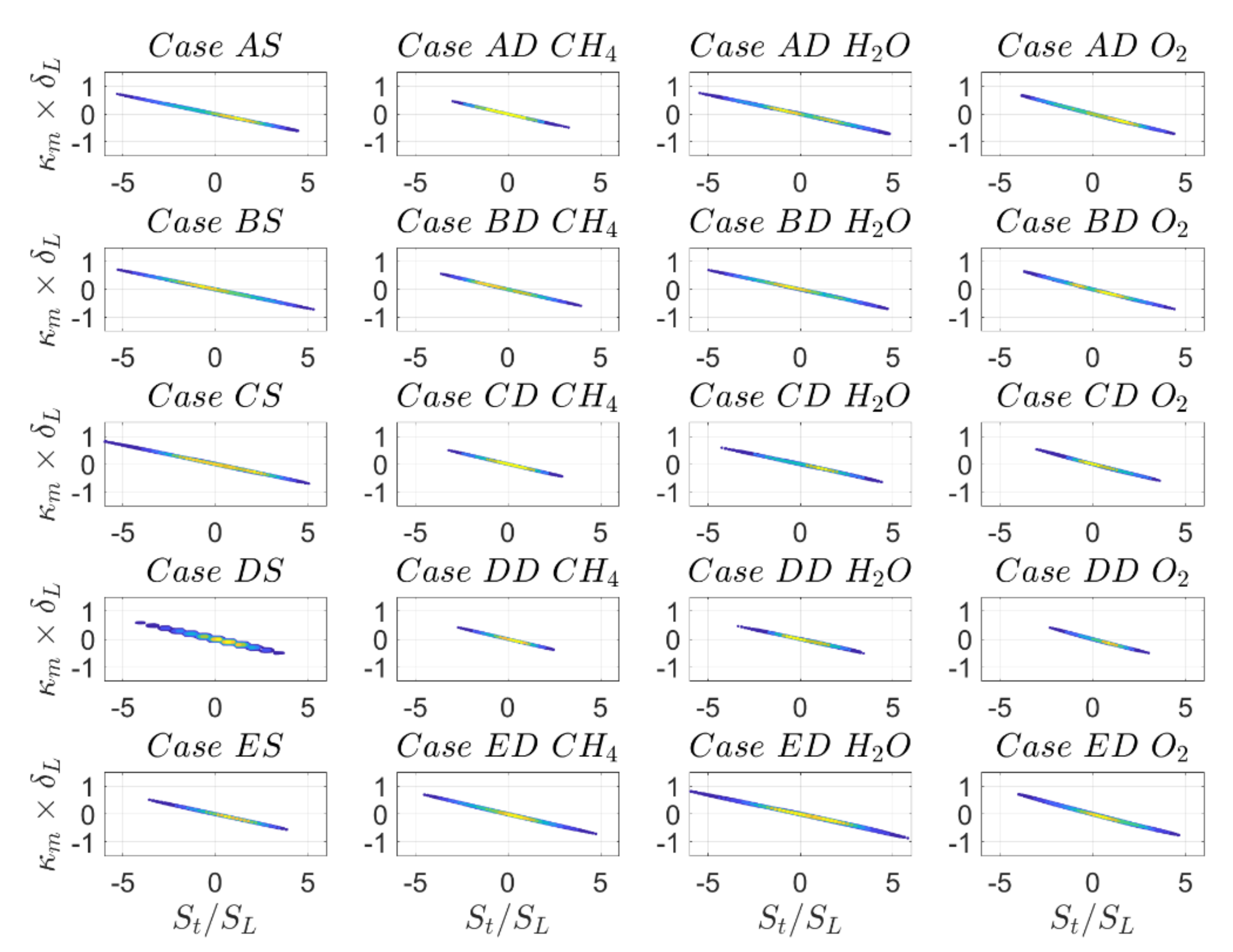
The behaviour observed from
Figure 5 is consistent with the probability density functions (PDF) of normalised displacement speed
on the
progress-variable isosurfaces, which are shown in
Figure 6 for cases A–E and the quantitative evaluation of mean
and standard deviation
of
for
are shown in
Table 6.
Table 6 indicates that the values of
can be split into three groups, cases A, B, cases C, D and case E, and this behaviour is consistent for all cases and in particular when comparing SC and DC.
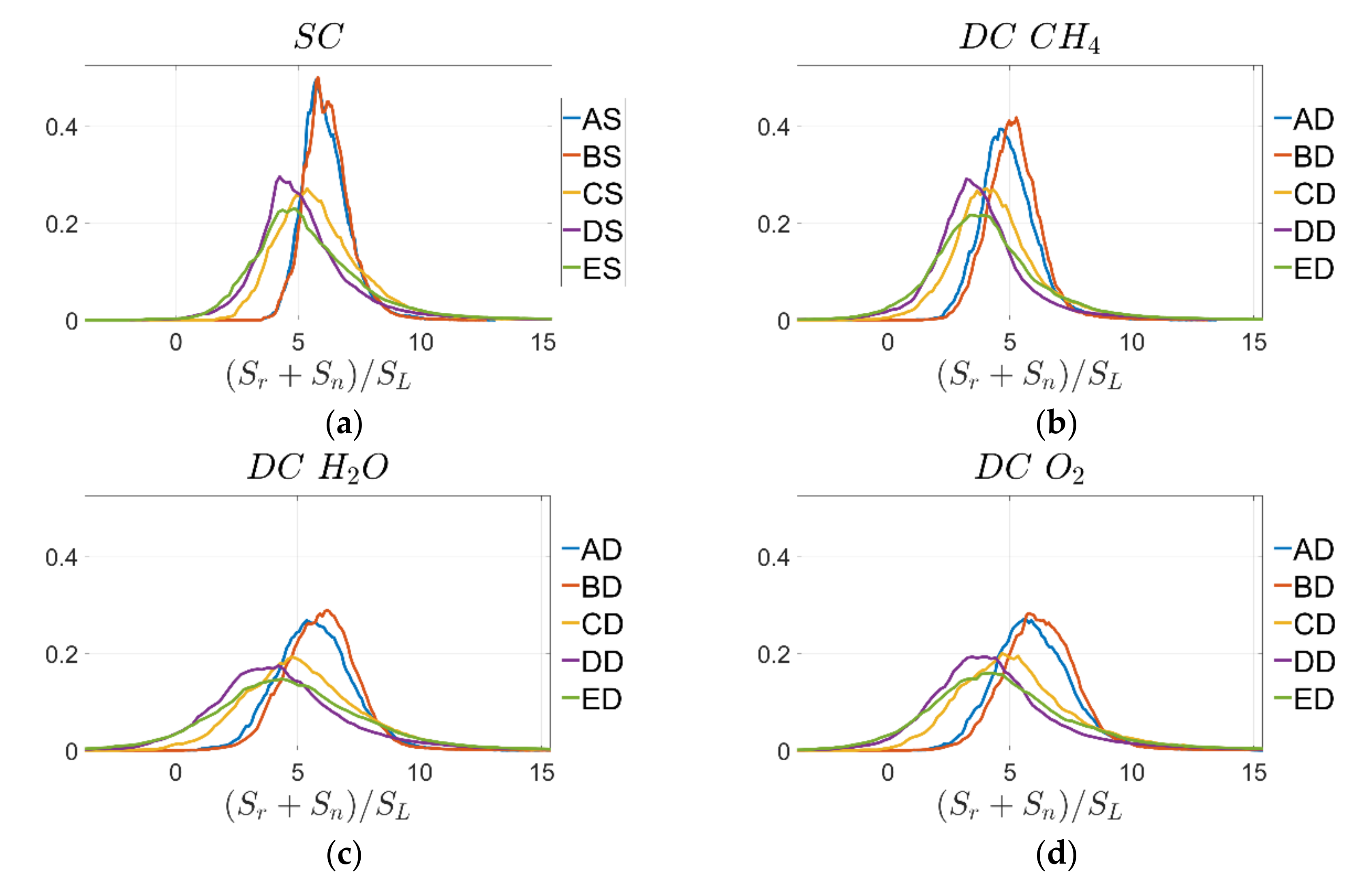
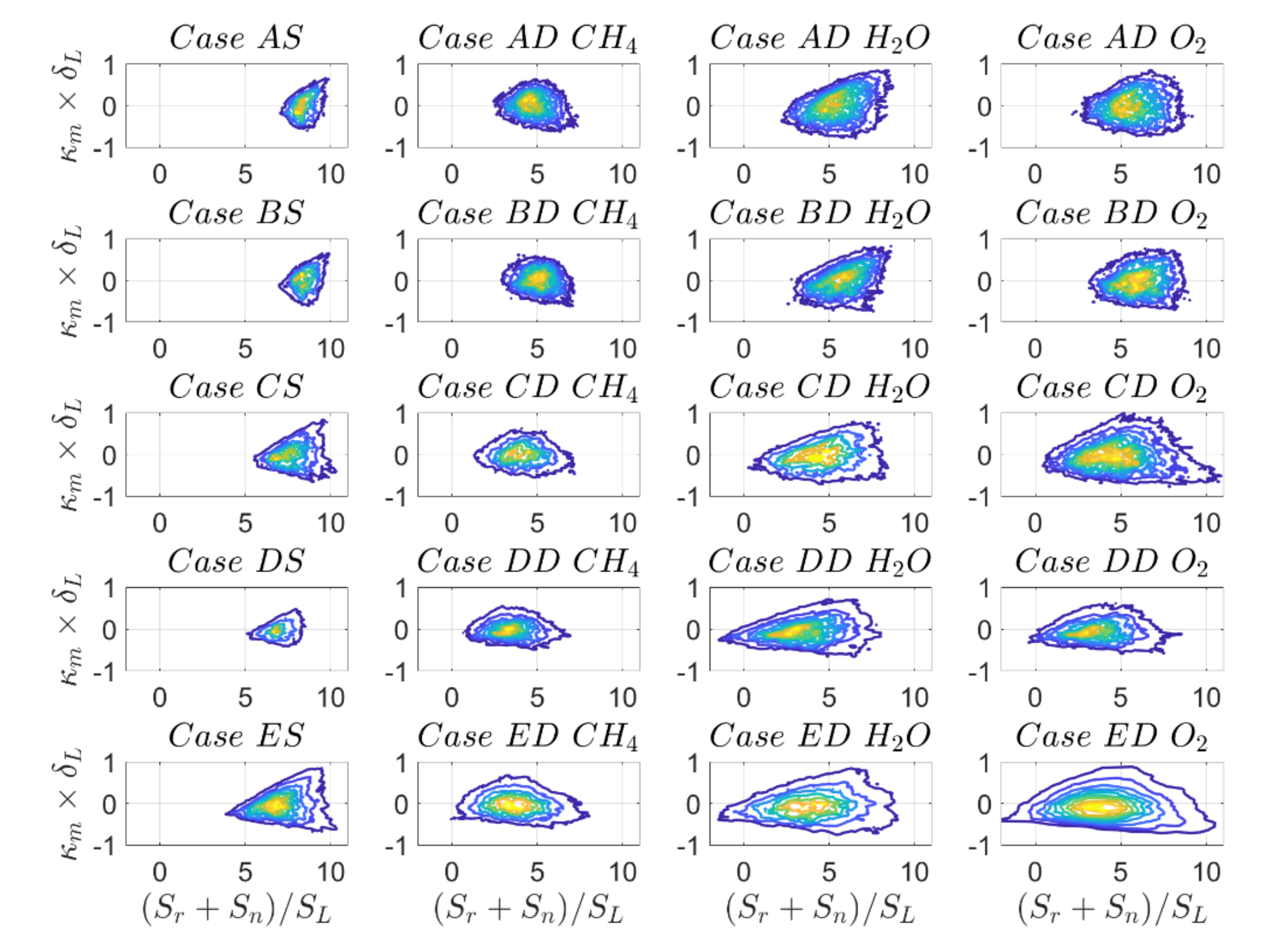
Similar trends can be observed for the PDF of normalised combined reaction and normal diffusion components of displacement speed
shown for the
isosurface in
Figure 7. It is noted that the DC PDFs for reaction progress variable based on
and
mass fractions tend to be wider than those for SC and DC based on
. This behaviour will be analysed in more detail by looking next at the individual contributions
, and the corresponding PDFs for
are shown in
Figure 7,
Figure 8 and
Figure 9, while the corresponding mean values and standard deviations for
and
are shown in
Table 7 and
Table 8. The mean value of
is close to zero for a statistically planar flame and therefore these numbers are not shown in the form of a table.
Figure 8 and
Table 7 indicate that the mean values of
are close to each other for all cases but there are differences based on the different definitions of reaction progress variable. This indicates that the differences in the PDFs of normal diffusion components are driven by the significant differences in the laminar profiles of reaction progress variable across the flame brush when using different species mass fractions for its definition as shown in
Figure 2b and
Figure 3a. It is also noted that the width of the PDFs of
tends to be larger for the DC cases compared to the SC simulations. A possible explanation is that
can be taken as a constant for SC simulations while
is temperature-dependent in the DC cases, and hence is likely to introduce additional variations for the DC setup due to the decoupling of reaction progress variable and temperature (see
Figure 3). Furthermore, as noted earlier, the SC simulations have higher viscosity and hence slightly smaller turbulence intensity at the time when statistics are taken.
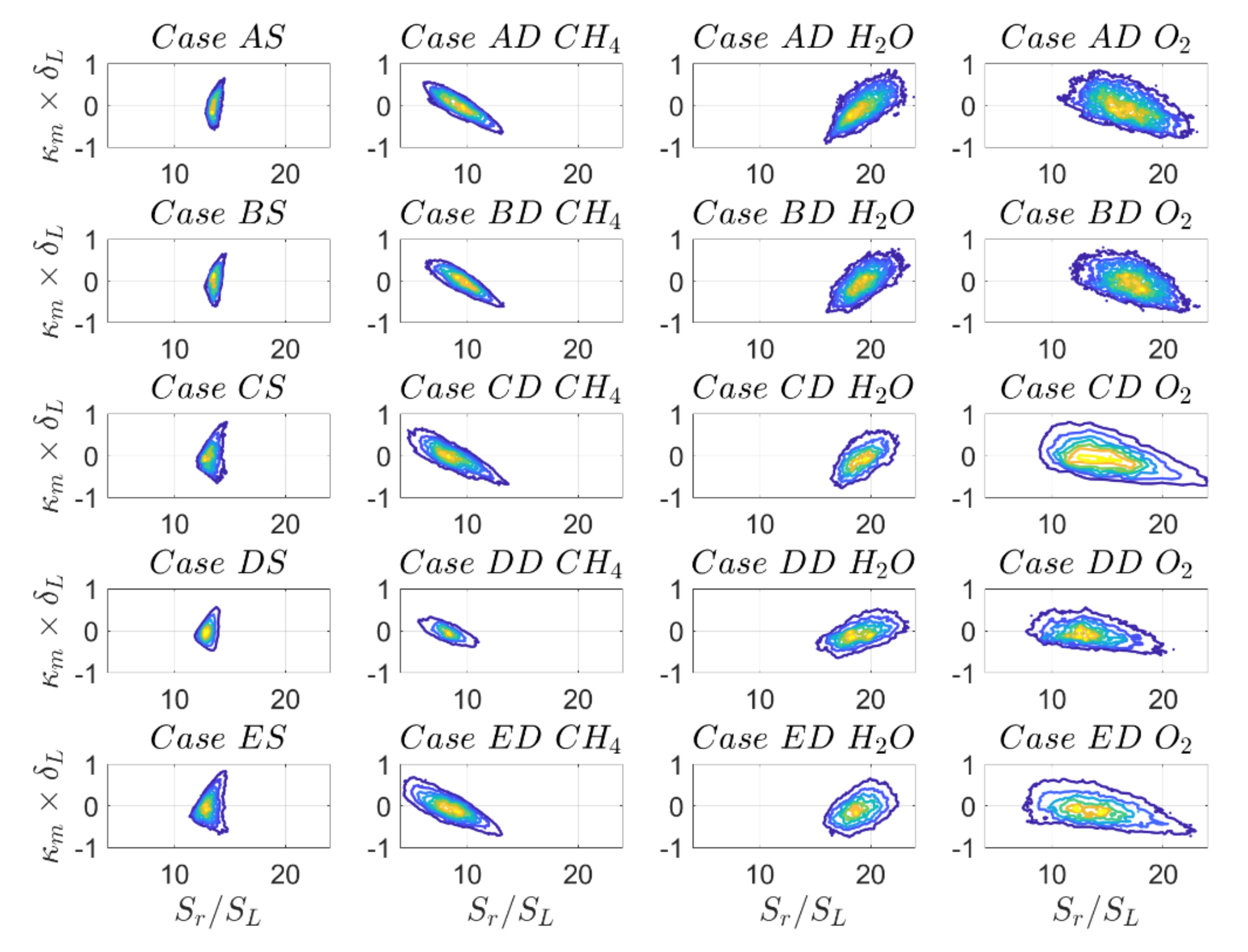
The PDFs of the normalised reaction component of displacement speed
for the
isosurface are shown in
Figure 9 and the first and second moments for the
reaction progress variable value are given in
Table 8. The mass fractions of
and
have different values at the location of maximum heat release. Thus, it is not surprising that they show moderate differences in mean values depending on the definition of reaction progress variable. For SC, the mean values of
are nearly identical for all cases considered here, while for the DC simulations there is more variability (in particular for reaction progress variable based on
and
mass fractions) depending on the initial turbulence intensity. This can be explained in the following manner. For simple chemistry, low Mach number and unity Lewis number simulations, reaction progress variable and non-dimensional temperature are identical and consequently the reaction rate
is a unique function of
. In contrast, for the DC case, the reaction rate of an individual species depends not only on temperature but also on the reaction progress of several other species. Furthermore, the standard enthalpy of formation of a pure element is zero which shows that
(and equally
) and
are not always directly related. Hence, a change of reaction progress variable in the DC case is less directly interlinked with a change in temperature. In all cases (including DC), the turbulent motion of the fluid causes variations of the surface density function
(which scales with the inverse flame thickness) in such a manner that the variance of
increases from case A to case E.
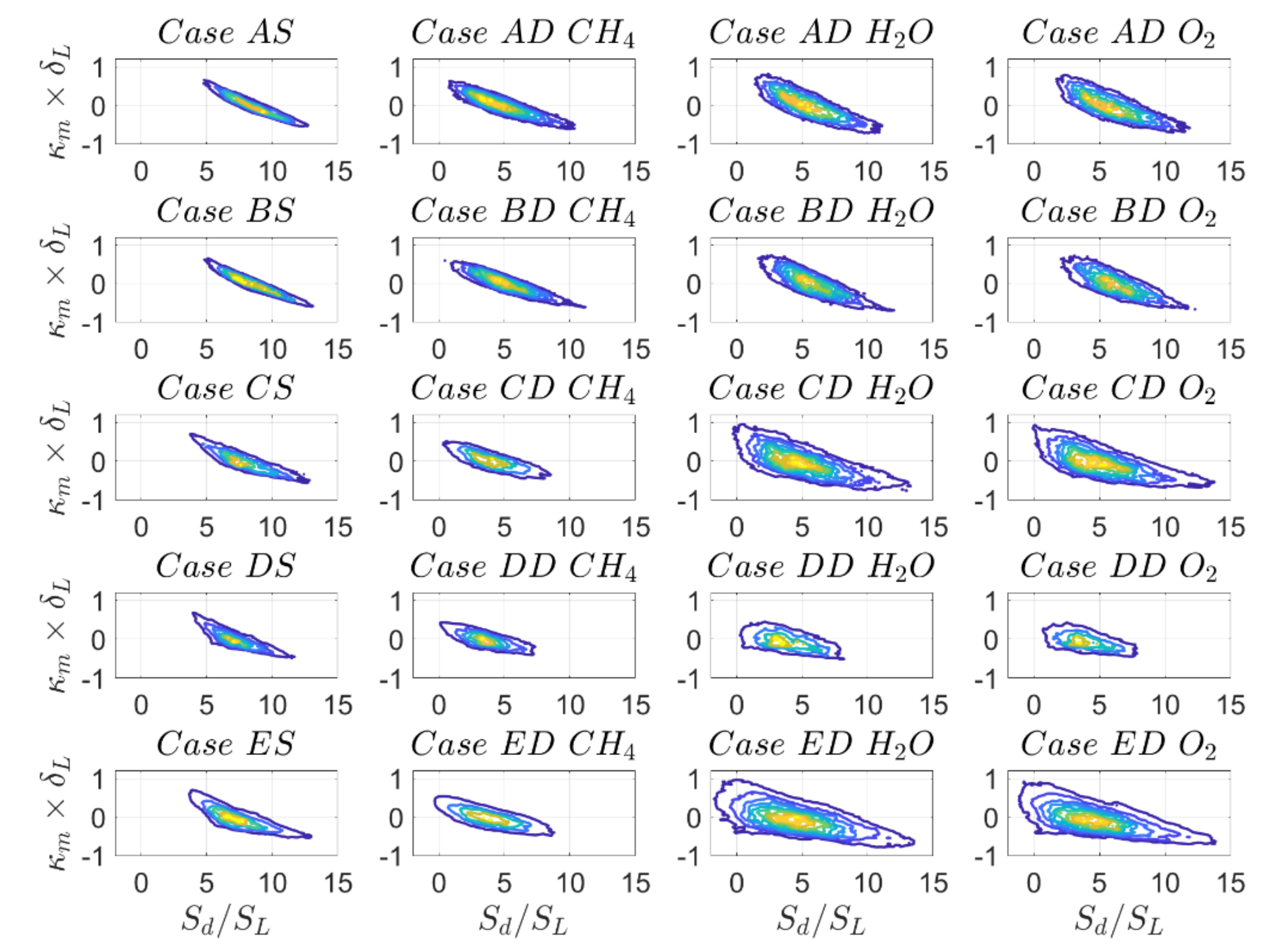
Despite the remarkable differences of the PDFs of
and
for the different definitions of reaction progress variable, the reaction–diffusion balance in the expression
remains qualitatively much more similar compared to the individual contributions as discussed before, because a more positive value of
is balanced by a more negative value of
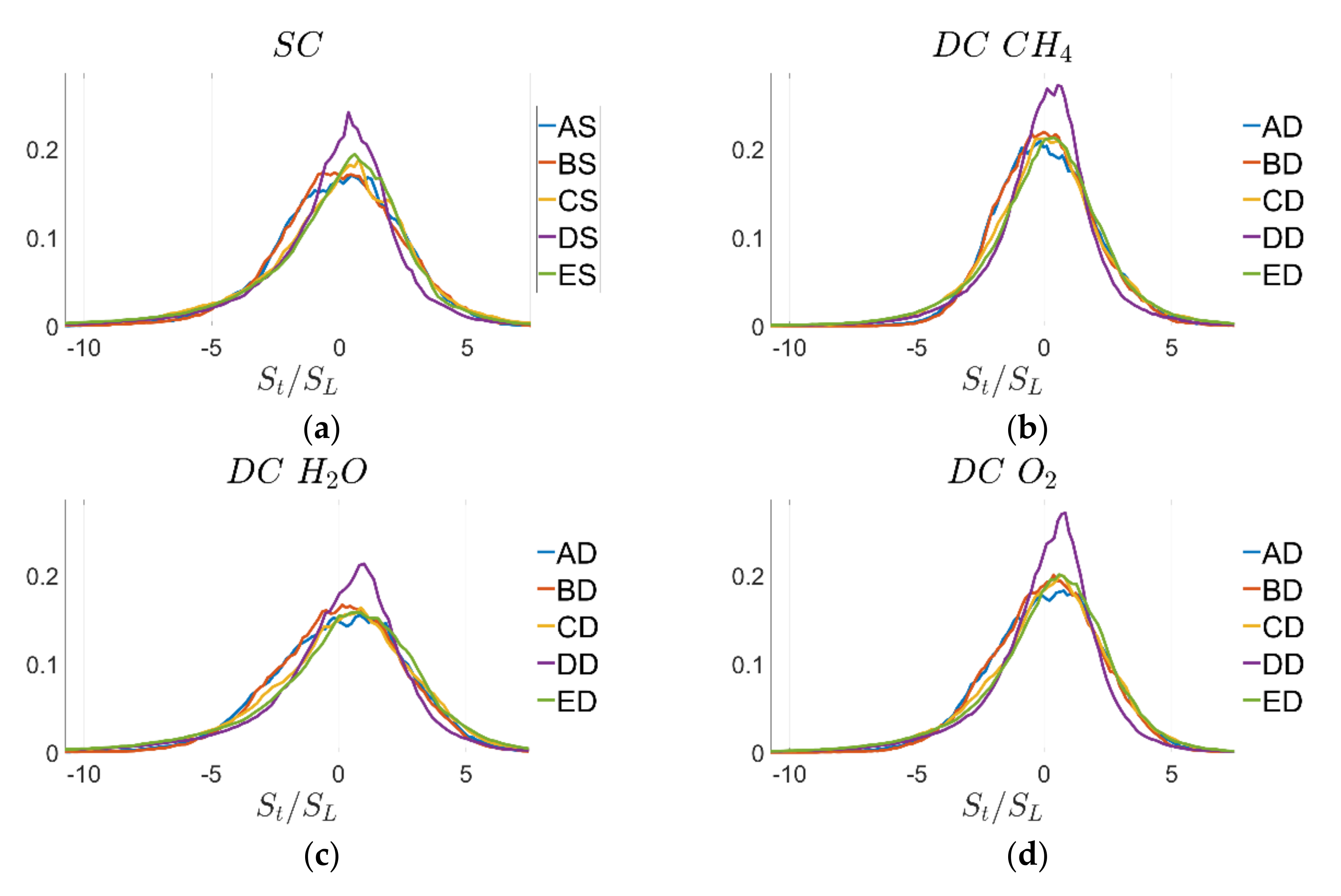
and vice versa. Finally, the PDFs of normalised tangential diffusion component of displacement speed
for the
isosurface are shown in
Figure 10. Due to the statistically planar nature of the flames considered here, their mean value is close to zero in all cases and the standard deviation has the same order of magnitude for all definitions of reaction progress variable but tends to increase with increasing turbulence intensity.
The statistics of remain qualitatively, and to a large extent quantitatively similar for SC and DC simulations and also for different definitions of reaction progress variable. However, there are marked qualitative differences for individual contributions of displacement speed and . To explain this behaviour, it is important to understand their interrelation with curvature and strain rate as well as the influence of the definition of reaction progress variable, which will be discussed in the next subsection.
3.2. Curvature and Tangential Strain Rate Effects on Local Displacement Speed and Its Components
Theoretical studies suggest that flame stretch is the controlling parameter of the flame structure in the limit of weak turbulence and weak flame wrinkling (for an overview see [
26,
47]). Flame stretch rate is, in turn, a function of flame curvature
and tangential strain rate
. The PDFs of
and
have been discussed in part 1 of this paper and do not provide additional insight here. Their joint PDFs are shown in
Figure 11 and the correlation coefficients between
and
are reported in
Table 9 for
.
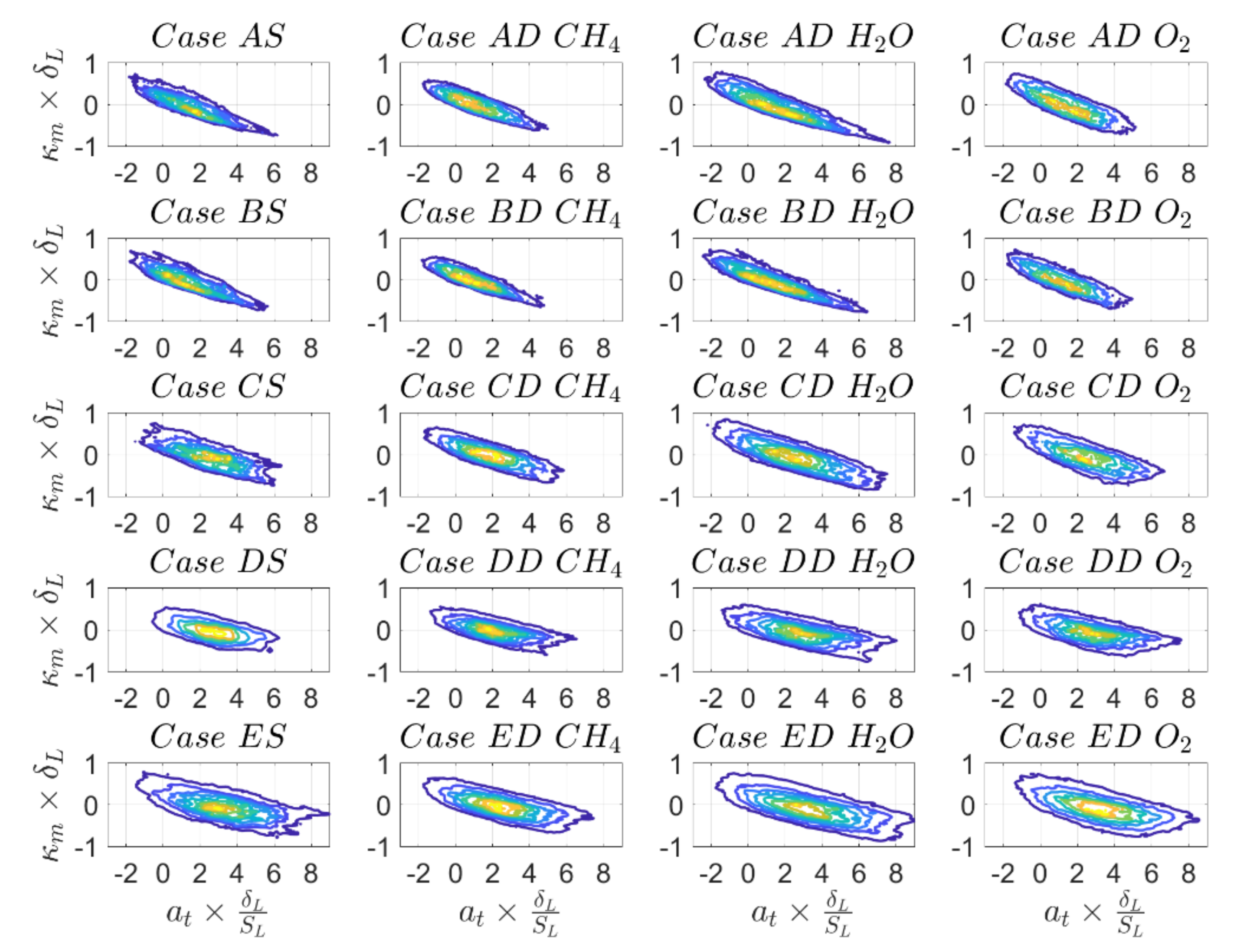
It can be seen from
Figure 11 that there is a negative correlation between
and
in all cases which is the strongest for cases A and B and weakest for cases D and E. The correlation strength is weakly dependent on the definition of reaction progress variable. These results are in very good agreement with an earlier analysis reported in [
49]. As
and
are not independent parameters, the dependence of
and its components with
can be deduced from the interrelation of
and its components with
and vice versa. Therefore, the following discussion is limited to correlations of displacement speed and its components with mean curvature.
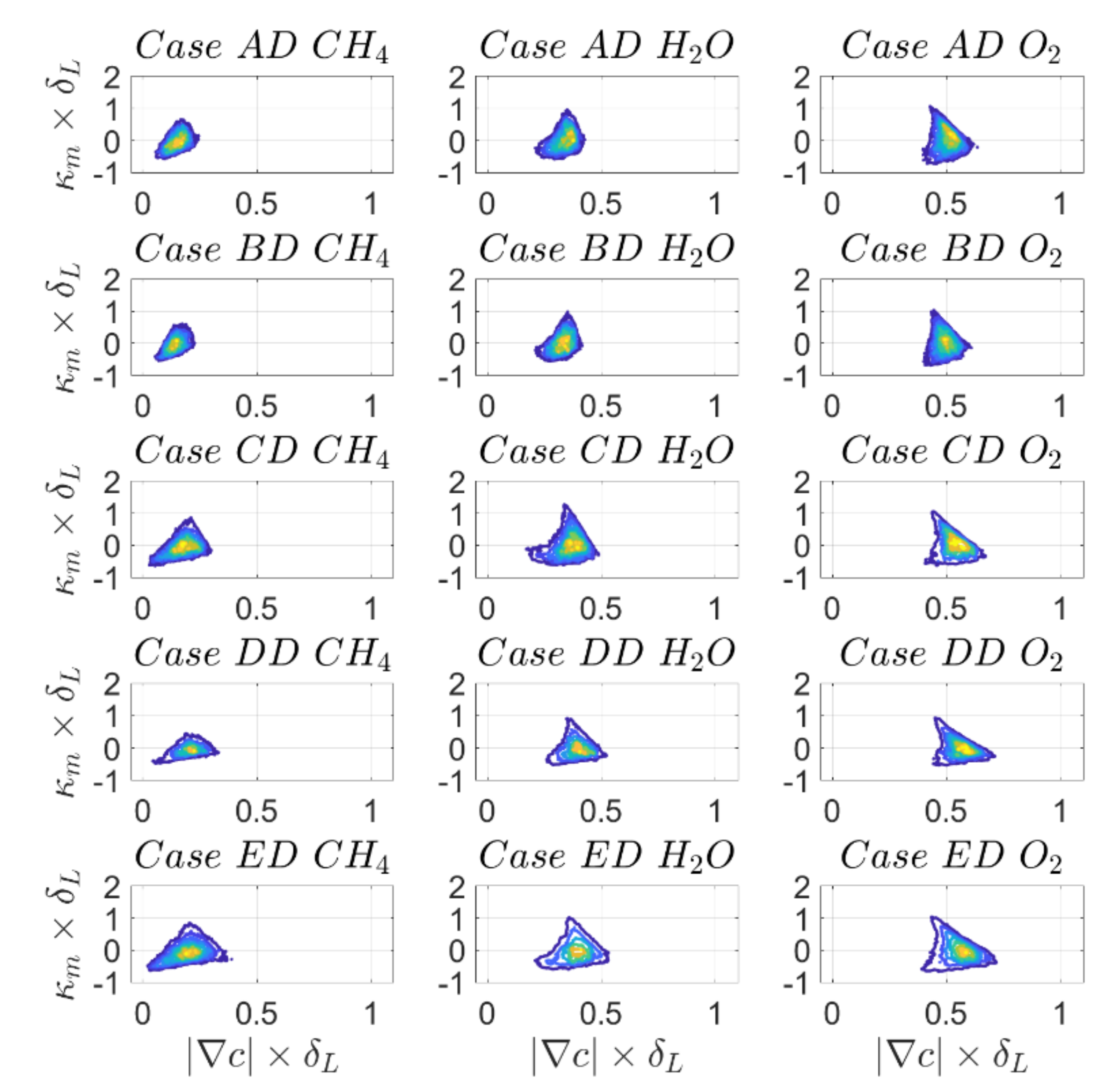
The joint PDFs of
and
with
are shown in
Figure 12 and
Figure 13, respectively for the
isosurface and the corresponding correlation coefficients are reported in
Table 10 for
. It can be seen from
Figure 12 that displacement speed is negatively correlated with mean curvature. While the correlation coefficients are close to
for cases A and B, the correlation becomes non-linear for increasing turbulence intensity, as can be discerned from the considerably smaller values of
in comparison to −1.0.
Table 10 also shows that the strength of linearity or non-linearity depends to some extent on the physio-chemical model and also on the definition of reaction progress variable in the case of DC. Tangential diffusion component of displacement speed
is deterministically negatively correlated with curvature (see Equation (8)) with
in all cases, and for all definitions of reaction progress variable, as illustrated in
Figure 14. This shows that the non-linear curvature dependence of
originates from the non-linear curvature dependence of
. For
, the correlation strength between
is weak in all cases.
Figure 13 and
Table 10 show that there is a weak positive correlation in the case of both SC and DC simulations based on the reaction progress variable defined in terms of
while the correlation turns out to be mildly negative for DC and reaction progress variable based on
and
mass fractions. Again, for SC, the behaviour is in good agreement with the DC results and detailed explanations are also provided for the change of correlation strength with the value of reaction progress variable [
49], while the present analysis focuses on the differences between SC and DC and also on the differences resulting from different definitions of reaction progress variable.
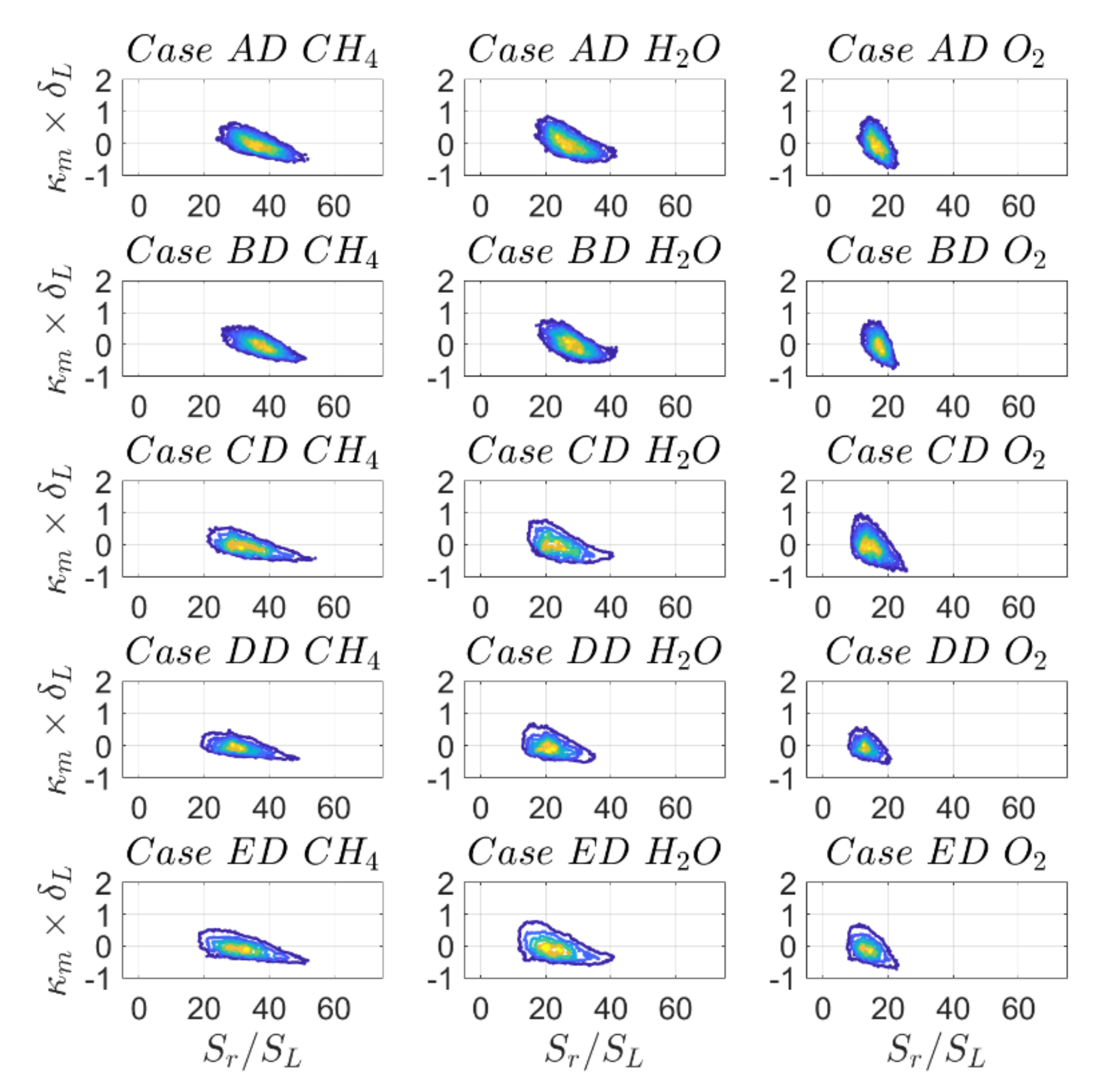
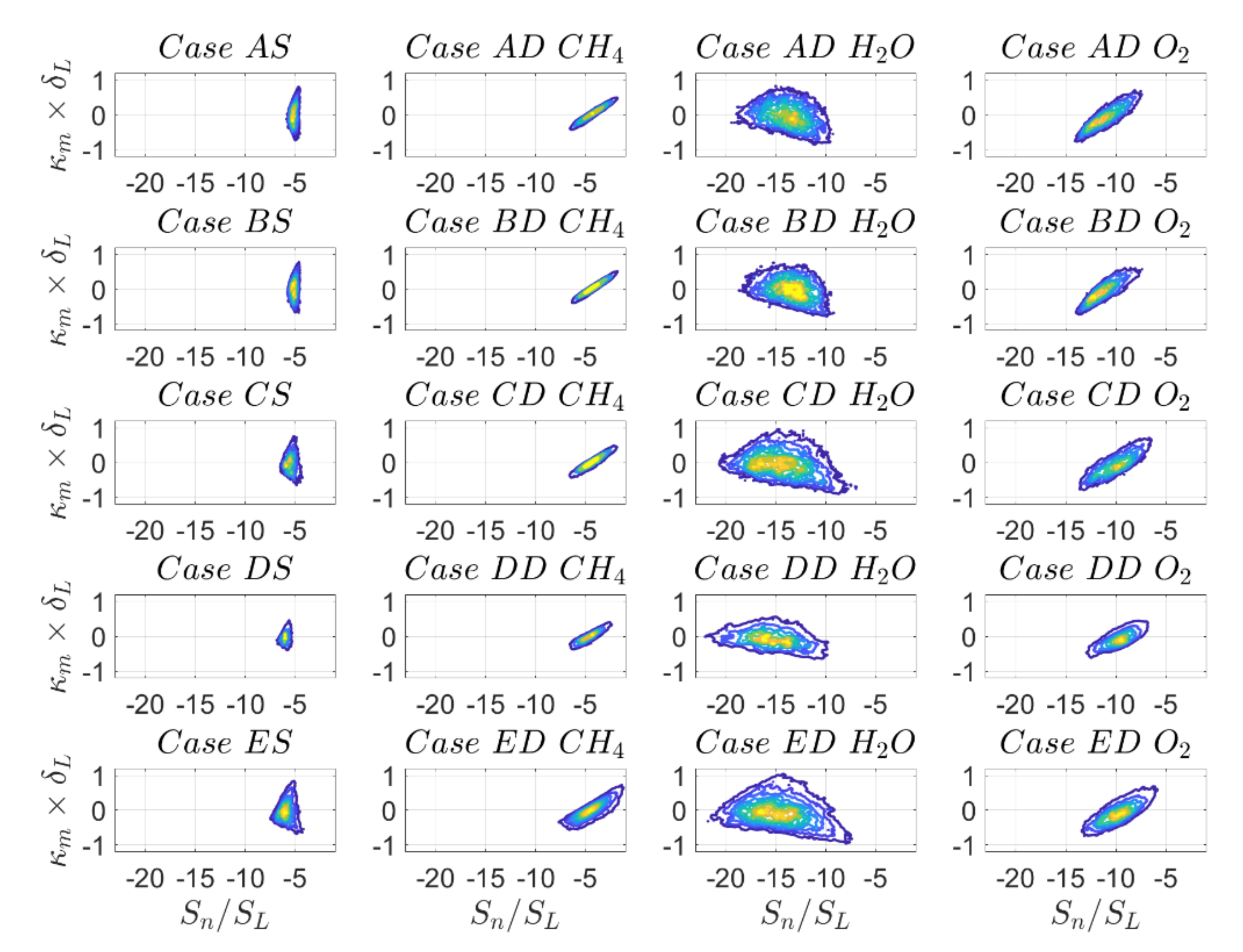
So far, results have revealed a reasonable qualitative agreement of displacement speed statistics with some quantitative variations when comparing DC with SC, which are of the same order of magnitude, as compared to different definitions of reaction progress variable in the case of DC. As illustrated in
Figure 15 and
Figure 16, this behaviour changes drastically when looking at the individual contributions of displacement speed
and
with mean curvature. For example, there is a weak positive correlation
in the case of SC and DC based on
mass fraction, whereas the correlation is clearly negative in the case of DC with reaction progress variable based on
and
mass fractions for the reaction progress variable isosurface
. In other words, there is a qualitative change in
with the variation of the definition of reaction progress variable, which warrants further explanation. Similar qualitative differences can be seen from the
joint PDFs shown in
Figure 16. The values of
and
are reported in
Table 11.
For low Mach number and unity Lewis number flames, the reaction rate
is independent of curvature and thus, according to Equation (8) the correlation between
and
is governed by the correlation between
and
. However, this statement is rendered invalid in the case of DC, where there might be a significant correlation between
and
, which also depends on the choice of reaction progress variable.
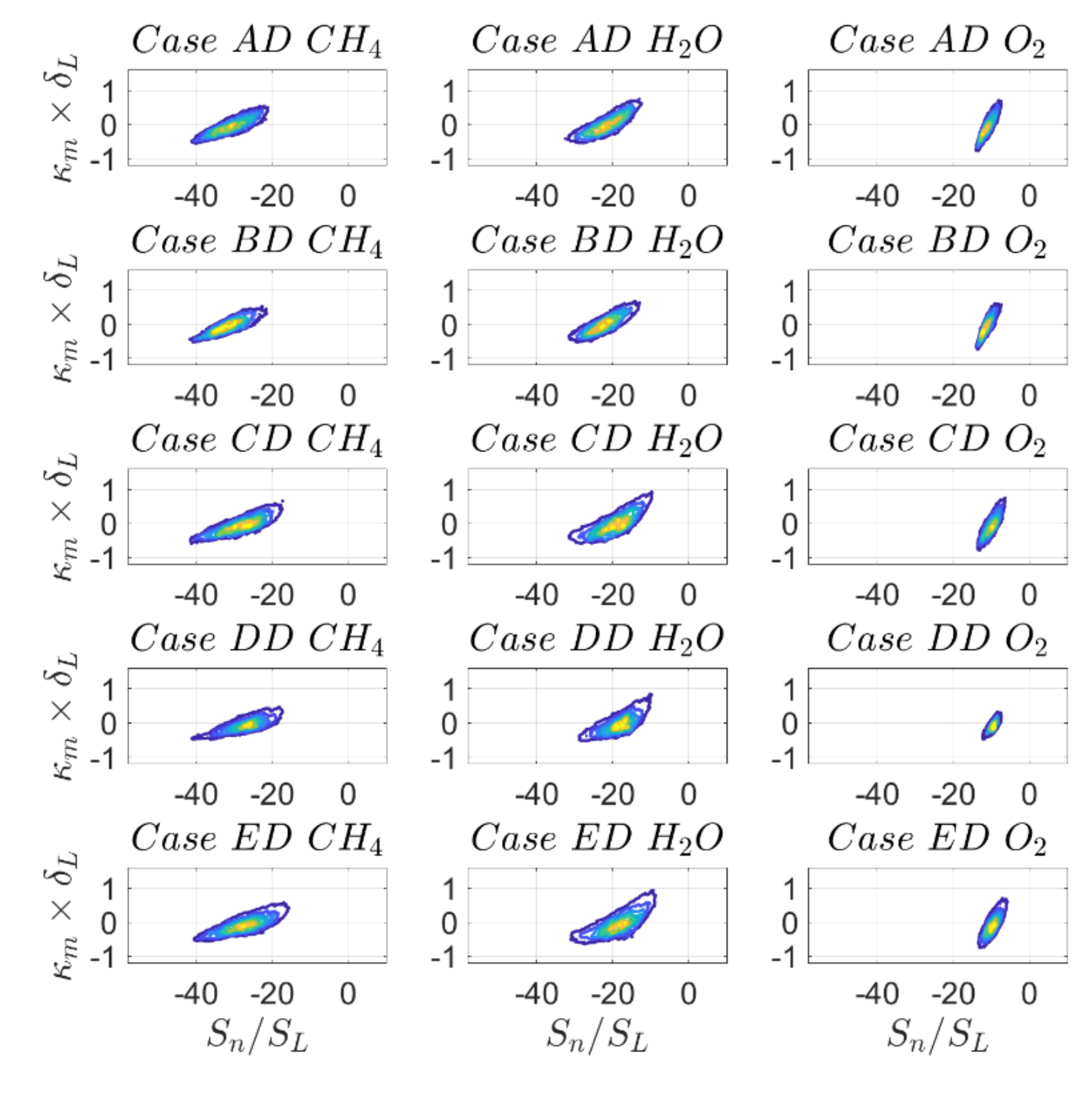
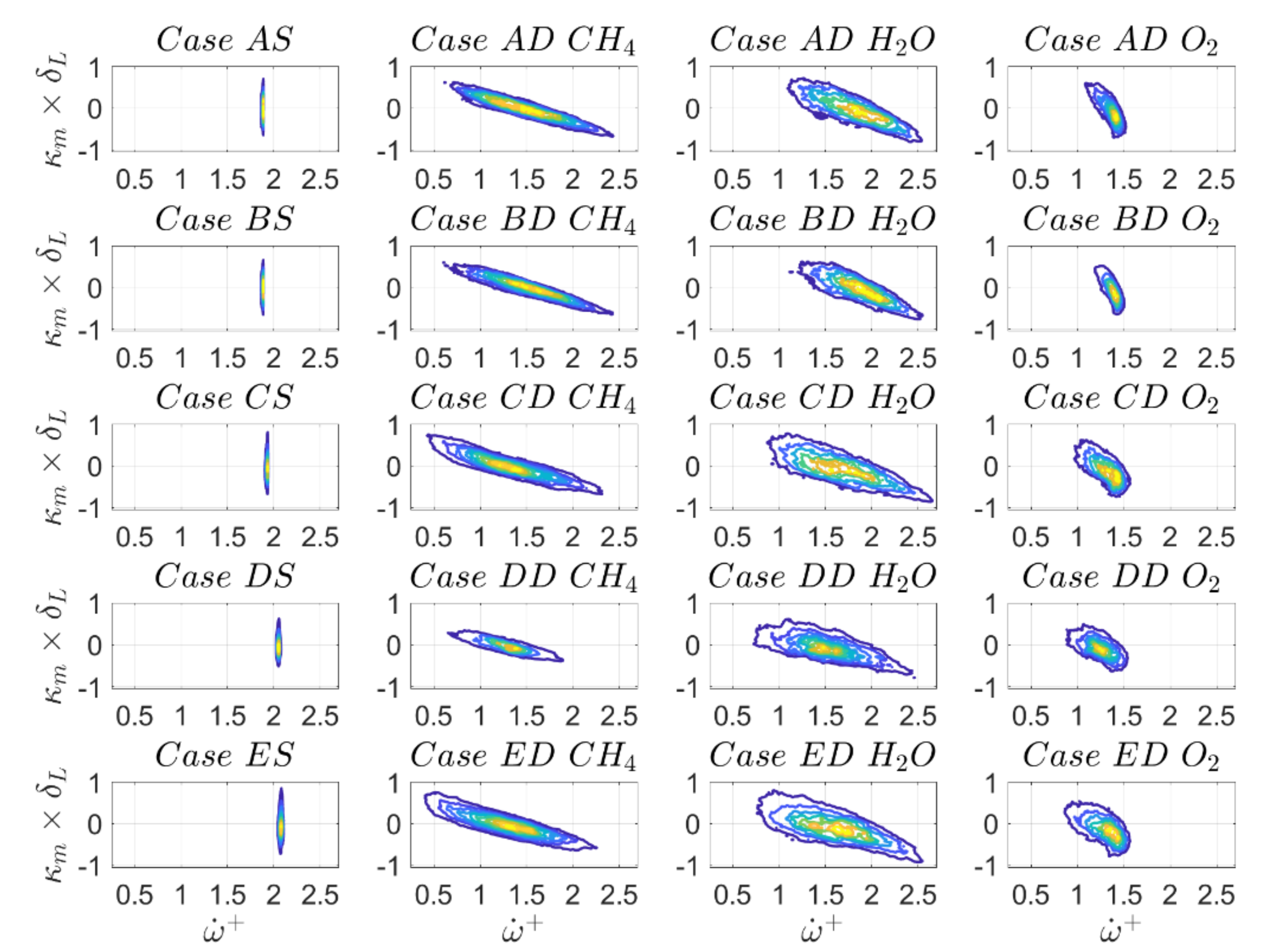
Figure 17 and
Figure 18 show the joint PDFs of normalised reaction rate
with mean curvature
and the joint PDFs for the surface density function
with
on the
isosurface, while the correlation coefficients are reported in
Table 12. For the ease of notation, the dimensionless reaction rate
will be referred to as
in the following. Indeed
Figure 17 shows that
in the case of SC. Together with
, this results in
. It is noted that for low Mach number and unity Lewis number flames density is constant on a given
isosurface.
For DC,
Figure 17 clearly indicates a negative correlation between reaction rate and curvature for all definitions of reaction progress variable on the
isosurface. In the case of SC, this would correspond to a scenario with Lewis number larger than unity [
31,
32] while the opposite would be expected for
. Indeed, it was discussed in the introduction that a methane flame is characterised by a Lewis number slightly larger than
according to the parameterisations given by Equations (5)–(7) [
45,
46,
47] which is qualitatively consistent with the trends shown in
Figure 17. However, the species
and
have considerably different Lewis numbers (
respectively, according to
Table 3) and therefore their behaviours warrant a more detailed discussion.
In
SC simulations [
31,
32], the focusing of reactants takes place at a faster rate than the rate of defocusing of heat at the positively curved zones, which leads to the simultaneous presence of high temperature and reactant concentrations and conversely for negatively curved regions. Hence, high (low) temperature values are associated with positive (negative) curvatures for
and the opposite is valid for
[
27,
28,
29,
30,
31,
32]. For the SC simulations, the reaction rate depends on temperature according to
where
is the normalised pre-exponential factor,
is the activation energy, such that a negative (positive) correlation between
and
induces a negative (positive) correlation between
and
and the same holds true for non-dimensional temperature which will be denoted
in the following discussion and in fact is identical to
. For a reaction mechanism involving
steps and
species given by:
the situation is much more complex and one obtains
where the specific forward
and backward
reaction rate coefficients
are given by an Arrhenius expression similar to Equation (9). Here, the
are called stoichiometric coefficients,
denotes the
different species,
are the molar masses,
the molar densities. Equation (11) shows that the reaction rate for species
in the context of DC depends on many more factors compared to the SC 1-step case shown in Equation (9) such that a negative (positive) correlation between
and
does not necessarily induce a correlation of same sign between
and
. A similar conclusion holds for
given by
A large reaction rate
of species
does not necessarily imply a high
because the enthalpy of formation of species
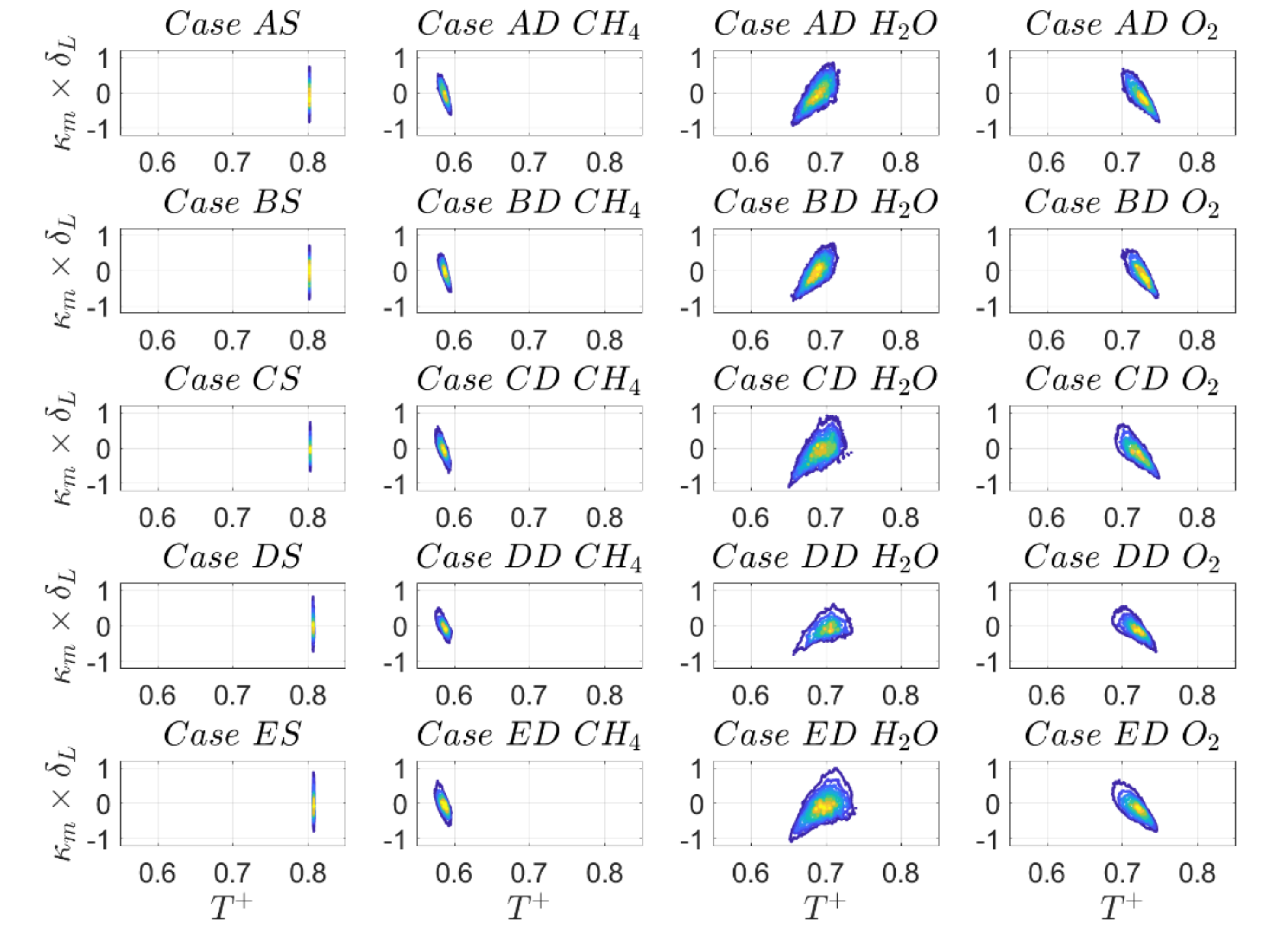
can be very small or even zero. Finally, all species share the identical temperature field, such that explanations holding true for SC cannot directly be applied to DC without caution. In fact, the joint PDFs between temperature and curvature for the
isosurface are shown in
Figure 19 and the correlation coefficients are reported in
Table 13 for the
isosurface. Indeed, by comparing
Figure 17 and
Figure 19, it becomes obvious that
while
for reaction progress variable based on
mass fraction, indicating that the value of temperature and rate of formation of species
can be partially decoupled.
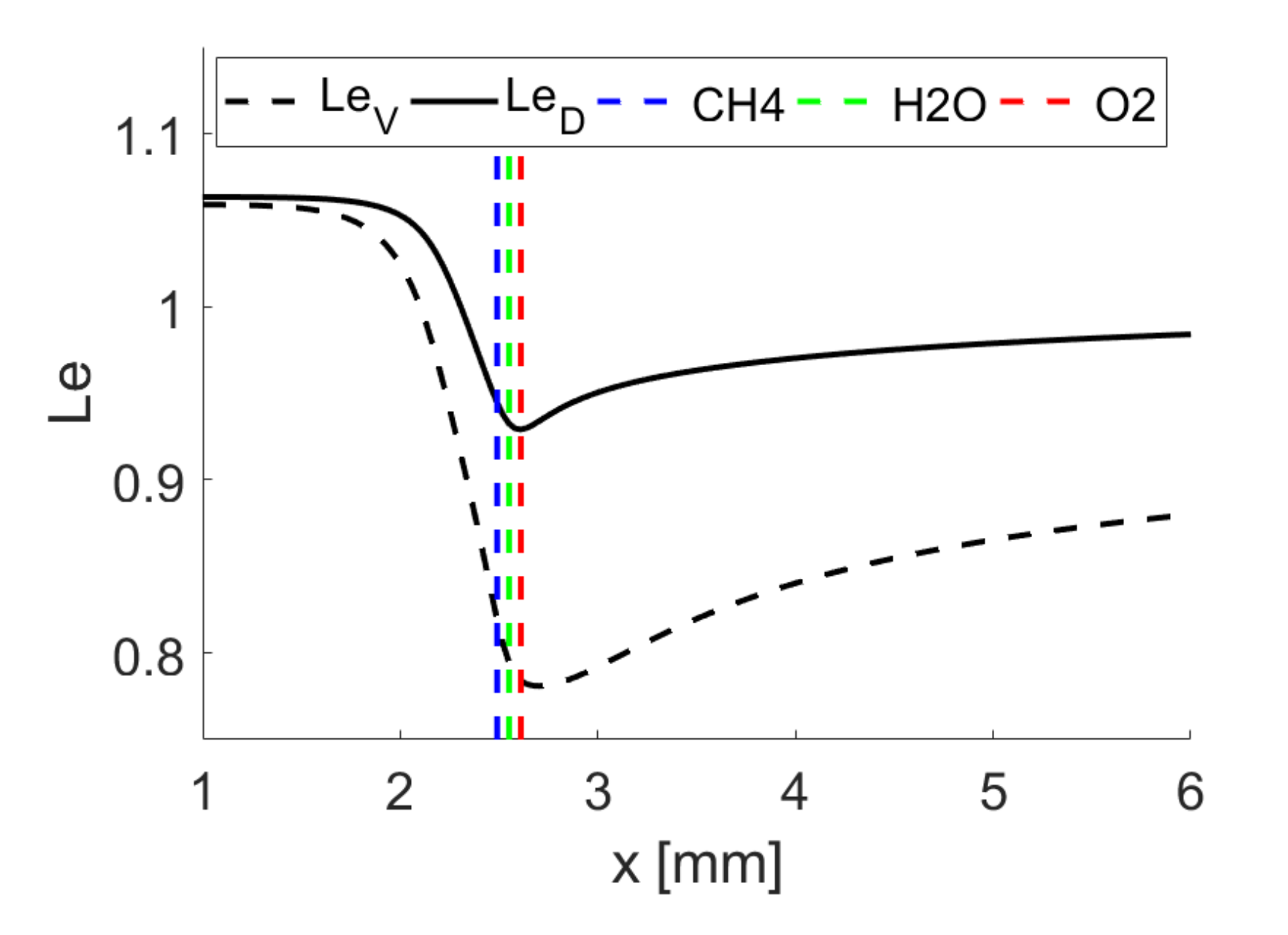
Water vapour is characterised by a higher diffusivity and Lewis number
(see
Table 3). For SC the thermo-diffusive mechanisms explained before lead to higher reaction rates in positively curved regions and higher flame wrinkling for
compared to
flames. As a result of this, positively (negatively) curved regions propagate faster (slower) and finger-like structures start to develop. A transfer of this mechanism to isosurfaces of individual
species in DC simulations is obviously not possible because all species transport equations are coupled. By contrast, as discussed next, it appears that the flame wrinkling of the
mass fraction-based reaction progress variable isosurface is smaller compared to those based on
and
isosurfaces. This is a result of preferential diffusion and its influence on intermediate reaction steps and can on one hand be attributed to higher diffusivity of
which smoothens high curvature magnitudes. On the other hand, this effect is further attenuated by higher (reduced) reactivity in the positively (negatively) curved regions. The turbulent flame areas
normalised by the cross-section of the computational domain
for
and
are exemplarily given by
and
respectively for case A. The values for temperature isosurfaces (representing the laminar flame temperature for
) for the three definitions of reaction progress variable are given by
.
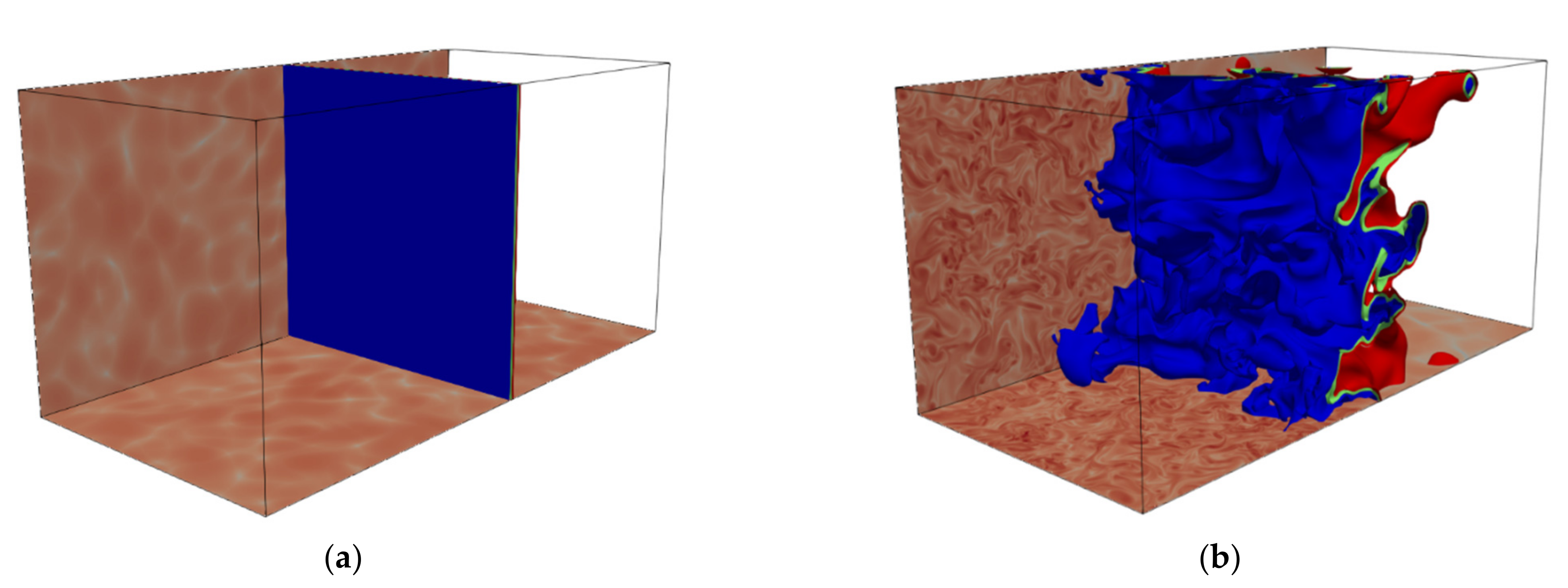
The reduced wrinkling of the
based reaction progress variable isosurface compared to
based reaction progress variable for case AD can be confirmed from
Figure 20a, which shows a simultaneous plot of isosurfaces of reaction progress variable
for
(red) and
(grey) with the
based isosurface overlapping the
-based isosurface. Similar observations in terms of flame wrinkling have been reported for detailed chemistry hydrogen-air simulations [
22].
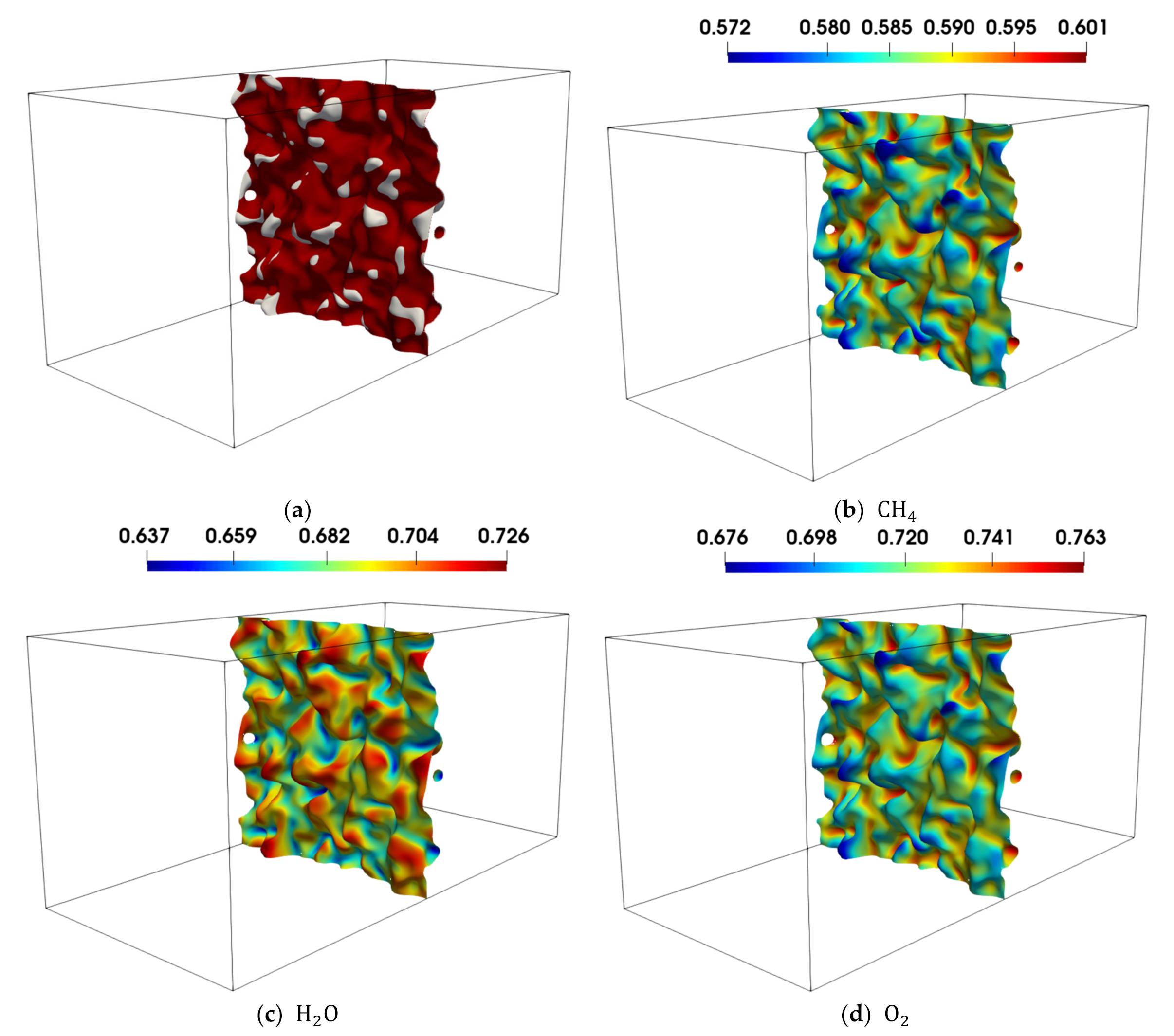
Figure 20 also shows isosurfaces of
coloured with non-dimensional temperature for case AD for reaction progress variable based on
and
mass fractions. The opposite correlation of temperature and curvature (observed in
Figure 19) between
,
and
can be clearly seen. While positively curved regions are characterised by lower temperature in the case of
and
based reaction progress variable isosurfaces, they show higher temperature for
. In order to understand this apparent contradiction, it is also important to recall that the different isosurfaces are characterised by different positions in the flame (see
Figure 2 and
Figure 3) and to note that the colour bars have different temperature ranges. As the
isosurface is less wrinkled than the corresponding temperature isosurface (because of higher mass diffusivity than thermal diffusivity), positively curved regions reach out less into to unburned gas side and hence they are characterised by higher temperatures. Similarly, negatively curved regions reach out less into the burned gas side and are consequently characterised by lower temperatures. The effect can be clearly seen in
Figure 21a and is consistent with the positive correlation between
and
for the
-based reaction progress variable isosurface.
Besides diffusion, flame propagation and wrinkling are also determined by reactive effects. At the positively curved locations, focussing of
takes place at a faster rate than the defocussing of
and heat and this gives rise to a higher likelihood of the reaction
which is a chain propagation reaction and promotes heat release due to
. This also decreases the wrinkling of
based reaction progress variable isosurface at the positively curved locations, whereas just the opposite mechanisms lead to less wrinkling and smaller temperatures in the negatively curved locations. The higher concentrations of mass fractions
and
in the positively curved regions of the
isosurface for
based reaction progress variable can be clearly seen in
Figure 21b–d and support the above argument. A similar argument can be made to explain the increased wrinkling of the
-based reaction progress variable isosurfaces and the negative correlation between temperature and curvature. The defocussing of
and
takes place at faster rates than that of
at the positively curved locations. This acts to promote the forward reaction of the equilibrium reaction
according to Le Chatelier. This can be substantiated from low concentrations of
and
at the positively curved locations of
isosurface for
-based reaction progress variable (not shown here).
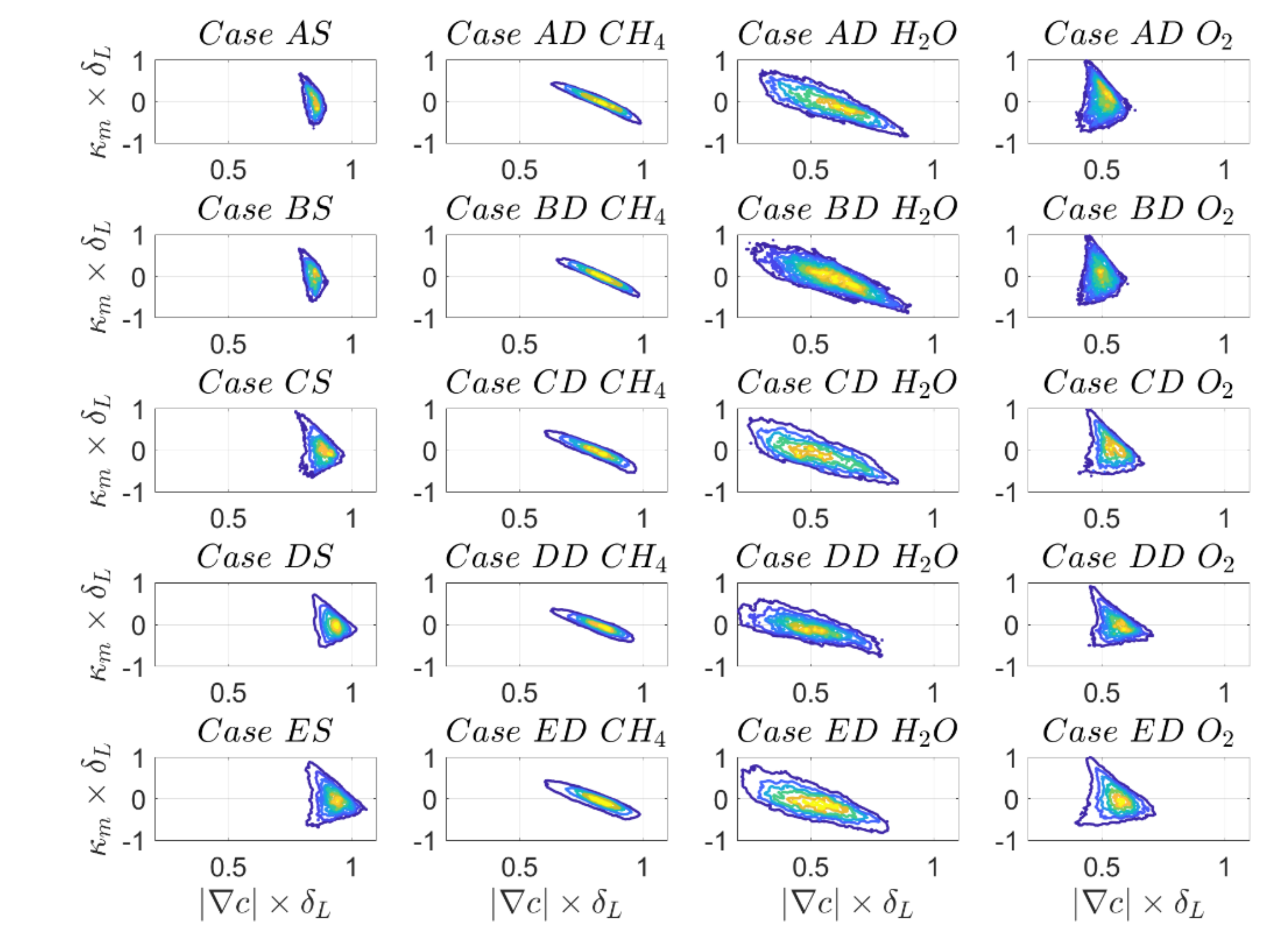
In order to finally understand the joint PDFs of
and
, besides reaction rate, it is important to look at the joint PDFs of
and
. An initial observation from
Figure 18 is that the joint PDFs for the
-based reaction progress variable behave to some extent similar to the SC case (i.e., they show a small correlation magnitude with a positive and a negative correlating branch for negative and positive curvatures, respectively) while the joint PDFs for reaction progress variable based on
and
mass fractions are negatively correlated and the positively correlating branch cannot be observed. It has been explained in part 1 of this paper that (for constant dilatation rate)
and
(where
is the normal strain rate) are negatively correlated. Further, a compressive (negative) normal strain rate causes local thinning which acts to increase the scalar gradient
. The negative correlation between
and
leads to the principally negative correlation between
and
. However,
can also assume small values at locations of high negative curvature because of secondary thickening effects induced by high values of dilation rate, which locally overcomes
to induce extensive normal strain rate (i.e.,
) [
44,
50,
51] leading to a positively correlating branch for SC and DC based on
-based reaction progress variable definition.
Based on the correlation behaviour of numerator and denominator in the definition of
and
, it is possible to infer the correlations of
and
with curvature. However, in contrast to low Mach number SC simulations where
can be assumed to be constant on a given isosurface, the same assumption does not hold any longer in the case of DC simulations, such that the denominator
potentially shows a more complex behaviour than
on its own (it is recalled that
deterministically depends on
such that different signs of
can be observed for different reaction progress variable definitions, see
Figure 19). The Pearson correlation coefficient between two random variables
is given by the covariance of both variables divided by the product of their standard deviations:
For the sign of the correlation, it is sufficient to look at the covariance between the random variables. Following Bohrnstedt and Goldberger [
52], the covariance of the product of two random variables
with a third random variable
is given by the expression
, where
denotes the expected value of a variable and
is the third-order moment of the three variables. It can be shown that the third-order moment vanishes under multivariate normality. In the present case, it is not negligible. The above identity shows that
In other words, the sign of the correlation
depends on the expected values of
and
and the covariances of these variables with
.
Table 14 and
Table 15 show the individual contributions of
and
, respectively, according to Equation (12), exemplarily for reaction progress variable based on
and
. These species have been selected because they show similar correlations trends for
and
but
shows the opposite sign for
and
, which requires additional explanation and a similar statement holds true for
.
By referring to the terms in Equation (12), it can be seen from
Table 14 that the higher magnitudes of
are mainly responsible for the change of sign of
in the case of
mass fraction-based reaction progress variable in comparison to
mass fraction-based reaction progress variable. The change of sign of
in the case of
-based reaction progress variable in comparison to
based reaction progress variable is the result of two effects. Firstly, the reaction progress variable based on
mass fraction has a small plateau for values slightly larger than
which results in small values of
and hence large values of
. Secondly, the positive correlation
induces a negative correlation
due to the ideal gas law. This, in turn, reinforces the negative correlation
for
-based reaction progress variable, while it results in a change of sign and weakening of correlation strength for
-based reaction progress variable because of opposing correlations of
and
with
. A similar explanation provides the explanations for the correlation behaviour between
and
(see
Table 15).
The foregoing discussion has shown that the statistics of
and
with curvature are sensitive to the definition of reaction progress variable and detailed explanations for this behaviour have been provided. Besides that, the statistics are also sensitive to the choice of isosurface level, which is also evident from the correlation coefficients reported in
Table 11,
Table 12 and
Table 13. Important qualitative differences have also been found for the joint PDFs of normalised reaction rate, surface density function and non-dimensional temperature with mean curvature. These differences have been explained based on preferential diffusion and its influence on intermediate reaction steps which give rise to different degrees of wrinkling of different isosurfaces for different definitions of reaction progress variable.
For further illustration, the joint PDFs of normalised reaction rate,
,
and
for different definitions of reaction progress variable are exemplarily shown in
Figure A1,
Figure A2,
Figure A3 and
Figure A4 in
Appendix A, but in contrast to earlier figures, the statistics are now taken on the same isosurface corresponding to the
based reaction progress variable value of
. It can be seen from
Figure A1,
Figure A2,
Figure A3 and
Figure A4 in
Appendix A that some of the qualitative differences observed earlier diminish if statistics are taken on the same
based reaction progress variable isosurface, and similar qualitative trends have been observed for other reaction progress variable definitions. For example,
becomes now consistently negative and, in addition, the negative correlation
vanishes.